Synergistic Effects of Heat-Killed Kefir Paraprobiotics and Flavonoid-Rich Prebiotics on Western Diet-Induced Obesity
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Sample Collection and Plasma Lipid Analysis
2.3. Real-Time RT-PCR
2.4. Isolation of Cellular Exopolysaccharide (EPS) from Kefir Lactic Acid Bacteria
2.5. 3T3-L1 Cell Culture
2.6. Statistical Analysis
3. Results
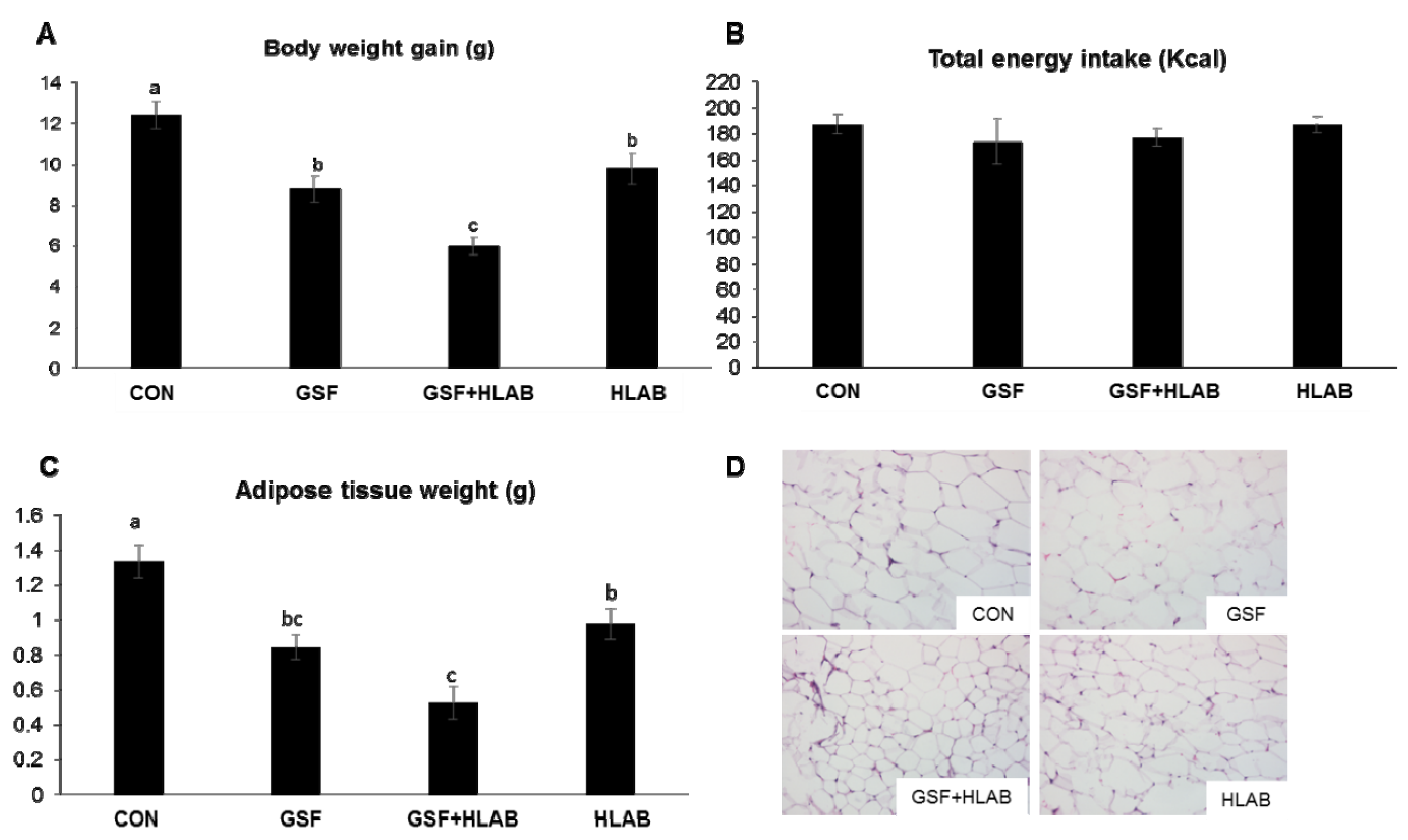
3.1. Effects of Wine GSF and HLAB Supplementation on Body Weight, Organ Weight, and Metabolic Parameters
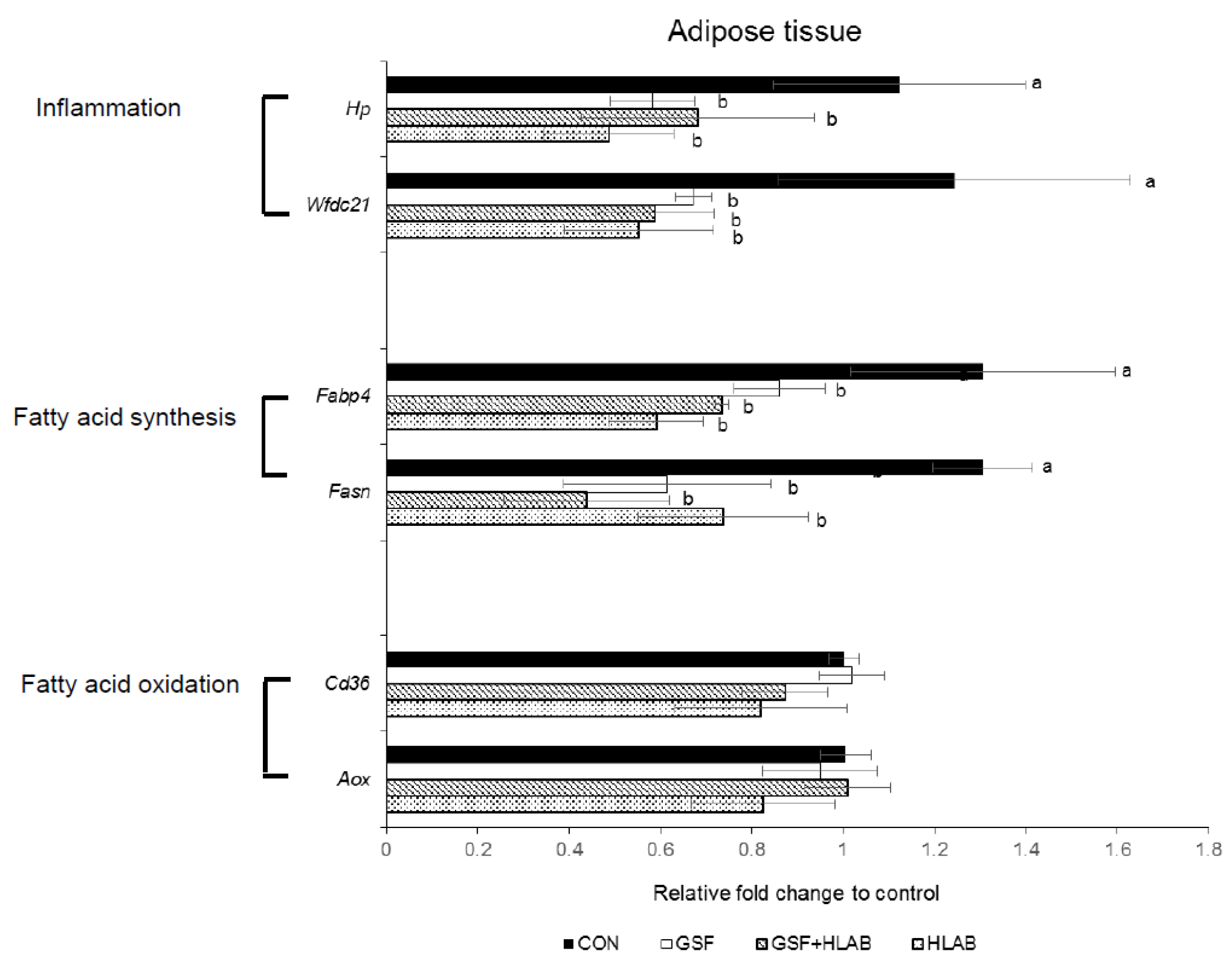
3.2. Analysis of Gene Expression in Adipose Tissue
3.3. Analysis of the Correlation between the Expression of Adipose mRNA and Metabolic Obesity Physiological Biomarkers
3.4. Inhibitory Activities of Cellular EPS of Kefir HLAB on Lipid Accumulation in 3T3-L1 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bluher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Bersoux, S.; Byun, T.H.; Chaliki, S.S.; Poole, K.G. Pharmacotherapy for obesity: What you need to know. Clevel. Clin. J. Med. 2017, 84, 951–958. [Google Scholar] [CrossRef]
- Markowiak, P.; Slizewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Pique, N.; Berlanga, M.; Minana-Galbis, D. Health Benefits of Heat-Killed (Tyndallized) Probiotics: An Overview. Int. J. Mol. Sci. 2019, 20, 2534. [Google Scholar] [CrossRef]
- Taverniti, V.; Guglielmetti, S. The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: Proposal of paraprobiotic concept). Genes Nutr. 2011, 6, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Inoue, N.; Ozawa, R.; Maekawa, T.; Izumo, T.; Kitagawa, Y.; Kiso, Y.; Shibata, H.; Ikeda, I. Effects of heat-killed Lactobacillus pentosus S-PT84 on postprandial hypertriacylglycerolemia in rats. Biosci. Biotechnol. Biochem. 2013, 77, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Bourrie, B.C.T.; Willing, B.P.; Cotter, P.D. The Microbiota and Health Promoting Characteristics of the Fermented Beverage Kefir. Front. Microbiol. 2016, 7, 647. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, H.; Jeong, D.; Kang, I.B.; Chon, J.W.; Kim, H.S.; Song, K.-Y.; Seo, K.-H. Kefir alleviates obesity and hepatic steatosis in high-fat diet-fed mice by modulation of gut microbiota and mycobiota: Targeted and untargeted community analysis with correlation of biomarkers. J. Nutr. Biochem. 