The Effects of Tocotrienol-Rich Vitamin E (Tocovid) on Diabetic Neuropathy: A Phase II Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Randomization and Masking
2.3. Procedures
2.4. Outcomes
2.5. Nerve Conduction Study
2.6. Serum NGF, MDA, TNFR-1, VCAM-1 and TXB2 Levels
2.7. Renal Profile, Lipid Profile and Liver Function Tests
2.8. HbA1c Assessment
2.9. Adherence
2.10. Statistical Analysis
3. Results
3.1. Validation of Nerve Conduction Study Methodology
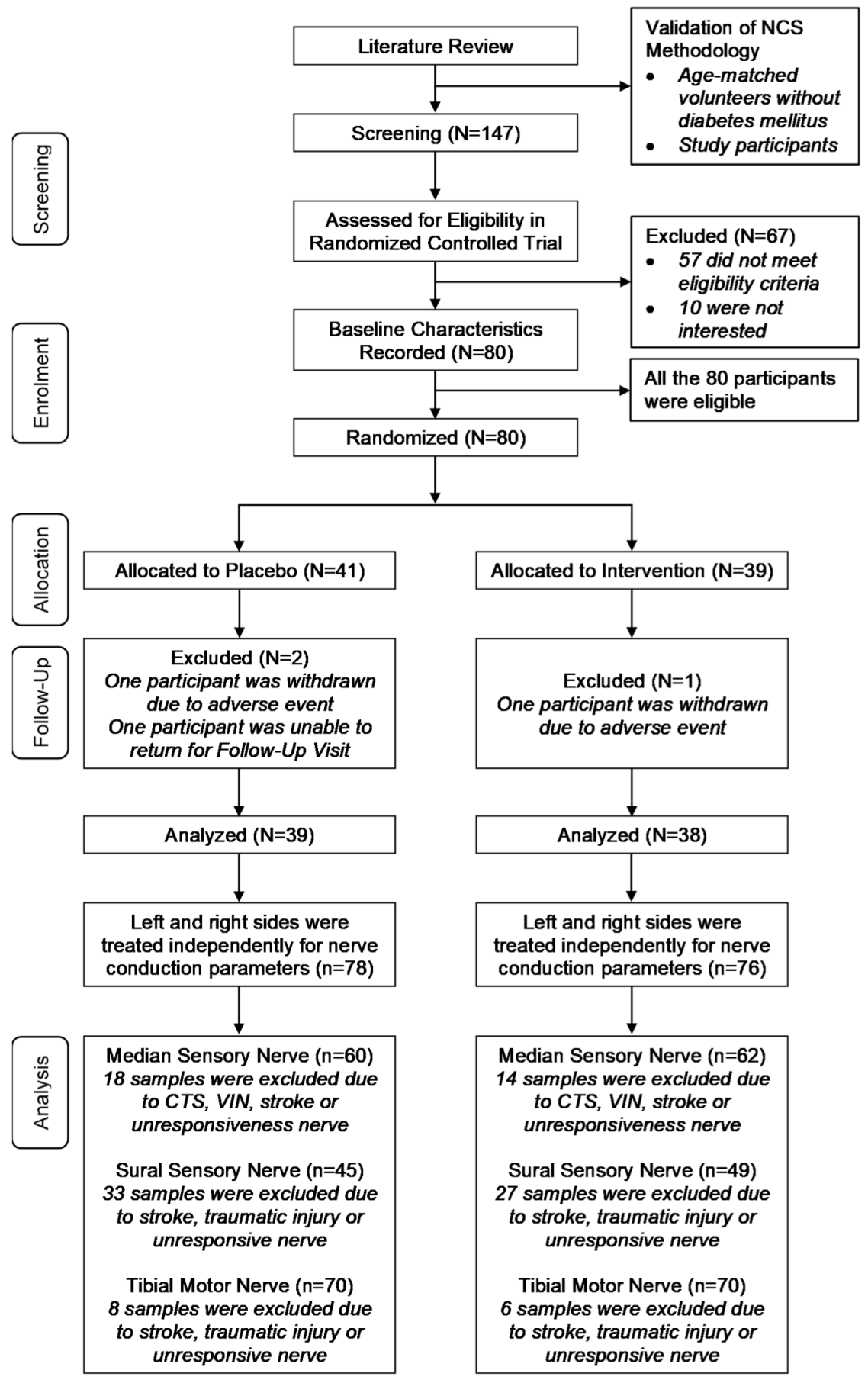
3.2. Randomized Controlled Trial
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tesfaye, S.; Boulton, A.J.; Dyck, P.J.; Freeman, R.; Horowitz, M.; Kempler, P. Diabetic neuropathies: Update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care 2010, 33, 2285–2293. [Google Scholar] [CrossRef] [PubMed]
- Pop-Busui, R.; Boulton, A.J.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A. Diabetic neuropathy: A position statement by the American Diabetes Association. Diabetes Care 2017, 40, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Sloan, G.; Shillo, P.; Selvarajah, D.; Wu, J.; Wilkinson, I.D.; Tracey, I. A new look at painful diabetic neuropathy. Diabetes Res. Clin. Pract. 2018, 144, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Volmer-Thole, M.; Lobmann, R. Neuropathy and Diabetic Foot Syndrome. Int. J. Mol. Sci. 2016, 17, 917. [Google Scholar] [CrossRef]
- Internal Clinical Guidelines T. National Institute for Health and Care Excellence: Clinical Guidelines. In Diabetic Foot Problems: Prevention and Management; National Institute for Health and Care Excellence: London, UK, 2015. [Google Scholar]
- Dewanjee, S.; Das, S.; Das, A.K.; Bhattacharjee, N.; Dihingia, A.; Dua, T.K. Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets. Eur. J. Pharmacol. 2018, 833, 472–523. [Google Scholar] [CrossRef]
- Cohen, K.; Shinkazh, N.; Frank, J.; Israel, I.; Fellner, C. Pharmacological treatment of diabetic peripheral neuropathy. Pharm. Ther. 2015, 40, 372. [Google Scholar]
- Boulton, A.J.; Kempler, P.; Ametov, A.; Ziegler, D. Whither pathogenetic treatments for diabetic polyneuropathy? Diabetes Metab. Res. Rev. 2013, 29, 327–333. [Google Scholar] [CrossRef]
- Peh, H.Y.; Tan, W.S.; Liao, W.; Wong, W.S. Vitamin E therapy beyond cancer: Tocopherol versus tocotrienol. Pharmacol. Ther. 2016, 162, 152–169. [Google Scholar] [CrossRef]
