The In Vitro Analysis of Prebiotics to Be Used as a Component of a Synbiotic Preparation
Abstract
1. Introduction
2. Materials and Methods
2.1. Prebiotic Substances
2.2. Microorganisms and Inoculum Preparation
2.3. Growth of Selected Lactobacillus spp. Strains in the Presence of Prebiotics
2.4. Identification of Metabolites Produced by the Probiotic Lactobacillus spp. in the Process of Prebiotics Fermentation
2.5. Enzymatic Identification of Optically Active Forms of Lactic Acid
2.6. Enzymatic Profile of Probiotic Lactobacillus spp. in the Presence of Prebiotics
2.7. The Influence of the Prebiotics on the Antagonistic Activity of Lactobacillus spp. Strains towards Pathogenic Bacteria
2.8. Statistical Analysis
3. Results
3.1. Growth of Selected Lactobacillus spp. Strains in the Presence of Prebiotics
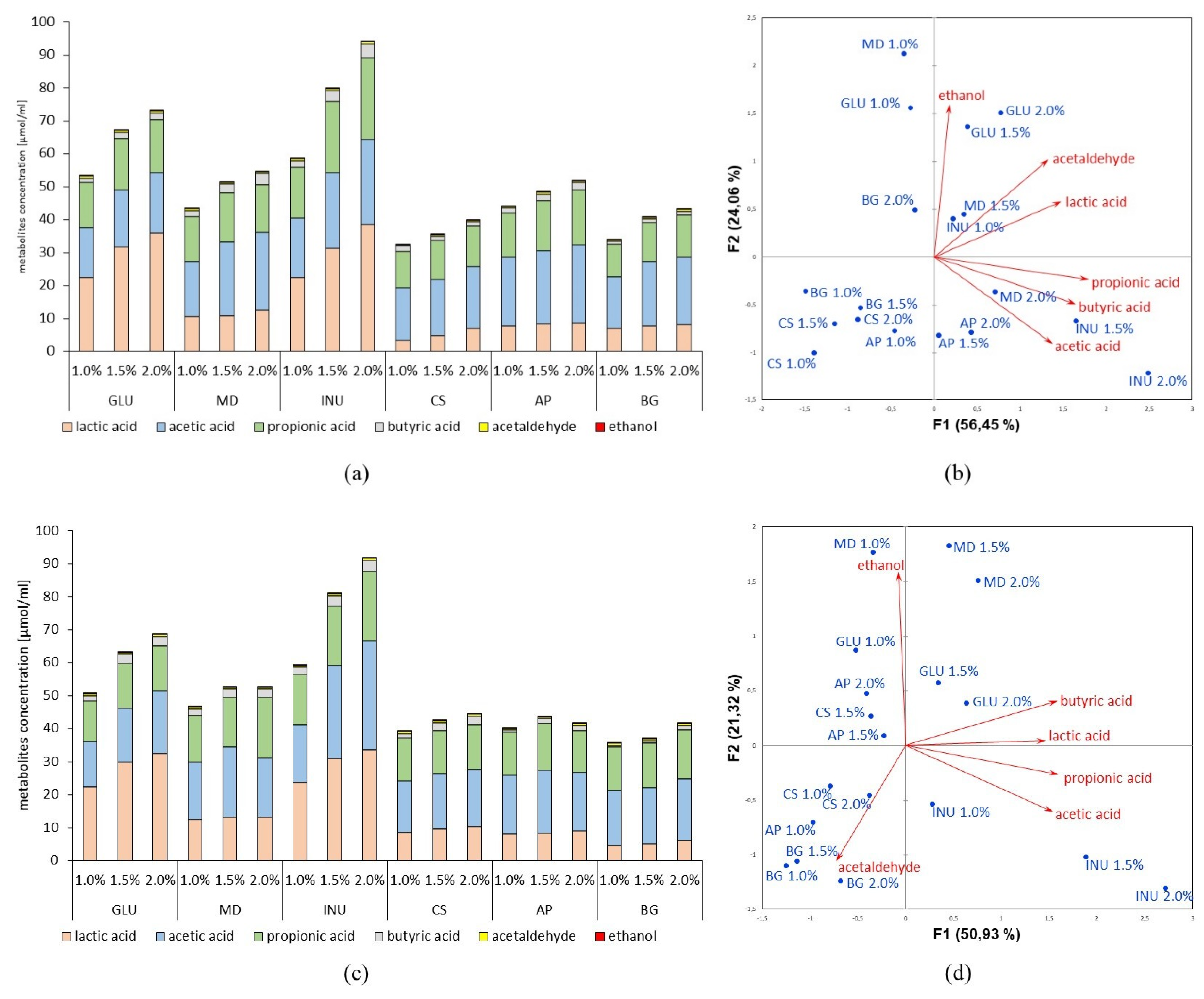
3.2. Identification of Metabolites Produced by the Probiotic Lactobacillus spp. in the Process of Prebiotics Fermentation
3.3. Enzymatic Identification of Optically Active Forms of Lactic Acid
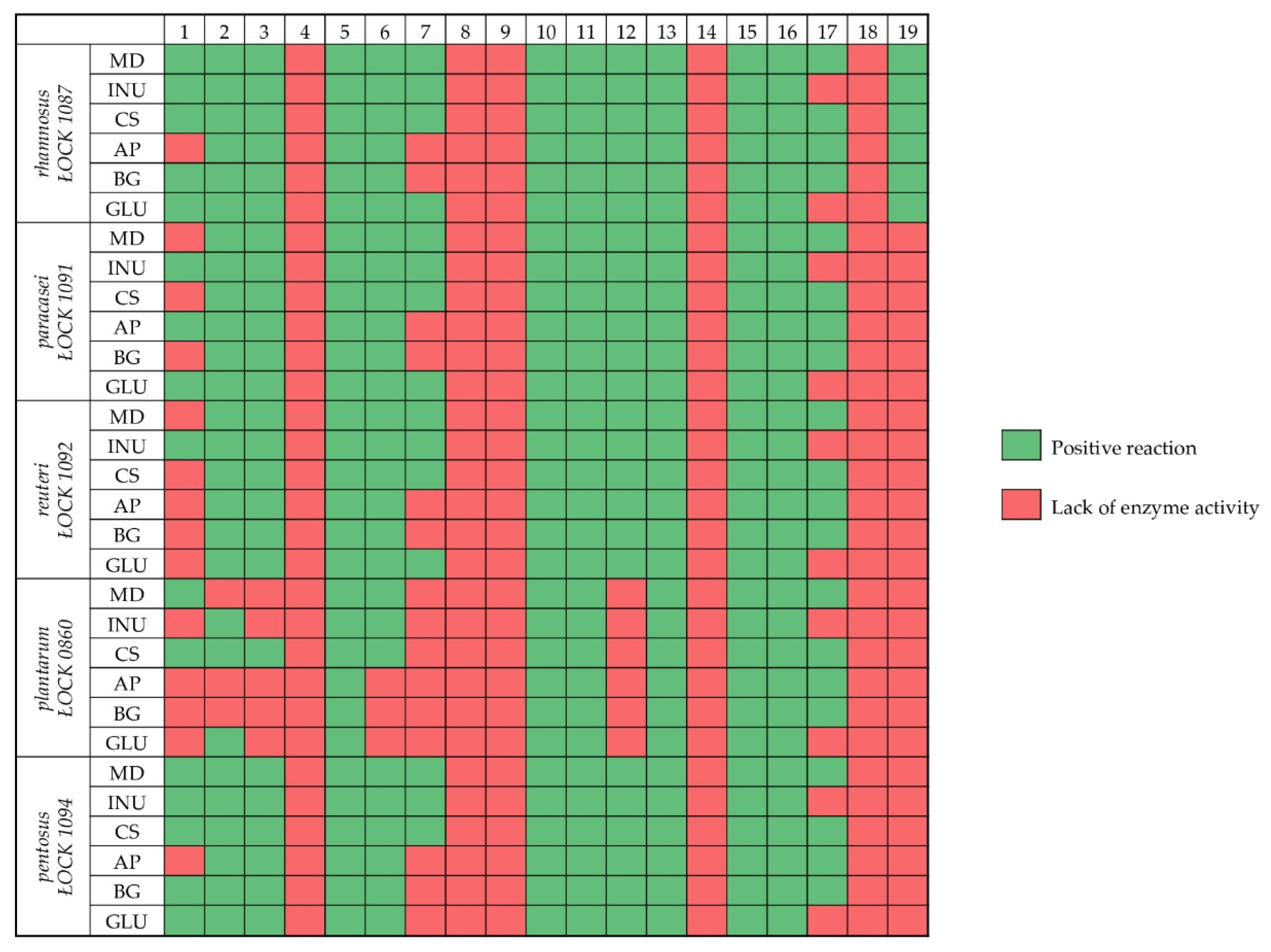
3.4. Enzymatic Profile of Probiotic Lactobacillus spp. in the Presence of Prebiotics
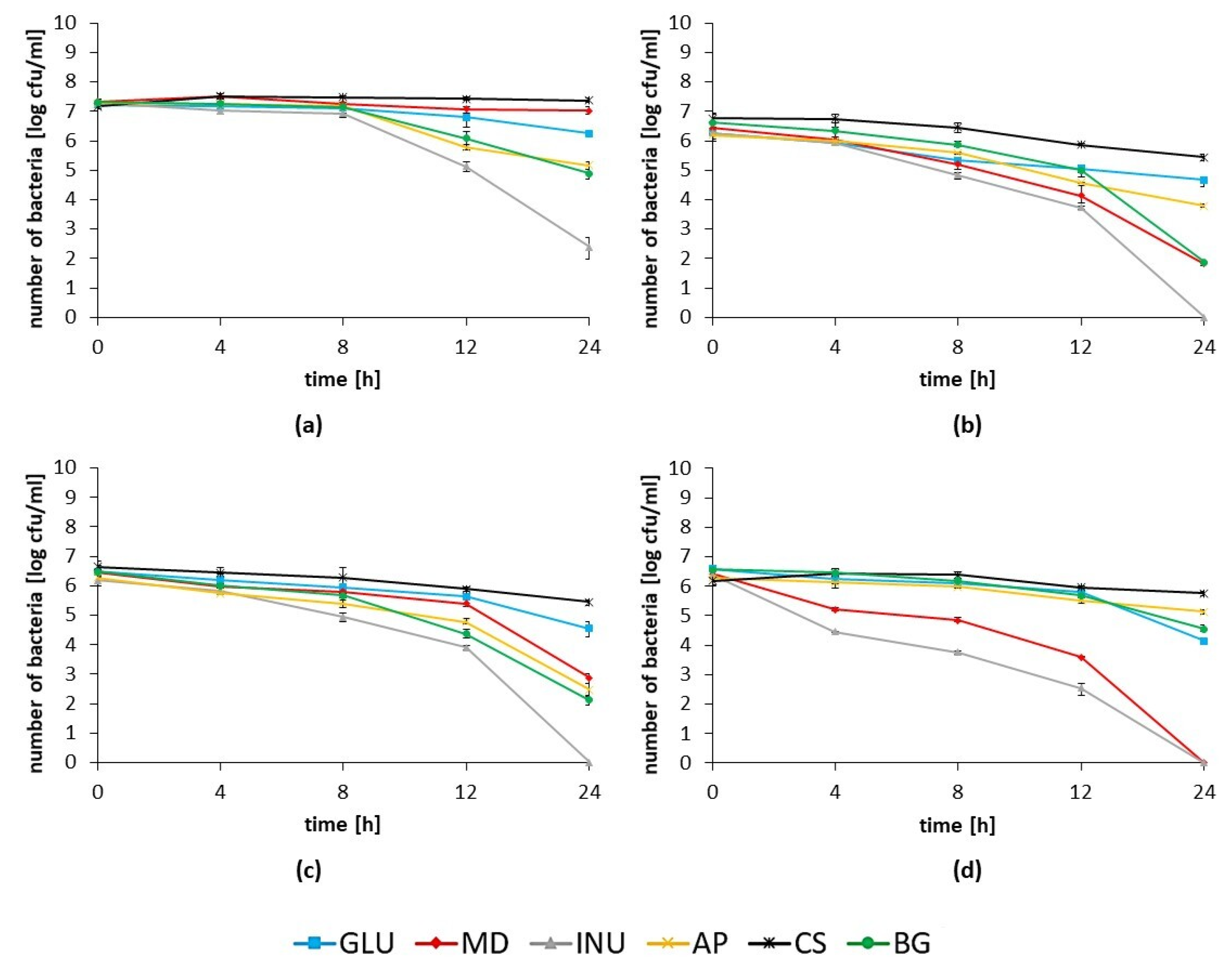
3.5. The Influence of the Prebiotics on the Antagonistic Activity of Lactobacillus spp. Strains towards Pathogenic Bacteria
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO; WHO. Guidelines for the Evaluation of Probiotics in Food; Food Agriculture Organization: Rome, Italy; World Health Organation: Geneva, Switzerland, 2002. [Google Scholar]
- Gibson, G.R.; Roberfroid, M.B. Dietary Modulation of the Human Colonic Microbiota: Introducing the Concept of Prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [PubMed]
- De Paulo Farias, D.; De Araújo, F.F.; Neri-Numa, I.A.; Pastore, G.M. Prebiotics: Trends in food, health and technological applications. Trends Food Sci. Technol. 2019, 93, 23–25. [Google Scholar] [CrossRef]
- Gomaa, E.Z. Effect of prebiotic substances on growth, fatty acid profile and probiotic characteristics of Lactobacillus brevis NM101-1. Microbiology (Russian Federation) 2017, 86, 618–628. [Google Scholar] [CrossRef]
