Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults
Abstract
1. Prebiotics
2. Prebiotics and Dietary Fibers
3. Mechanisms of Action of Prebiotics
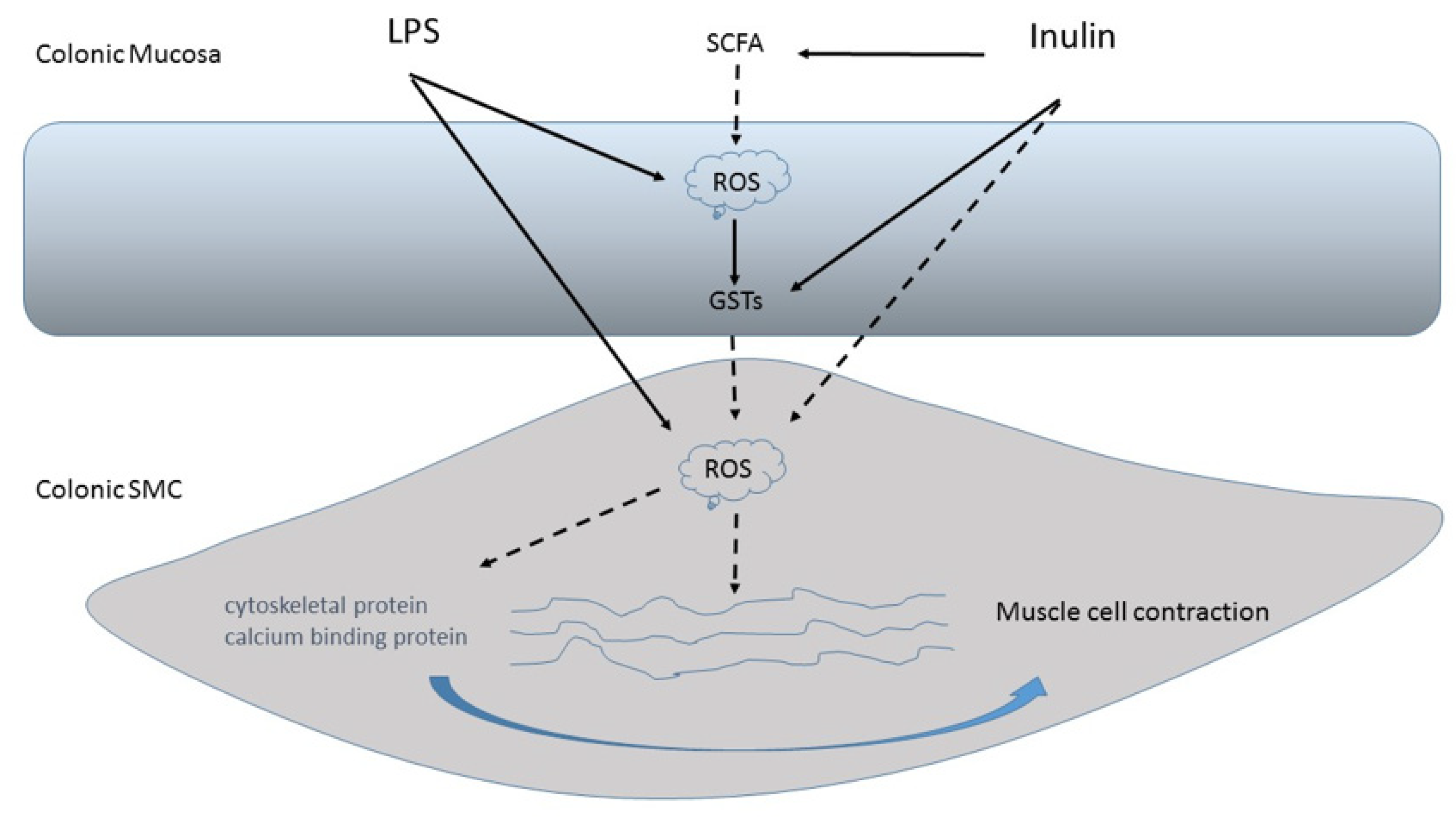
3.1. Fructans
3.2. GOS
3.3. Lactulose
3.4. New Molecules with Prebiotic Effects
4. Prebiotics and Gastrointestinal Disorders
5. Side Effects
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gibson, G.R.; Roberfroid, M. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1402. [Google Scholar] [CrossRef] [PubMed]
- Pineiro, M.; Asp, N.G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. Fao Technical Meeting on Prebiotics. J. Clin. Gastroenterol. 2008, 42, S156–S159. [Google Scholar] [CrossRef] [PubMed]
- Ministero Della Salute. Linee Guida su Probiotici e Prebiotici. Revisione Marzo 2018. Available online: https://sumailombardia.info/sanita-nazionale/ministero-salute/2018/05/07/linee-guida-su-probiotici-e-prebiotici-revisione-marzo-2018/ (accessed on 3 May 2018).
- De Vrese, M.; Schrezenmeri, J. Probiotics, prebiotics, and synbiotics. Adv. Biochem. Eng. Biotechnol. 2008, 111, 1–66. [Google Scholar] [PubMed]
- Macfarlane, S.; Macfarlane, G.; Cummings, J. Review article prebiotics in the gastrointestinal tract. Aliment. Pharmacol. Ther. 2006, 24, 701–714. [Google Scholar] [CrossRef]
- Gibson, G.R.; Probert, H.M.; Van Loo, J.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef]
- Roberfroid, M. Prebiotics: The concept revisited. J. Nutr. 2007, 137, 830S–837S. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Macfarlane, S.; Gibson, G.R. Validation of a Three-Stage Compound Continuous Culture System for Investigating the Effect of Retention Time on the Ecology and Metabolism of Bacteria in the Human Colon. Microb. Ecol. 1998, 35, 180–187. [Google Scholar] [CrossRef]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, S1–S63. [Google Scholar] [CrossRef]
- Van Loo, J.; Coussement, P.; De Leenheer, L.; Hoebregs, H.; Smits, G. On the presence of inulin and oligofructose as natural ingredients in the western diet. Crit. Rev. Food Sci. Nutr. 1995, 35, 525–552. [Google Scholar] [CrossRef]
- Zaman, S.A.; Sarbini, S.R. The potential of resistant starch as a prebiotic. Crit. Rev. Biotechnol. 2016, 36, 578–584. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Kim, S.S. Probiotics and Prebiotics: Present Status and Future Perspectives on Metabolic Disorders. Nutrients 2016, 18, 173. [Google Scholar] [CrossRef] [PubMed]
- Lunet, N.; Lacerda-Vieira, A.; Barros, H. Fruit and vegetables consumption and gastric cancer: A systematic review and meta-analysis of cohort studies. Cancer 2005, 53, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes: Proposed Definition of Dietary Fiber; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Slavin, J.L. Position of the American Dietetic Association: Health Implications of dietary fiber. J. Am. Diet. Assoc. 2008, 108, 1716–1731. [Google Scholar] [PubMed]
- Howlett, J.F.; Betteridge, V.A.; Champ, M.; Craig Stuart, A.S.; Meheust, A.; Jones, J.M. The definition of dietary fiber—discussions at the Ninth Vahouny Fiber Symposium: Building scientific agreement. Food Nutr. Res. 2010, 54, 5750. [Google Scholar] [CrossRef]
- Alimentarius Commission. Guidelines on Nutrition Labelling CAC/GL 2-1985 as Last Amended 2017, Joint FAO/WHO Food Standards Programme; Secretariat of the Codex Alimentarius Commission, FAO: Rome, Italy, 2017. [Google Scholar]
- Lo Presti, A.; Zorzi, F.; Del Chierico, F.; Altomare, A.; Cocca, S.; Avola, A.; De Biasio, F.; Russo, A.; Cella, E.; Reddel, S.; et al. Fecal and Mucosal Microbiota Profiling in Irritable Bowel Syndrome and Inflammatory Bowel Disease. Front. Microbiol. 2019, 10, 1655. [Google Scholar] [CrossRef]
- Den Besten, G.; Van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef]
- Hiel, S.; Bindels, L.B.; Pachikian, B.D.; Kalala, G.; Broers, V.; Zamariola, G.; Chang, B.P.I.; Kambashi, B.; Rodriguez, J.; Cani, P.D.; et al. Effects of a diet based on inulin-rich vegetables on gut health and nutritional behavior in healthy humans. Am. J. Clin. Nutr. 2019, 109, 1683–1695. [Google Scholar] [CrossRef]
- Kleessen, B.; Hartmann, L.; Blaut, M. Oligofructose and long-chain inulin: Influence on the gut microbial ecology of rats associated with a human faecal flora. Br. J. Nutr. 2001, 86, 291–300. [Google Scholar] [CrossRef]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011, 5, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Van der Beek, C.M.; Dejong, C.H.C.; Troost, F.J.; Masclee, A.A.M.; Lenaerts, K. Role of of short-chain fatty acids in colonic inflammation, carcinogenesis, and mucosal protection and healing. Nutr. Rev. 2017, 75, 286–305. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y.; et al. Fructooligosaccharide (FOS) and Galactooligosaccharide (GOS) increase Bifidobacterium but reduce butyrate producing bacteria with adverse glycemic metabolism in healthy young population. Sci. Rep. 2017, 7, 11789. [Google Scholar] [CrossRef]
- Watson, D.; O’Connell Motherway, M.; Schoterman, M.H.C.; Joost van Neerven, R.J.; Nauta, A.; van Sinderen, D. Selective carbohydrate utilization by lattobacilli and bifidobacterial. J. Appl. Microbiol. 2012, 114, 1132–1146. [Google Scholar] [CrossRef]
- Kanner, J.; Lapidot, T. The stomach as a bioreactor: Dietary lipid peroxidation in the gastric fluid and the effects of plant-derived antioxidants. Free Radic. Biol. Med. 2001, 31, 1388–1395. [Google Scholar] [CrossRef]