2017, 44, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.; Kim, D.-H.; Kang, I.-B.; Kim, H.; Song, K.-Y.; Kim, H.-S.; Seo, K.-H. Modulation of gut microbiota and increase in fecal water content in mice induced by administration of Lactobacillus kefiranofaciens DN1. Food Funct. 2017, 8, 680–686. [Google Scholar] [CrossRef]
- Kim, D.H.; Jeong, D.; Kang, I.B.; Kim, H.; Song, K.Y.; Seo, K.H. Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: Direct reduction of cholesterol and upregulation of PPAR-alpha in adipose tissue. Mol. Nutr. Food Res. 2017, 61, 1700252–1700260. [Google Scholar] [CrossRef]
- Kim, H.; Bartley, G.E.; Arvik, T.; Lipson, R.; Nah, S.Y.; Seo, K.; Yokoyama, W. Dietary Supplementation of Chardonnay Grape Seed Flour Reduces Plasma Cholesterol Concentration, Hepatic Steatosis, and Abdominal Fat Content in High-Fat Diet-Induced Obese Hamsters. J. Agric. Food Chem. 2014, 62, 1919–1925. [Google Scholar] [CrossRef]
- Seo, K.H.; Kim, D.H.; Jeong, D.; Yokoyama, W.; Kim, H. Chardonnay grape seed flour supplemented diets alter intestinal microbiota in diet-induced obese mice. J. Food Biochem. 2017, 41, e12396–e12404. [Google Scholar] [CrossRef]
- Seo, K.-H.; Bartley, G.E.; Tam, C.; Kim, H.-S.; Kim, D.-H.; Chon, J.-W.; Kim, H.; Yokoyama, W. Chardonnay Grape Seed Flour Ameliorates Hepatic Steatosis and Insulin Resistance via Altered Hepatic Gene Expression for Oxidative Stress, Inflammation, and Lipid and Ceramide Synthesis in Diet-Induced Obese Mice. PLoS ONE 2016, 11, e0167680. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.J.; Lee, H.G.; Seo, K.H.; Yokoyama, W.; Kim, H. Antiobesity Effect of Prebiotic Polyphenol-Rich Grape Seed Flour Supplemented with Probiotic Kefir-Derived Lactic Acid Bacteria. J. Agric. Food Chem. 2018, 66, 12498–12511. [Google Scholar] [CrossRef] [PubMed]
- Guri, A.J.; Hontecillas, R.; Si, H.; Liu, D.; Bassaganya-Riera, J. Dietary abscisic acid ameliorates glucose tolerance and obesity-related inflammation in db/db mice fed high-fat diets. Clin. Nutr. 2007, 26, 107–116. [Google Scholar] [CrossRef]
- Nie, Y.; Luo, F.; Wang, L.; Yang, T.; Shi, L.; Li, X.; Shen, J.; Xu, W.; Guo, T.; Lin, Q. Anti-hyperlipidemic effect of rice bran polysaccharide and its potential mechanism in high-fat diet mice. Food Funct. 2017, 8, 4028–4041. [Google Scholar] [CrossRef]