- Rajanandh, M.G.; Kosey, S.; Prathiksha, G. Assessment of antioxidant supplementation on the neuropathic pain score and quality of life in diabetic neuropathy patients—a randomized controlled study. Pharmacol. Rep. PR 2014, 66, 44–48. [Google Scholar] [CrossRef]
- Tutuncu, N.B.; Bayraktar, M.; Varli, K. Reversal of defective nerve conduction with vitamin E supplementation in type 2 diabetes: A preliminary study. Diabetes Care 1998, 21, 1915–1918. [Google Scholar] [CrossRef]
- Ogbera, A.O.; Ezeobi, E.; Unachukwu, C.; Oshinaike, O. Treatment of diabetes mellitus-associated neuropathy with vitamin E and Eve primrose. Indian J. Endocrinol. Metab. 2014, 18, 846–849. [Google Scholar] [CrossRef] [PubMed]
- Hor, C.P.; Fung, W.Y.; Ang, H.A.; Lim, S.C.; Kam, L.Y.; Sim, S.W. Efficacy of Oral Mixed Tocotrienols in Diabetic Peripheral Neuropathy: A Randomized Clinical Trial. JAMA Neurol. 2018, 75, 444–452. [Google Scholar] [PubMed]
- Won, J.C.; Park, T.S. Recent Advances in Diagnostic Strategies for Diabetic Peripheral Neuropathy. Endocrinol. Metab. 2016, 31, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Kakouros, N.; Rade, J.J.; Kourliouros, A.; Resar, J.R. Platelet function in patients with diabetes mellitus: From a theoretical to a practical perspective. Int. J. Endocrinol. 2011, 2011, 742719. [Google Scholar] [CrossRef]
- Vinik, A.I.; Erbas, T.; Park, T.S.; Nolan, R.; Pittenger, G.L. Platelet dysfunction in type 2 diabetes. Diabetes Care 2001, 24, 1476–1485. [Google Scholar] [CrossRef]
- American Diabetes Association. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2019. Diabetes Care 2019, 42, S13–S28. [Google Scholar] [CrossRef]
- Chen, S.; Andary, M.; Buschbacher, R.; Del Toro, D.; Smith, B.; So, Y. Electrodiagnostic reference values for upper and lower limb nerve conduction studies in adult populations. Muscle Nerve 2016, 54, 371–377. [Google Scholar] [CrossRef]
- Ferrante, M.A.; Teresa Spiegelberg, B.; Tsao, B.E. Principles of Nerve Conduction Studis and Needle EMG; American Association of Neuromuscular & Electrodiagnostic Medicine: Rochester, NY, USA, 2014; 35 p. [Google Scholar]
- Che, H.-L.; Tan, D.M.-Y.; Meganathan, P.; Gan, Y.-L.; Abdul Razak, G.; Fu, J.-Y. Validation of a HPLC/FLD method for quantification of tocotrienols in human plasma. Int. J. Anal. Chem. 2015, 2015, 357609. [Google Scholar] [CrossRef]
