Nutrients and Dietary Patterns Related to Osteoporosis
Abstract
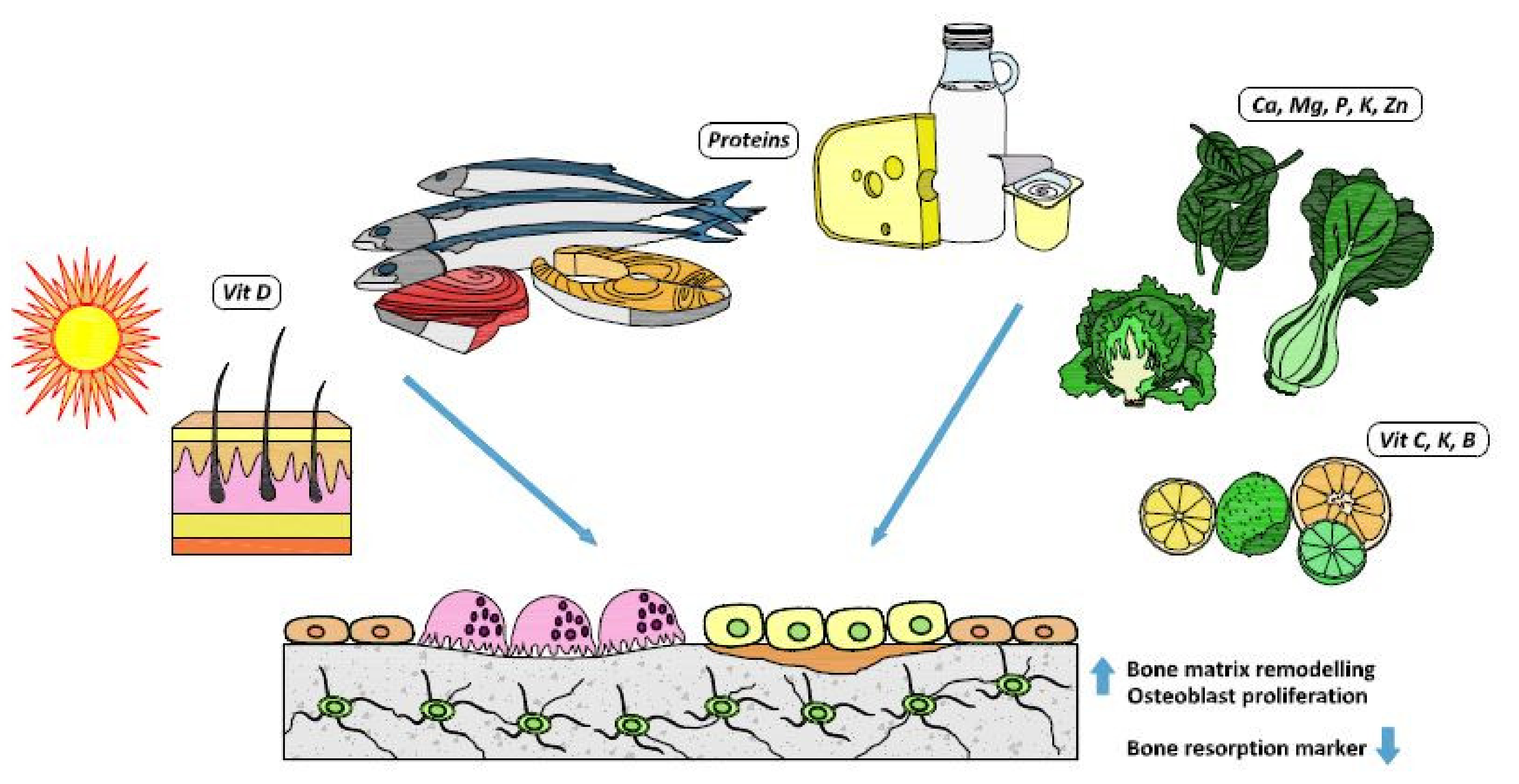
1. Introduction
2. Osteoporosis and Related Nutrients
2.1. Calcium, Vitamin D and Dairy Products
2.2. Other Minerals
2.3. Protein Intake
2.4. Vitamins K and C
2.5. Omega-3 Polyunsaturated Fatty Acids and Other Nutrients
3. Recent Studies on Reference Dietary Patterns
3.1. Mediterranean Diet
3.2. Western Diet
3.3. Asian Diet
3.4. Vegetarian Diets
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Black, D.M.; Rosen, C.J. Clinical practice. Postmenopausal osteoporosis. N. Engl. J. Med. 2016, 374, 254–262. [Google Scholar] [CrossRef]
- Kanis, J.A.; Cooper, C.; Rizzoli, R.; Reginster, J.-Y. European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2019, 30, 3–44. [Google Scholar] [CrossRef]
- Levis, S.; Lagari, V.S. The role of diet in osteoporosis prevention and management. Curr. Osteoporos. Rep. 2012, 10, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Chiodini, I.; Bolland, M.J. Calcium supplementation in osteoporosis: Useful or harmful? Eur. J. Endocrinol. 2018, 178, D13–D25. [Google Scholar] [CrossRef]
- Kanis, J.A.; Cooper, C.; Rizzoli, R.; Abrahamsen, B.; Al-Daghri, N.M.; Brandi, M.L.; Cannata-Andia, J.; Cortet, B.; Dimai, H.P.; Ferrari, S.; et al. Identification and management of patients at increased risk of osteoporotic fracture: Outcomes of an ESCEO expert consensus meeting. Osteoporos. Int. 2017, 28, 2023–2034. [Google Scholar] [CrossRef] [PubMed]
- Compston, J.; Cooper, A.; Cooper, C.; Gittoes, N.; Gregson, C.; Harvey, N.; Hope, S.; Kanis, J.A.; McCloskey, E.V.; Poole, K.E.S.; et al. UK clinical guideline for the prevention and treatment of osteoporosis. Arch. Osteoporos. 2017, 12, 43. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Reid, I.R. Should we prescribe calcium or vitamin D supplements to treat or prevent osteoporosis? Climacteric 2015, 18 (Suppl. 2), 22–31. [Google Scholar] [CrossRef]
- Adami, S.; Giannini, S.; Bianchi, G.; Sinigaglia, L.; Di Munno, O.; Fiore, C.E.; Minisola, S.; Rossini, M. Vitamin D status and response to treatment in post-menopausal osteoporosis. Osteoporos. Int. 2009, 20, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Mavrogianni, C.; van den Heuvel, E.; Singh-Povel, C.M.; Kiely, M.; Cashman, K.D. Reduced-fat Gouda-type cheese enriched with vitamin D3 effectively prevents vitamin D deficiency during winter months in postmenopausal women in Greece. Eur. J. Nutr. 2017, 56, 2367–2377. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.; Duffy, S.; O’Grady, M.; Jakobsen, J.; Galvin, K.; Teahan-Dillon, J.; Kerry, J.; Kelly, A.; O’Doherty, J.; Higgins, S.; et al. Vitamin D-enhanced eggs are protective of wintertime serum 25-hydroxyvitamin D in a randomized controlled trial of adults. Am. J. Clin. Nutr. 2016, 104, 629–637. [Google Scholar] [CrossRef]
- Tangestani, H.; Djafarian, K.; Emamat, H.; Arabzadegan, N.; Shab-Bidar, S. Efficacy of vitamin D fortified foods on bone mineral density and serum bone biomarkers: A systematic review and meta-analysis of interventional studies. Crit. Rev. Food Sci. Nutr. 2020, 60, 1094–1103. [Google Scholar] [CrossRef]