- Busserolles, J.; Gueux, E.; Rock, E.; Demigné, C.; Mazur, A.; Rayssiguier, Y. Oligofructose protects against the hypertriglyceridemic and pro-oxidative effects of a high fructose diet in rats. J. Nutr. 2003, 133, 1903–1908. [Google Scholar] [CrossRef] [PubMed]
- Welters, C.F.M.; Heineman, E.; Thunnissen, F.B.J.M.; Van den Bogaard, A.E.J.M.; Soeters, P.B.; Baeten, C.G.M.I. Effect of dietary inulin supplementation on inflammation of pouch mucosa in patients with an ileal pouch-anal anastomosis. Dis. Colon Rectum 2002, 45, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.B.; Neyrinck, A.M.; Bindels, L.B.; de Vos, W.M.; Gibson, G.R.; Thissen, J.P.; et al. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut J. 2013, 62, 1112–1121. [Google Scholar] [CrossRef]
- Ramirez-Farias, C.; Slezak, K.; Fuller, Z.; Duncan, A.; Holtrop, G.; Louis, P. Effect of inulin on the human gut microbiota: Stimulation of Bifidobacterium adolescentis and Faecalibacterium prausnitzii. Br. J. Nutr. 2009, 101, 541–550. [Google Scholar] [CrossRef]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Wang, J.; Sailer, M.; Theis, S.; Verbeke, K.; Raes, J. Prebiotic inulin-type fructans induce specific changes in the human gut microbiota. Gut J. 2017, 66, 1968–1974. [Google Scholar] [CrossRef] [PubMed]
- Rivière, A.; Selak, M.; Lantin, D.; Leroy, F.; De Vuyst, L. Bifidobacteria and Butyrate-Producing Colon Bacteria: Importance and Strategies for Their Stimulation in the Human Gut. Front. Microbiol. 2016, 7, 979. [Google Scholar] [CrossRef] [PubMed]
- Bouhnik, Y.; Vahedi, K.; Achour, L.; Attar, A.; Salfati, J.; Pochart, P.; Marteau, P.; Flourié, B.; Bornet, F.; Rambaud, J.C. Short-chain fructo-oligosaccharide administration dose-dependently increases fecal bifidobacteria in healthy humans. J. Nutr. 1999, 129, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Paineau, D.; Bornet, F. The capacity of short-chain fructo-oligosaccharides to stimulate faecal bifidobacteria: A dose-response relationship study in healthy humans. Nutr. J. 2006, 5, 8. [Google Scholar] [CrossRef]
- Gibson, G.R.; Beatty, E.R.; Wang, X.; Cummings, J.H. Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterol 1995, 108, 975–982. [Google Scholar] [CrossRef]
- Ramnani, P.; Gaudier, E.; Bingham, M.; Van Bruggen, P.; Tuohy, K.M.; Gibson, G.R. Prebiotic effect of fruit and vegetable shots containing Jerusalem artichoke inulin: A human intervention study. Br. J. Nutr. 2010, 104, 233–240. [Google Scholar] [CrossRef]
- Kleessen, B.; Schwarz, S.; Boehm, A.; Fuhrmann, H.; Richter, A.; Henle, T.; Krueger, M. Jerusalem artichoke and chicory inulin in bakery products affect faecal microbiota of healthy volunteers. Br. J. Nutr. 2007, 98, 540–549. [Google Scholar] [CrossRef]
- Carotti, S.; Guarino, M.P.L.; Vespasiani-Gentilucci, U.; Morini, S. Starring role of toll-like receptor-4 activation in the gut-liver axis. World J. Gastrointest. Pathophysiol. 2015, 6, 99–109. [Google Scholar] [CrossRef]
- Pasqualetti, V.; Altomare, A.; Guarino, M.P.L.; Locato, V.; Cocca, S.; Cimini, S.; Palma, R.; Alloni, R.; De Gara, L.; Cicala, M. Antioxidant activity of inulin and its role in the prevention of human colonic muscle cell impairment induced by lipopolysaccharide mucosal exposure. PLoS ONE 2014, 9, e98031. [Google Scholar] [CrossRef]
- Van den Ende, W.; Valluru, R. Sucrose, sucrosyl oligosaccharides, and oxidative stress: Scavenging and salvaging? J. Exp. Bot. 2009, 60, 9–18. [Google Scholar] [CrossRef]
- Stoyanova, S.; Geuns, J.; Hideg, E.; Van den Ende, W. The food additives inulin and stevioside counteract oxidative stress. Int. J. Food Sci. Nutr. 2011, 62, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Guarino, M.P.L.; Altomare, A.; Barera, S.; Locato, V.; Cocca, S.; Franchin, C.; Arrigoni, G.; Vannini, C.; Grossi, S.; Campomenosi, P.; et al. Effect of Inulin on Proteome Changes Induced by Pathogenic Lipopolysaccharide in Human Colon. PLoS ONE 2017, 12, e0169481. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J. Effect of resistant starch on fecal bulk and fermentation dependent events in humans. Am. J. Clin. Nutr. 1995, 62, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Miene, C.; Weise, A.; Glei, M. Impact of Polyphenol Metabolites Produced by Colonic Microbiota on Expression of COX-2 and GSTT2 in Human Colon Cells (LT97). Nutr. Cancer 2011, 63, 653–662. [Google Scholar] [CrossRef]
- Brummer, Y.; Kaviani, M.; Tosh, S.M. Structural and functional characteristics of dietary fibre in beans, lentils, peas and chickpeas. Food Res. Int. 2015, 67, 117–125. [Google Scholar] [CrossRef]
- Vulevic, J.; Rastall, R.A.; Gibson, G.R. Developing a quantitative approach for determining the in vitro prebiotic potential of dietary oligosaccharides. Fems Microbiol. Lett. 2004, 236, 153–159. [Google Scholar] [CrossRef]
- Depeint, F.; Tzortzis, G.; Vulevic, J.; I’Anson, K.; Gibson, G.R. Prebiotic evaluation of a novel galactooligosaccharide mixture produced by the enzymatic activity of Bifidobacterium bifidum NCIMB 41171, in healthy humans: A randomized, double-blind, crossover, placebo-controlled intervention study. Am. J. Clin. Nutr. 2008, 87, 785–791. [Google Scholar] [CrossRef]
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P.; Tzortzis, G.; Toward, R.E.; Gibson, G.R. Influence of galactooligosaccharide mixture (B-GOS) on gut microbiota, immune parameters and metabolomics in elderly persons. Br. J. Nutr. 2015, 114, 586–595. [Google Scholar] [CrossRef]
- Cheng, W.; Lu, J.; Lin, W.; Wei, X.; Li, H.; Zhao, X.; Jiang, A.; Yuan, J. Effects of a galacto-oligosaccharide-rich diet on fecal microbiota and metabolite profiles in mice. Food Funct. 2018, 9, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Petuely, F. Bifidusflora bei Flaschenkindern durch bifidogene Substanzen (Bifidusfaktor). Zeit Kinderheilk 1957, 79, 174–179. [Google Scholar] [CrossRef]
