COVID-19 Lockdown and Changes of the Dietary Pattern and Physical Activity Habits in a Cohort of Patients with Type 2 Diabetes Mellitus
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
2.2. Study Design
2.3. Data Analysis
2.4. Ethics Approval and Consent to Participate
3. Results and Discussion
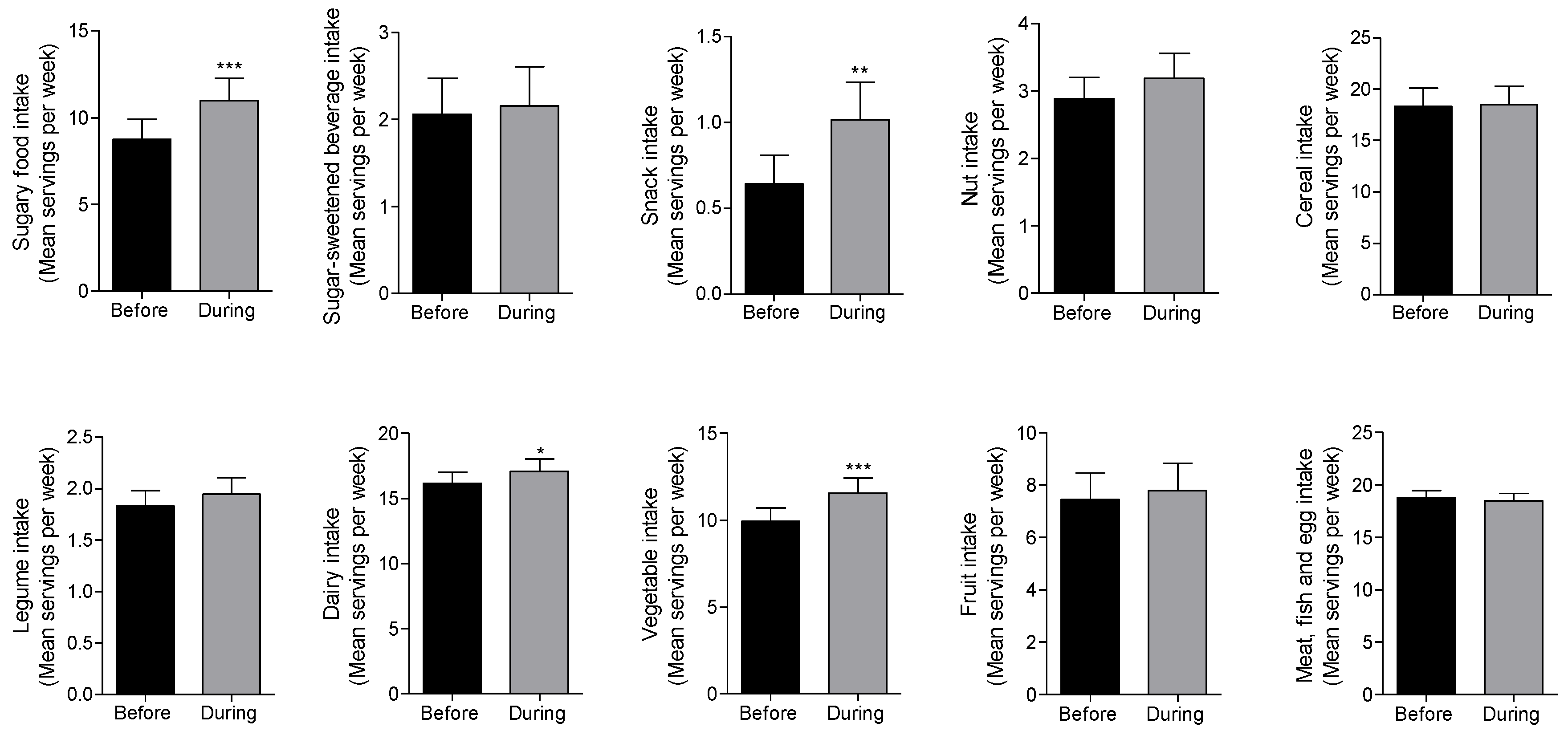
3.1. Changes in Dietary Patterns of Patients with Type 2 Diabetes during the COVID-19 Lockdown
3.2. Changes of Dietary Habits of Patients with Type 2 Diabetes Mellitus According to Their Sociodemographic Characteristics before and during Lockdown
3.3. Gender
3.4. Age
3.5. Body-Mass Index (BMI)
3.6. Capillary Glycated Haemoglobin (HbA1c)
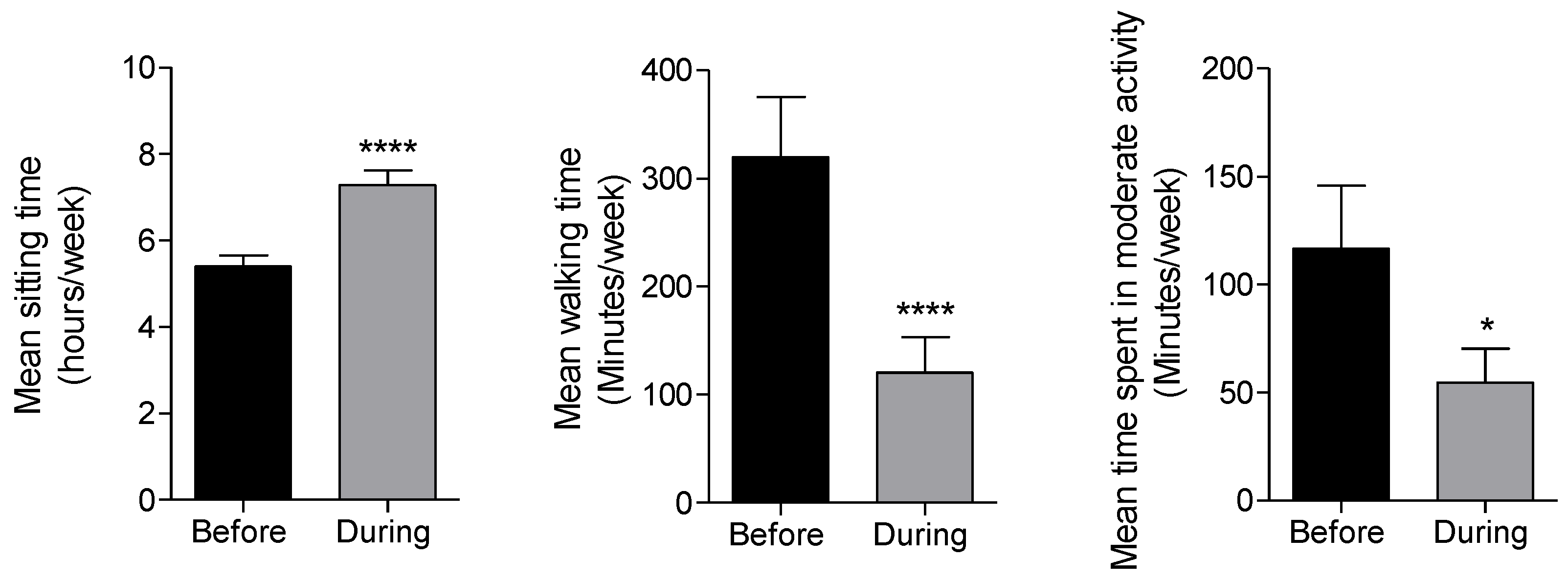
3.7. Changes in Physical Activity Habits during the COVID-19 Lockdown of Patients with T2DM
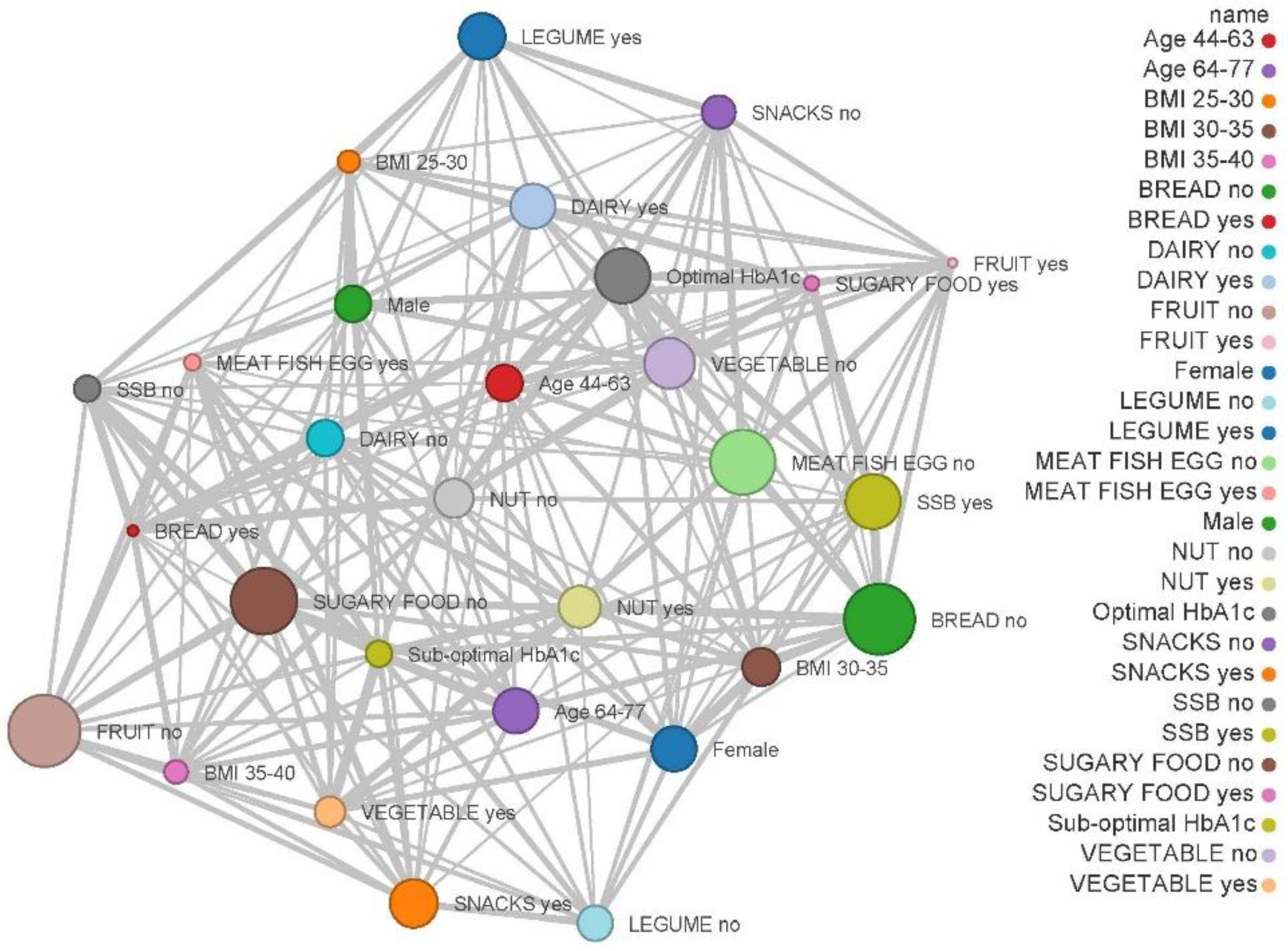
3.8. Relationship between Food Cravings due to the COVID-19 Lockdown and Sociodemographic, Dietary and Physical Activity Characteristics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carlos, W.G.; Dela Cruz, C.S.; Cao, B.; Pasnick, S.; Jamil, S. Novel Wuhan (2019-nCoV) Coronavirus. Am. J. Respir. Crit. Care Med. 2020, 201, P7–P8. [Google Scholar] [CrossRef] [PubMed]
- WHO. Coronavirus Disease (COVID-2019) Situation Reports. Situation Report—155. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200623-covid-19-sitrep-155.pdf?sfvrsn=ca01ebe_2 (accessed on 23 July 2020).
- WHO. Coronavirus Disease (COVID-19) Advice for the Public. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 23 July 2020).
