Traditional Dietary Patterns and Risk of Mortality in a Longitudinal Cohort of the Salus in Apulia Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population:
2.2. Dietary and Lifestyle Variables
2.3. Food Group and Dietary Patterns
2.3.1. Food Groups
2.3.2. A Priori Dietary Pattern
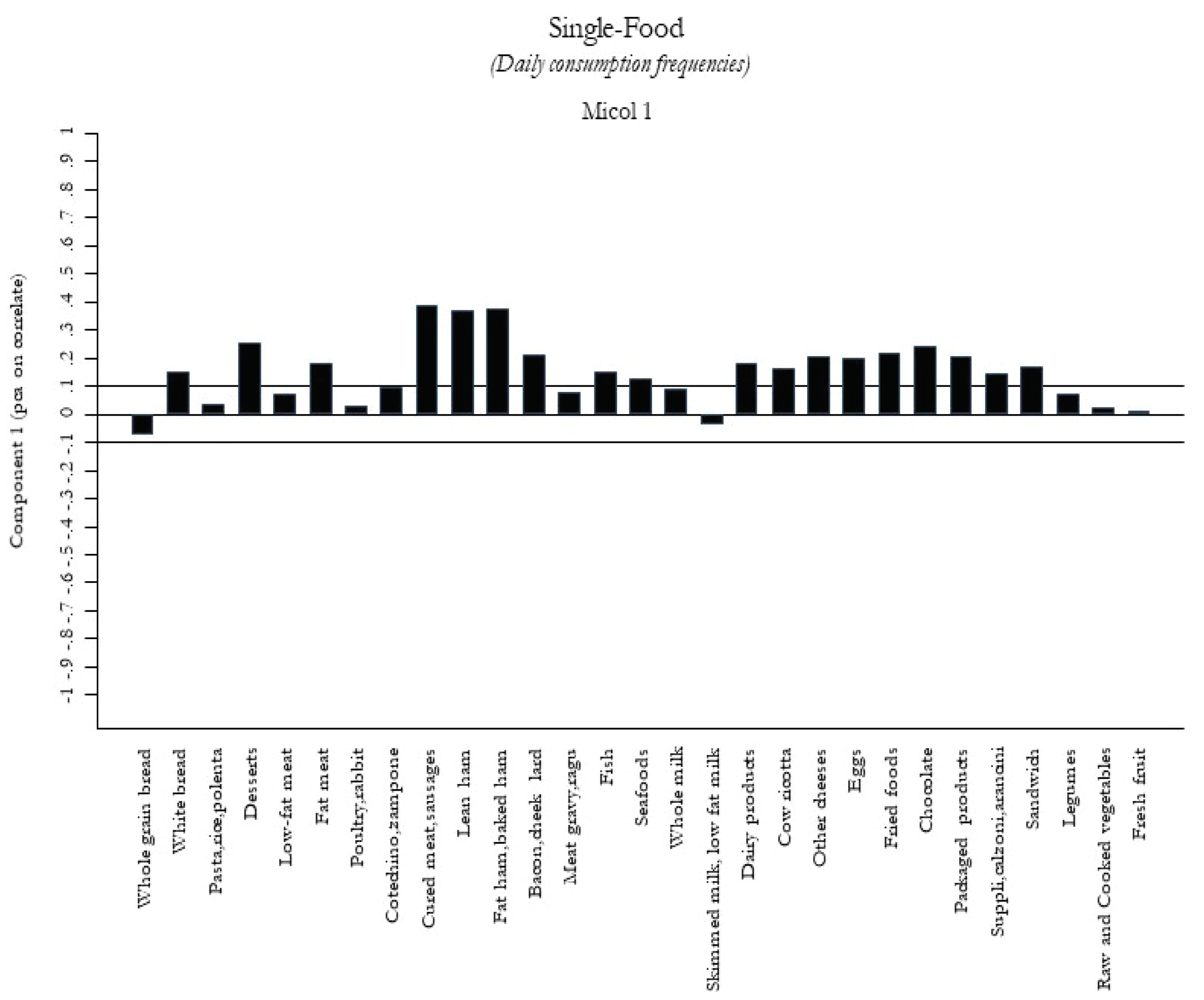
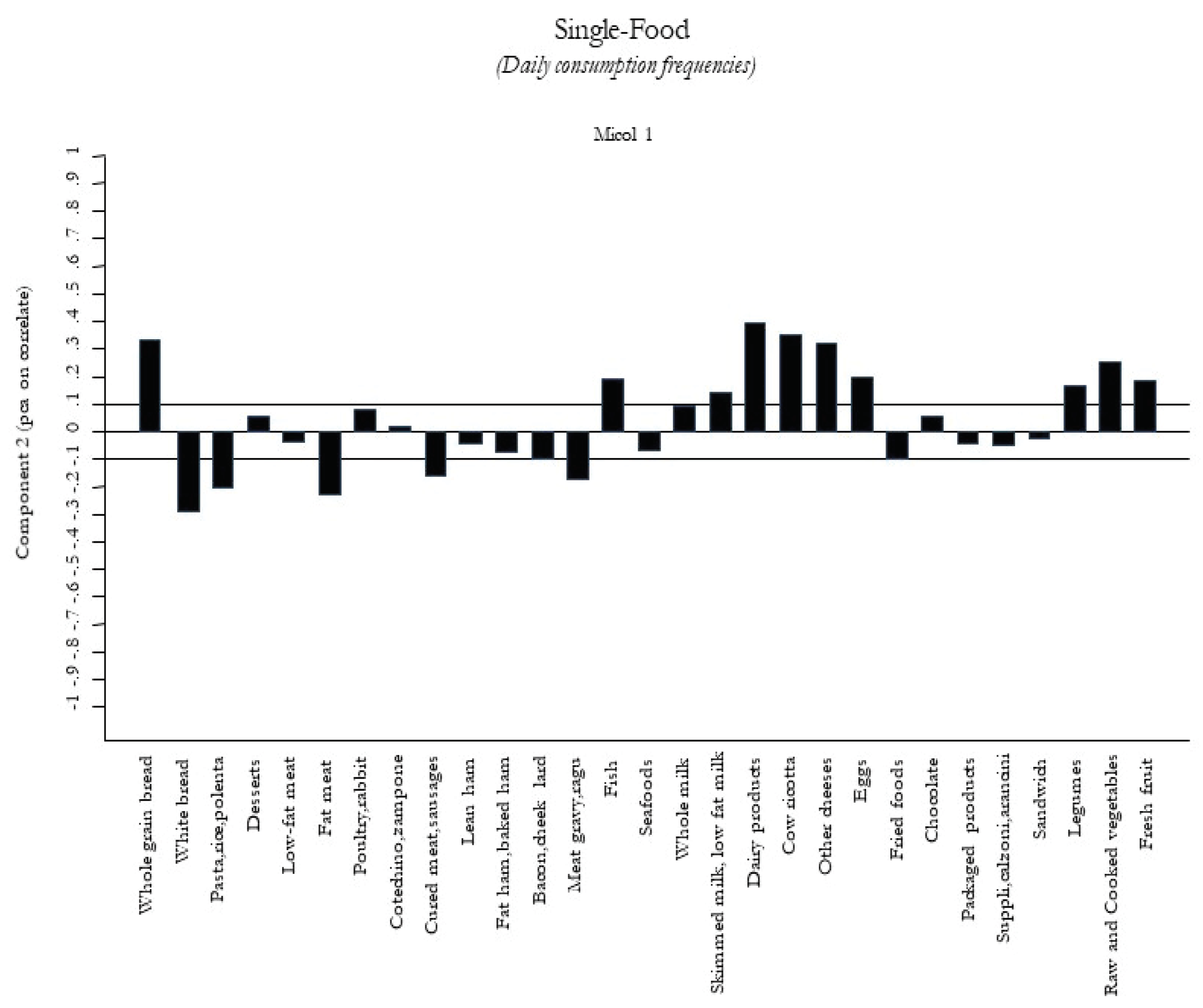
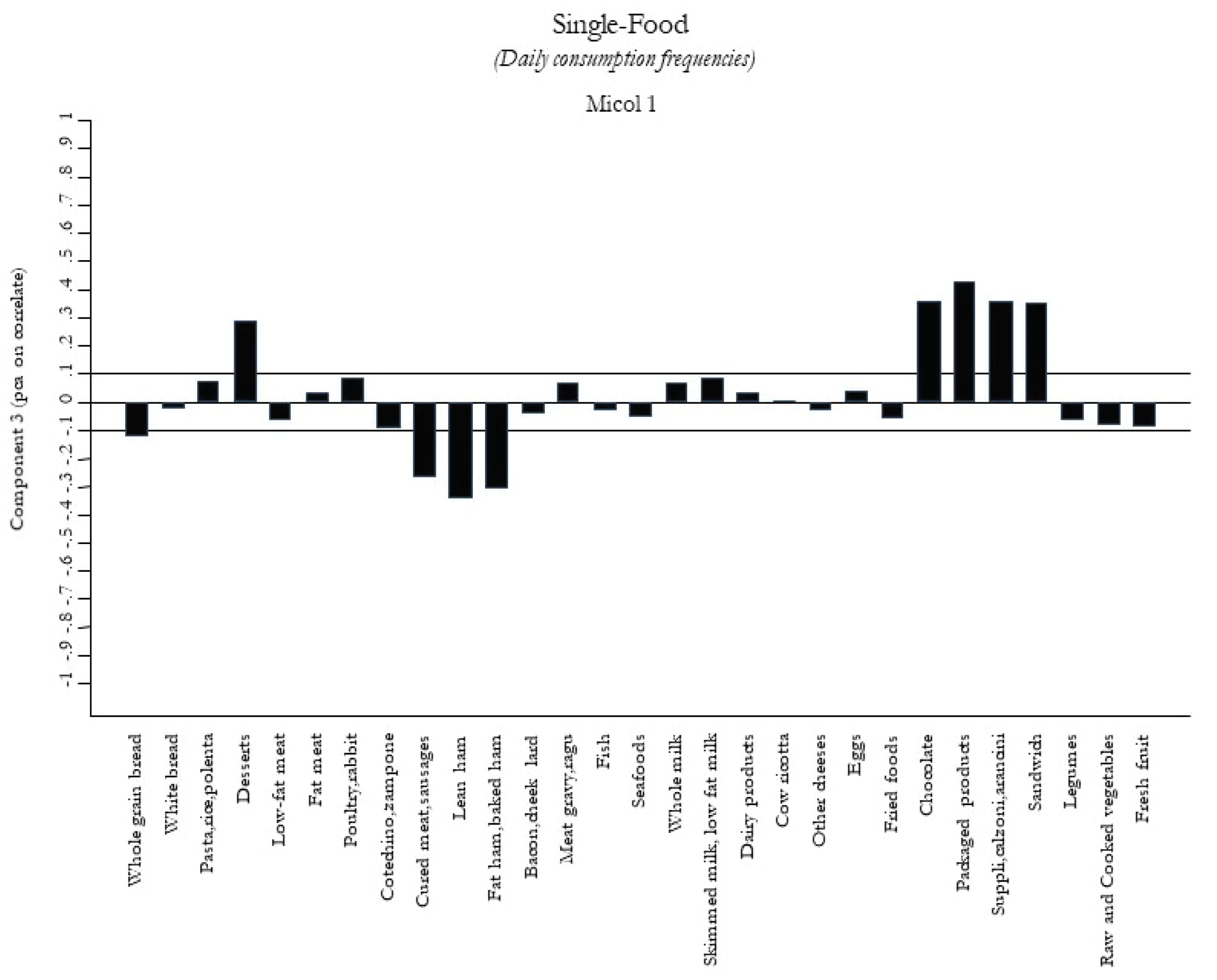
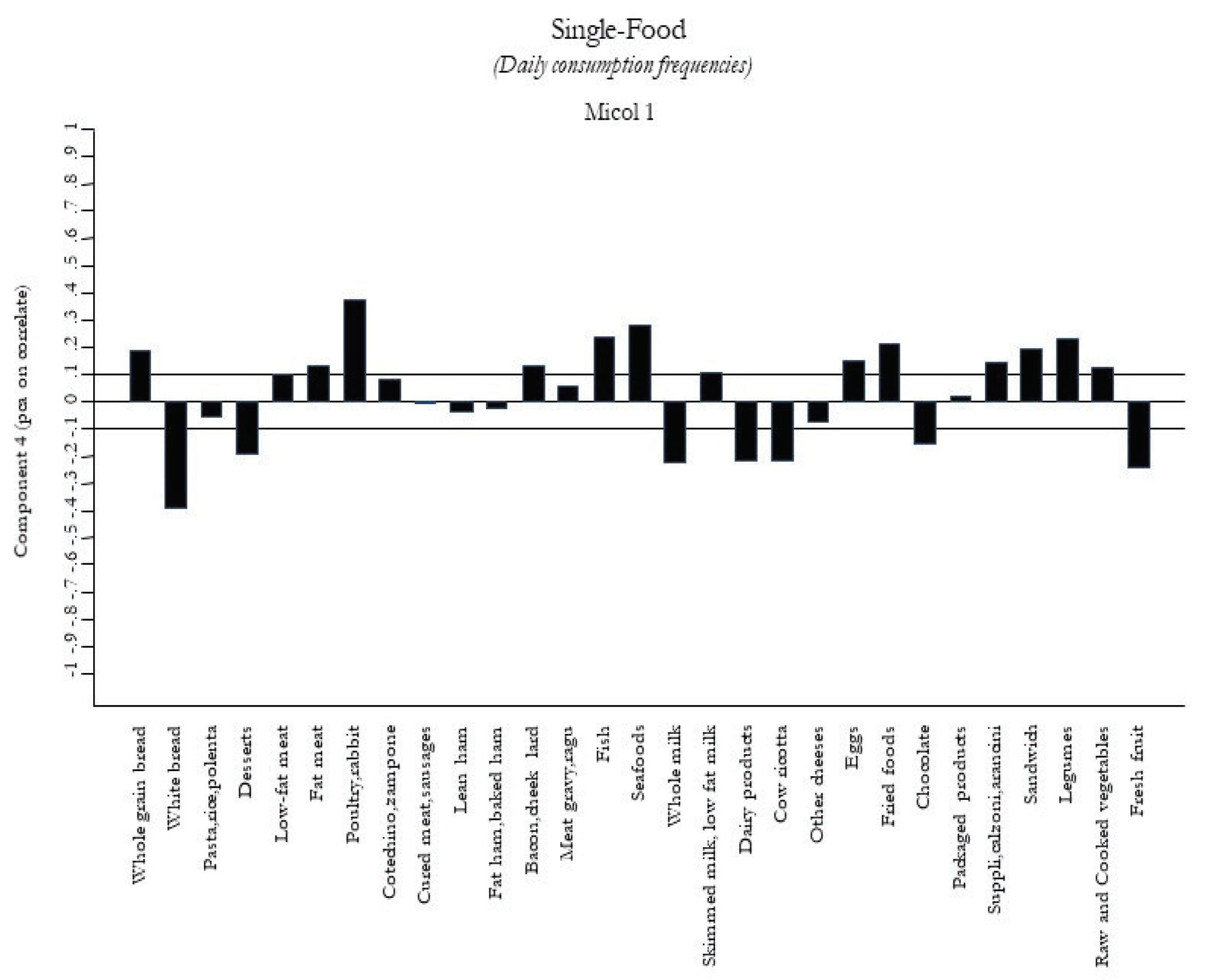
2.3.3. A Posteriori Dietary Patterns
2.3.4. Predictive Variables for the Outcomes
2.3.5. Mortality Assessment
2.4. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, X.; Ouyang, Y.; Liu, J.; Zhu, M.; Zhao, G.; Bao, W.; Hu, F.B. Fruit and vegetable consumption and mortality from all causes, cardiovascular disease, and cancer: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2014, 349, g4490. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef] [PubMed]
- Mayhew, A.J.; De Souza, R.J.; Meyre, D.; Anand, S.S.; Mente, A. A systematic review and meta-analysis of nut consumption and incident risk of CVD and all-cause mortality. Br. J. Nutr. 2015, 115, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-G.; Sun, J.-W.; Yang, Y.; Ma, X.; Wang, Y.-Y.; Xiang, Y.-B. Fish consumption and all-cause mortality: A meta-analysis of cohort studies. Eur. J. Clin. Nutr. 2015, 70, 155–161. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef]
- Guenther, P.M.; Casavale, K.O.; Reedy, J.; Kirkpatrick, S.I.; Hiza, H.A.; Kuczynski, K.J.; Kahle, L.L.; Krebs-Smith, S.M. Update of the healthy eating index: HEI-2010. J. Acad. Nutr. Diet. 2013, 113, 569–580. [Google Scholar] [CrossRef]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef]
- Folsom, A.R.; Parker, E.; Harnack, L.J. Degree of concordance with DASH diet guidelines and incidence of hypertension and fatal cardiovascular disease. Am. J. Hypertens. 2007, 20, 225–232. [Google Scholar] [CrossRef]
