1. Introduction
Though they are considered weeds and are rarely a cultivated crop, nettles grow in a broad range of climate conditions and are widespread throughout Europe and North America, North Africa, and in parts of Asia. Nettles (genus
Urtica) have been used for centuries in traditional medicine in several cultures and have been widely studied as intervention for several medical conditions. Dietary supplements based on
Urtica spp extracts are widely available. About 25 different species exist within genus Urtica.
Urtica dioica is the species commonly found in North America, New Zealand, Turkey, and Europe [
1]. Actively growing
U. dioica shoots are harvested before flowering for consumption as an herb or spinach alternative [
2,
3].
Most studies on
U. dioica have been performed using its ethanolic or water extract particularly the extract of the leaves and stem which is rich in polyphenols. This extract has been shown to be richer in individual polyphenols than other plants such as dandelion and cranberry juice [
1,
3].
U. dioica extract has been studied for several different disease conditions including autoimmune diseases, such as rheumatoid arthritis, allergy, eczema, atherosclerosis, cancer, as well as age-related degenerative brain disorders. It has also been studied for alleviating bladder disorders, hemorrhaging and inflammation [
4,
5,
6]. More recent studies show that
U. dioica extract possess anti-diabetic properties [
5,
6,
7]. A comprehensive review on studies using the extract as intervention for diabetes shows that over 21 studies [
7]. Obesity induced insulin resistance caused by accumulation of certain lipid metabolites is a key pathophysiologic feature of type 2 diabetes mellitus (T2DM) [
8,
9]. Progression of insulin resistance into overt type 2 diabetes can be prevented or delayed by timely intervention. Treatment of insulin resistance is directed at increasing insulin sensitivity in peripheral tissues such as skeletal muscle and adipose tissue. Because of the high societal and economic cost of treating diabetes, timely intervention for insulin resistance has important medical, economic, social, and human implications. Therefore, conventional medications that address insulin resistance are a major focus for drug development [
10].
Our previous studies using cell culture models showed that
U. dioica extract activates the proteins AKT1 and AKT2 which are components of the insulin signaling pathway by enhancing their phosphorylation in presence of excess fat metabolites [
8,
9]. Rather than use the extract, in this study we incorporated whole
U. dioica vegetable in the diet. Our laboratory focus is on whole foods or class of foods that promote health rather than purified supplements. We thus designed a diet to focus on whole-vegetable effects and formulated the control diets to be isocaloric to the diet containing the vegetable. Furthermore, we tested the efficacy of the vegetable in (i) preventing fat accumulation and insulin resistance when used from the beginning of the study or (ii) reversing fat accumulation and insulin resistance by including it in the diet after obesity is induced with the high fat (HF) diet. We report on effects of the vegetable on a HF diet induced fat accumulation, insulin resistance, inflammation, endotoxemia and the expression of genes encoding proteins that are involved or impact the insulin signaling pathway in the skeletal muscle of C57BL/6J mice.
2. Materials and Methods
2.1. Acquisition of Plant Sample
U. dioica L. (UT) from spring growth was harvested as the total herb above the root mass. The plant was collected from the Green Farmacy Garden a medicinal garden located in Fulton Maryland; latitude 39.1454180, longitude −76.9213600. A voucher sample was deposited at the University Of Maryland herbarium. The stem and leaves were washed, chopped, oven dried (50 °C) for one day, and powdered using a coffee grinder. The powdered sample was subjected to nutrition analysis before being incorporated into the diets.
2.2. Nutrition Analysis
The dried powdered sample was analyzed by Medallion Labs (Minneapolis, MN) using the Association of Analytical Chemists (AOAC) methods for calorific value and proximate analysis: moisture, ash, total lipid, protein, carbohydrate, soluble fiber, insoluble fiber, and fatty acid profile. A list of methods used are shown in
Supplementary Table S1.
2.3. Formulation of Study Diets
We formulated three study diets, the low fat (LF) control, HF control and the HF containing 9%
U. dioica (HFUT). All diets were formulated to be isocaloric (3961 kcal/Kg) as shown in
Table 1. Diets were formulated to be have the same amount of sucrose and protein considering that the
U. dioica used in this study had 16.1% protein. The LF diet contained 10% fat while the HF and HFUT contained 45% fat.
2.4. Study Animals
All animal experiments and procedures were performed in accordance to a protocol approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Maryland. Thirty six (36) male C57BL/6J mice at 7 weeks age were ordered from Jackson Laboratories, Inc. (Bar Harbor, Maine, USA). All mice were singly housed in shoebox cages with corncob bedding in controlled environmental conditions (22 °C), 12 h light dark cycle with ad libitum access to food and water.
