Age-related Smell and Taste Impairments and Vitamin D Associations in the U.S. Adults National Health and Nutrition Examination Survey
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
Study Sample
2.2. Study Variables
2.2.1. Main Exposure
2.2.2. Main Outcomes
Smell and Taste Assessments
2.2.3. Covariates
2.3. Statistical Analysis
3. Results
3.1. Study Sample
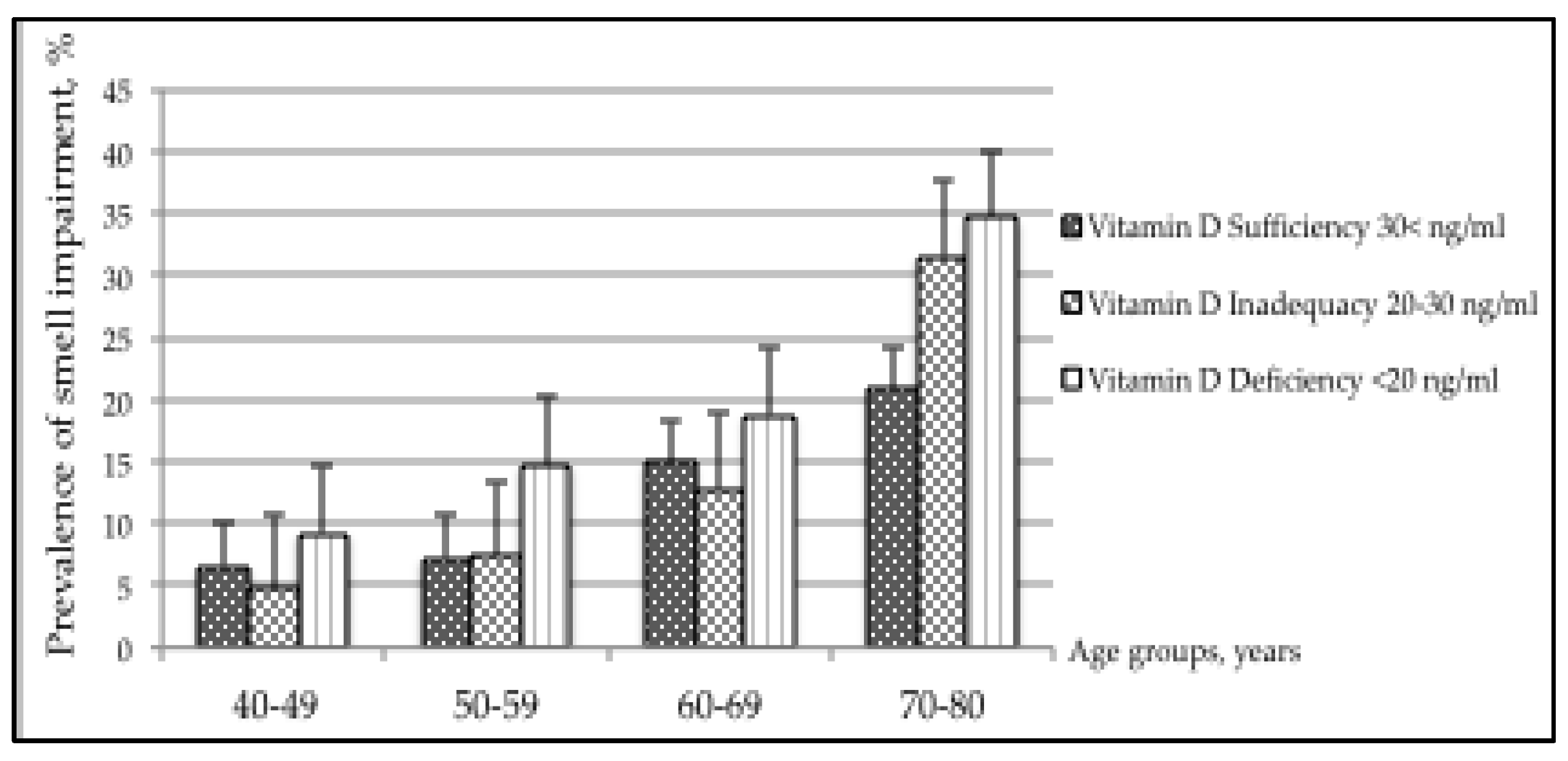
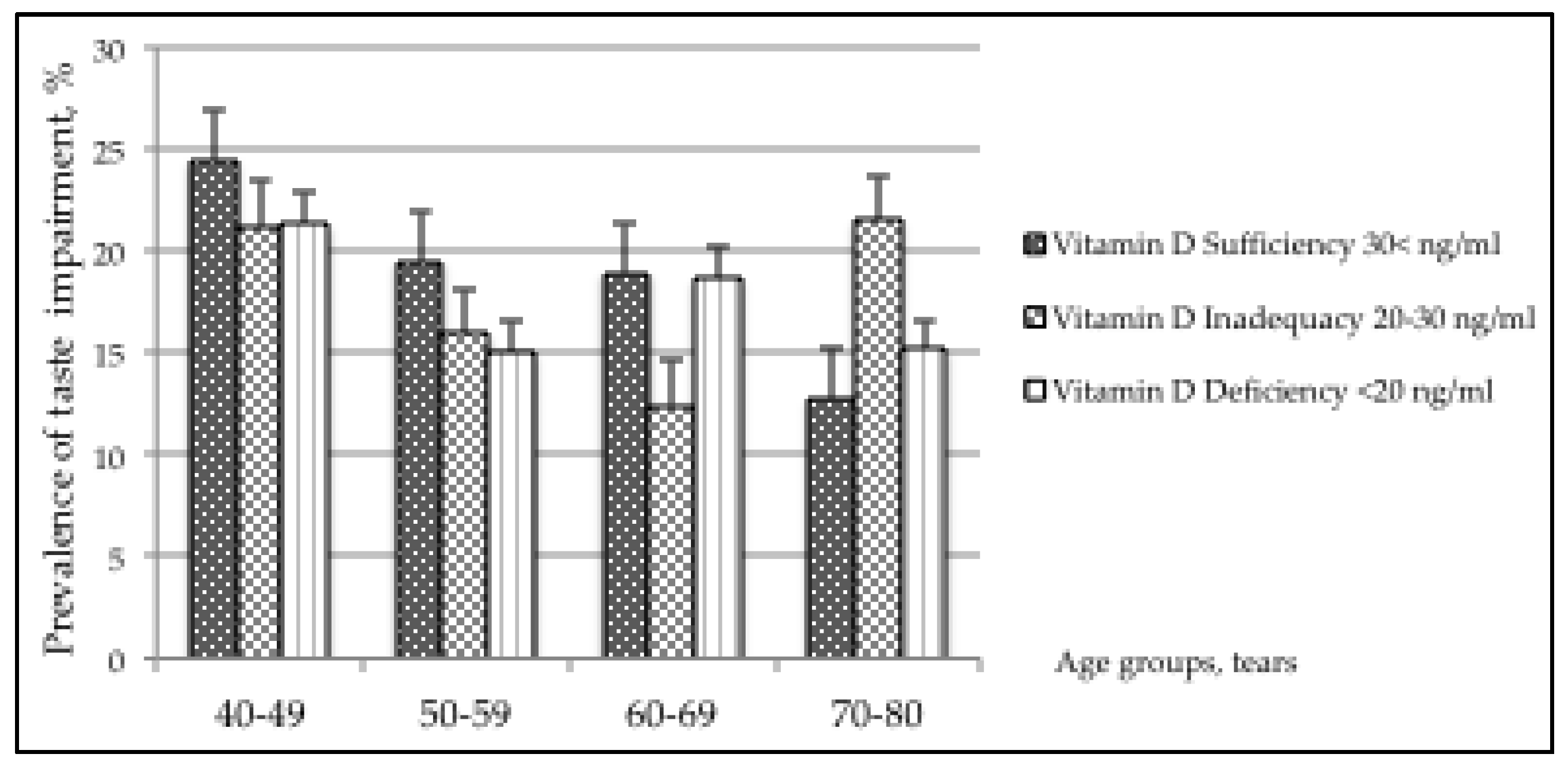
3.2. Smell and Taste Impairments across Age Groups
3.3. Smell Impairment and the Association with Vitamin D
3.4. Taste Impairment and the Association with Vitamin D
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Smoliner, C.; Fischedick, A.; Sieber, C.C.; Wirth, R. Olfactory function and malnutrition in geriatric patients. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2013, 68, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Doty, R.L. Age-Related Deficits in Taste and Smell. Otolaryngol. Clin. North Am. 2018, 51, 815–825. [Google Scholar] [CrossRef]
- Attems, J.; Walker, L.; Jellinger, K.A. Olfaction and Aging: A Mini Review. Gerontology 2015, 61, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Zong, G.; Doty, R.L.; Sun, Q. Prevalence and risk factors of taste and smell impairment in a nationwide representative sample of the US population: A cross-sectional study. BMJ Open 2016, 6, e013246. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Deafness and Other Communication Disorders (NIDCD). Taste and Smell. 2019. Available online: https://www.nidcd.nih.gov/health/taste-smell (accessed on 29 February 2020).
