Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Samples
2.2. Animal Care and Handling
2.3. Bone Tissue Analysis
2.4. Cell Culture and Differentiation
2.5. Total RNA Extraction and Quantitative PCR (Q-PCR)
2.6. Statistical Analysis
3. Results
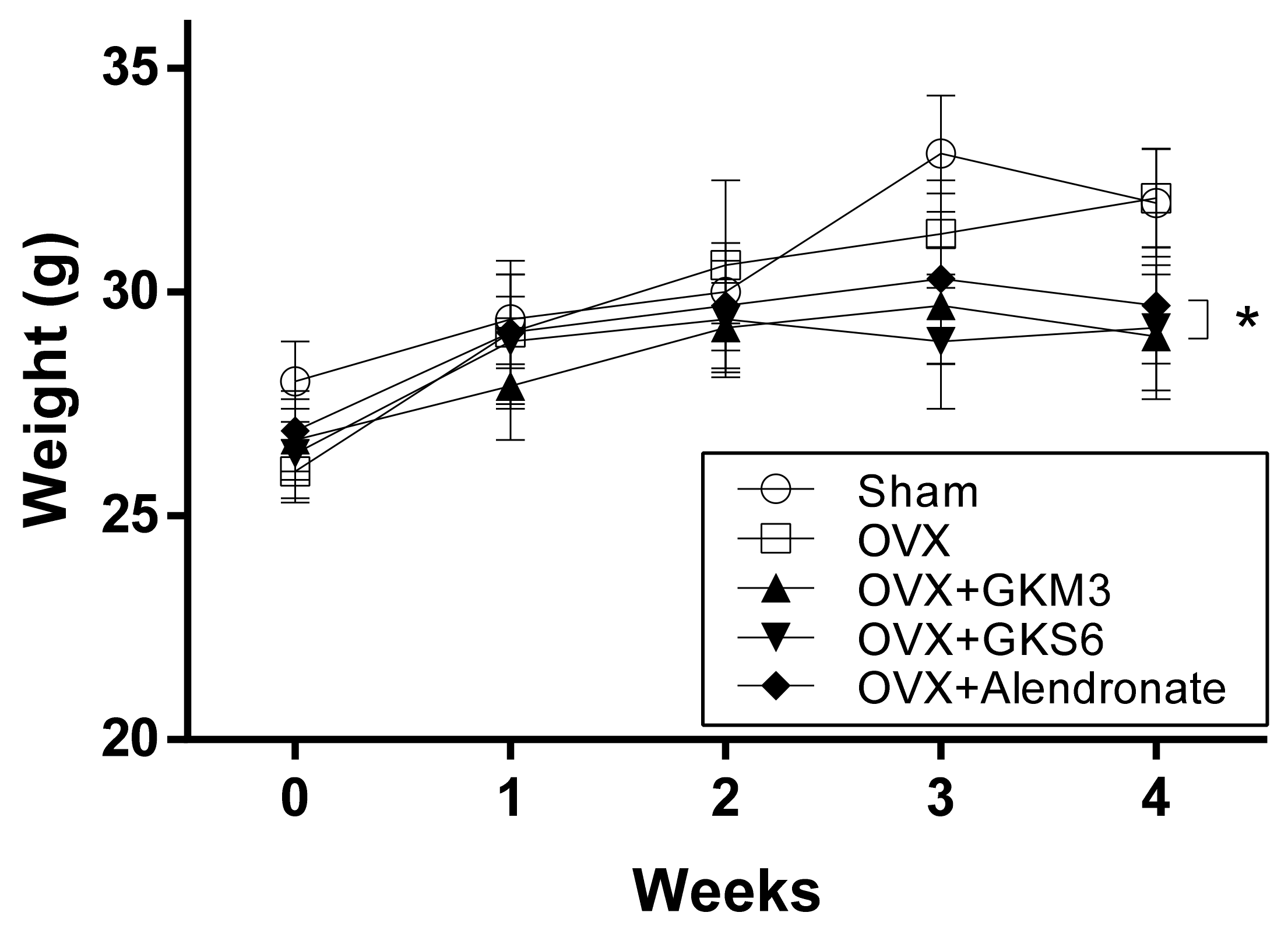
3.1. Effects of Probiotics on Body Weight of OVX Mice
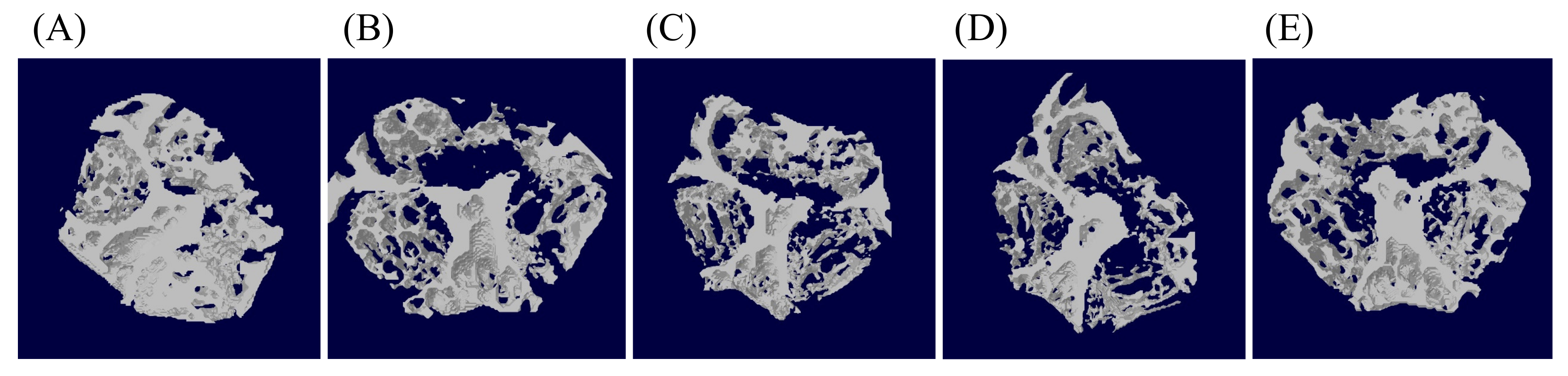
3.2. Effects of Probiotics on Bone Morphometric Parameters
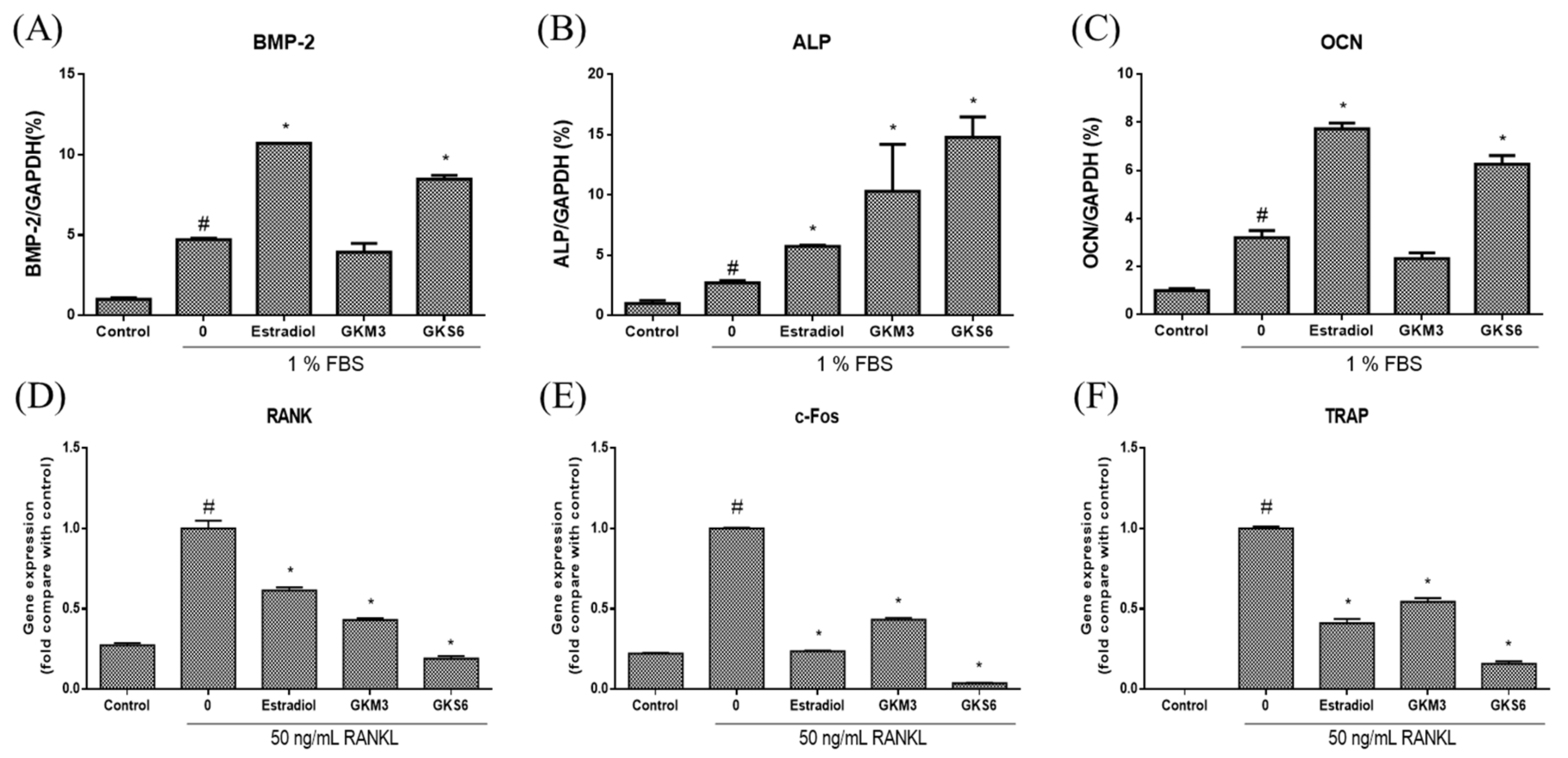
3.3. Effect of Probiotics on Bone Metabolism-Related Gene Expressions
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Boonen, S.; Dejaeger, E.; Vanderschueren, D.; Venken, K.; Bogaerts, A.; Verschueren, S.; Milisen, K. Osteoporosis and osteoporotic fracture occurrence and prevention in the elderly: A geriatric perspective. Best Pract. Res. Clin. Endocrinol. Metab. 2008, 22, 765–785. [Google Scholar] [CrossRef] [PubMed]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Nakahama, K. Cellular communications in bone homeostasis and repair. Cell. Mol. Life Sci. 2010, 67, 4001–4009. [Google Scholar] [CrossRef] [PubMed]
- Canalis, E.; Economides, A.N.; Gazzerro, E. Bone morphogenetic proteins, their antagonists, and the skeleton. Endocr. Rev. 2003, 24, 218–235. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, M.M.; Takayanagi, H. The immune system, bone and RANKL. Arch. Biochem. Biophys. 2014, 561, 118–123. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, N.K.; Lee, S.Y. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol. Cells 2017, 40, 706–713. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, N. Regulation of NFATc1 in Osteoclast Differentiation. J. Bone Metab. 2014, 21, 233–241. [Google Scholar] [CrossRef]