- Peltier, E.; Minder, R. Spain Imposes Nationwide Lockdown to Fight Coronavirus. The New York Times, 15 March 2020. [Google Scholar]
- Academia Española de Nutrición y Dietética, Recomendaciones De Alimentación Y Nutrición Para La Población Española Ante La Crisis Sanitaria Del COVID-19. 2020. Available online: https://academianutricionydietetica.org/NOTICIAS/alimentacioncoronavirus.pdf (accessed on 23 July 2020).
- Lana, R.M.; Coelho, F.C.; Gomes, M.; Cruz, O.G.; Bastos, L.S.; Villela, D.A.M.; Codeco, C.T. The novel coronavirus (SARS-CoV-2) emergency and the role of timely and effective national health surveillance. Cad. Saude Publica 2020, 36, e00019620. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; Barrea, L.; Savastano, S.; Colao, A. Nutritional recommendations for CoVID-19 quarantine. Eur. J. Clin. Nutr. 2020, 74, 850–851. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Yannakoulia, M.; Chan, J.L.; Mantzoros, C.S. Management of the metabolic syndrome and type 2 diabetes through lifestyle modification. Annu. Rev. Nutr. 2009, 29, 223–256. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Yang, L.; Xi, X.; Lin, X.; He, D.; Wang, W. Avoiding hospital-related infections during the COVID-19 epidemic. Crit. Care 2020, 24, 206. [Google Scholar] [CrossRef]
- Holmes, E.A.; O’Connor, R.C.; Perry, V.H.; Tracey, I.; Wessely, S.; Arseneault, L.; Ballard, C.; Christensen, H.; Cohen Silver, R.; Everall, I.; et al. Multidisciplinary research priorities for the COVID-19 pandemic: A call for action for mental health science. Lancet Psychiatry 2020, 7, 547–560. [Google Scholar] [CrossRef]
