β-Hydroxybutyrate Increases Exercise Capacity Associated with Changes in Mitochondrial Function in Skeletal Muscle
Abstract
1. Introduction
2. Materials and Methods
2.1. Chronic β-HB Supplementation
2.2. Exercise Performance Test
2.3. Grip Strength Test
2.4. Blood Ketone Measurements
2.5. Transmission Electron Microscopy
2.6. Skeletal Muscle Fiber Permeabilization (PmFB)
2.7. Mitochondrial Oxygen Consumption
2.8. Western Blot Analysis
2.9. RNA Isolation and RT-PCR
2.10. Statistical Analysis
3. Results
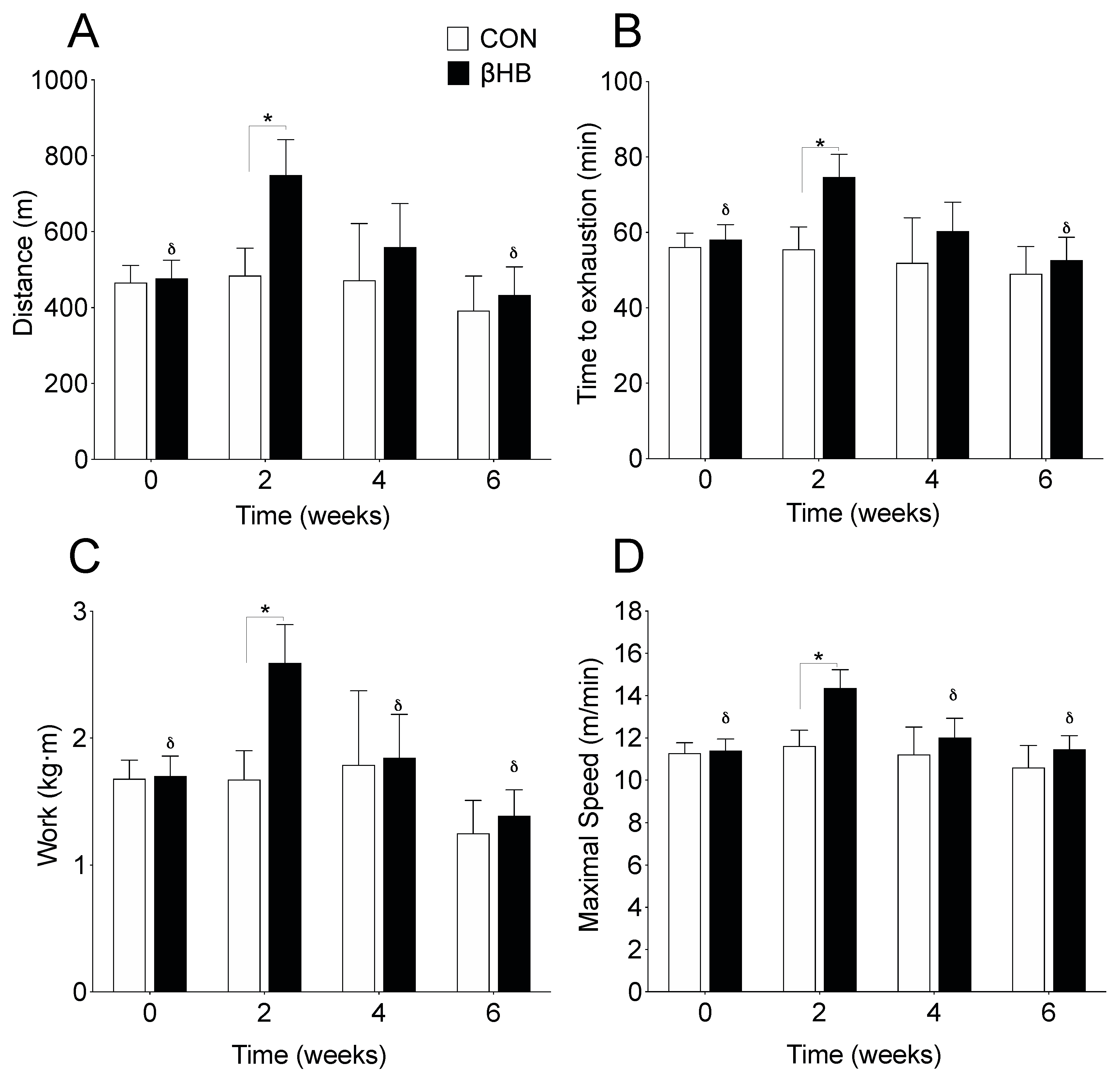
3.1. β-HB Supplementation Improves Endurance Capacity
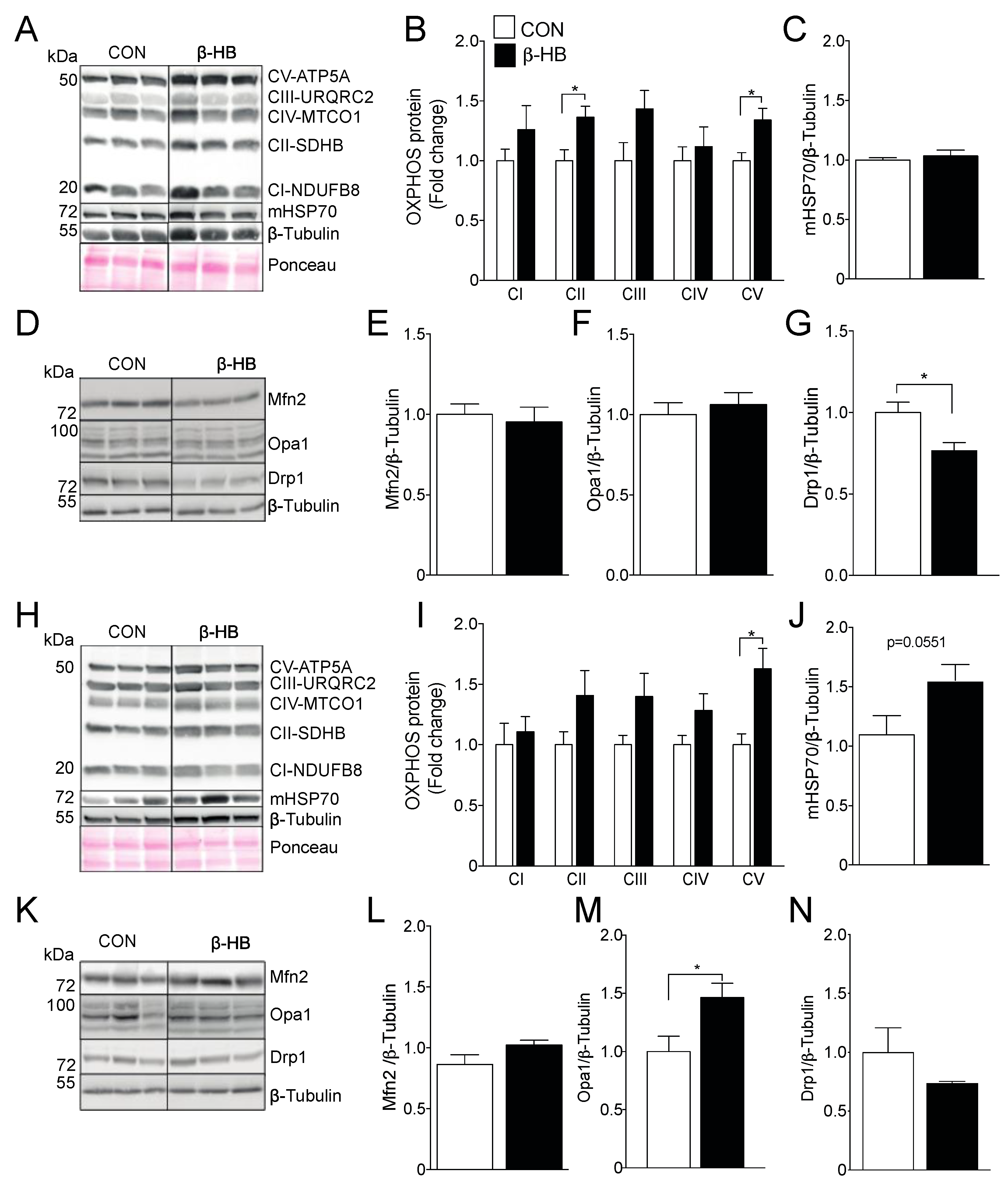
3.2. β-HB Long-Term Supplementation Altered Electron Transport Chain Subunit Protein Content
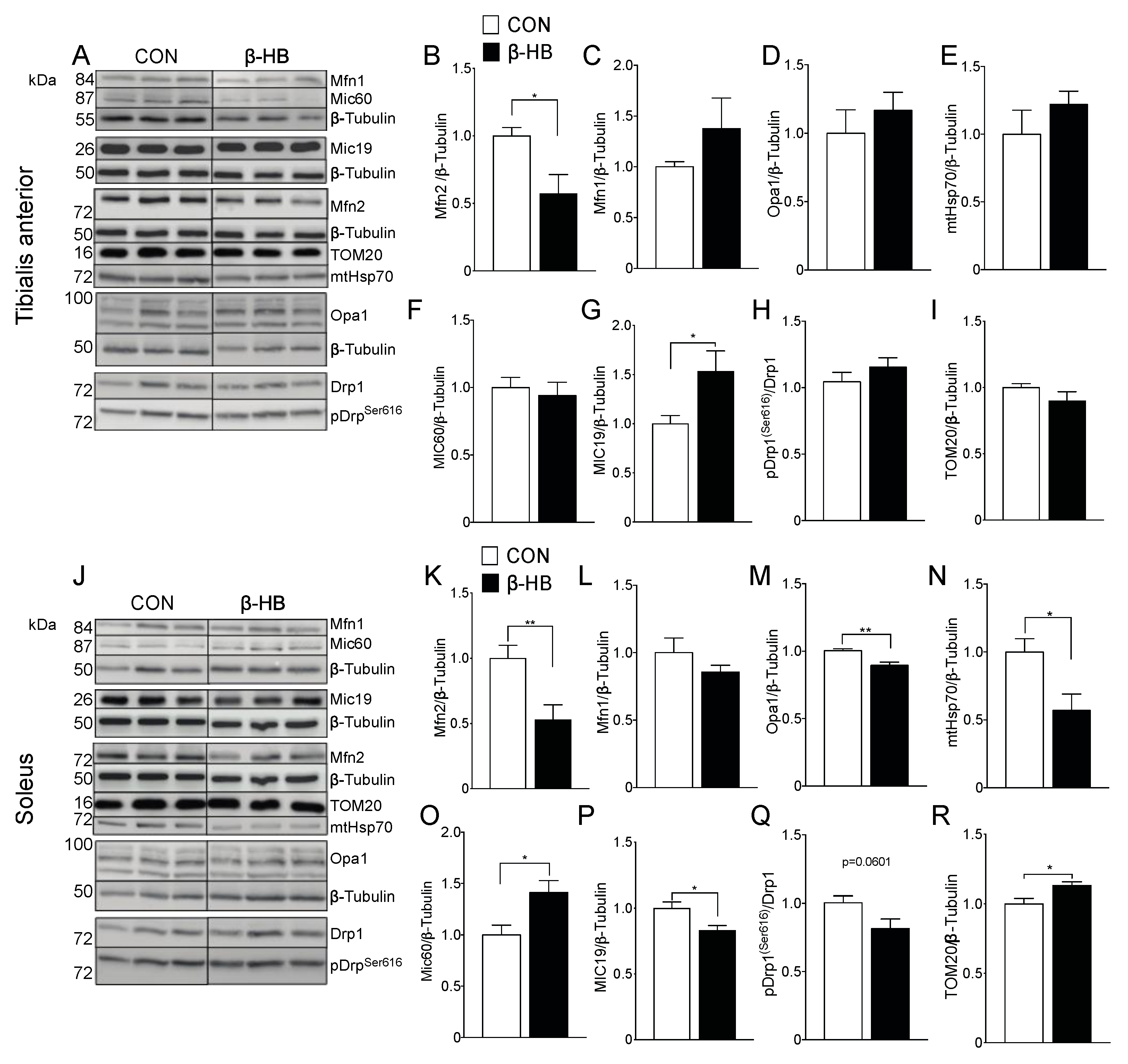
3.3. β-HB Long-Term Supplementation Induces a Mild Change in Mitochondrial Dynamics Proteins
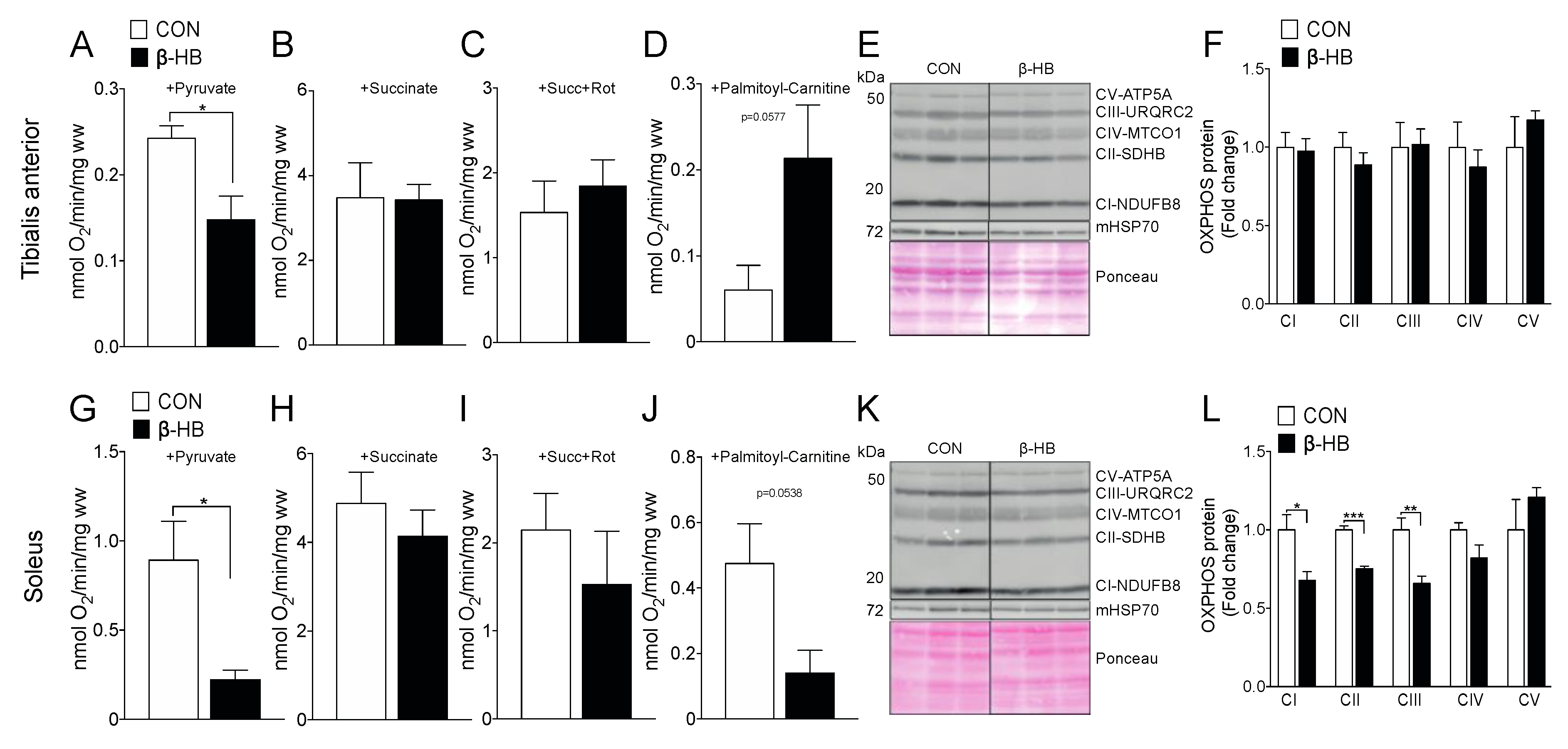
3.4. β-HB Short-Term Supplementation Modifies Mitochondrial Function and Substrate Utilization
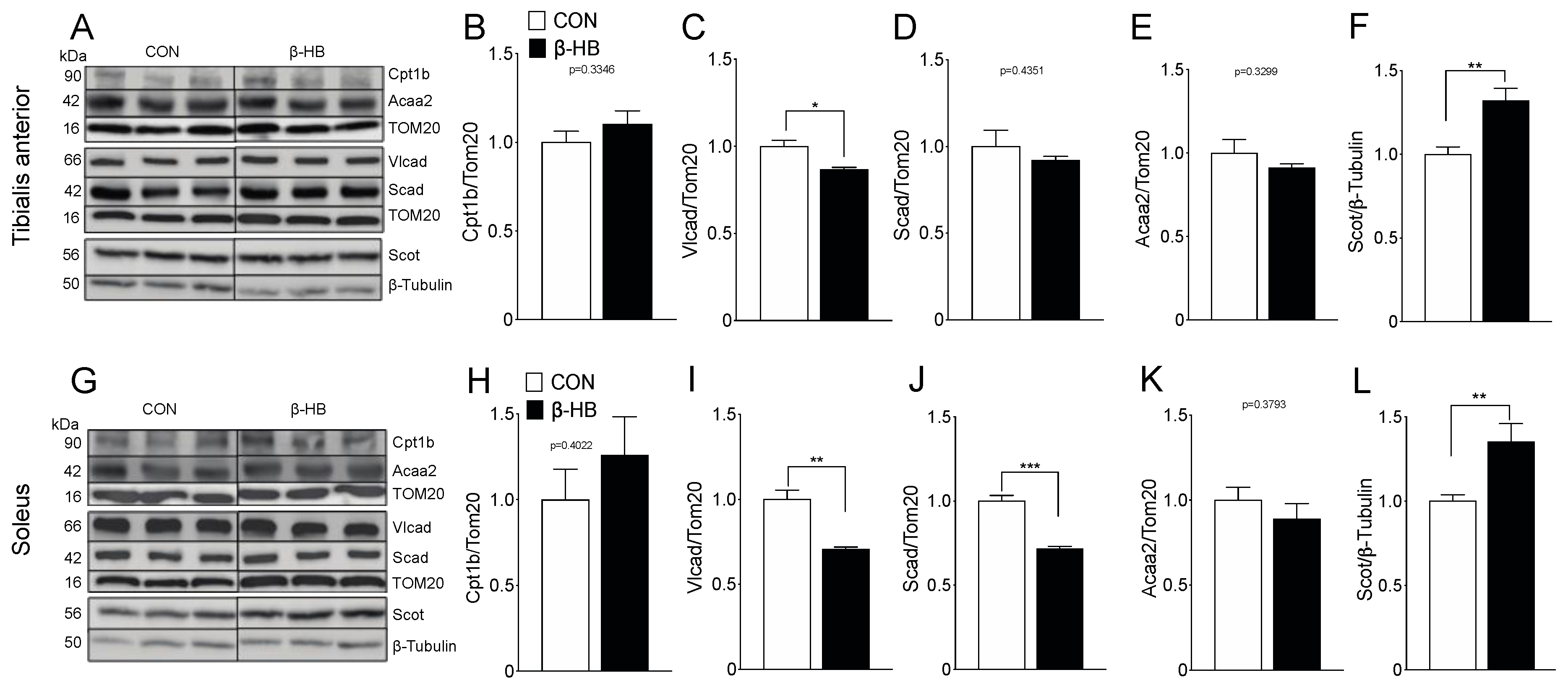
3.5. Lipid Metabolism and Ketone Oxidation Protein Content Are Altered After Short-Term β-HB Supplementation
3.6. β-HB Short-Term Supplementation Alters Mitochondrial Dynamic Fusion Protein Content
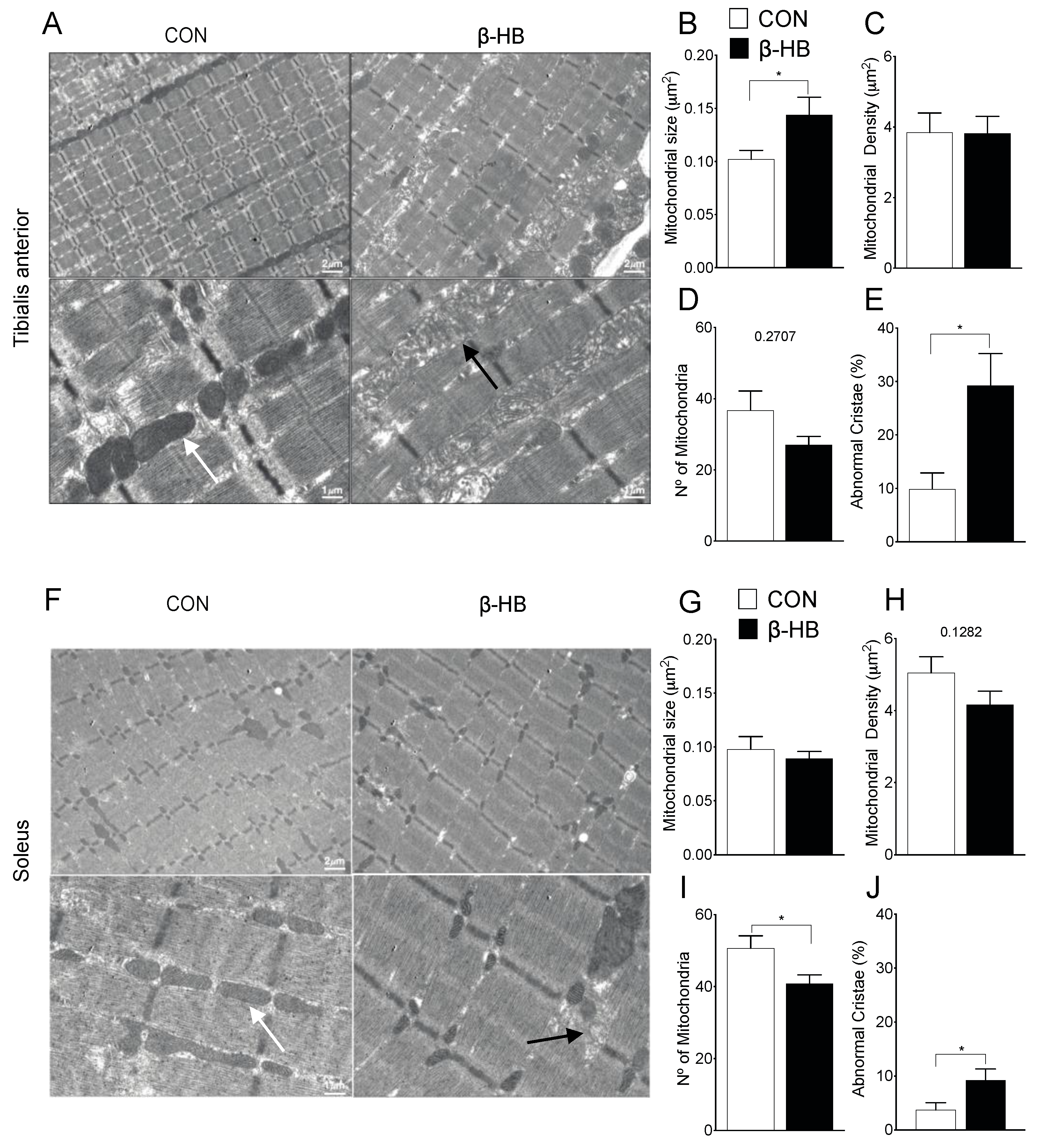
3.7. β-HB Short-Term Supplementation Disrupts Mitochondrial Morphology of SkM with Different Phenotypes
4. Discussion
4.1. Sustained Supplementation of β-HB Increases Physical Performance without Affecting Body Weight
4.2. Short-Term β-HB Supplementation Reduces Pyruvate Oxidation
4.3. β-HB Supplementation Modifies Mitochondrial Morphology
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Puchalska, P.; Crawford, P.A. Multi-dimensional Roles of Ketone Bodies in Fuel Metabolism, Signaling, and Therapeutics. Cell Metab. 2017, 25, 262–284. [Google Scholar] [CrossRef] [PubMed]
- VanItallie, T.B.; Nufert, T.H. Ketones: Metabolism’s ugly duckling. Nutr. Rev. 2003, 61, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Laffel, L. Ketone bodies: A review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes/Metab. Res. Rev. 1999, 15, 412–426. [Google Scholar] [CrossRef]
- Newman, J.C.; Verdin, E. β-Hydroxybutyrate: A Signaling Metabolite. Annu. Rev. Nutr. 2017, 37, 51–76. [Google Scholar] [CrossRef]
- Nagao, M.; Toh, R.; Irino, Y.; Mori, T.; Nakajima, H.; Hara, T.; Honjo, T.; Satomi-Kobayashi, S.; Shinke, T.; Tanaka, H.; et al. β-Hydroxybutyrate elevation as a compensatory response against oxidative stress in cardiomyocytes. Biochem. Biophys. Res. Commun. 2016, 475, 322–328. [Google Scholar] [CrossRef]
- Kong, G.; Huang, Z.; Ji, W.; Wang, X.; Liu, J.; Wu, X.; Huang, Z.; Li, R.; Zhu, Q. The ketone metabolite β-Hydroxybutyrate attenuates oxidative stress in spinal cord injury by suppression of class I histone deacetylases. J. Neurotrauma 2017, 34, 2645–2655. [Google Scholar] [CrossRef]
