A Dose-Dependent Effect of Carnipure® Tartrate Supplementation on Endurance Capacity, Recovery, and Body Composition in an Exercise Rat Model
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Experimental Design
2.3. Biochemical Analysis
2.4. Statistical Analysis
3. Results and Discussion
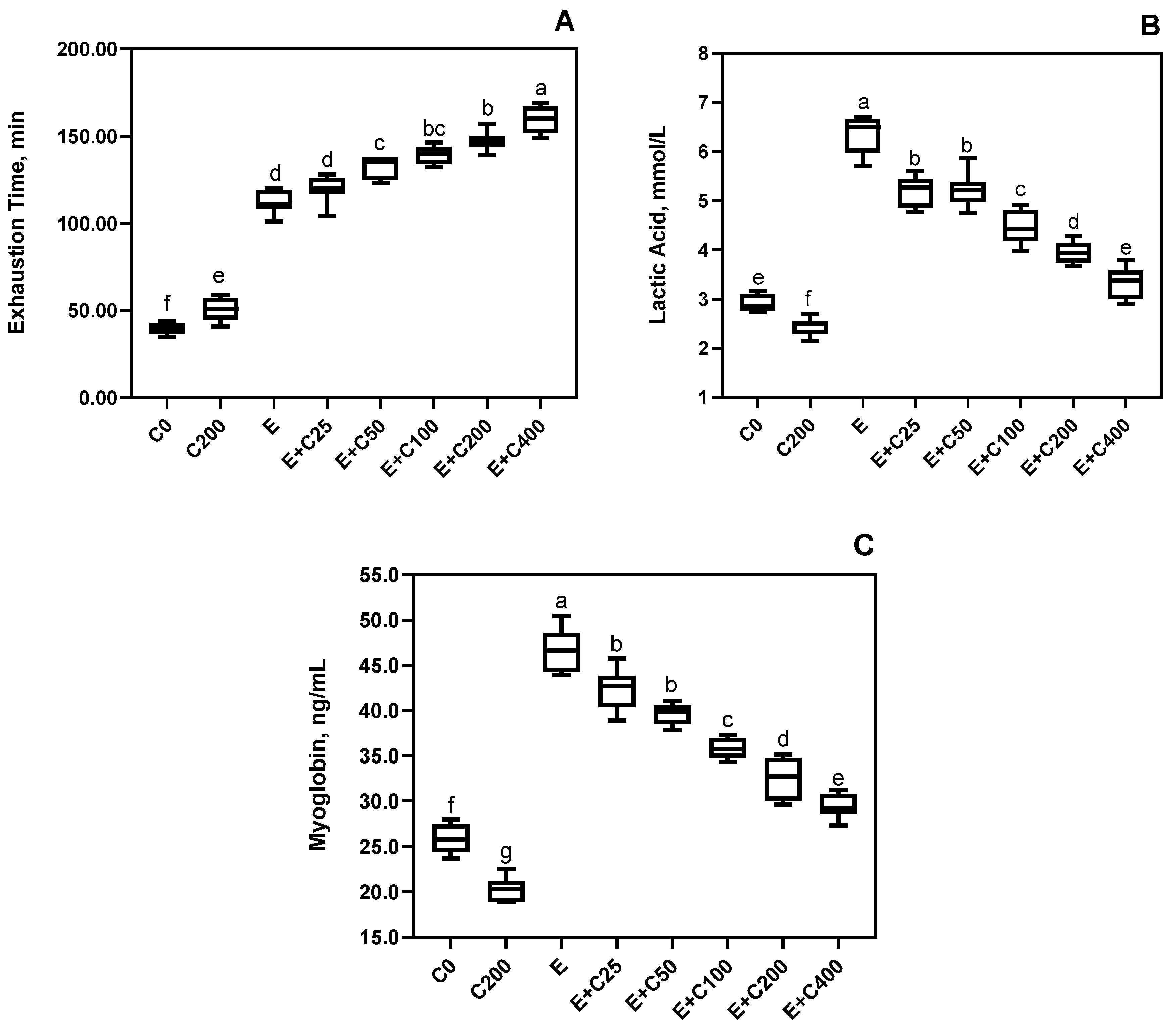
3.1. Effects on Exercise Performance, Recovery, and Fatigue
3.2. Effects of Carnipure® Tartrate Supplementation on Body Weight and Composition
3.3. Antioxidant Capacity
3.4. Effects on Glucose and Lipid Levels
3.5. L-Carnitine Plasma Levels
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CT | Carnipure® Tartrate |
| MDA | Malondialdehyde |
| SOD | Super-oxide dismutase |
| CAT | Catalase |
| GSHPx | Glutathione peroxidase |
| TCA | Tricarboxylic acid cycle |
| BMI | Body mass index |
| AST | Aspartate aminotransferase |
| ALT | Alanine aminotransferase |
| ROS | Reactive oxygen species |
References
- Elokda, A.S.; Nielsen, D.H. Effects of exercise training on the glutathione antioxidant system. Eur. J. Cardiovasc. Prev. Rehabil. 2007, 14, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Von Stengel, S.; Engelke, K.; Kalender, W.A. Exercise decreases the risk of metabolic syndrome in elderly females. Med. Sci. Sports Exerc. 2009, 41, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Rosety-Rodriguez, M.; Rosety, I.; Fornieles-Gonzalez, G.; Diaz-Ordonez, A.J.; Camacho, A.; Rosety, M.A.; Ordonez, F.J. A 6-week training program increased muscle antioxidant system in elderly diabetic fatty rats. Med. Sci. Monit. 2012, 18, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Silva, E.P., Jr.; Borges, L.S.; Mendes-da-Silva, C.; Hirabara, S.M.; Lambertucci, R.H. l-Arginine supplementation improves rats’ antioxidant system and exercise performance. Free Radic. Res. 2017, 51, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Orhan, C.; Tuzcu, M.; Sahin, N.; Erten, F.; Juturu, V. Capsaicinoids improve the consequences of physical activity. Toxicol. Rep. 2018, 5, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Munoz, V.R.; Gaspar, R.C.; Esteca, M.V.; Baptista, I.L.; Vieira, R.F.L.; Ramos da Silva, A.S.; de Moura, L.P.; Cintra, D.E.; Ropelle, E.R.; Pauli, J.R. Physical exercise increases ROCK activity in the skeletal muscle of middle-aged rats. Mech. Ageing Dev. 2020, 111213. [Google Scholar] [CrossRef]
- Pedralli, M.L.; Marschner, R.A.; Kollet, D.P.; Neto, S.G.; Eibel, B.; Tanaka, H.; Lehnen, A.M. Different exercise training modalities produce similar endothelial function improvements in individuals with prehypertension or hypertension: A randomized clinical trial Exercise, endothelium and blood pressure. Sci. Rep. 2020, 20, 7628. [Google Scholar] [CrossRef]