- Pabari, K.; Pithva, S.; Kothari, C.; Purama, R.K.; Kondepudi, K.K.; Vyas, B.R.M.; Kothari, R.; Ambalam, P. Evaluation of Probiotic Properties and Prebiotic Utilization Potential of Weissella paramesenteroides Isolated From Fruits. Probiotics Antimicrob. Proteins 2020. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Kumar, P.; Saxena, M.J. Feed Additives in Animal Health. In Nutraceuticals in Veterinary Medicine; Gupta, R.C., Srivastava, A., Lall, R., Eds.; Springer: Cham, Switzerland, 2019; pp. 345–362. [Google Scholar]
- Sharma, S.; Bano, A.; Gupta, A.; Bajpai, P.; Lohani, M.; Pathak, N. Pre- and Probiotics: Using Functional Foods in the Fight Against Microbial Resistance to Antibiotics. In Antibacterial Drug Discovery to Combat MDR; Ahmad, I., Ahmad, S., Rumbaugh, K., Eds.; Springer: Singapore, 2019; pp. 397–425. [Google Scholar]
- Rawi, M.H.; Zaman, S.A.; Pa’ee, K.F.; Leong, S.S.; Sarbini, S.R. Prebiotics metabolism by gut-isolated probiotics. J. Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, types, sources, mechanisms, and clinical applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Nicolaus, B.; Poli, A.; Orlando, P. The prebiotic source influences the growth, biochemical features and survival under simulated gastrointestinal conditions of the probiotic Lactobacillus acidophilus. Anaerobe 2012, 18, 280–285. [Google Scholar] [CrossRef]
- Anadón, A.; Ares, I.; Martínez-Larrañaga, M.R.; Martínez, M.A. Prebiotics and Probiotics in Feed and Animal Health. In Nutraceuticals in Veterinary Medicine; Gupta, R.C., Srivastava, A., Lall, R., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Dhewa, T.; Pant, S.; Mishra, V. Development of freeze dried synbiotic formulation using a probiotic strain of Lactobacillus plantarum. J. Food Sci. Technol. 2014, 51, 83–89. [Google Scholar] [CrossRef]
- Yoo, J.Y.; Kim, S.S. Probiotics and Prebiotics: Present Status and Future Perspectives on Metabolic Disorders. Nutrients 2016, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Institute of Fermentation Technology and Microbiology. Pure Cultures Collection (ŁOCK 105). Available online: http://mikrobiologia.p.lodz.pl/kolekcja-lock/ (accessed on 20 April 2020).