- Babizhayev, M.A.; Deyev, A.I.; Yegorov, Y.E. Olfactory dysfunction and cognitive impairment in age-related neurodegeneration: Prevalence related to patient selection, diagnostic criteria and therapeutic treatment of aged clients receiving clinical neurology and community-based care. Curr. Clin. Pharmacol. 2011, 6, 236–259. [Google Scholar] [CrossRef] [PubMed]
- Henkin, R.I. Zinc in taste function: A critical review. Biol. Trace Elem. Res. 1984, 6, 263–280. [Google Scholar] [CrossRef] [PubMed]
- Aden, E.; Carlsson, M.; Poortvliet, E.; Stenlund, H.; Linder, J.; Edstrom, M.; Forsgren, L.; Haglin, L. Dietary intake and olfactory function in patients with newly diagnosed Parkinson’s disease: A case-control study. Nutr. Neurosci. 2011, 14, 25–31. [Google Scholar] [CrossRef]
- Hummel, T.; Whitcroft, K.L.; Rueter, G.; Haehner, A. Intranasal vitamin A is beneficial in post-infectious olfactory loss. Eur. Arch. Oto-Rhino-Laryngol. 2017, 274, 2819–2825. [Google Scholar] [CrossRef]
- Henkin, R.I.; Hoetker, J.D. Deficient dietary intake of vitamin E in patients with taste and smell dysfunctions: Is vitamin E a cofactor in taste bud and olfactory epithelium apoptosis and in stem cell maturation and development? Nutrition 2003, 19, 1013–1021. [Google Scholar] [CrossRef]
- Derin, S.; Koseoglu, S.; Sahin, C.; Sahan, M. Effect of vitamin B12 deficiency on olfactory function. Int. Forum Allergy Rhinol. 2016, 6, 1051–1055. [Google Scholar] [CrossRef]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef]
- Lee, S.H.; Suh, E.; Park, K.C.; Haam, J.H.; Kim, K.; Koo, H.S.; Choi, B.H.; Won, B.Y.; Park, K.H.; Park, K.S.; et al. Association of serum 25-hydroxyvitamin D and serum total cholesterol with depressive symptoms in Korean adults: The Fifth Korean National Health and Nutrition Examination Survey (KNHANES V, 2010–2012). Public Health Nutr. 2017, 20, 1836–1843. [Google Scholar] [CrossRef]
- Kienreich, K.; Grubler, M.; Tomaschitz, A.; Schmid, J.; Verheyen, N.; Rutters, F.; Dekker, J.M.; Pilz, S. Vitamin D, arterial hypertension & cerebrovascular disease. Indian J. Med. Res. 2013, 137, 669–679. [Google Scholar]
- Michaelsson, K.; Melhus, H.; Larsson, S.C. Serum 25-Hydroxyvitamin D Concentrations and Major Depression: A Mendelian Randomization Study. Nutrients 2018, 10, 1987. [Google Scholar] [CrossRef]
- Schietzel, S.; Fischer, K.; Brugger, P.; Orav, E.J.; Renerts, K.; Gagesch, M.; Freystaetter, G.; Stahelin, H.B.; Egli, A.; Bischoff-Ferrari, H.A. Effect of 2000 IU compared with 800 IU vitamin D on cognitive performance among adults age 60 years and older: A randomized controlled trial. Am. J. Clin. Nutr. 2019, 110, 246–253. [Google Scholar] [CrossRef]
- Glaser, S.D.; Veenstra, T.D.; Jirikowski, G.F.; Prufer, K. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat olfactory system. Cell. Mol. Neurobiol. 1999, 19, 613–624. [Google Scholar] [CrossRef]
- Soh, Z.; Saito, M.; Kurita, Y.; Takiguchi, N.; Ohtake, H.; Tsuji, T. A comparison between the human sense of smell and neural activity in the olfactory bulb of rats. Chem. Senses 2014, 39, 91–105. [Google Scholar] [CrossRef]
- Homem, C.C.; Repic, M.; Knoblich, J.A. Proliferation control in neural stem and progenitor cells. Nat. Rev. Neurosci. 2015, 16, 647–659. [Google Scholar] [CrossRef]
- Shirazi, H.A.; Rasouli, J.; Ciric, B.; Rostami, A.; Zhang, G.X. 1,25-Dihydroxyvitamin D3 enhances neural stem cell proliferation and oligodendrocyte differentiation. Exp. Mol. Pathol. 2015, 98, 240–245. [Google Scholar] [CrossRef]
- Veleva, D. The Role of Vitamin D on Cell Proliferation in the Olfactory System and the Brain. Ph.D. Thesis, Doctorate Griffith University, Queensland, Australia, 2010. [Google Scholar]
- Kruse, R.A.; Cambron, J.A. A possible correlation between vitamin D deficiency and loss of smell: 2 case reports. J. Chiropr. Med. 2011, 10, 310–315. [Google Scholar] [CrossRef][Green Version]
- National Center for Health Statistics. About the National Health and Nutrition Examination Survey. 2019. Available online: http://www.cdc.gov/nchs/nhanes/about_nhanes.htm (accessed on 29 February 2020).
