Non-IgE-Mediated Gastrointestinal Food Allergies in Children: An Update
Abstract
1. Introduction
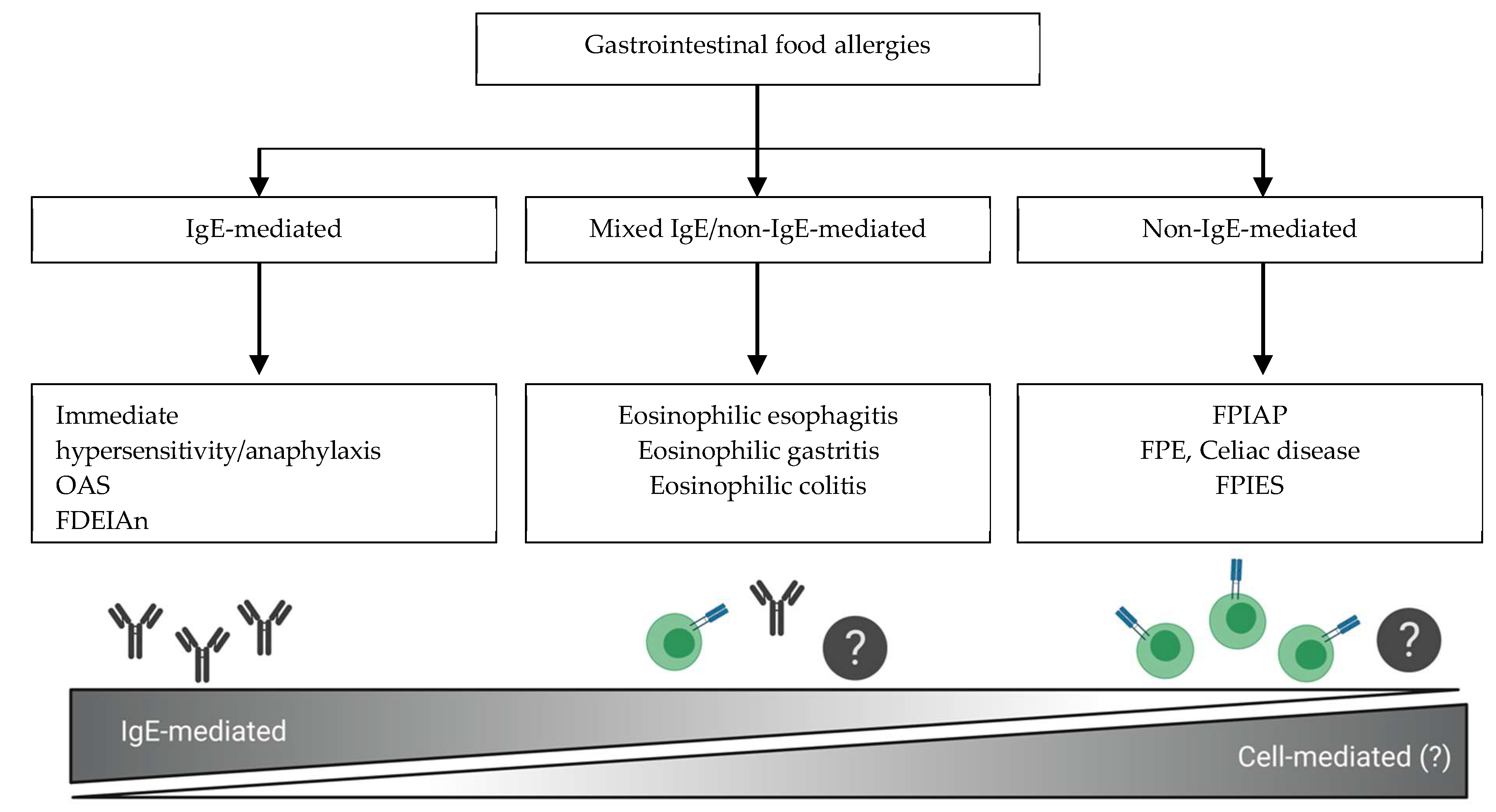
2. Classification and Terminology
3. Epidemiology
4. Pathophysiology
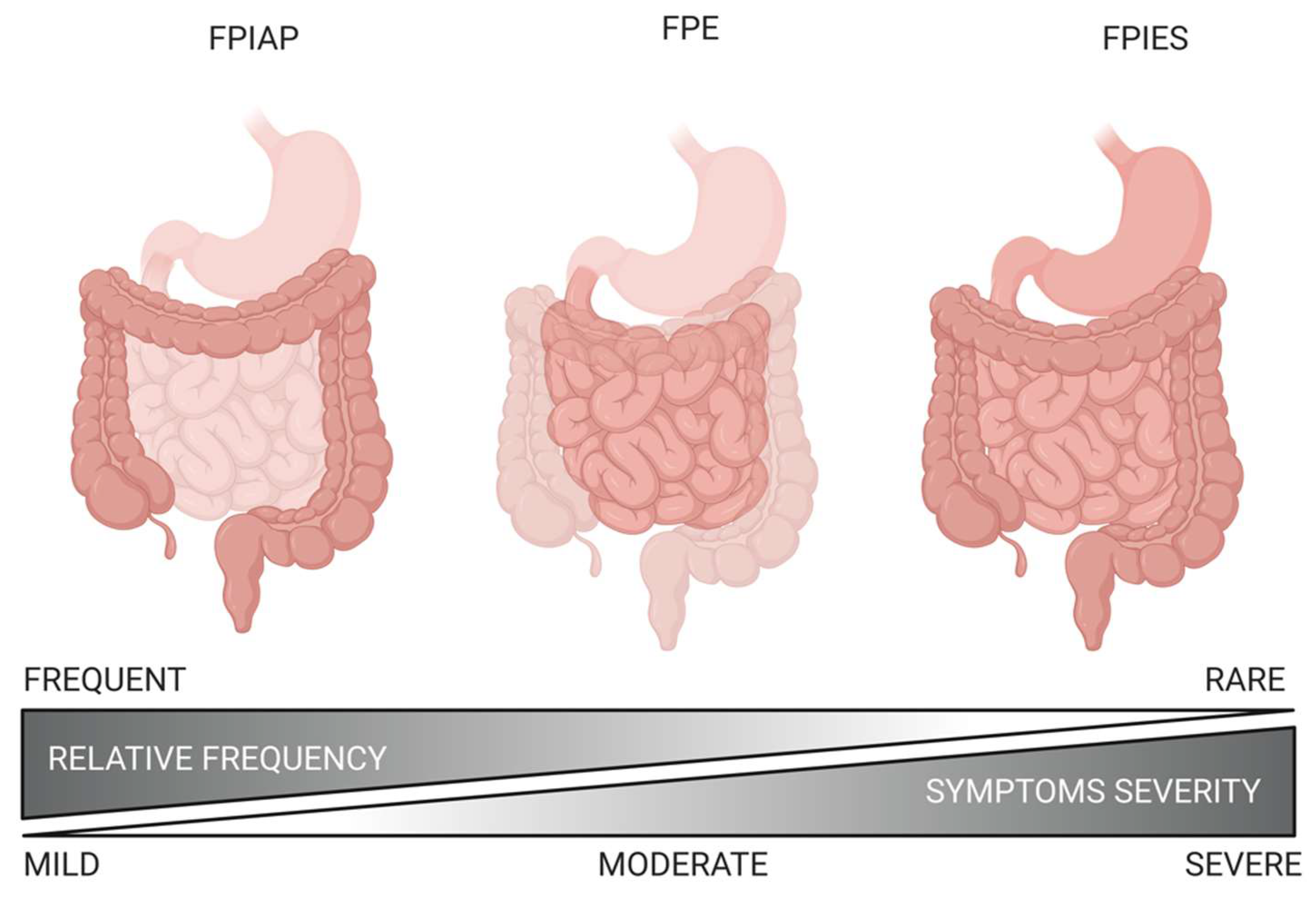
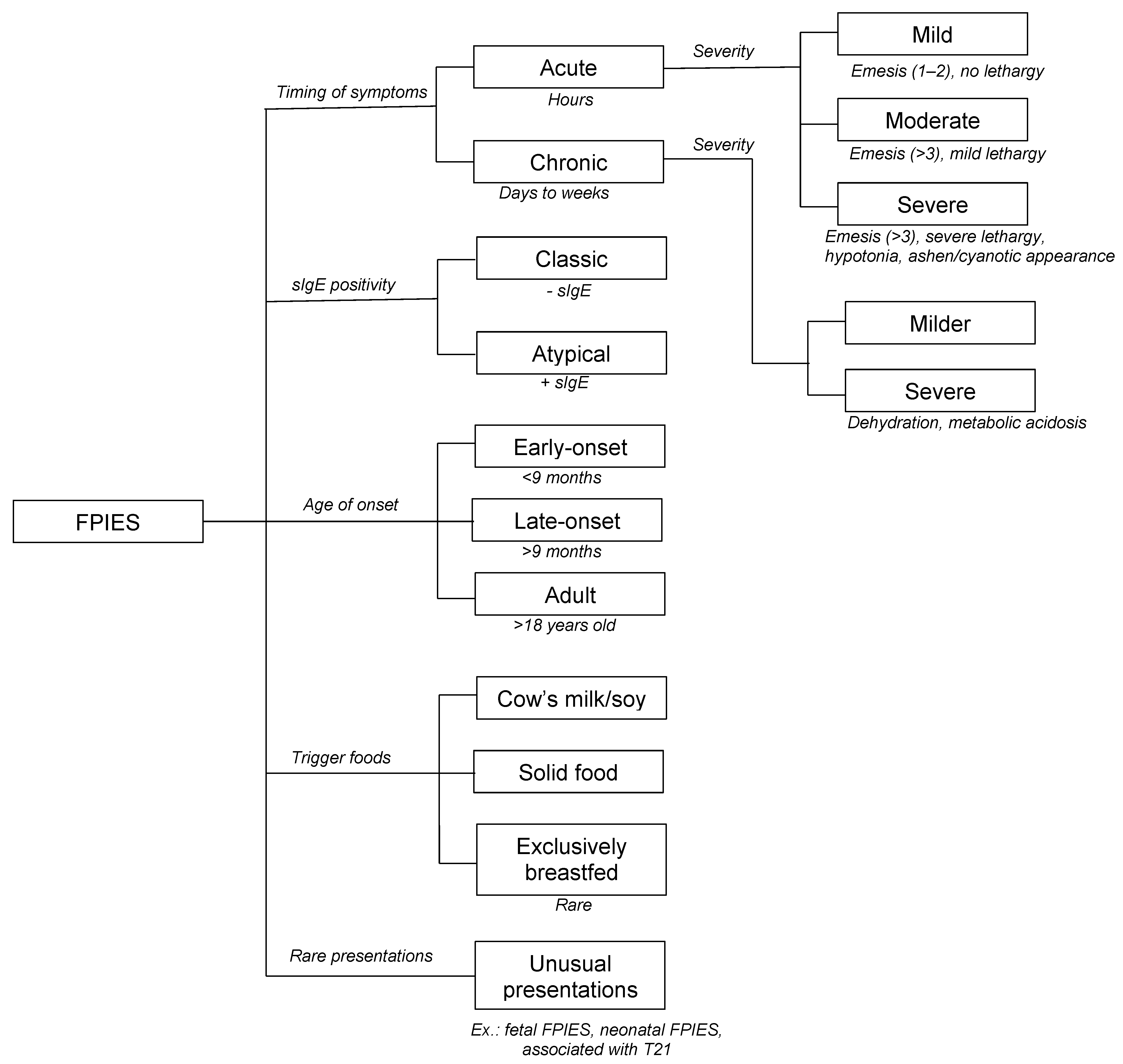
5. Clinical Manifestations
6. Reported Food Triggers
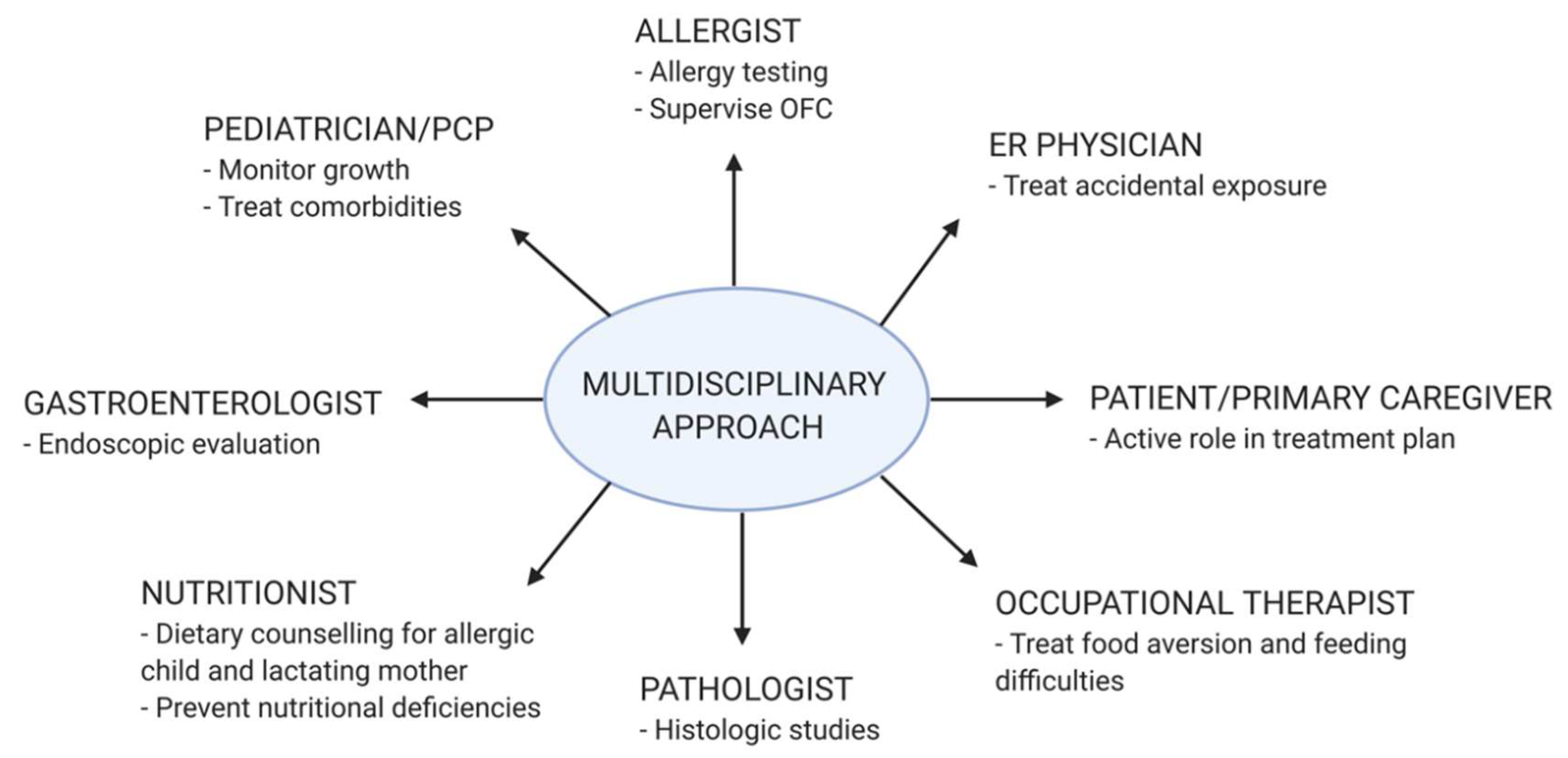
7. Diagnosis
8. Oral Food Challenge
9. Paraclinical Investigations and Biomarkers
9.1. Laboratory Findings
9.2. Allergy Testing
9.3. Stool Studies
9.4. Radiologic Evaluation
9.5. Endoscopic Evaluation
9.6. Other Biomarkers
10. Differential Diagnosis
11. Natural History
12. Management
12.1. Food Elimination
12.2. Nutritional Impact
12.3. Culprit Food Reintroduction
12.4. Introduction of Weaning Foods
13. Quality of Life
14. Role of Food Allergens in Other Common Pediatric Gastrointestinal Disorders
15. Future Perspectives
16. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| APT | atopy patch test |
| CRP | C reactive protein |
| FODMAPS | fermentable oligosaccharides, disaccharides, monosaccharides and polyols |
| FPE | food protein-induced enteropathy |
| FPIAP | food protein-induced allergic proctocolitis |
| FPIES | food protein-induced enterocolitis syndrome |
| FTT | failure to thrive |
| GERD | gastroesophageal reflux disease |
| IBS | irritable bowel syndrome |
| IgE | immunoglobulin E |
| IgE-GA | IgE-mediated food allergy |
| LTT | lymphocyte transformation test |
| MPV | mean platelet volume |
| NEC | necrotizing enterocolitis |
| non-IgE-GI-FA | non-immunoglobulin E-mediated gastrointestinal food allergic disorders |
| OFC | oral food challenge |
| QoL | quality of life |
| sIgE | serum food-specific IgE |
References
- Heine, R.G. Gastrointestinal Food Allergies. Chem. Immunol. Allergy 2015, 101, 171–180. [Google Scholar] [CrossRef]
- Fenton, M. Guidelines for the diagnosis and management of food allergy in the United States. Clin. Transl. Allergy 2011, 1, S10. [Google Scholar] [CrossRef][Green Version]
- Sampson, H.A.; Aceves, S.; Bock, S.A.; James, J.; Jones, S.; Lang, D.; Nadeau, K.; Nowak-Węgrzyn, A.; Oppenheimer, J.; Perry, T.T.; et al. Food allergy: A practice parameter update—2014. J. Allergy Clin. Immunol. 2014, 134, 1016–1025.e43. [Google Scholar] [CrossRef] [PubMed]
- Comberiati, P.; Landi, M.; Martelli, A.; Piacentini, G.; Capristo, C.; Paiola, G.; Peroni, D. Awareness of allergic enterocolitis among primary-care paediatricians: A web-based pilot survey. Allergol. et Immunopathol. 2016, 44, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Greenhawt, M.; Bird, J.A.; Nowak-Wegrzyn, A. Trends in Provider Management of Patients with Food Protein–Induced Enterocolitis Syndrome. J. Allergy Clin. Immunol. Pr. 2017, 5, 1319–1324.e12. [Google Scholar] [CrossRef] [PubMed]
- Tordesillas, L.; Berin, M.C.; Sampson, H.A. Immunology of Food Allergy. Immunity 2017, 47, 32–50. [Google Scholar] [CrossRef]
- Waserman, S.; Bégin, P.; Watson, W. IgE-mediated food allergy. Allergy, Asthma Clin. Immunol. 2018, 14, 55. [Google Scholar] [CrossRef]
- Burks, A.W.; Tang, M.; Sicherer, S.; Muraro, A.; Eigenmann, P.A.; Ebisawa, M.; Fiocchi, A.; Chiang, W.; Beyer, K.; Wood, R.; et al. ICON: Food allergy. J. Allergy Clin. Immunol. 2012, 129, 906–920. [Google Scholar] [CrossRef]
- Koutri, E.; Papadopoulou, A. Eosinophilic Gastrointestinal Diseases in Childhood. Ann. Nutr. Metab. 2018, 73, 18–28. [Google Scholar] [CrossRef]
- Jean-Christoph, C.; Szajewska, H.; Shamir, R.; Nowak-Wegrzyn, A. Non-IgE-mediated gastrointestinal food allergies in children. Pediatr. Allergy Immunol. 2016, 28, 6–17. [Google Scholar] [CrossRef]
- Elizur, A.; Cohen, M.; Goldberg, M.R.; Rajuan, N.; Cohen, A.; Leshno, M.; Katz, Y. Cow’s milk associated rectal bleeding: A population based prospective study. Pediatr. Allergy Immunol. 2012, 23, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.M.; Virkud, Y.V.; Seay, H.; Hickey, A.; Ndahayo, R.; Rosow, R.; Southwick, C.; Elkort, M.; Gupta, B.; Kramer, E.; et al. Prospective Assessment of Pediatrician-Diagnosed Food Protein–Induced Allergic Proctocolitis by Gross or Occult Blood. J. Allergy Clin. Immunol. Pr. 2020, 8, 1692.e1–1699.e1. [Google Scholar] [CrossRef] [PubMed]
- Arvola, T.; Galanakis, E.; Bitsori, M.; Dimitriou, H.; Giannakopoulou, C.; Karkavitsas, N.S.; Kalmanti, M. Rectal Bleeding in Infancy: Clinical, Allergological, and Microbiological Examination. Pediatrics 2006, 117, 760–768. [Google Scholar] [CrossRef] [PubMed]
- A Xanthakos, S.; Schwimmer, J.B.; Melin-Aldana, H.; Rothenberg, M.E.; Witte, D.P.; Cohen, M.B. Prevalence and Outcome of Allergic Colitis in Healthy Infants with Rectal Bleeding: A Prospective Cohort Study. J. Pediatr. Gastroenterol. Nutr. 2005, 41, 16–22. [Google Scholar] [CrossRef]
- Verkasalo, M.; Kuitunen, P.; Savilahti, E.; Tiilikainen, A. CHANGING PATTERN OF COW’S MILK INTOLERANCE: An Analysis of the Occurrence and Clinical Course in the 60s and mid-70s. Acta Paediatr. 1981, 70, 289–295. [Google Scholar] [CrossRef]
- Savilahti, E. Food-Induced Malabsorption Syndromes. J. Pediatr. Gastroenterol. Nutr. 2000, 30, S61–S66. [Google Scholar] [CrossRef]
- Kokkonen, J.; Haapalahti, M.; Tikkanen, S.; Karttunen, R.; Savilahti, E. Gastrointestinal complaints and diagnosis in children: A population-based study. Acta Paediatr. 2004, 93, 880–886. [Google Scholar] [CrossRef]
- Vitoria, J.C.; Sojo, A.; Rodriguez-Soriano, J. Changing pattern of cow’s milk protein intolerance. Acta Paediatr. Scand 1990, 79, 566–567. [Google Scholar] [CrossRef]
- Katz, Y.; Goldberg, M.R.; Rajuan, N.; Cohen, A.; Leshno, M. The prevalence and natural course of food protein–induced enterocolitis syndrome to cow’s milk: A large-scale, prospective population-based study. J. Allergy Clin. Immunol. 2011, 127, 647–653.e3. [Google Scholar] [CrossRef]
- Alonso, S.B.; Ezquiaga, J.G.; Berzal, P.T.; Tardón, S.D.; José, M.M.S.; López, P.A.; Bermejo, T.B.; Teruel, S.Q.; Zudaire, L.; Ángel, E.; et al. Food protein–induced enterocolitis syndrome: Increased prevalence of this great unknown—results of the PREVALE study. J. Allergy Clin. Immunol. 2019, 143, 430–433. [Google Scholar] [CrossRef]
- Mehr, S.; Frith, K.; Barnes, E.H.; Campbell, D.E.; Allen, K.J.; Gold, M.; Joshi, P.; Kakakios, A.; Loh, R.; Peake, J.; et al. Food protein–induced enterocolitis syndrome in Australia: A population-based study, 2012–2014. J. Allergy Clin. Immunol. 2017, 140, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Nowak-Wegrzyn, A.; Warren, C.M.; Brown-Whitehorn, T.; Cianferoni, A.; Schultz-Matney, F.; Gupta, R.S. Food protein-induced enterocolitis syndrome in the US population-based study. J. Allergy Clin. Immunol. 2019, 144, 1128–1130. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Eigenmann, P.A.; Sampson, H.A. Clinical features of food protein–induced enterocolitis syndrome. J. Pediatr. 1998, 133, 214–219. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Sampson, H.A.; Wood, R.A.; Sicherer, S.H. Food protein-induced enterocolitis syndrome caused by solid food proteins. Pediatrics 2003, 111, 829–835. [Google Scholar] [CrossRef]