- Śliżewska, K.; Chlebicz, A. Lactic Bacterial Strain of Lactobacillus pentosus. PL Patent 233261 B1, 30 September 2019. [Google Scholar]
- Śliżewska, K.; Chlebicz, A. Lactic Bacterial Strain of Lactobacillus reuteri. PL Patent 233263 B1, 30 September 2019. [Google Scholar]
- Śliżewska, K.; Chlebicz, A. Lactic Bacterial Strain of Lactobacillus rhamnosus. PL Patent 233582 B1, 29 November 2019. [Google Scholar]
- Śliżewska, K.; Chlebicz, A. Lactic Bacterial Strain of Lactobacillus paracasei. PL Patent 233262 B1, 30 September 2019. [Google Scholar]
- Śliżewska, K.; Motyl, I.; Libudzisz, Z.; Otlewska, A.; Burchardt, H.; Klecha, J.; Henzler, J. Lactobacillus plantarum lactic bacteria strain. PL Patent 221959 B1, 30 June 2016. [Google Scholar]
- Hayek, S.A.; Ibrahim, S.A. Current Limitations and Challenges with Lactic Acid Bacteria: A Review. Food Nutr. Sci. 2013, 4, 73–87. [Google Scholar] [CrossRef]
- Watson, D.; O’Connell Motherway, M.; Schoterman, M.H.C.; van Neerven, R.J.J.; Nauta, A.; Van Sinderen, D. Selective carbohydrate utilization by lactobacilli and bifidobacteria. J. Appl. Microbiol. 2013, 114, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- Iraporda, C.; Rubel, I.A.; Manrique, G.D.; Abraham, A.G. Influence of inulin rich carbohydrates from Jerusalem artichoke (Helianthus tuberosus L.) tubers on probiotic properties of Lactobacillus strains. LWT Food Sci. Technol. 2019, 101, 738–746. [Google Scholar] [CrossRef]
- McLaughlin, H.P.; Motherway, M.O.C.; Lakshminarayanan, B.; Stanton, C.; Paul Ross, R.; Brulc, J.; Menon, R.; O’Toole, P.W.; van Sinderen, D. Carbohydrate catabolic diversity of bifidobacteria and lactobacilli of human origin. Int. J. Food Microbiol. 2015, 203, 109–121. [Google Scholar] [CrossRef]
- Kunová, G.; Rada, V.; Lisová, I.; Ročková, Š.; Vlková, E. In vitro fermentability of prebiotic oligosaccharides by lactobacilli. Czech J. Food Sci. 2018, 29, 49–54. [Google Scholar] [CrossRef]
- Önal Darilmaz, D.; Sönmez, Ş.; Beyatli, Y. The effects of inulin as a prebiotic supplement and the synbiotic interactions of probiotics to improve oxalate degrading activity. Int. J. Food Sci. Technol. 2019, 54, 12–131. [Google Scholar] [CrossRef]