- Hirschey, M.D.; Newman, J.; He, W.; Shirakawa, K.; Le Moan, N.; Grueter, C.A.; Lim, H.; Saunders, L.R.; Stevens, R.D.; Newgard, C.B.; et al. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013, 339, 211–214. [Google Scholar] [CrossRef]
- Yamanashi, T.; Iwata, M.; Kamiya, N.; Tsunetomi, K.; Kajitani, N.; Wada, N.; Iitsuka, T.; Yamauchi, T.; Miura, A.; Pu, S.; et al. Beta-hydroxybutyrate, an endogenic NLRP3 inflammasome inhibitor, attenuates stress-induced behavioral and inflammatory responses. Sci. Rep. 2017, 7, 7677. [Google Scholar] [CrossRef]
- Cavaleri, F.; Bashar, E. Potential synergies of β-hydroxybutyrate and butyrate on the modulation of metabolism, inflammation, cognition, and general health. J. Nutr. Metab. 2018, 2018, 7195760. [Google Scholar] [CrossRef]
- Goldberg, E.L.; Asher, J.L.; Molony, R.D.; Shaw, A.C.; Zeiss, C.J.; Wang, C.; Morozova-Roche, L.A.; Herzog, R.I.; Iwasaki, A.; Dixit, V.D. β-Hydroxybutyrate deactivates neutrophil NLRP3 inflammasome to relieve gout flares. Cell Rep. 2017, 18, 2077–2087. [Google Scholar] [CrossRef]
- Thomsen, H.H.; Rittig, N.; Johannsen, M.; Møller, A.B.; Jørgensen, J.O.; Jessen, N.; Møller, N. Effects of 3-hydroxybutyrate and free fatty acids on muscle protein kinetics and signaling during LPS-induced inflammation in humans: Anticatabolic impact of ketone bodies. Am. J. Clin. Nutr. 2018, 108, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, R.; Moller, N.; Botker, H.E.; Eiskjaer, H.; Pryds, K.; Mellemkjaer, S.; Hansson, N.H.; Harms, H.J.; Gormsen, L.C.; Froekiaer, J.; et al. P3193The ketone body 3-hydroxybutyrate increases cardiac output without affecting myocardial external efficiency. Eur. Heart J. 2018, 39, ehy563-P3193. [Google Scholar] [CrossRef]
- Klos, M.; Morgenstern, S.; Hicks, K.; Suresh, S.; Devaney, E.J. The effects of the ketone body β-hydroxybutyrate on isolated rat ventricular myocyte excitation-contraction coupling. Arch. Biochem. Biophys. 2019, 662, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Gormsen, L.C.; Svart, M.; Thomsen, H.H.; Søndergaard, E.; Vendelbo, M.H.; Christensen, N.; Tolbod, L.P.; Harms, H.J.; Nielsen, R.; Wiggers, H.; et al. Ketone Body Infusion With 3-Hydroxybutyrate Reduces Myocardial Glucose Uptake and Increases Blood Flow in Humans: A Positron Emission Tomography Study. J. Am. Heart Assoc. 2017, 6, e005066. [Google Scholar] [CrossRef]
- Fritzen, A.M.; Lundsgaard, A.-M.; Kiens, B. Dietary Fuels in Athletic Performance. Annu. Rev. Nutr. 2019, 39, 45–73. [Google Scholar] [CrossRef]