- Maciag, M.C.; Bartnikas, L.M.; Sicherer, S.H.; Herbert, L.J.; Young, M.C.; Matney, F.; Westcott-Chavez, A.A.; Petty, C.R.; Phipatanakul, W.; Bingemann, T.A. A Slice of Food Protein–Induced Enterocolitis Syndrome (FPIES): Insights from 441 Children with FPIES as Provided by Caregivers in the International FPIES Association. J. Allergy Clin. Immunol. Pr. 2020, 8, 1702–1709. [Google Scholar] [CrossRef]
- Jean-Christoph, C.; Ford, L.S.; Sickles, L.; Järvinen, K.M.; Sicherer, S.H.; Sampson, H.A.; Nowak-Wegrzyn, A. Clinical features and resolution of food protein–induced enterocolitis syndrome: 10-year experience. J. Allergy Clin. Immunol. 2014, 134, 382.e4–389.e4. [Google Scholar] [CrossRef]
- Lucarelli, S.; Di Nardo, G.; Lastrucci, G.; D’Alfonso, Y.; Marcheggiano, A.; Federici, T.; Frediani, S.; Frediani, T.; Cucchiara, S. Allergic proctocolitis refractory to maternal hypoallergenic diet in exclusively breast-fed infants: A clinical observation. BMC Gastroenterol. 2011, 11, 82. [Google Scholar] [CrossRef]
- Kuitunen, P.; Visakorpi, J.K.; Savilahti, E.; Pelkonen, P. Malabsorption syndrome with cow’s milk intolerance. Clinical findings and course in 54 cases. Arch. Dis. Child 1975, 50, 351–356. [Google Scholar]
- Wakiguchi, H.; Hasegawa, S.; Kaneyasu, H.; Kajimoto, M.; Fujimoto, Y.; Hirano, R.; Katsura, S.; Matsumoto, K.; Ichiyama, T.; Ohga, S. Long-lasting non-IgE-mediated gastrointestinal cow’s milk allergy in infants with Down syndrome. Pediatr. Allergy Immunol. 2015, 26, 821–823. [Google Scholar] [CrossRef]
- Kusters, M.A.A.; Verstegen, R.H.; Gemen, E.F.A.; De Vries, E. Intrinsic defect of the immune system in children with Down syndrome: A review. Clin. Exp. Immunol. 2009, 156, 189–193. [Google Scholar] [CrossRef]
- Nateghi Rostami, M.; Douraghi, M.; Mohammadi, A.M.; Nikmanesh, B. Altered serum pro-inflammatory cytokines in children with Down’s syndrome. Eur. Cytokine Netw. 2012, 23, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.D.; Evans, N.; Pandey, A.; Hraha, T.H.; Smith, K.P.; Markham, N.; Rachubinski, A.L.; Wolter-Warmerdam, K.; Hickey, F.; Espinosa, J.M.; et al. Trisomy 21 causes changes in the circulating proteome indicative of chronic autoinflammation. Sci. Rep. 2017, 7, 14818. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.P.; Magnusson, J.; Ahlstedt, S.; Dahlman-Höglund, A.; Hanson, L.Å.; Magnusson, O.; Bengtsson, U.; Telemo, E. Local allergic reaction in food-hypersensitive adults despite a lack of systemic food-specific IgE. J. Allergy Clin. Immunol. 2002, 109, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Van Sickle, G.J.; Powell, G.K.; McDonald, P.J.; Goldblum, R.M. Milk- and soy protein-induced enterocolitis: Evidence for lymphocyte sensitization to specific food proteins. Gastroenterology 1985, 88, 1915–1921. [Google Scholar] [CrossRef]
- Hoffman, K.M.; Ho, D.G.; Sampson, H.A. Evaluation of the usefulness of lymphocyte proliferation assays in the diagnosis of allergy to cow’s milk. J. Allergy Clin. Immunol. 1997, 99, 360–366. [Google Scholar] [CrossRef]
- Morita, H.; Nomura, I.; Orihara, K.; Yoshida, K.; Akasawa, A.; Tachimoto, H.; Ohtsuka, Y.; Namai, Y.; Futamura, M.; Shoda, T.; et al. Antigen-specific T-cell responses in patients with non–IgE-mediated gastrointestinal food allergy are predominantly skewed to TH2. J. Allergy Clin. Immunol. 2013, 131, 590.e6–592.e6. [Google Scholar] [CrossRef]
- Caubet, J.C.; Bencharitiwong, R.; Ross, A.; Sampson, H.A.; Berin, M.C.; Nowak-Wegrzyn, A. Humoral and cellular responses to casein in patients with food protein-induced enterocolitis to cow’s milk. J. Allergy Clin. Immunol. 2017, 139, 572–583. [Google Scholar] [CrossRef]
- Goswami, R.; Blazquez, A.B.; Kosoy, R.; Rahman, A.; Nowak-Wegrzyn, A.; Berin, M.C. Systemic innate immune activation in food protein–induced enterocolitis syndrome. J. Allergy Clin. Immunol. 2017, 139, 1885.e9–1896.e9. [Google Scholar] [CrossRef]
- Benlounes, N.; Dupont, C.; Candalh, C.; Blaton, M.; Darmon, N.; Desjeux, J.; Heyman, M. The threshold for immune cell reactivity to milk antigens decreases in cow’s milk allergy with intestinal symptoms. J. Allergy Clin. Immunol. 1996, 98, 781–789. [Google Scholar] [CrossRef]
- González-Delgado, P.; Caparrós, E.; Moreno, M.V.; Clemente, F.; Flores, E.; Velásquez, L.; Rubio, G.; Fernandez, J. Clinical and immunological characteristics of a pediatric population with food protein-induced enterocolitis syndrome (FPIES) to fish. Pediatr. Allergy Immunol. 2016, 27, 269–275. [Google Scholar] [CrossRef]
- Shek, L.P.; Bardina, L.; Castro, R.; Sampson, H.A.; Beyer, K. Humoral and cellular responses to cow milk proteins in patients with milk-induced IgE-mediated and non-IgE-mediated disorders. Allergy 2005, 60, 912–919. [Google Scholar] [CrossRef] [PubMed]
- Konstantinou, G.N.; Bencharitiwong, R.; Grishin, A.; Caubet, J.C.; Bardina, L.; Sicherer, S.H.; Sampson, H.A.; Nowak-Wegrzyn, A. The role of casein-specific IgA and TGF-beta in children with food protein-induced enterocolitis syndrome to milk. Pediatr. Allergy Immunol. 2014, 25, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Beyer, K.; Castro, R.; Birnbaum, A.; Benkov, K.; Pittman, N.; Sampson, H.A. Human milk-specific mucosal lymphocytes of the gastrointestinal tract display a TH2 cytokine profile. J. Allergy Clin. Immunol. 2002, 109, 707–713. [Google Scholar] [CrossRef]
- Kapel, N.; Matarazzo, P.; Haouchine, D.; Abiola, N.; Guerin, S.; Magne, D.; Gobert, J.G.; Dupont, C. Fecal tumor necrosis factor alpha, eosinophil cationic protein and IgE levels in infants with cow’s milk allergy and gastrointestinal manifestations. Clin. Chem. Lab. Med. 1999, 37, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.L.; Hwang, J.B.; Park, J.J.; Kim, S.G. Expression of transforming growth factor beta1, transforming growth factor type I and II receptors, and TNF-alpha in the mucosa of the small intestine in infants with food protein-induced enterocolitis syndrome. J. Allergy Clin. Immunol. 2002, 109, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Benlounes, N.; Candalh, C.; Matarazzo, P.; Dupont, C.; Heyman, M. The time-course of milk antigen–induced TNF-α secretion differs according to the clinical symptoms in children with cow’s milk allergy. J. Allergy Clin. Immunol. 1999, 104, 863–869. [Google Scholar] [CrossRef]
- Heyman, M.; Darmon, N.; Dupont, C.; Dugas, B.; Hirribaren, A.; Blaton, M.A.; Desjeux, J.F. Mononuclear cells from infants allergic to cow’s milk secrete tumor necrosis factor alpha, altering intestinal function. Gastroenterology 1994, 106, 1514–1523. [Google Scholar] [CrossRef]
- Jean-Christoph, C.; Nowak-Wegrzyn, A. Current understanding of the immune mechanisms of food protein-induced enterocolitis syndrome. Expert Rev. Clin. Immunol. 2011, 7, 317–327. [Google Scholar] [CrossRef]
- Mori, F.; Barni, S.; Cianferoni, A.; Pucci, N.; De Martino, M.; Novembre, E. Cytokine Expression in CD3+ Cells in an Infant with Food Protein-Induced Enterocolitis Syndrome (FPIES): Case Report. Clin. Dev. Immunol. 2009, 2009, 1–4. [Google Scholar] [CrossRef]
- Kimura, M.; Ito, Y.; Shimomura, M.; Morishita, H.; Meguro, T.; Adachi, Y.; Seto, S. Cytokine profile after oral food challenge in infants with food protein-induced enterocolitis syndrome. Allergol. Int. 2017, 66, 452–457. [Google Scholar] [CrossRef]
