The Relationship between Vitamin K and Osteoarthritis: A Review of Current Evidence
Abstract
1. Introduction
2. The Relationship Ship between Vitamin K Status and OA
2.1. Case-Control Studies
2.2. Cross-Sectional Studies
2.3. Prospective Studies
2.4. Clinical Trial
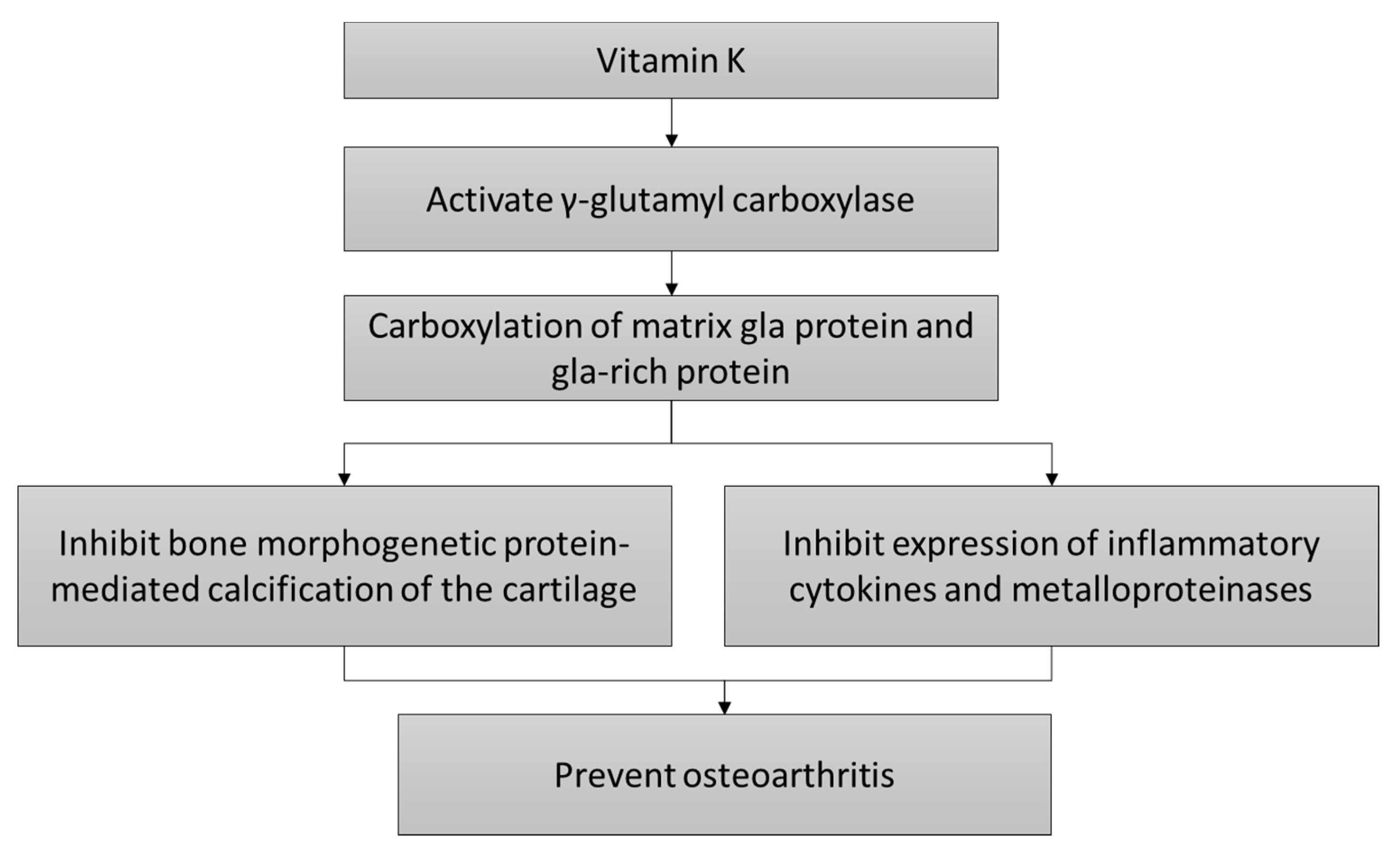
3. Mechanism of Action
4. Perspectives
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Malemud, C.J. Biologic basis of osteoarthritis: State of the evidence. Curr. Opin. Rheumatol. 2015, 27, 289–294. [Google Scholar] [CrossRef]
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef]
- Vina, E.R.; Kwoh, C.K. Epidemiology of osteoarthritis: Literature update. Curr. Opin. Rheumatol. 2018, 30, 160–167. [Google Scholar] [CrossRef]
- Palazzo, C.; Nguyen, C.; Lefevre-Colau, M.-M.; Rannou, F.; Poiraudeau, S. Risk factors and burden of osteoarthritis. Ann. Phys. Rehabil. Med. 2016, 59, 134–138. [Google Scholar] [CrossRef]
- Taruc-Uy, R.L.; Lynch, S.A. Diagnosis and treatment of osteoarthritis. Prim. Care 2013, 40, 821–836. [Google Scholar] [CrossRef]
- Al-Saadi, H.M.; Pang, K.-L.; Ima-Nirwana, S.; Chin, K.-Y. Multifaceted Protective Role of Glucosamine against Osteoarthritis: Review of Its Molecular Mechanisms. Sci. Pharm. 2019, 87, 34. [Google Scholar] [CrossRef]
- Liu, X.; Machado, G.C.; Eyles, J.P.; Ravi, V.; Hunter, D.J. Dietary supplements for treating osteoarthritis: A systematic review and meta-analysis. Br. J. Sports Med. 2018, 52, 167–175. [Google Scholar] [CrossRef]
- Chin, K.Y. The spice for joint inflammation: Anti-inflammatory role of curcumin in treating osteoarthritis. Drug Des. Devel. Ther. 2016, 10, 3029–3042. [Google Scholar] [CrossRef]
- Chin, K.-Y.; Wong, S.K.; Japar Sidik, F.Z.; Abdul Hamid, J.; Abas, N.H.; Mohd Ramli, E.S.; Afian Mokhtar, S.; Rajalingham, S.; Ima Nirwana, S. The Effects of Annatto Tocotrienol Supplementation on Cartilage and Subchondral Bone in an Animal Model of Osteoarthritis Induced by Monosodium Iodoacetate. Int. J. Environ. Res. Public Health 2019, 16, 2897. [Google Scholar] [CrossRef]
- Chin, K.-Y.; Ima-Nirwana, S. The Role of Vitamin E in Preventing and Treating Osteoarthritis–A Review of the Current Evidence. Front. Pharmacol. 2018, 9, 946. [Google Scholar] [CrossRef]
- Chin, K.Y.; Pang, K.L. Therapeutic Effects of Olive and Its Derivatives on Osteoarthritis: From Bench to Bedside. Nutrients 2017, 9, 60. [Google Scholar] [CrossRef]
- Manoy, P.; Yuktanandana, P.; Tanavalee, A.; Anomasiri, W.; Ngarmukos, S.; Tanpowpong, T.; Honsawek, S. Vitamin D Supplementation Improves Quality of Life and Physical Performance in Osteoarthritis Patients. Nutrients 2017, 9, 799. [Google Scholar] [CrossRef]
- Hawellek, T.; Hubert, J.; Hischke, S.; Krause, M.; Bertrand, J.; Pap, T.; Püschel, K.; Rüther, W.; Niemeier, A. Articular cartilage calcification of the hip and knee is highly prevalent, independent of age but associated with histological osteoarthritis: Evidence for a systemic disorder. Osteoarthr. Cartil. 2016, 24, 2092–2099. [Google Scholar] [CrossRef]
- Roemhildt, M.L.; Gardner-Morse, M.G.; Morgan, C.F.; Beynnon, B.D.; Badger, G.J. Calcium phosphate particulates increase friction in the rat knee joint. Osteoarthr. Cartil. 2014, 22, 706–709. [Google Scholar] [CrossRef][Green Version]
- Roemhildt, M.L.; Beynnon, B.D.; Gardner-Morse, M. Mineralization of articular cartilage in the Sprague-Dawley rat: Characterization and mechanical analysis. Osteoarthr. Cartil. 2012, 20, 796–800. [Google Scholar] [CrossRef][Green Version]