- Tai, V.; Leung, W.; Grey, A.; Reid, I.R.; Bolland, M.J. Calcium intake and bone mineral density: Systematic review and meta-analysis. BMJ 2015, 351, h4183. [Google Scholar] [CrossRef] [PubMed]
- Feskanich, D.; Meyer, H.E.; Fung, T.T.; Bischoff-Ferrari, H.A.; Willett, W.C. Milk and other dairy foods and risk of hip fracture in men and women. Osteoporos. Int. 2018, 29, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Garcia, R.; Mendoza, N.; Palacios, S.; Salas, N.; Quesada-Charneco, M.; Garcia-Martin, A.; Fonolla, J.; Lara-Villoslada, F.; Muñoz-Torres, M. Effects of daily intake of calcium and vitamin D-enriched milk in healthy postmenopausal women: A randomized, controlled, double-blind nutritional study. J. Women’s Health 2018, 27, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Biver, E. Effects of fermented milk products on bone. Calcif. Tissue Int. 2018, 102, 489–500. [Google Scholar] [CrossRef]
- Li, J.-Y.; Chassaing, B.; Tyagi, A.M.; Vaccaro, C.; Luo, T.; Adams, J.; Darby, T.M.; Weitzmann, M.N.; Mulle, J.G.; Gewirtz, A.T.; et al. Sex steroid deficiency-associated bone loss is microbiota dependent and prevented by probiotics. J. Clin. Investig. 2016, 126, 2049–2063. [Google Scholar] [CrossRef]
- Gennari, C. Calcium and vitamin D nutrition and bone disease of the elderly. Public Health Nutr. 2001, 4, 547–559. [Google Scholar] [CrossRef]
- Rizzoli, R.; Biver, E. Are probiotics the new calcium and vitamin D for bone health? Curr. Osteoporos. Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Boonen, S.; Brandi, M.-L.; Bruyere, O.; Cooper, C.; Kanis, J.A.; Kaufman, J.-M.; Ringe, J.D.; Weryha, G.; Reginster, J.-Y. Vitamin D supplementation in elderly or postmenopausal women: A 2013 update of the 2008 recommendations from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Curr. Med. Res. Opin. 2013, 29, 305–313. [Google Scholar] [CrossRef]
- Willett, W.C.; Ludwig, D.S. Milk and Health. N. Engl. J. Med. 2020, 382, 644–654. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Kiel, D.P.; Dawson-Hughes, B.; Orav, J.E.; Li, R.; Spiegelman, D.; Dietrich, T.; Willett, W.C. Dietary calcium and serum 25-hydroxyvitamin D status in relation to BMD among U.S. adults. J. Bone Miner. Res. 2009, 24, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Baron, J.A.; Burckhardt, P.; Li, R.; Spiegelman, D.; Specker, B.; Orav, J.E.; Wong, J.B.; Staehelin, H.B.; et al. Calcium intake and hip fracture risk in men and women: A meta-analysis of prospective cohort studies and randomized controlled trials. Am. J. Clin. Nutr. 2007, 86, 1780–1790. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Baron, J.A.; Kanis, J.A.; Orav, E.J.; Staehelin, H.B.; Kiel, D.P.; Burckhardt, P.; Henschkowski, J.; Spiegelman, D.; et al. Milk intake and risk of hip fracture in men and women: A meta-analysis of prospective cohort studies. J. Bone Miner. Res. 2011, 26, 833–839. [Google Scholar] [CrossRef]
- Wallace, T.C.; Jun, S.; Zou, P.; McCabe, G.P.; Craig, B.A.; Cauley, J.A.; Weaver, C.M.; Bailey, R.L. Dairy intake is not associated with improvements in bone mineral density or risk of fractures across the menopause transition: Data from the Study of Women’s Health Across the Nation. Menopause 2020. [Google Scholar] [CrossRef]