- Compadre, C.M.; Singh, A.; Thakkar, S.; Zheng, G.; Breen, P.J.; Ghosh, S. Molecular dynamics guided design of tocoflexol: A new radioprotectant tocotrienol with enhanced bioavailability. Drug Dev. Res. 2014, 75, 10–22. [Google Scholar] [CrossRef]
- Liu, Z.; Lee, H.-J.; Garofalo, F.; Jenkins, D.J.; El-Sohemy, A. Simultaneous measurement of three tocopherols, all-trans-retinol, and eight carotenoids in human plasma by isocratic liquid chromatography. J. Chromatogr. Sci. 2011, 49, 221–227. [Google Scholar] [CrossRef]
- Jordan, P.; Brubacher, D.; Moser, U.; Stahelin, H.B.; Gey, K.F. Vitamin E and vitamin A concentrations in plasma adjusted for cholesterol and triglycerides by multiple regression. Clin. Chem. 1995, 41, 924–927. [Google Scholar] [CrossRef]
- Dunnigan, S.K.; Ebadi, H.; Breiner, A.; Katzberg, H.D.; Lovblom, L.E.; Perkins, B.A. Conduction slowing in diabetic sensorimotor polyneuropathy. Diabetes Care 2013, 36, 3684–3690. [Google Scholar] [CrossRef]
- Yoshida, Y.; Saito, Y.; Jones, L.S.; Shigeri, Y. Chemical reactivities and physical effects in comparison between tocopherols and tocotrienols: Physiological significance and prospects as antioxidants. J. Biosci. Bioeng. 2007, 104, 439–445. [Google Scholar] [CrossRef]
- Ghafoorunissa Hemalatha, S.; Rao, M.V. Sesame lignans enhance antioxidant activity of vitamin E in lipid peroxidation systems. Mol. Cell. Biochem. 2004, 262, 195–202. [Google Scholar] [CrossRef]
- Suzuki, Y.J.; Tsuchiya, M.; Wassall, S.R.; Choo, Y.M.; Govil, G.; Kagan, V.E. Structural and dynamic membrane properties of alpha-tocopherol and alpha-tocotrienol: Implication to the molecular mechanism of their antioxidant potency. Biochemistry 1993, 32, 10692–10699. [Google Scholar] [CrossRef]
- Serbinova, E.; Kagan, V.; Han, D.; Packer, L. Free radical recycling and intramembrane mobility in the antioxidant properties of alpha-tocopherol and alpha-tocotrienol. Free Radic. Biol. Med. 1991, 10, 263–275. [Google Scholar] [CrossRef]
- Mutalib, M.S.A.; Khaza’ai, H.; Wahle, K.W.J. Palm-tocotrienol rich fraction (TRF) is a more effective inhibitor of LDL oxidation and endothelial cell lipid peroxidation than α-tocopherol in vitro. Food Res. Int. 2003, 36, 405–413. [Google Scholar] [CrossRef]
- Kamat, J.P.; Devasagayam, T.P. Tocotrienols from palm oil as potent inhibitors of lipid peroxidation and protein oxidation in rat brain mitochondria. Neurosci. Lett. 1995, 195, 179–182. [Google Scholar] [CrossRef]