- Rees, S.; Williams, A. Promoting and supporting self-management for adults living in the community with physical chronic illness: A systematic review of the effectiveness and meaningfulness of the patient-practitioner encounter. JBI Libr. Syst. Rev. 2009, 7, 492–582. [Google Scholar] [CrossRef]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef]
- Galarregui, C.; Zulet, M.A.; Cantero, I.; Marin-Alejandre, B.A.; Monreal, J.I.; Elorz, M.; Benito-Boillos, A.; Herrero, J.I.; Tur, J.A.; Abete, I.; et al. Interplay of Glycemic Index, Glycemic Load, and Dietary Antioxidant Capacity with Insulin Resistance in Subjects with a Cardiometabolic Risk Profile. Int. J. Mol. Sci. 2018, 19, 3662. [Google Scholar] [CrossRef]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef]
- Moreno, S.; Rodriguez, S.; Fernandez, M.C.; Tamez, J.; Cepeda-Benito, A. Clinical validation of the trait and state versions of the Food Craving Questionnaire. Assessment 2008, 15, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Escobar, M.; Martinez-Uribe, L. Network Coincidence Analysis: The netCoin R Package. J. Stat. Softw. 2020, 93, 32. [Google Scholar] [CrossRef]
- Moynihan, A.B.; van Tilburg, W.A.; Igou, E.R.; Wisman, A.; Donnelly, A.E.; Mulcaire, J.B. Eaten up by boredom: Consuming food to escape awareness of the bored self. Front. Psychol. 2015, 6, 369. [Google Scholar] [CrossRef] [PubMed]
- WHO. Mental Health and COVID-19. Available online: https://www.who.int/teams/mental-health-and-substance-use/covid-19 (accessed on 23 July 2020).