- König, H.; Fröhlich, J. Lactic acid bacteria. In Biology of Microorganisms on Grapes, in Must and in Wine; Springer: Berlin/Heidelberg, Germany, 2017; pp. 3–41. ISBN 9783319600215. [Google Scholar]
- Chen, C.C.; Lai, C.C.; Huang, H.L.; Huang, W.Y.; Toh, H.S.; Weng, T.C.; Chuang, Y.C.; Lu, Y.C.; Tang, H.J. Antimicrobial activity of Lactobacillus species against carbapenem-resistant Enterobact. Front. Microbiol. 2019, 10, 789. [Google Scholar] [CrossRef]
- Tingirikari, J.M.R. In-Vitro Prebiotic Analysis of Microbiota Accessible Pectic Polysaccharides. Curr. Microbiol. 2019, 76, 1452–1460. [Google Scholar] [CrossRef]
- Sharma, S.; Kanwar, S.S. Effect of prebiotics on growth behavior of Lactobacillus plantarum and their impact on adherence of strict anaerobic pathogens to intestinal cell lines. J. Food Saf. 2018, 38, e12384. [Google Scholar] [CrossRef]
- De Souza Oliveira, R.P.; Perego, P.; de Oliveira, M.N.; Converti, A. Effect of inulin on the growth and metabolism of a probiotic strain of Lactobacillus rhamnosus in co-culture with Streptococcus thermophilus. LWT Food Sci. Technol. 2012, 47, 358–363. [Google Scholar] [CrossRef]
- Tagaino, R.; Washio, J.; Abiko, Y.; Tanda, N.; Sasaki, K.; Takahashi, N. Metabolic property of acetaldehyde production from ethanol and glucose by oral Streptococcus and Neisseria. Sci. Rep. 2019, 9, 10446. [Google Scholar] [CrossRef] [PubMed]
- Cizeikiene, D.; Juodeikiene, G.; Damasius, J. Use of wheat straw biomass in production of L-lactic acid applying biocatalysis and combined lactic acid bacteria strains belonging to the genus Lactobacillus. Biocatal. Agric. Biotechnol. 2018, 15, 185–191. [Google Scholar] [CrossRef]
- Trontel, A.; Batušić, A.; Gusić, I.; Slavica, A.; Šantek, B.; Novak, S. Production of D- and L-lactic acid by mono- and mixed cultures of Lactobacillus sp. Food Technol. Biotechnol. 2011, 49, 75–82. [Google Scholar]
- Mack, D.R. D(-)-lactic acid-producing probiotics, D(-)-lactic acidosis and infants. Can. J. Gastroenterol. 2004, 18, 75–82. [Google Scholar] [CrossRef]
- Mayeur, C.; Gratadoux, J.-J.; Bridonneau, C.; Chegdani, F.; Larroque, B.; Kapel, N.; Corcos, O.; Thomas, M.; Joly, F. Faecal D/L Lactate Ratio Is a Metabolic Signature of Microbiota Imbalance in Patients with Short Bowel Syndrome. PLoS ONE 2013, 8, e54335. [Google Scholar] [CrossRef]
