Dietary Supplementation with Curcumin Reduce Circulating Levels of Glycogen Synthase Kinase-3β and Islet Amyloid Polypeptide in Adults with High Risk of Type 2 Diabetes and Alzheimer’s Disease
Abstract
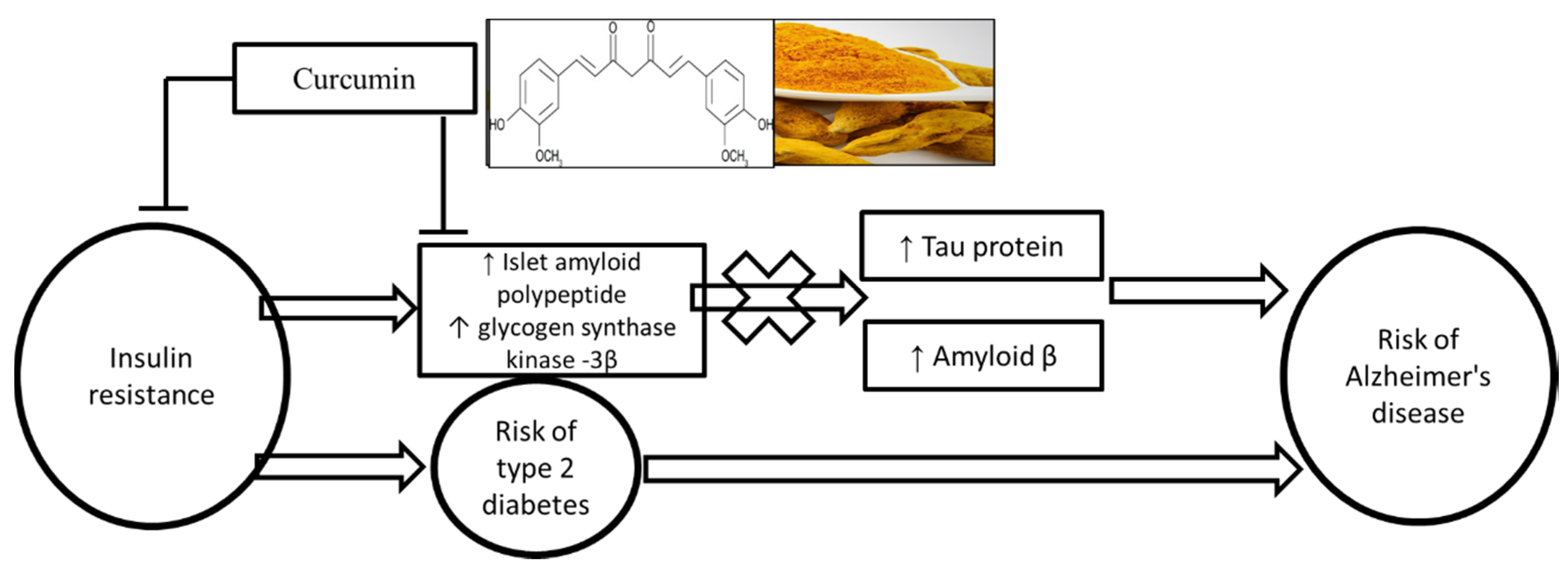
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Data Collection and Outcome Measures
Questionnaires (Diet, Physical Activity and Medical History)
2.4. Body Composition and Anthropometric Measures
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
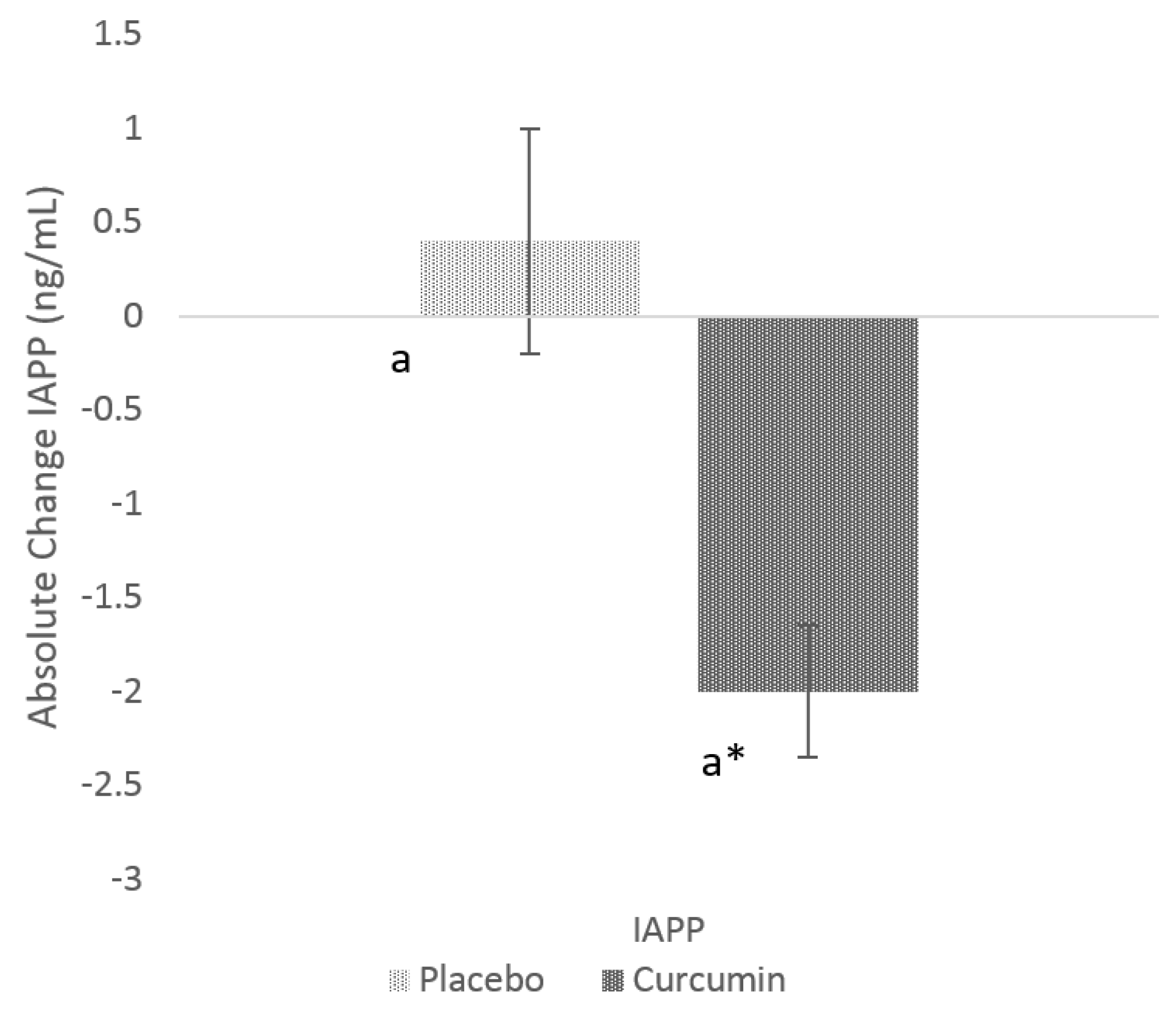
3.2. Effects of Curcumin on GSK-3β and IAPP
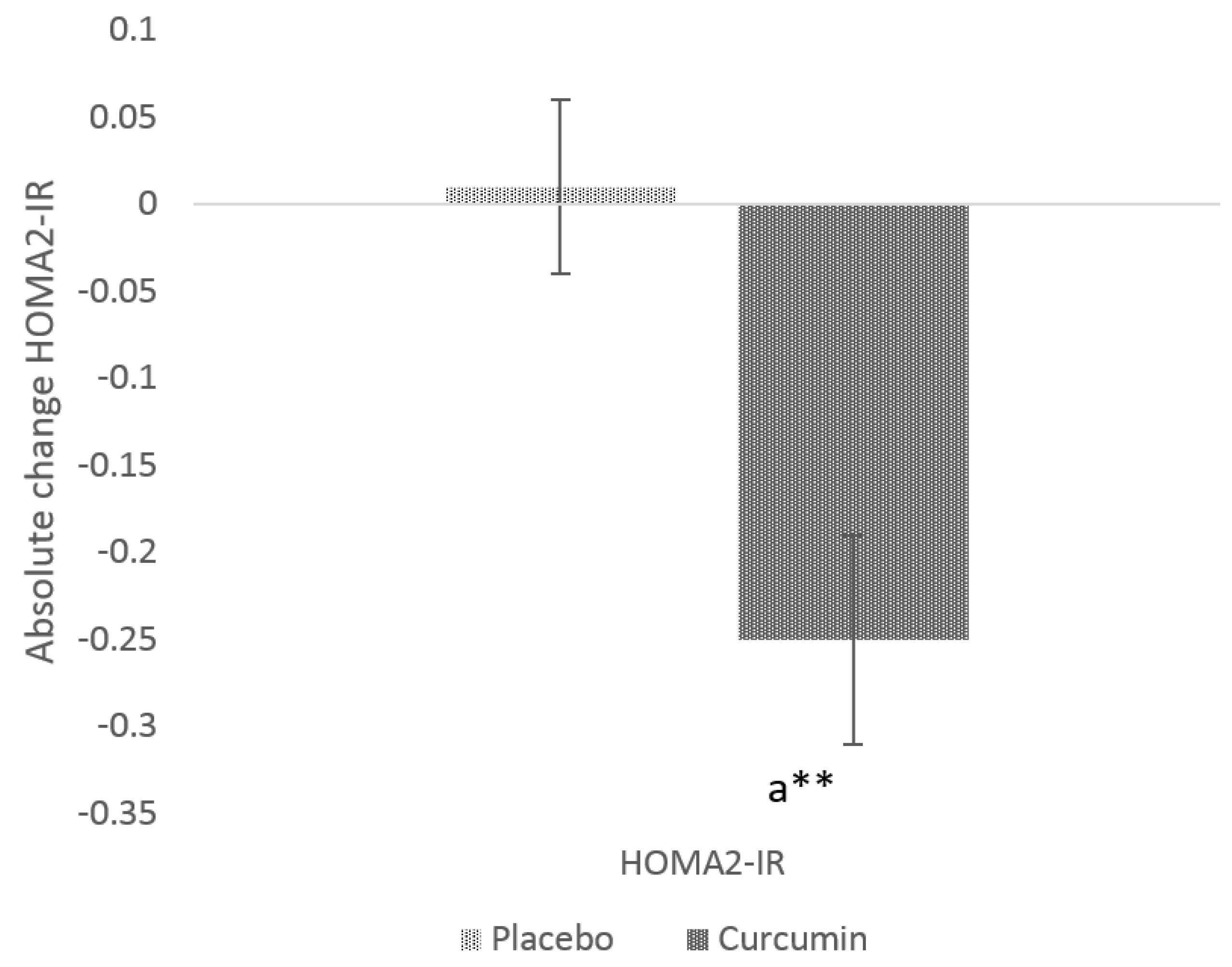
3.3. Glycaemic Indices
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AUSDRISK | Australian Type 2 Diabetes Risk |
| AD | Alzheimer’s disease |
| GSK-3β | glycogen synthase kinase-3 β |
| HOMA-IR | homeostasis model for assessment of insulin resistance |
| IAPP | islet amyloid polypeptide |
| T2D | type 2 diabetes |
References
- Ferreira, L.S.S.; Fernandes, C.S.; Vieira, M.N.N.; De Felice, F.G. Insulin resistance in Alzheimer’s disease. Front. Neurosci. 2018, 12, 830. [Google Scholar] [CrossRef]
- de la Monte, S.M.; Wands, J.R. Alzheimer’s disease is type 3 diabetes-evidence reviewed. J. Diabetes Sci. Technol. 2008, 2, 1101–1113. [Google Scholar] [CrossRef]
- Luchsinger, J.A.; Reitz, C.; Honig, L.S.; Tang, M.X.; Shea, S.; Mayeux, R. Aggregation of vascular risk factors and risk of incident Alzheimer disease. Neurology 2005, 65, 545–551. [Google Scholar] [CrossRef]
- Luchsinger, J.A.; Tang, M.-X.; Shea, S.; Mayeux, R. Hyperinsulinemia and risk of Alzheimer disease. Neurology 2004, 63, 1187–1192. [Google Scholar] [CrossRef]
- Westermark, P.; Andersson, A.; Westermark, G.T. Islet amyloid polypeptide, islet amyloid, and diabetes mellitus. Physiol. Rev. 2011, 91, 795–826. [Google Scholar] [CrossRef]
- N Fawver, J.; Ghiwot, Y.; Koola, C.; Carrera, W.; Rodriguez-Rivera, J.; Hernandez, C.; TDineley, K.; Kong, Y.; Li, J.; Jhamandas, J.; et al. Islet Amyloid Polypeptide (IAPP): A Second Amyloid in Alzheimer’s Disease. Curr. Alzheimer Res. 2014, 11, 928–940. [Google Scholar] [CrossRef]