- Ruiz-Roso, M.B.; de Carvalho Padilha, P.; Mantilla-Escalante, D.C.; Ulloa, N.; Brun, P.; Acevedo-Correa, D.; Arantes Ferreira Peres, W.; Martorell, M.; Aires, M.T.; de Oliveira Cardoso, L.; et al. Covid-19 Confinement and Changes of Adolescent’s Dietary Trends in Italy, Spain, Chile, Colombia and Brazil. Nutrients 2020, 12, 1807. [Google Scholar] [CrossRef] [PubMed]
- Centre for the Promotion of Imports from Developing Countries. All hands on deck for the fresh sector (COVID-19). 2020. Available online: https://www.cbi.eu/news/all-hands-deck-fresh-sector-covid-19 (accessed on 23 July 2020).
- Yamamoto, J.; Ishihara, J.; Kotemori, A.; Yoshizaki, T.; Goto, A.; Mizoue, T.; Noda, M.; Sawada, N.; Tsugane, S. Association Between Okinawan Vegetables Consumption and Risk of Type 2 Diabetes in Japanese Communities: The JPHC Study. J. Epidemiol. 2020, 30, 227–235. [Google Scholar] [CrossRef]
- Del Rio-Celestino, M.; Font, R. The Health Benefits of Fruits and Vegetables. Foods 2020, 9, 369. [Google Scholar] [CrossRef]
- WHO. Food and Nutrition Tips during Self-Quarantine. 2020. Available online: https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/technical-guidance/food-and-nutrition-tips-during-self-quarantine (accessed on 23 July 2020).
- Forouhi, N.G. Association between consumption of dairy products and incident type 2 diabetes—Insights from the European Prospective Investigation into Cancer study. Nutr. Rev. 2015, 73, 15–22. [Google Scholar] [CrossRef]
- O’Connor, L.M.; Lentjes, M.A.H.; Luben, R.N.; Khaw, K.-T.; Wareham, N.J.; Forouhi, N.G. Dietary dairy product intake and incident type 2 diabetes: A prospective study using dietary data from a 7-day food diary. Diabetologia 2014, 57, 909–917. [Google Scholar] [CrossRef]
- Conserjería de Sanidad, Nutritional Recommendations from the Health Service of Madrid. 2013. Available online: http://www.madrid.org/cs/Satellite?blobcol=urldata&blobheader=application%2Fpdf&blobheadername1=Content-disposition&blobheadername2=cadena&blobheadervalue1=filename%3DRecomendaciones+dietetico+nutricionales.pdf&blobheadervalue2=language%3Des%26site%3DHospitalRamonCajal&blobkey=id&blobtable=MungoBlobs&blobwhere=1352862624022&ssbinary=true (accessed on 23 July 2020).
- Grzymislawska, M.; Puch, E.A.; Zawada, A.; Grzymislawski, M. Do nutritional behaviors depend on biological sex and cultural gender? Adv. Clin. Exp. Med. 2020, 29, 165–172. [Google Scholar] [CrossRef]
- Fidler Mis, N.; Braegger, C.; Bronsky, J.; Campoy, C.; Domellof, M.; Embleton, N.D.; Hojsak, I.; Hulst, J.; Indrio, F.; Lapillonne, A.; et al. Sugar in Infants, Children and Adolescents: A Position Paper of the European Society for Paediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 681–696. [Google Scholar] [CrossRef]
- Selvin, E.; Parrinello, C.M. Age-related differences in glycaemic control in diabetes. Diabetologia 2013, 56, 2549–2551. [Google Scholar] [CrossRef] [PubMed]
- Sundararajan, K.; Campbell, M.K.; Choi, Y.H.; Sarma, S. The relationship between diet quality and adult obesity: Evidence from Canada. J. Am. Coll. Nutr. 2014, 33, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Drenowatz, C.; Shook, R.P.; Hand, G.A.; Hébert, J.R.; Blair, S.N. The independent association between diet quality and body composition. Sci. Rep. 2014, 4, 4928. [Google Scholar] [CrossRef] [PubMed]