- Michaëlsson, K.; Wolk, A.; Langenskiöld, S.; Basu, S.; Warensjö Lemming, E.; Melhus, H.; Byberg, L. Milk intake and risk of mortality and fractures in women and men: Cohort studies. BMJ 2014, 349, g6015. [Google Scholar] [CrossRef]
- Hidayat, K.; Du, X.; Shi, B.-M.; Qin, L.-Q. Systematic review and meta-analysis of the association between dairy consumption and the risk of hip fracture: Critical interpretation of the currently available evidence. Osteoporos. Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, E.G.H.M.; Steijns, J.M.J.M. Dairy products and bone health: How strong is the scientific evidence? Nutr. Res. Rev. 2018, 31, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.H.; Kim, J.H.; Hong, A.R.; Lee, J.H.; Kim, S.W.; Shin, C.S. Dietary potassium intake is beneficial to bone health in a low calcium intake population: The Korean National Health and Nutrition Examination Survey (KNHANES) (2008–2011). Osteoporos. Int. 2017, 28, 1577–1585. [Google Scholar] [CrossRef] [PubMed]
- Lambert, H.; Frassetto, L.; Moore, J.B.; Torgerson, D.; Gannon, R.; Burckhardt, P.; Lanham-New, S. The effect of supplementation with alkaline potassium salts on bone metabolism: A meta-analysis. Osteoporos. Int. 2015, 26, 1311–1318. [Google Scholar] [CrossRef]
- Erem, S.; Atfi, A.; Razzaque, M.S. Anabolic effects of vitamin D and magnesium in aging bone. J. Steroid Biochem. Mol. Biol. 2019, 193, 105400. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press: Washington, DC, USA, 2006; Volume 55, ISBN 978-0-309-15742-1. [Google Scholar]
- De Baaij, J.H.F.; Hoenderop, J.G.J.; Bindels, R.J.M. Magnesium in man: Implications for health and disease. Physiol. Rev. 2015, 95, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Uwitonze, A.M.; Razzaque, M.S. Role of magnesium in vitamin D activation and function. J. Am. Osteopath. Assoc. 2018, 118, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Rosanoff, A.; Weaver, C.M.; Rude, R.K. Suboptimal magnesium status in the United States: Are the health consequences underestimated? Nutr. Rev. 2012, 70, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Brodowski, J. Levels of ionized magnesium in women with various stages of postmenopausal osteoporosis progression evaluated on the basis of densitometric examinations. Przegl. Lek. 2000, 57, 714–716. [Google Scholar]
- Okyay, E.; Ertugrul, C.; Acar, B.; Sisman, A.R.; Onvural, B.; Ozaksoy, D. Comparative evaluation of serum levels of main minerals and postmenopausal osteoporosis. Maturitas 2013, 76, 320–325. [Google Scholar] [CrossRef]
- Veronese, N.; Stubbs, B.; Solmi, M.; Noale, M.; Vaona, A.; Demurtas, J.; Maggi, S. Dietary magnesium intake and fracture risk: Data from a large prospective study. Br. J. Nutr. 2017, 117, 1570–1576. [Google Scholar] [CrossRef]
- Hayhoe, R.P.G.; Lentjes, M.A.H.; Luben, R.N.; Khaw, K.-T.; Welch, A.A. Dietary magnesium and potassium intakes and circulating magnesium are associated with heel bone ultrasound attenuation and osteoporotic fracture risk in the EPIC-Norfolk cohort study. Am. J. Clin. Nutr. 2015, 102, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Stevenson, J.C.; Bauer, J.M.; van Loon, L.J.C.; Walrand, S.; Kanis, J.A.; Cooper, C.; Brandi, M.-L.; Diez-Perez, A.; Reginster, J.