- D’Alessandro, A.; De Pergola, G. Mediterranean diet pyramid: A proposal for Italian people. Nutrients 2014, 6, 4302–4316. [Google Scholar] [CrossRef]
- Zupo, R.; Lampignano, L.; Lattanzio, A.; Mariano, F.; Osella, A.R.; Bonfiglio, C.; Giannelli, G.; De Pergola, G. Association between adherence to the Mediterranean diet and circulating Vitamin D levels. Int. J. Food Sci. Nutr. 2020. [Google Scholar] [CrossRef]
- Veronese, N.; Notarnicola, M.; Cisternino, A.M.; Inguaggiato, R.; Guerra, V.; Reddavide, R.; Donghia, R.; Rotolo, O.; Zinzi, I.; Leandro, G.; et al. Trends in adherence to the Mediterranean diet in South Italy: A cross sectional study. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Attili, A.F.; Carulli, N.; Roda, E.; Barbara, B.; Capocaccia, L.; Menotti, A.; Okoliksanyi, L.; Ricci, G.; Capocaccia, R.; Festi, D.; et al. Epidemiology of gallstone disease in Italy: Prevalence data of the Multicenter Italian Study on Cholelithiasis (MI COL.). Am. J. Epidemiol. 1995, 141, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Misciagna, G.; Leoci, C.; Guerra, V.; Chiloiro, M.; Elba, S.; Petruzzi, J.; Mossa, A.; Noviello, M.R.; Coviello, A.; Minutolo, M.C.; et al. Epidemiology of cholelithiasis in southern Italy. Part II: Risk factors. Eur. J. Gastroenterol. Hepatol. 1996, 8, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Osella, A.R.; Misciagna, G.; Leone, A.; Di Leo, A.; Fiore, G. Epidemiology of hepatitis C Virus infection in an area of Southern Italy. J. Hepatol. 1997, 27, 30–35. [Google Scholar] [CrossRef]
- World Health Organization. Multimorbidity; World Health Organization: Geneva, Switzerland, 2016; Available online: http://apps.who.int/iris/handle/10665/252275 (accessed on 19 July 2019).
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the Mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the MedDietScore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef]
- Morris, M.C.; Tangney, C.; Wang, Y.; Sacks, F.M.; Barnes, L.L.; Bennett, D.A.; Aggarwal, N.T. MIND diet slows cognitive decline with aging. Alzheimer’s Dement. 2015, 11, 1015–1022. [Google Scholar] [CrossRef]