- Chiellini, C.; Bertacca, A.; Novelli, S.E.; Gorgun, C.Z.; Ciccarone, A.; Giordano, A.; Xu, H.; Soukas, A.; Costa, M.; Gandini, D.; et al. Obesity modulates the expression of haptoglobin in the white adipose tissue via TNFalpha. J. Cell Physiol. 2002, 190, 251–258. [Google Scholar] [CrossRef]
- Elkahoui, S.; Bartley, G.E.; Yokoyama, W.H.; Friedman, M. Dietary Supplementation of Potato Peel Powders Prepared from Conventional and Organic Russet and Non-organic Gold and Red Potatoes Reduces Weight Gain in Mice on a High-Fat Diet. J. Agric. Food Chem. 2018, 66, 6064–6072. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, Z.; Li, Y.; Zhou, L.; Ding, Q.; Xu, L. Isolated exopolysaccharides from Lactobacillus rhamnosus GG alleviated adipogenesis mediated by TLR2 in mice. Sci. Rep. 2016, 6, 36083. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Kwon, J.H.; Lee, H.G.; Seo, K.H.; Kim, H. Combination of Whole Grapeseed Flour and Newly Isolated Kefir Lactic Acid Bacteria Reduces High-Fat-Induced Hepatic Steatosis. Mol. Nutr. Food Res. 2019, 63, e1801040. [Google Scholar] [PubMed]
- Mazloom, K.; Siddiqi, I.; Covasa, M. Probiotics: How Effective Are They in the Fight against Obesity? Nutrients 2019, 11, 258. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Van Hul, M. Novel opportunities for next-generation probiotics targeting metabolic syndrome. Curr. Opin. Biotechnol. 2015, 32, 21–27. [Google Scholar] [CrossRef]
- Hiippala, K.; Jouhten, H.; Ronkainen, A.; Hartikainen, A.; Kainulainen, V.; Jalanka, J.; Satokari, R. The Potential of Gut Commensals in Reinforcing Intestinal Barrier Function and Alleviating Inflammation. Nutrients 2018, 10, 988. [Google Scholar] [CrossRef] [PubMed]
- Kanmani, P.; Kim, H. Protective Effects of Lactic Acid Bacteria Against TLR4 Induced Inflammatory Response in Hepatoma HepG2 Cells Through Modulation of Toll-Like Receptor Negative Regulators of Mitogen-Activated Protein Kinase and NF-kappaB Signaling. Front. Immunol. 2018, 9, 1537. [Google Scholar] [CrossRef]
- Kanmani, P.; Ansari, A.; Villena, J.; Kim, H. Immunobiotics Beneficially Modulate TLR4 Signaling Triggered by Lipopolysaccharide and Reduce Hepatic Steatosis In Vitro. J. Immunol. Res. 2019, 2019, 3876896. [Google Scholar] [CrossRef]