-Y. The role of dietary protein and vitamin D in maintaining musculoskeletal health in postmenopausal women: A consensus statement from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Maturitas 2014, 79, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Bonjour, J.-P. Nutritional disturbance in acid-base balance and osteoporosis: A hypothesis that disregards the essential homeostatic role of the kidney. Br. J. Nutr. 2013, 110, 1168–1177. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Biver, E.; Bonjour, J.-P.; Coxam, V.; Goltzman, D.; Kanis, J.A.; Lappe, J.; Rejnmark, L.; Sahni, S.; Weaver, C.; et al. Benefits and safety of dietary protein for bone health-an expert consensus paper endorsed by the European Society for Clinical and Economical Aspects of Osteopororosis, Osteoarthritis, and Musculoskeletal Diseases and by the International Osteoporosis Fo. Osteoporos. Int. 2018, 29, 1933–1948. [Google Scholar] [CrossRef]
- Groenendijk, I.; den Boeft, L.; van Loon, L.J.C.; de Groot, L.C. High versus low dietary protein intake and bone health in older adults: A systematic review and meta-analysis. Comput. Struct. Biotechnol. J. 2019, 17, 1101–1112. [Google Scholar] [CrossRef] [PubMed]
- Rizzoli, R.; Bischoff-Ferrari, H.; Dawson-Hughes, B.; Weaver, C. Nutrition and bone health in women after the menopause. Women’s Health (Lond. Engl.) 2014, 10, 599–608. [Google Scholar] [CrossRef] [PubMed]
- New, S.A. Intake of fruit and vegetables: Implications for bone health. Proc. Nutr. Soc. 2003, 62, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Ahmadieh, H.; Arabi, A. Vitamins and bone health: Beyond calcium and vitamin D. Nutr. Rev. 2011, 69, 584–598. [Google Scholar] [CrossRef]
- Mott, A.; Bradley, T.; Wright, K.; Cockayne, E.S.; Shearer, M.J.; Adamson, J.; Lanham-New, S.A.; Torgerson, D.J. Effect of vitamin K on bone mineral density and fractures in adults: An updated systematic review and meta-analysis of randomised controlled trials. Osteoporos. Int. 2019, 30, 1543–1559. [Google Scholar] [CrossRef]
- Veronese, N.; Bano, G.; Bertozzo, G.; Granziera, S.; Solmi, M.; Manzato, E.; Sergi, G.; Cohen, A.T.; Correll, C.U. Vitamin K antagonists’ use and fracture risk: Results from a systematic review and meta-analysis. J. Thromb. Haemost. 2015, 13, 1665–1675. [Google Scholar] [CrossRef] [PubMed]
- Finck, H.; Hart, A.R.; Jennings, A.; Welch, A.A. Is there a role for vitamin C in preventing osteoporosis and fractures? A review of the potential underlying mechanisms and current epidemiological evidence. Nutr. Res. Rev. 2014, 27, 268–283. [Google Scholar] [CrossRef]
- Malmir, H.; Shab-Bidar, S.; Djafarian, K. Vitamin C intake in relation to bone mineral density and risk of hip fracture and osteoporosis: A systematic review and meta-analysis of observational studies. Br. J. Nutr. 2018, 119, 847–858. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, C.; Bo, Y.; You, J.; Zhu, Y.; Duan, D.; Cui, H.; Lu, Q. Dietary vitamin C intake and the risk of hip fracture: A dose-response meta-analysis. Osteoporos. Int. 2018, 29, 79–87. [Google Scholar] [CrossRef]
- Griel, A.E.; Kris-Etherton, P.M.; Hilpert, K.F.; Zhao, G.; West, S.G.; Corwin, R.L. An increase in dietary n-3 fatty acids decreases a marker of bone resorption in humans. Nutr. J. 2007, 6, 2. [Google Scholar] [CrossRef]