- Abaffy, T.; Cooper, G.J. GSK3 involvement in amylin signaling in isolated rat soleus muscle. Peptides 2004, 25, 2119–2125. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, W. Islet amyloid polypeptide: Another key molecule in Alzheimer’s pathogenesis? Prog. Neurobiol. 2017, 153, 100–120. [Google Scholar] [CrossRef]
- Hooper, C.; Killick, R.; Lovestone, S. The GSK3 hypothesis of Alzheimer’s disease. J. Neurochem. 2008, 104, 1433–1439. [Google Scholar] [CrossRef]
- Gao, C.; Holscher, C.; Liu, Y.; Li, L. GSK3: A key target for the development of novel treatments for type 2 diabetes mellitus and Alzheimer disease. Rev. Neurosci. 2011, 23, 1–11. [Google Scholar] [CrossRef]
- Beurel, E.; Grieco, S.F.; Jope, R.S. Glycogen synthase kinase-3 (GSK3): Regulation, actions, and diseases. Pharmacol. Ther. 2015, 148, 114–131. [Google Scholar] [CrossRef]
- Thota, R.N.; Acharya, S.H.; Abbott, K.A.; Garg, M.L. Curcumin and long-chain Omega-3 polyunsaturated fatty acids for Prevention of type 2 Diabetes (COP-D): Study protocol for a randomised controlled trial. Trials 2016, 17, 565. [Google Scholar] [CrossRef]
- Weisberg, S.P.; Leibel, R.; Tortoriello, D.V. Dietary curcumin significantly improves obesity-associated inflammation and diabetes in mouse models of diabesity. Endocrinology 2008, 149, 3549–3558. [Google Scholar] [CrossRef]
- Shen, L.; Ji, H.F. The pharmacology of curcumin: Is it the degradation products? Trends Mol. Med. 2012, 18, 138–144. [Google Scholar] [CrossRef]
- Pivari, F.; Mingione, A.; Brasacchio, C.; Soldati, L. Curcumin and Type 2 Diabetes Mellitus: Prevention and Treatment. Nutrients 2019, 11, 1837. [Google Scholar] [CrossRef]
- Mishra, S.; Palanivelu, K. The effect of curcumin (turmeric) on Alzheimer’s disease: An overview. Ann. Indian Acad. Neurol. 2008, 11, 13–19. [Google Scholar] [CrossRef]