- McClung, M.; Clemmesen, B.; Daifotis, A.; Gilchrist, N.L.; Eisman, J.; Weinstein, R.S.; Fuleihan, G.E.-H.; Reda, C.; Yates, A.J.; Ravn, P. Alendronate prevents postmenopausal bone loss in women without osteoporosis. A double-blind, randomized, controlled trial. Alendronate Osteoporosis Prevention Study Group. Ann. Intern. Med. 1998, 128, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Gertz, B.J.; Holland, S.D.; Kline, W.F.; Matuszewski, B.K.; Porras, A.G. Clinical pharmacology of alendronate sodium. Osteoporos. Int. 1993, 3, S13–S16. [Google Scholar] [CrossRef] [PubMed]
- McClung, M.R.; Balske, A.; Burgio, D.E.; Wenderoth, D.; Recker, R.R. Treatment of postmenopausal osteoporosis with delayed-release risedronate 35 mg weekly for 2 years. Osteoporos. Int. 2013, 24, 301–310. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Delmas, P.D.; Ensrud, K.E.; Adachi, J.D.; Harper, K.D.; Sarkar, S.; Gennari, C.; Reginster, J.Y.; Pols, H.A.; Recker, R.R.; Harris, S.T.; et al. Efficacy of raloxifene on vertebral fracture risk reduction in postmenopausal women with osteoporosis: Four-year results from a randomized clinical trial. J. Clin. Endocrinol. Metab. 2002, 87, 3609–3617. [Google Scholar] [CrossRef] [PubMed]
- Yavropoulou, M.P.; Makras, P.; Anastasilakis, A.D. Bazedoxifene for the treatment of osteoporosis. Expert Opin. Pharmacother. 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- George, E.L.; Lin, Y.-L.; Saunders, M.M. Bisphosphonate-related osteonecrosis of the jaw: A mechanobiology perspective. Bone Rep. 2018, 8, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Lyytinen, H.; Pukkala, E.; Ylikorkala, O. Breast cancer risk in postmenopausal women using estrogen-only therapy. Obstet. Gynecol. 2006, 108, 1354–1360. [Google Scholar] [CrossRef]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, prebiotics and synbiotics—A review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef]
- Fuller, R. Probiotics in man and animals. J. Appl. Bacteriol. 1989, 66, 365–378. [Google Scholar]