- Karlsson, M.R.; Rugtveit, J.; Brandtzaeg, P. Allergen-responsive CD4+CD25+ regulatory T cells in children who have outgrown cow’s milk allergy. J. Exp. Med. 2004, 199, 1679–1688. [Google Scholar] [CrossRef] [PubMed]
- Mehr, S.; Lee, E.; Hsu, P.; Anderson, D.; De Jong, E.; Bosco, A.; Campbell, D. Innate immune activation occurs in acute food protein-induced enterocolitis syndrome reactions. J. Allergy Clin. Immunol. 2019, 144, 600–602.e2. [Google Scholar] [CrossRef]
- McDonald, P.J.; Goldblum, R.M.; Van Sickle, G.J.; Powell, G.K. Food Protein-induced Enterocolitis: Altered Antibody Response to Ingested Antigen. Pediatr. Res. 1984, 18, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Khoshoo, V.; Bhan, M.K.; Kumar, R.; Arora, N.K.; Stintzing, G. Is cow’s milk protein sensitive enteropathy a cell mediated immunological phenomenon? Acta Paediatr. Scand 1991, 80, 1092–1093. [Google Scholar] [CrossRef] [PubMed]
- Nagata, S.; Yamashiro, Y.; Ohtsuka, Y.; Shioya, T.; Oguchi, S.; Shimizu, T.; Maeda, M. Quantitative Analysis and Immunohistochemical Studies on Small Intestinal Mucosa of Food-Sensitive Enteropathy. J. Pediatr. Gastroenterol. Nutr. 1995, 20, 44–48. [Google Scholar] [CrossRef]
- Augustin, M.T.; Kokkonen, J.; Karttunen, T.J. Duodenal cytotoxic lymphocytes in cow’s milk protein sensitive enteropathy and coeliac disease. Scand J. Gastroenterol. 2005, 40, 1398–1406. [Google Scholar] [CrossRef]
- Goldman, H.; Proujansky, R. Allergic Proctitis and Gastroenteritis in Children. Am. J. Surg. Pathol. 1986, 10, 75–86. [Google Scholar] [CrossRef]
- Odze, R.D.; Bines, J.; Leichtner, A.M.; Goldman, H.; Antonioli, D.A. Allergic proctocolitis in infants: A prospective clinicopathologic biopsy study. Hum. Pathol. 1993, 24, 668–674. [Google Scholar] [CrossRef]
- Untersmayr, E.; Schöll, I.; Swoboda, I.; Beil, W.J.; Förster-Waldl, E.; Walter, F.; Riemer, A.; Kraml, G.; Kinaciyan, T.; Spitzauer, S.; et al. Antacid medication inhibits digestion of dietary proteins and causes food allergy: A fish allergy model in BALB/c mice. J. Allergy Clin. Immunol. 2003, 112, 616–623. [Google Scholar] [CrossRef]
- Díaz, J.J.; Espín, B.; Segarra, O.; Domínguez-Ortega, G.; Blasco-Alonso, J.; Cano, B.; Rayo, A.; Moreno, A. Food Protein-induced Enterocolitis Syndrome. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 232–236. [Google Scholar] [CrossRef]
- Caubet, J.; Cianferoni, A.; Groetch, M.; Nowak-Wegrzyn, A. Food protein-induced enterocolitis syndrome. Clin. Exp. Allergy 2019, 49, 1178–1190. [Google Scholar] [CrossRef] [PubMed]
- Delahaye, C.; Chauveau, A.; Kiefer, S.; Dumond, P. Food protein-induced enterocolitis syndrome (FPIES) in 14 children. Arch. Pediatr. 2017, 24, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Barnes, E.H.; Mehr, S.; Campbell, D.E. Differentiating Acute Food Protein–Induced Enterocolitis Syndrome From Its Mimics: A Comparison of Clinical Features and Routine Laboratory Biomarkers. J. Allergy Clin. Immunol. Pr. 2019, 7, 471.e3–478.e3. [Google Scholar] [CrossRef]
- Su, K.-W.; Patil, S.; Stockbridge, J.; Martin, V.; Virkud, Y.; Huang, J.L.; Shreffler, W.; Yuan, Q. Food aversion and poor weight gain in food protein-induced enterocolitis syndrome: A retrospective study. J. Allergy Clin. Immunol. 2020, 145, AB52. [Google Scholar] [CrossRef]
- Blackman, A.C.; Anvari, S.; Davis, C.M.; Anagnostou, A. Emerging triggers of food protein–induced enterocolitis syndrome. Ann. Allergy, Asthma Immunol. 2019, 122, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Ludman, S.; Harmon, M.; Whiting, D.; Du Toit, G. Clinical presentation and referral characteristics of food protein-induced enterocolitis syndrome in the United Kingdom. Ann. Allergy, Asthma Immunol. 2014, 113, 290–294. [Google Scholar] [CrossRef]
- Sopo, S.M.; Giorgio, V.; Iacono, I.D.; Novembre, E.; Mori, F.; Onesimo, R. A multicentre retrospective study of 66 Italian children with food protein-induced enterocolitis syndrome: Different management for different phenotypes. Clin. Exp. Allergy 2012, 42, 1257–1265. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Chehade, M.; Groetch, M.E.; Spergel, J.M.; Wood, R.A.; Allen, K.; Atkins, D.; Bahna, S.; Barad, A.V.; Berin, C.; et al. International consensus guidelines for the diagnosis and management of food protein-induced enterocolitis syndrome: Executive summary-Workgroup Report of the Adverse Reactions to Foods Committee, American Academy of Allergy, Asthma & Immunology. J. Allergy Clin. Immunol. 2017, 139, 1111 e4–1126 e4. [Google Scholar]
- Sicherer, S.H. Food protein-induced enterocolitis syndrome: Case presentations and management lessons. J. Allergy Clin. Immunol. 2005, 115, 149–156. [Google Scholar] [CrossRef]
- Monti, G.; Castagno, E.; Liguori, S.A.; Lupica, M.M.; Tarasco, V.; Viola, S.; Tovo, P.-A. Food protein–induced enterocolitis syndrome by cow’s milk proteins passed through breast milk. J. Allergy Clin. Immunol. 2011, 127, 679–680. [Google Scholar] [CrossRef]
- Kaya, A.; Toyran, M.; Civelek, E.; Mısırlıoglu, E.D.; Kırsaçlıoglu, C.T.; Kocabaş, C.N. Food Protein-Induced Enterocolitis Syndrome In Two Exclusively Breastfed Infants. Pediatr. Allergy Immunol. 2016, 27, 749–750. [Google Scholar] [CrossRef]
- Nomura, I.; Morita, H.; Hosokawa, S.; Hoshina, H.; Fukuie, T.; Watanabe, M.; Ohtsuka, Y.; Shoda, T.; Terada, A.; Takamasu, T.; et al. Four distinct subtypes of non–IgE-mediated gastrointestinal food allergies in neonates and infants, distinguished by their initial symptoms. J. Allergy Clin. Immunol. 2011, 127, 685.e8–688.e8. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.J.; Nowak-Wegrzyn, A.; Vadas, P. FPIES in adults. Ann. Allergy, Asthma Immunol. 2018, 121, 736–738. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, B.N.; Boyle, R.J.; Gore, C.; Simpson, A.; Custovic, A. Food protein–induced enterocolitis syndrome can occur in adults. J. Allergy Clin. Immunol. 2012, 130, 1199–1200. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.A.; Smith, W.B. Non–IgE-mediated gastrointestinal food hypersensitivity syndrome in adults. J. Allergy Clin. Immunol. Pr. 2014, 2, 355.e1–357.e1. [Google Scholar] [CrossRef]
- Gonzalez-Delgado, P.; Caparrós, E.; Moreno, M.V.; Cueva, B.; Fernández, J. Food protein–induced enterocolitis-like syndrome in a population of adolescents and adults caused by seafood. J. Allergy Clin. Immunol. Pr. 2019, 7, 670–672. [Google Scholar] [CrossRef]
- Ichimura, S.; Kakita, H.; Asai, S.; Mori, M.; Takeshita, S.; Ueda, H.; Muto, T.; Kondo, T.; Yamada, Y. A Rare Case of Fetal Onset, Food Protein-Induced Enterocolitis Syndrome. Neonatology 2019, 116, 376–379. [Google Scholar] [CrossRef]
- Faber, M.R.; Rieu, P.; A Semmekrot, B.; Krieken, J.H.J.M.; Tolboom, J.J.M.; Draaisma, J.M.T. Allergic colitis presenting within the first hours of premature life. Acta Paediatr. 2007, 94, 1514–1515. [Google Scholar] [CrossRef]
- Walker-Smith, J. Cow milk-sensitive enteropathy: Predisposing factors and treatment. J. Pediatr. 1992, 121, S111–S115. [Google Scholar] [CrossRef]
- Walker-Smith, J.; Harrison, M.; Kilby, A.; Phillips, A.; France, N. Cows’ milk-sensitive enteropathy. Arch. Dis. Child. 1978, 53, 375–380. [Google Scholar] [CrossRef]