- Evans, M.; Cogan, K.E.; Egan, B. Metabolism of ketone bodies during exercise and training: Physiological basis for exogenous supplementation. J. Physiol. 2017, 595, 2857–2871. [Google Scholar] [CrossRef]
- Sansone, M.; Sansone, A.; Borrione, P.; Romanelli, F.; Di Luigi, L.; Sgrò, P. Effects of Ketone Bodies on Endurance Exercise. Curr. Sports Med. Rep. 2018, 17, 444–453. [Google Scholar] [CrossRef]
- De Oliveira Caminhotto, R.; Komino, A.C.M.; de Fatima Silva, F.; Andreotti, S.; Sertié, R.A.L.; Reis, G.B.; Lima, F.B. Oral β-hydroxybutyrate increases ketonemia, decreases visceral adipocyte volume and improves serum lipid profile in Wistar rats. Nutr. Metab. 2017, 14, 31. [Google Scholar] [CrossRef]
- Kesl, S.L.; Poff, A.M.; Ward, N.P.; Fiorelli, T.N.; Ari, C.; Van Putten, A.J.; Sherwood, J.W.; Arnold, P.; D’Agostino, D.P. Effects of exogenous ketone supplementation on blood ketone, glucose, triglyceride, and lipoprotein levels in Sprague–Dawley rats. Nutr. Metab. 2016, 13, 9. [Google Scholar] [CrossRef]
- Kephart, W.; Holland, M.; Mumford, P.; Mobley, B.; Lowery, R.; Roberts, M.; Wilson, J. The Effects of Intermittent Ketogenic Dieting as well as Ketone Salt supplementation on Body Composition and Circulating Health Biomarkers in Exercising Rodents. FASEB J. 2016, 30, lb383. [Google Scholar] [CrossRef]
- Kephart, W.C.; Mumford, P.W.; Mao, X.; Romero, M.A.; Hyatt, H.W.; Zhang, Y.; Mobley, C.B.; Quindry, J.C.; Young, K.C.; Beck, D.T.; et al. The 1-Week and 8-Month Effects of a Ketogenic Diet or Ketone Salt Supplementation on Multi-Organ Markers of Oxidative Stress and Mitochondrial Function in Rats. Nutrients 2017, 9, 1019. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.J.; Knight, N.S.; Cole, M.A.; Cochlin, L.E.; Carter, E.; Tchabanenko, K.; Pichulik, T.; Gulston, M.K.; Atherton, H.J.; Schroeder, M.A.; et al. Novel ketone diet enhances physical and cognitive performance. FASEB J. 2016, 30, 4021–4032. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.J.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.J.; Stubbs, B.; West, J.; McLure, S.W.; et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metab. 2016, 24, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.J.; Clarke, K. Acute nutritional ketosis: Implications for exercise performance and metabolism. Extrem. Physiol. Med. 2014, 3, 17. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, T.; Myette-Cote, E.; Durrer, C.; Little, J.P. Nutritional ketone salts increase fat oxidation but impair high-intensity exercise performance in healthy adult males. Appl. Physiol. Nutr. Metab. 2017, 42, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Winder, W.W.; Baldwin, K.M.; Holloszy, J.O. Enzymes involved in ketone utilization in different types of muscle: Adaptation to exercise. Eur. J. Biochem. 1974, 47, 461–467. [Google Scholar] [CrossRef]