- Sarker, M.R.; Franks, S.; Sumien, N.; Thangthaeng, N.; Filipetto, F.; Forster, M. Curcumin Mimics the Neurocognitive and Anti-Inflammatory Effects of Caloric Restriction in a Mouse Model of Midlife Obesity. PLoS ONE 2015, 10, e0140431. [Google Scholar] [CrossRef]
- Kim, G.Y.; Kim, K.H.; Lee, S.H.; Yoon, M.S.; Lee, H.J.; Moon, D.O.; Lee, C.M.; Ahn, S.C.; Park, Y.C.; Park, Y.M. Curcumin inhibits immunostimulatory function of dendritic cells: MAPKs and translocation of NF-kappa B as potential targets. J. Immunol. 2005, 174, 8116–8124. [Google Scholar] [CrossRef]
- Dong, S.; Zeng, Q.; Mitchell, E.S.; Xiu, J.; Duan, Y.; Li, C.; Tiwari, J.K.; Hu, Y.; Cao, X.; Zhao, Z. Curcumin enhances neurogenesis and cognition in aged rats: Implications for transcriptional interactions related to growth and synaptic plasticity. PLoS ONE 2012, 7, e31211. [Google Scholar] [CrossRef]
- Yu, S.Y.; Zhang, M.; Luo, J.; Zhang, L.; Shao, Y.; Li, G. Curcumin ameliorates memory deficits via neuronal nitric oxide synthase in aged mice. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2013, 45, 47–53. [Google Scholar] [CrossRef]
- Cheng, K.K.; Yeung, C.F.; Ho, S.W.; Chow, S.F.; Chow, A.H.; Baum, L. Highly stabilized curcumin nanoparticles tested in an in vitro blood-brain barrier model and in Alzheimer’s disease Tg2576 mice. AAPS J. 2013, 15, 324–336. [Google Scholar] [CrossRef]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar] [CrossRef]
- Hoppe, J.B.; Frozza, R.L.; Pires, E.N.; Meneghetti, A.B.; Salbego, C. The curry spice curcumin attenuates beta-amyloid-induced toxicity through beta-catenin and PI3K signaling in rat organotypic hippocampal slice culture. Neurol. Res. 2013, 35, 857–866. [Google Scholar] [CrossRef]
- Ng, T.P.; Chiam, P.C.; Lee, T.; Chua, H.C.; Lim, L.; Kua, E.H. Curry consumption and cognitive function in the elderly. Am. J. Epidemiol. 2006, 164, 898–906. [Google Scholar] [CrossRef]