2.5. Randomization and Feeding Regime
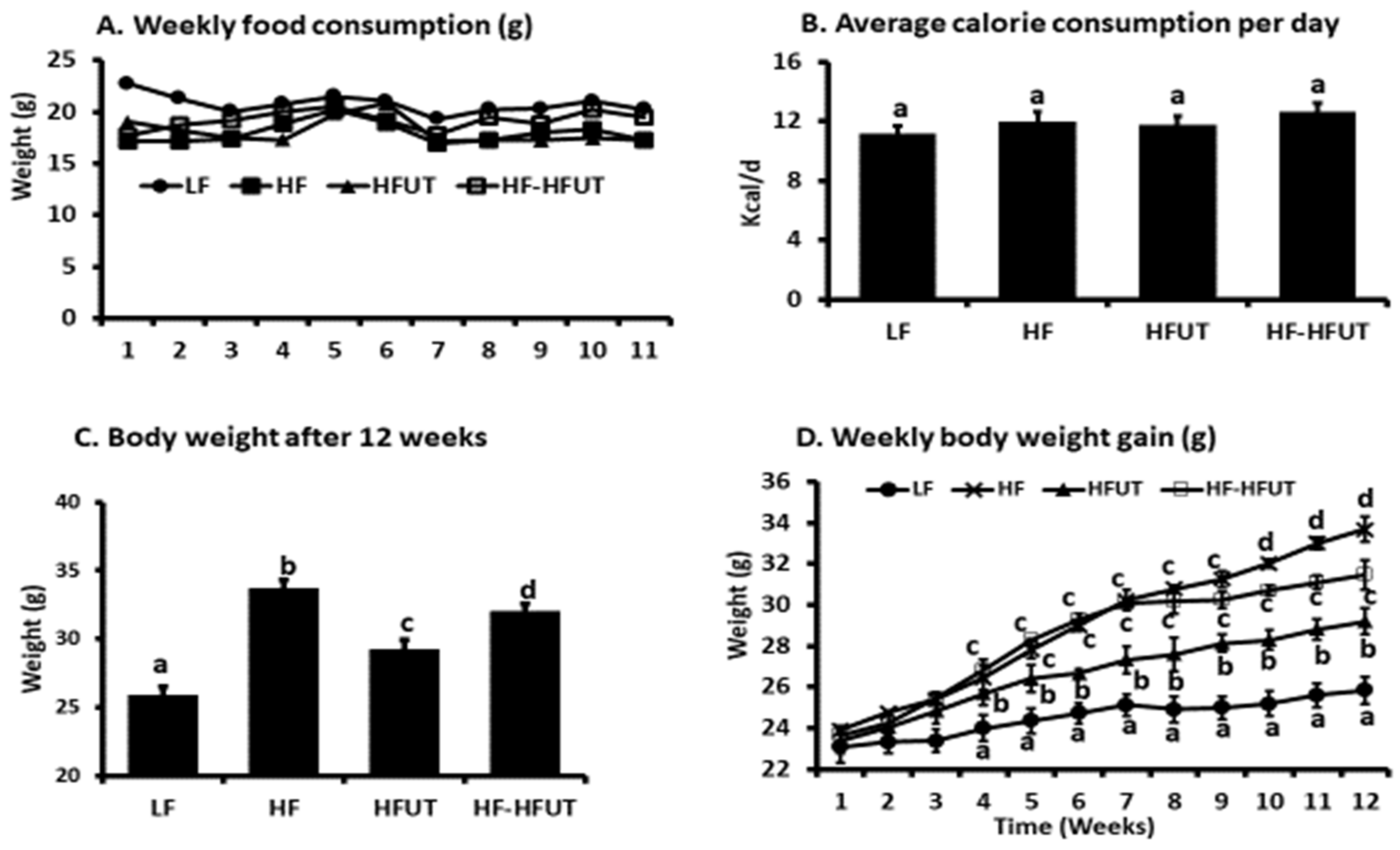
After one week baseline feeding of all mice on the LF diet, mice were randomized based on body weight and the homeostatic model assessment of insulin resistance (HOMA-IR) into four groups of 9. Group 1 was fed the LF diet for 12 weeks, group 2 was fed the HF diet for 12 weeks, group 3 was fed the HF diet containing
Urtica dioica vegetable (HFUT) diet for 12 weeks and, group 4 was fed the HF diet for 6 weeks and then switched to the HFUT diet for the next 6 weeks. Food intake (the difference of weight administered and leftover plus spillage) and body weight were monitored and recorded twice weekly. Sample size of
n = 9 for each treatment group was used based on a power analysis in our previous animal study on obesity induced insulin resistance [
9] which showed 8 to be a number sufficient to generate statistically significant results. To attain a statistical power of 0.90, 8 samples are needed for a one tailed test and 10 samples for a two tailed test.
2.6. Determination of Insulin Sensitivity
Fasting (6 h) plasma glucose and insulin were determined at baseline and after 6 and 12 weeks of feeding. Blood was collected by mandibular bleeding. Insulin levels were determined by a mouse ELISA kit (Crystal Chem, Downers Grove, IL). Blood glucose was measured using a portable glucometer (Milipitas, CA, USA). Insulin resistance was assessed by calculating HOMA-IR using the formula: HOMA-IR = fasting glucose (mg/dL) × fasting insulin (ng/mL)/405 as shown before [
11].
2.7. Tissue Collection and Processing
At 20 weeks of age, following 12 weeks of dietary intervention, animals were anesthetized by isoflurane inhalation. Terminal blood was collected by heart puncture followed by euthanasia by cervical dislocation. Serum was separated and stored at −80 °C to be used in later tests. Abdominal fat pads (epididymal, perirenal, and retroperitoneal) were excised and their weight was determined and summed as total abdominal fat. All tissues collected were snap frozen in liquid nitrogen and then stored at −80 °C for later analysis.
2.8. Histopathology
For histopathology, tissue samples were collected and fixed in 10% buffered formalin or snap frozen in liquid nitrogen. The samples fixed in formalin were dehydrated in alcohol and embedded in paraffin before sectioning at a thickness of 2–3 μm. Sections were stained with Oil red O (ORO). The liver snap frozen samples were cut directly in 3–4 μm sections and stained with ORO without any prior fixation (alcohol).