- Haimoto, H.; Watanabe, S.; Komeda, M.; Wakai, K. The impact of carbohydrate intake and its sources on hemoglobin A1c levels in Japanese patients with type 2 diabetes not taking anti-diabetic medication. Diabetes Metab. Syndr. Obes. 2018, 11, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Leitzmann, M.; Tonstad, S.; Vatten, L.J. Physical activity and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis. Eur. J. Epidemiol. 2015, 30, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Umpierre, D.; Ribeiro, P.A.; Kramer, C.K.; Leitao, C.B.; Zucatti, A.T.; Azevedo, M.J.; Gross, J.L.; Ribeiro, J.P.; Schaan, B.D. Physical activity advice only or structured exercise training and association with HbA1c levels in type 2 diabetes: A systematic review and meta-analysis. JAMA 2011, 305, 1790–1799. [Google Scholar] [CrossRef] [PubMed]
- Bernard, P.; Dore, I.; Romain, A.J.; Hains-Monfette, G.; Kingsbury, C.; Sabiston, C. Dose response association of objective physical activity with mental health in a representative national sample of adults: A cross-sectional study. PLoS ONE 2018, 13, e0204682. [Google Scholar] [CrossRef]
- The Lancet Public Health. Time to tackle the physical activity gender gap. Lancet Public Health 2019, 4, e360. [Google Scholar] [CrossRef]
- Naja, F.; Hamadeh, R. Nutrition amid the COVID-19 pandemic: A multi-level framework for action. Eur. J. Clin. Nutr. 2020, 1–5. [Google Scholar] [CrossRef]
- Watson, N.A.; Dyer, K.A.; Buckley, J.D.; Brinkworth, G.D.; Coates, A.M.; Parfitt, G.; Howe, P.R.C.; Noakes, M.; Murphy, K.J. Reductions in food cravings are similar with low-fat weight loss diets differing in protein and carbohydrate in overweight and obese adults with type 2 diabetes: A randomized clinical trial. Nutr. Res. 2018, 57, 56–66. [Google Scholar] [CrossRef]
- Boswell, R.G.; Kober, H. Food cue reactivity and craving predict eating and weight gain: A meta-analytic review. Obes. Rev. 2016, 17, 159–177. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | Sample (%) |
|---|---|
| Gender (n = 72) | |
| Male | 48.6 |
| Female | 51.4 |
| Age (years) (n = 72) | |
| 44 to 63 | 48.6 |
| 64 to 77 | 51.4 |
| Body-mass-index (kg/m2) (n = 63) | |
| 25.0 to <30 | 16.7 |
| 30 to <35 | 63.0 |
| 35 to <40 | 20.4 |
| Capillary HbA1c (n = 59) | |
| <6.5% | 69.5 |
| ≥6.5% | 30.5 |
| Gender (n = 72) | Age (Years) (n = 72) | Body-Mass-Index (kg/m2) (n = 63) | Capillary HbA1c (n = 59) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | 44 to 63 | 64 to 77 | 25.0 to <30 | 30 to <35 | 35 to <40 | <6.5% | ≥6.5% | ||
| Sugary food | Mean (95% CI) before lockdown | 10.1 (6.1–14.1) | 7.2 (4.6–9.7) | 10.2 (6.1–14.3) | 8.3 (5.2–11.4) | 11.7 (4.2–19.1) | 7.9 (4.3–11.5) | 9.3 (5.2–13.3) | 7.8 (5.1–10.5) | 13.1 (6.4–19.8) |
| Mean (95% CI) during lockdown | 12.3 (7.6–16.9) | 9.4 (6.8–12.0) | 14.2 (9.3–19.0) | 9.2 (6.3–12.1) | 13.9 (5.0–22.8) | 9.7 (6.2–13.2) | 12.9 (8.3–17.5) | 10.3 (7.1–13.6) | 14.7 (7.8–21.6) | |
| p-value | 0.0013 ** | 0.0036 ** | <0.0001 **** | 0.1035 | 0.0649 | 0.0307 * | 0.001 *** | <0.0001 **** | 0.1337 | |
