Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic?
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Variable Assessment
2.3. Statistical Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Keum, N.; Lee, D.H.; Greenwood, D.C.; Manson, J.E.; Giovannucci, E. Vitamin D supplements and total cancer incidence and mortality: A meta-analysis of randomized controlled trials. Ann. Oncol. 2019, 30, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Schöttker, B.; Haug, U.; Schomburg, L.; Köhrle, J.; Perna, L.; Müller, H.; Holleczek, B.; Brenner, H. Strong associations of 25-hydroxyvitamin D levels with all-cause, cardiovascular, cancer and respiratory disease mortality in a large cohort study. Am. J. Clin. Nutr. 2013, 97, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Schöttker, B.; Jorde, R.; Peasey, A.; Thorand, B.; Jansen, E.H.; De Groot, L.; Streppel, M.; Gardiner, J.; Ordóñez-Mena, J.M.; Perna, L.; et al. Consortium on health and ageing: Network of cohorts in Europe and the United States. Vitamin D and mortality: Meta-analysis of individual participant data from a large consortium of cohort studies from Europe and the United States. BMJ 2014, 348, g3656. [Google Scholar] [CrossRef]
- Schöttker, B.; Brenner, H. Vitamin D as a resilience factor, helpful for survival of potentially fatal conditions: A hypothesis emerging from recent findings of the ESTHER cohort study and the CHANCES consortium. Nutrients 2015, 7, 3264–3278. [Google Scholar] [CrossRef]
- Brenner, H.; Jansen, L.; Saum, K.U.; Holleczek, B.; Schöttker, B. Vitamin D supplementation trials aimed at reducing mortality have much higher power when focusing on people with low serum 25-hydroxyvitamin D concentrations. J. Nutr. 2017, 147, 1325–1333. [Google Scholar] [CrossRef]
- Schöttker, B.; Hagen, L.; Zhang, Y.; Gào, X.; Holleczek, B.; Gao, X.; Brenner, H. Serum 25-hydroxyvitamin D levels as an aging marker: Strong associations with age and all-cause mortality independent from telomere length, epigenetic age acceleration, and 8-isoprostane levels. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 74, 121–128. [Google Scholar] [CrossRef]
- Löw, M.; Stegmaier, C.; Ziegler, H.; Rothenbacher, D.; Brenner, H. Epidemiological investigations of the chances of preventing, recognizing early and optimally treating chronic diseases in an elderly population (ESTHER study). Dtsch. Med. Wochenschr. 2004, 129, 2643–2647. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar] [CrossRef]
- Desquilibet, L.; Mariotti, F. Dose-response analyses using restricted cubic spline functions in public health research. Stat. Med. 2010, 29, 1037–1057. [Google Scholar] [CrossRef]
- World Health Organization Health Statistics and Information Systems. Metrics: Population Attributable Fraction (PAF). Available online: https://www.who.int/healthinfo/global_burden_disease/metrics_paf/en/ (accessed on 2 May 2020).