- Adachi, H.; Ishii, N. Effects of tocotrienols on life span and protein carbonylation in Caenorhabditis elegans. J. Gerontol. Series A Biol. Sci. Med Sci. 2000, 55, B280–B285. [Google Scholar] [CrossRef]
- Valls-Canals, J.; Povedano, M.; Montero, J.; Pradas, J. Diabetic polyneuropathy. Axonal or demyelinating? Electromyogr. Clin. Neurophysiol. 2002, 42, 3–6. [Google Scholar]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Rahimi-Madiseh, M.; Malekpour-Tehrani, A.; Bahmani, M.; Rafieian-Kopaei, M. The research and development on the antioxidants in prevention of diabetic complications. Asian Pac. J. Trop. Med. 2016, 9, 825–831. [Google Scholar] [CrossRef]
- Niki, E. Lipid peroxidation products as oxidative stress biomarkers. BioFactors 2008, 34, 171–180. [Google Scholar] [CrossRef]
- Cermenati, G.; Abbiati, F.; Cermenati, S.; Brioschi, E.; Volonterio, A.; Cavaletti, G. Diabetes-induced myelin abnormalities are associated with an altered lipid pattern: Protective effects of LXR activation. J. Lipid Res. 2012, 53, 300–310. [Google Scholar] [CrossRef]
- Kanakapura, G.V.; Bangalore, P.C.; Lakshmi, B.P. Antioxidant status in diabetic neuropathy. Trials 2017, 6, 8. [Google Scholar]
- Vincent, A.M.; Callaghan, B.C.; Smith, A.L.; Feldman, E.L. Diabetic neuropathy: Cellular mechanisms as therapeutic targets. Nat. Rev. Neurol. 2011, 7, 573–583. [Google Scholar] [CrossRef]
- Singh, R.; Kishore, L.; Kaur, N. Diabetic peripheral neuropathy: Current perspective and future directions. Pharmacol. Res. 2014, 80, 21–35. [Google Scholar] [CrossRef]
- Gohda, T.; Maruyama, S.; Kamei, N.; Yamaguchi, S.; Shibata, T.; Murakoshi, M. Circulating TNF Receptors 1 and 2 Predict Mortality in Patients with End-stage Renal Disease Undergoing Dialysis. Sci. Rep. 2017, 7, 43520. [Google Scholar] [CrossRef]
- Brownlee, M. The Pathobiology of Diabetic Complications. Unifying Mech. 2005, 54, 1615–1625. [Google Scholar]
- Pop-Busui, R.; Ang, L.; Holmes, C.; Gallagher, K.; Feldman, E.L. Inflammation as a Therapeutic Target for Diabetic Neuropathies. Curr. Diabetes Rep. 2016, 16, 29. [Google Scholar] [CrossRef]
- Bluher, M.; Unger, R.; Rassoul, F.; Richter, V.; Paschke, R. Relation between glycaemic control, hyperinsulinaemia and plasma concentrations of soluble adhesion molecules in patients with impaired glucose tolerance or Type II diabetes. Diabetologia 2002, 45, 210–216. [Google Scholar] [CrossRef]