- Winder, W.W.; Baldwin, K.M.; Holloszy, J.O. Exercise-induced increase in the capacity of rat skeletal muscle to oxidize ketones. Can. J. Physiol. Pharmacol. 1975, 53, 86–91. [Google Scholar] [CrossRef]
- Winder, W.W.; Baldwin, K.M.; Holloszy, J.O. Exercise-induced adaptive increase in rate of oxidation of beta-hydroxybutyrate by skeletal muscle. Proc. Soc. Exp. Biol. Med. 1973, 143, 753–755. [Google Scholar] [CrossRef]
- Glancy, B.; Hartnell, L.M.; Malide, D.; Yu, Z.-X.; Combs, C.A.; Connelly, P.S.; Subramaniam, S.; Balaban, R.S. Mitochondrial reticulum for cellular energy distribution in muscle. Nature 2015, 523, 617–620. [Google Scholar] [CrossRef]
- Liesa, M.; Shirihai, O.S. Mitochondrial Dynamics in the Regulation of Nutrient Utilization and Energy Expenditure. Cell Metab. 2013, 17, 491–506. [Google Scholar] [CrossRef]
- Twig, G.; Elorza, A.; Molina, A.J.A.; Mohamed, H.; Wikstrom, J.D.; Walzer, G.; Stiles, L.; Haigh, S.E.; Katz, S.; Las, G.; et al. Fission and selective fusion govern mitochondrial segregation and elimination by autophagy. EMBO J. 2008, 27, 433–446. [Google Scholar] [CrossRef] [PubMed]
- Rambold, A.S.; Kostelecky, B.; Elia, N.; Lippincott-Schwartz, J. Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc. Natl. Acad. Sci. USA 2011, 108, 10190–10195. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, E.; Griparic, L.; Shurland, D.-L.; van der Bliek, A.M. Dynamin-related protein Drp1 Is required for mitochondrial division in mammalian Cells. MBoC 2001, 12, 2245–2256. [Google Scholar] [CrossRef] [PubMed]
- Gomes, L.C.; Di Benedetto, G.; Scorrano, L. During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat. Cell Biol. 2011, 13, 589–598. [Google Scholar] [CrossRef]
- Eisner, V.; Lenaers, G.; Hajnóczky, G. Mitochondrial fusion is frequent in skeletal muscle and supports excitation–contraction coupling. J. Cell Biol. 2014, 205, 179–195. [Google Scholar] [CrossRef]
- Mishra, P.; Chan, D.C. Metabolic regulation of mitochondrial dynamics. J. Cell Biol. 2016, 212, 379–387. [Google Scholar] [CrossRef]
- Mishra, P.; Varuzhanyan, G.; Pham, A.H.; Chan, D.C. Mitochondrial dynamics is a distinguishing feature of skeletal muscle fiber types and regulates organellar compartmentalization. Cell Metab. 2015, 22, 1033–1044. [Google Scholar] [CrossRef]
- Putti, R.; Sica, R.; Migliaccio, V.; Lionetti, L. Diet impact on mitochondrial bioenergetics and dynamics. Front. Physiol. 2015, 6, 109. [Google Scholar] [CrossRef]
- Diaz-Vegas, A.; Sanchez-Aguilera, P.; Krycer, J.R.; Morales, P.E.; Monsalves-Alvarez, M.; Cifuentes, M.; Rothermel, B.A.; Lavandero, S. Is Mitochondrial Dysfunction a Common Root of Noncommunicable Chronic Diseases? Endocr. Rev. 2020, 41, bnaa005. [Google Scholar] [CrossRef]