- GBD 2017 Causes of Death Collaborators*. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Gubbels Bupp, M.R.; Potluri, T.; Fink, A.L.; Klein, S.L. The confluence of sex hormones and aging on immunity. Front. Immunol. 2018, 9, 1269. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Kozloski, M. Sex differences in age trajectories of physiological dysregulation: Inflammation, metabolic syndrome, and allostatic load. J. Gerontol. A Biol. Sci. Med. Sci. 2011, 66, 493–500. [Google Scholar] [CrossRef]
- Straub, R.H. The complex role of estrogens in inflammation. Endocr. Rev. 2007, 28, 521–574. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mishra, A.; Brinton, R.D. Transitions in metabolic and immune systems from pre-menopause to post-menopause: Implications for age-associated neurodegenerative diseases. F1000Research 2020, 9. [Google Scholar] [CrossRef]
- Meftahi, G.H.; Jangravi, Z.; Sahraei, H.; Bahari, Z. The possible pathophysiology mechanism of cytokine storm in elderly adults with COVID-19 infection: The contribution of “inflame-aging”. Inflamm. Res. 2020, 1–15. [Google Scholar] [CrossRef]
- Daneshkhah, A.; Agrawal, V.; Eshein, A.; Subramanian, H.; Kumar Roy, H.; Backman, V. The possible role of vitamin d in suppressing cytokine storm and associated mortality in COVID-19 patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Quesada-Gomez, J.M.; Castillo, M.E.; Bouillon, R. Vitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with Coronavirus SARS-CoV-2 infections. J. Steroid Biochem. Mol. Biol. 2020, 105719. [Google Scholar] [CrossRef]