2.9. Analysis of Triglycerides in Liver and Colon Contents
About 100 mg of colon contents or 150 mg of liver were separately homogenized in ND40 reagent containing protease inhibitors and analyzed by a triglyceride kit (Cayman Chemical, Ann Arbor MI, USA). Briefly, after centrifuging at 4 °C, the supernatant was diluted 3-fold and reacted with lipoprotein lipase to release glycerol and fatty acids. The glycerol was then quantified by a colorimetric enzymatic reaction according the kit manufacturer’s instructions.
2.10. Serum Analysis
Serum samples were analyzed for total triglycerides using a colorimetric kit (Cayman Chemical, Ann Arbor, MI) according to manufactures specifications. Serum LDL cholesterol and HDL cholesterol were quantified using mouse ELISA kits. Endotoxin levels were determined by quantifying serum LPS levels using a mouse ELISA kit. All ELISA kits were from (Cusabio, Wuhan China).
2.11. RNA Extraction and cDNA Synthesis
RNA from 50 mg of gastrocnemius skeletal muscle, and liver and 100 mg adipose (epididymal fat pad) was separately extracted and purified using the RNAeasy mini kit (Qiagen, German Town, MD, USA) according to the manufacturer’s specifications. Prior to extraction the samples were disrupted in TRIzol with bead beating using the FastPrep®-24 (MP Biomedical, Solon, Ohio, USA). Both RNA quantity and quality were determined using the Qubit 4 fluorometer (Thermofisher Scientific, RockVille, MD, USA).
Only RNA samples with a concentration over 2.3 ng/uL and an RNA integrity number greater than 7.8 were used for cDNA synthesis using the RT2 first strand kit (Qiagen, German Town, MD, USA) with 1000 ng as starting RNA per sample. Similarly, only an RNA with an integrity number greater than 7.8 was used in cDNA synthesis for qPCR of targeted genes.
2.12. Analysis of Target Genes in Skeletal Muscle, Adipose Tissue, and Liver by qPCR
Primer sets (IDT Technologies, Coralville IA, USA) were used for analyses of selected genes. The genes selected for this analysis were not exhaustive but we selected genes that have been shown to impact adipogenesis, lipogenesis, fatty acid oxidation, and gluconeogenesis. RT-PCR cycling conditions on the CFX 96 (Bio-rad, Hercules CA, USA), were 2 min at 50 °C and 2 min at 95 °C, followed by 40 cycles of two-step PCR denaturation at 95 °C for 15 s and annealing extension at 60 °C for 1 min. Duplicate assay samples contained 10 ng cDNA and 6 μmol/L primers in 2× PowerUp™ SYBR™ Green Master Mix (Thermofisher Scientific, RockVille, MD, USA) in a final volume of 20 μL. Means of duplicates were taken, and relative amount of target mRNA was normalized to β-actin levels as an endogenous control gene. Data were analyzed according to the 2
−ΔΔCT method, and fold difference was calculated between LF, HF, and HFUT groups. A list of primers used is shown in
Supplementary Table S1.
2.13. Analysis of Insulin Signaling Pathway Genes, by RT2 Profiler PCR Array
The differential expression of 84 genes that are involved in, respond to or impact the insulin signaling pathway was determined using the configured RT2 Profiler TM PCR Array (Qiagen, German Town, MD, USA), focused on the insulin signaling pathway. About 1000 ng of cDNA from each sample was mixed with SYBR green qPCR master mix and the mixture was then aliquoted in wells of the RT2 profiler PCR array. The array was centrifuged for 1 min at 1000× g at room temperature before qPCR. Cycling conditions on the CFX 96 (Bio-rad, Hercules CA, USA), were one cycle of 95 °C for 1 min, 40 cycles of 15 s at 95 °C and 60 °C for 60 s. A melting curve analysis was used to verify PCR specificity. The threshold cycle (Cq) values for each gene and that of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as reference gene were used to calculate ΔΔCq values and relative gene expression was determined by calculating 2-ΔΔCT. For genes that were downregulated, a reciprocal of 2−ΔΔCT was calculated. Results were also analysed by the GeneGlobe data analysis center (Qiagen, German Town, MD, USA).
2.14. Statistical Analyses
Data are expressed or graphed as mean ± SEM. Differences between two groups were assessed using the unpaired two tailed student t-test. Data sets with more than two groups were assessed using the MIXED procedure of SAS 9.4 for analysis of variance between treatments. Main effects were considered significant at p ≤ 0.05. Data was analyzed for outliers using studentized residuals. Significant differences observed were followed up using the Bonferroni test of multiple comparisons.
4. Discussion
Although diet supplements based on
Urtica dioica extracts are widely available in the United States, not much is known about the benefits of this plant when consumed as a whole vegetable. In many international cultures
U. dioica shoots are harvested before flowering for use as an herb or spinach alternative. Some recipes incorporate
U. dioica leaf flour in bread, pasta, and noodle dough. Besides being high in polyphenols, on a dry weight basis,
U. dioica leaf is comparable to common bean (
Phaseolus vulgaris) and chicken as a source of essential amino acids [
1,
2,
3]. In the current study, we show that including the vegetable in a HF diet, attenuates fat accumulation in adipose tissue and whole-body insulin resistance. In our previous study [
8] in which we incorporated 0.5%
U. dioica ethanolic extract into the HF diet, we did not observe a reduction in body weight or body composition; the extract improved insulin signaling despite not having an effect on fat accumulation. Our current findings on reduction of body weight and fat accumulation when the whole vegetable is used instead of the extract, are thus novel and raise new questions on mechanisms involved. It also suggests that other components of the vegetable besides the phytochemicals in the ethanol extract may impact positive effects on body weight.