- Agnoli, C.; Krogh, V.; Grioni, S.; Sieri, S.; Palli, D.; Masala, G.; Sacerdote, C.; Vineis, P.; Tumino, R.; Frasca, G.; et al. A priori-defined dietary patterns are associated with reduced risk of stroke in a large Italian cohort. J. Nutr. 2011, 141, 1552–1558. [Google Scholar] [CrossRef]
- Smith, A.D.A.C.; Emmett, P.; Newby, P.K.; Northstone, K. Dietary patterns obtained through principal components analysis: The effect of input variable quantification. Br. J. Nutr. 2012, 109, 1881–1891. [Google Scholar] [CrossRef]
- Ishwaran, H.; Kogalur, U.B.; Blackstone, E.H.; Lauer, M.S. Random survival forests. Ann. Appl. Stat. 2008, 2, 841–860. [Google Scholar] [CrossRef]
- Dietrich, S.; Floegel, A.; Troll, M.; Kuhn, T.; Rathmann, W.; Peters, A.; Sookthai, D.; Von Bergen, M.; Kaaks, R.; Adamski, J.; et al. Random survival forest in practice: A method for modelling complex metabolomics data in time to event analysis. Int. J. Epidemiol. 2016, 45, 1406–1420. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Milias, G.A.; Pitsavos, C.; Stefanadis, C. MedDietScore: A computer program that evaluates the adherence to the Mediterranean dietary pattern and its relation to cardiovascular disease risk. Comput. Methods Prog. Biomed. 2006, 83, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Yubero-Serrano, E.M.; Lopez-Moreno, J.; Delgado, F.G.; Lopez-Miranda, J. Extra virgin olive oil: More than a healthy fat. Eur. J. Clin. Nutr. 2018, 72, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Hu, F.B.; Martínez-González, M.A.; Fitó, M.; Bulló, M.; Estruch, R.; Ros, E.; Corella, D.; Recondo, J.; Gómez-Gracia, E.; et al. Olive oil intake and risk of cardiovascular disease and mortality in the PREDIMED Study. BMC Med. 2014, 12, 78. [Google Scholar] [CrossRef] [PubMed]