- Vijay, K. Toll-like receptors in immunity and inflammatory diseases: Past, present, and future. Int. Immunopharmacol. 2018, 59, 391–412. [Google Scholar] [CrossRef]
- Lunagariya, N.A.; Patel, N.K.; Jagtap, S.C.; Bhutani, K.K. Inhibitors of Pancreatic Lipase: State of the Art and Clinical Perspectives. EXCLI J. 2014, 13, 897–921. [Google Scholar] [PubMed]
- Lim, J.; Kale, M.; Kim, D.H.; Kim, H.S.; Chon, J.W.; Seo, K.H.; Lee, H.G.; Yokoyama, W.; Kim, H. Antiobesity Effect of Exopolysaccharides Isolated from Kefir Grains. J. Agric. Food Chem. 2017, 65, 10011–10019. [Google Scholar] [CrossRef] [PubMed]
- Wachi, S.; Kanmani, P.; Tomosada, Y.; Kobayashi, H.; Yuri, T.; Egusa, S.; Shimazu, T.; Suda, T.; Aso, H.; Sugawara, M.; et al. Lactobacillus delbrueckii TUA4408L and its extracellular polysaccharides attenuate enterotoxigenic Escherichia coli-induced inflammatory response in porcine intestinal epitheliocytes via Toll-like receptor-2 and 4. Mol. Nutr. Food Res. 2014, 58, 2080–2093. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Lee, S.M.; Li, J.; Tran, G.; Jabri, B.; Chatila, T.A.; Mazmanian, S.K. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 2011, 332, 974–977. [Google Scholar] [CrossRef] [PubMed]
- Castro-Bravo, N.; Wells, J.M.; Margolles, A.; Ruas-Madiedo, P. Interactions of Surface Exopolysaccharides From Bifidobacterium and Lactobacillus Within the Intestinal Environment. Front. Microbiol. 2018, 9, 2426. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.H.; Kim, D.H.; Yokoyama, W.H.; Kim, H. Synbiotic Effect of Whole Grape Seed Flour and Newly Isolated Kefir Lactic Acid Bacteria on Intestinal Microbiota of Diet-Induced Obese Mice. J. Agric. Food Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, R.; Mateus, N.; de Freitas, V. Study of the interaction of pancreatic lipase with procyanidins by optical and enzymatic methods. J. Agric. Food Chem. 2010, 58, 11901–11906. [Google Scholar] [CrossRef]
- Nishiyama, H.; Nagai, T.; Kudo, M.; Okazaki, Y.; Azuma, Y.; Watanabe, T.; Goto, S.; Ogata, H.; Sakurai, T. Supplementation of pancreatic digestive enzymes alters the composition of intestinal microbiota in mice. Biochem. Biophys. Res. Commun. 2018, 495, 273–279. [Google Scholar] [CrossRef]

| Ingredient | CON | 2.5% GSF |
|---|---|---|
| Lard fat | 225 | 225 |
| Soybean oil | 27 | 25 |
| Cholesterol | 0.8 | 0.8 |
| MCC a | 50 | 35.7 |