The benefits of the
U. dioica vegetable are likely attributed to the high fiber content of 53.3% (
Supplementary Table S2), phytochemical components, high protein content, unique fatty acid profile and high percentage of linoleic and linolenic acids (
Supplementary Table S3). The soluble and insoluble fiber may contribute to beneficial effects through the action of the gut microbiota. The wide range of compounds in the ethanolic extract have been elucidated by a number of researchers [
1,
12,
13] and have been shown to include the phenolic acids; p-hydroxybenzoic acid, gentisic acid, protocatechuic acid, vanillic acid, quinic acid, ferulic acid, p-coumaric acid, caffeic acid and, 5-
O-caffeolylquinic acid. The second large group of compounds are flavonols which include kaempferol, kaempferol 3-
O-glucoside, quercitrin, quercetin 3-
O-glucoside, quercetin 3-
O-rutinoside (rutin), and isorhamnetin. Others are the biflavonoids amentoflavone and the catechin a flavan-3-ol. All these phytochemicals and the fiber content may individually impact obesity and insulin resistance in ways that are yet unknown.
The connections between obesity and metabolic diseases are well established. Obesity leads to elevated levels of circulating free fatty acids that are converted into metabolites that induce insulin resistance [
8,
9] and leads to low-grade elevation in plasma of gut derived lipolysaccharide (LPS) (metabolic endotoxemia) [
14,
15]. When bound to its receptor Toll-Like Receptor 4, LPS stimulates whole-body and tissue specific metabolic perturbations by initiating a signaling cascade that results in pro-and anti-inflammatory pathways and initiates obesity and insulin resistance. The HF diet significantly raised LPS levels in blood but supplementation with
U. dioica vegetable did not lower LPS levels (
Table 2). It is likely that the vegetable attenuates fat accumulation and insulin resistance by mechanisms not related to bacterial LPS.
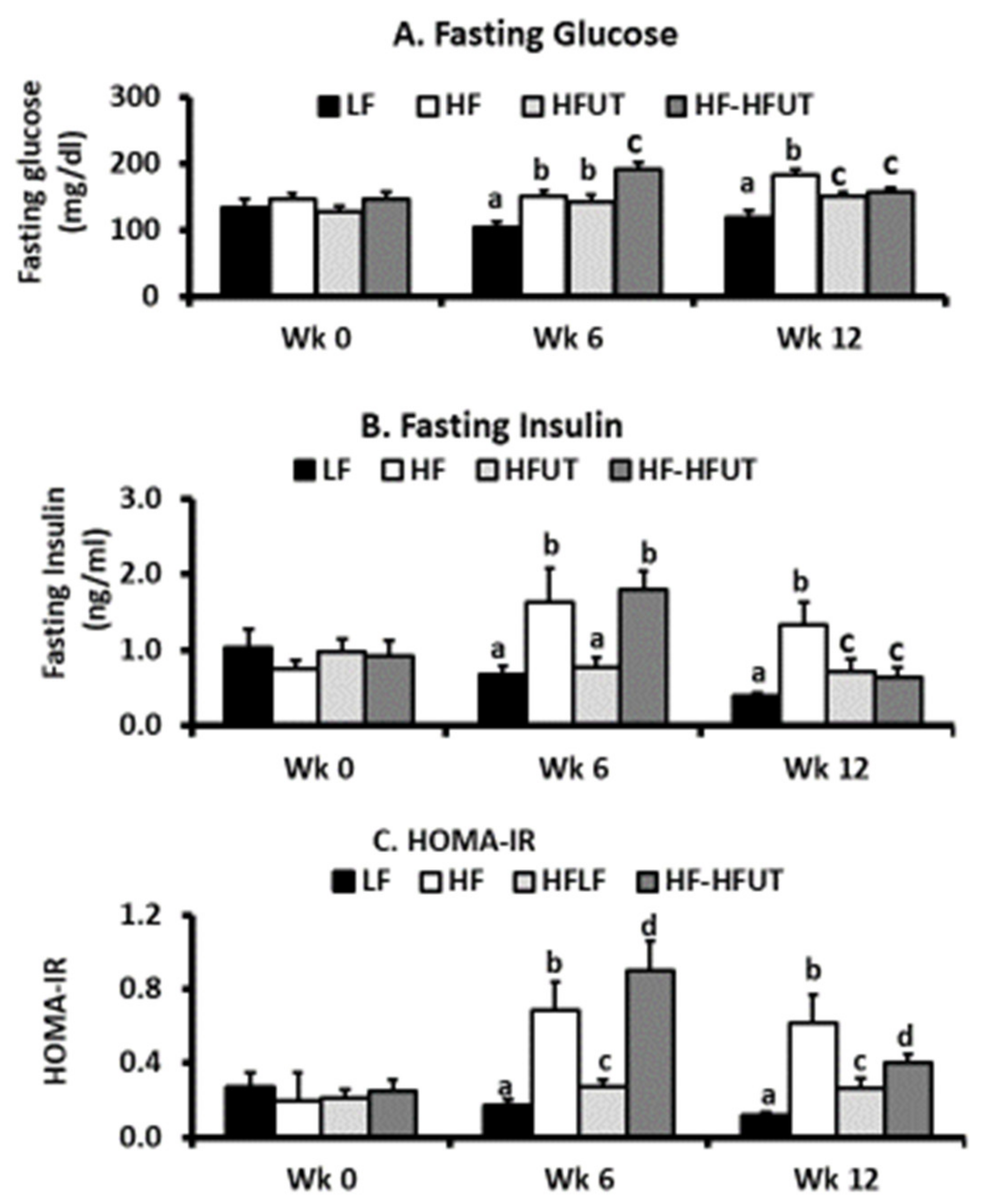
In agreement with our previous study using the water/ethanol extract of
U. dioica [
8], UT vegetable prevented the development of insulin resistance in mice fed the HF diet and also reversed insulin resistance when obese mice were switched to the vegetable containing diet (
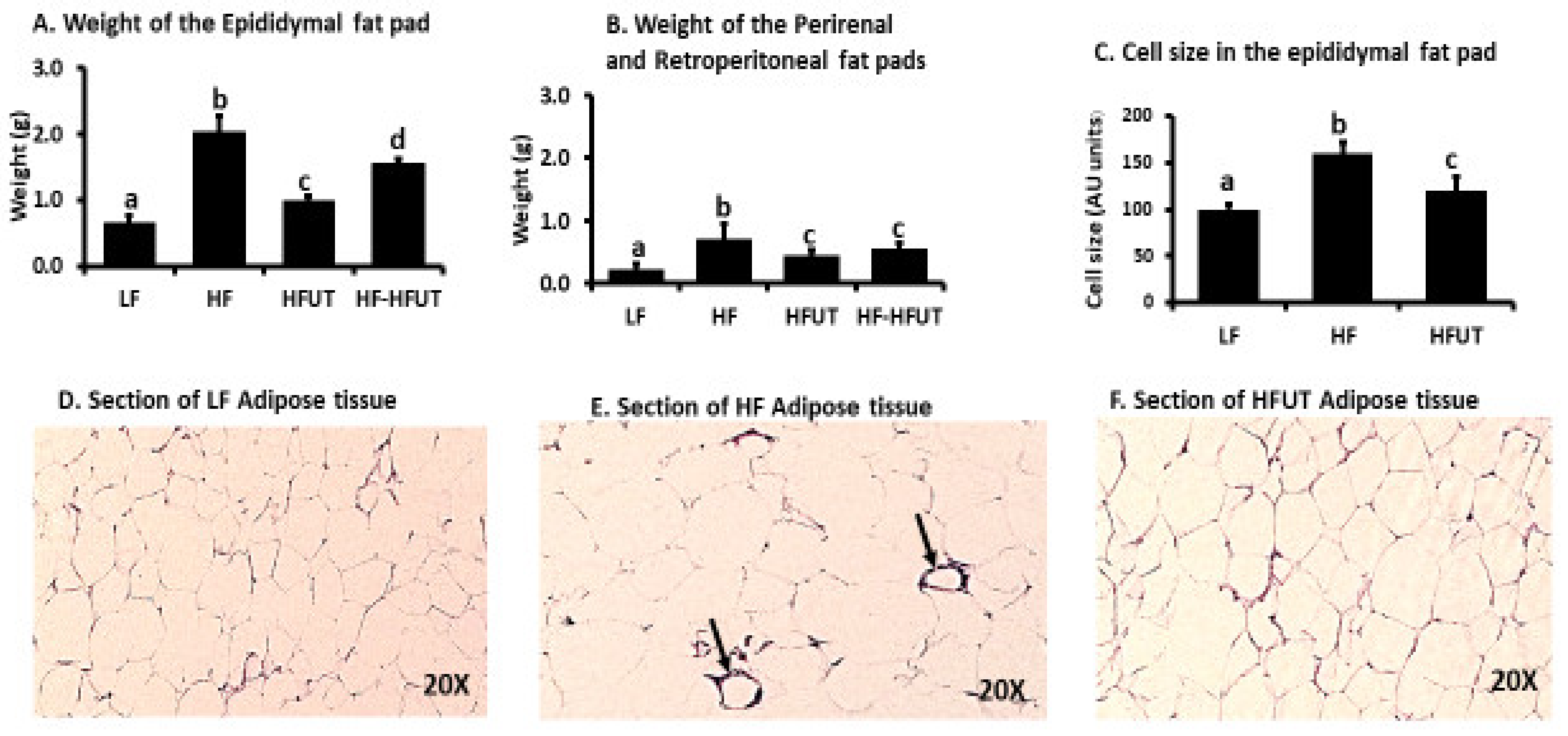
Figure 2). The HFUT diet significantly attenuated fat accumulation in adipose tissue and resulted in smaller adipocytes with no evidence of inflammatory markers (crown like structures) (