- Yang, D.K.; Lee, S.J.; Adam, G.O.; Kim, S.J. Aralia continentalis kitagawa Extract Attenuates the Fatigue Induced by Exhaustive Exercise through Inhibition of Oxidative Stress. Antioxidants 2020, 9, 379. [Google Scholar] [CrossRef]
- Kerling, A.; Hartung, D.; Stubbs, B.; Kück, M.; Tegtbur, U.; Grams, L. Impact of aerobic exercise on muscle mass in patients with major depressive disorder: A randomized controlled trial. Neuropsychiatr. Dis. Treat. 2018, 14, 1969–1974. [Google Scholar] [CrossRef]
- Lee, H.; Kim, K.; Kim, B.; Shin, J.; Rajan, S.; Wu, J.; Chen, X.; Brow, M.D.; Lee, S.; Park, J.Y. A cellular mechanism of muscle memory facilitates mitochondrial remodelling following resistance training. J. Physiol. 2018, 596, 4413–4426. [Google Scholar] [CrossRef]
- Irving, B.A.; Davis, C.K.; Brock, D.W.; Weltman, J.Y.; Swift, D.; Barrett, E.J.; Gaesser, G.A.; Weltman, A. Effect of exercise training intensity on abdominal visceral fat and body composition. Med. Sci. Sports Exer. 2008, 40, 1863–1872. [Google Scholar] [CrossRef] [PubMed]
- Nakagata, T.; Yamada, Y.; Naito, H. Estimating energy cost of body weight resistance exercise using a multistage exercise Test. J. Strength Cond. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tscholl, P.; Alonso, J.M.; Dollé, G.; Junge, A.; Dvorak, J. The use of drugs and nutritional supplements in top-level track and field athletes. Am. J. Sports Med. 2010, 38, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Fielding, R.; Riede, L.; Lugo, J.P.; Bellamine, A. L-Carnitine Supplementation in Recovery after Exercise. Nutrients 2018, 10, 349. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Randell, R. Fat burners: Nutrition supplements that increase fat metabolism. Obes. Rev. 2011, 12, 841–851. [Google Scholar] [CrossRef]
- Kim, J.H.; Pan, J.H.; Lee, E.S.; Kim, Y.J. L-Carnitine enhances exercise endurance capacity by promoting muscle oxidative metabolism in mice. Biochem. Biophys. Res. Commun. 2015, 464, 568–573. [Google Scholar] [CrossRef]
- Odo, S.; Tanabe, K.; Yohda, M.; Yamauchi, M. Liver-oriented acute metabolic effects of a low dose of l-carnitine under fat-mobilizing conditions: Pilot Human Clinical Trial. J. Nutr. Sci. Vitaminol. 2020, 66, 136–149. [Google Scholar] [CrossRef]
- Flanagan, J.L.; Simmons, P.A.; Vehige, J.; Willcox, M.D.; Garrett, Q. Role of carnitine in disease. Nutr. Metab. 2010, 7, 30. [Google Scholar] [CrossRef]
- Bremer, J. Carnitine—Metabolism and functions. Physiol. Rev. 1983, 63, 1420–1479. [Google Scholar] [CrossRef]
- Constantin-Teodosiu, D.; Carlin, J.I.; Cederblad, G.; Harris, R.C.; Hultman, E. Acetyl group accumulation and pyruvate dehydrogenase activity in human muscle during incremental exercise. Acta Physiol. Scand. 1991, 143, 367–372. [Google Scholar] [CrossRef]