- Ji, Y.; Park, S.; Chung, Y.; Kim, B.; Park, H.; Huang, E.; Jeong, D.; Jung, H.Y.; Kim, B.; Hyun, C.K.; et al. Amelioration of obesity-related biomarkers by Lactobacillus sakei CJLS03 in a high-fat diet-induced obese murine model. Sci. Rep. 2019, 9, 6821. [Google Scholar] [CrossRef]
- Zhao, X.; Yi, R.; Zhou, X.; Mu, J.; Long, X.; Pan, Y.; Song, J.L.; Park, K.Y. Preventive effect of Lactobacillus plantarum KSFY02 isolated from naturally fermented yogurt from Xinjiang, China, on d-galactose-induced oxidative aging in mice. J. Dairy Sci. 2019. [Google Scholar] [CrossRef]
- Fang, Y.; Chen, H.Q.; Zhang, X.; Zhang, H.; Xia, J.; Ding, K.; Fang, Z.Y. Probiotic administration of lactobacillus rhamnosus GR-1 attenuates atherosclerotic plaque formation in ApoE-/- mice fed with a high-fat diet. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 3533–3541. [Google Scholar] [CrossRef]
- Borchers, A.T.; Selmi, C.; Meyers, F.J.; Keen, C.L.; Gershwin, M.E. Probiotics and immunity. J. Gastroenterol. 2009, 44, 26–46. [Google Scholar] [CrossRef]
- Hsu, C.L.; Hou, Y.H.; Wang, C.S.; Lin, S.W.; Jhou, B.Y.; Chen, C.C.; Chen, Y.L. Antiobesity and Uric Acid-Lowering Effect of Lactobacillus plantarum GKM3 in High-Fat-Diet-Induced Obese Rats. J. Am. Coll. Nutr. 2019, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wright, L.E.; Christian, P.J.; Rivera, Z.; Van Alstine, W.G.; Funk, J.L.; Bouxsein, M.L.; Hoyer, P.B. Comparison of skeletal effects of ovariectomy versus chemically induced ovarian failure in mice. J. Bone Miner. Res. 2008, 23, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Sophocleous, A.; Idris, A.I. Rodent models of osteoporosis. Bonekey Rep. 2014, 3, 614. [Google Scholar] [CrossRef] [PubMed]
- Jee, W.S.; Yao, W. Overview: Animal models of osteopenia and osteoporosis. J. Musculoskelet. Neuronal Interact 2001, 1, 193–207. [Google Scholar] [PubMed]