- Sun, Q.; Tang, D.D.; Yin, E.G.; Wei, L.L.; Chen, P.; Deng, S.P. Diagnostic Significance of Serum Levels of Nerve Growth Factor and Brain Derived Neurotrophic Factor in Diabetic Peripheral Neuropathy. Med. Sci. Monit. 2018, 24, 5943–5950. [Google Scholar] [CrossRef]
| Nerve Conduction Parameter | Study Subjects | Normal Controls | p-Value |
|---|---|---|---|
| Median Sensory Nerve | n = 20 ‡ | n = 20 § | |
| Conduction Velocity (m/s) a | 42.84 ± 5.80 | 53.47 ± 4.51 | <0.001 * |
| Peak Velocity (m/s) a | 34.21 ± 4.02 | 41.65 ± 3.68 | <0.001 * |
| NP Amplitude (µV) b | 18.40 (8.15) | 31.20 (18.60) | <0.001 * |
| Sural Sensory Nerve | n = 24 † | n = 24 † | |
| Conduction Velocity (m/s) b | 42.75 (4.93) | 50.45 (4.50) | <0.001 * |
| Peak Velocity (m/s) a | 33.56 ± 2.90 | 39.18 ± 3.22 | <0.001 * |
| PP Amplitude (µV) a | 7.12 ± 4.87 | 13.65 ± 5.35 | <0.001 * |
| Tibial Motor Nerve | n = 24 † | n = 24 † | |
| Conduction Velocity (m/s) b | 41.90 (8.17) | 49.55 (3.90) | <0.001 * |
| Distal Amplitude at Ankle (mV) a | 7.65 ± 3.62 | 12.69 ± 3.50 | <0.001 * |
| Baseline Characteristic | Placebo (n = 41) | Tocovid (n = 39) | p-Value | Total (n = 80) |
|---|---|---|---|---|
| Gender, n (%) | 0.870 † | |||
| Male | 27 (65.9) | 25 (64.1) | 52 (65.0) | |
| Female | 14 (34.1) | 14 (35.9) | 28 (35.0) | |
| Race, n (%) | 0.808 ‡ | |||
| Malay | 19 (46.3) | 20 (51.3) | 39 (48.8) | |
| Chinese | 12 (29.3) | 10 (25.6) | 22 (27.5) | |
| Indian | 7 (17.1) | 8 (20.5) | 15 (18.8) | |
| Others | 3 (7.3) | 1 (2.6) | 4 (5.0) | |
| Age (years) b | 64.00 (15.00) | 63.00 (12.00) | 0.836 | 63.50 (14.00) |
| Duration of DM (years) b | 13.00 (11.00) | 14.00 (10.00) | 0.916 | 14.00 (10.00) |
| HbA1c (%) b | 7.40 (1.85) | 7.20 (1.70) | 0.509 | 7.40 (1.70) |
| SBP (mmHg) a | 129.39 ± 12.99 | 133.90 ± 12.76 | 0.122 | 131.59 ± 13.00 |
| DBP (mmHg) a | 77.90 ± 9.12 | 78.08 ± 8.83 | 0.931 | 77.99 ± 8.92 |
| BMI (kg/m2) a | 28.36 ± 5.07 | 27.99 ± 4.17 | 0.722 | 28.18 ± 4.63 |
| Serum Biomarkers | ||||
| NGF (ng/mL) b | 11.47 (4.11) | 11.11 (4.58) | 0.59 | 11.26 (3.62) |
| MDA (ng/mL) b | 881.03 (697.85) | 794.22 (726.90) | 0.881 | 878.54 (713.99) |
| VCAM-1 (ng/mL) b | 347.15 (311.90) | 412.47 (275.07) | 0.438 | 382.40 (274.14) |
| TNFR-1 (pg/mL) b | 66.87 (56.64) | 80.37 (63.71) | 0.22 | 71.54 (59.51) |
| TXB2 (pg/mL) b | 83.96 (73.01) | 90.11 (61.67) | 0.634 | 85.78 (67.90) |
| α-Tocopherol (µmol/L) | 40.46 ± 18.73 | 42.05 ± 25.72 | 0.753 | 41.23 ± 22.23 |
| Safety Tests | ||||
| eGFR a | 64.34 ± 21.92 | 67.23 ± 20.86 | 0.548 | 65.75 ± 21.33 |