- Terada, A.; Hara, H.; Katoaka, M.; Mitsuoka, T. Effect of lactulose on the composition and metabolic activity of the human fecal flora. Microialb Ecol. Health Dis. 1992, 5, 43–50. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Attar, A.; Joly, F.A.; Riottot, M.; Dyard, F.; Flourié, B. Lactulose ingestion increases faecal bifidobacterial counts: A randomized double—blind study in healthy humans. Eur. J. Clin. Nutr. 2004, 58, 462–466. [Google Scholar] [CrossRef]
- Ballongue, J.C.; Schumann, C.; Quignon, P. Effects of lactulose and lactitol on colonic microflora and enzymatic activity. Scand. J. Gastroenterol. 1997, 32, 41–44. [Google Scholar] [CrossRef]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Vicaut, E.; Neut, C.; Flourié, B.; Brouns, F.; Bornet, F.R. The capacity of nondigestible carbohydrates to stimulate fecal bifidobacteria in healthy humans: A double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am. J. Clin. Nutr. 2004, 80, 1658–1664. [Google Scholar] [CrossRef]
- Tuohy, K.M.; Ziemer, C.J.; Klinder, A.; Knöbel, Y.; Pool Zobel, B.L.; Gibson, G.R. A human volunteer study to determine the prebiotic effects of lactulose powder on human colonic microbiota. Microb. Ecol. Health Disease 2002, 14, 165–173. [Google Scholar] [CrossRef]
- Bothe, M.K.; Maathuis, A.J.H.; Bellmann, S.; van der Vossen, J.M.B.M.; Berressem, D.; Koehler, A.; Schwejda-Guettes, S.; Gaigg, B.; Kuchinka Koch, A.; Stover, J.F. Dose—dependent prebiotic effect of lactulose in a computer-controlled in vitro model of the human large intestine. Nutrients 2017, 9, 767. [Google Scholar] [CrossRef]
- Sakai, Y.; Seki, N.; Hamano, H.; Ochi, H.; Abe, F.; Shimizu, F.; Masuda, K.; Iino, H. A study of the prebiotic effect of lactulose at low dosages in healthy Japanese women. Biosci. Microbiota Food Health 2019, 38, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Ruszkowsi, J.; Witkowsi, J.M. Lactulose: Patient—and dose—dependent prebiotic properties in humans. Anaerobe 2019, 59, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Lecerf, J.M.; De´peint, F.; Clerc, E.; Dugenet, Y.; Niamba, C.N.; Rhazi, L.; Cayzeele, A.; Abdelnour, G.; Jaruga, A.; Younes, H.; et al. Xylo-oligosaccharide (XOS) in combination with inulin modulates both the intestinal environment and immune status in healthy subjects, while XOS alone only shows prebiotic properties. Br. J. Nutr. 2012, 108, 1847–1858. [Google Scholar] [CrossRef] [PubMed]
- Mäkeläinen, H.; Saarinen, M.; Stowell, J.; Rautonen, N.; Ouwehand, A.C. Xylo—oligosaccharides and lactitol promote the growth of Bifidobacterium lactis and Lactobacillus species in pure cultures. Benef. Microbes 2010, 1, 139–148. [Google Scholar] [CrossRef]
- Mäkeläinen, H.; Forssten, S.; Saarinen, M.; Stowell, J.; Rautonen, N.; Ouwehand, A.C. Xylo—oligosaccharides enhance the growth of bifidobacterial and Bifidobacterium lactis in a simulated colon model. Benef. Microbes 2010, 1, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-H.; Chou, L.M.; Chien, J.-S.; Lin, C.I. Prebiotic effects of xilooligosaccharides on the improvement of microbiota balance in human subjects. Gastroenterol. Res. Pract. 2016, 6, 1–6. [Google Scholar]
- Finegold, S.M.; Li, Z.; Summanen, P.H. Xylooligosaccharides increases bifidobacterial but not lactobacilli in human gut microbiota. Food Funct. 2014, 5, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Švejstil, R.; Musilová, Š.; Rada, V. Raffinose—series oligosaccharides in soybean products. Sci. Agric. Bohem. 2015, 46, 73–77. [Google Scholar]
- Pérez López, E.; Cela, D.; Costabile, A.; Mateos Aparicio, I.; Rupérez, P. In vitro fermentability and prebiotic potential of soyabean Okara by human faecal microbiota. Br. J. Nutr. 2016, 116, 1116–1124. [Google Scholar] [CrossRef]
- Yan, M.; Xingzhuang, W.; Vigna, G.; Xianjum, M. Effects of soybean oligosaccharides on intestinal microbial communities and immune modulation in mice. Saudi J. Biol. Sci. 2017, 24, 114–121. [Google Scholar]
- Kaulpiboon, J.; Rudeekulthamrong, P.; Watanasatitarpa, S.; Ito, K.; Pongsawasdi, P. Synthesis of long-chain isomaltooligosaccharides from tapioca starch and an in vitro investigation of their prebiotic properties. J. Mol. Catal. B 2015, 120, 127–135. [Google Scholar] [CrossRef]
- Ketabi, A.; Dieleman, L.A.; Gänzle, M.G. Influence of isomalto-oligosaccharides on intestinal microbiota in rats. J. Appl. Microbiol. 2011, 110, 1297. [Google Scholar] [CrossRef] [PubMed]
- Rycroft, C.E.; Jones, M.R.; Gibson, G.R.; Rastall, R.A. A comparative in vitro evaluation of the fermentation properties of prebiotic oligosaccharides. J. Appl. Microbiol. 2001, 91, 878. [Google Scholar] [CrossRef]
- Singh, D.P.; Singh, J.; Boparai, R.K.; Zhu, J.H.; Mantri, S.; Khare, P.; Khardori, R.; Kondepudi, K.K.; Chopra, K.; Bishnoi, M. Isomalto-oligosaccharides, a prebiotic, functionally augment green tea effects against high fat diet-induced metabolic alterations via preventing gut dysbacteriosis in mice. Pharmacol. Res. 2017, 123, 103–113. [Google Scholar] [CrossRef]
- Nath, A.; Haktanirlar, G.; Varga, Á.; Molnar, M.A.; Albert, K.; Galambos, I.; Koris, A.; Vatai, G. Biological activities of lactose-derived prebiotics and symbiotic with probiotics on gastrointestinal system. Medicina 2008, 54, 18. [Google Scholar] [CrossRef] [PubMed]
- Maydeo, A. Lactitol or lactulose in the treatment of chronic constipation: Result of a systematic. J. Indian Med Assoc. 2010, 108, 789–792. [Google Scholar] [PubMed]