- Montazeri-Najafabady, N.; Ghasemi, Y.; Dabbaghmanesh, M.H.; Talezadeh, P.; Koohpeyma, F.; Gholami, A. Supportive Role of Probiotic Strains in Protecting Rats from Ovariectomy-Induced Cortical Bone Loss. Probiotics Antimicrob. Proteins 2018. [Google Scholar] [CrossRef]
- Chiang, S.S.; Pan, T.M. Antiosteoporotic effects of Lactobacillus -fermented soy skim milk on bone mineral density and the microstructure of femoral bone in ovariectomized mice. J. Agric. Food Chem. 2011, 59, 7734–7742. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Jung, W.-K.; Kim, J.-A.; Choi, I.-W.; Kim, S.J. Beneficial effects of fucoidan on osteoblastic MG63 cell differentiation. Food Chem. 2009, 116, 990–994. [Google Scholar] [CrossRef]
- Collin-Osdoby, P.; Osdoby, P. RANKL-mediated osteoclast formation from murine RAW 264.7 cells. Methods Mol. Biol. (Clifton) 2012, 816, 187–202. [Google Scholar] [CrossRef]
- Collins, F.L.; Rios-Arce, N.D.; Schepper, J.D.; Parameswaran, N.; McCabe, L.R. The Potential of Probiotics as a Therapy for Osteoporosis. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Jones, R.M.; Mulle, J.G.; Pacifici, R. Osteomicrobiology: The influence of gut microbiota on bone in health and disease. Bone 2018, 115, 59–67. [Google Scholar] [CrossRef]
- Chiang, S.S.; Liao, J.W.; Pan, T.M. Effect of bioactive compounds in lactobacilli-fermented soy skim milk on femoral bone microstructure of aging mice. J. Sci. Food Agric. 2012, 92, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Witte, M.M.; Resuehr, D.; Chandler, A.R.; Mehle, A.K.; Overton, J.M. Female mice and rats exhibit species-specific metabolic and behavioral responses to ovariectomy. Gen. Comp. Endocrinol. 2010, 166, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Pang, W.Y.; Wang, X.L.; Mok, S.K.; Lai, W.P.; Chow, H.K.; Leung, P.C.; Yao, X.S.; Wong, M.S. Naringin improves bone properties in ovariectomized mice and exerts oestrogen-like activities in rat osteoblast-like (UMR-106) cells. Br. J. Pharmacol. 2010, 159, 1693–1703. [Google Scholar] [CrossRef] [PubMed]