- Sun, D.; Krishnan, A.; Zaman, K.; Lawrence, R.; Bhattacharya, A.; Fernandes, G. Dietary n-3 fatty acids decrease osteoclastogenesis and loss of bone mass in ovariectomized mice. J. Bone Miner. Res. 2003, 18, 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Mangano, K.M.; Sahni, S.; Kerstetter, J.E.; Kenny, A.M.; Hannan, M.T. Polyunsaturated fatty acids and their relation with bone and muscle health in adults. Curr. Osteoporos. Rep. 2013, 11, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Zhang, X.; Li, Z.; Bai, H.; Chen, L. Effects of omega-3 fatty acids on bone turnover markers in postmenopausal women: Systematic review and meta-analysis. Climacteric 2017, 20, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Fonolla-Joya, J.; Reyes-García, R.; García-Martín, A.; López-Huertas, E.; Muñoz-Torres, M. Daily intake of milk enriched with n-3 fatty acids, oleic acid, and calcium improves metabolic and bone biomarkers in postmenopausal women. J. Am. Coll. Nutr. 2016, 35, 529–536. [Google Scholar] [CrossRef]
- Lavado-García, J.; Roncero-Martin, R.; Moran, J.M.; Pedrera-Canal, M.; Aliaga, I.; Leal-Hernandez, O.; Rico-Martin, S.; Canal-Macias, M.L. Long-chain omega-3 polyunsaturated fatty acid dietary intake is positively associated with bone mineral density in normal and osteopenic Spanish women. PLoS ONE 2018, 13, e0190539. [Google Scholar] [CrossRef]
- Sadeghi, O.; Djafarian, K.; Ghorabi, S.; Khodadost, M.; Nasiri, M.; Shab-Bidar, S. Dietary intake of fish, n-3 polyunsaturated fatty acids and risk of hip fracture: A systematic review and meta-analysis on observational studies. Crit. Rev. Food Sci. Nutr. 2019, 59, 1320–1333. [Google Scholar] [CrossRef]
- Fratoni, V.; Brandi, M.L. B vitamins, homocysteine and bone health. Nutrients 2015, 7, 2176–2192. [Google Scholar] [CrossRef]
- Van Wijngaarden, J.P.; Doets, E.L.; Szczecinska, A.; Souverein, O.W.; Duffy, M.E.; Dullemeijer, C.; Cavelaars, A.E.J.M.; Pietruszka, B.; Van’t Veer, P.; Brzozowska, A.; et al. Vitamin B12, folate, homocysteine, and bone health in adults and elderly people: A systematic review with meta-analyses. J. Nutr. Metab. 2013, 2013, 486186. [Google Scholar] [CrossRef]
- Nielsen, F.H.; Lukaski, H.C.; Johnson, L.K.; Roughead, Z.K.F. Reported zinc, but not copper, intakes influence whole-body bone density, mineral content and T score responses to zinc and copper supplementation in healthy postmenopausal women. Br. J. Nutr. 2011, 106, 1872–1879. [Google Scholar] [CrossRef]
- Benetou, V.; Orfanos, P.; Pettersson-Kymmer, U.; Bergstrom, U.; Svensson, O.; Johansson, I.; Berrino, F.; Tumino, R.; Borch, K.B.; Lund, E.; et al. Mediterranean diet and incidence of hip fractures in a European cohort. Osteoporos. Int. 2013, 24, 1587–1598. [Google Scholar] [CrossRef]
- Benetou, V.; Orfanos, P.; Feskanich, D.; Michaëlsson, K.; Pettersson-Kymmer, U.; Byberg, L.; Eriksson, S.; Grodstein, F.; Wolk, A.; Jankovic, N.; et al. Mediterranean diet and hip fracture incidence among older adults: The CHANCES project. Osteoporos. Int. 2018, 29, 1591–1599. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Butler, L.M.; van Dam, R.M.; Ang, L.-W.; Yuan, J.-M.; Koh, W.-P. Adherence to a vegetable-fruit-soy dietary pattern or the Alternative Healthy Eating Index is associated with lower hip fracture risk among Singapore Chinese. J. Nutr. 2014, 144, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Zupo, R.; Lampignano, L.; Lattanzio, A.; Mariano, F.; Osella, A.R.; Bonfiglio, C.; Giannelli, G.; De Pergola, G. Association between adherence to the Mediterranean Diet and circulating Vitamin D levels. Int. J. Food Sci. Nutr. 2020, 1–7. [Google Scholar] [CrossRef]