- Finney, M.; Smullen, J.; Foster, H.A.; Brokx, S.; Storey, D.M. Effects of low doses of lactitol on faecal microflora, pH, short chain fatty acids and gastrointestinal symptomology. Eur. J. Nutr. 2007, 46, 307–314. [Google Scholar] [CrossRef]
- Fujita, K.; Ito, T.; Kishino, E. Characteristics and applications of lactosucrose. J. Eng. Thermophys. 2009, 57, 13–21. [Google Scholar]
- Cardoso, T.; Marques, C.; Andreotti Dagostin, J.L.; Masson, M.L. Lactobionic Acid as a Potential Food Ingredient: Recent Studies and Applications. J. Food Sci. 2019, 84, 1672–1681. [Google Scholar] [CrossRef]
- Goderska, K. The antioxidant and prebiotic properties of lactobionic acid. Appl. Microbiol. Biotechnol. 2019, 103, 3737–3751. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G. Lactose and lactose derivatives as bioactive ingredients in human nutrition. Int. Dairy J. 2008, 18, 458–465. [Google Scholar] [CrossRef]
- Van Dokkum, W.; Wezendonk, L.J.W.; Van Aken-Schneijder, P.; Kistemaker, I.C. The tolerance of lactobionic acid in man. TNO Nutr. Food Res. 1994, 95, 1–22. [Google Scholar]
- Mukherjee, R.; Yun, J.W. Lactobionic acid reduces body weight gain in diet induced obese rats by targeted inhibition of galectin-1. Biochem. Biophys. Res. Commun. 2015, 463, 1311–1316. [Google Scholar] [CrossRef]
- Maier, T.V.; Lucio, M.; Ho Lee, L.; VerBerkmoes, N.C.; Brislawn, C.J.; Bernhardt, J.; Lamendella, R.; McDermott, J.E.; Bergeron, N.; Heinzmann, S.S.; et al. Impact of Dietary Resistant Starch on the Human Gut Microbiome, Metaproteome, and Metabolome. Am. Soc. Microbiol. 2017, 8, e01343-17. [Google Scholar] [CrossRef]
- Tachon, S.; Zhou, J.; Keenan, M.; Martin, R.; Marco, M.L. The intestinal microbiota in aged mice is modulated by dietary resistant starch and correlated with improvements in host responses. FEMS Microbiol. Ecol. 2013, 83, 299–309. [Google Scholar] [CrossRef]
- Zhu, C.-L.; Zhao, X.-H. In vitro fermentation of a retrograded maize starch by healthy adult fecal extract and impacts of exogenous microorganisms on three acids production. Starke 2013, 65, 330–337. [Google Scholar] [CrossRef]
- Kalmokoff, M.; Zwicker, B.; O’Hara, M.; Matias, F.; Green, J.; Shastri, P.; Green-Johnson, J.; Brooks, S.P. Temporal change in the gut community of rats fed with high amylose corn starch is driven by endogenous urea rather than strictly on carbohydrate availability. J. Appl. Microbiol. 2013, 11, 1516–1528. [Google Scholar] [CrossRef] [PubMed]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar]
- Al-Ghazzewi, F.H.; Khanna, S.; Tester, R.F.; Piggott, J. The potential use of hydrolised konjac glucomannan as a prebiotic. J. Sci. Food Agric. 2007, 87, 1758–1766. [Google Scholar] [CrossRef]
- Chen, H.L.; Cheng, H.C.; Liu, Y.J.; Wu, W.T. Konjac acts as a natural laxative by increasing stool bulk and improving colonic ecology in healthy adults. Nutrition 2006, 22, 1112–1119. [Google Scholar] [CrossRef]
- Chen, H.L.; Cheng, H.C.; Wu, W.T.; Liu, Y.J.; Liu, S.Y. Supplementation of konjac glucomannan into a low-fiber Chinese diet promoted bowel movement and improved colonic ecology in constipated adults: A placebo controlled, diet controlled trial. J. Am. Coll. Nutr. 2008, 27, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Connolly, M.L.; Lovegrove, J.A.; Tuohy, K.M. Konjac glucomannan hydrolysate beneficially modulates bacterial composition and activity within the faecal microbiota. J. Funct. Foods 2010, 2, 219–224. [Google Scholar] [CrossRef]
- Harmayani, E.; Aprilia, V.; Marsono, Y. Characterization of glucomannan from Amorphophallus oncophyllus and its prebiotic activity in vivo. Carbohydr. Polym. 2014, 112, 475–479. [Google Scholar] [CrossRef]
- Ariestanti, C.A.; Seechamnanturakit, V.; Harmayani, E.; Wichienchot, S. Optimization on production of konjac oligo-glucomannan and their effect on the gut microbiota. Food Sci. Nutr. 2019, 7, 788–796. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef] [PubMed]
- Etxeberria, U.; Fernández-Quintela, A.; Milagro, F.I.; Aguirre, L.; Martínez, J.A.; Portillo, M.P. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 2013, 61, 9517–9533. [Google Scholar] [CrossRef] [PubMed]
- Pierre, J.F.; Heneghan, A.F.; Feliciano, R.P.; Shanmuganayagam, D.; Roenneburg, D.A.; Krueger, C.G.; Reed, J.D.; Kudsk, K.A. Cranberry proanthocyanidins improve the gut mucous layer morphology and function in mice receiving elemental enteral nutrition. Jpen J. Parenter. Enter. Nutr. 2013, 37, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, J.; Collins, B.; Wolf, P.G.; Martinez, K.; Shen, W.; Chuang, C.C.; Zhong, W.; Cooney, P.; Cockrell, C.; Chang, E.; et al. Table grape consumption reduces adiposity and markers of hepatic lipogenesis and alters gut microbiota in butter fat-fed mice. J. Nutr. Biochem. 2016, 27, 123–135. [Google Scholar] [CrossRef]
- Roopchand, D.E.; Carmody, R.N.; Kuhn, P.; Moskal, K.; Rojas-Silva, P.; Turnbaugh, P.J.; Raskin, I. Dietary polyphenols promote growth of the gut bacterium akkermansia muciniphila and attenuate high-fat diet-induced metabolic syndrome. Diabetes 2015, 64, 2847–2858. [Google Scholar] [CrossRef]
- Moreno-Indias, I.; Sanchez-Alcoholado, L.; Perez-Martinez, P.; Andrés-Lacueva, C.; Cardona, F.; Tinahones, F.; Queipo-Ortuño, M.I. Red wine polyphenols modulate fecal microbiota and reduce markers of the metabolic syndrome in obese patients. Food Funct. 2016, 7, 1775–1787. [Google Scholar] [CrossRef]
- Namasivayam, N. Chemoprevention in experimental animals. Ann. N. Y. Acad. Sci. 2011, 1215, 60–71. [Google Scholar] [CrossRef]