- Mitchell, F. Vitamin-D and COVID-19: Do deficient risk a poorer outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef]
- Li, X.; He, J.; Yu, M.; Sun, J. The efficacy of vitamin D therapy for patients with COPD: A meta-analysis of randomized controlled trials. Ann. Palliat. Med. 2020, 9, 286–297. [Google Scholar] [CrossRef]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.; Solway, J. Association of vitamin D deficiency and treatment with COVID-19 incidence. medRxiv 2020, 20095893. [Google Scholar] [CrossRef]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020. [Google Scholar] [CrossRef]
- Lindqvist, P.G.; Epstein, E.; Landin-Olsson, M.; Ingvar, C.; Nielsen, K.; Stenbeck, M.; Olsson, H. Avoidance of sun exposure is a risk factor for all-cause mortality: Results from the Melanoma in Southern Sweden cohort. J. Intern. Med. 2014, 276, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Metzler, B.; Siostrzonek, P.; Binder, R.K.; Bauer, A.; Reinstadler, S.J. Decline of acute coronary syndrome admissions in Austria since the outbreak of COVID-19: The pandemic response causes cardiac collateral damage. Eur. Heart J. 2020, 41, 1852–1853. [Google Scholar] [CrossRef] [PubMed]
- Tapper, E.B.; Asrani, S.K. COVID-19 pandemic will have a long-lasting impact on the quality of cirrhosis care. J. Hepatol. 2020, 73, 441–445. [Google Scholar] [CrossRef]