- National Institutes of Health. Office of Dietary Supplements, Fact Sheet for Health Professionals, Vitamin D. 2019. Available online: https://ods.od.nih.gov/factsheets/Vitamin%20D-HealthProfessional/ (accessed on 20 March 2020).
- Analytical Note for 25-Hydroxyvitamin D Data Analysis Using NHANES III (1988–1994), NHANES 2001–2006, and NHANES 2007–2010. 2015. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/VitaminD/AnalyticalNote.aspx (accessed on 20 March 2020).
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Dietary Reference Intakes for Calcium and Vitamin D; Ross, A.C., Taylor, C.L., Yaktine, A.L., Del Valle, H.B., Eds.; National Academies Press: Washington, DC, USA, 2011. Available online: https://www.ncbi.nlm.nih.gov/books/NBK56070/ (accessed on 20 March 2020).
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Norman, A.W. Vitamin D nutrition is at a crossroads. Public Health Nutr. 2011, 14, 744–745. [Google Scholar] [CrossRef] [PubMed]
- Doty, R.L.; Shaman, P.; Dann, M. Development of the University of Pennsylvania Smell Identification Test: A standardized microencapsulated test of olfactory function. Physiol. Behav. 1984, 32, 489–502. [Google Scholar] [CrossRef]
- Rawal, S.; Hoffman, H.J.; Honda, M.; Huedo-Medin, T.B.; Duffy, V.B. The Taste and Smell Protocol in the 2011-2014 US National Health and Nutrition Examination Survey (NHANES): Test-Retest Reliability and Validity Testing. Chemosens. Percept. 2015, 8, 138–148. [Google Scholar] [CrossRef]
- Analytic Notes for Dietary Interview—Total Nutrient Intakes, First Day Using NHANES 2013–2014. 2015. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2013-2014/DR1TOT_H.htm (accessed on 20 March 2020).
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression; Wiley: New York, NY, USA, 2000; pp. 91–116. [Google Scholar]
- Kim, J.E.; Oh, E.; Park, J.; Youn, J.; Kim, J.S.; Jang, W. Serum 25-hydroxyvitamin D3 level may be associated with olfactory dysfunction in de novo Parkinson’s disease. J. Clin. Neurosci. 2018, 57, 131–135. [Google Scholar] [CrossRef]
- Mayne, P.E.; Burne, T.H.J. Vitamin D in Synaptic Plasticity, Cognitive Function, and Neuropsychiatric Illness. Trends Neurosci. 2019, 42, 293–306. [Google Scholar] [CrossRef]
- DeLuca, G.C.; Kimball, S.M.; Kolasinski, J.; Ramagopalan, S.V.; Ebers, G.C. Review: The role of vitamin D in nervous system health and disease. Neuropathol. Appl. Neurobiol. 2013, 39, 458–484. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Tuohimaa, P. Neurosteroid hormone vitamin D and its utility in clinical nutrition. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 12–19. [Google Scholar] [CrossRef]
- Langub, M.C.; Herman, J.P.; Malluche, H.H.; Koszewski, N.J. Evidence of functional vitamin D receptors in rat hippocampus. Neuroscience 2001, 104, 49–56. [Google Scholar] [CrossRef]
- Obiefuna, S.; Donohoe, C. Neuroanatomy, Nucleus Gustatory, StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2020. [Google Scholar]