- Thota, R.N.; Acharya, S.H.; Garg, M.L. Curcumin and/or omega-3 polyunsaturated fatty acids supplementation reduces insulin resistance and blood lipids in individuals with high risk of type 2 diabetes: A randomised controlled trial. Lipids Health Dis. 2019, 18, 31. [Google Scholar] [CrossRef]
- Sesti, G. Pathophysiology of insulin resistance. Best Pract. Res. Clin. Endocrinol. Metab. 2006, 20, 665–679. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, N.Q.; Yan, F.; Jin, H.; Zhou, S.Y.; Shi, J.S.; Jin, F. Diabetes mellitus and Alzheimer’s disease: GSK-3β as a potential link. Behav. Brain Res. 2018, 339, 57–65. [Google Scholar] [CrossRef]
- Feng, Z.-C.; Donnelly, L.; Li, J.; Krishnamurthy, M.; Riopel, M.; Wang, R. Inhibition of Gsk3β activity improves β-cell function in c-KitWv/+ male mice. Lab. Investig. 2012, 92, 543–555. [Google Scholar] [CrossRef][Green Version]
- Gleason, J.E.; Szyleyko, E.A.; Eisenmann, D.M. Multiple redundant Wnt signaling components function in two processes during C. elegans vulval development. Dev. Biol. 2006, 298, 442–457. [Google Scholar] [CrossRef]
- Jope, R.S.; Johnson, G.V. The glamour and gloom of glycogen synthase kinase-3. Trends Biochem. Sci. 2004, 29, 95–102. [Google Scholar] [CrossRef]
- Xiong, Z.; Hongmei, Z.; Lu, S.; Yu, L. Curcumin mediates presenilin-1 activity to reduce β-amyloid production in a model of Alzheimer’s disease. Pharmacol. Rep. 2011, 63, 1101–1108. [Google Scholar] [CrossRef]
- Huang, H.-C.; Tang, D.; Xu, K.; Jiang, Z.-F. Curcumin attenuates amyloid-β-induced tau hyperphosphorylation in human neuroblastoma SH-SY5Y cells involving PTEN/Akt/GSK-3β signaling pathway. J. Recept. Signal Transduct. 2014, 34, 26–37. [Google Scholar] [CrossRef]
- Bustanji, Y.; Taha, M.O.; Almasri, I.M.; Al-Ghussein, M.A.S.; Mohammad, M.K.; Alkhatib, H.S. Inhibition of glycogen synthase kinase by curcumin: Investigation by simulated molecular docking and subsequent in vitro/in vivo evaluation. J. Enzym. Inhib. Med. Chem. 2009, 24, 771–778. [Google Scholar] [CrossRef]