- Iyngkaran, N.; Robinson, M.J.; Prathap, K.; Sumithran, E.; Yadav, M. Cows’ milk protein-sensitive enteropathy. Combined clinical and histological criteria for diagnosis. Arch. Dis. Child. 1978, 53, 20–26. [Google Scholar] [CrossRef][Green Version]
- Iyngkaran, N.; Abdin, Z.; Davis, K.; Boey, C.; Prathap, K.; Yadav, M.; Lam, S.; Puthucheary, S. Acquired carbohydrate intolerance and cow milk protein-sensitive enteropathy in young infants. J. Pediatr. 1979, 95, 373–378. [Google Scholar] [CrossRef]
- Lozinsky, A.C.; Morais, M.B. Eosinophilic colitis in infants. J. Pediatr. (Rio J) 2014, 90, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Lake, A.M. Food-Induced Eosinophilic Proctocolitis. J. Pediatr. Gastroenterol. Nutr. 2000, 30, S58–S60. [Google Scholar] [CrossRef] [PubMed]
- Cetinkaya, P.G.; Kahveci, M.; Karaatmaca, B.; Esenboga, S.; Sahiner, U.M.; Sekerel, B.E.; Soyer, O. Predictors for late tolerance development in food protein-induced allergic proctocolitis. Allergy Asthma Proc. 2020, 41, e11–e18. [Google Scholar] [CrossRef]
- Erdem, S.; Nacaroglu, H.; Karaman, S.; Erdur, C.; Karkıner, C.; Can, D. Tolerance development in food protein-induced allergic proctocolitis: Single centre experience. Allergol. et Immunopathol. 2017, 45, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Kaya, A.; Toyran, M.; Civelek, E.; Misirlioğlu, E.; Kirsaclioglu, C.; Kocabaş, C.N.; Kırsaçlıoğlu, C. Characteristics and Prognosis of Allergic Proctocolitis in Infants. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 1. [Google Scholar] [CrossRef] [PubMed]
- Sierra Salinas, C.; Blasco Alonso, J.; Olivares Sanchez, L.; Barco Galvez, A.; del Rio Mapelli, L. Dietary protein-induced proctocolitis in childhood. Am. J. Gastroenterol. 2008, 103, 2605–2612. [Google Scholar]
- Ravelli, A.; Villanacci, V.; Chiappa, S.; Bolognini, S.; Manenti, S.; Fuoti, M. Dietary protein-induced proctocolitis in childhood. Am. J. Gastroenterol. 2008, 103, 2605–2612. [Google Scholar] [CrossRef] [PubMed]
- Høst, A.; Halken, S.; Jacobsen, H.P.; Christensen, A.E.; Herskind, A.M.; Plesner, K.B. Clinical course of cow’s milk protein allergy/intolerance and atopic diseases in childhood. Pediatr. Allergy Immunol. 2002, 13, 23–28. [Google Scholar] [CrossRef]
- Vanto, T.; Helppila, S.; Juntunen-Backman, K.; Kalimo, K.; Klemola, T.; Korpela, R.; Koskinen, P. Prediction of the development of tolerance to milk in children with cow’s milk hypersensitivity. J. Pediatr. 2004, 144, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.F.; Dias, A.; Pinheiro, J.A. Predictive factors for the persistence of cow’s milk allergy. Pediatr. Allergy Immunol. 2010, 21, 1127–1134. [Google Scholar] [CrossRef]
- Wang, K.Y.; Lee, J.; Cianferoni, A.; Ruffner, M.A.; Dean, A.; Molleston, J.M.; Pawlowski, N.A.; Heimall, J.; Saltzman, R.W.; Ram, G.S.; et al. Food Protein–Induced Enterocolitis Syndrome Food Challenges: Experience from a Large Referral Center. J. Allergy Clin. Immunol. Pr. 2019, 7, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.-B.; Sohn, S.M.; Kim, A.S. Prospective follow-up oral food challenge in food protein-induced enterocolitis syndrome. Arch. Dis. Child. 2008, 94, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Ruffner, M.A.; Ruymann, K.; Barni, S.; Cianferoni, A.; Brown-Whitehorn, T.; Spergel, J.M. Food Protein-induced Enterocolitis Syndrome: Insights from Review of a Large Referral Population. J. Allergy Clin. Immunol. Pr. 2013, 1, 343–349. [Google Scholar] [CrossRef]
- Burks, A.W.; Casteel, H.B.; Fiedorek, S.C.; Williams, L.W.; Pumphrey, C.L. Prospective oral food challenge study of two soybean protein isolates in patients with possible milk or soy protein enterocolitis. Pediatr. Allergy Immunol. 1994, 5, 40–45. [Google Scholar] [CrossRef]
- Mehr, S.; Kakakios, A.; Frith, K.; Kemp, A.S. Food Protein-Induced Enterocolitis Syndrome: 16-Year Experience. Pediatrics 2009, 123, 459–464. [Google Scholar] [CrossRef]
- Mehr, S.; Kakakios, A.M.; Kemp, A.S. Rice: A common and severe cause of food protein-induced enterocolitis syndrome. Arch. Dis. Child. 2009, 94, 220–223. [Google Scholar] [CrossRef]
- Hojsak, I.; Kljaić-Turkalj, M.; Mišak, Z.; Kolacek, S. Rice protein-induced enterocolitis syndrome. Clin. Nutr. 2006, 25, 533–536. [Google Scholar] [CrossRef]
- Sopo, S.M.; Monaco, S.; Badina, L.; Barni, S.; Longo, G.; Novembre, E.; Viola, S.; Monti, G. Food protein-induced enterocolitis syndrome caused by fish and/or shellfish in Italy. Pediatr. Allergy Immunol. 2015, 26, 731–736. [Google Scholar] [CrossRef]
- Vazquez-Ortiz, M.; Machinena, A.; Dominguez, O.; Alvaro, M.; Calvo-Campoverde, K.; Giner, M.T.; Jiménez-Feijoo, R.; Lozano, J.; Piquer, M.; Dias, M.; et al. Food protein–induced enterocolitis syndrome to fish and egg usually resolves by age 5 years in Spanish children. J. Allergy Clin. Immunol. Pr. 2017, 5, 512.e1–515.e1. [Google Scholar] [CrossRef] [PubMed]
- Mehr, S.; Frith, K.; Campbell, D.E. Epidemiology of food protein-induced enterocolitis syndrome. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Manuel, P.D.; Walker-Smith, J.A.; Soeparto, P. Cow’s milk sensitive enteropathy in Indonesian infants. Lancet 1980, 2, 1365–1366. [Google Scholar] [CrossRef]
- Ajami, R.I.P.; Sari, S.K.C.; Mazas, Y.A.; Calvo, J.B.; Abadía, M.I.G. Experience in food protein-induced enterocolitis syndrome in a paediatric allergy clinic. Anales de Pediatría 2020. [Google Scholar] [CrossRef]
- Yilmaz, E.A.; Soyer, O.; Cavkaytar, O.; Karaatmaca, B.; Buyuktiryaki, B.; Sahiner, U.M.; Sekerel, B.E.; Sackesen, C. Characteristics of children with food protein-induced enterocolitis and allergic proctocolitis. Allergy Asthma Proc. 2017, 38, 54–62. [Google Scholar] [CrossRef]
- Meyer, R.W.; Lozinsky, A.C.; Fleischer, D.M.; Vieira, M.C.; Du Toit, G.; Vandenplas, Y.; Dupont, C.; Knibb, R.; Uysal, P.; Cavkaytar, O.; et al. Diagnosis and management of Non-IgE gastrointestinal allergies in breastfed infants—An EAACI Position Paper. Allergy 2019, 75, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Nowak-Wegrzyn, A.; Assa’ad, A.H.; Bahna, S.L.; Bock, S.H.; Sicherer, S.A.; Teuber, S.S.; on behalf of the Adverse Reactions to Food Committee of American Academy of Allergy, and Immunology. Work Group report: Oral food challenge testing. J. Allergy Clin. Immunol. 2009, 123, S365–S383. [Google Scholar] [CrossRef] [PubMed]
- Culy, C.R.; Bhana, N.; Plosker, G.L. Ondansetron: A review of its use as an antiemetic in children. Pediatr. Drugs 2001, 3, 441–479. [Google Scholar] [CrossRef]
- Holbrook, T.; Keet, C.A.; Frischmeyer-Guerrerio, P.A.; Wood, R.A. Use of ondansetron for food protein–induced enterocolitis syndrome. J. Allergy Clin. Immunol. 2013, 132, 1219–1220. [Google Scholar] [CrossRef]
- Sopo, S.M.; Battista, A.; Greco, M.; Monaco, S. Ondansetron for Food Protein-Induced Enterocolitis Syndrome. Int. Arch. Allergy Immunol. 2014, 164, 137–139. [Google Scholar] [CrossRef]
- Feuille, E.; Nowak-Wegrzyn, A. Food Protein-Induced Enterocolitis Syndrome, Allergic Proctocolitis, and Enteropathy. Curr. Allergy Asthma Rep. 2015, 15, 50. [Google Scholar] [CrossRef] [PubMed]
- Sopo, S.M.; Monaco, S.; Bersani, G.; Romano, A.; Fantacci, C. Proposal for management of the infant with suspected food protein-induced allergic proctocolitis. Pediatr. Allergy Immunol. 2018, 29, 215–218. [Google Scholar] [CrossRef]