- Li, X.; Lui, F. Anosmia, StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2020. [Google Scholar]
- Kershaw, J.C.; Mattes, R.D. Nutrition and taste and smell dysfunction. World J. Otorhinolaryngol. Head Neck Surg. 2018, 4, 3–10. [Google Scholar] [CrossRef]
- Schiffman, S.S.; Graham, B.G. Taste and smell perception affect appetite and immunity in the elderly. Eur. J. Clin. Nutr. 2000, 54, S54–S63. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, V.F.; Vestergaard, E.T.; Hejlesen, O.; Andersson, C.U.N.; Cichosz, S.L. Prevalence of taste and smell impairment in adults with diabetes: A cross-sectional analysis of data from the National Health and Nutrition Examination Survey (NHANES). Prim. Care Diabetes 2018, 12, 453–459. [Google Scholar] [CrossRef] [PubMed]
| Taste Assessment n = 2636 | p-Value | Smell Assessment n = 2216 | p-Value | |||
|---|---|---|---|---|---|---|
| Normal | Impairment | Normal | Impairment | |||
| Vitamin D (25(OH)D) (ng/mL) | n = 2142 (81.7%) | n = 494 (18.3%) | 0.926 | n = 1866 (87.8%) | n = 350 (12.2%) | 0.123 |
| Sufficiency >30 | 848(45.4) | 188(47.0) | 723(44.9) | 127(45.7) | ||
| Inadequacy 20–30 | 770(35.4) | 178(34.1) | 673(35.8) | 124(29.5) | ||
| Deficiency <20 | 524(19.2) | 128(18.9) | 470(19.3) | 99(24.8) | ||
| Gender | 0.760 | 0.004 | ||||
| Male | 1032(48.8) | 234(48.0) | 1016(53.4) | 216(61.3) | ||
| Female | 1110(51.2) | 260(52.0) | 850(46.6) | 134(38.7) | ||
| Age groups (years) | 0.114 | <0.001 | ||||
| 40–49 | 577(27.6) | 163(35.6) | 600(33.0) | 44(16.5) | ||
| 50–59 | 557(30.2) | 131(27.9) | 500(30.7) | 67(22.1) | ||
| 60–69 | 539(24.0) | 110(21.7) | 459(22.5) | 105(28.4) | ||
| 70–80 | 469(18.2) | 90(14.8) | 307(13.8) | 134(33.0) | ||
| Race/ethnicity | 0.023 | 0.037 | ||||
| Non-Hispanic White | 1064(75.3) | 240(72.9) | 879(73.8) | 137(66.1) | ||
| Hispanic American | 428(9.8) | 99(10.0) | 391(10.4) | 77(12.5) | ||
| Non-Hispanic Black | 386(8.6) | 117 (12.5) | 364(9.5) | 85(13.1) | ||
| Other race | 264(6.3) | 38(4.6) | 232(6.2) | 51(8.3) | ||
| Education | 0.557 | <0.001 | ||||
| ≤12th grade with no diploma | 404(13.0) | 93(12.5) | 338(11.8) | 113(24.5) | ||
| High-school graduate/equivalent | 469(21.4) | 134(25.0) | 423(22.0) | 79(20.7) | ||
| Some college/equivalent | 655(31.5) | 145 (30.8) | 566(31.2) | 80(27.9) | ||
| College graduate and above | 614(34.1) | 122(31.8) | 539(35.0) | 78(26.9) | ||
| PIR (percentiles) | 0.166 | 0.008 | ||||
| <185% | 831(27.3) | 210(28.4) | 709(26.5) | 161(32.9) | ||
| 185–350% | 519(23.6) | 123(27.7) | 451(23.6) | 93(26.6) | ||
| 350%< | 792(49.1) | 161(43.9) | 706(49.9) | 96(27.5) | ||
| Smoking status | 0.355 | 0.378 | ||||
| Never | 1141(53.5) | 235(50.4) | 1,028(56.2) | 191(53.6) | ||
| Former | 633(30.1) | 149(29.5) | 527(28.3) | 109(33.4) | ||
| Current | 368(16.4) | 110(20.1) | 311(15.5) | 50(13.0) | ||