- Baddour, K.; Kudrick, L.D.; Neopaney, A.; Sabik, L.M.; Peddada, S.D.; Nilsen, M.L.; Johnson, J.T.; Ferris, R.L.; Mady, L.J. Potential impact of the COVID-19 pandemic on financial toxicity in cancer survivors. Head Neck 2020, 42, 1332–1338. [Google Scholar] [CrossRef]
- Søreide, K.; Hallet, J.; Matthews, J.B.; Schnitzbauer, A.A.; Line, P.D.; Lai, P.B.S.; Otero, J.; Callegaro, D.; Warner, S.G.; Baxter, N.N.; et al. Immediate and long-term impact of the COVID-19 pandemic on delivery of surgical services. Br. J. Surg. 2020. [Google Scholar] [CrossRef]
- Zentrum für Krebsregisterdaten (ZfKD) im Robert Koch-Institut; Gesellschaft der Epidemiologischen Krebsregister e.V. (GEKID) (Eds.) Krebs in Deutschland für 2015/2016; Robert Koch-Institut: Berlin, Germany, 2019. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
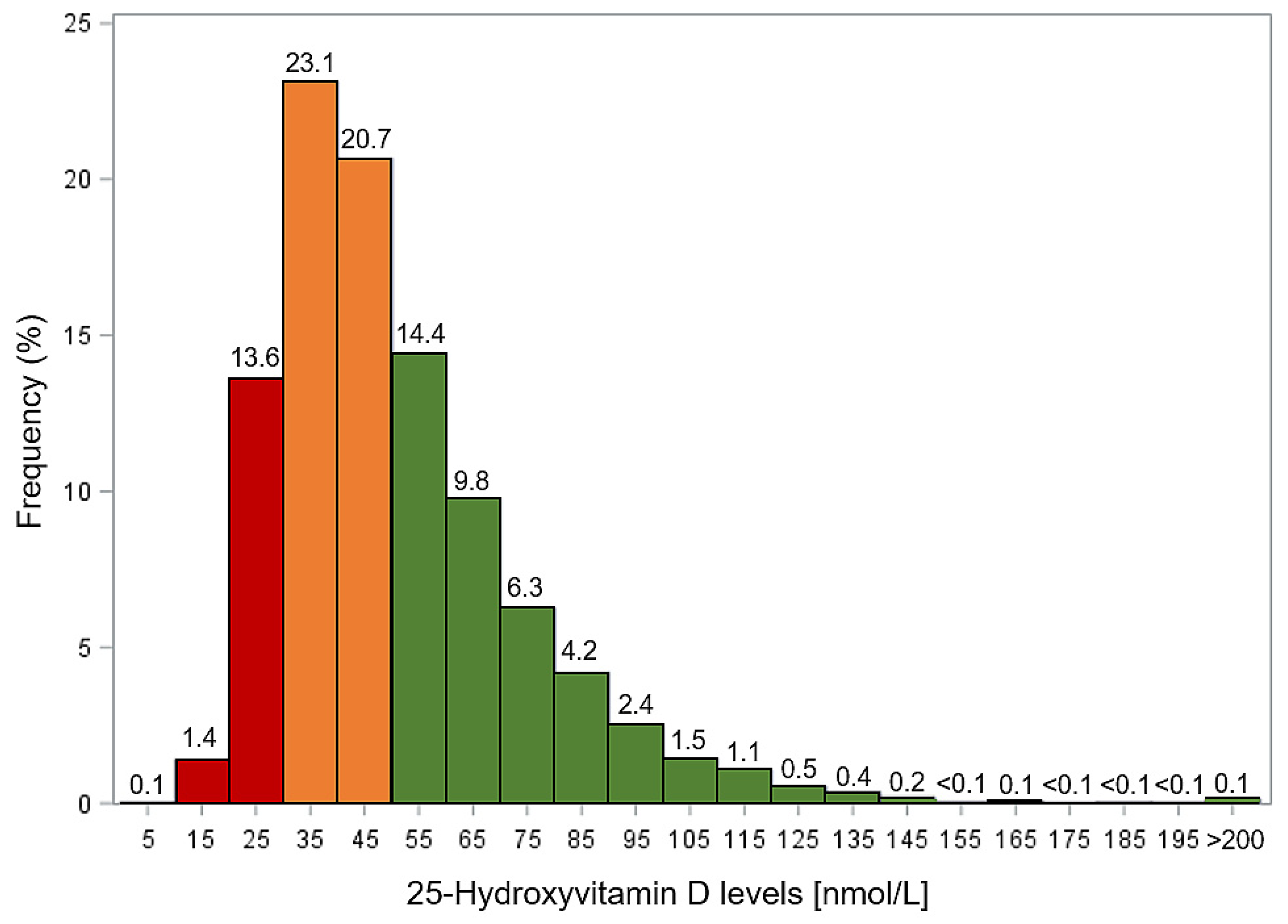
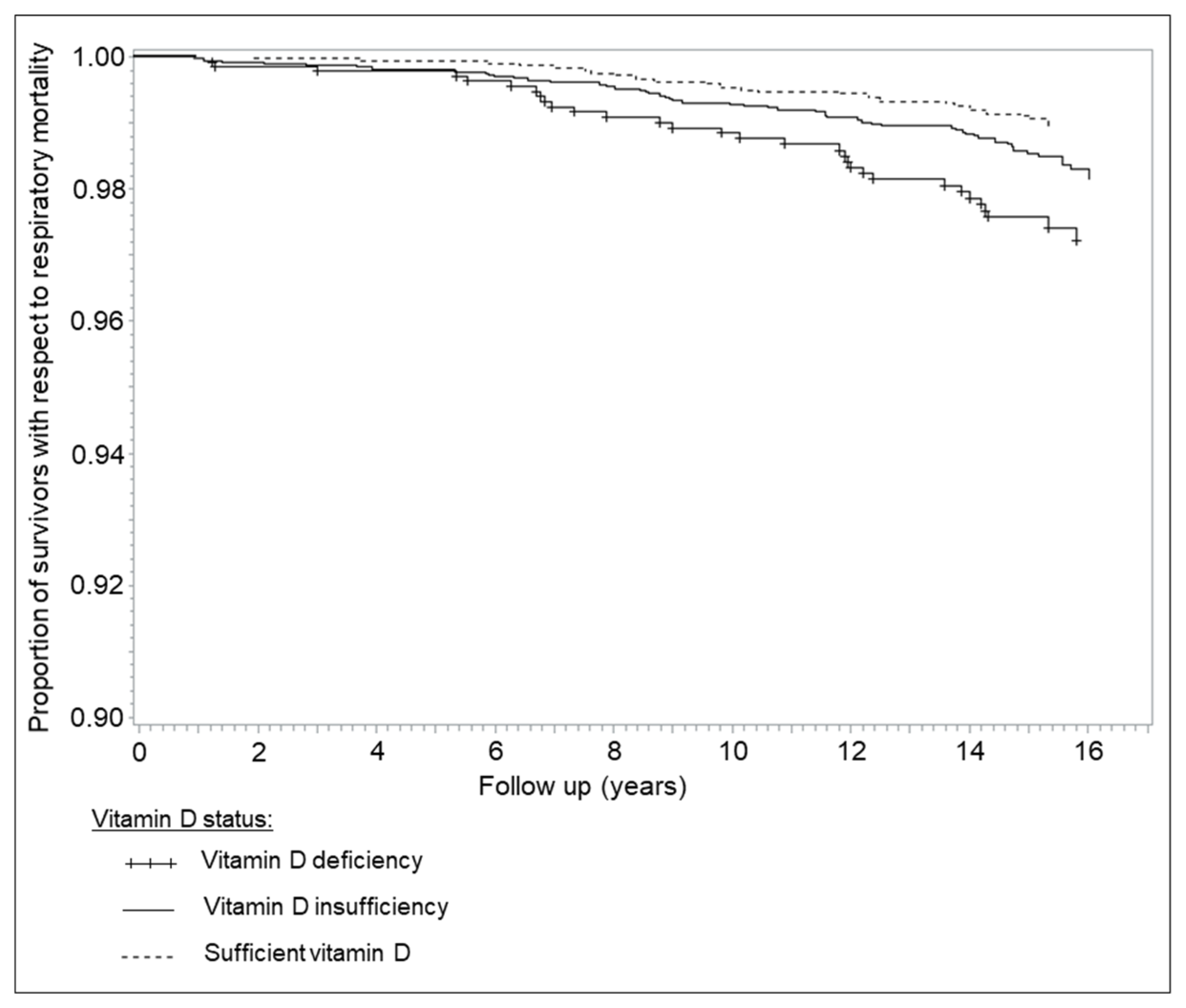
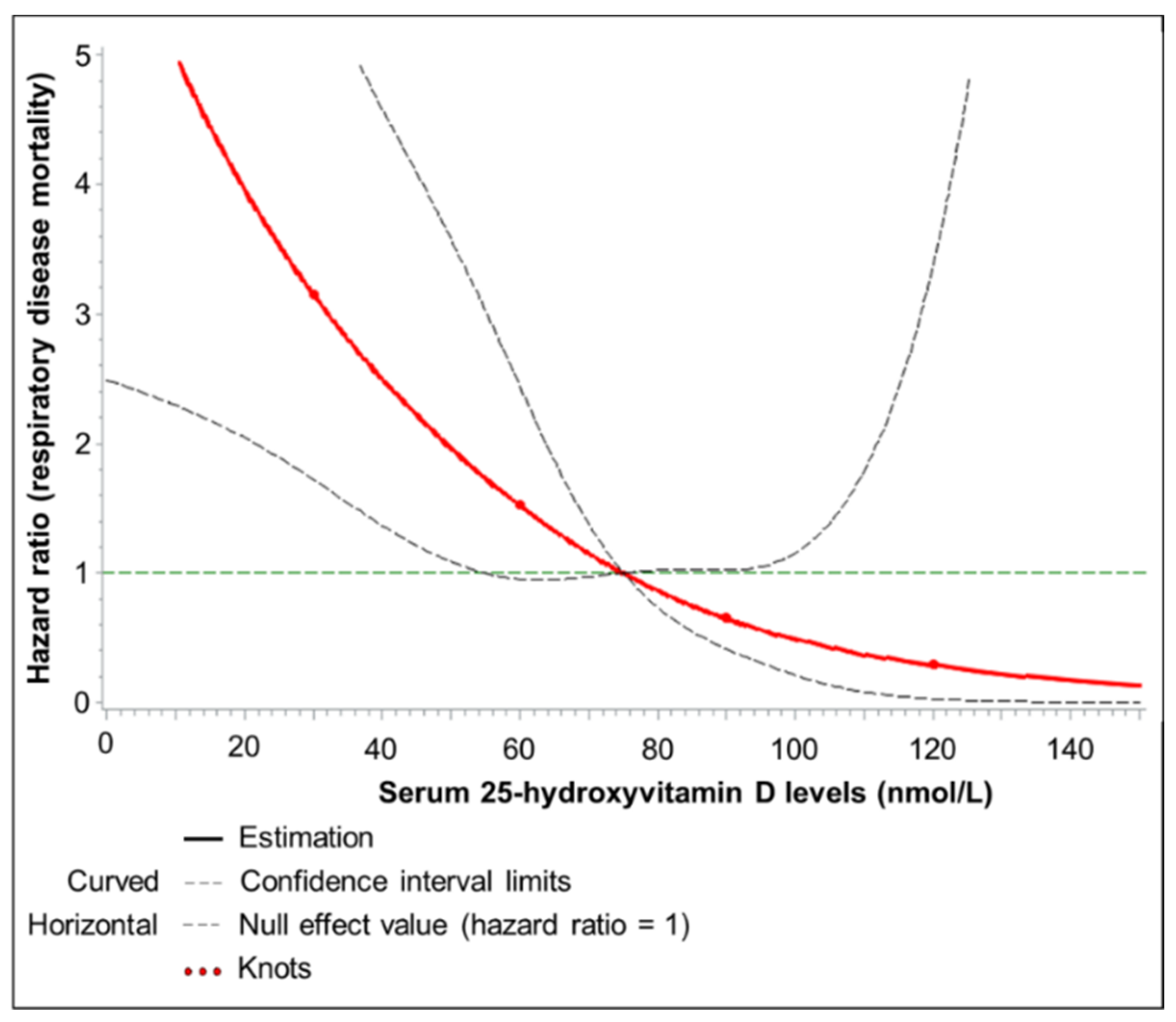
| Characteristic | Proportion of Characteristic in Total Population (%) | Prevalence of Vitamin D Insufficiency among Subjects with Characteristic (%) | Prevalence of Vitamin D Deficiency among Subjects with Characteristic (%) | Median 25(OH)D (Interquartile Range) |
|---|---|---|---|---|
| Total cohort | 100.0 | 43.8 | 15.1 | 45.6 (34.3–61.6) |
| Sex | ||||
| Female | 56.2 | 52.6 | 15.7 | 42.1 (32.9–53.9) |
| Male | 43.8 | 32.6 | 14.2 | 52.0 (37.0–71.0) |
| Age (years) | ||||
| 50–64 | 61.3 | 42.4 | 14.2 | 46.7 (35.1–63.2) |
| 65–69 | 22.9 | 45.2 | 15.5 | 44.3 (33.9–60.1) |
| 70–75 | 15.8 | 47.3 | 17.7 | 42.3 (32.3–55.9) |
| Month of recruitment (Season) | ||||
| January–February | 21.7 | 45.7 | 24.4 | 39.5 (30.3–53.5) |