- Small, G.W.; Siddarth, P.; Li, Z.; Miller, K.J.; Ercoli, L.; Emerson, N.D.; Martinez, J.; Wong, K.P.; Liu, J.; Merrill, D.A.; et al. Memory and Brain Amyloid and Tau Effects of a Bioavailable Form of Curcumin in Non-Demented Adults: A Double-Blind, Placebo-Controlled 18-Month Trial. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry 2018, 26, 266–277. [Google Scholar] [CrossRef]
- Leighton, B.; Cooper, G.J. Pancreatic amylin and calcitonin gene-related peptide cause resistance to insulin in skeletal muscle in vitro. Nature 1988, 335, 632–635. [Google Scholar] [CrossRef]
- Roth, J.D. Amylin and the regulation of appetite and adiposity: Recent advances in receptor signaling, neurobiology and pharmacology. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 8–13. [Google Scholar] [CrossRef]
- Edvinsson, L.; Goadsby, P.J.; Uddman, R. Amylin: Localization, effects on cerebral arteries and on local cerebral blood flow in the cat. Sci. World J. 2001, 1, 168–180. [Google Scholar] [CrossRef]
- Lorenzo, A.; Razzaboni, B.; Weir, G.C.; Yankner, B.A. Pancreatic islet cell toxicity of amylin associated with type-2 diabetes mellitus. Nature 1994, 368, 756–760. [Google Scholar] [CrossRef]
- May, P.C.; Boggs, L.N.; Fuson, K.S. Neurotoxicity of human amylin in rat primary hippocampal cultures: Similarity to Alzheimer’s disease amyloid-beta neurotoxicity. J. Neurochem. 1993, 61, 2330–2333. [Google Scholar] [CrossRef]
- Hull, R.L.; Westermark, G.T.; Westermark, P.; Kahn, S.E. Islet Amyloid: A Critical Entity in the Pathogenesis of Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2004, 89, 3629–3643. [Google Scholar] [CrossRef]
- Zhu, H.; Tao, Q.; Ang, T.F.A.; Massaro, J.; Gan, Q.; Salim, S.; Zhu, R.Y.; Kolachalama, V.B.; Zhang, X.; Devine, S.; et al. Association of Plasma Amylin Concentration with Alzheimer Disease and Brain Structure in Older Adults. JAMA Netw. Open 2019, 2, e199826. [Google Scholar] [CrossRef] [PubMed]