| Alcohol (drinks/day) | 0.014 | 0.004 | ||||
| Never/< 12 drinks a year | 735(28.4) | 156(25.0) | 584(25.3) | 172(40.7) | ||
| ≤2 | 674(32.1) | 136(28.9) | 608(33.2) | 87(24.9) | ||
| ≥3 | 383(21.9) | 90(22.5) | 344(22.3) | 50(20.6) | ||
| ≥4 | 350(17.6) | 112(23.7) | 330(19.2) | 41(13.7) | ||
| Season of examination * | 0.085 | 0.909 | ||||
| November–April | 1062(45.4) | 226(41.1) | 910(46.6) | 157(43.1) | ||
| May–October | 1080(54.6) | 268(58.1) | 956(56.4) | 193(56.9) | ||
| BMI (kg/m2) ** | 29.6(6.7) | 29.5(7.5) | 0.847 | 29.4(6.7) | 30.2(8.1) | 0.215 |
| Chronic diseases | ||||||
| Diabetes | 446(16.7) | 103(18.8) | 0.465 | 365(15.8) | 87(21.0) | 0.038 |
| CVD | 124(5.4) | 34(7.4) | 0.026 | 87(4.3) | 29(7.6) | 0.021 |
| Cancer | 313(17.1) | 55(13.5) | 0.221 | 220(13.3) | 58 (24.9) | 0.002 |
| Asthma | 307(14.1) | 79(16.0) | 0.406 | 251(13.4) | 47(16.6) | 0.227 |
| Dietary intake of Vitamin D(mcg/day) | 0.546 | 0.981 | ||||
| Intake <3.1 | 1069(49.2) | 252(50.6) | 933(50.1) | 183(50.2) | ||
| Intake ≥3.1 | 1073(50.8) | 242(49.4) | 933(49.9) | 167(49.8) | ||
| Energy intake (kcal/day) ** | 2085(922) | 2084(951) | 0.987 | 2094(886) | 1930(974) | 0.044 |
| Vitamin D (25(OH)D) (ng/mL) | Age-Adjusted OR Model | p-Value | Adjusted OR Initial Model | p-Value | Adjusted OR Final Model | p-Value |
|---|---|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | OR (95% CI) | ||||
| Sufficiency >30 | 1.00 | 1.00 | 1.00 | |||
| Inadequacy 20–30 | 1.05(0.70–1.59) | 0.796 | 0.85(0.56–1.29) | 0.406 | 0.87(0.59–1.30) | 0.482 |
| Deficiency <20 | 1.79(1.36–2.37) | 0.000 | 1.36(1.01–1.82) | 0.042 | 1.39(1.02–1.89) | 0.038 |
| Gender | ||||||
| Male | 1.00 | 1.00 | 1.00 | |||
| Female | 0.52(0.36–0.76) | 0.002 | 0.40(0.24–0.67) | 0.002 | 0.48(0.31–0.73) | 0.002 |
| Age groups (years) | ||||||
| 40–49 | 1.00 | 1.00 | 1.00 | |||
| 50–59 | 1.44(0.76–2.73) | 0.247 | 1.39(0.70–2.75) | 0.326 | 1.34(0.68–2.64) | 0.370 |
| 60–69 | 2.53(1.36–4.71) | 0.006 | 2.30(1.15–4.64) | 0.022 | 2.36(1.17–4.76) | 0.020 |
| 70–80 | 4.78(2.93–7.81) | 0.000 | 4.28(2.25–8.16) | 0.000 | 4.30(2.41–7.68) | 0.000 |
| Race/ethnicity | ||||||
| Non-Hispanic White | 1.00 | 1.00 | 1.00 | |||
| Hispanic American | 1.80(1.26–2.57) | 0.003 | 1.37(0.92–2.06) | 0.113 | 1.35(0.87–2.10) | 0.171 |
| Non-Hispanic Black | 1.84(1.41–2.38) | 0.000 | 1.51(0.99–2.29) | 0.053 | 1.57(1.06–2.32) | 0.027 |
| Other race | 1.91(1.19–3.07) | 0.011 | 2.15(1.29–3.59) | 0.006 | 2.96(1.21–3.17) | 0.010 |
| Education | ||||||
| ≤12th grade no diploma | 1.00 | 1.00 | 1.00 | |||
| High-school graduate | 0.46(0.27–0.76) | 0.005 | 0.47(0.27–0.81) | 0.009 | 0.49(0.29–0.84) | 0.012 |
| Some college | 0.43(0.33–0.57) | 0.000 | 0.44(0.32–0.62) | 0.000 | 0.47(0.33–0.65) | 0.000 |
| College graduate/above | 0.40(0.27–0.60) | 0.000 | 0.44(0.27–0.71) | 0.002 | 0.44(0.27–0.71) | 0.003 |
| PIR (percentiles) | ||||||