- Timmons, J.A.; Poucher, S.M.; Constantin-Teodosiu, D.; Worrall, V.; Macdonald, I.A.; Greenhaff, P.L. Increased acetyl group availability enhances contractile function of canine skeletal muscle during ischemia. J. Clin. Investig. 1996, 97, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Calo, L.A.; Pagnin, E.; Davis, P.A.; Semplicini, A.; Nicolai, R.; Calvani, M.; Pessina, A.C. Antioxidant effect of L-carnitine and its short chain esters: Relevance for the protection from oxidative stress related cardiovascular damage. Int. J. Cardiol. 2006, 107, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant and antiradical activities of L-carnitine. Life Sci. 2006, 78, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Boyacioglu, M.; Turgut, H.; Akgullu, C.; Eryilmaz, U.; Kum, C.; Onbasili, O.A. The effect of L-carnitine on oxidative stress responses of experimental contrast-induced nephropathy in rats. J. Vet. Med. Sci. 2014, 76, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Meng, F.; Li, H.; Hua, X.; Wu, L.; Yuan, X. L-carnitine alleviates oxidative stress-related damage via MAPK signaling in human lens epithelial cells exposed to H2O2. Int. J. Mol. Med. 2019, 44, 1515–1522. [Google Scholar] [CrossRef]
- Marconi, C.; Sassi, G.; Carpinelli, A.; Cerretelli, P. Effects of l-carnitine loading on the aerobic and anaerobic performance of endurance athletes. Eur. J. Appl. Physiol. Occup. Physiol. 1985, 54, 131–135. [Google Scholar] [CrossRef]
- Colombani, P.; Wenk, C.; Kunz, I.; Krahenbuhl, S.; Kuhnt, M.; Arnold, M.; Frey-Rindova, P.; Frey, W.; Langhans, W. Effects of L-carnitine supplementation on physical performance and energy metabolism of endurance-trained athletes: A double-blind crossover field study. Eur. J. Appl. Physiol. Occup. Physiol. 1996, 73, 434–439. [Google Scholar] [CrossRef]
- Broad, E.M.; Maughan, R.J.; Galloway, S.D. Carbohydrate, protein, and fat metabolism during exercise after oral carnitine supplementation in humans. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 567–584. [Google Scholar] [CrossRef]
- Orer, G.E.; Guzel, N.A. The effects of acute L-carnitine supplementation on endurance performance of athletes. J. Strength Cond. Res. 2014, 28, 514–519. [Google Scholar] [CrossRef]
- Rivas-Estany, E.; Sixto-Fernandez, S.; Barrera-Sarduy, J.; Hernandez-Garcia, S.; Gonzalez-Guerra, R.; Stusser-Beltranena, R. Effects of long-term exercise training on left ventricular function and remodeling in patients with anterior wall myocardial infarction. Arch. Cardiol. Méx. 2013, 83, 167–173. [Google Scholar]
- Feng, R.; Wang, L.; Li, Z.; Yang, R.; Liang, Y.; Sun, Y.; Yu, Q.; Ghartey-Kwansah, G.; Sun, Y.; Wu, Y.; et al. A systematic comparison of exercise training protocols on animal models of cardiovascular capacity. Life Sci. 2019, 217, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Dragan, G.I.; Wagner, W.; Ploesteanu, E. Studies concerning the ergogenic value of protein supply and l-carnitine in elite junior cyclists. Physiologie 1988, 25, 129–132. [Google Scholar] [PubMed]