- McCarthy, G.M.; Westfall, P.R.; Masuda, I.; Christopherson, P.A.; Cheung, H.S.; Mitchell, P.G. Basic calcium phosphate crystals activate human osteoarthritic synovial fibroblasts and induce matrix metalloproteinase-13 (collagenase-3) in adult porcine articular chondrocytes. Ann. Rheum. Dis. 2001, 60, 399–406. [Google Scholar] [CrossRef]
- El-Maadawy, S.; Kaartinen, M.T.; Schinke, T.; Murshed, M.; Karsenty, G.; McKee, M.D. Cartilage formation and calcification in arteries of mice lacking matrix Gla protein. Connect. Tissue Res. 2003, 44 (Suppl. 1), 272–278. [Google Scholar] [CrossRef]
- Yagami, K.; Suh, J.Y.; Enomoto-Iwamoto, M.; Koyama, E.; Abrams, W.R.; Shapiro, I.M.; Pacifici, M.; Iwamoto, M. Matrix GLA protein is a developmental regulator of chondrocyte mineralization and, when constitutively expressed, blocks endochondral and intramembranous ossification in the limb. J. Cell Biol. 1999, 147, 1097–1108. [Google Scholar] [CrossRef]
- Xue, W.; Wallin, R.; Olmsted-Davis, E.A.; Borrás, T. Matrix GLA protein function in human trabecular meshwork cells: Inhibition of BMP2-induced calcification process. Investig. Ophthalmol. Vis. Sci. 2006, 47, 997–1007. [Google Scholar] [CrossRef]
- Misra, D.; Booth, S.L.; Crosier, M.D.; Ordovas, J.M.; Felson, D.T.; Neogi, T. Matrix Gla protein polymorphism, but not concentrations, is associated with radiographic hand osteoarthritis. J. Rheumatol. 2011, 38, 1960–1965. [Google Scholar] [CrossRef]
- Theuwissen, E.; Smit, E.; Vermeer, C. The role of vitamin K in soft-tissue calcification. Adv. Nutr. 2012, 3, 166–173. [Google Scholar] [CrossRef]
- Shearer, M.J.; Fu, X.; Booth, S.L. Vitamin K nutrition, metabolism, and requirements: Current concepts and future research. Adv. Nutr. 2012, 3, 182–195. [Google Scholar] [CrossRef]
- Office of Dietary Supplements-National Institute of Health. Vitamin K Fact Sheets for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/vitaminK-HealthProfessional/#en3 (accessed on 9 February 2020).
- Roberts, N.B.; Holding, J.D.; Walsh, H.P.; Klenerman, L.; Helliwell, T.; King, D.; Shearer, M. Serial changes in serum vitamin K1, triglyceride, cholesterol, osteocalcin and 25-hydroxyvitamin D3 in patients after hip replacement for fractured neck of femur or osteoarthritis. Eur. J. Clin. Investig. 1996, 26, 24–29. [Google Scholar] [CrossRef]
- El-Brashy, A.E.W.S.; El-Tanawy, R.M.; Hassan, W.A.; Shaban, H.M.; Bhnasawy, M.M. Potential role of vitamin K in radiological progression of early knee osteoarthritis patients. Egypt. Rheumatol. 2016, 38, 217–223. [Google Scholar] [CrossRef][Green Version]
- Rafael, M.S.; Cavaco, S.; Viegas, C.S.; Santos, S.; Ramos, A.; Willems, B.A.; Herfs, M.; Theuwissen, E.; Vermeer, C.; Simes, D.C. Insights into the association of Gla-rich protein and osteoarthritis, novel splice variants and gamma-carboxylation status. Mol. Nutr. Food Res. 2014, 58, 1636–1646. [Google Scholar] [CrossRef]
- Bing, W.; Feng, L. Attenuate Synovial Fluid Uncarboxylated Matrix Gla-Protein (ucMGP) Concentrations Are Linked with Radiographic Progression in Knee Psteoarthritis. Adv. Clin. Exp. Med. 2015, 24, 1013–1017. [Google Scholar] [CrossRef]
- Silaghi, C.N.; Fodor, D.; Cristea, V.; Craciun, A.M. Synovial and serum levels of uncarboxylated matrix Gla-protein (ucMGP) in patients with arthritis. Clin. Chem. Lab. Med. 2011, 50, 125–128. [Google Scholar] [CrossRef]