| <185% | 1.00 | 1.00 | -- | -- | ||
| 185%-350% | 0.98(0.71–1.35) | 0.894 | 1.20(0.90–1.60) | 0.194 | -- | -- |
| 350%< | 0.66(0.49–0.90) | 0.011 | 0.92(0.66–1.28) | 0.602 | -- | -- |
| Smoking status | ||||||
| Never | 1.00 | 1.00 | -- | -- | ||
| Former | 1.05(0.69–1.59) | 0.815 | 0.89(0.58–1.37) | 0.571 | -- | -- |
| Current | 1.13(0.70–1.82) | 0.597 | 0.88(0.56–1.38) | 0.544 | -- | -- |
| Alcohol (drinks/day) | ||||||
| Never/ <12 drinks a year | 1.00 | 1.00 | 1.00 | |||
| ≤2 | 0.58(0.40–0.84) | 0.006 | 0.59(0.40–0.88) | 0.012 | 0.58(0.40–0.85) | 0.009 |
| ≥3 | 0.81(0.56–1.17) | 0.242 | 0.88(0.61–1.28) | 0.489 | 0.82(0.55–1.20) | 0.280 |
| ≥4 | 0.63(0.33–1.19) | 0.145 | 0.67(0.35–1.27) | 0.202 | 0.61(0.31–1.20) | 0.144 |
| Season of examination * | ||||||
| November–April | 1.00 | 1.00 | -- | -- | ||
| May–October | 0.95(0.67–1.34) | 0.751 | 1.12(0.84–1.49) | 0.408 | -- | -- |
| BMI (kg/m2) | 1.02(0.99–1.05) | 0.125 | 1.02(0.99–1.05) | 0.084 | -- | -- |
| Chronic diseases | ||||||
| Diabetes | 1.18(0.84–1.66) | 0.317 | 0.78(0.58–1.05) | 0.098 | -- | -- |
| CVD | 1.10(0.60–2.00) | 0.752 | 1.01(0.52–1.95) | 0.982 | -- | -- |
| Cancer | 1.83(1.94–2.57) | 0.083 | 2.06(1.22–3.45) | 0.010 | 2.00(1.16–3.46) | 0.016 |
| Asthma | 1.28(0.83–1.98) | 0.251 | 1.46(0.95–2.22) | 0.073 | -- | -- |
| Dietary intake of Vitamin D (mcg/day) | ||||||
| <3.1 | 1.0 | 1.0 | -- | -- | ||
| ≥3.1 | 0.93(0.66–1.32) | 0.669 | 0.99(0.68–1.46) | 0.996 | -- | -- |
| Energy intake (kcal/day) | 0.99(0.99–1.00) | 0.299 | 0.99(0.99–1.00) | 0.112 | -- | -- |
| Vitamin D (25(OH)D) (ng/mL) | Age-Adjusted OR Model | p-Value | Adjusted OR Final Model | p-Value |
|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | |||
| Sufficiency > 30 | 1.00 | 1.00 | ||
| Inadequacy 20–30 | 0.88(0.60–1.28) | 0.466 | 0.86(0.59–1.26) | 0.412 |
| Deficiency <20 | 0.88(0.58–1.32) | 0.515 | 0.79(0.51–1.24) | 0.288 |
| Gender | ||||
| Male | 1.00 | -- | -- | |
| Female | 1.03(0.84–1.26) | 0.739 | -- | -- |
| Age groups (years) | ||||
| 40–49 | 1.00 | 1.00 | ||
| 50–59 | 0.72(0.50–1.03) | 0.069 | 0.71(0.50–1.00) | 0.055 |
| 60–69 | 0.70(0.49–1.00) | 0.051 | 0.69(0.47–1.01) | 0.056 |
| 70–80 | 0.63(0.43–0.92) | 0.021 | 0.61(0.42–0.89) | 0.014 |
| Race/ethnicity | ||||
| Non-Hispanic White | 1.00 | 1.00 | ||
| Hispanic American | 0.96(0.63–1.46) | 0.836 | 1.01(0.66–1.55) | 0.953 |
| Non-Hispanic Black | 1.44(1.00–2.08) | 0.050 | 1.58(1.07–2.34) | 0.024 |
| Other race | 0.70(0.40–1.21) | 0.187 | 0.76(0.42–1.35) | 0.319 |
| Education | ||||
| ≤12th grade | 1.00 | -- | -- | |
| High-school graduate | 1.21(0.81–1.81) | 0.326 | -- | -- |
| Some college | 1.02(0.68–1.50) | 0.936 | -- | -- |
| College graduate or above | 0.96(0.62–1.48) | 0.934 | -- | -- |
| PIR (percentiles) | ||||
| <185% | 1.00 | -- | -- | |
| 185–350% | 1.12(0.78–1.62) | 0.518 | -- | -- |
| 350%< | 0.84(0.60–1.20) | 0.328 | -- | -- |
| Smoking status | ||||