- Lin, Y.T.; Kwon, Y.I.; Labbe, R.G.; Shetty, K. Inhibition of Helicobacter pylori and Associated Urease by Oregano and Cranberry Phytochemical Synergies. Appl. Environ. Microbiol. 2005, 71, 8558–8564. [Google Scholar] [CrossRef]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef]
- Jin, J.S.; Touyama, M.; Hisada, T.; Benno, Y. Effects of green tea consumption on human fecal microbiota with special reference to Bifidobacterium species. Microbiol. Immunol. 2012, 56, 729–739. [Google Scholar] [CrossRef]
- Okubo, T.; Ishihara, N.; Oura, A.; Serit, M.; Kim, M.; Yamamoto, T.; Misuoka, T. In vivo effects of tea polyphenol intake on human intestinal microflora and metabolism. Biosci. Biotechnol. Biochem. 1992, 56, 588–591. [Google Scholar] [CrossRef]
- Tzortzis, G.; Vulevic, J. Galacto-oligosaccharide prebiotics. In Prebiotics and Probiotics Science and Technology; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Belorkar, S.A.; Gupta, A.K. Oligosaccharides: A boon from nature’s desk. Amb Express 2016, 6, 82. [Google Scholar] [CrossRef] [PubMed]
- Joossens, M.; De Preter, V.; Ballet, V.; Verbeke, K.; Rutgeerts, P.; Vermeire, S. Effect of oligofructose-enriched inulin (OF-IN) on bacterial composition and disease activity of patients with Crohn’s Disease: Results from a double-blinded randomized controlled trial. Gut 2012, 61, 958. [Google Scholar] [CrossRef] [PubMed]
- Vigsnæs, L.K.; Holck, J.; Meyer, A.S.; Rask Licht, T. In vitro fermentation of sugar beet arabino-oligosaccharides by fecal microbiota obtained from patients with ulcerative colitis to selectively stimulate the growth of Bifidobacterium spp. and Lactobacillus spp. Appl. Environ. Microbiol. 2011, 77, 8336–8344. [Google Scholar] [CrossRef] [PubMed]
- Goffin, D.; Delzenne, N.; Blecker, C.; Hanon, E.; Deroanne, C.; Paquot, M. Will Isomalto-Oligosaccharides, a Well-Established Functional Food in Asia, Break through the European and American Market? The Status of Knowledge on these Prebiotics. Crit. Rev. Food Sci. Nutr. 2011, 51, 394. [Google Scholar] [CrossRef] [PubMed]
- Seibel, J.; Buchholz, K. Tools in oligosaccharide synthesis: Current research and application. Adv. Carbohydr. Chem. Biochem. 2010, 63, 101–163. [Google Scholar]
- Jalanka, J.; Major, G.; Murray, K.; Singh, G.; Nowak, A.; Kurtz, C.; Silos-Santiago, I.; Johnston, J.M.; de Vos, W.M.; Spiller, R. The Effect of Psyllium Husk on Intestinal Microbiota in Constipated Patients and Healthy Controls. Int. J. Mol. Sci. 2019, 20, 433. [Google Scholar] [CrossRef]
- Fernandez-Banares, F.; Hinojosa, J.; Sanchez-Lombrana, J.; Navarro, E.; Martinez-Salmeron, J.; Garcia-Puges, A.; Gonzalez-Huix, F.; Riera, J.; Gonzalez-Lara, V.; Domiguez-Abascal, F. Randomized clinical trial of Plantago ovata Seeds (Dietary fiber) as compared with mesalamine in maintaining remission in ulcerative cholitis. Am. J. Gastroenterol. 1999, 94, 427–433. [Google Scholar]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef]
- Ooi, S.L.; Correa, D.; Pak, S.C. Probiotics, prebiotics, and low FODMAP diet for irritable bowel syndrome. What is the current evidence? Complementary Ther. Med. 2019, 43, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.; Anderson, J.L.; Barrett, J.S.; Muir, J.G.; Irving, P.M.; Whelan, K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J. Nutr. 2012, 142, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Whelan, K. Altered gastrointestinal microbiota in irritable bowel syndrome and its modification by diet: Probiotics, prebiotics and the low FODMAP diet. Proc. Nutr. Soc. 2016, 75, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Rajilic-Stojanovic, M.; Biagi, E.; Heilig, H.G.; Kajander, K.; Kekkonen, R.A.; Tims, S.; de Vos, W.M. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1792–1801. [Google Scholar] [CrossRef] [PubMed]
- Jalanka-Tuovinen, J.; Salonen, A.; Nikkila, J.; Immonen, O.; Kekkonen, R.; Lahti, L.; Palva, A.; de Vos, W.M. Intestinal microbiota in healthy adults: Temporal analysis reveals individual and common core and relation to intestinal symptoms. PLoS ONE 2011, 6, e23035. [Google Scholar] [CrossRef]
- Hunter, J.O.; Tuffnell, Q.; Lee, A.J. Controlled trial of oligofructose in the management of irritable bowel syndrome. J. Nutr. 1999, 129, 1451S–1453S. [Google Scholar] [CrossRef]
- Olesen, M.; Gudmand-Høyer, E. Efficacy, safety, and tolerability of fructooligosaccharides in the treatment of irritable bowel syndrome. Am. J. Clin. Nutr. 2000, 72, 1570–1575. [Google Scholar] [CrossRef]
- Paineau, D.; Payen, F.; Panserieu, S.; Coulombier, G.; Sobaszek, A.; Lartigau, I.; Brabet, M.; Galmiche, J.-P.; Tripodi, D.; Sacher-Huvelin, S.; et al. The effects of regular consumption of short-chain fructo-oligosaccharides on digestive comfort of subjects with minor functional bowel disorders. Br. J. Nutr. 2008, 99, 311–318. [Google Scholar] [CrossRef]
- Wilson, B.; Rossi, M.; Dimidi, E.; Whelan, K. Prebiotics in irritable bowel syndrome and other functional bowel disorders in adults: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2019, 109, 1098–1111. [Google Scholar] [CrossRef]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides:definition, specificity, function, and application in gastrointestinal disorders. Gastroenterology 2017, 32, 64–68. [Google Scholar]
- Whelan, K. Probiotics and prebiotics in the management of irritable bowel syndrome: A review of recent clinical trials and systematic reviews. Curr. Opin. Clin. Nutr. Metab. Care 2011, 14, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Cammarota, G.; Ianiro, G.; Cianci, R.; Bibbò, S.; Gasbarrini, A.; Currò, D. The involvement of gut microbiota in inflammatory bowel disease pathogenesis: Potential for therapy. Pharmacol. Ther. 2015, 149, 191–212. [Google Scholar] [CrossRef] [PubMed]