- Murray, K.F.; Christie, D.L. Dietary protein intolerance in infants with transient methemoglobinemia and diarrhea. J. Pediatr. 1993, 122, 90–92. [Google Scholar] [CrossRef]
- Baudon, J.J.; Fontaine, J.L.; Mougenot, J.F.; Navarro, J.; Polonovski, C.; Laplane, R. Digestive intolerance to cow’s milk proteins in infants. Biological and histological study. Arch. Fr. Pediatr. 1975, 32, 787–801. [Google Scholar] [PubMed]
- Lake, A.M.; Whitington, P.F.; Hamilton, S.R. Dietary protein-induced colitis in breast-fed infants. J. Pediatr. 1982, 101, 906–910. [Google Scholar] [CrossRef]
- Molnár, K.; Pintér, P.; Győrffy, H.; Cseh, A.; Muller, K.E.; Arató, A.; Veres, G. Characteristics of allergic colitis in breast-fed infants in the absence of cow’s milk allergy. World J. Gastroenterol. 2013, 19, 3824–3830. [Google Scholar] [CrossRef]
- Kimura, M.; Shimomura, M.; Morishita, H.; Meguro, T.; Seto, S. Eosinophilia in infants with food protein-induced enterocolitis syndrome in Japan. Allergol. Int. 2017, 66, 310–316. [Google Scholar] [CrossRef]
- Giavi, S.; Megremis, S.; Papadopoulos, N.G. Lymphocyte stimulation test for the diagnosis of non-IgE-mediated cow’s milk allergy: A step closer to a noninvasive diagnostic tool? Int. Arch. Allergy Immunol. 2011, 157, 1–2. [Google Scholar] [CrossRef]
- I Fogg, M.; Pawlowski, N.A.; Spergel, J.M.; Brown-Whitehorn, T.A. Atopy patch test for the diagnosis of food protein-induced enterocolitis syndrome. Pediatr. Allergy Immunol. 2006, 17, 351–355. [Google Scholar] [CrossRef]
- Järvinen, K.M.; Caubet, J.-C.; Sickles, L.; Ford, L.S.; Sampson, H.A.; Nowak-Węgrzyn, A. Poor utility of atopy patch test in predicting tolerance development in food protein-induced enterocolitis syndrome. Ann. Allergy, Asthma Immunol. 2012, 109, 221–222. [Google Scholar] [CrossRef]
- Concha, S.; Cabalin, C.; Iturriaga, C.; Perez-Mateluna, G.; Gomez, C.; Cifuentes, L.; Harris, P.R.; Gana, J.C.; Borzutzky, A. Diagnostic validity of fecal occult blood test in infants with food protein-induced allergic proctocolitis. Rev. Chil. Pediatr. 2018, 89, 630–637. [Google Scholar] [PubMed]
- Khan, S. Testing for Fecal Calprotectin in Food Protein–Induced Enterocolitis Syndrome. J. Investig. Allergol. Clin. Immunol. 2018, 28, 287–288. [Google Scholar] [CrossRef] [PubMed]
- Beser, O.F.; Sancak, S.; Erkan, T.; Kutlu, T.; Cokugras, H.; Cokugras, F.C. Can Fecal Calprotectin Level Be Used as a Markers of Inflammation in the Diagnosis and Follow-Up of Cow’s Milk Protein Allergy? Allergy Asthma Immunol. Res. 2014, 6, 33–38. [Google Scholar]
- Baldassarre, M.E.; Laforgia, N.; Fanelli, M.; Laneve, A.; Grosso, R.; Lifschitz, C. Lactobacillus GG Improves Recovery in Infants with Blood in the Stools and Presumptive Allergic Colitis Compared with Extensively Hydrolyzed Formula Alone. J. Pediatr. 2010, 156, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Trillo Belizon, C.; Ortega Paez, E.; Medina Claros, A.F.; Rodriguez Sanchez, I.; Reina Gonzalez, A.; Vera Medialdea, R.; Ramon Salguero, J.M. Faecal calprotectin as an aid to the diagnosis of non-IgE mediated cow’s milk protein allergy. An. Pediatr. (Barc) 2016, 84, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Ma, J.; Geng, S.; Wang, J.; Liu, J.; Zhang, J.; Sheng, X. Fecal Calprotectin Concentrations in Healthy Children Aged 1-18 Months. PLoS ONE 2015, 10, e0119574. [Google Scholar] [CrossRef]
- Zhu, Q.; Li, F.; Wang, J.; Shen, L.; Sheng, X. Fecal Calprotectin in Healthy Children Aged 1-4 Years. PLoS ONE 2016, 11, e0150725. [Google Scholar] [CrossRef]
- Richards, D.G.; Somers, S.; Issenman, R.M.; Stevenson, G.W. Cow’s milk protein/soy protein allergy: Gastrointestinal imaging. Radiology 1988, 167, 721–723. [Google Scholar] [CrossRef]
- Powell, G.K. Enterocolitis in low-birth-weight infants associated with milk and soy protein intolerance. J. Pediatr. 1976, 88, 840–844. [Google Scholar] [CrossRef]
- Jayasooriya, S.; Fox, A.T.; Murch, S.H. Do Not Laparotomize Food Protein-Induced Enterocolitis Syndrome. Pediatr. Emerg. Care 2007, 23, 173–175. [Google Scholar] [CrossRef]
- Jimbo, K.; Ohtsuka, Y.; Kono, T.; Arai, N.; Kyoudo, R.; Hosoi, K.; Aoyagi, Y.; Kudo, T.; Asai, N.; Shimizu, T. Ultrasonographic study of intestinal Doppler blood flow in infantile non-IgE-mediated gastrointestinal food allergy. Allergol. Int. 2019, 68, 199–206. [Google Scholar] [CrossRef]
- Variend, S.; Placzek, M.; Raafat, F.; A Walker-Smith, J. Small intestinal mucosal fat in childhood enteropathies. J. Clin. Pathol. 1984, 37, 373–377. [Google Scholar] [CrossRef]
- McCalla, R.; Savilahtl, E.; Perkkiö, M.; Kuitunen, P.; Backman, A. Morphology of the Jejunum in Children with Eczema due to Food Allergy. Allergy 1980, 35, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Sorea, S.; Dabadie, A.; Bridoux-Henno, L.; Balancon-Morival, M.; Jouan, H.; Le Gall, E. Hemorrhagic colitis in exclusively breast-fed infants. Arch. Pediatr. 2003, 10, 772–775. [Google Scholar] [CrossRef]
- Kimura, M.; Ito, Y.; Tokunaga, F.; Meguro, T.; Shimomura, M.; Morishita, H.; Seto, S. Increased C-reactive protein and fever in Japanese infants with food protein-induced enterocolitis syndrome. Pediatr. Int. 2016, 58, 826–830. [Google Scholar] [CrossRef]
- Kimura, M.; Shimomura, M.; Morishita, H.; Meguro, T.; Seto, S. Serum C-reactive protein in food protein-induced enterocolitis syndrome versus food protein-induced proctocolitis in Japan. Pediatr. Int. 2016, 58, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Pecora, V.; Prencipe, G.; Valluzzi, R.L.; Dahdah, L.; Insalaco, A.; Cianferoni, A.; De Benedetti, F.; Fiocchi, A. Inflammatory events during food protein-induced enterocolitis syndrome reactions. Pediatr. Allergy Immunol. 2017, 28, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Yagi, H.; Takizawa, T.; Sato, K.; Inoue, T.; Nishida, Y.; Ishige, T.; Tatsuki, M.; Hatori, R.; Kobayashi, Y.; Yamada, Y.; et al. Severity scales of non-IgE-mediated gastrointestinal food allergies in neonates and infants. Allergol. Int. 2018, 68, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Geng, L.; Xu, Z.; Chen, P.; Friesen, C.A.; Gong, S.; Li, D.-Y. Severe Food Protein-Induced Enterocolitis Syndrome to Cow’s Milk in Infants. Nutrients 2016, 8, 1. [Google Scholar] [CrossRef]
- Maines, E.; Di Palma, A.; Burlina, A. Food triggers and inherited metabolic disorders: A challenge to the pediatrician. Ital. J. Pediatr. 2018, 44, 18. [Google Scholar] [CrossRef]
- Lee, J.H.; Choe, Y.H.; Lee, S.K.; Seo, J.M.; Kim, J.H.; Suh, Y.L. Allergic proctitis and abdominal distention mimicking Hirschsprung’s disease in infants. Acta Paediatr. 2007, 96, 1784–1789. [Google Scholar] [CrossRef]
- Onesimo, R.; Iacono, I.D.; Giorgio, V.; Limongelli, M.G.; Sopo, S.M. Can food protein induced enterocolitis syndrome shift to immediate gastrointestinal hypersensitivity? A report of two cases. Eur. Ann. Allergy Clin. Immunol. 2011, 43, 61–63. [Google Scholar] [PubMed]
- Kessel, A.; Dalal, I. The pendulum between food protein-induced enterocolitis syndrome and IgE-mediated milk allergy. Acta Paediatr. 2011, 100, e183–e185. [Google Scholar] [CrossRef] [PubMed]