- Ostadmohammadi, V.; Jamilian, M.; Bahmani, F.; Asemi, Z. Vitamin D and probiotic co-supplementation affects mental health, hormonal, inflammatory and oxidative stress parameters in women with polycystic ovary syndrome. J. Ovarian Res. 2019, 12, 5. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson, C.; Engdahl, C.; Fak, F.; Andersson, A.; Windahl, S.H.; Farman, H.H.; Moverare-Skrtic, S.; Islander, U.; Sjogren, K. Probiotics protect mice from ovariectomy-induced cortical bone loss. PLoS ONE 2014, 9, e92368. [Google Scholar] [CrossRef]
- Ogasawara, T.; Kawaguchi, H.; Jinno, S.; Hoshi, K.; Itaka, K.; Takato, T.; Nakamura, K.; Okayama, H. Bone Morphogenetic Protein 2-Induced Osteoblast Differentiation Requires Smad-Mediated Down-Regulation of Cdk6. Mol. Cell. Biol. 2004, 24, 6560. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, N. Signaling Pathways in Osteoclast Differentiation. Chonnam Med. J. 2016, 52, 12–17. [Google Scholar] [CrossRef]
- Ji, Y.; Chung, Y.M.; Park, S.; Jeong, D.; Kim, B.; Holzapfel, W.H. Dose-dependent and strain-dependent anti-obesity effects of Lactobacillus sakei in a diet induced obese murine model. PeerJ 2019, 7, e6651. [Google Scholar] [CrossRef]
| Gene | Forward | Reverse |
|---|---|---|
| BMP-2 | 5′−GGGTTGGAACTCCAGACTGT−3′ | 5′−GAAGAGTGAGTGGACCCCAG−3′ |
| ALP | 5′−CCACGTCTTCACATTTGGTG−3′ | 5′−AGACTGCGCCTAGTAGTTGT−3′ |
| OCN | 5′−TGAGAGCTCTCACACTCCTCGCCCTATTGG−3′ | 5′−GCTCCCAGCCATCGATACAGGTAGCGC−3′ |
| RANK | 5′−AAACCTTGGACCAACTGCAC−3′ | 5′−ACCATCTTCTCCTCCCHAGT−3′ |
| c-Fos | 5′−ATGGGCTCTCCTGTCAACAC−3′ | 5′−GGCTGCCAAAATAAACTCCA−3′ |
| TRAP | 5′−ACTTCCCCAGCCCTTACTACCG−3′ | 5′−TCAGCACATAGCCCACACCG−3′ |
| GAPDH | 5′−ACTTTGTCAAGCTCATTTCC−3′ | 5′−TGCAGCGAACTTTATTGATG−3′ |
| BMP-2: bone morphogenetic protein-2; ALP: alkaline phosphatase; OCN: osteocalcin; RANK: receptor activator of nuclear factor-κB; TRAP: tartrate-resistant acid phosphatase | ||