- Silk, D.B.A.; Davis, A.; Vulevic, J.; Tzortzis, G.; Gibson, G.R. Clinical trial: The effects of a trans-galactooligosaccharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2009, 29, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Vulevic, J.; Tzortzis, G.; Juric, A.; Gibson, G.R. Effect of a prebiotic galactooligosaccharide mixture (B-GOS®) on gastrointestinal symptoms in adults selected from a general population who suffer with bloating, abdominal pain, or flatulence. Neurogastroenterol. Motil. 2018, 11, e13440. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Ren, Y.; Lu, J.; Bartlett, M.; Chen, L.; Zhang, Y.; Guo, X.; Liu, C. A Novel Prebiotic Blend Product Prevents Irritable Bowel Syndrome in Mice by Improving Gut Microbiota and Modulating Immune Response. Nutrients 2017, 9, 1341. [Google Scholar] [CrossRef]
- Ford, A.C.; Harris, L.A.; Lacy, B.E.; Quigley, E.M.; Moayyedi, P. Systematic review with meta-analysis: The efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2018, 48, 1044–1060. [Google Scholar] [CrossRef]
- Pandey, K.R.; Naik, S.R.; Vakil, B.V. Probiotics, prebiotics and synbiotics-a review. J. Food Sci. Technol. 2015, 52, 7577–7587. [Google Scholar] [CrossRef]
- Carroll, I.M.; Ringel-Kulka, T.; Siddle, J.P.; Ringel, Y. Alterations in Composition and Diversity of the Intestinal Microbiota in Patients with Diarrhea-Predominant Irritable Bowel Syndrome. Neurogastroenterol. Motil. 2012, 24, 521.e248–530.e248. [Google Scholar] [CrossRef]
- Hedin, C.; Whelan, K.; Lindsay, J.O. Evidence for the use of probiotics and prebiotics in inflammatory bowel disease: A review of clinical trials. Proc. Nutr. Soc. 2007, 66, 307–315. [Google Scholar] [CrossRef]
- Sutherland, L.; Singleton, J.; Sessions, J.; Hanauer, S.; Krawitt, E.; Rankin, G.; Summers, R.; Mekhjian, H.; Greenberger, N.; Kelly, M. Double blind, placebo controlled trial of metronidazole in Crohn’s disease. Gut 1991, 32, 1071–1075. [Google Scholar] [CrossRef]
- Borgaonkar, M.; MacIntosh, D.; Fardy, J.; Simms, L. Anti-tuberculous therapy for maintaining remission of Crohn’s disease. Cochrane Database Syst. Rev. 2000, CD000299. [Google Scholar] [CrossRef]
- Looijer–van Langen, M.A.C.; Dieleman, L.A. Prebiotics in Chronic Intestinal Inflammation. Inflamm. Bowel Dis. 2009, 15, 454–462. [Google Scholar] [CrossRef]
- Benjamin, J.L.; Hedin, C.R.H.; Koutsoumpas, A.; Ng, S.C.; McCarthy, N.E.; Hart, A.L.; Kamm, M.A.; Sanderson, J.D.; Knight, S.C.; Forbes, A.; et al. Randomised, double-blind, placebo-controlled trial of fructo-oligosaccharides in active Crohn’s disease. Gut 2011, 60, 923–929. [Google Scholar] [CrossRef]
- Lindsay, J.O.; Whelan, K.; Stagg, A.J.; Gobin, P.; Al-Hassi, H.O.; Rayment, N.; Kamm, M.A.; Knight, S.C.; Forbes, A. Clinical Microbiological and Immunological Effects of Fructo-Oligosaccharide in patients with Crohn’s Disease. Gut 2006, 55, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Casellas, F.; Borruel, N.; Torrejon, A.; Varela, E.; Antolin, M.; Guarner, F.; Malagelada, J.-R. Oral oligofructose-enriched inulin supplementation in acute ulcerative colitis is well tolerated and associated with lowered faecal calprotectin. Aliment. Pharmacol. Ther. 2007, 25, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Joossens, M.; Huys, G.; Van Steen, K.; Cnockaert, M.; Vermeire, S.; Rutgeerts, P.; Verbele, K.; Vandamme, P.; De Preter, V. High-Throughput Method for Comparative Analysis of Denaturing Gradient Gel Electrophoresis Profiles from Human Fecal Samples reveals significant increases in two bifidobacterial species after inuli-type prebiotic intake. FEMS Microbiol. Ecol. 2011, 75, 343–349. [Google Scholar] [CrossRef]
- Astó, E.; Méndez, I.; Audivert, S.; Farran-Codina, A.; Espadaler, J. The Efficacy of Probiotics, Prebiotic Inulin-Type Fructans, and Synbiotics in Human Ulcerative Colitis: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 293. [Google Scholar] [CrossRef]
- Hafer, A.; Kramer, S.; Duncker, S.; Kruger, M.; Manns, M.P.; Bischoff, S.C. Effect of oral lactulose on clinical and immunohistocemical parameters in patients with inflammatory bowel disease: A pilot study. BMC Gastroenterol. 2007, 7, 36. [Google Scholar] [CrossRef]
- Fellerman, K.; Wehkamp, J.; Herrlinger, K.R.; Stange, E.F. Chron’s disease: A defensin deficiency syndrome? Eur. J. Gastroenterol. Hepatol. 2003, 15, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Szilagy, A.; Rivard, J.; Shrier, I. Diminished efficacy of colonic adaptation to lactulose occurs in patients with inflammatory bowel disease in remission. Dig. Dis. Sci. 2002, 47, 2811–2822. [Google Scholar] [CrossRef]
- Hallert, C.; Kaldma, M.; Petersson, B. Ispaghula Husk may relieve gastrointestinal symptoms in ulcerative colitis in remission. Scand. J. Gastroenterol. 1991, 26, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Charrier, J.A.; Martin, R.J.; McCutcheon, K.L.; Raggio, A.M.; Goldsmith, F.; Goita, M.; Senevirathne, R.N.; Brown, I.L.; Pelkman, C.; Zhou, J.; et al. High fat diet partially attenuates fermentation responses in rats fed resistant starch from high amylose maize. Obesity 2013, 21, 2350–2355. [Google Scholar] [CrossRef] [PubMed]
- Toden, S. Resistant starch prevent colonic DNA damage induced by high dietary cooked red meat or casein in rats. Cancer Biol. Ther. 2006, 5, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.J.; Robien, K.; Slavin, J.L. Effect of prebiotics on biomarkers of colorectal cancer in humans: A systematic review. Nutr. Rev. 2012, 70, 436–443. [Google Scholar] [CrossRef] [PubMed]