| Interaction p-value | 0.96 | 0.003 ** | 0.69 | 0.4 | ||||||
| Snacks | Mean (95% CI) before lockdown | 0.57 (0.1–1.1) | 0.7 (0.3–1.2) | 0.9 (0.5–1.5) | 0.3 (0.1–0.5) | 0.4 (−0.1–0.8) | 0.9 (0.3–1.5) | 0.3 (−0.04–0.6) | 0.7 (0.3–1.1) | 0.3 (−0.1–0.5) |
| Mean (95% CI) during lockdown | 0.7 (0.2–1.3) | 1.3 (0.6–1.9) | 1.5 (0.7–2.4) | 0.5 (0.2–0.8) | 0.7 (0.1–1.5) | 1.3 (0.5–2.1) | 0.7 (−0.02–1.3) | 1.1 (0.5–1.6) | 0.6 (−0.1–1.3) | |
| p-value | 0.07 | 0.0025 ** | 0.0037 ** | 0.0326 | 0.1362 | 0.0133 * | 0.0896 | 0.0046** | 0.1106 | |
| Interaction p-value | 0.097 | 0.078 | 0.85 | 0.94 | ||||||
| Sugar-sweetened beverages (SSB) | Mean (95% CI) before lockdown | 2.3 (1.0–3.6) | 1.8 (0.7–2.8) | 2.6 (1.1–4.1) | 1.2 (0.4–2.0) | 1.9 (0.5–3.3) | 2.4 (0.7–4.0) | 1.3 (0.2–2.3) | 1.6 (0.5–2.7) | 2.2 (0.7–3.7) |
| Mean (95% CI) during lockdown | 2.6 (1.1–4.1) | 1.7 (0.6–2.7) | 2.6 (1.1–4.1) | 1.4 (0.3–2.5) | 2.2 (0.1–4.3) | 2.5 (0.8–4.2) | 1.2 (0.1–2.3) | 1.5 (0.4–2.5) | 2.9 (1.0–4.9). | |
| p-value | 0.2985 | 0.3242 | >0.9999 | 0.4452 | 0.6165 | 0.3265 | 0.8474 | 0.1643 | 0.105 | |
| Interaction p-value | 0.19 | 0.49 | 0.47 | 0.009 ** | ||||||
| Diary | Mean (95% CI) before lockdown | 18.3 (14.7–20.8) | 14.5 (12.2–16.8) | 15.7 (13.3–18.2) | 15.5 (13.2–17.9) | 14.3 (11.6–17.1) | 16.0 (13.2–18.8) | 16.9 (13.6–20.2) | 16.3 (14.0–18.5) | 15.2 (12.5–18.0) |
| Mean (95% CI) during lockdown | 19.0 (16.29–21.7) | 15.5 (12.9–18.2) | 16.3 (16.6–18.9) | 16.9 (14.1–19.8) | 14.5 (11.9–17.2) | 17.5 (14.1–20.8) | 17.8 (14.0–24.6) | 17.5 (14.8–20.2) | 15.7 (13.1–18.2) | |
| p-value | 0.0871 | 0.0514 | 0.1431 | 0.0335 | 0.486 | 0.0275 * | 0.2513 | 0.0342 * | 0.2698 | |
| Interaction p-value | 0.7 | 0.26 | 0.4 | 0.38 | ||||||
| Legume | Mean (95% CI) before lockdown | 2.0 (1.5–2.4) | 1.7 (1.3–2.1) | 1.8 (1.4–2.3) | 1.8 (1.3–2.3) | 2.1 (1.5–2.7) | 1.7 (1.3–2.1) | 1.9 (1.0–2.7) | 1.9 (1.5–2.4) | 1.7 (1.1–2.3) |
| Mean (95% CI) during lockdown | 2.1 (1.5–2.4) | 1.8 (1.4–2.3) | 1.8 (1.3–2.3) | 2.0 (1.5–2.5) | 2.4 (1.6–3.2) | 1.7 (1.3–2.1) | 2.0 (1.1–2.8) | 2.0 (1.6–2.5) | 1.9 (1.2–2.5) | |
| p-value | 0.1835 (1.6–2.6) | 0.1687 | 0.6621 | 0.0506 | 0.0961 | 0.6632 | 0.3299 | 0.2099 | 0.1631 | |
| Interaction p-value | 0.65 | 0.16 | 0.25 | 0.43 | ||||||
| Vegetable | Mean (95% CI) before lockdown | 9.1 (6.7–11.4) | 10.7 (8.7–12.8) | 9.1 (6.9–11.3) | 11.5 (9.1–14.0) | 13.2 (9.4–17.0) | 8.9 (6.6–11.2) | 10.3 (6.9–13.6) | 9.2 (7.2–11.2) | 12.7 (8.9–16.4) |
| Mean (95% CI) during lockdown | 10.8 (8.1–13.5) | 12.2 (9.9–14.4) | 10.8 (8.2–13.4) | 13.3 (10.7–16.0) | 14.6 (9.6–19.6) | 10.7 (8.1–13.3) | 12.4 (9.1–15.6) | 10.6 (8.4–12.8) | 15.4 (11.3–19.5) | |
| p-value | 0.0049 ** | 0.001 ** | 0.003 ** | 0.0033 ** | 0.0707 | 0.0018 ** | 0.0279 * | 0.0071 ** | 0.0029 ** | |
| Interaction p-value | 0.7 | 0.74 | 0.7 | 0.14 | ||||||
| Fruit | Mean (95% CI) before lockdown | 7.7 (4.8–5.4) | 7.4 (4.6–10.2) | 9.1 (5.7–12.6) | 6.9 (4.2–9.7) | 10.7 (4.5–17.0) | 8.3 (5.1–11.6) | 5.4 (2.2–8.5) | 7.5 (4.6–10.3) | 9.5 (4.9–14.1) |
| Mean (95% CI) during lockdown | 8.5 (10.7–11.6) | 7.5 (4.6–10.39 | 9.7 (6.2–13.2) | 7.2 (4.4–9.9) | 11.4 (5.3–17.5) | 8.9 (5.6–12.3) | 5.3 (2.1–8.4) | 7.7 (4.7–10.6) | 10.2 (5.7–14.6) | |