- Gorostiaga, E.M.; Maurer, C.A.; Eclache, J.P. Decrease in respiratory quotient during exercise following l-carnitine supplementation. Int. J. Sports Med. 1989, 10, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Soop, M.; Björkman, O.; Cederblad, G.; Hagenfeldt, L.; Wahren, J. Influence of carnitine supplementation on muscle substrate and carnitine metabolism during exercise. J. Appl. Physiol. 1988, 64, 2394–2399. [Google Scholar] [CrossRef] [PubMed]
- Trappe, S.W.; Costill, D.L.; Goodpaster, B.; Vukovich, M.D.; Fink, W.J. The effects of l-carnitine supplementation on performance during interval swimming. Int. J. Sports Med. 1994, 15, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Broad, E.M.; Maughan, R.J.; Galloway, S.D. Effects of four weeks l-carnitine L-tartrate ingestion on substrate utilization during prolonged exercise. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Harper, P.; Elwin, C.E.; Cederblad, G. Pharmacokinetics of intravenous and oral bolus doses of l-carnitine in healthy subjects. Eur. J. Clin. Pharmacol. 1988, 35, 555–562. [Google Scholar] [CrossRef]
- Evans, A.M.; Fornasini, G. Pharmacokinetics of L-carnitine. Clin. Pharmacokinet. 2003, 42, 941–967. [Google Scholar] [CrossRef]
- Brass, E.P.; Hoppel, C.L.; Hiatt, W.R. Effect of intravenous l-carnitine on carnitine homeostasis and fuel metabolism during exercise in humans. Clin. Pharmacol. Ther. 1994, 55, 681–692. [Google Scholar] [CrossRef]
- Van Hall, G. Lactate as a fuel for mitochondrial respiration. Acta Physiol. Scand. 2000, 168, 643–656. [Google Scholar] [CrossRef]
- Mallard, A.; Briskey, D.; Richards, A.; Mills, D.; Rao, A. The effect of orally dosed levagen+™ (palmitoylethanolamide) on exercise recovery in healthy males-A Double-Blind, Randomized, Placebo-Controlled Study. Nutrients 2020, 12, 596. [Google Scholar] [CrossRef]
- Brevetti, G.; Chiariello, M.; Ferulano, G.; Policicchio, A.; Nevola, E.; Rossini, A.; Angelini, C. Increases in walking distance in patients with peripheral vascular disease treated with l-carnitine: A double-blind, cross-over study. Circulation 1988, 77, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Siliprandi, N.; Di Lisa, F.; Menabó, R. Clinical use of carnitine. Past, present and future. Adv. Exp. Med. Biol. 1990, 272, 175–181. [Google Scholar] [PubMed]
- Giamberardino, M.A.; Dragani, L.; Valente, R.; Di Lisa, F.; Saggini, R.; Vecchiet, L. Effects of prolonged l-carnitine administration on delayed muscle pain and CK release after eccentric effort. Int. J. Sports Med. 1996, 17, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Müller, D.M.; Seim, H.; Kiess, W.; Löster, H.; Richter, T. Effects of oral l-carnitine supplementation on in vivo long-chain fatty acid oxidation in healthy adults. Metabolism 2002, 51, 1389–1391. [Google Scholar] [PubMed]