Figure 3). This is in line with previous work which shows that when adipocytes enlarge (hypertrophy) in obesity, they release TNFα and insulin-like growth factor which stimulate hyperplasia. Reversal of obesity decreases the number and size of the adipocytes followed by apoptosis of adipocytes [
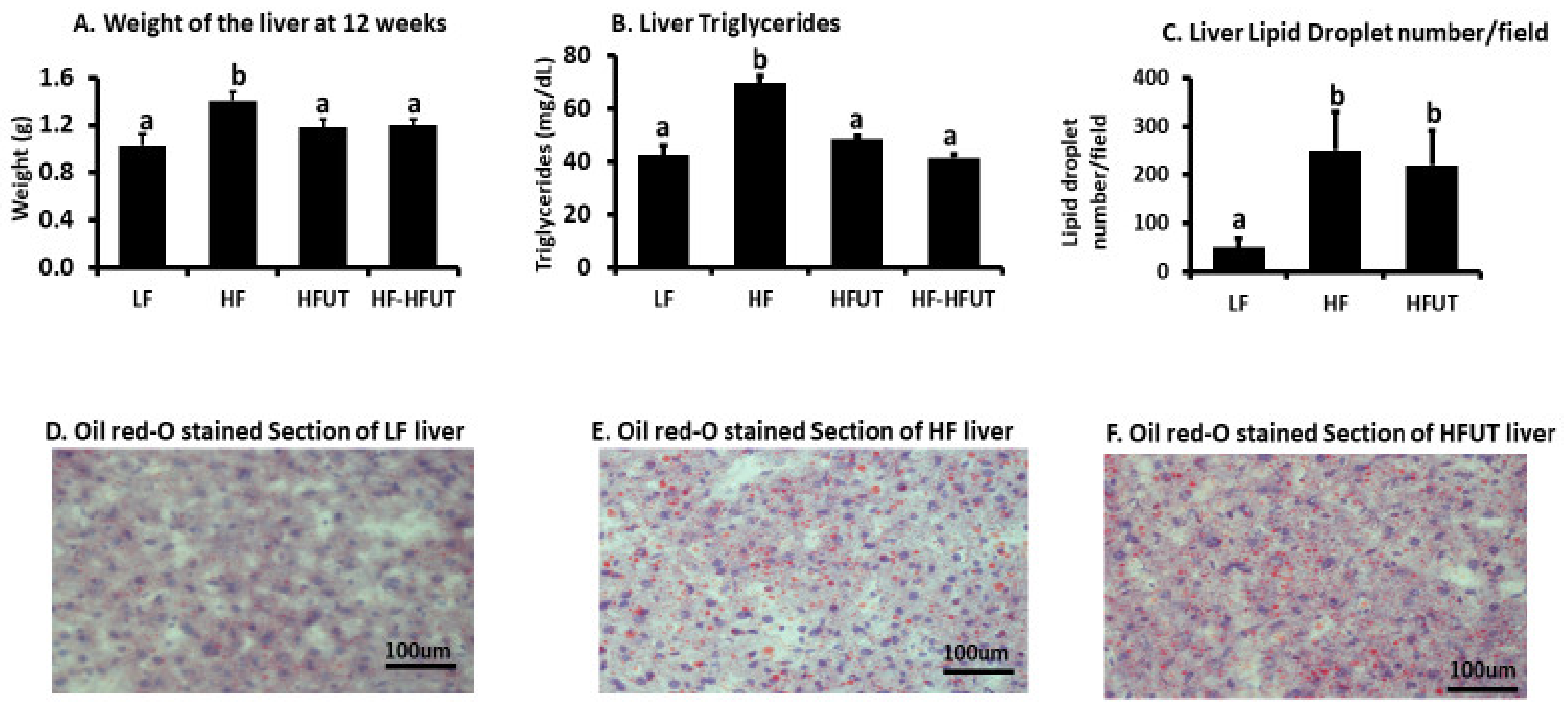
16]. The positive results of fat accumulation in liver were inconclusive; while liver weight and liver triglycerides were lowered by the
U. dioica (
Figure 4), the effect on serum triglycerides and cholesterol levels was not different from those of the HF diet (
Table 2). However, the mice were not fasted before euthanasia. The results may have been different if terminal blood and tissues were collected after fasting. An unexpected observation was the higher amount of LDL cholesterol in the LF diet compared to the HF diet. The LF diet had a higher amount of carbohydrates compared to the HF diet. It has been previously shown that increased dietary carbohydrates, particularly simple sugars and starches with high glycemic index, can increase levels of small, dense LDL and HDL [
17]. The increased triglycerides observed in colon contents of HF diet fed mice was not lowered by the diet with
U. dioica vegetable (
Table 2). It is likely that while
U. dioica impacts fat metabolism and assimilation in ways that are yet unknown, it does not affect fat digestion or absorption in the digestive tract.