- Shen, C.-L.; von Bergen, V.; Chyu, M.-C.; Jenkins, M.R.; Mo, H.; Chen, C.-H.; Kwun, I.-S. Fruits and dietary phytochemicals in bone protection. Nutr. Res. 2012, 32, 897–910. [Google Scholar] [CrossRef] [PubMed]
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; Feriche, B.; Lorenzo, M.L.; Olea, F. Mediterranean diet and bone mineral density in two age groups of women. Int. J. Food Sci. Nutr. 2013, 64, 155–161. [Google Scholar] [CrossRef]
- Palomeras-Vilches, A.; Viñals-Mayolas, E.; Bou-Mias, C.; Jordà-Castro, M.; Agüero-Martínez, M.; Busquets-Barceló, M.; Pujol-Busquets, G.; Carrion, C.; Bosque-Prous, M.; Serra-Majem, L.; et al. Adherence to the mediterranean diet and bone fracture risk in middle-aged women: A case control study. Nutrients 2019, 11, 2508. [Google Scholar] [CrossRef]
- Pérez-Rey, J.; Roncero-Martín, R.; Rico-Martín, S.; Rey-Sánchez, P.; Pedrera-Zamorano, J.D.; Pedrera-Canal, M.; López-Espuela, F.; Lavado García, J.M. Adherence to a mediterranean diet and bone mineral density in Spanish premenopausal women. Nutrients 2019, 11, 555. [Google Scholar] [CrossRef]
- Savanelli, M.C.; Barrea, L.; Macchia, P.E.; Savastano, S.; Falco, A.; Renzullo, A.; Scarano, E.; Nettore, I.C.; Colao, A.; Di Somma, C. Preliminary results demonstrating the impact of Mediterranean diet on bone health. J. Transl. Med. 2017, 15, 81. [Google Scholar] [CrossRef]
- Da Silva, T.R.; Martins, C.C.; Ferreira, L.L.; Spritzer, P.M. Mediterranean diet is associated with bone mineral density and muscle mass in postmenopausal women. Climacteric 2019, 22, 162–168. [Google Scholar] [CrossRef]
- Malmir, H.; Saneei, P.; Larijani, B.; Esmaillzadeh, A. Adherence to Mediterranean diet in relation to bone mineral density and risk of fracture: A systematic review and meta-analysis of observational studies. Eur. J. Nutr. 2018, 57, 2147–2160. [Google Scholar] [CrossRef]
- García-Martínez, O.; Rivas, A.; Ramos-Torrecillas, J.; De Luna-Bertos, E.; Ruiz, C. The effect of olive oil on osteoporosis prevention. Int. J. Food Sci. Nutr. 2014, 65, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Roncero-Martín, R.; Aliaga Vera, I.; Moreno-Corral, L.J.; Moran, J.M.; Lavado-Garcia, J.M.; Pedrera-Zamorano, J.D.; Pedrera-Canal, M. Olive oil consumption and bone microarchitecture in Spanish women. Nutrients 2018, 10, 968. [Google Scholar] [CrossRef]
- Fernández-Real, J.M.; Bulló, M.; Moreno-Navarrete, J.M.; Ricart, W.; Ros, E.; Estruch, R.; Salas-Salvadó, J. A Mediterranean diet enriched with olive oil is associated with higher serum total osteocalcin levels in elderly men at high cardiovascular risk. J. Clin. Endocrinol. Metab. 2012, 97, 3792–3798. [Google Scholar] [CrossRef] [PubMed]