- Masala, G.; Ceroti, M.; Pala, V.; Krogh, V.; Vineis, P.; Sacerdote, C.; Saieva, C.; Salvini, S.; Sieri, S.; Berrino, F.; et al. A dietary pattern rich in olive oil and raw vegetables is associated with lower mortality in Italian elderly subjects. Br. J. Nutr. 2007, 98, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Lampousi, A.-M.; Portillo, M.P.; Romaguera, D.; Hoffmann, G.; Boeing, H. Olive oil in the prevention and management of type 2 diabetes mellitus: A systematic review and meta-analysis of cohort studies and intervention trials. Nutr. Diabetes 2017, 7, e262. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Cayssials, V.; Clèries, R.; Redondo, M.L.; Huerta, J.; Rodríguez-Barranco, M.; Sánchez-Cruz, J.-J.; Mokoroa, O.; Gil, L.; Amiano, P.; et al. Moderate egg consumption and all-cause and specific-cause mortality in the Spanish European Prospective into Cancer and Nutrition (EPIC-Spain) study. Eur. J. Nutr. 2018, 58, 2003–2010. [Google Scholar] [CrossRef]
- Guo, J.; Hobbs, D.A.; Cockcroft, J.R.; Elwood, P.C.; Pickering, J.E.; Lovegrove, J.; Givens, D.I. Association between egg consumption and cardiovascular disease events, diabetes and all-cause mortality. Eur. J. Nutr. 2017, 57, 2943–2952. [Google Scholar] [CrossRef]
- Solfrizzi, V.; Agosti, P.; Lozupone, M.; Custodero, C.; Schilardi, A.; Valiani, V.; Santamato, A.; Sardone, R.; Dibello, V.; Di Lena, L.; et al. Nutritional interventions and cognitive-related outcomes in patients with late-life cognitive disorders: A systematic review. Neurosci. Biobehav. Rev. 2018, 95, 480–498. [Google Scholar] [CrossRef]
- Mutungi, G.; Ratliff, J.; Puglisi, M.; Torres-Gonzalez, M.; Vaishnav, U.; Leite, J.O.; Quann, E.; Volek, J.S.; Fernandez, M.-L. Dietary cholesterol from eggs increases plasma HDL cholesterol in overweight men consuming a carbohydrate-restricted diet. J. Nutr. 2008, 138, 272–276. [Google Scholar] [CrossRef]
- Rong, Y.; Chen, L.; Zhu, T.; Song, Y.; Yu, M.; Shan, Z.; Sands, A.; Hu, F.B.; Liu, L. Egg consumption and risk of coronary heart disease and stroke: Dose-response meta-analysis of prospective cohort studies. BMJ 2013, 346, e8539. [Google Scholar] [CrossRef]