- Evans, M.; Patchett, E.; Nally, R.; Kearns, R.; Larney, M.; Egan, B. Effect of acute ingestion of β-hydroxybutyrate salts on the response to graded exercise in trained cyclists. Eur. J. Sport Sci. 2018, 18, 376–386. [Google Scholar] [CrossRef]
- Svensson, M.; Rosvall, P.; Boza-Serrano, A.; Andersson, E.; Lexell, J.; Deierborg, T. Forced treadmill exercise can induce stress and increase neuronal damage in a mouse model of global cerebral ischemia. Neurobiol. Stress 2016, 5, 8–18. [Google Scholar] [CrossRef] [PubMed]
- La Fuente, F.P.; Quezada, L.; Sepúlveda, C.; Monsalves-Alvarez, M.; Rodríguez, J.M.; Sacristán, C.; Chiong, M.; Llanos, M.; Espinosa, A.; Troncoso, R. Exercise regulates lipid droplet dynamics in normal and fatty liver. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2019, 1864, 158519. [Google Scholar] [CrossRef] [PubMed]
- Massett, M.P.; Berk, B.C. Strain-dependent differences in responses to exercise training in inbred and hybrid mice. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2005, 288, R1006–R1013. [Google Scholar] [CrossRef]
- McCosh, R.B.; Kreisman, M.J.; Breen, K.M. Frequent Tail-tip Blood Sampling in Mice for the Assessment of Pulsatile Luteinizing Hormone Secretion. J. Vis. Exp. 2018, e57894. [Google Scholar] [CrossRef] [PubMed]
- Bartsakoulia, M.; Pyle, A.; Troncoso-Chandía, D.; Vial-Brizzi, J.; Paz-Fiblas, M.V.; Duff, J.; Griffin, H.; Boczonadi, V.; Lochmüller, H.; Kleinle, S.; et al. A novel mechanism causing imbalance of mitochondrial fusion and fission in human myopathies. Hum. Mol. Genet. 2018, 27, 1186–1195. [Google Scholar] [CrossRef]
- Del Campo, A.; Contreras-Hernández, I.; Castro-Sepúlveda, M.; Campos, C.A.; Figueroa, R.; Tevy, M.F.; Eisner, V.; Casas, M.; Jaimovich, E. Muscle function decline and mitochondria changes in middle age precede sarcopenia in mice. Aging (Albany NY) 2018, 10, 34–55. [Google Scholar] [CrossRef]
- Pereira, R.O.; Tadinada, S.M.; Zasadny, F.M.; Oliveira, K.J.; Pires, K.M.P.; Olvera, A.; Jeffers, J.; Souvenir, R.; Mcglauflin, R.; Seei, A.; et al. OPA1 deficiency promotes secretion of FGF21 from muscle that prevents obesity and insulin resistance. EMBO J. 2017, 36, 2126–2145. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3, research0034.1–research0034.11. [Google Scholar] [CrossRef]
- Kammoun, M.; Cassar-Malek, I.; Meunier, B.; Picard, B. A Simplified Immunohistochemical Classification of Skeletal Muscle Fibres in Mouse. Eur. J. Histochem. 2014, 58, 2254. [Google Scholar] [CrossRef]
- Zierath, J.R.; Hawley, J.A. Skeletal Muscle Fiber Type: Influence on Contractile and Metabolic Properties. PLoS Biol. 2004, 2, e348. [Google Scholar] [CrossRef] [PubMed]