- Neogi, T.; Booth, S.L.; Zhang, Y.Q.; Jacques, P.F.; Terkeltaub, R.; Aliabadi, P.; Felson, D.T. Low vitamin K status is associated with osteoarthritis in the hand and knee. Arthritis Rheum 2006, 54, 1255–1261. [Google Scholar] [CrossRef]
- Ishii, Y.; Noguchi, H.; Takeda, M.; Sato, J.; Yamamoto, N.; Wakabayashi, H.; Kanda, J.; Toyabe, S. Distribution of vitamin K2 in subchondral bone in osteoarthritic knee joints. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 1813–1818. [Google Scholar] [CrossRef]
- Oka, H.; Akune, T.; Muraki, S.; En-yo, Y.; Yoshida, M.; Saika, A.; Sasaki, S.; Nakamura, K.; Kawaguchi, H.; Yoshimura, N. Association of low dietary vitamin K intake with radiographic knee osteoarthritis in the Japanese elderly population: Dietary survey in a population-based cohort of the ROAD study. J. Orthop. Sci. 2009, 14, 687–692. [Google Scholar] [CrossRef]
- Muraki, S.; Akune, T.; En-Yo, Y.; Yoshida, M.; Tanaka, S.; Kawaguchi, H.; Nakamura, K.; Oka, H.; Yoshimura, N. Association of dietary intake with joint space narrowing and osteophytosis at the knee in Japanese men and women: The ROAD study. Mod. Rheumatol. 2014, 24, 236–242. [Google Scholar] [CrossRef]
- Naito, K.; Watari, T.; Obayashi, O.; Katsube, S.; Nagaoka, I.; Kaneko, K. Relationship between serum undercarboxylated osteocalcin and hyaluronan levels in patients with bilateral knee osteoarthritis. Int. J. Mol. Med. 2012, 29, 756–760. [Google Scholar] [CrossRef][Green Version]
- Shea, M.K.; Kritchevsky, S.B.; Hsu, F.C.; Nevitt, M.; Booth, S.L.; Kwoh, C.K.; McAlindon, T.E.; Vermeer, C.; Drummen, N.; Harris, T.B.; et al. The association between vitamin K status and knee osteoarthritis features in older adults: The Health, Aging and Body Composition Study. Osteoarthr. Cartil. 2015, 23, 370–378. [Google Scholar] [CrossRef]
- Shea, M.K.; Kritchevsky, S.B.; Loeser, R.F.; Booth, S.L. Vitamin K status and mobility limitation and disability in older adults: The Health, Aging, and Body Composition Study. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 75, 792–797. [Google Scholar] [CrossRef]
- den Hollander, W.; Boer, C.G.; Hart, D.J.; Yau, M.S.; Ramos, Y.F.M.; Metrustry, S.; Broer, L.; Deelen, J.; Cupples, L.A.; Rivadeneira, F.; et al. Genome-wide association and functional studies identify a role for matrix Gla protein in osteoarthritis of the hand. Ann. Rheum. Dis. 2017, 76, 2046–2053. [Google Scholar] [CrossRef]
- Shea, M.K.; Loeser, R.F.; McAlindon, T.E.; Houston, D.K.; Kritchevsky, S.B.; Booth, S.L. Association of Vitamin K Status Combined With Vitamin D Status and Lower-Extremity Function: A Prospective Analysis of Two Knee Osteoarthritis Cohorts. Arthritis Care Res. 2018, 70, 1150–1159. [Google Scholar] [CrossRef]
- Misra, D.; Booth, S.L.; Tolstykh, I.; Felson, D.T.; Nevitt, M.C.; Lewis, C.E.; Torner, J.; Neogi, T. Vitamin K deficiency is associated with incident knee osteoarthritis. Am. J. Med. 2013, 126, 243–248. [Google Scholar] [CrossRef]
- Neogi, T.; Felson, D.T.; Sarno, R.; Booth, S.L. Vitamin K in hand osteoarthritis: Results from a randomised clinical trial. Ann. Rheum. Dis. 2008, 67, 1570–1573. [Google Scholar] [CrossRef]
- Ea, H.-K.; Nguyen, C.; Bazin, D.; Bianchi, A.; Guicheux, J.; Reboul, P.; Daudon, M.; Lioté, F. Articular cartilage calcification in osteoarthritis: Insights into crystal-induced stress. Arthritis Rheum. 2011, 63, 10–18. [Google Scholar] [CrossRef]
- Wallin, R.; Schurgers, L.J.; Loeser, R.F. Biosynthesis of the vitamin K-dependent matrix Gla protein (MGP) in chondrocytes: A fetuin-MGP protein complex is assembled in vesicles shed from normal but not from osteoarthritic chondrocytes. Osteoarthr. Cartil. 2010, 18, 1096–1103. [Google Scholar] [CrossRef]