- Banzato, C.; Piacentini, G.L.; Comberiati, P.; Mazzei, F.; Boner, A.L.; Peroni, D.G. Unusual shift from IgE-mediated milk allergy to food protein-induced enterocolitis syndrome. Eur. Ann. Allergy Clin. Immunol. 2013, 45, 209–211. [Google Scholar] [PubMed]
- Barni, S.; Mori, F.; Bianchi, A.; Pucci, N.; Novembre, E. Shift from IgE-mediated cow’s milk allergy to food protein-induced enterocolitis syndrome in 2 infants. Pediatr. Allergy Immunol. 2018, 29, 446–447. [Google Scholar] [CrossRef]
- Duffey, H.; Egan, M. Development of food protein–induced enterocolitis syndrome (FPIES) to egg after immunoglobulin E–mediated egg allergy. Ann. Allergy, Asthma Immunol. 2018, 121, 379–380. [Google Scholar] [CrossRef]
- Katz, Y.; Rajuan, N.; Goldberg, M.R.; Eisenberg, E.; Heyman, E.; Cohen, A.; Leshno, M. Early exposure to cow’s milk protein is protective against IgE-mediated cow’s milk protein allergy. J. Allergy Clin. Immunol. 2010, 126, 77 e1–82 e1. [Google Scholar] [CrossRef]
- Flinterman, A.E.; Knulst, A.C.; Meijer, Y.; Pasmans, S.G.M.A.; Bruijnzeel-Koomen, C.A.F.M. Acute allergic reactions in children with AEDS after prolonged cow’s milk elimination diets. Allergy 2006, 61, 370–374. [Google Scholar] [CrossRef]
- Du Toit, G.; Roberts, G.; Sayre, P.H.; Bahnson, H.T.; Radulovic, S.; Santos, A.F.; Brough, H.A.; Phippard, D.; Basting, M.; Feeney, M.; et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. New Engl. J. Med. 2015, 372, 803–813. [Google Scholar] [CrossRef]
- Walker-Smith, J. Food sensitive enteropathy: Overview and update. Pediatr. Int. 1994, 36, 545–549. [Google Scholar] [CrossRef]
- Kokkonen, J.; Haapalahti, M.; Laurila, K.; Karttunen, T.J.; Maki, M. Cow’s milk protein-sensitive enteropathy at school age. J. Pediatr. 2001, 139, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Kokkonen, J.; Tikkanen, S.; Savilahti, E. Residual Intestinal Disease After Milk Allergy in Infancy. J. Pediatr. Gastroenterol. Nutr. 2001, 32, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Walker, W.A. Cow’s milk protein-sensitive enteropathy at school age: A new entity or a spectrum of mucosal immune responses with age. J. Pediatr. 2001, 139, 765–766. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, G.; Cremon, C.; Frediani, S.; Lucarelli, S.; Villa, M.P.; Stanghellini, V.; La Torre, G.; Martemucci, L.; Barbara, G. Allergic Proctocolitis Is a Risk Factor for Functional Gastrointestinal Disorders in Children. J. Pediatr. 2018, 195, 128–133.e1. [Google Scholar] [CrossRef]
- Meyer, R.; Schwarz, C.; Shah, N. A review on the diagnosis and management of food-induced gastrointestinal allergies. Curr. Allergy Clin. Immunol. 2012, 25, 10–17. [Google Scholar]
- De Boissieu, D.; Matarazzo, P.; Dupont, C. Allergy to extensively hydrolyzed cow milk proteins in infants: Identification and treatment with an amino acid-based formula. J. Pediatr. 1997, 131, 744–747. [Google Scholar] [CrossRef]
- A Vanderhoof, J.; Murray, N.D.; Kaufman, S.S.; Mack, D.R.; Antonson, D.L.; Corkins, M.R.; Perry, D.; Kruger, R. Intolerance to protein hydrolysate infant formulas: An underrecognized cause of gastrointestinal symptoms in infants. J. Pediatr. 1997, 131, 741–744. [Google Scholar] [CrossRef]
- American Academy of Pediatrics. Committee on Nutrition. Hypoallergenic infant formulas. Pediatrics 2000, 106, 346–349. [Google Scholar] [CrossRef]
- Vandenplas, Y. Prevention and Management of Cow’s Milk Allergy in Non-Exclusively Breastfed Infants. Nutrients 2017, 9, 731. [Google Scholar] [CrossRef]
- Vandenplas, Y.; Koletzko, S.; Isolauri, E.; Hill, D.; Oranje, A.P.; Brueton, M.; Staiano, A.; Dupont, C. Guidelines for the diagnosis and management of cow’s milk protein allergy in infants. Arch. Dis. Child. 2007, 92, 902–908. [Google Scholar] [CrossRef]
- Sopo, S.M.; Buonsenso, D.; Monaco, S.; Crocco, S.; Longo, G.; Calvani, M. Food protein-induced enterocolitis syndrome (FPIES) and well cooked foods: A working hypothesis. Allergol. et Immunopathol. 2013, 41, 346–348. [Google Scholar] [CrossRef] [PubMed]
- Sopo, S.M.; Romano, A.; Bersani, G.; Fantacci, C.; Badina, L.; Longo, G.; Monti, G.; Viola, S.; Tripodi, S.; Barilaro, G.; et al. Cooking influence in tolerance acquisition in egg-induced acute food protein enterocolitis syndrome. Allergol. et Immunopathol. 2019, 47, 221–226. [Google Scholar] [CrossRef]
- Uncuoglu, A.; Yologlu, N.; Simsek, I.; Uyan, Z.; Aydogan, M. Tolerance to baked and fermented cow’s milk in children with IgE-mediated and non-IgE-mediated cow’s milk allergy in patients under two years of age. Allergol. et Immunopathol. 2017, 45, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Upton, J.E.M.; Nowak-Węgrzyn, A. The Impact of Baked Egg and Baked Milk Diets on IgE- and Non-IgE-Mediated Allergy. Clin. Rev. Allergy Immunol. 2018, 55, 118–138. [Google Scholar] [CrossRef] [PubMed]
- Camargo, L.S.; Da Silveira, J.A.C.; Taddei, J.A.; Neto, U.F. ALLERGIC PROCTOCOLITIS IN INFANTS: Analysis of the evolution of the nutritional status. Arq. de Gastroenterol. 2016, 53, 262–266. [Google Scholar] [CrossRef]
- Venter, C.; Groetch, M. Nutritional management of food protein-induced enterocolitis syndrome. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 255–262. [Google Scholar] [CrossRef][Green Version]
- Meyer, R.W.; De Koker, C.; Dziubak, R.; Venter, C.; Dominguez-Ortega, G.; Cutts, R.; Yerlett, N.; Skrapak, A.-K.; Fox, A.T.; Shah, N. Malnutrition in children with food allergies in the UK. J. Hum. Nutr. Diet. 2013, 27, 227–235. [Google Scholar] [CrossRef]
- Meyer, R.W.; Rommel, N.; Van Oudenhove, L.; Fleming, C.; Dziubak, R.; Shah, N. Feeding difficulties in children with food protein-induced gastrointestinal allergies. J. Gastroenterol. Hepatol. 2014, 29, 1764–1769. [Google Scholar] [CrossRef]
- Meyer, R.W.; Wright, K.; Vieira, M.C.; Chong, K.W.; Chatchatee, P.; Vlieg-Boerstra, B.J.; Groetch, M.; Dominguez-Ortega, G.; Heath, S.; Lang, A.; et al. International survey on growth indices and impacting factors in children with food allergies. J. Hum. Nutr. Diet. 2018, 32, 175–184. [Google Scholar] [CrossRef]
- Meyer, R.W.; De Koker, C.; Dziubak, R.; Skrapac, A.-K.; Godwin, H.; Reeve, K.; Lozinsky, A.C.; Shah, N. A practical approach to vitamin and mineral supplementation in food allergic children. Clin. Transl. Allergy 2015, 5, 11. [Google Scholar] [CrossRef]
- Meyer, R.W. Nutritional disorders resulting from food allergy in children. Pediatr. Allergy Immunol. 2018, 29, 689–704. [Google Scholar] [CrossRef] [PubMed]
- Foong, R.-X.M.; Meyer, R.W.; Dziubak, R.; Lozinsky, A.C.; Godwin, H.; Reeve, K.; Hussain, S.T.; Nourzaie, R.; Shah, N. Establishing the prevalence of low vitamin D in non-immunoglobulin-E mediated gastrointestinal food allergic children in a tertiary centre. World Allergy Organ. J. 2017, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Mailhot, G.; Perrone, V.; Alos, N.; Dubois, J.; Delvin, E.; Paradis, L.; Roches, A.D. Cow’s Milk Allergy and Bone Mineral Density in Prepubertal Children. Pediatrics 2016, 137. [Google Scholar] [CrossRef]
- Lazare, F.B.; Brand, D.A.; Marciano, T.A.; Daum, F. Rapid Resolution of Milk Protein Intolerance in Infancy. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Luyt, D.; Ball, H.; Makwana, N.; Green, M.R.; Bravin, K.; Nasser, S.M.; Clark, A. BSACI guideline for the diagnosis and management of cow’s milk allergy. Clin. Exp. Allergy 2014, 44, 642–672. [Google Scholar] [CrossRef]