- Gram, M.; Vigelsø, A.; Yokota, T.; Hansen, C.N.; Helge, J.W.; Hey-Mogensen, M.; Dela, F. Two weeks of one-leg immobilization decreases skeletal muscle respiratory capacity equally in young and elderly men. Exp. Gerontol. 2014, 58, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Adhihetty, P.J.; Taivassalo, T.; Haller, R.G.; Walkinshaw, D.R.; Hood, D.A. The effect of training on the expression of mitochondrial biogenesis- and apoptosis-related proteins in skeletal muscle of patients with mtDNA defects. Am. J. Physiol.-Endocrinol. Metab. 2007, 293, E672–E680. [Google Scholar] [CrossRef]
- Cogliati, S.; Frezza, C.; Soriano, M.E.; Varanita, T.; Quintana-Cabrera, R.; Corrado, M.; Cipolat, S.; Costa, V.; Casarin, A.; Gomes, L.C.; et al. Mitochondrial cristae shape determines respiratory chain supercomplexes assembly and respiratory efficiency. Cell 2013, 155, 160–171. [Google Scholar] [CrossRef]
- Arase, K.; Fisler, J.S.; Shargill, N.S.; York, D.A.; Bray, G.A. Intracerebroventricular infusions of 3-OHB and insulin in a rat model of dietary obesity. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 1988, 255, R974–R981. [Google Scholar] [CrossRef]
- Ari, C.; Kovács, Z.; Juhasz, G.; Murdun, C.; Goldhagen, C.R.; Koutnik, A.P.; Poff, A.M.; Kesl, S.L.; D’Agostino, D.P. Exogenous Ketone Supplements Reduce Anxiety-Related Behavior in Sprague-Dawley and Wistar Albino Glaxo/Rijswijk Rats. Front. Mol. Neurosci. 2017, 9, 137. [Google Scholar] [CrossRef]
- Poff, A.; Kesl, S.; Koutnik, A.; Ward, N.; Ari, C.; Deblasi, J.; D’Agostino, D. Characterizing the metabolic effects of exogenous ketone supplementation—An alternative or adjuvant to the ketogenic diet. FASEB J. 2017, 31, 970.7. [Google Scholar] [CrossRef]
- Kovács, Z.; D’Agostino, D.P.; Dobolyi, A.; Ari, C. Adenosine A1 Receptor Antagonism Abolished the Anti-seizure Effects of Exogenous Ketone Supplementation in Wistar Albino Glaxo Rijswijk Rats. Front. Mol. Neurosci. 2017, 10, 235. [Google Scholar] [CrossRef]
- Stubbs, B.J.; Cox, P.J.; Evans, R.D.; Santer, P.; Miller, J.J.; Faull, O.K.; Magor-Elliott, S.; Hiyama, S.; Stirling, M.; Clarke, K. On the metabolism of exogenous ketones in humans. Front. Physiol. 2017, 8, 848. [Google Scholar] [CrossRef]
- Scofield, R.F.; Brady, P.S.; Schumann, W.C.; Kumaran, K.; Ohgaku, S.; Margolis, J.M.; Landau, B.R. On the lack of formation of l-(+)-3-hydroxybutyrate by liver. Arch. Biochem. Biophys. 1982, 214, 268–272. [Google Scholar] [CrossRef]