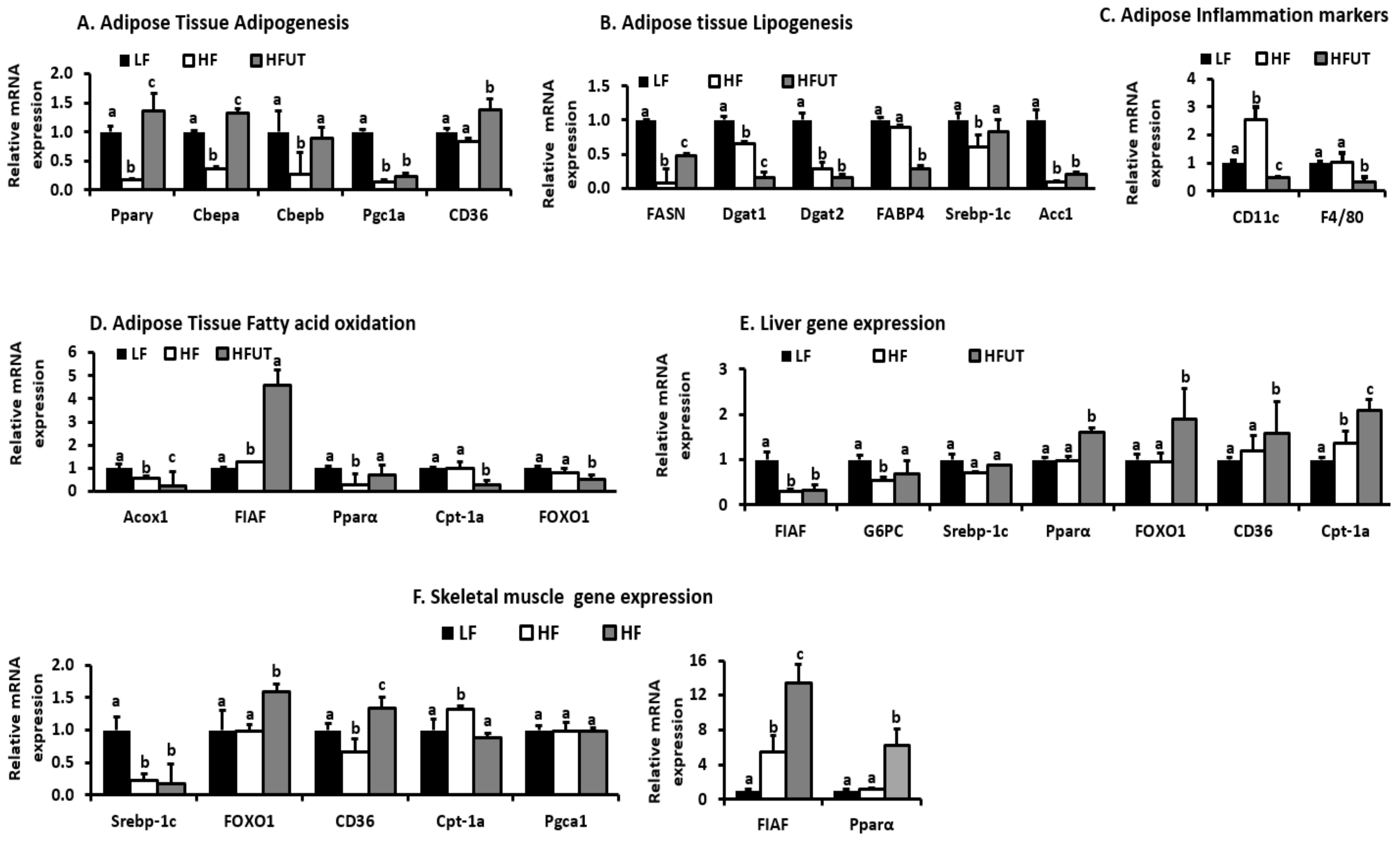
We evaluated the effect of HFUT on the expression of genes that encode proteins involved in inflammation, fatty acid transport, fatty acid oxidation, adipogenesis, and lipogenesis. The HF diet induced a more than 2-fold increase in CD11c compared to the LF diet group. The HFUT reversed this and down regulated CD11c to levels below those of the LF diet group. HFUT also reduced expression of F4/80 another marker of inflammation in adipose (
Figure 5C). A specific subset of macrophages that expresses CD11c and produces high levels of pro-inflammatory cytokines is recruited to obese adipose and muscle tissue. These macrophages are linked to the development of obesity-associated insulin resistance. Using a conditional cell ablation system, to deplete CD11c
+ cells in obese mouse models Patsouris et al. [
18] showed that depleting CD11c
+ cells results in rapid normalization of insulin sensitivity and leads to a marked decrease gene expression and protein levels of inflammatory markers. CD11c plays an important role in T-cell accumulation and activation in adipose tissue contributes to insulin resistance associated with obesity. CD11c messenger RNA positively correlates with MCP-1 in visceral adipose of obese humans with metabolic syndrome compared with lean humans [
19]. Thus CD11c
+ cells are a potential therapeutic target for treatment of obesity-related insulin resistance and type II diabetes.
Compared with the HF diet, HFUT increased the expression of genes that are involved in fatty acid transport and oxidation in both adipose tissue and skeletal muscle. Specifically, the expression of Angiopoietin-like protein or fasting induced adipocyte factor (FIAF) increased more than 4 fold in adipose and by 13 fold in muscle. FIAF is a circulating inhibitor of lipoprotein lipase and restrains its ability to import and store fatty acids in peripheral tissues. Furthermore, FIAF expression regulates energy expenditure via modulation of AMP-activated protein kinase (AMPK) activity in muscle and adipose. Elevated FIAF levels have been shown to protect against diet-induced obesity [
20]. This may be a contributing factor in the lower fat accumulation observed in HFUT fed mice. The HFUT increased the expression of Ppar-α in skeletal muscle by more than 6-fold and increased it by 1.6 fold in liver. In adipose tissue, the HFUT prevented the HF induced reduction in expression of Ppar-α and increased Ppar-γ expression by 1.4 fold (
Figure 5D–F). Activators of both PPARα and PPARγ have been shown to improve insulin sensitivity and normalize impaired glucose tolerance in both humans and rodent models of IR. In liver, PPAR-α regulates genes involved in lipid and lipoprotein metabolism. When activated, it promotes fatty acid oxidation by inducing expression of mitochondrial acyl-CoA dehydrogenases, acetyl-CoA production, promotes glucose sparing and ketone body synthesis by upregulating mitochondrial hydroxymethylglutaryl-CoA synthase (HMGS), a rate-limiting ketogenesis enzyme [
21,
22]. It also reduces pyruvate kinase, induces PDK4 expression and reduces weight gain in rodents [
21,
22]. PPARα activation further reduces plasma triglyceride-rich lipoproteins by increasing the activity of LPL, which hydrolyzes lipoprotein triglycerides. Thus, through enhanced fatty acid oxidation, UT vegetable may impact fat accumulation mechanisms and result in the leaner phenotype. The HF diet lowered PPARγ expression and the HFUT prevented this and enhanced the expression to levels higher than those of the LF group. PPARγ is highly expressed in adipose tissue where its activation alters fat topography, adipose phenotype and upregulates genes involved in fatty acid metabolism and triglyceride storage.