- Barbier de La Serre, C.; Ellis, C.L.; Lee, J.; Hartman, A.L.; Rutledge, J.C.; Raybould, H.E. Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 299, G440–G448. [Google Scholar] [CrossRef] [PubMed]
- Candela, M.; Guidotti, M.; Fabbri, A.; Brigidi, P.; Franceschi, C.; Fiorentini, C. Human intestinal microbiota: Cross-talk with the host and its potential role in colorectal cancer. Crit. Rev. Microbiol. 2011, 37, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hernot, D.C.; Boileau, T.W.; Bauer, L.L.; Middelbos, I.S.; Murphy, M.R.; Swanson, K.S.; Fahey Jr, G.C. In vitro fermentation profiles, gas production rates, and microbiota modulation as affected by certain fructans, galactooligosaccharides, and polydextrose. J. Agric. Food Chem. 2009, 57, 1354–1361. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhang, Y.; Zheng, P.; Chen, X.; Yang, Y. Starch structure modulates metabolic activity and gut microbiota profile. Anaerobe J. 2013, 24, 71–78. [Google Scholar] [CrossRef]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.; Brummer, R.J. Review article: The role of butyrate on colonic function. Aliment. Pharmacol. Ther. 2008, 27, 104–119. [Google Scholar] [CrossRef]
- Zhang, J.; Yi, M.; Zha, L.; Chen, S.; Li, Z.; Li, C.; Gong, M.; Deng, H.; Chu, X.; Chen, J.; et al. Sodium Butyrate Induces Endoplasmic Reticulum Stress and Autophagy in Colorectal Cells: Implications for Apoptosis. PLoS ONE 2016, 11, e0147218. [Google Scholar] [CrossRef]
- Kumar Singh, A.S.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Lauro, M.R.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients 2019, 11, 2216. [Google Scholar] [CrossRef] [PubMed]
- Sengottuvelan, M.; Nalini, N. Dietary supplementation of resveratrol suppresses colonic tumour incidence in 1,2-dimethylhydrazine-treated rats by modulating biotransforming enzymes and aberrant crypt foci development. Br. J. Nutr. 2006, 96, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Niness, K.R. Inulin and oligofructose: What are they? J. Nutr. 1999, 129, 1402S–1406S. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Quigley, E.M.; Lacy, B.E.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.; Moayyedi, P. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: Systematic review and meta-analysis. Am. J. Gastroenterol. 2014, 109, 1547–1561. [Google Scholar] [CrossRef] [PubMed]
- Akram, W.; Garud, N.; Joshi, R. Role of inulin as prebiotics on inflammatory bowel disease. Drug Discov. Ther. 2019, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Seki, N.; Hamano, K.; Ochi, H.; Abe, F.; Masuda, K.; Iino, H. Prebiotic effect of two grams of lactulose in healthy Japanese women: A randomized, double-blind, placebo-controlled crossover trial. Benef. Microbes 2019, 10, 629–639. [Google Scholar] [CrossRef]
- Mizota, T.; Mori, T.; Yaeshima, T.; Yanagida, T.; Iwatsuki, K.; Ichibashi, M.; Tamura, Y.; Fukuwatari, Y. Effects of low dosages of lactulose on the intestinal function of healthy adults. Milchwissenschaft 2002, 57, 312–315. [Google Scholar]
- Wotzka, S.Y.; Kreuzer, M.; Maier, L.; Zünd, M.; Schlumberger, M.; Nguyen, B.; Fox, M.; Pohi, D.; Heinrich, H.; Rogler, G.; et al. Microbiota stability in healthy individuals after single-dose lactulose challenge-a randomized controlled study. PLoS ONE 2018, 13, e0206214. [Google Scholar] [CrossRef]
- Asha, M.Z.; Khalil, S.F.H. Sultan Efficacy and Safety of Probiotics, Prebiotics and Synbiotics in the Treatment of Irritable Bowel Syndrome: A systematic review and meta-analysis. Qaboos Univ. Med. J. 2020, 20, e13–e24. [Google Scholar] [CrossRef]
- Van Dijk, M.; Pot, G.K. The effects of nutritional interventions on recurrence in survivors of colorectal adenomas and cancer: A systematic review of randomised controlled trials. Eur. J. Clin. Nutr. 2016, 70, 566–573. [Google Scholar] [CrossRef]
| Prebiotic | Effect | Reference |
|---|---|---|
| Inulin-type fructans | Stimulate the activity of the glutathione S-transferases of antioxidant enzymes | [29] |
| Inulin | Induces scavenger activity in the radical O2 | [30] |
| Inulin | Prevents lipid peroxidation in the stomach | [46] |
| Inulin and other “sugar-like” elements | Replace vitamin C as a dietary supplement and/or limit its degradation | [47] |
| Inulin-type prebiotics | Inhibit the degradation of ascorbate | [47] |
| Inulin | Causes a protective effect on LPS-induced damage of colonic muscle | [41,42] |
| Type of Prebiotics | Structure | Mechanisms of Action |
|---|---|---|
| Fructans (Inulin and Fructooligosaccharides, FOS) | Fructosyl-fructose β (2X1) glycosidic bonds (FOS DP 2–9; inulin DP 2–60) [25]. | ↑ Lactobacilli and Bifidobacterium (especially B. longum subsp. Longum, B. pseudocatenulatum, B. bifidum and B. adolescentis) growth [22,26]. ↑ SCFAs production [26]. Act indirectly as a scavenger of Reactive Oxygen Species (ROS), thanks to the action of SCFAs and can stimulate the activity of the glutathione S-transferases (GSTs) of antioxidant enzymes [42]. Contrast the oxidative stress induced by LPS in human colonic mucosa [42]. |
| Galactooligosaccharides (GOS) | Commercially produced by the enzymatic activity of β-galactosidase transferase on lactose (DP 2–8). It is a galactose polymer with a terminal β-linked glucose monomer [105]. | ↑ Bifidobacterium [50] and fecal Bifidobacteria concentration growth [51]. Can modulate immune function: ↑ Cytokine IL-10, interleukin 8 (IL-8) and C-reactive protein, improve Natural Killer (NK) cell activity, and ↓ IL-1β expression [51]. Improve lipid metabolism [52]. Enrich the mouse microbiota of Alloprevotella, Bacteroides, and Parasutterella [52]. |
