Recommended Intake of Key Food Groups and Cardiovascular Risk Factors in Australian Older, Rural-Dwelling Adults
Abstract
1. Introduction
2. Materials and Methods
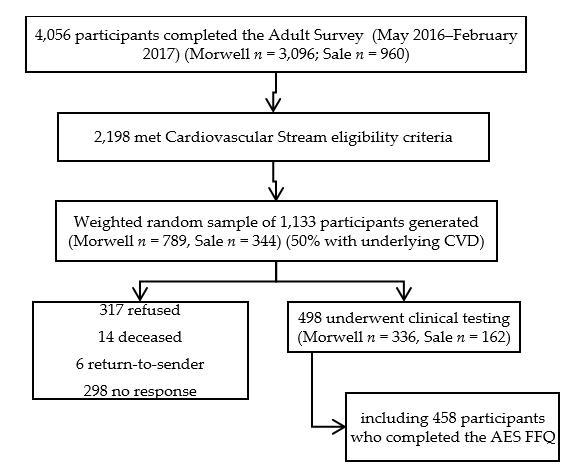
2.1. Participants
2.2. Measures
2.2.1. Cardiometabolic Risk Factors
2.2.2. Dietary Intake Assessment
2.3. Statistical Analysis
2.4. Ethics
3. Results
3.1. Participant Characteristics
3.2. Dietary Intake
3.3. Associations between Diet Quality and Cardiometabolic Risk Factors
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GBD 2017 Risk Factor Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1923–1994. [Google Scholar]
- GBD 2017 Diet Collaborators. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- National Health & Medical Research Council. Eat for Health: Australian Dietary Guidelines; National Health and Medical Research Council: Canberra, Australia, 2013. [Google Scholar]
- Australian Bureau of Statistics. Australian Health Survey: Consumption of food groups from the Australian Dietary Guidelines. Australian Bureau of Statistics: Canberra, Australia, 2016. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/4364.0.55.0122011-12 (accessed on 3 December 2019).
- Fayet-Moore, F.; Cassettari, T.; Tuck, K.; McConnell, A.; Petocz, P. Dietary fibre intake in Australia. Paper I: Associations with demographic, socio-economic, and anthropometric factors. Nutrients 2018, 10, 599. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, M.G.; Milte, C.M.; Crawford, D.; McNaughton, S.A. A revised Australian Dietary Guideline Index and its association with key sociodemographic factors, health behaviors and body mass index in peri-retirement aged adults. Nutrients 2016, 8, 160. [Google Scholar] [CrossRef] [PubMed]
- Bennett, E.; Peters, S.A.E.; Woodward, M. Sex differences in macronutrient intake and adherence to dietary recommendations: Findings from the UK Biobank. BMJ Open 2018, 8, e020017. [Google Scholar] [CrossRef] [PubMed]
- Heart Foundation of Australia. Heart Health Map for Latrobe. 2019. Available online: https://www.heartfoundation.org.au/for-professionals/heart-maps/australian-heart-maps (accessed on 19 December 2019).
- Collins, C.E.; Burrows, T.L.; Rollo, M.E.; Boggess, M.M.; Watson, J.F.; Guest, M.; Duncanson, K.; Pezdirc, K.; Hutchesson, M.J. The comparative validity and reproducibility of a diet quality index for adults: The Australian Recommended Food Score. Nutrients 2015, 7, 785–798. [Google Scholar] [CrossRef] [PubMed]
- Hazelwood Health Study. Adult Survey Volume 1 Comparison of Morwell and Sale. 2017. Available online: https://hazelwoodhealthstudy.org.au/study-findings/study-reports (accessed on 3 December 2019).
- Australian Bureau of Statistics. 033.0.55.001-Census of Population and Housing: Socio-Economic Indexes for Areas (SEIFA), Australia. 2016. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/2033.0.55.001~2016~Main%20Features~IRSAD~20 (accessed on 3 December 2019).
- World Health Organization. Body Mass Index. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 13 November 2019).
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro III, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare. The Active Australia Survey: A guide and manual for implementation, analysis and reporting. 2003. Available online: https://www.aihw.gov.au/reports/physical-activity/active-australia-survey/contents/table-of-contents (accessed on 3 December 2019).
- Australian Government Publishing Service. AUSNUT Australian Food and Nutrient Database; Australian New Zealand Food Authority; Australian Government Publishing Service: Canberra, Australia, 1999. [Google Scholar]
- Australian Government National Health & Medical Research Council. Nutrient Reference Values for Australia and New Zealand. 2017. Available online: www.nrv.gov.au (accessed on 3 December 2019).
- Banna, J.C.; McCrory, M.A.; Fialkowski, M.K.; Boushey, C. Examining plausibility of self-reported energy intake data: Considerations for method selection. Front. Nutr. 2017, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare. A picture of overweight and obesity in Australia 2017. Canberra. 2017. Available online: www.aihw.gov.au (accessed on 3 December 2019).
- Australian Bureau of Statistics. National Health Survey: First results, 2014–2015. Canberra; 2015. Catalogue number: 4364.0. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/4364.0.55.0012014-15 (accessed on 3 December 2019).
- Australian Bureau of Statistics. Australian Health Survey: Nutrition First Results-Foods and Nutrients, 2011–2012. Canberra; 2014. Catalogue Number 4364.0.55.007. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/lookup/4364.0.55.007main+features12011-12 (accessed on 23 March 2020).