- Nuesch, R.; Rossetto, M.; Martina, B. Plasma and urine carnitine concentrations in well-trained athletes at rest and after exercise. Influence of l-carnitine intake. Drugs Exp. Clin. Res. 1999, 25, 167–171. [Google Scholar]
- Pedersen, B.K. Muscles and their myokines. J. Exp. Biol. 2011, 214, 337–346. [Google Scholar] [CrossRef]
- Naclerio, F.; Larumbe-Zabala, E.; Cooper, R.; Allgrove, J.; Earnest, C.P. A multi-ingredient containing carbohydrate, proteins L-glutamine and L-carnitine attenuates fatigue perception with no effect on performance, muscle damage or immunity in soccer players. PLoS ONE 2015, 10, e0125188. [Google Scholar] [CrossRef]
- Talenezhad, N.; Mohammadi, M.; Ramezani-Jolfaie, N.; Mozaffari-Khosravi, H.; Salehi-Abargouei, A. Effects of l-carnitine supplementation on weight loss and body composition: A systematic review and meta-analysis of 37 randomized controlled clinical trials with dose-response analysis. Clin. Nutr. ESPEN 2020, 37, 9–23. [Google Scholar] [CrossRef]
- Adeva-Andany, M.M.; Calvo-Castro, I.; Fernandez-Fernandez, C.; Donapetry-Garcia, C.; Pedre-Pineiro, A.M. Significance of l-carnitine for human health. IUBMB Life 2017, 69, 578–594. [Google Scholar] [CrossRef]
- Germain, P.; Staels, B.; Dacquet, C.; Spedding, M.; Laudet, V. Overview of nomenclature of nuclear receptors. Pharmacol. Rev. 2006, 58, 685–704. [Google Scholar] [CrossRef] [PubMed]
- Elmslie, J.L.; Porter, R.J.; Joyce, P.R.; Hunt, P.J.; Mann, J.I. Carnitine does not improve weight loss outcomes in valproate-treated bipolar patients consuming an energy-restricted, low-fat diet. Bipolar. Disord. 2006, 8, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P.; Salvadeo, S.A.; Ferrari, I.; Gravina, A.; Mereu, R.; Cicero, A.F. Comparison of orlistat treatment and placebo in obese type 2 diabetic patients. Expert Opin. Pharmacother. 2010, 11, 1971–1982. [Google Scholar] [CrossRef]
- Derosa, G.; Maffioli, P.; Salvadeo, S.A.; Ferrari, I.; Gravina, A.; Mereu, R. Effects of combination of sibutramine and L-carnitine compared with sibutramine monotherapy on inflammatory parameters in diabetic patients. Metabolism 2011, 60, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Barzegar, A.; Alipour, B.; Panahi, F.; Karamzad, N. Effect of L-carnitine supplementation on serum adipokines (leptin and visfatin) levels in obese type II diabetes mellitus women with hypocaloric diet. Life Sci. J. 2013, 10, 359–365. [Google Scholar]
- Ching, P.L.; Willett, W.C.; Rimm, E.B.; Colditz, G.A.; Gortmaker, S.L.; Stampfer, M.J. Activity level and risk of overweight in male health professionals. Am. J. Public Health 1996, 86, 25–30. [Google Scholar] [CrossRef]