| Treatments | Dose (mg/Kg) | BV/TV (%) | Tb.Th | Tb.N | Tb.Sp | BMD |
|---|---|---|---|---|---|---|
| (μm) | (No./mm) | (μm) | (g/cm3) | |||
| Sham | - | 40.9 ± 1.6 d | 112.6 ± 2.6 d | 3.58 ± 0.17 b | 282.2 ± 56.0 ab | 0.67 ± 0.05 d |
| OVX | - | 32.0 ± 2.1 a | 100.1 ± 6.6 a | 3.11 ± 0.16 a | 379.2 ± 51.8 c | 0.51 ± 0.03 a |
| +GKM3 | 20.5 | 32.5 ± 2.3 ab | 104.6 ± 3.5 abc | 3.14 ± 0.13 a | 337.7 ± 44.7 ab | 0.55 ± 0.05 ab |
| +GKS6 | 20.5 | 32.9 ± 2.7 abc | 106.9 ± 5.2 bcd | 3.11 ± 0.12 a | 288.4 ± 37.6 ab | 0.58 ± 0.02 bc |
| +Alen | 2.5 | 34.0 ± 2.1 abc | 106.9 ± 5.6 bcd | 3.20 ± 0.14 a | 255.1 ± 41.3 a | 0.59 ± 0.03 bc |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.-C.; Lin, S.-W.; Li, I.-C.; Chen, Y.-P.; Tzu, S.-Y.; Chou, W.; Chen, C.-C.; Lin, W.-C.; Chen, Y.-L.; Lin, W.-H. Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation. Nutrients 2020, 12, 1914. https://doi.org/10.3390/nu12071914
Yang L-C, Lin S-W, Li I-C, Chen Y-P, Tzu S-Y, Chou W, Chen C-C, Lin W-C, Chen Y-L, Lin W-H. Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation. Nutrients. 2020; 12(7):1914. https://doi.org/10.3390/nu12071914
Chicago/Turabian StyleYang, Li-Chan, Shih-Wei Lin, I-Chen Li, Yen-Po Chen, Shih-Yang Tzu, Wei Chou, Chin-Chu Chen, Wen-Chuan Lin, Yen-Lien Chen, and Wen-Hsin Lin. 2020. "Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation" Nutrients 12, no. 7: 1914. https://doi.org/10.3390/nu12071914
APA StyleYang, L.-C., Lin, S.-W., Li, I.-C., Chen, Y.-P., Tzu, S.-Y., Chou, W., Chen, C.-C., Lin, W.-C., Chen, Y.-L., & Lin, W.-H. (2020). Lactobacillus plantarum GKM3 and Lactobacillus paracasei GKS6 Supplementation Ameliorates Bone Loss in Ovariectomized Mice by Promoting Osteoblast Differentiation and Inhibiting Osteoclast Formation. Nutrients, 12(7), 1914. https://doi.org/10.3390/nu12071914