- Yao, Y.; Zebboudj, A.F.; Shao, E.; Perez, M.; Boström, K. Regulation of bone morphogenetic protein-4 by matrix GLA protein in vascular endothelial cells involves activin-like kinase receptor 1. J. Biol. Chem. 2006, 281, 33921–33930. [Google Scholar] [CrossRef]
- Zebboudj, A.F.; Imura, M.; Boström, K. Matrix GLA protein, a regulatory protein for bone morphogenetic protein-2. J. Biol. Chem. 2002, 277, 4388–4394. [Google Scholar] [CrossRef]
- Fu, X.; Wu, J.; Cheng, X.; Deng, R.; Wang, W.; Wang, X.; Wu, T. Effects of gamma-glutamyl carboxylase gene overexpression on the differentiation of chondrocytes from osteoarthritis rabbits. Int. J. Clin. Exp. Med. 2017, 10, 9096–9102. [Google Scholar]
- Viegas, C.S.B.; Cavaco, S.; Neves, P.L.; Ferreira, A.; João, A.; Williamson, M.K.; Price, P.A.; Cancela, M.L.; Simes, D.C. Gla-rich protein is a novel vitamin K-dependent protein present in serum that accumulates at sites of pathological calcifications. Am. J. Pathol. 2009, 175, 2288–2298. [Google Scholar] [CrossRef]
- Cavaco, S.; Viegas, C.S.B.; Rafael, M.S.; Ramos, A.; Magalhães, J.; Blanco, F.J.; Vermeer, C.; Simes, D.C. Gla-rich protein is involved in the cross-talk between calcification and inflammation in osteoarthritis. Cell Mol. Life Sci. 2016, 73, 1051–1065. [Google Scholar] [CrossRef]
- Viegas, C.S.B.; Costa, R.M.; Santos, L.; Videira, P.A.; Silva, Z.; Araújo, N.; Macedo, A.L.; Matos, A.P.; Vermeer, C.; Simes, D.C. Gla-rich protein function as an anti-inflammatory agent in monocytes/macrophages: Implications for calcification-related chronic inflammatory diseases. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Riphagen, I.J.; Keyzer, C.A.; Drummen, N.E.A.; de Borst, M.H.; Beulens, J.W.J.; Gansevoort, R.T.; Geleijnse, J.M.; Muskiet, F.A.J.; Navis, G.; Visser, S.T.; et al. Prevalence and Effects of Functional Vitamin K Insufficiency: The PREVEND Study. Nutrients 2017, 9, 1334. [Google Scholar] [CrossRef]
- McNinch, A. Vitamin K deficiency bleeding: Early history and recent trends in the United Kingdom. Early Hum. Dev. 2010, 86 (Suppl. 1), 63–65. [Google Scholar] [CrossRef]
- Institute of Medicine (US) Panel on Micronutrients. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Wu, S.; Chen, X.; Jin, D.-Y.; Stafford, D.W.; Pedersen, L.G.; Tie, J.-K. Warfarin and vitamin K epoxide reductase: A molecular accounting for observed inhibition. Blood 2018, 132, 647–657. [Google Scholar] [CrossRef]
- Violi, F.; Lip, G.Y.; Pignatelli, P.; Pastori, D. Interaction Between Dietary Vitamin K Intake and Anticoagulation by Vitamin K Antagonists: Is It Really True?: A Systematic Review. Medicine 2016, 95, e2895. [Google Scholar] [CrossRef]
- Schurks, M.; Glynn, R.J.; Rist, P.M.; Tzourio, C.; Kurth, T. Effects of vitamin E on stroke subtypes: Meta-analysis of randomised controlled trials. BMJ 2010, 341, c5702. [Google Scholar] [CrossRef]
- Hanzawa, F.; Sakuma, E.; Nomura, S.; Uchida, T.; Oda, H.; Ikeda, S. Excess alpha-tocopherol decreases extrahepatic phylloquinone in phylloquinone-fed rats but not menaquinone-4 in menaquinone-4-fed rats. Mol. Nutr. Food Res. 2014, 58, 1601–1609. [Google Scholar] [CrossRef]
- Booth, S.L.; Golly, I.; Sacheck, J.M.; Roubenoff, R.; Dallal, G.E.; Hamada, K.; Blumberg, J.B. Effect of vitamin E supplementation on vitamin K status in adults with normal coagulation status. Am. J. Clin. Nutr. 2004, 80, 143–148. [Google Scholar] [CrossRef]