- Goderska, K.; Nowak, J.; Czarnecki, Z. Comparison of the growth of Lactobacillus acidophilus and Bifidobacterium bifidum species in media supplemented with selected saccharides including prebiotics. Technol. Aliment. 2008, 7, 5–20. [Google Scholar]
- Kanjan, P.; Hongpattarakere, T. Prebiotic efficacy and mechanism of inulin combined with inulin-degrading Lactobacillus paracasei I321 in competition with Salmonella. Carbohydr. Polym. 2017, 169, 236–244. [Google Scholar] [CrossRef]
- Chuah, L.O.; Foo, H.L.; Loh, T.C.; Mohammed Alitheen, N.B.; Yeap, S.K.; Abdul Mutalib, N.E.; Abdul Rahim, R.; Yusoff, K. Postbiotic metabolites produced by Lactobacillus plantarum strains exert selective cytotoxicity effects on cancer cells. BMC Complement. Altern. Med. 2019, 19, 114. [Google Scholar] [CrossRef]
- Likotrafiti, E.; Valavani, P.; Argiriou, A.; Rhoades, J. In vitro evaluation of potential antimicrobial synbiotics using Lactobacillus kefiri isolated from kefir grains. Int. Dairy J. 2015, 45, 23–30. [Google Scholar] [CrossRef]
- Chaiyasut, C.; Pattananandecha, T.; Sirilun, S.; Suwannalert, P.; Peerajan, S.; Sivamaruthi, B.S. Synbiotic preparation with lactic acid bacteria and inulin as a functional food: In vivo evaluation of microbial activities, and preneoplastic aberrant crypt foci. Food Sci. Technol. 2017, 37, 328–336. [Google Scholar] [CrossRef]
- Shokryazdan, P.; Jahromi, M.F.; Liang, J.B.; Sieo, C.C.; Kalavathy, R.; Idrus, Z.; Ho, Y.W. In Vitro Assessment of Bioactivities of Lactobacillus Strains as Potential Probiotics for Humans and Chickens. J. Food Sci. 2017, 82, 2734–2745. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.A.; Jerônimo, G.T.; da Costa Marchiori, N.; de Oliveira, H.M.; Owatari, M.S.; Jesus, G.F.A.; Garcia, P.; do Nascimento Vieira, F.; Martins, M.L.; Mouriño, J.L.P. Autochthonous probiotic Lactobacillus sp. in the diet of bullfrog tadpoles Lithobates catesbeianus improves weight gain, feed conversion and gut microbiota. Aquac. Nutr. 2017, 23, 910–916. [Google Scholar] [CrossRef]