- Daval, M.; Bedrood, S.; Gurlo, T.; Huang, C.J.; Costes, S.; Butler, P.C.; Langen, R. The effect of curcumin on human islet amyloid polypeptide misfolding and toxicity. Amyloid 2010, 17, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.Q.; Townsend, M. Insulin resistance and amyloidogenesis as common molecular foundation for type 2 diabetes and Alzheimer’s disease. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2009, 1792, 482–496. [Google Scholar] [CrossRef] [PubMed]
- Rasgon, N.L.; Kenna, H.A.; Wroolie, T.E.; Kelley, R.; Silverman, D.; Brooks, J.; Williams, K.E.; Powers, B.N.; Hallmayer, J.; Reiss, A. Insulin resistance and hippocampal volume in women at risk for Alzheimer’s disease. Neurobiol. Aging 2011, 32, 1942–1948. [Google Scholar] [CrossRef] [PubMed]
- Willette, A.A.; Bendlin, B.B.; Starks, E.J.; Birdsill, A.C.; Johnson, S.C.; Christian, B.T.; Okonkwo, O.C.; La Rue, A.; Hermann, B.P.; Koscik, R.L.; et al. Association of Insulin Resistance With Cerebral Glucose Uptake in Late Middle-Aged Adults at Risk for Alzheimer Disease. JAMA Neurol. 2015, 72, 1013–1020. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Sasaki, K.; Tanizaki, Y.; Hata, J.; Fujimi, K.; Matsui, Y.; Sekita, A.; Suzuki, S.O.; Kanba, S.; Kiyohara, Y.; et al. Insulin resistance is associated with the pathology of Alzheimer disease. Hisayama Study 2010, 75, 764–770. [Google Scholar]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Luechapudiporn, R.; Phisalaphong, C.; Jirawatnotai, S. Curcumin extract for prevention of type 2 diabetes. Diabetes Care 2012, 35, 2121–2127. [Google Scholar] [CrossRef]
- Na, L.X.; Li, Y.; Pan, H.Z.; Zhou, X.L.; Sun, D.J.; Meng, M.; Li, X.X.; Sun, C.H. Curcuminoids exert glucose-lowering effect in type 2 diabetes by decreasing serum free fatty acids: A double-blind, placebo-controlled trial. Mol. Nutr. Food Res. 2013, 57, 1569–1577. [Google Scholar] [CrossRef]

| Characteristics | Total (n = 29) | Placebo (n = 15) | Curcumin (n = 14) | p Value |
|---|---|---|---|---|
| Age (years) | 52.3 ± 1.9 | 50.4 ± 2.6 | 54.5 ± 2.9 | 0.2998 |
| Males/females (n/n) | 12/17 | 6/9 | 6/8 | - |