| March–April | 13.9 | 51.6 | 21.2 | 38.9 (31.1–51.7) |
| May–June | 14.2 | 47.8 | 12.4 | 45.3 (35.1–60.1) |
| July–August | 17.0 | 35.6 | 6.9 | 53.9 (41.8–72.9) |
| September–October | 17.0 | 37.0 | 8.1 | 52.5 (40.1–71.5) |
| November–Deccember | 16.2 | 47.0 | 15.6 | 44.1 (33.7–58.2) |
| School education | ||||
| ≤9 years | 75.0 | 44.7 | 15.3 | 45.1 (34.1–60.8) |
| 9–11 years | 14.1 | 44.3 | 13.5 | 46.3 (34.9–61.8) |
| ≥12 years | 11.0 | 36.1 | 15.4 | 49.2 (34.9–68.4) |
| Smoking | ||||
| Never | 50.5 | 47.9 | 14.5 | 44.5 (34.3–58.7) |
| Former | 32.6 | 37.6 | 12.2 | 50.1 (36.8–67.8) |
| Current | 16.9 | 43.2 | 21.4 | 41.3 (31.3–57.9) |
| BMI (kg/m2) | ||||
| <30 | 74.5 | 42.1 | 14.1 | 46.8 (34.9–63.4) |
| ≥30 | 25.5 | 48.9 | 17.9 | 41.9 (32.5–56.0) |
| Physical activity a | ||||
| Low | 67.1 | 45.9 | 16.7 | 43.9 (33.2–58.3) |
| Moderate or high | 32.9 | 39.6 | 11.6 | 49.5 (36.9–67.3) |
| Fish consumption at least once per week | ||||
| No | 33.5 | 44.6 | 15.6 | 44.6 (33.4–60.9) |
| Yes | 66.5 | 43.2 | 14.2 | 46.3 (34.9–62.4) |
| Cause of Death | 25(OH)D [nmol/L] | Participants | Number of Deaths | Mortality a | Age, Sex and Season Adjusted Model | Full Model | ||
|---|---|---|---|---|---|---|---|---|
| Hazard Ratio (95% CI) | p-Value | Hazard Ratio (95% CI) b | p-Value | |||||
| Any cause | >50 | 3924 | 873 | 15.6 | Ref | Ref | ||
| 30–50 | 4186 | 1010 | 17.1 | 1.28 (1.16–1.41) | <0.0001 | 1.20 (1.09–1.32) | 0.0002 | |
| <30 | 1438 | 480 | 24.9 | 1.91 (1.70–2.15) | <0.0001 | 1.67 (1.48–1.89) | <0.0001 | |