| p-value | 0.0133 * | 0.744 | 0.0608 | 0.4508 | 0.1264 | 0.0577 | 0.7894 | 0.4484 | 0.0617 | |
| Interaction p-value | 0.075 | 0.81 | 0.42 | 0.3 | ||||||
| Nut | Mean (95% CI) before lockdown | 2.9 (1.9–3.8) | 2.8 (2.0–3.7) | 3.4 (2.4–4.3) | 2.6 (1.7–3.5) | 2.2 (0.9–3.5) | 3.6 (2.6–4.7) | 2.7 (1.3–4.0) | 2.9 (2.0–3.8) | 3.1 (1.8–4.4) |
| Mean (95% CI) during lockdown | 3.4 (2.2–4.8) | 2.9 (1.9–3.8) | 3.5 (2.5–4.6) | 2.9 (1.9–3.9) | 3.4 (1.8–5.0) | 3.4 (2.3–4.6) | 2.8 (1.4–4.1) | 3.0 (2.1–4.0) | 3.7 (2.5–5.1) | |
| p-value | 0.0974 | 0.7111 | 0.2636 | 0.3248 | 0.0335 * | 0.2835 | 0.577 | 0.5119 | 0.2355 | |
| Interaction p-value | 0.17 | 0.67 | 0.0032 ** | 0.23 | ||||||
| Meat. Fish and Egg | Mean (95% CI) before lockdown | 18.0 (16.0–19.9) | 19.4 (14.5–21.4) | 20.0 (17.9- 22.3) | 17.7 (15.7–19.7) | 18.9 (16.0–21.9) | 18.9 (16.3–24.6) | 18.2 (15.6–20.7) | 19.5 (15.6–21.3) | 16.9 (13.8–20.0) |
| Mean (95% CI) during lockdown | 17.8 (15.1–19.9) | 19.1 (17.2–21.0) | 19.9 (17.7–22.2) | 17.3 (15.2–19.4) | 19.8 (16.0–23.6) | 19.0 (16.6–24.5) | 16.8 (14.1–19.5) | 18.9 (17.1–20.8) | 17.3 (13.5–21.0) | |
| p-value | 0.7255 | 0.5203 | 0.8217 | 0.4622 | 0.2625 | 0.8567 | 0.0388 * | 0.2704 | 0.6352 | |
| Interaction p-value | 0.82 | 0.94 | 0.022 ** | 0.31 | ||||||
| Cereal | Mean (95% CI) before lockdown | 17.7 (12.3–23.2) | 18.6 (14.2–23.1) | 17.8 (12.5.−23.1) | 20.0 (14.4–25.6) | 21.8 (108–32.8) | 16.9 (12.1–21.8) | 17.2 (11.6–22.7) | 16.1 (12.5–20.0) | 20.8 (13.3–28.3) |
| Mean (95% CI) during lockdown | 19.7 (13.8–25.7) | 17.0 (13.0–21.1) | 18.5 (12.3–24.5) | 19.3 (14.2–24.4) | 23.7 (11.3–36.2) | 17.1 (12.9–21.4) | 15.4 (10.4–20.4) | 15.8 (12.6–19.0) | 21.9 (13.2–30.5) | |
| p-value | 0.0585 | 0.2806 | 0.5591 | 0.6729 | 0.3852 | 0.895 | 0.3906 | 0.7338 | 0.7126 | |
| Interaction p-value | 0.049 * | 0.66 | 0.45 | 0.55 | ||||||
| Gender (n = 72) | Age (Years) (n = 72) | Body-Mass-Index (kg/m2) (n = 63) | Capillary HbA1c (n = 59) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | 44 to 63 | 64 to 77 | 25.0 to <30 | 30 to <35 | 35 to <40 | <6.5% | ≥6.5% | ||
| Moderate physical activity | Mean (95% CI) before lockdown | 60.5 (30.9–90.1) | 44.5 (26.4–62.5) | 190.6 (70.4–310.8) | 68.2 (15.0–121.5) | 81.3 (−39.1–201.8) | 102.9 (44.9–161.0) | 124.0 (15.6–232.4) | 125.7 (55.6–195.8) | 57.2 (4.1–110.3) |
| Mean (95% CI) during lockdown | 20.6 (8.5–35.3) | 21.2 (3.9–38.6) | 33.8 (6.9–60.7) | 48.4 (13.2–83.6) | 32.7 (−6.4–71.8) | 60.9 (18.9–102.9) | 26.3 (−9.0–61.5) | 43.6 (15.6–71.6) | 40.3 (−10.6–91.1) | |
| t-test p-value | 0.0007 *** | 0.0005 *** | 0.0119 * | 0.3694 | 0.2803 | 0.0658 | 0.0933 | 0.0118 * | 0.5591 | |
| Interaction p-value | 0.13 | 0.027 * | 0.56 | 0.2 | ||||||
| Walking (min/week) | Mean (95% CI) before lockdown | 363.2 (178.5–547.9) | 268.4 (140.1–396.) | 231.9 (147–316.8) | 339.9 | 434.7 (195.2–647.1) | 270.4 (110.6–430.3) | 214.8 (119.2–3.10). | 291.3 (165.8–416.8) | 289.5 (124.0–455.0) |
| Mean (95% CI) during lockdown | 108.8 (37.6–180) | 128.3 (16.5–240.1) | 89.7 (16.2–163.2) | 124.8 | 138.0 (−24.3–300.3) | 113.3 (−19.8–246.3) | 95.9 (25.0–166.7) | 127.2 (26.28–228.1) | 85.4 (−12.9–183.7) | |