- Tsai, Y.-C.; Chou, Y.-C.; Wu, A.-B.; Hu, C.-M.; Chen, C.-Y.; Chen, F.-A.; Lee, J.-A. Stereoselective effects of 3-hydroxybutyrate on glucose utilization of rat cardiomyocytes. Life Sci. 2006, 78, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Haces, M.L.; Hernández-Fonseca, K.; Medina-Campos, O.N.; Montiel, T.; Pedraza-Chaverri, J.; Massieu, L. Antioxidant capacity contributes to protection of ketone bodies against oxidative damage induced during hypoglycemic conditions. Exp. Neurol. 2008, 211, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.C.; Covarrubias, A.J.; Zhao, M.; Yu, X.; Gut, P.; Ng, C.-P.; Huang, Y.; Haldar, S.; Verdin, E. Ketogenic diet reduces midlife mortality and improves memory in aging mice. Cell Metab. 2017, 26, 547–557.e8. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.N.; Wallace, M.A.; Tomilov, A.A.; Zhou, Z.; Marcotte, G.R.; Tran, D.; Perez, G.; Gutierrez-Casado, E.; Koike, S.; Knotts, T.A.; et al. A ketogenic diet extends longevity and healthspan in adult mice. Cell Metab. 2017, 26, 539–546.e5. [Google Scholar] [CrossRef]
- Egan, B.; D’Agostino, D.P. Fueling Performance: Ketones enter the Mix. Cell Metab. 2016, 24, 373–375. [Google Scholar] [CrossRef]
- Ferreira, J.C.; Rolim, N.P.; Bartholomeu, J.B.; Gobatto, C.A.; Kokubun, E.; Brum, P.C. Maximal Lactate Steady State in Running Mice: Effect of Exercise Training. Clin. Exp. Pharmacol. Physiol. 2007, 34, 760–765. [Google Scholar] [CrossRef]
- Ma, S.; Huang, Q.; Tominaga, T.; Liu, C.; Suzuki, K. An 8-Week Ketogenic Diet Alternated Interleukin-6, Ketolytic and Lipolytic Gene Expression, and Enhanced Exercise Capacity in Mice. Nutrients 2018, 10, 1696. [Google Scholar] [CrossRef]
- Diekman, E.F.; Ferdinandusse, S.; van der Pol, L.; Waterham, H.R.; Ruiter, J.P.N.; Ijlst, L.; Wanders, R.J.; Houten, S.M.; Wijburg, F.A.; Blank, A.C.; et al. Fatty acid oxidation flux predicts the clinical severity of VLCAD deficiency. Genet. Med. 2015, 17, 989–994. [Google Scholar] [CrossRef]
- Waisbren, S.E.; Levy, H.L.; Noble, M.; Matern, D.; Gregersen, N.; Pasley, K.; Marsden, D. Short-chain acyl-CoA dehydrogenase (SCAD) deficiency: An examination of the medical and neurodevelopmental characteristics of 14 cases identified through newborn screening or clinical symptoms. Mol. Genet. Metab. 2008, 95, 39–45. [Google Scholar] [CrossRef]
- Sebastián, D.; Palacín, M.; Zorzano, A. Mitochondrial Dynamics: Coupling Mitochondrial Fitness with Healthy Aging. Trends Mol. Med. 2017, 23, 201–215. [Google Scholar] [CrossRef]
- Izuta, Y.; Imada, T.; Hisamura, R.; Oonishi, E.; Nakamura, S.; Inagaki, E.; Ito, M.; Soga, T.; Tsubota, K. Ketone body 3-hydroxybutyrate mimics calorie restriction via the Nrf2 activator, fumarate, in the retina. Aging Cell 2018, 17, e12699. [Google Scholar] [CrossRef] [PubMed]
- Krebs, H.A. Rate control of the tricarboxylic acid cycle. Adv. Enzym. Regul. 1970, 8, 335–353. [Google Scholar] [CrossRef]
- Caffin, F.; Prola, A.; Piquereau, J.; Novotova, M.; David, D.; Garnier, A.; Fortin, D.; Alavi, M.; Veksler, V.; Ventura-Clapier, R.; et al. Altered skeletal muscle mitochondrial biogenesis but improved endurance capacity in trained OPA1-deficient mice. J. Physiol. 2013, 591, 6017–6037. [Google Scholar] [CrossRef]