| GSF b | 0 | 25 |
| Casein | 200 | 200 |
| Corn starch | 91.2 | 82.5 |
| Fructose | 355 | 355 |
| l-cystine | 3 | 3 |
| Choline bitartrate | 3 | 3 |
| Mineral mix | 35 | 35 |
| Vitamin mix | 10 | 10 |
| Total diet (g) | 1000 | 1000 |
| Fat % | 46.7 | 46.8 |
| Protein % | 16.7 | 16.8 |
| Carbohydrate % | 36.6 | 36.4 |
| Gene | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| Aox | GTTGATCACGCACATCTTGG | TGGCTTCGAGTGAGGAAGTT |
| Cd361 | CCGGGCCACGTAGAAAACA | CCTCCAAACACAGCCAGGAC |
| Fasn2 | AGCACTGCCTTCGGTTCAGTC | AAGAGCTGTGGAGGCCACTTG |
| Hp3 | CGAGAAGAAAAACTTGACGA | TCACGTACACACCATACTCAG |
| Wfdc21 | TGAGACCTCTGCAGCTTTTAG | ACAGATGTGACTGCATCCAATA |
| 36B44 | TCTAGGACCCGAGAAGACCTC | GTTGTCAAACACCTGCTGGAT |
| CON | GSF | GSF+HLAB | HLAB | |
|---|---|---|---|---|
| Total cholesterol (mg/dL) | 136 ± 8.8 | 129 ± 5.7 | 118 ± 4.7 | 129 ± 8.1 |
| HDL cholesterol (mg/dL) | 79 ± 2.9 | 74 ± 3.6 | 68 ± 6.4 | 78 ± 5.4 |
| LDL cholesterol (mg/dL) | 17 ± 0.7 | 16 ± 1.0 | 18 ± 2.4 | 17 ± 1.0 |
| Triglycerides (mg/dL) | 99 ± 8.9 a | 78 ± 5.7 a | 72 ± 4.7 b | 88 ± 8.1 a |
| Glucose (mg/dL) | 203 ± 18.6 | 189 ± 18.2 | 209 ± 21.6 | 209 ± 25.8 |
| Bodyweight Gain | Adipose Tissue Weight | Triglycerides | |
|---|---|---|---|
| Fabp4 | 0.57 * | 0.66 # | 0.38 |
| Fasn | 0.46 * | 0.50 * | 0.23 |
| Wfdc21 | 0.66 # | 0.61 # | 0.40 |
| Hp | 0.56 * | 0.79 # | 0.57 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seo, K.-H.; Jeong, J.; Kim, H. Synergistic Effects of Heat-Killed Kefir Paraprobiotics and Flavonoid-Rich Prebiotics on Western Diet-Induced Obesity. Nutrients 2020, 12, 2465. https://doi.org/10.3390/nu12082465
Seo K-H, Jeong J, Kim H. Synergistic Effects of Heat-Killed Kefir Paraprobiotics and Flavonoid-Rich Prebiotics on Western Diet-Induced Obesity. Nutrients. 2020; 12(8):2465. https://doi.org/10.3390/nu12082465
Chicago/Turabian StyleSeo, Kun-Ho, Jaewoon Jeong, and Hyunsook Kim. 2020. "Synergistic Effects of Heat-Killed Kefir Paraprobiotics and Flavonoid-Rich Prebiotics on Western Diet-Induced Obesity" Nutrients 12, no. 8: 2465. https://doi.org/10.3390/nu12082465
APA StyleSeo, K.-H., Jeong, J., & Kim, H. (2020). Synergistic Effects of Heat-Killed Kefir Paraprobiotics and Flavonoid-Rich Prebiotics on Western Diet-Induced Obesity. Nutrients, 12(8), 2465. https://doi.org/10.3390/nu12082465