| Cardiovascular disease c | >50 | 3924 | 313 | 5.6 | Ref | Ref | ||
| 30–50 | 4186 | 344 | 5.8 | 1.18 (1.00–1.39) | 0.0452 | 1.10 (0.94–1.30) | 0.2329 | |
| <30 | 1438 | 158 | 8.2 | 1.73 (1.41–2.12) | <0.0001 | 1.52 (1.23–1.86) | <0.0001 | |
| Cancer c | >50 | 3924 | 328 | 5.9 | Ref | Ref | ||
| 30–50 | 4186 | 344 | 5.8 | 1.17 (1.00–1.38) | 0.0491 | 1.10 (0.94–1.29) | 0.2320 | |
| <30 | 1438 | 153 | 7.9 | 1.58 (1.28–1.92) | <0.0001 | 1.38 (1.13–1.70) | 0.0020 | |
| Respiratory disease c | >50 | 3924 | 34 | 0.6 | Ref | Ref | ||
| 30–50 | 4186 | 58 | 1.0 | 2.24 (1.45–3.52) | 0.0004 | 2.06 (1.32–3.21) | 0.0015 | |
| <30 | 1438 | 31 | 1.6 | 3.69 (2.18–6.21) | <0.0001 | 3.04 (1.79–5.17) | <0.0001 | |
| Cause of Death | 25(OH)D [nmol/L] | Women | Men | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ntotal | Ndeaths | Mortality a | Hazard Ratio (95% CI) b | p-Value | Ntotal | Ndeaths | Mortality a | Hazard Ratio (95% CI) b | p-Value | For Interaction | ||
| Any cause | >50 | 1701 | 235 | 9.4 | Ref | 2223 | 638 | 20.8 | Ref | |||
| 30–50 | 2821 | 558 | 13.7 | 1.24 (1.06–1.45) | 0.0070 | 1365 | 452 | 24.7 | 1.19 (1.05–1.35) | 0.0071 | 0.713 | |
| <30 | 843 | 246 | 21.0 | 1.76 (1.45–2.12) | <0.0001 | 595 | 234 | 30.8 | 1.66 (1.42–1.96) | <0.0001 | 0.703 | |
| Cardiovascular disease | >50 | 1701 | 81 | 3.2 | Ref | 2223 | 232 | 7.5 | Ref | |||
| 30–50 | 2821 | 190 | 4.7 | 1.17 (0.90–1.53) | 0.245 | 1365 | 154 | 8.4 | 1.11 (0.90–1.38) | 0.3159 | 0.770 | |