- Palermo, A.; Tuccinardi, D.; D’Onofrio, L.; Watanabe, M.; Maggi, D.; Maurizi, A.R.; Greto, V.; Buzzetti, R.; Napoli, N.; Pozzilli, P.; et al. Vitamin K and osteoporosis: Myth or reality? Metabolism 2017, 70, 57–71. [Google Scholar] [CrossRef]
| Reference | Study Design | Outcomes |
|---|---|---|
| Roberts et al. 1996 [24] | Patients with femoral neck fracture (n = 13): 10 women, median age 80 years; 3 men, median age 85 years. OA patients (n = 16): 10 women, median age 71 years; 6 men, median age 64 years. Healthy volunteers (n = 25): 16 women and 9 men, median age 77 years. Blood was sampled from the patients before and after surgery. Serum vitamin K1 measured by HPLC. | Vitamin K1: ↓ in patients with femoral neck fracture and OA vs. normal controls; ↓ after surgery. Carboxylated and undercarboxylated osteocalcin: ~ between case and control. |
| Silaghi et al. 2012 [28] | Non-inflammatory arthritis group: n = 17, median age 55 years, patients with OA or chondrocalcinosis Inflammatory arthritis group: n = 9, median age 55 years, patients with juvenile idiopathic arthritis, ankylosing spondylitis Control: n = 30, median age 42 years, no radiographic evidence of arthritis Uncarboxylated MGP measured by ELISA | Serum uncarboxylated MGP: ↑ in the controls vs. patients with arthritis; ↑ in the non-inflammatory arthritis group vs. inflammatory arthritis group. Synovial uncarboxylated MGP: ↑ in the inflammatory arthritis group vs. non-inflammatory arthritis group Synovial/serum uncarboxylated MGP ratio: ↑ in the inflammatory group vs. non-inflammatory group. |
| Rafael et al. 2014 [26] | OA samples from patients undergoing total knee replacement surgery. Control samples from the autopsy of subjects with no history of joint disease. Immunostaining technique. | Undercarboxylated GRP: ↑ in the OA cartilage matrix and synovial membrane vs. to carboxylated one. Carboxylated MGP: ↑ in the control cartilage matrix and synovial membrane vs. undercarboxylated ones. |
| Bing and Feng 2015 [27] | Case: 178 knee OA patients, aged 62.8 ± 7.4 years, 114 women: 64 men Control: 160 healthy outpatients in a hospital, aged 63.2 ± 8 years, 94 women: 66 men OA assessment: knee radiographs with KL criteria (at least ≥ 2 at one knee to be selected as cases) Uncarboxylated MGP measured using ELISA | Serum uncarboxylated MGP: ↓ in the OA patients vs. control. Synovial fluid uncarboxylated MGP level correlated negatively with the disease severity among the OA patients. |
| El-Brashy et al. 2016 [25] | Case: 40 knee OA patients (36 women: 10 men), age 50.4 ± 4.9 years, KL grade 2 or less Control 20 healthy individuals (85 women: 15 men), age 48.9 ± 4.6 years, Plasma phylloquinone level was measured using ELISA OA assessed using pain scale, The Western Ontario McMaster Scale (WOMAC), Thomas Grading Score and musculoskeletal ultrasound | Plasma vitamin K: ↓ in knee OA patients vs. control WOMAC score: ↑ in patients with vitamin K deficiency vs those who were sufficient. A significant positive correlation was found between plasma vitamin K level and medial cartilage thickness. |
| Reference | Study Design | Findings |
|---|---|---|
| Neogi et al. 2006 [29] | Framingham Offspring Study. Subjects: n = 672, 53% women. Mean age 66 years OA assessed using Framingham OA atlas. Phylloquinone level assessed using HPLC. | ↓ hand OA and large osteophyte for subjects with the Q4 (1.81–21.5 nmol/L) of phylloquinone level vs. Q2 (0.59–1.02 nmol/L) & Q1 (0.05–0.58 nmol/L). ↓ joint space narrowing for hand OA in Q4 vs. Q1. A threshold effect of 1 nmol/L was observed, in which ↓ OA prevalence after that level. Knee joint OA not associated with phylloquinone level |