- Gerosa, S.; Skoet, J. Milk Availability: Trends in Production and Demand and Medium-Term Outlook. 2012. Available online: https://ageconsearch.umn.edu/record/289000/ (accessed on 12 December 2019).
- Tognon, G.; Nilsson, L.M.; Shungin, D.; Lissner, L.; Jansson, J.-H.; Renström, F.; Wennberg, M.; Winkvist, A.; Johansson, I. Nonfermented milk and other dairy products: Associations with all-cause mortality. Am. J. Clin. Nutr. 2017, 105, 1502–1511. [Google Scholar] [CrossRef] [PubMed]
- Deghan, M.; Mente, A.; Yusuf, S. Associations of fats and carbohydrates with cardiovascular disease and mortality-PURE and simple?—Authors’ reply. Lancet 2018, 391, 1681–1682. [Google Scholar] [CrossRef]
- Fontecha, J.; Juárez, M. Chapter 19—Recent advances in dairy ingredients and cardiovascular diseases with special reference to milk fat components. In Dairy in Human Health and Disease across the Lifespan; Watson, R.R., Collier, R.J., Preedy, V.R., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 251–261. Available online: http://www.sciencedirect.com/science/article/pii/B9780128098684000194 (accessed on 12 December 2019).
- Goulet, O. Potential role of the intestinal microbiota in programming health and disease: Figure 1. Nutr. Rev. 2015, 73, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Butel, M.-J. Probiotics, gut microbiota and health. Med. Mal. Infect. 2014, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Bamia, C.; Trichopoulos, D. Anatomy of health effects of Mediterranean diet: Greek EPIC prospective cohort study. BMJ 2009, 338, b2337. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Guinee, T.P. Salting and the role of salt in cheese. Int. J. Dairy Technol. 2004, 57, 99–109. [Google Scholar] [CrossRef]
- Carnovale, E.; Marletta, L. Tabelle di Composizione Degli Alimenti; Edra: Perignano, Italy, 1997; Available online: https://play.google.com/store/books/details?id=TfswAAAACAAJ (accessed on 12 December 2019).