| Urea b | 6.15 (3.63) | 5.65 (2.60) | 0.606 | 5.75 (3.30) |
| AST b | 20.00 (9.50) | 20.00 (11.00) | 0.928 | 20.00 (10.00) |
| ALT b | 21.00 (15.50) | 23.00 (17.50) | 0.976 | 21.00 (17.00) |
| TC a | 4.56 ± 0.98 | 4.91 ± 1.11 | 0.14 | 4.73 ± 1.05 |
| HDL-C b | 1.20 (0.48) | 1.30 (0.40) | 0.37 | 1.30 (0.40) |
| Median Sensory Nerve † | Placebo (n = 60) | Tocovid (n = 62) | p-Value |
| Conduction Velocity (m/s) | |||
| At Baseline b | 44.10 (9.07) | 43.45 (10.43) | 0.838 |
| At Eight Weeks a | 43.44 ± 5.94 | 46.29 ± 6.65 | 0.014 * |
| Change b | 0.00 (2.90) | 1.25 (3.35) | <0.001 * |
| Peak Velocity (m/s) | |||
| At Baseline a | 35.21 ± 4.86 | 35.45 ± 4.91 | 0.778 |
| At Eight Weeks b | 35.40 (5.65) | 35.40 (8.55) | 0.218 |
| Change b | −0.15 (2.33) | 0.95 (2.40) | <0.001 * |
| NP Amplitude (µV) | |||
| At Baseline b | 17.80 (11.93) | 20.55 (14.25) | 0.259 |
| At Eight Weeks b | 17.75 (13.65) | 22.35 (15.60) | 0.102 |
| Change a | 1.15 ± 4.93 | 1.70 ± 5.95 | 0.58 |
| PP Amplitude (µV) | |||
| At Baseline b | 28.95 (17.78) | 32.15 (19.70) | 0.251 |
| At Eight Weeks b | 29.25 (20.28) | 34.90 (21.78) | 0.032 * |
| Change b | 1.40 (8.30) | 3.15 (11.35) | 0.213 |
| Sural Sensory Nerve‡ | Placebo (n = 45) | Tocovid (n = 49) | p-Value |
| Conduction Velocity (m/s) | |||
| At Baseline a | 43.15 ± 5.53 | 43.53 ± 5.92 | 0.751 |
| At Eight Weeks a | 42.29 ± 4.51 | 45.32 ± 5.30 | 0.004 * |
| Change b | −0.60 (2.10) | 1.60 (1.80) | <0.001 * |
| Peak Velocity (m/s) | |||
| At Baseline a | 34.36 ± 3.73 | 34.32 ± 4.49 | 0.955 |
| At Eight Weeks b | 33.30 (3.70) | 35.00 (7.65) | 0.133 |
| Change a | −0.54 ± 1.62 | 1.14 ± 1.64 | <0.001 * |
| PP Amplitude (µV) | |||
| At Baseline b | 8.70 (8.05) | 7.00 (8.45) | 0.5 |
| At Eight Weeks b | 7.40 (8.60) | 7.70 (11.35) | 0.904 |
| Change a | 0.43 ± 2.99 | 1.11 ± 2.57 | 0.243 |
| NP Amplitude (µV) | |||
| At Baseline b | 10.10 (5.50) | 9.80 (9.40) | 0.498 |
| At Eight Weeks b | 8.50 (5.60) | 9.20 (9.25) | 0.31 |
| Change b | −0.40 (2.10) | 0.10 (4.95) | 0.47 |
| Tibial Motor Nerve § | Placebo (n = 70) | Tocovid (n = 70) | p-Value |
| Conduction Velocity (m/s) | |||
| At Baseline a | 40.17 ± 6.14 | 41.44 ± 5.69 | 0.206 |
| At Eight Weeksa | 39.02 ± 5.79 | 42.40 ± 4.89 | <0.001 * |
| Change b | −0.90 (3.50) | 0.75 (2.25) | <0.001 * |
| Distal Amplitude at Ankle (mV) | |||
| At Baseline a | 7.87 ± 4.78 | 8.32 ± 4.08 | 0.547 |