- Pala, R.; Genc, E.; Tuzcu, M.; Orhan, C.; Sahin, N.; Er, B.; Sahin, K. L-Carnitine supplementation increases expression of PPAR-γ and glucose transporters in skeletal muscle of chronically and acutely exercised rats. Cell Mol. Biol. 2018, 64, 1–6. [Google Scholar] [CrossRef]
- Parandak, K.; Arazi, H.; Khoshkhahesh, F.; Nakhostin-Roohi, B. The effect of two-week L-carnitine supplementation on exercise-induced oxidative stress and muscle damage. Asian J. Sports Med. 2014, 5, 123–128. [Google Scholar]
- Sen, C.K. Antioxidants and redox regulation of cellular signaling: Introduction. Med. Sci. Sports Exerc. 2001, 33, 368–370. [Google Scholar] [CrossRef]
- Sener, G.; Paskaloglu, K.; Satiroglu, H.; Alican, I.; Kaçmaz, A.; Sakarcan, A. L-carnitine ameliorates oxidative damage due to chronic renal failure in rats. J. Cardiovasc. Pharmacol. 2004, 43, 698–705. [Google Scholar] [CrossRef]
- Rani, P.J.; Panneerselvam, C. Carnitine as a free radical scavenger in aging. Exp. Gerontol. 2001, 36, 1713–1726. [Google Scholar] [CrossRef]
- Stephens, F.B.; Constantin-Teodosiu, D.; Laithwaite, D.; Simpson, E.J.; Greenhaff, P.L. An acute increase in skeletal muscle carnitine content alters fuel metabolism in resting human skeletal muscle. J. Clin. Endocrinol. Metab. 2006, 91, 5013–5018. [Google Scholar] [CrossRef] [PubMed]
- Muoio, D.M.; Noland, R.C.; Kovalik, J.P.; Seiler, S.E.; Davies, M.N.; DeBalsi, K.L.; Covington, J.D. Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility. Cell Metab. 2012, 15, 764–777. [Google Scholar] [CrossRef] [PubMed]
- Frosig, C.; Rose, A.J.; Treebak, J.T.; Kiens, B.; Richter, E.A.; Wojtaszewski, J.F. Effects of endurance exercise training on insulin signaling in human skeletal muscle: Interactions at the level of phosphatidylinositol 3-kinase, Akt, and AS160. Diabetes 2007, 56, 2093–2102. [Google Scholar] [CrossRef] [PubMed]
- Casciani, C.U.; Caruso, U.; Cravotto, E.; Corsi, M.; Pola, P.; Savi, L.; Grilli, M. Effect of L-carnitine on lipid pattern in haemodialysis. Lancet 1980, 316, 1309–1310. [Google Scholar] [CrossRef]
- Vacha, G.M.; Giorcelli, G.; Siliprandi, N.; Corsi, M. Favorable effects of L-carnitine treatment on hypertriglyceridemia in hemodialysis patients: Decisive role of low levels of high-density lipoprotein-cholesterol. Am. J. Clin. Nutr. 1983, 38, 532–540. [Google Scholar] [CrossRef]
- Malaguarnera, M.; Vacante, M.; Avitabile, T.; Malaguarnera, M.; Cammalleri, L.; Motta, M. L-Carnitine supplementation reduces oxidized LDL cholesterol in patients with diabetes. Am. J. Clin. Nutr. 2009, 89, 71–76. [Google Scholar] [CrossRef]
- Askarpour, M.; Hadi, A.L.; Symonds, M.E.; Miraghajani, M.; Omid, S.; Sheikhi, A.; Ghaedi, E. Efficacy of l-carnitine supplementation for the management of blood lipids: A systematic review and dose-response meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1151–1167. [Google Scholar] [CrossRef]