| t-test p-value | 0.0006 *** | 0.0005 *** | 0.0007 *** | <0.0001 **** | 0.0037 ** | 0.0002 *** | 0.0142 * | 0.0003 *** | 0.0004 *** | |
| Interaction p-value | 0.14 | 0.077 * | 0.13 | 0.56 | ||||||
| Sitting (hours/week) | Mean (95% CI) before lockdown | 5.2 (4.6–5.6) | 5.6 (4.8–6.3) | 5.9 (5.1–6.7) | 5.2 (4.5–5.9) | 6.0 (5.0–7.0) | 5.2 (4.3–6.1) | 5.4 (4.5–6.2) | 5.4 (4.7–6.0) | 5.0 (4.2–5.9) |
| Mean (95% CI) during lockdown | 7.7 (6.9–8.5)3 | 6.8 (5.6–8.0) | 7.3 (6.0–8.6) | 7.3 (6.4–8.2) | 7.5 (5.9–9.0) | 6.7 (5.7–7.7) | 7.2 (6.1–8.1). | 6.9 (6.2–7.6) | 7.2 (6.0–8.5) | |
| t-test p-value | <0.0001 **** | 0.0213* | 0.0332 * | <0.0001 **** | 0.0516 | 0.001 ** | 0.0043** | <0.0001 **** | 0.0002 *** | |
| Interaction p-value | 0.07 | 0.35 | 0.52 | 0.25 | ||||||
| Variables | FCQ-T | FCQ-S | ||
|---|---|---|---|---|
| Correlation | p-Value | Correlation | p-Value | |
| Gender | 0.227 | 0.018 | −0.207 | 0.040 |
| Age | −0.110 | 0.193 | −0.069 | 0.292 |
| Body-mass-index (kg/m2) | 0.229 | 0.035 | −0.284 | 0.012 |
| Capillary HbA1c | 0.176 | 0.091 | 0.265 | 0.021 |
| Sugary food intake | 0.093 | 0.220 | 0.082 | 0.248 |
| Snacks food intake | 0.227 | 0.029 | 0.163 | 0.089 |
| SSB intake | 0.149 | 0.110 | 0.150 | 0.108 |
| Diary intake | −0.221 | 0.032 | −0.221 | 0.032 |
| Legume intake | −0.176 | 0.073 | −0.153 | 0.103 |
| Vegetable intake | 0.308 | 0.005 | 0.417 | 0.0001 |
| Fruit intake | 0.210 | 0.039 | −0.029 | 0.406 |
| Nut intake | 0.043 | 0.362 | −0.027 | 0.413 |
| Meat, fish and egg intake | −0.142 | 0.120 | −0.222 | 0.032 |
| Cereal intake | 0.000 | 0.500 | 0.460 | 0.012 |
| Moderate activity | 0.132 | 0.136 | 0.180 | 0.066 |
| Walking time | 0.109 | 0.184 | 0.203 | 0.044 |
| Sitting time | −0.085 | 0.242 | −0.166 | 0.085 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Roso, M.B.; Knott-Torcal, C.; Matilla-Escalante, D.C.; Garcimartín, A.; Sampedro-Nuñez, M.A.; Dávalos, A.; Marazuela, M. COVID-19 Lockdown and Changes of the Dietary Pattern and Physical Activity Habits in a Cohort of Patients with Type 2 Diabetes Mellitus. Nutrients 2020, 12, 2327. https://doi.org/10.3390/nu12082327
Ruiz-Roso MB, Knott-Torcal C, Matilla-Escalante DC, Garcimartín A, Sampedro-Nuñez MA, Dávalos A, Marazuela M. COVID-19 Lockdown and Changes of the Dietary Pattern and Physical Activity Habits in a Cohort of Patients with Type 2 Diabetes Mellitus. Nutrients. 2020; 12(8):2327. https://doi.org/10.3390/nu12082327
Chicago/Turabian StyleRuiz-Roso, María Belén, Carolina Knott-Torcal, Diana C. Matilla-Escalante, Alba Garcimartín, Miguel A. Sampedro-Nuñez, Alberto Dávalos, and Mónica Marazuela. 2020. "COVID-19 Lockdown and Changes of the Dietary Pattern and Physical Activity Habits in a Cohort of Patients with Type 2 Diabetes Mellitus" Nutrients 12, no. 8: 2327. https://doi.org/10.3390/nu12082327
APA StyleRuiz-Roso, M. B., Knott-Torcal, C., Matilla-Escalante, D. C., Garcimartín, A., Sampedro-Nuñez, M. A., Dávalos, A., & Marazuela, M. (2020). COVID-19 Lockdown and Changes of the Dietary Pattern and Physical Activity Habits in a Cohort of Patients with Type 2 Diabetes Mellitus. Nutrients, 12(8), 2327. https://doi.org/10.3390/nu12082327