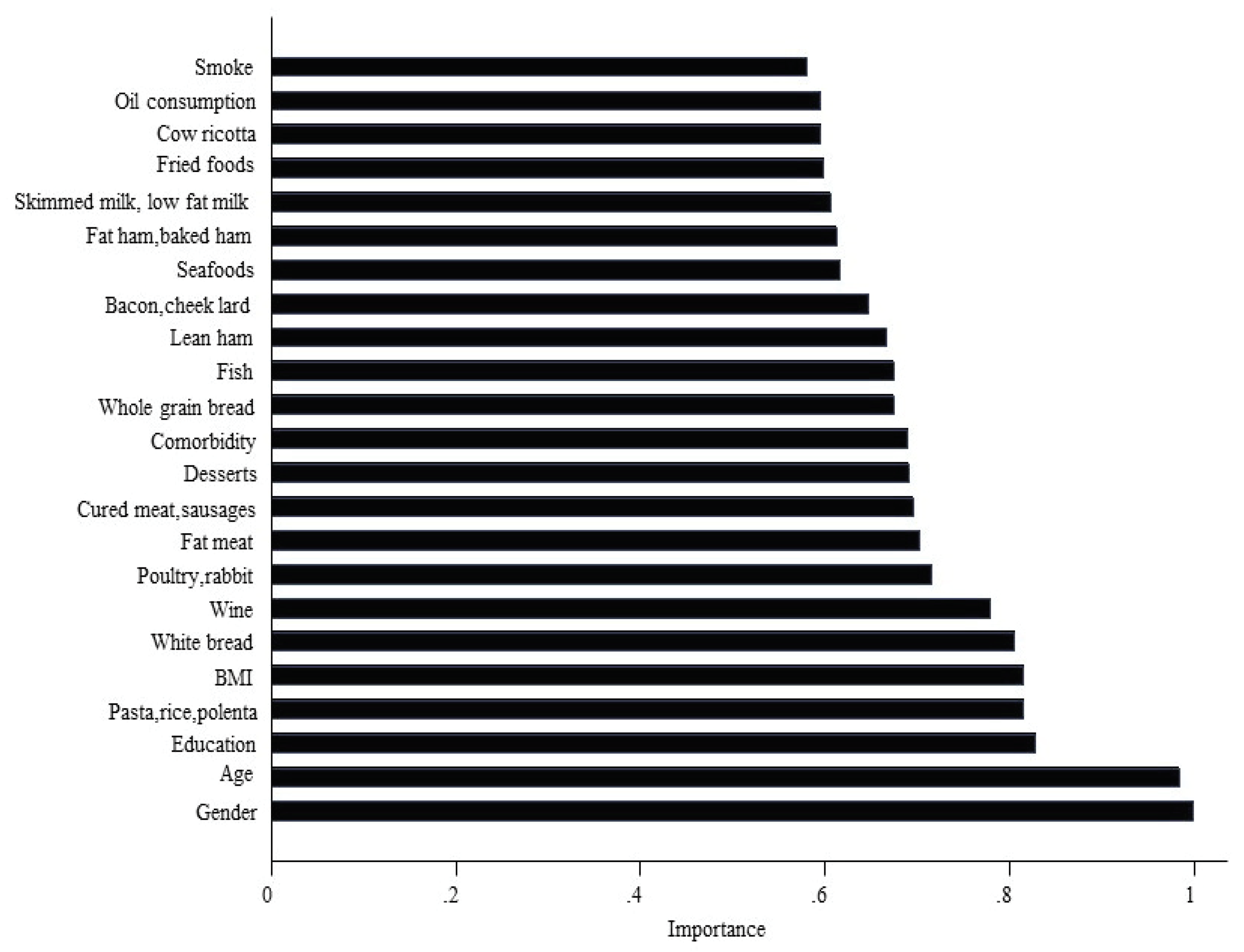
| Variables | Frequencies * | HR of Mortality Mutually Adjusted (95% CI) |
|---|---|---|
| Gender (Female) (%) | 1043 (42.19) | 0.65 (0.56 to 0.75) |
| Age (yrs) (M±SD) | 48.00 ± 10.71 | 1.12 (1.11 to 1.13) |
| Education (%) | ||
| Low (≤ 5years) | 1657 (67.03) | 1 |
| Medium (8 years) | 457 (18.49) | 0.95 (0.79 to 1.16) |
| High (> 8years) | 358 (14.48) | 0.89 (0.70 to 1.13) |
| Smoking (%) | 788 (31.88) | 1.63 (1.40 to 1.89) |
| Body Mass Index (kg/m²) | 27.48±4.55 | 1.04 (1.02 to 1.05) |
| Wine (≥ 1 lt/day) (%) | 289 (11.69) | 0.96 (0.79 to 1.16) |
| Comorbidity (≥ 2) (%) | 154 (6.23) | 1.20 (0.97 to 1.49) |
| Frequent use of olive oil (%) | 864 (34.95) | 0.84 (0.73 to 0.96) |
| Foods | Total Sample (M ± SD) | Men (M ± SD) | Women (M ± SD) |
|---|---|---|---|
| Whole grain bread (Pane integrale) | 0.09 ± 0.34 | 0.08 ± 0.32 | 0.11 ± 0.37 |
| Semolina type and other bread (Pane bianco) | 1.28 ± 0.66 | 1.31 ± 0.65 | 1.24 ± 0.67 |
| Pasta, rice, polenta (Pasta, riso, polenta) | 0.52 ± 0.26 | 0.53 ± 0.26 | 0.50 ± 0.25 |
| Desserts (Torte, crostate) | 0.17 ± 0.17 | 0.16 ± 0.16 | 0.20 ± 0.18 |
| Low-fat meat (Carne magra) | 0.34 ± 0.11 | 0.35 ± 0.11 | 0.34 ± 0.11 |
| Fatty meat (Carne di maiale) | 0.12 ± 0.14 | 0.13 ± 0.14 | 0.09 ± 0.14 |
| Poultry, rabbit (Pollame e coniglio) | 0.25 ± 0.15 | 0.25 ± 0.15 | 0.25 ± 0.15 |
| Cotechino, zampone (Cotechino, zampone) | 0.005 ± 0.033 | 0.005 ± 0.03 | 0.006 ± 0.04 |
| Cured meat, sausages (Salumi, salsiccia) | 0.13 ± 0.15 | 0.14 ± 0.15 | 0.11 ± 0.15 |
| Lean ham (Prosciutto magro) | 0.13 ± 0.14 | 0.14 ± 0.15 | 0.12 ± 0.14 |
| Fatty ham, ham (Prosciutto grasso o cotto) | 0.13 ± 0.14 | 0.14 ± 0.14 | 0.13 ± 0.14 |
| Bacon, cheek lard (Pancetta e guanciale) | 0.04 ± 0.07 | 0.04 ± 0.08 | 0.03 ± 0.07 |
| Meat sauce (Sughi di carne, ragù) | 0.32 ± 0.12 | 0.33 ± 0.12 | 0.32 ± 0.11 |
| Fish (Pesce) | 0.23 ± 0.15 | 0.23 ± 0.15 | 0.23 ± 0.15 |
| Seafoods (Frutti di mare) | 0.07 ± 0.10 | 0.07 ± 0.12 | 0.06 ± 0.08 |
| Whole milk (Latte intero) | 0.25 ± 0.45 | 0.25 ± 0.45 | 0.26 ± 0.45 |
| Skimmed milk, low fat milk (Latte scremato o parzialmente scremato) | 0.26 ± 0.44 | 0.24 ± 0.43 | 0.28 ± 0.46 |
| Sheep ricotta, fiordilatte, mozzarella, smoked provola (Ricotta di pecora, fiordilatte, mozzarella, provola affumicata) | 0.29 ± 0.18 | 0.27 ± 0.19 | 0.31 ± 0.16 |
| Cow ricotta (Ricotta di vacca) | 0.15 ± 0.15 | 0.13 ± 0.14 | 0.17 ± 0.15 |
| Other cheeses (Altri formaggi) | 0.27 ± 0.18 | 0.27 ± 0.19 | 0.27 ± 0.18 |
| Eggs (Uova) | 0.20 ± 0.16 | 0.19 ± 0.16 | 0.20 ± 0.16 |
| Fried foods (Alimenti fritti) | 0.18 ± 0.15 | 0.19 ± 0.15 | 0.18 ± 0.15 |
| Chocolate (Cioccolata) | 0.07 ± 0.12 | 0.06 ± 0.13 | 0.07 ± 0.12 |
| Croissant, buns, pizzas (cornetti, maritozzi, pizza) | 0.04 ± 0.11 | 0.04 ± 0.13 | 0.03 ± 0.10 |
| Typical local rotisserie (Supplì, calzone, arancini) | 0.08 ± 0.09 | 0.08 ± 0.10 | 0.08 ± 0.09 |
| Sandwiches (Tramezzini) | 0.01 ± 0.06 | 0.02 ± 0.07 | 0.01 ± 0.04 |