| Ethnicity—no (%) | ||||
| Caucasian | 23 | 12 (80) | 11 (78.6) | - |
| Asian | 3 | 1 (6.7) | 2 (14.3) | - |
| Others | 3 | 2 (13.3) | 1 (7.1) | - |
| Anthropometry measures | ||||
| Body weight (kg) | 88.8 ± 3.0 | 90.7 ± 4.9 | 86.7 ± 3.5 | 0.5206 |
| Muscle mass (kg) | 33.3 ± 1.4 | 32.4 ± 1.7 | 34.4 ± 2.4 | 0.4998 |
| Body fat mass (kg) | 32.4 ± 2.2 | 33.7 ± 3.5 | 31.1 ± 3.0 | 0.5785 |
| Body mass index (kg. m−2) | 31.3 ± 1.0 | 32.3 ± 1.7 | 30.2 ± 1.1 | 0.3276 |
| Waist circumference(cm) | 105.4 ± 2.4 | 106.0 ± 3.9 | 104.9 ± 2.9 | 0.8246 |
| Percent body fat (%) | 34.7 ± 1.8 | 35.3 ± 2.2 | 34.8 ± 2.5 | 0.5467 |
| Plasma outcome measures | ||||
| Fasting glucose (mmol/L) | 5.4 ± 0.1 | 5.2 ± 0.1 | 5.6 ± 0.2 | 0.1121 |
| Fasting serum insulin (mIU/L) | 9.9 (4.9) | 10.3 (7.9) | 9.1 (4.6) | 0.6005 |
| HOMA2-IR | 1.3 (0.6) | 1.3 (1.1) | 1.2 (0.6) | 0.7268 |
| IAPP (ng/mL) | 4.5 (2.6) | 4.1 (2.6) | 3.9 (3.1) | 0.8948 |
| GSK-3β (ng/mL) | 3.0 (1.7) | 2.7 (1.8) | 3.4 (2.7) | 0.1625 |
| Dietary intakes (kj) | 9047.3 ± 424.9 | 8497.1 ± 599.6 | 9682.1 ± 573.06 | 0.1685 |
| Physical Activity (METs-minutes/week) | 2432 (4920) | 3894 (5214) | 1765 (1597) | 0.1161 |
| Outcome Measures | Treatment Group | Mean Change | p Value | Mean Difference between Treatment Groups | p Value |
|---|---|---|---|---|---|
| Body weight (kg) | Placebo | 0.64 ± 0.4 | 0.1731 | ||
| Curcumin | −0.1 ± 0.4 | 0.8272 | −0.7 ± 0.6 | 0.2292 | |
| Muscle mass (kg) | Placebo | 0.1 (0.1) | 0.8902 | ||
| Curcumin | 0.25 (0.7) | 0.4440 | 0.1(0.8) | 0.5257 | |
| Body fat mass (kg) | Placebo | 0.1 (0.4) | 0.7577 | ||
| Curcumin | −0.85 (0.9) | 0.8487 | −0.5 (2.1) | 0.3478 | |
| Body mass index (kg/m2) | Placebo | 0.20 (0.2) | 0.1945 | ||
| Curcumin | 0.03 (0.2) | 0.8296 | 0 (0.7) | 0.3573 | |
| Waist circumference (cm) | Placebo | 0.87 ± 0.7 | 0.2258 | ||
| Curcumin | −0.10 ± 0.8 | 0.8940 | −0.1 ± 1.0 | 0.3557 | |
| Percent body fat (%) | Placebo | 0.5 (1.8) | 0.4388 | ||
| Curcumin | −0.6 (1.6) | 0.5980 | 0 (2) | 0.7699 | |