- Jang, H.J.; Song, M.W.; Lee, N.K.; Paik, H.D. Antioxidant effects of live and heat-killed probiotic Lactobacillus plantarum Ln1 isolated from kimchi. J. Food Sci. Technol. 2018, 55, 3174–3180. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Kim, S.J.; Lee, J.Y.; Yoon, S.R.; You, S.Y.; Kim, S.H. Multifunctional properties of Lactobacillus plantarum strains WiKim83 and WiKim87 as a starter culture for fermented food. Food Sci. Nutr. 2019, 7, 2505–2516. [Google Scholar] [CrossRef]
- Cheon, M.J.; Lim, S.M.; Lee, N.K.; Paik, H.D. Probiotic Properties and Neuroprotective Effects of Lactobacillus buchneri KU200793 Isolated from Korean Fermented Foods. Int. J. Mol. Sci. 2020, 21, 1227. [Google Scholar] [CrossRef]
- Aziz, G.; Fakhar, H.; Rahman, S.; Tariq, M.; Zaidi, A. An assessment of the aggregation and probiotic characteristics of Lactobacillus species isolated from native (desi) chicken gut. J. Appl. Poult. Res. 2019, 28, 846–857. [Google Scholar] [CrossRef]
- O’Donnell, M.M.; Harris, H.M.B.; Lynch, D.B.; Ross, R.P.; O’Toole, P.W. Lactobacillus ruminis strains cluster according to their mammalian gut source. BMC Microbiol. 2015, 15, 80. [Google Scholar]
- Pisano, M.B.; Viale, S.; Conti, S.; Fadda, M.E.; Deplano, M.; Melis, M.P.; Deiana, M.; Cosentino, S. Preliminary evaluation of probiotic properties of Lactobacillus strains isolated from Sardinian dairy products. Biomed. Res. Int. 2014, 2014, 286390. [Google Scholar] [CrossRef]
- Ng, S.Y.; Koon, S.S.; Padam, B.S.; Chye, F.Y. Evaluation of probiotic potential of lactic acid bacteria isolated from traditional Malaysian fermented Bambangan (Mangifera pajang). CYTA J. Food 2015, 13, 563–572. [Google Scholar]

| Strains | Source |
|---|---|
| Lb. paracasei ŁOCK 1091 | Caecal content of sow |
| Lb. pentosus ŁOCK 0860 | Broiler chicken dung |
| Lb. plantarum ŁOCK 0860 | Plant silage |
| Lb. reuteri ŁOCK 1092 | Piglet’s caecal content |
| Lb. rhamnosus ŁOCK 1087 | Turkey dung |
| Carbon Source 1 | Concentration [% (w/v)] | Lactobacillus spp. Strain [log CFU/mL] 2 | ||||
|---|---|---|---|---|---|---|
| rhamnosus ŁOCK 1087 | paracasei ŁOCK 1091 | reuteri ŁOCK 1092 | plantarum ŁOCK 0860 | pentosus ŁOCK 1094 | ||
| GLU | 1.0 | 8.00 ± 1.19 a,b,c | 7.71 ± 1.03 a,b | 7.53 ± 1.28 a | 7.99 ± 1.06 a,b,c | 8.55 ± 1.14 |
| 1.5 | 9.17 ± 0.73 | 8.59 ± 0.78 | 8.19 ± 0.72 | 8.52 ± 0.56 | 8.44 ± 0.77 | |