| Oka et al. 2009 [31] | Research on Osteoarthritis Against Disease (ROAD) study. Subjects: 719 Japanese subjects, 62.4% women. Mean age 72.1 ± 6.3 years for men, 72.0 ± 7 yrs for women. OA assessed through radiographs using KL grade. Vitamin K intake assessed through Brief Dietary History Questionnaire | Vitamin K intake was associated with ↓ OA grade ≥ 2 (OR 0.75 95% CI 0.63–0.89) or ≥ 3 (OR 0.67 95% CI 0.53–0.84). Based on sex, the relationship of OA grade ≥ 2 remained for men (OR 0.76 95% CI 0.59–0.95) and women (OR 0.74 95% CI 0.58–0.96). For women, the relationship of OA grade ≥ 3 (OR 0.61 95% CI 0.45–0.81) was associated with vitamin K intake. |
| Misra et al. 2011 [20] | 376 subjects participated in a randomized controlled trial on the effects of vitamin K supplementation on bone and vascular health. 59% women, age 71 ± 5.5 years. | No association between serum MGP levels and hand OA. Homozygote rs1800802 minor allele (GG) was associated with a lower risk of hand OA vs. having 1 major allele at this locus (OR 0.56, 95% CI 0.32–0.99). The same allele was associated with a lower risk of joint space narrowing (OR 0.25, 95% CI 0.11–0.58) and osteophyte formation (OR 0.31, 95% CI 0.17–0.58). Homozygote rs4236 major allele (TT) was associated with a lower risk of joint space narrowing compared to genotypes with at least one minor allele (OR 0.52, 95% CI 0.35–0.78). |
| Naito et al. 2012 [33] | 25 patients with KL grade 3/4 for bilateral knee OA (age: 76 + 7.8 years, BMI; 24.9 + 4.7) were recruited. Serum undercarboxylated osteocalcin was measured by ELISA. | Serum undercarboxylated osteocalcin: marginal ↑ increase in patients with KL grade 6/7/8. Positive correlation between undercarboxylated osteocalcin and bone metabolism markers (serum N-terminal telopeptide and bone alkaline phosphatase) and synovitis marker (serum hyaluronan), but not with cartilage metabolism markers (urinary CTX-II and C-terminal type II procollagen peptide). |
| Ishii et al. 2013 [30] | 58 bones from patients (13 men; 45 women) undergoing (aged 73 ± 8 years) total knee arthroplasty (all grade 4 OA) were collected. Vitamin K2 in the medial and lateral femoral and tibial condyles were compared. Vitamin K2 was analysed using HPLC. | ↑ Vitamin K2 in the lateral femoral and tibial condyles than the medial condyles; ↑ in the femoral lateral/medial condyles than the tibial lateral/medial condyles but only the lateral parts were significant. The difference in age and sex was not significant. |
| Muraki et al. 2014 [32] | Research on Osteoarthritis/Osteoporosis Against Disability Study (ROAD) Subjects: 827 subjects (305 men, 522 women), mean age 69.2 ± 9.3 years. A knee OA computer-aided diagnostic system was used to analyse minimum joint space width (mJSW) and osteophyte area. Vitamin K intake (previous month) assessed via Brief Dietary History Questionnaire | Vitamin K intake was significantly associated with mJSW but not osteophyte area. In women, vitamin K, B1, B2, B6, and C intakes were associated with mJSW, after adjusting for age, BMI and total energy. In women, vitamin E, B1, B2, niacin, and B6 were associated with osteophyte area. |