- Baker, R.D.; Greer, F.R. Diagnosis and Prevention of Iron Deficiency and Iron-Deficiency Anemia in Infants and Young Children (0-3 Years of Age). Pediatrics 2010, 126, 1040–1050. [Google Scholar] [CrossRef]
- Foong, R.X.; Meyer, R.; Godwin, H.; Dziubak, R.; Lozinsky, A.C.; Reeve, K.; Knibb, R.; Shah, N. Parental perception of their child’s quality of life in children with non-immunoglobulin-E-mediated gastrointestinal allergies. Pediatr. Allergy Immunol. 2017, 28, 251–256. [Google Scholar] [CrossRef]
- Meyer, R.W.; Godwin, H.; Dziubak, R.; Panepinto, J.A.; Foong, R.-X.M.; Bryon, M.; Lozinsky, A.C.; Reeve, K.; Shah, N. The impact on quality of life on families of children on an elimination diet for Non-immunoglobulin E mediated gastrointestinal food allergies. World Allergy Organ. J. 2017, 10, 8. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Katz, Y.; Mehr, S.; Koletzko, S. Non–IgE-mediated gastrointestinal food allergy. J. Allergy Clin. Immunol. 2015, 135, 1114–1124. [Google Scholar] [CrossRef]
- Iacono, G.; Carroccio, A.; Cavataio, F.; Montalto, G.; Kazmierska, I.; Lorello, D.; Soresi, M.; Notarbartolo, A. Gastroesophageal reflux and cow’s milk allergy in infants: A prospective study. J. Allergy Clin. Immunol. 1996, 97, 822–827. [Google Scholar]
- Nielsen, R.G.; Bindslev-Jensen, C.; Kruse-Andersen, S.; Husby, S. Severe Gastroesophageal Reflux Disease and Cow Milk Hypersensitivity in Infants and Children: Disease Association and Evaluation of a New Challenge Procedure. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Ravelli, A.M.; Tobanelli, P.; Volpi, S.; Ugazio, A.G. Vomiting and gastric motility in infants with cow’s milk allergy. J. Pediatr. Gastroenterol. Nutr. 2001, 32, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, O.; Mancini, V.; Thapar, N.; Giorgio, V.; Elawad, M.; Hill, S.; Shah, N.; Lindley, K.J. Cow’s Milk Challenge Increases Weakly Acidic Reflux in Children with Cow’s Milk Allergy and Gastroesophageal Reflux Disease. J. Pediatr. 2012, 161, 476–481.e1. [Google Scholar] [CrossRef]
- Vandenplas, Y. Management of paediatric GERD. Nat. Rev. Gastroenterol. Hepatol. 2013, 11, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Loo, E.X.L.; Wang, D.Y.; Siah, K.T.H. Association between Irritable Bowel Syndrome and Allergic Diseases: To Make a Case for Aeroallergen. Int. Arch. Allergy Immunol. 2019, 181, 31–42. [Google Scholar] [CrossRef]
- Choung, R.S.; A Murray, J. The Role for Food Allergies in the Pathogenesis of Irritable Bowel Syndrome: Understanding Mechanisms of Intestinal Mucosal Responses Against Food Antigens. Gastroenterology 2019, 157, 15–17. [Google Scholar] [CrossRef]
- Stefanini, G.F.; Saggioro, A.; Alvisi, V.; Angelini, G.; Capurso, L.; Di Lorenzo, G.; Dobrilla, G.; Dodero, M.; Galimberti, M.; Gasbarrini, G.; et al. Oral Cromolyn Sodium in Comparison with Elimination Diet in the Irritable Bowel Syndrome, Diarrheic Type Multicenter Study of 428 Patients. Scand. J. Gastroenterol. 1995, 30, 535–541. [Google Scholar] [CrossRef]
- Fritscher-Ravens, A.; Pflaum, T.; Mösinger, M.; Ruchay, Z.; Röcken, C.; Milla, P.J.; Das, M.; Böttner, M.; Wedel, T.; Schuppan, D.; et al. Many Patients With Irritable Bowel Syndrome Have Atypical Food Allergies Not Associated With Immunoglobulin E. Gastroenterology 2019, 157, 109.e5–118.e5. [Google Scholar] [CrossRef]
- Quinlan, P.T.; Lockton, S.; Irwin, J.; Lucas, A.L. The Relationship between Stool Hardness and Stool Composition in Breast- and Formula-Fed Infants. J. Pediatr. Gastroenterol. Nutr. 1995, 20, 81–90. [Google Scholar] [CrossRef]
- Iacono, G.; Bonventre, S.; Scalici, C.; Maresi, E.; Di Prima, L.; Soresi, M.; Di Ges??, G.; Noto, D.; Carroccio, A. Food intolerance and chronic constipation: Manometry and histology study. Eur. J. Gastroenterol. Hepatol. 2006, 18, 143–150. [Google Scholar] [CrossRef]
- Nocerino, R.; Pezzella, V.; Cosenza, L.; Amoroso, A.; Di Scala, C.; Amato, F.; Iacono, G.; Canani, R.B. The Controversial Role of Food Allergy in Infantile Colic: Evidence and Clinical Management. Nutrients 2015, 7, 2015–2025. [Google Scholar] [CrossRef]
- Gordon, M.; Biagioli, E.; Sorrenti, M.; Lingua, C.; Moja, L.; Banks, S.S.; Ceratto, S.; Savino, F. Dietary modifications for infantile colic. Cochrane Database Syst. Rev. 2018, 10, CD011029. [Google Scholar] [CrossRef] [PubMed]
- Natsume, O.; Kabashima, S.; Nakazato, J.; Yamamoto-Hanada, K.; Narita, M.; Kondo, M.; Saito-Abe, M.; Kishino, A.; Takimoto, T.; Inoue, E.; et al. Two-step egg introduction for prevention of egg allergy in high-risk infants with eczema (PETIT): A randomised, double-blind, placebo-controlled trial. Lancet 2017, 389, 276–286. [Google Scholar] [CrossRef]
- Qamer, S.; Deshmukh, M.; Patole, S. Probiotics for cow’s milk protein allergy: A systematic review of randomized controlled trials. Eur. J. Nucl. Med. Mol. Imaging 2019, 178, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Canani, R.B.; Nocerino, R.; Terrin, G.; Coruzzo, A.; Cosenza, L.; Leone, L.; Troncone, R. Effect of Lactobacillus GG on tolerance acquisition in infants with cow’s milk allergy: A randomized trial. J. Allergy Clin. Immunol. 2012, 129, 580–582.e5. [Google Scholar] [CrossRef] [PubMed]
- Leonard, S.A.; Jean-Christoph, C.; Kim, J.S.; Groetch, M.; Nowak-Wegrzyn, A. Baked Milk- and Egg-Containing Diet in the Management of Milk and Egg Allergy. J. Allergy Clin. Immunol. Pr. 2015, 3, 13–23. [Google Scholar] [CrossRef]
| . | FPIES | FPE | FPIAP |
|---|---|---|---|
| Age of presentation | Cow’s milk/soy: First weeks-months of life Solids: 4–7 months Can also occur in adults | 2–24 months Can also occur in older children | First weeks-months of life (<6 months) Can also occur in older children |
| Top culprit foods | Cow’s milk, soy (C > A) Rice, poultry, fish, fruits, vegetables (A > C) | Cow’s milk, soy Wheat, egg | Cow’s milk, soy Egg, corn, wheat |
| Multiple foods | Frequent ≥3 foods: 5–10% | Rare | Occasional |
| Feeding at onset | Formula | Formula | Exclusively BF (>50%) |
| Clinical presentation | (A): repeated vomiting, diarrhea, dehydration (shock: 15%), lethargy, pallor, hypothermia (C): intermittent vomiting, diarrhea, FTT | Diarrhea, intermittent vomiting, FTT, malabsorption (steatorrhea), bloody stools (rare) | Blood/mucus streaked stools, mild diarrhea Otherwise well-appearing |
| Co-morbid atopy | 40–60% Familial: 40–80% | 20–40% | 25–50% Familial: 30–60% |
| Laboratory anomalies | Anemia (C) Eosinophilia (C) Neutrophilia (A, C) Thrombocytosis (A) Methemoglobinemia (A, C) Metabolic acidosis (A, C) | Anemia Hypoalbuminemia Iron deficiency | Mild anemia Hypoalbuminemia (rare) Eosinophilia |
| Stool studies | Occult blood (A, C) PMN (A, C) Eosinophils (A, C) Reducing substances (C) | Fecal fat Low d-Xylose excretion | Gross/occult blood Eosinophils |
| Endoscopy/Histology | Friable mucosa Ulceration Villous atrophy Crypt abscesses Inflammatory cell infiltrates | Villous atrophy Crypt hyperplasia Lymphocytic infiltrate | Mild, focal colitis Eosinophilic infiltration Lymphonodular hyperplasia |