- García-Gavilán, J.F.; Bulló, M.; Canudas, S.; Martínez-González, M.A.; Estruch, R.; Giardina, S.; Fitó, M.; Corella, D.; Ros, E.; Salas-Salvadó, J. Extra virgin olive oil consumption reduces the risk of osteoporotic fractures in the PREDIMED trial. Clin. Nutr. 2018, 37, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Sahni, S.; Mangano, K.M.; McLean, R.R.; Hannan, M.T.; Kiel, D.P. Dietary approaches for bone health: Lessons from the framingham osteoporosis study. Curr. Osteoporos. Rep. 2015, 13, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Movassagh, E.Z.; Vatanparast, H. Current evidence on the association of dietary patterns and bone health: A scoping review. Adv. Nutr. 2017, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Skinner, J.; Guile, G.R.; Cassidy, A.; Spector, T.D.; MacGregor, A.J. Diet and bone mineral density study in postmenopausal women from the TwinsUK registry shows a negative association with a traditional English dietary pattern and a positive association with wine. Am. J. Clin. Nutr. 2011, 94, 1371–1375. [Google Scholar] [CrossRef]
- Cao, J.J. Effects of obesity on bone metabolism. J. Orthop. Surg. Res. 2011, 6, 30. [Google Scholar] [CrossRef]
- Heaney, R.P. Role of dietary sodium in osteoporosis. J. Am. Coll. Nutr. 2006, 25, 271S–276S. [Google Scholar] [CrossRef]
- Calvo, M.S.; Moshfegh, A.J.; Tucker, K.L. Assessing the health impact of phosphorus in the food supply: Issues and considerations. Adv. Nutr. 2014, 5, 104–113. [Google Scholar] [CrossRef]
- Nicoll, R.; McLaren Howard, J. The acid-ash hypothesis revisited: A reassessment of the impact of dietary acidity on bone. J. Bone Miner. Metab. 2014, 32, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Messina, M. Soy foods, isoflavones, and the health of postmenopausal women. Am. J. Clin. Nutr. 2014, 100, 423S–430S. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Lee, S.-K.; Chun, O.K. Soy isoflavones and osteoporotic bone loss: A review with an emphasis on modulation of bone remodeling. J. Med. Food 2016, 19, 1–14. [Google Scholar] [CrossRef]
- Shin, S.; Joung, H. A dairy and fruit dietary pattern is associated with a reduced likelihood of osteoporosis in Korean postmenopausal women. Br. J. Nutr. 2013, 110, 1926–1933. [Google Scholar] [CrossRef]
- Hirata, H.; Kitamura, K.; Saito, T.; Kobayashi, R.; Iwasaki, M.; Yoshihara, A.; Watanabe, Y.; Oshiki, R.; Nishiwaki, T.; Nakamura, K. Association between dietary intake and bone mineral density in japanese postmenopausal women: The Yokogoshi cohort study. Tohoku J. Exp. Med. 2016, 239, 95–101. [Google Scholar] [CrossRef]
- Perna, S.; Avanzato, I.; Nichetti, M.; D’Antona, G.; Negro, M.; Rondanelli, M. Association between dietary patterns of meat and fish consumption with bone mineral density or fracture risk: A systematic literature. Nutrients 2017, 9, 1029. [Google Scholar] [CrossRef] [PubMed]
- Tucker, K.L. Vegetarian diets and bone status. Am. J. Clin. Nutr. 2014, 100, 329S–335S. [Google Scholar] [CrossRef] [PubMed]
- Ho-Pham, L.T.; Nguyen, N.D.; Nguyen, T.V. Effect of vegetarian diets on bone mineral density: A Bayesian meta-analysis. Am. J. Clin. Nutr. 2009, 90, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Appleby, P.; Roddam, A.; Allen, N.; Key, T. Comparative fracture risk in vegetarians and nonvegetarians in EPIC-Oxford. Eur. J. Clin. Nutr. 2007, 61, 1400–1406. [Google Scholar] [CrossRef]