| Lactulose | Synthetic disaccharide Galactose–fructose β (1–4)-linked [55]. | At a low dosage (2–3 g/day), ↑ Bifidobacterium count [53,59], but not Lactobacilli, and determines a low production of SCFAs; 5 g/day determines the correct balance among the microbial population (Bifidobacteria, Lactobacilli and Anaerostipes) and SCFAs production, while 10 g/day ↓ butyrate production and ↑ acetate [59]. |
| Lactobionic acid | A gluconic acid bonded to a galactose [78]. | ↑ Lactobacilli and Bifidobacterium growth [79]. Has anti-inflammatory properties, ↓ obesity and improves metabolic parameters [82]. |
| Xilooligosaccharides (XOS) | Xylose units linked by β (1–4) bonds, with a DP of 2 to 10 [106]. | ↑ Bifidobacteria (especially Bifidobacterium lactis and Bifidobacterium adolescentis [63]), Lactobacilli [65] and butyrate fecal concentrations [62]. ↓ Clostridium growth [65]. No changes in lactobacilli counts, stool pH and SCFAs production [66]. |
| Arabinooligosaccharides (AOS) | α (1–6)-linked backbone of L. Arabinosyl residues, which can be single- or double-substituted with α (1–2)- and/or α (1–3)-linked L-arabinosyl residues [107] | ↑ Lactobacilli and Bifidobacterium growth [108]. ↓ Firmicutes, Bacteroidetes and Desulfovibrio [108]. ↑ Production of acetate that determines a decrease in pH, probably contributing to the amelioration of inflammation and prevention of flare-ups in UC patients [108]. |
| Soybean oligosaccharides | Tri, tetra or pentasaccharide galactose–sucrose α (1–6)-linked [67] | ↑ Lactobacilli and Bifidobacterium growth [68]. ↓ Clostridia and Bacteroidetes [68]. ↑ Immunological functions [69]. |
| Isomaltooligosaccharides (IMO) | Gluco-oligosaccharides, with an α (1–6) bond and DP between 2 and 10 (di-, tri- and tetrasaccharides) [71,109,110]. | ↑ Lactobacilli and Bifidobacterium [70,71,72,73], Akkermansia, and Roseburia [73] growth. Improve Firmicutes / Bacteroidetes and Prevotella / Bacteroidetes ratios [73]. Show positive effects on visceral adipose tissue, on the production of pro-inflammatory cytokines and on lipid and glycemic control, improving insulin, glucagon and leptin levels [73]. |
| Resistant starch | Glucose polysaccharides consisting of amylose (α (1–4) bonds) and amylopectin (α (1–6) bonds) [12]. | ↑ Bifidobacteria, Bacteroidetes, Akkermansia and Allobactum species [12]. ↑ SCFAs production [84,85,86]. |
| Glucomannan | Mannose and glucose at a molar ratio of 1.6:1, with little residues of galactose or acetyl groups [88]. | ↑ Lactobacilli and Bifidobacterium growth [88]. ↓ Clostridium perfringens and Escherichia Coli growth [89,90,91]. ↑ SCFAs production [92]. ↓ Cecal pH value [92]. Improves blood cholesterol, glycemia and reduces constipation [88]. |
| Psyllium | Highly branched and gel-forming arabinoxylan, a polymer rich in arabinose and xylose [111]. | ↑ Fecalibacterium and Phascolarctobacterium growth, associated with SCFAs production [111]. ↓ Christensenella, associated with hard stools [111]. ↑ Butyrate fecal concentration [112]. |
| Polyphenols | Hydroxylated aromatic rings or phenol rings [113]. | ↑ Lactobacilli and Bifidobacterium [99,102,103,104] Akkermansia [96,97,98], Roseburia and F. Prausnitzii [99] growth. ↓ Clostridium growth [102,104]. Offset Helicobacter Pylori-inhibiting urease [101]. Inhibit pro-inflammatory mediators: cyclooxygenase-2 (COX2), IL-6, Tumor Necrosis Factor-α (TNF-α), Nuclear Factor kB (NFkB) and Vascular-Endothelial Growth Factor (VEGF) [47,100]. Reduce serum triacylglycerol and C- reactive protein [102]. |
| Meta-Analysis | Eligible RCTs | Prebiotics Analyzed | Effects | |
|---|---|---|---|---|
| IBS | Wilson et al. 2019 [123] | 11 | non-inulin-type fructan prebiotics | Improvement of flatulence severity score |
| inulin-type fructans | No benefits | |||
| Ford et al. 2018 [158] | 2 | Fructooligosaccharides | No results on symptoms | |
| 1 | GOS | Reduction in mean global symptoms but not in abdominal pain | ||
| Asha MZ et Al. 2020 [163] | 3 | partially-hydrolysed guar gum and fructooligosac | no benefits | |
| IBD | Astó et al. 2019 [140] | 2 | Inulin—type fructans | Prebiotics help mesalazine to mitigate intestinal inflammation |
| Colon cancer | van Dijk M. 2016 [164] | 4 | prebiotic fibers | insufficient data |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarino, M.P.L.; Altomare, A.; Emerenziani, S.; Di Rosa, C.; Ribolsi, M.; Balestrieri, P.; Iovino, P.; Rocchi, G.; Cicala, M. Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults. Nutrients 2020, 12, 1037. https://doi.org/10.3390/nu12041037
Guarino MPL, Altomare A, Emerenziani S, Di Rosa C, Ribolsi M, Balestrieri P, Iovino P, Rocchi G, Cicala M. Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults. Nutrients. 2020; 12(4):1037. https://doi.org/10.3390/nu12041037
Chicago/Turabian StyleGuarino, Michele Pier Luca, Annamaria Altomare, Sara Emerenziani, Claudia Di Rosa, Mentore Ribolsi, Paola Balestrieri, Paola Iovino, Giulia Rocchi, and Michele Cicala. 2020. "Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults" Nutrients 12, no. 4: 1037. https://doi.org/10.3390/nu12041037
APA StyleGuarino, M. P. L., Altomare, A., Emerenziani, S., Di Rosa, C., Ribolsi, M., Balestrieri, P., Iovino, P., Rocchi, G., & Cicala, M. (2020). Mechanisms of Action of Prebiotics and Their Effects on Gastro-Intestinal Disorders in Adults. Nutrients, 12(4), 1037. https://doi.org/10.3390/nu12041037