| Shea et al. 2015 [34] | 791 community-dwelling elderly (mean age 74+3 years, BMI 27.7 + 4.8, 67% women) from Healthy, Aging and Body Composition Study (Health ABC). 40% African Americans and 60% Caucasians. Median follow up period of 37 months. OA assessed via Magnetic resonance imaging on both knees Plasma phylloquinone measured using reversed-phase HPLC Vitamin K intake assessed via The Health ABC food frequency questionnaire MGP assessed via ELISA | ↑ plasma dephosphorylated uncarboxylated MGP (highest vs lowest quartiles) was associated with ↑ risk of meniscus damage (OR: 1.6, 95%CI 1.1–2.3), osteophytes (OR: 1.7, 95%CI 1.1–2.5), bone marrow lesions (OR: 1.9, 95%CI 1.3–2.8) and subarticular cyst (OR: 1.5, 95%CI 1.0–2.1). African Americans in the lowest uncarboxylated MGP quartile had the highest risk of having articular damage. Caucasians with non-detectable plasma vitamin K were more likely to have meniscal damage. |
| Shea et al. 2019 [35] | Health ABC: n = 1323 (635 men) for plasma phylloquinone HPLC measurement; n = 716 for uncarboxylated MGP measurement. Age 74.2 ± 2.8 years, 40% black/60% white. Mobility limitation defined as 2 consecutive semiannual reports of difficulties either walking 1/4 miles or climbing 10 steps. Mobility disability defined as 2 consecutive semiannual reports of huge difficulties/inability to walk 1/4 miles. | Subjects with circulating vitamin K level < 0.5 nmol/L had more risk for mobility limitation (OR: 1.49 (96% CI 1.04–2.13)) and disability (OR: 1.95 (96% CI 1.08–3.54)) compared to those with > 1.0 nmol/L uncarboxylated MGP was not associated with both variables. |
| Reference | Study Design | Findings |
|---|---|---|
| Misra et al. 2013 [38] | 1180 subjects from Multicenter Osteoarthritis (MOST) study, 62% women, mean age 62 + 8 years. Knee radiographs or MRI scan obtained at baseline and 30 months later. Plasma phylloquinone measured at baseline using HPLC. New incidence of OA referred to KL grade 0/1 to > 2, cartilage lesion based on Whole-Organ Magnetic Resonance Imaging Score from 0 > 1 and osteophytes from 0/1 to >2. | Subclinical vitamin K deficiency (9.2% of the overall population) was associated with incident radiographic knee OA (RR: 1.56 95% CI 1.08–2.25) cartilage lesion (RR: 2.39 95% CI 1.05–5.40) but not with osteophytes (RR 2.35 95%CI 0.54–10.13). It was also associated with OA at one or both knees (RR 1.33 95% CI 1.01–1.75 and RR 2.12 95% CI 1.06–4.24). |
| Shea et al. 2015 [34] | Cross-sectional and longitudinal studies. 791 community-dwelling elderly (mean age 74 + 3 years, BMI 27.7 + 4.8, 67% W) From Healthy, Aging and Body Composition Study (Health ABC). 40% African Americans and 60% Caucasians. Median follow up period of 37 months. OA assessed using magnetic resonance imaging on both knees. Vitamin K assessed using plasma phylloquinone measured using reversed-phase HPLC. Dietary intake assessed using the Health ABC food frequency questionnaire. MGP assessed using ELISA. | Non-detectable plasma vitamin K level (<0.2 nmol/L) at baseline predicted articular cartilage (OR: 1.7, 95%CI 1.0–3.0) and meniscus damage (OR 2.6, 95%CI 1.3–5.2) after three years, compared to those with sufficient vitamin K. Plasma uncarboxylated MGP did not predict knee pain at baseline. |