- Fabiani, R.; Naldini, G.; Chiavarini, M. Dietary patterns in relation to low bone mineral density and fracture risk: A systematic review and meta-analysis. Adv. Nutr. 2019, 10, 219–236. [Google Scholar] [CrossRef]
| Source | Calcium (mg) | Phosphorous (mg) | Potassium (mg) | Vitamin D (IU) | Protein (g) |
|---|---|---|---|---|---|
| Whole fat milk | 119 | 93 | 151 | * | 3.3 |
| Skimmed milk | 122 | 101 | 156 | * | 3.4 |
| Swiss cheese | 791 | 567 | 77 | 20 | 26.9 |
| Cheddar cheese | 721 | 512 | 98 | 24 | 24.9 |
| Cream cheese | 98 | 106 | 138 | * | 5.9 |
| Yogurt low fat | 183 | 144 | 234 | * | 5.3 |
| Ice cream | 128 | 105 | 199 | 0 | 3.5 |
| Wild salmon | 9 | 0 | 360 | 600–1000 | 38 |
| Eggs | 56 | 197 | 138 | 20 | 12.5 |
| Dietary Pattern | Name of the Study | No. Participants (Sex), Mean Age/Age Range | Effect on BMD | Effect on Fracture Risk |
|---|---|---|---|---|
| Mediterranean Diet | EPIC Study [61] | 188,795 (139,981 women and 48,814 men) 48.6 years (±10.8) | 7% decrease in hip fracture incidence | |
| CHANCES Project [62] | 140,775 (116,176 women, 24,599 men) 60 years and older | 4% decrease in hip fracture risk | ||
| Meta-analysis, Malmir et al. [71] | 358,746 13–80 years | Positive association with lumbar spine, femoral neck and total hip BMD | 21% reduced risk of hip fracture | |
| Asian Diet | Singapore Chinese Health Study [63]; vegetables–fruit–soy | 63,257 (35,241 women and 27,913 men) 45–74 years | 34% reduced risk of hip fracture | |
| Osteoporosis Korean Health and Nutrition Examination Survey 2008–10 [85] | 3735 (postmenopausal Women) 64 ± 9 years | Dairy and fruit pattern decreased the risk of osteoporosis of the lumbar spine (53%); white rice, kimchi and seaweed dietary pattern was negatively associated with bone health | ||
| Western Diet | Co-twin controlled study, United Kingdom [78] | 4928 (postmenopausal Women) 56 ± 12 years | Inversely associated with BMD in femoral neck | |
| Framingham Offspring Study, United States, [76] | 2740 (1534 women and 1206 men) 29–86 years | Higher intakes of red meat and processed food were inversely associated with femoral neck BMD | ||
| Vegetarian Diet | Bayesian meta-analysis [89] | 2749 (1880 women and 869 men) 20–79 years | 4% lower BMD (95% CI: 2%, 7%) at femoral neck and lumbar spine | |
| EPIC Oxford study [90] | 34,696 (26,749 women and 7947 men) 20–89 years | Fracture incidence rate ratios: 1.00 (0.89–1.13) for vegetarians and 1.30 (1.02–1.66) for vegans |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Garach, A.; García-Fontana, B.; Muñoz-Torres, M. Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients 2020, 12, 1986. https://doi.org/10.3390/nu12071986
Muñoz-Garach A, García-Fontana B, Muñoz-Torres M. Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients. 2020; 12(7):1986. https://doi.org/10.3390/nu12071986
Chicago/Turabian StyleMuñoz-Garach, Araceli, Beatriz García-Fontana, and Manuel Muñoz-Torres. 2020. "Nutrients and Dietary Patterns Related to Osteoporosis" Nutrients 12, no. 7: 1986. https://doi.org/10.3390/nu12071986
APA StyleMuñoz-Garach, A., García-Fontana, B., & Muñoz-Torres, M. (2020). Nutrients and Dietary Patterns Related to Osteoporosis. Nutrients, 12(7), 1986. https://doi.org/10.3390/nu12071986