| 2.0 | 10.06 ± 0.68 | 10.50 ± 0.50 | 8.53 ± 0.84 | 9.70 ± 1.09 | 9.98 ± 0.71 | |
| MD | 1.0 | 9.44 ± 0.93 | 9.89 ±0.64 | 8.93 ± 1.34 | 9.74 ± 0.98 | 8.79 ± 0.70 |
| 1.5 | 9.70 ± 0.75 | 9.91 ± 1.03 | 9.49 ± 0.89 | 10.54 ± 0.50 b,c,d | 9.83 ± 0.46 | |
| 2.0 | 9.80 ± 0.97 | 10.37 ± 0.54 | 10.05 ± 0.56 | 10.38 ± 0.89 | 9.64 ± 1.15 | |
| INU | 1.0 | 9.02 ± 0.84 | 9.43 ± 0.60 | 10.37 ± 0.52 | 10.15 ± 0.82 | 9.70 ± 1.04 |
| 1.5 | 9.20 ± 1.37 | 9.61 ± 0.92 | 11.04 ± 0.37 d | 10.48 ± 0.80 | 10.08 ± 1.06 | |
| 2.0 | 10.91 ± 0.92 c,d | 10.46 ± 0.59 | 11.09 ± 0.47 d | 10.71 ± 0.81 c,d | 10.38 ± 1.14 | |
| CS | 1.0 | 8.74 ± 0.30 | 8.24 ± 0.78 | 8.24 ± 0.63 | 9.58 ± 0.80 | 8.42 ± 0.67 |
| 1.5 | 9.60 ± 0.89 | 8.99 ± 0.46 | 9.55 ± 0.80 | 10.18 ± 0.59 | 9.68 ± 1.14 | |
| 2.0 | 10.10 ± 0.72 | 9.00 ± 1.01 | 9.26 ± 1.18 | 10.38 ± 0.69 | 9.47 ± 1.00 | |
| AP | 1.0 | 8.92 ± 1.04 | 9.57 ± 0.87 | 10.01 ± 0.29 | 10.14 ± 0.72 | 10.12 ± 1.06 |
| 1.5 | 9.43 ± 0.71 | 9.70 ± 0.75 | 10.51 ± 0.57 | 10.55 ± 0.76 b,c,d | 10.84 ± 0.15 c.d | |
| 2.0 | 9.86 ± 0.85 | 9.93 ± 1.01 | 9.93 ± 1.01 | 10.12 ± 1.10 | 9.90 ± 1.20 | |
| BG | 1.0 | 8.33 ± 1.05 | 9.43 ± 0.62 | 8.64 ± 0.84 | 8.44 ± 0.80 | 8.97 ± 0.67 |
| 1.5 | 9.23 ± 0.67 | 9.41 ± 0.75 | 9.24 ± 1.29 | 9.02 ± 0.74 | 8.85 ± 1.15 | |
| 2.0 | 9.21 ± 0.97 | 10.33 ± 0.46 | 9.88 ± 0.32 | 10.12 ± 0.57 | 9.43 ± 0.80 | |
| Lactobacillus spp. Strain | Lactic Acid Isomer | Carbon Source 1 | |||||
|---|---|---|---|---|---|---|---|
| GLU | MD | INU | CS | AP | BG | ||
| Concentration [g/l] 2 | |||||||
| rhamnosus ŁOCK 1087 | D(-) | 0.942 ± 0.027 d, e | 0.119 ± 0.008 a, b | 0.402 ± 0.075 a, b, c, d | 0.087 ± 0.004 a | 0.166 ± 0.048 a, b | 0.088 ± 0.014 a |
| L(+) | 4.570 ± 1.106 g | 0.407 ± 0.003 a, b, c | 6.634 ± 0.996 h, i | 0.106 ± 0.003 a, b | 0.083 ± 0.007 a | 0.105 ± 0.009 a, b | |
| Total | 5.512 ± 1.079 B | 0.527 ± 0.011 A | 6.634 ± 0.921 B | 0.193 ± 0.001 A | 0.249 ± 0.019 A | 0.193 ± 0.022 A | |
| paracasei ŁOCK 1091 | D(-) | 4.511 ± 0.412 g | 0.112 ± 0.015 a, b | 2.200 ± 0.301 f | 0.023 ± 0.002 a | 0.094 ± 0.003 a | 0.049 ± 0.006 a |
| L(+) | 2.424 ± 0.110 f | 0.547 ± 0.019 a, b, c, d, e | 5.701 ± 0.105 h | 0.032 ± 0.002 a | 0.169 ± 0.049 a, b | 0.057 ± 0.005 a | |