| <30 | 843 | 90 | 7.7 | 1.78 (1.30–2.44) | 0.0003 | 595 | 68 | 9.0 | 1.37 (1.02–1.83) | 0.0335 | 0.198 | |
| Cancer | >50 | 1701 | 95 | 3.8 | Ref | 2223 | 233 | 7.6 | Ref | |||
| 30–50 | 2821 | 188 | 4.6 | 1.12 (0.87–1.44) | 0.3922 | 1365 | 156 | 8.5 | 1.09 (0.88–1.34) | 0.4262 | 0.770 | |
| <30 | 843 | 64 | 5.5 | 1.22 (0.87–1.70) | 0.2450 | 595 | 89 | 11.7 | 1.58 (1.22–2.07) | 0.0007 | 0.198 | |
| Respiratory disease | >50 | 1701 | 3 | 0.1 | Ref | 2223 | 31 | 1.0 | Ref | |||
| 30–50 | 2821 | 23 | 0.6 | 4.28 (1.27–1.42) | 0.0189 | 1365 | 35 | 1.9 | 1.89 (1.14–3.15) | 0.0136 | 0.201 | |
| <30 | 843 | 16 | 1.4 | 8.47 (2.38–30.12) | 0.0010 | 595 | 15 | 2.0 | 2.25 (1.14–4.42) | 0.0190 | 0.041 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brenner, H.; Holleczek, B.; Schöttker, B. Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic? Nutrients 2020, 12, 2488. https://doi.org/10.3390/nu12082488
Brenner H, Holleczek B, Schöttker B. Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic? Nutrients. 2020; 12(8):2488. https://doi.org/10.3390/nu12082488
Chicago/Turabian StyleBrenner, Hermann, Bernd Holleczek, and Ben Schöttker. 2020. "Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic?" Nutrients 12, no. 8: 2488. https://doi.org/10.3390/nu12082488
APA StyleBrenner, H., Holleczek, B., & Schöttker, B. (2020). Vitamin D Insufficiency and Deficiency and Mortality from Respiratory Diseases in a Cohort of Older Adults: Potential for Limiting the Death Toll during and beyond the COVID-19 Pandemic? Nutrients, 12(8), 2488. https://doi.org/10.3390/nu12082488