In liver and muscle, the HFUT diet increased FOXO1 gene by almost 2 fold (
Figure 5E,F). FOXO proteins are important in metabolism and energy homeostasis. FOXO1 is particularly inhibits hepatic gluconeogenesis by insulin action. Insulin inhibits FOXO1 activity through the PI3K/ AKT signaling pathway resulting in an inhibition of gluconeogenesis by suppressing expression of glucose-6-phosphatase (G6PC) and Pck1 [
23]. FOXO1 stimulates fatty acid uptake and oxidation in muscle cells. Other genes involved in fatty acid oxidation and energy expenditure and impacted by HFUT were fatty acid translocase (FAT/
CD36), carnitine palmitoyl transferase (CPT-1a) (by more than 2 fold in liver), FABP and Acc2. Overexpression of Cpt1 in skeletal muscle has been shown to enhance fatty acid oxidation and improve high-fat diet-induced insulin resistance [
24]. The expression of CD36 was enhanced by HFUT in all three tissues. CD 36 promotes adipocyte differentiation and adipogenesis.
Overall, the HFUT diet increased the expression of genes involved in adipogenesis (Pparγ, CEBPα, SREBF1c, FABP4, FASN, and CD36), lowered genes involved in lipogenesis (Acc1, Dgat1, Dgat2, FABP) and increased genes involved in fatty acid oxidation (FIAF, Pparα, Cpt-1). Thus the combination of reduction in lipogenesis genes and increase in and fatty acid oxidation genes contributed to the leaner frame in HFUT fed mice.
The analysis of genes that impact the insulin signaling pathway in skeletal muscle using the RT
2 array showed that HFUT diet did not induce significant changes in the expression in most of the 84 genes. (
Supplementary Table S4). In agreement with our previous study [
8] we found no significant change in the expression of AKT1, AKT2, and AKT3 isoforms and GLUT4 (Slc27a4). In our previous studies [
8,
9] we showed that
U. dioica extract enhanced insulin signaling by enhancing the phosphorylation of AKT1 and AKT2 but had no effect on their gene or protein expression. The notable few changes observed using the RT
2 array include the reduction in uncoupling protein 1 (mitochondrial proton carrier) (UCP1), increase in eukaryotic translation initiation factor 4E binding protein 1 and increase in glucose-6-phosphatase, catalytic (GPC6). These genes are expressed in very low amounts in skeletal muscle and hence their changed expression may not impact mechanisms. Furthermore, we were unsuccessful in confirming the changed expression of UCP-1 and G6PC using qPCR even after using primers validated by IDT. The results of Srebp1C were replicated in the qPCR assays. The HFUT diet downregulated glucose 6-phosphatase (G6PC), the gene that encodes the enzyme that dephosphorylates the glucose molecule to enable its transport out of the cells. Primarily expressed in the liver, the enzyme is present in muscle cells in very low amounts. The downregulation of this gene by HFUT diet may augment its role in keeping glucose within the muscle cells and thus result in lower blood glucose levels. The HF diet lowered the levels of eukaryotic translation initiation factor 4E binding protein 1 (Eif4ebp1) gene but supplementing the HFUT prevented this. Increased insulin resistance in 4E-BP1 and 4E-BP2 KO mice is linked to increased ribosomal protein S6 kinase (S6K) activity and impairment of AKT signaling in muscle, liver, and adipose tissue [
25]. The HFUT diet downregulated UCP1 in skeletal muscle. UCP1 is mainly expressed in brown adipose tissue where its role in thermogenesis has been widely studied. Although it is now known that UCP1 is also expressed in white adipose and skeletal muscle, its physiological functions in these tissues is not well established and different studies had shown conflicting results. Our findings on downregulation of UCP1 in skeletal muscle contrast with a number of studies that have linked overexpression of UCP1 to increased energy expenditure, reduced fat mass and improved glucose tolerance [
26,
27,
28]. However, the overexpression of UCP1 does not reduce susceptibility to obesity when the mice are not challenged thermogenically [
26]. In fact, obesity-resistant UCP1-deficient mice have increased resistance to obesity due to the inactivation of a major thermogenic mechanism that results in an increased expenditure of energy [
26].
Our data clearly shows a relationship between the effects of U. dioica vegetable and favorable metabolic parameters but raises several unanswered questions. The limitation of our study is that we assess mRNA changes (transcript abundance) as a proxy for protein levels but have not yet quantified changes in the proteins encoded by these genes. Protein expression levels are highly regulated and protein levels are more conserved than the mRNA levels. In addition to changes in mRNA expression, differences in the translational efficiency of the mRNAs are important. In further studies we shall evaluate protein expression levels, identify the active plant components responsible for the positive effects and metabolites produced in tissues to gain a better understanding of the potential of U. dioica as a metabolically favorable functional whole food for obesity and insulin resistance.