- Fathizadeh, H.; Milajerdi, A.; Reiner, Ž.; Kolahdooz, F.; Chamani, M.; Amirani, E.; Asemi, Z. The Effects of L-Carnitine Supplementation on Serum Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Curr. Pharm. Des. 2019, 25, 3266–3281. [Google Scholar] [CrossRef]
- Orzali, A.; Donzelli, F.; Enzi, G.; Rubaltelli, F.F. Effect of carnitine on lipid metabolism in the newborn. I. Carnitine supplementation during total parenteral nutrition in the first 48 h of life. Biol. Neonatol. 1983, 43, 186–190. [Google Scholar] [CrossRef]
- Penn, D.; Schmidt-Sommerfeld, E. Carnitine and carnitine esters in plasma and adipose tissue of chronic uremic patients undergoing hemodialysis. Metabolism 1983, 32, 806–809. [Google Scholar] [CrossRef]
- Abolfathi, M.; Mohd-Yusof, B.N.; Hanipah, Z.N.; Mohd Redzwan, S.; Yusof, L.M.; Khosroshahi, M.Z. The effects of carnitine supplementation on clinical characteristics of patients with non-alcoholic fatty liver disease: A systematic review and meta-analysis of randomized controlled trials. Complement. Ther. Med. 2020, 48, 102273. [Google Scholar] [CrossRef] [PubMed]
- Bucioli, S.A.; De Abreu, L.C.; Valenti, V.E.; Vannucchi, H. Carnitine supplementation effects on nonenzymatic antioxidants in young rats submitted to exhaustive exercise stress. J. Strength Cond. Res. 2012, 26, 1695–1700. [Google Scholar] [CrossRef] [PubMed]

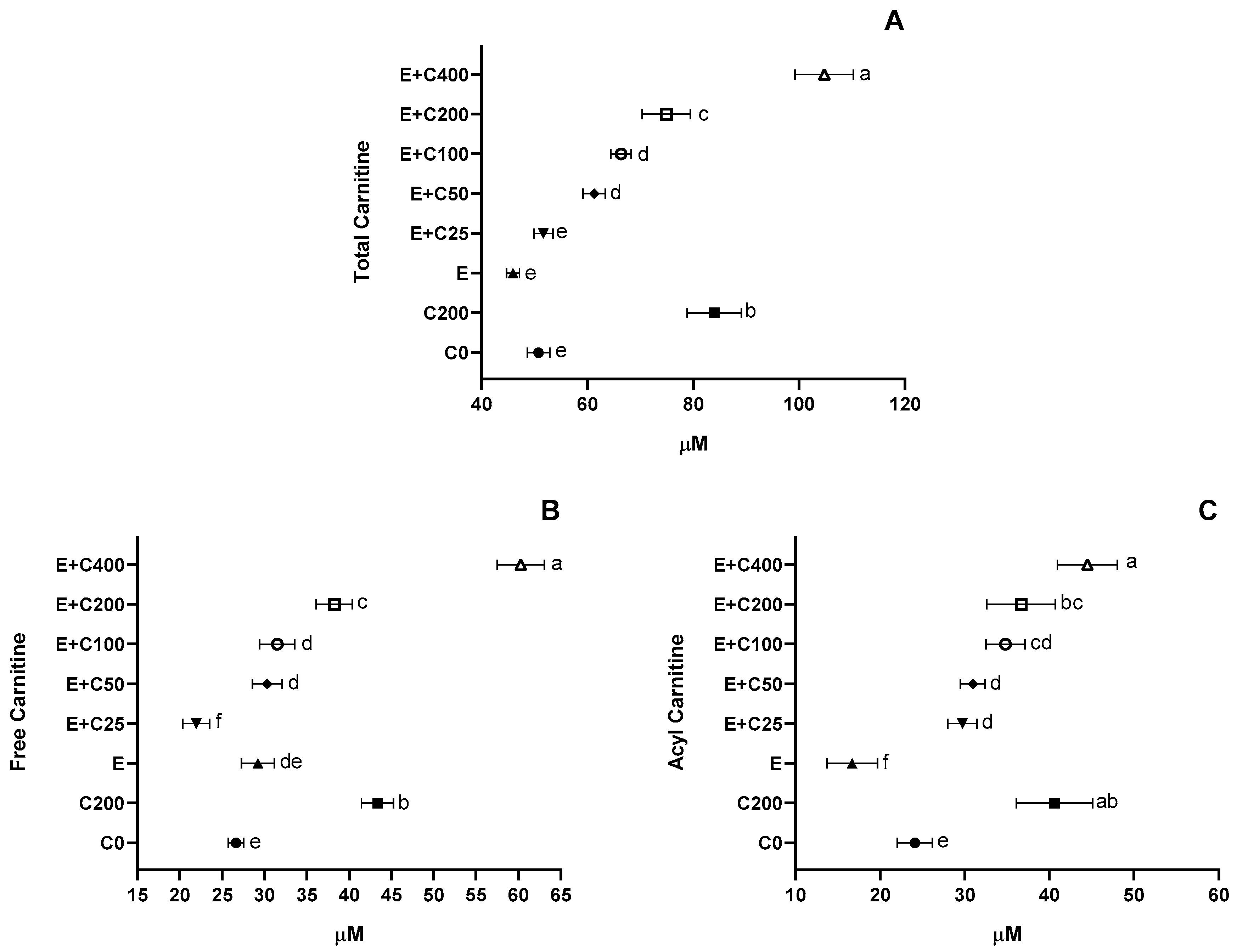
| Ingredient | % |
|---|---|
| Maize | 26.00 |
| Wheat | 14.00 |
| Vegetable oil | 3.00 |
| Soybean meal, 48% CP 1 | 33.10 |
| Sunflower meal, 30% CP | 8.00 |
| Wheat bran | 7.00 |
| Molasses | 5.00 |
| Limestone | 0.80 |
| Salt | 0.80 |
| DL-Methionine | 0.80 |
| Dicalcium phosphate | 1.20 |
| Vitamin and mineral premix 2 | 0.30 |
| Analysis (%) | |
| Crude protein | 24.27 |
| Ether extract | 4.55 |
| Crude cellulose | 4.04 |
| Ash | 6.91 |
| Ca | 0.75 |
| P | 0.41 |
| Items | Groups | |||||||
|---|---|---|---|---|---|---|---|---|
| Control | Car200 | Exercise (E) | E+Car25 | E+Car50 | E+Car100 | E+Car200 | E+Car400 | |
| MDA, µmol/L | 0.873 ± 0.017 a | 0.693 ± 0.021 b | 0.768 ± 0.015b | 0.599 ± 0.021 c | 0.549 ± 0.014 c,d | 0.553 ± 0.018 c,d | 0.489 ± 0.019 d,e | 0.419 ± 0.009 e |
| SOD, U/mg protein | 83.88 ± 2.21 e | 95.87 ± 2.47 c,d | 85.21 ± 2.99 d,e | 96.49 ± 0.78 c,d | 102.89 ± 2.69 b,c | 109.68 ± 2.49 a,b | 114.56 ± 2.70 a | 120.16 ± 3.58 a |
| CAT, U/mg protein | 135.51 ± 2.61 e | 147.19 ± 2.01 d,e | 138.47 ± 3.69 e | 144.55 ± 3.21 d,e | 152.72 ± 2.50 c,d | 162.11 ± 1.97 b,c | 170.77 ± 2.57 b | 186.10 ± 2.50 a |
| GSHPx, U/mg protein | 36.87 ± 1.27 f | 48.74 ± 1.11 d | 42.56 ± 1.43 e | 49.75 ± 1.08 d | 55.91 ± 0.85 c | 59.57 ± 0.68 b,c | 62.58 ± 1.03 b | 69.06 ± 1.53 a |
| Items | Groups | |||||||
|---|---|---|---|---|---|---|---|---|
| Control | Car200 | Exercise (E) | E+Car25 | E+Car50 | E+Car100 | E+Car200 | E+Car400 | |