| Never | 1.00 | -- | -- | |
| Former | 1.10(0.80–1.51) | 0.524 | -- | -- |
| Current | 1.25(0.89–1.76) | 0.181 | -- | -- |
| Alcohol (drinks/day) | ||||
| Never/< 12 drinks a year | 1.00 | 1.00 | ||
| ≤2 | 0.98(0.72-1.37) | 0.937 | 0.99(0.72–1.39) | 0.995 |
| ≥3 | 1.12(0.73-1.72) | 0.578 | 1.15(0.74–1.77) | 0.508 |
| ≥4 | 1.39(1.04-1.86) | 0.029 | 1.43(1.07–1.89) | 0.018 |
| Season of examination * | ||||
| November–April | 1.00 | -- | -- | |
| May–October | 1.22 (0.98–1.50) | 0.063 | -- | -- |
| BMI (kg/m2) | 0.99(0.97–1.03) | 0.811 | -- | -- |
| Chronic diseases | ||||
| Diabetes | 1.24(0.82–1.83) | 0.288 | -- | -- |
| CVD | 1.67(1.24–2.24) | 0.002 | 1.71(1.33–2.20) | 0.000 |
| Cancer | 0.83(0.48–1.45) | 0.494 | -- | -- |
| Asthma | 1.17(0.80–1.69) | 0.395 | -- | -- |
| Dietary intake of Vitamin D (mcg/day) | ||||
| <3.1 | 1.00 | -- | -- | |
| ≥3.1 | 0.97(0.80–1.16) | 0.712 | -- | -- |
| Energy intake (kcal/day) | 0.99(0.99–1.00) | 0.789 | -- | -- |
| Vitamin D (25(OH)D) (ng/mL) | Crude OR | p-Value | Adjusted OR* | p-Value |
|---|---|---|---|---|
| Age group 40–49 years, n = 740 | OR (95% CI) | OR (95% CI) | ||
| Sufficiency >30 | 1.00 | 1.00 | ||
| Inadequacy 20–30 | 0.83(0.43–1.62) | 0.562 | 0.83(0.45–1.55) | 0.542 |
| Deficiency <20 | 0.84(0.39–1.82) | 0.633 | 0.74(0.37–1.46) | 0.357 |
| Age group 50–59 years, n = 688 | ||||
| Sufficiency >30 | 1.00 | 1.00 | ||
| Inadequacy 20–30 | 0.78(0.36–1.69) | 0.508 | 0.76(0.35–1.67) | 0.474 |
| Deficiency <20 | 0.73(0.31–1.74) | 0.453 | 0.62(0.25–1.52) | 0.272 |
| Age group 60–69 years, n = 649 | ||||
| Sufficiency >30 | 1.00 | 1.00 | ||
| Inadequacy 20–30 | 0.61(0.33–1.09) | 0.088 | 0.59(0.32–1.09) | 0.088 |
| Deficiency <20 | 0.98(0.52–1.88) | 0.955 | 0.95(0.46–1.93) | 0.871 |
| Age group 70–80 years, n = 559 | ||||
| Sufficiency >30 | 1.00 | 1.00 | ||
| Inadequacy 20–30 | 1.88(1.27–2.78) | 0.004 | 1.96(1.35–2.85) | 0.002 |
| Deficiency <20 | 1.22(0.51–2.93) | 0.635 | 1.15(0.45–2.98) | 0.753 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bigman, G. Age-related Smell and Taste Impairments and Vitamin D Associations in the U.S. Adults National Health and Nutrition Examination Survey. Nutrients 2020, 12, 984. https://doi.org/10.3390/nu12040984
Bigman G. Age-related Smell and Taste Impairments and Vitamin D Associations in the U.S. Adults National Health and Nutrition Examination Survey. Nutrients. 2020; 12(4):984. https://doi.org/10.3390/nu12040984
Chicago/Turabian StyleBigman, Galya. 2020. "Age-related Smell and Taste Impairments and Vitamin D Associations in the U.S. Adults National Health and Nutrition Examination Survey" Nutrients 12, no. 4: 984. https://doi.org/10.3390/nu12040984
APA StyleBigman, G. (2020). Age-related Smell and Taste Impairments and Vitamin D Associations in the U.S. Adults National Health and Nutrition Examination Survey. Nutrients, 12(4), 984. https://doi.org/10.3390/nu12040984