- Milder, J.B.; Liang, L.-P.; Patel, M. Acute oxidative stress and systemic Nrf2 activation by the ketogenic diet. Neurobiol. Dis. 2010, 40, 238–244. [Google Scholar] [CrossRef]
- Anderson, E.J.; Yamazaki, H.; Neufer, P.D. Induction of Endogenous Uncoupling Protein 3 Suppresses Mitochondrial Oxidant Emission during Fatty Acid-supported Respiration. J. Biol. Chem. 2007, 282, 31257–31266. [Google Scholar] [CrossRef] [PubMed]
| Parameter | CON | β-HB | p-Value | |
|---|---|---|---|---|
| Body weight (g) | Basal | 22.5 ± 0.48 | 23.8 ± 0.40 | 0.073 |
| 2 weeks | 23 ± 0.61 | 23 ± 0.25 | 0.987 | |
| 4 weeks | 24 ± 0.60 | 24 ± 0.36 | 0.788 | |
| 6 weeks | 24 ± 0.76 | 24 ± 0.46 | 0.945 | |
| β-hydroxybutyrate (mM) | Basal | 0.4 ± 0.8 | 0.4 ± 0.04 | 0.765 |
| 2 weeks | 0.4 ± 0.04 | 0.7 ± 0.03 | 0.001 * | |
| 4 weeks | 0.4 ± 0.04 | 0.7 ± 0.08 | 0.056 | |
| 6 weeks | 0.5 ± 0.06 | 0.8 ± 0.09 | 0.037 * | |
| Calorie intake (kcal) | 2 weeks | 10.6 ± 0.41 | 9.2 ± 0.62 | 0.120 |
| 4 weeks | 11.1 ± 0.54 | 10.5 ± 0.75 | 0.532 | |
| 6 weeks | 11.2 ± 0.21 | 11.6 ± 0.29 | 0.379 | |
| Food intake (g) | 2 weeks | 2.7 ± 0.10 | 2.2 ± 0.15 | 0.020 * |
| 4 weeks | 2.8 ± 0.14 | 2.5 ± 0.19 | 0.266 | |
| 6 weeks | 2.9 ± 0.05 | 2.9 ± 0.06 | 0.832 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monsalves-Alvarez, M.; Morales, P.E.; Castro-Sepulveda, M.; Sepulveda, C.; Rodriguez, J.M.; Chiong, M.; Eisner, V.; Lavandero, S.; Troncoso, R. β-Hydroxybutyrate Increases Exercise Capacity Associated with Changes in Mitochondrial Function in Skeletal Muscle. Nutrients 2020, 12, 1930. https://doi.org/10.3390/nu12071930
Monsalves-Alvarez M, Morales PE, Castro-Sepulveda M, Sepulveda C, Rodriguez JM, Chiong M, Eisner V, Lavandero S, Troncoso R. β-Hydroxybutyrate Increases Exercise Capacity Associated with Changes in Mitochondrial Function in Skeletal Muscle. Nutrients. 2020; 12(7):1930. https://doi.org/10.3390/nu12071930
Chicago/Turabian StyleMonsalves-Alvarez, Matías, Pablo Esteban Morales, Mauricio Castro-Sepulveda, Carlos Sepulveda, Juan Manuel Rodriguez, Mario Chiong, Verónica Eisner, Sergio Lavandero, and Rodrigo Troncoso. 2020. "β-Hydroxybutyrate Increases Exercise Capacity Associated with Changes in Mitochondrial Function in Skeletal Muscle" Nutrients 12, no. 7: 1930. https://doi.org/10.3390/nu12071930
APA StyleMonsalves-Alvarez, M., Morales, P. E., Castro-Sepulveda, M., Sepulveda, C., Rodriguez, J. M., Chiong, M., Eisner, V., Lavandero, S., & Troncoso, R. (2020). β-Hydroxybutyrate Increases Exercise Capacity Associated with Changes in Mitochondrial Function in Skeletal Muscle. Nutrients, 12(7), 1930. https://doi.org/10.3390/nu12071930