| GLU, mg/dL | 115.29 ± 2.01 a | 109.43 ± 1.25 a,b | 104.43 ± 1.32 b,c | 101.14 ± 1.97 c | 103.43 ± 1.46 b,c | 101.43 ± 1.36 c | 98.43 ± 1.32 c,d | 92.43 ± 1.25 d |
| TG, mg/dL | 172.86 ± 4.08 a | 165.00 ± 2.32 a,b | 156.14 ± 4.08 b,c | 150.57 ± 3.21 c | 135.29 ± 2.54 d | 124.43 ± 1.46 d,e | 120.14 ± 2.11 e | 113.43 ± 1.39 e |
| TC, mg/dL | 149.57 ± 2.10 a | 142.62 ± 1.45 b | 138.22 ± 0.67 b,c | 135.10 ± 0.56 c | 127.57 ± 1.25 d | 115.08 ± 0.62 e | 110.23 ± 0.78 e | 102.27 ± 1.50 f |
| BUN, mg/dL | 21.59 ± 0.82 | 21.40 ± 0.65 | 20.67 ± 0.45 | 21.23 ± 0.90 | 21.34 ± 0.85 | 21.60 ± 0.82 | 21.31 ± 1.06 | 21.41 ± 0.69 |
| TP, g/dL | 6.50 ± 0.18 | 6.49 ± 0.13 | 6.50 ± 0.23 | 6.13 ± 0.43 | 5.93 ± 0.29 | 6.07 ± 0.44 | 6.37 ± 0.18 | 6.29 ± 0.20 |
| TBIL, mg/dL | 0.23 ± 0.01 | 0.24 ± 0.01 | 0.23 ± 0.01 | 0.22 ± 0.01 | 0.22 ± 0.01 | 0.23 ± 0.01 | 0.24 ± 0.01 | 0.23 ± 0.02 |
| Items | Groups | |||||||
|---|---|---|---|---|---|---|---|---|
| Control | Car200 | Exercise (E) | E+Car25 | E+Car50 | E+Car100 | E+Car200 | E+Car400 | |
| ALB, g/dL | 3.40 ± 0.08 | 3.46 ± 0.05 | 3.47 ± 0.09 | 3.23 ± 0.16 | 3.09 ± 0.21 | 3.56 ± 0.05 | 3.39 ± 0.07 | 3.44 ± 0.06 |
| GLOB, g/dL | 3.04 ± 0.12 b | 3.10 ± 0.08 a,b | 3.41 ± 0.10 a,b | 3.27 ± 0.11 a,b | 3.29 ± 0.12 a,b | 3.53 ± 0.11 a | 3.11 ± 0.11 a,b | 3.16 ± 0.05 a,b |
| ALT, U/L | 103.43 ± 5.12 | 100.86 ± 4.07 | 99.43 ± 3.73 | 100.00 ± 4.62 | 101.00 ± 3.13 | 99.57 ± 4.49 | 104.86 ± 4.40 | 100.14 ± 2.96 |
| AST, U/L | 121.78 ± 6.73 | 122.94 ± 7.48 | 121.68 ± 3.64 | 121.31 ± 5.17 | 120.50 ± 3.77 | 122.57 ± 3.29 | 122.57 ± 3.72 | 121.57 ± 4.95 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahin, K.; Orhan, C.; Kucuk, O.; Sahin, N.; Tuzcu, M.; Er, B.; Durkee, S.; Bellamine, A. A Dose-Dependent Effect of Carnipure® Tartrate Supplementation on Endurance Capacity, Recovery, and Body Composition in an Exercise Rat Model. Nutrients 2020, 12, 1519. https://doi.org/10.3390/nu12051519
Sahin K, Orhan C, Kucuk O, Sahin N, Tuzcu M, Er B, Durkee S, Bellamine A. A Dose-Dependent Effect of Carnipure® Tartrate Supplementation on Endurance Capacity, Recovery, and Body Composition in an Exercise Rat Model. Nutrients. 2020; 12(5):1519. https://doi.org/10.3390/nu12051519
Chicago/Turabian StyleSahin, Kazim, Cemal Orhan, Osman Kucuk, Nurhan Sahin, Mehmet Tuzcu, Besir Er, Shane Durkee, and Aouatef Bellamine. 2020. "A Dose-Dependent Effect of Carnipure® Tartrate Supplementation on Endurance Capacity, Recovery, and Body Composition in an Exercise Rat Model" Nutrients 12, no. 5: 1519. https://doi.org/10.3390/nu12051519
APA StyleSahin, K., Orhan, C., Kucuk, O., Sahin, N., Tuzcu, M., Er, B., Durkee, S., & Bellamine, A. (2020). A Dose-Dependent Effect of Carnipure® Tartrate Supplementation on Endurance Capacity, Recovery, and Body Composition in an Exercise Rat Model. Nutrients, 12(5), 1519. https://doi.org/10.3390/nu12051519