| Allergy evaluation | Negative; sIgE+ in 25% | Negative (not recommended) | Negative (not recommended) |
| Diagnosis | Clinical +/− OFC | Clinical & histological | Clinical +/− OFC |
| Treatment | Avoidance of offending foods | Avoidance of offending foods | Avoidance of offending foods (maternal exclusion diet if BF) |
| Time to improvement | (A) 4–12 h (<24 h) (C) 3–10 days | Several weeks (1–2 weeks) | 72 h (up to 2 weeks) |
| Natural history | Resolution < 3–5 y Later if sIgE+ or solid foods | Resolution < 1–2 years | Resolution < 1–2 years |
| FPIES | FPE | FPIAP | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Country | USA 1 | UK 2 | Spain 3 | Italy 4 | Australia 5 | Turkey 6 | Finland 7 | USA 8 | Turkey 9 |
| N (total) | N = 1340 | N = 54 | N = 336 | N = 66 | N = 265 | N = 27 | N = 54 | N = 95 | N = 359 |
| % | % | % | % | % | % | % | % | % | |
| Cow’s milk | 19–67 | 46 | 26–38 | 67 | 20–33 | 74 | 100 | 65 | 91–100 |
| Soy | 8–41 | 11 | 0–1 | 4 | 5–34 | - | 11 | 3 * | 0–3 * |
| Rice | 19–53 | 4 | 1–10 | 4 | 40–45 | 4 | - | - | - |
| Oat | 16–37 | 6 | 0–1 | - | 6–9 | - | - | - | - |
| Wheat | 1–16 | 11 | 0–1 | 2 | 0–3 | 4 | 37 | - | 0–4 |
| Corn | 2–8 | 2 | 0–3 | 2 | 0–1 | - | - | 6 | - |
| Eggs | 0–23 | 13 | 10–21 | 6 | 0–12 | - | 4 | 18 | 7–22 |
| Fish/Shellfish | 1–15 | 15 | 34–54 | 12 | 3–5 | 15 | - | - | 0–2 |
| Poultry | 5–10 | 7 | 1–4 | 3 | 3–8 | - | - | - | 0–3 |
| Meat | 3–18 | 4 | 1 | - | 3–4 | - | 2 | - | 0–10 |
| Sweet potato | 4–22 | - | - | - | 3–6 | - | - | - | - |
| Potato | 2–8 | 2 | 0–1 | - | 0–2 | 4 | - | - | 0–2 |
| Squash | 0–12 | - | - | - | - | - | - | - | - |
| Carrot | 0–7 | 4 | 0 | - | 0–1 | - | - | - | 0–1 |
| Banana | 4–24 | 6 | 0–1 | 3 | 3–4 | 4 | 4 | - | - |
| Avocado | 0–16 | - | - | - | 0–2 | - | - | - | - |
| Apple | 0–11 | 2 | 0–1 | - | 0–2 | - | - | - | 0–1 |
| Pear | 0–9 | - | 0–1 | - | 0–3 | - | - | - | - |
| Acute FPIES 1 | |
| Major Criteria, PLUS | Minor Criteria (≥3 Occurring with Episode) |
| 1. Vomiting 1–4 h after suspect food ingestion AND 2. Absence IgE-mediated allergic symptoms | 1. ≥2 episodes with same food 2. 1 episode with a different food 3. Lethargy 4. Pallor 5. Need for ER visit 6. Need for IV fluid support 7. Diarrhea within 24 h (usually 5–10 h) 8. Hypotension 9. Hypothermia |
| Chronic FPIES 2 | |
| Symptoms and severity | Criteria |
| Milder (lower doses with intermittent ingestion): 1. Intermittent vomiting and/or diarrhea 2. FTT 3. No dehydration or metabolic acidosis Severe (higher doses with chronic ingestion): 1. Intermittent but progressive vomiting and diarrhea (occasionally with blood) 2. Possible dehydration and metabolic acidosis | 1. Resolution of symptoms within days after elimination of offending food(s) 2. Acute recurrence of symptoms (vomiting in 1–4 h, diarrhea in <24 h, usually 5–10 h) when the food is reintroduced 3. Confirmatory OFC, or presumptive diagnosis if OFC not performed |
| FPE 3 | |
| 1. Generally <9 months of age at diagnosis, but can also present in older children 2. Repeated exposure to causative foods elicits gastrointestinal symptoms (predominantly vomiting and FTT), without alternative cause 3. Histologic confirmation of the diagnosis in a symptomatic child by biopsy of the small bowel showing villous injury, crypt hyperplasia and inflammation 4. Clinical and histological improvement after removal of offending food(s) 5. Exclusion of alternative causes | |
| FPIAP 4 | |
| 1. Mild rectal bleeding in an otherwise healthy infant 2. Resolution of symptoms after elimination of offending food(s) (if exclusively breastfed, resolution after a maternal elimination diet) 3. Recurrence of symptoms upon reintroduction of culprit food(s) in the diet (preferable) 4. Exclusion of other causes of rectal bleeding | |
| Acute FPIES | Chronic FPIES | FPE | FPIAP | |
|---|---|---|---|---|
| Allergic | Anaphylaxis Eosinophilic gastroenteropathies | FPIAP FPE Eosinophilic gastroenteropathies | Celiac disease Chronic FPIES Eosinophilic gastroenteropathies | FPIES FPE Eosinophilic gastroenteropathies |
| Infectious | Sepsis Viral/bacterial/ parasitic gastroenteritis | Viral/bacterial/ parasitic gastroenteritis | Viral/bacterial/ parasitic gastroenteritis | Viral/bacterial/ parasitic gastroenteritis |
| Gastrointestinal | Hirschsprung Pyloric stenosis Intussusception Volvulus NEC | GERD Hirschsprung Pyloric stenosis VEOIBD Cystic fibrosis | VEOIBD Cystic fibrosis | Anal fissure Swallowed maternal blood NEC Intussusception Volvulus Meckel diverticulum Intestinal duplication kyst Infantile polyp VEOIBD |
| Metabolic | Inborn errors of metabolism T1DM | Inborn errors of metabolism T1DM | Inborn errors of metabolism Congenital disaccharidase deficiency T1DM | - |
| Hematologic | Congenital methemoglobinemia | Congenital methemoglobinemia | - | Coagulation defect Thrombocytopenia |
| Neuro- logic | Cyclic vomiting Intracranial mass | Cyclic vomiting Intracranial mass | - | - |
| Cardiovascular | Congenital heart defect Cardiomyopathy Arrythmia | Congenital heart defect Cardiomyopathy | - | Vascular malformation |
| Endocri- nologic | Congenital adrenal hypoplasia | Congenital adrenal hypoplasia | Congenital adrenal hypoplasia | - |
| Immunologic | - | PID Autoimmune enteropathy | PID Autoimmune enteropathy | - |
| Psychologic | Food aversion | Food aversion | Food aversion Neglect | - |
| FPIES | FPE | FPIAP | |
|---|---|---|---|
| Cow’s milk | 1st choice: EHF (10–20% reactivity) AAF if failure of EHF Soy formula: 40–60% co-reactivity † Rice formula: co-reactivity unknown May tolerate baked cow’s milk | First choice: EHF (20% reactivity) AAF if failure of EHF Soy formula: 10–30% co-reactivity † | If BF: 1st choice: maternal elimination diet Alternative: EHF If on formula: 1st choice: EHF (10–20% reactivity) AAF if failure of EHF Soy formula: 10–30% co-reactivity † |
| Soy | Cow’s milk: 40% co-reactivity † | - | May be eliminated if no improvement with cow’s milk exclusion alone |
| Rice | Oats: 25–40% co-reactivity Wheat: 0–5% co-reactivity Corn: 1%: co-reactivity | - | - |
| Chicken | Avoid all poultry *: up to 40% co-reactivity | - | - |
| Egg | May tolerate baked eggs | - | May be eliminated if no improvement with cow’s milk/soy exclusion alone |
| Fish | Avoid all fish *: up to 80% co-reactivity between white & red fish Shellfish: 50% co-reactivity | - | - |
| Maternal elimination diet in BF | No, unless symptomatic | Unknown | Yes |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Labrosse, R.; Graham, F.; Caubet, J.-C. Non-IgE-Mediated Gastrointestinal Food Allergies in Children: An Update. Nutrients 2020, 12, 2086. https://doi.org/10.3390/nu12072086
Labrosse R, Graham F, Caubet J-C. Non-IgE-Mediated Gastrointestinal Food Allergies in Children: An Update. Nutrients. 2020; 12(7):2086. https://doi.org/10.3390/nu12072086
Chicago/Turabian StyleLabrosse, Roxane, François Graham, and Jean-Christoph Caubet. 2020. "Non-IgE-Mediated Gastrointestinal Food Allergies in Children: An Update" Nutrients 12, no. 7: 2086. https://doi.org/10.3390/nu12072086
APA StyleLabrosse, R., Graham, F., & Caubet, J.-C. (2020). Non-IgE-Mediated Gastrointestinal Food Allergies in Children: An Update. Nutrients, 12(7), 2086. https://doi.org/10.3390/nu12072086