| El-Brashy et al. 2016 [25] | A case-control study with 12 months longitudinal follow up. Case: 40 knee OA patients (36 women: 10 men), age 50.4 ± 4.9 years, KL grade 2 or less Control 20 healthy individuals (85 women: 15 men), age 48.9 ± 4.6 years Plasma phylloquinone level was measured using ELISA. OA assessed using pain scale, The Western Ontario McMaster Scale (WOMAC), Thomas Grading Score and musculoskeletal ultrasound. | ↑ WOMAC score and pain scale of patients with deficiency vs. those with sufficient level. ↓ cartilage thickness at medial, lateral and sulcus condyles in patients with vitamin K deficiency. The same observation was obtained for Thomas score at the medial compartment and total score. Vitamin K deficiency (0.5 nmol/L) among the patients was associated with radiographic OA progression (RR: 2.08, 95% CI 1.30–3.32). The best cut-off for vitamin K on radiographic OA progression was 1.74 nmol/L, on ultrasound was 1.28 nmol/L. |
| Shea et al. 2018 [37] | Two prospective cohort studies: Health, ABC and Osteoarthritis Initiative (OAI). In Health ABC: n = 1069, 60% women, aged 75 ± 3 years. Plasma phylloquinone and 25-hydroxyvitamin D were determined. Physical function: short physical performance battery and usual 20-meter gait speed. In OAI: n = 4475, 58% women, aged 61 ± 9 yrs. Vitamin K and D intakes were determined using the Block Brief 2000 food frequency questionnaire. Physical function: 20-meter gait speed and chair stand completion time. Follow-up period for both: 4–5 years. | In Health ABC, adequate circulating K (≥ 1 nmol/L) and D (≥ 50 nmol/L) predicted better physical performance battery scores and gait speed on follow up. Changes in physical performance score were not associated with vitamin D status. Both variables showed no correlation with gait speed. In OAI, adequate vitamin K (≥ 90 µg/day for women or 120 µg/day for men) and D intake (≥ 600 IU for age < 70 years, ≥ 800 IU for age ≥ 70 years) were associated with overall 20-m gait speed and chair stand completion time on follow up, but not 400-meter walk time. |
| Shea et al. 2019 [35] | Health ABC. n = 1323 (635 men) for plasma phylloquinone HPLC measurement, n = 716 for uncarboxylated MGP measurement. Age 74.2 ± 2.8 years, 40% black/60% white. Mobility limitation defined as 2 consecutive semiannual reports of difficulties either walking 1/4 miles or climbing 10 steps. Mobility disability defined as 2 consecutive semiannual reports of huge difficulties/inability to walk 1/4 miles. Median follow-up: 6.4 (8.6) years for limitation and 10.3 (5.8) years for disability. | Subjects with circulating vitamin K level <0.5 nmol/L were more likely to develop mobility limitation (OR: 1.27 (96% CI 1.05–1.53)) and disability (OR: 1.34 (96% CI 1.01–1.76)) compared to those with 1.0 nmol/L. After adjustment for knee pain, the association with disability was attenuated significantly (OR: 1.26 (96% CI 0.96–1.67)). Plasma uncarboxylated MGP was not associated with mobility limitation but was associated with incident mobility disability non-linearly (n-shape). |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chin, K.-Y. The Relationship between Vitamin K and Osteoarthritis: A Review of Current Evidence. Nutrients 2020, 12, 1208. https://doi.org/10.3390/nu12051208
Chin K-Y. The Relationship between Vitamin K and Osteoarthritis: A Review of Current Evidence. Nutrients. 2020; 12(5):1208. https://doi.org/10.3390/nu12051208
Chicago/Turabian StyleChin, Kok-Yong. 2020. "The Relationship between Vitamin K and Osteoarthritis: A Review of Current Evidence" Nutrients 12, no. 5: 1208. https://doi.org/10.3390/nu12051208
APA StyleChin, K.-Y. (2020). The Relationship between Vitamin K and Osteoarthritis: A Review of Current Evidence. Nutrients, 12(5), 1208. https://doi.org/10.3390/nu12051208