| Total | 6.935 ± 0.522 C | 0.659 ± 0.004 B | 7.901 ± 0.196 D | 0.056 ± 0.005 A | 0.263 ± 0.046 A, B | 0.106 ± 0.012 A | |
| reuteri ŁOCK 1092 | D(-) | 0.950 ± 0.029 e | 0.091 ± 0.007 a | 0.517 ± 0.111 a, b, c, d, e | 0.025 ± 0.002 a | 0.026 ± 0.001 a | 0.017 ± 0.001 a |
| L(+) | 4.698 ± 0.381 g | 0.472 ± 0.001 a, b, c, d, e | 7.282 ± 0.206 j | 0.076 ± 0.009 a | 0.071 ± 0.009 a | 0.078 ± 0.005 a | |
| Total | 5.648 ± 0.411 C | 0.563 ± 0.007 B | 7.799 ± 0.317 D | 0.101 ± 0.011 A | 0.097 ± 0.009 A | 0.095 ± 0.005 A | |
| plantarum ŁOCK 0860 | D(-) | 1.003 ± 0.014 e | 0.067 ± 0.002 a | 0.501 ± 0.007 a, b, c, d, e | 0.008 ± 0.001 a | 0.034 ± 0.005 a | 0.020 ± 0.003 a |
| L(+) | 6.285 ± 0.089 i | 0.487 ± 0.013 a, b, c, d, e | 7.044 ± 0.089 j | 0.059 ± 0.002 a | 0.095 ± 0.002 a | 0.085 ± 0.011 a | |
| Total | 7.287 ± 0.076 C | 0.554 ± 0.011 B | 7.546 ± 0.082 D | 0.068 ± 0.009 A | 0.129 ± 0.017 A | 0.104 ± 0.008 A | |
| pentosus ŁOCK 1094 | D(-) | 0.798 ± 0.085 c, d, e | 0.051 ± 0.040 a | 0.284 ± 0.052 a, b, c | 0.046 ± 0.005 a | 0.085 ± 0.004 a | 0.027 ± 0.002 a |
| L(+) | 4.237 ± 0.183 g | 0.642 ± 0.084 b, c, d, e | 7.248 ± 0.304 j | 0.074 ± 0.011 a | 0.061 ± 0.005 a | 0.054 ± 0.004 a | |
| Total | 5.035 ± 0.103 C | 0.692 ± 0.44 B | 7.531 ± 0.252 D | 0.120 ± 0.016 A | 0.146 ± 0.001 A | 0.081 ± 0.008 A | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Śliżewska, K.; Chlebicz-Wójcik, A. The In Vitro Analysis of Prebiotics to Be Used as a Component of a Synbiotic Preparation. Nutrients 2020, 12, 1272. https://doi.org/10.3390/nu12051272
Śliżewska K, Chlebicz-Wójcik A. The In Vitro Analysis of Prebiotics to Be Used as a Component of a Synbiotic Preparation. Nutrients. 2020; 12(5):1272. https://doi.org/10.3390/nu12051272
Chicago/Turabian StyleŚliżewska, Katarzyna, and Agnieszka Chlebicz-Wójcik. 2020. "The In Vitro Analysis of Prebiotics to Be Used as a Component of a Synbiotic Preparation" Nutrients 12, no. 5: 1272. https://doi.org/10.3390/nu12051272
APA StyleŚliżewska, K., & Chlebicz-Wójcik, A. (2020). The In Vitro Analysis of Prebiotics to Be Used as a Component of a Synbiotic Preparation. Nutrients, 12(5), 1272. https://doi.org/10.3390/nu12051272