| Fasting glucose (mmol/L) | Placebo | −0.06 ± 0.1 | 0.3625 | ||
| Curcumin | −0.07 ± 0.1 | 0.6041 | −0.004 ± 0.1 | 0.9747 | |
| Fasting serum insulin (µIU/L) | Placebo | 0.1 ± 0.4 | 0.8251 | ||
| Curcumin | −1.9 ± 0.6 | 0.0076 | −2.0 ± 0.4 | 0.0115 | |
| Dietary intakes (kj) | Placebo | −134.5 ± 479.2 | 0.7830 | ||
| Curcumin | 298.4 ± 487.9 | 0.5520 | −433.0 ± 686.6 | 0.5338 | |
| Physical activity (Metabolic equivalent-minute/week) | Placebo | −473 (3880) | 0.2202 | ||
| Curcumin | 104.5 (1508) | 0.6249 | 0 (2618) | 0.2386 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thota, R.N.; Rosato, J.I.; Dias, C.B.; Burrows, T.L.; Martins, R.N.; Garg, M.L. Dietary Supplementation with Curcumin Reduce Circulating Levels of Glycogen Synthase Kinase-3β and Islet Amyloid Polypeptide in Adults with High Risk of Type 2 Diabetes and Alzheimer’s Disease. Nutrients 2020, 12, 1032. https://doi.org/10.3390/nu12041032
Thota RN, Rosato JI, Dias CB, Burrows TL, Martins RN, Garg ML. Dietary Supplementation with Curcumin Reduce Circulating Levels of Glycogen Synthase Kinase-3β and Islet Amyloid Polypeptide in Adults with High Risk of Type 2 Diabetes and Alzheimer’s Disease. Nutrients. 2020; 12(4):1032. https://doi.org/10.3390/nu12041032
Chicago/Turabian StyleThota, Rohith N, Jessica I Rosato, Cintia B Dias, Tracy L Burrows, Ralph N Martins, and Manohar L Garg. 2020. "Dietary Supplementation with Curcumin Reduce Circulating Levels of Glycogen Synthase Kinase-3β and Islet Amyloid Polypeptide in Adults with High Risk of Type 2 Diabetes and Alzheimer’s Disease" Nutrients 12, no. 4: 1032. https://doi.org/10.3390/nu12041032
APA StyleThota, R. N., Rosato, J. I., Dias, C. B., Burrows, T. L., Martins, R. N., & Garg, M. L. (2020). Dietary Supplementation with Curcumin Reduce Circulating Levels of Glycogen Synthase Kinase-3β and Islet Amyloid Polypeptide in Adults with High Risk of Type 2 Diabetes and Alzheimer’s Disease. Nutrients, 12(4), 1032. https://doi.org/10.3390/nu12041032