| Legumes: peas, beans, lentils, fava beans, chickpeas (Legumi: piselli, fagioli, lenticchie, fave, ceci) | 0.32 ± 0.11 | 0.32 ± 0.11 | 0.31 ± 0.12 |
| Raw vegetables, Cooked vegetables (Verdure crude e cotte) | 0.41 ± 0.23 | 0.39 ± 0.23 | 0.43 ± 0.24 |
| Fresh fruit (Frutta fresca) | 1.81 ± 0.50 | 1.76 ± 0.55 | 1.88 ± 0.40 |
| Foods in Questionnaire | HR of Mortality for one Frequency Per Day, Adjusted for Covariates (95% CI) |
|---|---|
| Whole grain bread | 1.10 (0.93 to 1.30) |
| Semolina type band other bread | 0.92 (0.83 to 1.02) |
| Pasta, rice, polenta | 0.92 (0.71 to 1.18) |
| Desserts | 0.89 (0.58 to 1.37) |
| Low-fat meat | 0.86 (0.48 to 1.53) |
| Fatty meat | 0.60 (0.37 to 0.99) |
| Poultry, rabbit | 1.02 (0.67 to 1.54) |
| Cotechino, zampone | 0.16 (0.01 to 2.07) |
| Cured meat, sausages | 0.78 (0.47 to 1.28) |
| Lean ham | 0.82 (0.50 to 1.35) |
| Fatty ham, ham | 0.59 (0.35 to 0.99) |
| Bacon, cheek lard | 0.72 (0.25 to 2.09) |
| Meat sauce | 1.55 (0.85 to 2.82) |
| Fish | 0.80 (0.53 to 1.22) |
| Seafood | 0.63 (0.33 to 1.22) |
| Whole milk | 1.07 (0.93 to 1.23) |
| Skimmed milk, low-fat milk | 1.00 (0.87 to 1.15) |
| Sheep ricotta, fiordilatte, mozzarella, smoked provola | 1.36 (0.99 to 1.86) |
| Cow ricotta | 1.45 (0.94 to 2.24) |
| Other cheeses | 1.05 (0.73 to 1.52) |
| Eggs | 0.63 (0.42 to 0.95) |
| Fried foods | 1.11 (0.72 to 1.71) |
| Chocolate | 1.05 (0.56 to 1.97) |
| Croissant, buns, pizzas | 0.55 (0.24 to 1.27) |
| Typical local rotisserie | 0.89 (0.42 to 1.87) |
| Sandwiches | 0.79 (0.16 to 3.75) |
| Legumes: peas, beans, lentils, fava beans, chickpeas | 1.17 (0.68 to 2.01) |
| Raw vegetables, cooked vegetables | 1.06 (0.80 to 1.41) |
| Fresh fruit | 1.00 (0.88 to 1.15) |
| Variables | HR * with Olive oil as Adjustment (95% CI) |
|---|---|
| Food groups (sum of frequencies) | (per one frequency per day, not mutually adjusted) |
| Legumes and vegetables (Legumi e vegetali) | 1.07 (0.85 to 1.36) |
| Fresh cheese (Ricotta di pecora e di vacca, fiordilatte, mozzarella, provola affumicata) | 1.29 (1.03 to 1.63) |
| Red meat (Carne rossa: carne magra e grassa) | 0.89 (0.66 to 1.19) |
| Processed meat (Carni processate: salumi, salsicce, pancetta, guanciale, cotechino, zampone, prosciutto magro e grasso o cotto) | 0.85 (0.70 to 1.03) |
| Fish and seafood (pesce e frutti di mare) | 0.78 (0.57 to 1.07) |
| Sweets (Torte, crostate, cioccolata, prodotti confezionati) | 0.89 (0.58 to 1.37) |
| First 5 foods after backward selection | (per one frequency per day, mutually adjusted) |
| Fatty meat (Carne grassa) | 0.64 (0.39 to 1.06) |
| Fatty ham, baked ham (Carne grassa e cotta) | 0.59 (0.35 to 0.99) |
| Meat sauce (Sughi di carne, ragù) | 1.76 (0.97 to 3.20) |
| Sheep ricotta, fiordilatte, mozzarella, smoked provola (Ricotta di pecora, fiordilatte, mozzarella, provola affumicata) | 1.41 (1.04 to 1.92) |
| Eggs (Uova) | 0.62 (0.41 to 0.94) |
| First 5 RSF foods with highest importance | (per one frequency per day, mutually adjusted) |