| At Eight Weeks a | 8.78 ± 5.07 | 9.44 ± 4.12 | 0.398 |
| Change b | 0.60 (1.83) | 0.80 (1.95) | 0.291 |
| Analyte | Placebo (n = 39) | Tocovid (n = 38) | p-Value |
|---|---|---|---|
| Serum Biomarkers | |||
| NGF (ng/mL) a | 10.02 ± 2.28 | 11.04 ± 2.05 | 0.047 * |
| MDA (ng/mL) b | 1158.37 (794.77) | 959.57 (903.02) | 0.551 |
| VCAM-1 (ng/mL) a | 257.40 ± 113.23 | 278.28 ± 129.36 | 0.453 |
| TNFR-1 (pg/mL) b | 23.24 (55.52) | 21.88 (45.43) | 0.874 |
| TXB2 (pg/mL) b | 162.57 (164.41) | 150.70 (132.24) | 0.418 |
| HbA1c (%) a | 7.81 ± 1.29 | 7.43 ± 1.11 | 0.174 |
| SBP (mmHg) a | 129.00 ± 13.95 | 133.16 ± 14.70 | 0.207 |
| DBP (mmHg) b | 75.00 (14.00) | 73.00 (16.00) | 0.386 |
| BMI (kg/m2) a | 27.81 ± 4.89 | 27.57 ± 4.29 | 0.823 |
| α-Tocopherol (µmol/L) | 37.90 ± 17.61 | 67.10 ± 38.59 | <0.001 * |
| Safety Tests | |||
| eGFR b | 62.00 (39.00) | 63.50 (42.00) | 0.303 |
| Urea a | 6.90 ± 2.61 | 6.63 ± 2.47 | 0.651 |
| AST b | 19.00 (8.00) | 18.50 (8.25) | 0.394 |
| ALT b | 20.00 (12.00) | 19.00 (14.00) | 0.298 |
| TC a | 4.43 ± 1.10 | 4.95 ± 1.03 | 0.036 * |
| HDL-C b | 1.20 (0.30) | 1.30 (0.50) | 0.569 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ng, Y.T.; Phang, S.C.W.; Tan, G.C.J.; Ng, E.Y.; Botross Henien, N.P.; M. Palanisamy, U.D.; Ahmad, B.; Abdul Kadir, K. The Effects of Tocotrienol-Rich Vitamin E (Tocovid) on Diabetic Neuropathy: A Phase II Randomized Controlled Trial. Nutrients 2020, 12, 1522. https://doi.org/10.3390/nu12051522
Ng YT, Phang SCW, Tan GCJ, Ng EY, Botross Henien NP, M. Palanisamy UD, Ahmad B, Abdul Kadir K. The Effects of Tocotrienol-Rich Vitamin E (Tocovid) on Diabetic Neuropathy: A Phase II Randomized Controlled Trial. Nutrients. 2020; 12(5):1522. https://doi.org/10.3390/nu12051522
Chicago/Turabian StyleNg, Yeek Tat, Sonia Chew Wen Phang, Gerald Chen Jie Tan, En Yng Ng, Nevein Philip Botross Henien, Uma Devi M. Palanisamy, Badariah Ahmad, and Khalid Abdul Kadir. 2020. "The Effects of Tocotrienol-Rich Vitamin E (Tocovid) on Diabetic Neuropathy: A Phase II Randomized Controlled Trial" Nutrients 12, no. 5: 1522. https://doi.org/10.3390/nu12051522
APA StyleNg, Y. T., Phang, S. C. W., Tan, G. C. J., Ng, E. Y., Botross Henien, N. P., M. Palanisamy, U. D., Ahmad, B., & Abdul Kadir, K. (2020). The Effects of Tocotrienol-Rich Vitamin E (Tocovid) on Diabetic Neuropathy: A Phase II Randomized Controlled Trial. Nutrients, 12(5), 1522. https://doi.org/10.3390/nu12051522