| Semolina type and other bread (Pane bianco) | 0.94 (0.84 to 1.04) |
| Pasta, rice, polenta (Pasta, riso, polenta) | 0.93 (0.73 to 1.20) |
| Desserts (Dolci, crostate, etc.) | 0.96 (0.62 to 1.50) |
| Fatty meat (Carne grassa) | 0.63 (0.38 to 1.04) |
| Poultry, rabbit (Pollame, coniglio) | 0.99 (0.65 to 1.50) |
| Components of PCA | (per pca-score, not mutually adjusted) |
| Energy-Rich Foods Pattern | 0.96 (0.92 to 1.01) |
| Farmhouse Diet Pattern | 1.05 (1.00 to 1.10) |
| Sweets Pattern | 1.01 (0.95 to 1.08) |
| Winter Pattern | 0.97 (0.92 to 1.02) |
| Elderly Pattern | 1.01 (0.96 to 1.07) |
| A priori indices | (per index point, not mutually adjusted, excluding olive oil as covariate) |
| DASH | 1.03 (0.97 to 1.10) |
| MedDiet score | 0.98 (0.97 to 1.00) |
| MIND | 0.96 (0.92 to 1.00) |
| Italian MedDiet index | 0.95 (0.90 to 1.00) |
| Parameters | Total Mortality | Cancer Mortality | Cardiovascular and Stroke Mortality |
|---|---|---|---|
| HR (95% CI) | HR (95% CI) | HR (95% CI) | |
| Components PCA § | |||
| Farmhouse Diet Pattern | 1.05 (1.00 to 1.10) | 1.03 (0.93 to 1.13) | 0.98 (0.88 to 1.10) |
| Foods § | |||
| Fatty meat | 0.89 (0.76 to 1.05) | 0.97 (0.71 to 1.33) | 0.79 (0.55 to 1.15) |
| Dairy products | |||
| Sheep and cow ricotta, fiordilatte, mozzarella, smoked provola | 1.29 (1.03 to 1.63) | 1.42 (0.90 to 2.22) | 0.96 (0.57 to 1.61) |
| Eggs | 0.86 (0.76 to 0.99) | 0.87 (0.66 to 1.13) | 0.73 (0.55 to 0.98) |
| Fatty ham, baked ham | 0.59 (0.35 to 0.99) | 0.90 (0.35 to 2.32) | 0.44 (0.14 to 1.45) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zupo, R.; Sardone, R.; Donghia, R.; Castellana, F.; Lampignano, L.; Bortone, I.; Misciagna, G.; De Pergola, G.; Panza, F.; Lozupone, M.; et al. Traditional Dietary Patterns and Risk of Mortality in a Longitudinal Cohort of the Salus in Apulia Study. Nutrients 2020, 12, 1070. https://doi.org/10.3390/nu12041070
Zupo R, Sardone R, Donghia R, Castellana F, Lampignano L, Bortone I, Misciagna G, De Pergola G, Panza F, Lozupone M, et al. Traditional Dietary Patterns and Risk of Mortality in a Longitudinal Cohort of the Salus in Apulia Study. Nutrients. 2020; 12(4):1070. https://doi.org/10.3390/nu12041070
Chicago/Turabian StyleZupo, Roberta, Rodolfo Sardone, Rossella Donghia, Fabio Castellana, Luisa Lampignano, Ilaria Bortone, Giovanni Misciagna, Giovanni De Pergola, Francesco Panza, Madia Lozupone, and et al. 2020. "Traditional Dietary Patterns and Risk of Mortality in a Longitudinal Cohort of the Salus in Apulia Study" Nutrients 12, no. 4: 1070. https://doi.org/10.3390/nu12041070
APA StyleZupo, R., Sardone, R., Donghia, R., Castellana, F., Lampignano, L., Bortone, I., Misciagna, G., De Pergola, G., Panza, F., Lozupone, M., Passantino, A., Veronese, N., Guerra, V., Boeing, H., & Giannelli, G. (2020). Traditional Dietary Patterns and Risk of Mortality in a Longitudinal Cohort of the Salus in Apulia Study. Nutrients, 12(4), 1070. https://doi.org/10.3390/nu12041070