- McLean, R.M.; Farmer, V.L.; Nettleton, A.; Cameron, C.M.; Cook, N.R.; Campbell, N.R.C.; TRUE Consortium (International Consortium for Quality Research on Dietary Sodium/Salt). Assessment of dietary sodium intake using a food frequency questionnaire and 24-hour urinary sodium excretion: A systematic literature review. J. Clin. Hypertens. 2017, 19, 1214–1230. [Google Scholar] [CrossRef] [PubMed]
- Waijers, P.M.; Feskens, E.J.; Ocke, M.C. A critical review of predefined diet quality scores. Br. J. Nutr. 2007, 97, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Mendelian Randomization of Dairy Consumption Working Group; CHARGE Consortium. Dairy Intake and Body Composition and Cardiometabolic Traits among Adults: Mendelian Randomization Analysis of 182041 Individuals from 18 Studies. Clin. Chem. 2019, 65, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, M.; Mente, A.; Rangarajan, S.; Sheridan, P.; Mohan, V.; Iqbal, R.; Gupta, R.; Lear, S.; Wentzel-Viljoen, E.; Avezum, A.; et al. Association of dairy intake with cardiovascular disease and mortality in 21 countries from five continents (PURE): A prospective cohort study. Lancet 2018, 392, 2288–2297. [Google Scholar] [CrossRef]
- Alexander, D.D.; Bylsma, L.C.; Vargas, A.J.; Cohen, S.S.; Doucette, A.; Mohamed, M.; Irvin, S.R.; Miller, P.E.; Watson, H.; Fryzek, J.P. Dairy consumption and CVD: A systematic review and meta-analysis. Br. J. Nutr. 2016, 115, 737–750. [Google Scholar] [CrossRef] [PubMed]
- Patterson, A.; Hure, A.; Burrows, T.; Jackson, J.; Collins, C. Diet quality and 10-year healthcare costs by BMI categories in the mid-age cohort of the Australian Longitudinal Study on Women’s Health. J. Hum. Nutr. Diet. 2018, 31, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Aljadani, H.M.; Sibbritt, D.; Patterson, A.; Collins, C. The Australian Recommended Food Score did not predict weight gain in middle-aged Australian women during six years of follow-up. Aust. N. Z. J. Public Health 2013, 37, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Australian Bureau of Statistics. National Health Survey: First results 2017–18. Sugar sweetened drinks and diet drinks. 2019. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/4364.0.55.001~2017-18~Main%20Features~Sugar%20sweetened%20and%20diet%20drink%20consumption~110 (accessed on 23 March 2020).
- Narain, A.; Kwok, C.S.; Mamas, M.A. Soft drink intake and the risk of metabolic syndrome: A systematic review and meta-analysis. Int. J. Clin. Pract. 2017, 71, e12927. [Google Scholar] [CrossRef] [PubMed]
- Satija, A.; Yu, E.; Willett, W.C.; Hu, F.B. Understanding nutritional epidemiology and its role in policy. Adv. Nutr. 2015, 6, 5–18. [Google Scholar] [CrossRef] [PubMed]
| Men (n = 256) | Women (n = 202) | p | |
|---|---|---|---|
| Age (years) | 70 ± 9 | 73 ± 7 | <0.001 |
| Area level disadvantage: n (%) first IRSAD quintile | 164 (64.1) | 141 (69.8) | 0.482 |
| School education to year 10 or below | 73 (28.6) | 99 (49.3) | <0.001 |
| Meeting physical activity guidelines (self-reported) | 123 (48.4) | 91 (45.3) | 0.510 |
| Body Mass Index (kg/m2) | 29.9 ± 5.0 | 30.3 ± 6.5 | 0.36 |
| Waist Circumference (cm) | 108.9 ± 13.4 | 100.3 ± 15.1 | <0.001 |
| Waist to Hip ratio | 1.04 ± 0.06 | 0.90 ± 0.07 | <0.001 |
| Diabetes | 53 (20.8) | 29 (14.6) | 0.110 |
| History of CVD | 131 (49.6) | 95 (45.6) | 0.408 |
| Current smoker | 19 (7.5) | 14 (7.0) | 0.096 |
| Systolic/Diastolic Blood Pressure (mmHg) | 133/72 ±17/11 | 135/72 ± 19/11 | 0.878 |
| Heart rate (bpm) | 63 ± 11 | 67 ± 11 | 0.003 |
| Total cholesterol (mmol/L) | 4.36 ± 0.99 | 4.85 ± 0.98 | <0.001 |
| HDL cholesterol (mmol/L) | 1.21 ± 0.33 | 1.47 ± 0.37 | <0.001 |
| LDL cholesterol (mmol/L) | 2.33 ± 0.88 | 2.58 ± 0.89 | 0.004 |
| HbA1c (%) | 6.1 ± 1.1 | 5.9 ± 0.8 | 0.128 |
| Estimated glomerular filtration rate (eGFR) (mL/min/1.73 m2) | 71.2 ± 15.6 a | 71.3 ± 15.7 b | 0.955 |
| Men (n = 256) | Women (n = 202) | p | |
|---|---|---|---|
| Energy (kJ/day) | 9727 (9326–10128) | 7989 (7537–8441) | <0.001 |
| ARFS-total (score/73) | 29.0 (27.8–30.3) | 33.1 (31.6–34.5) | <0.001 |
| ARFS-Vegetable (score/21) | 11.4 (10.8–12.0) | 13.5 (12.8–14.2) | <0.001 |
| ARFS-Fruit (score/12) | 4.7 (4.4–5.1) | 5.5 (5.1–5.9) | 0.004 |
| ARFS-Grain (score/13) | 4.0 (3.8–4.2) | 4.2 (4.0–4.5) | 0.198 |
| ARFS-Meat (score/7) | 2.7 (2.5–2.9) | 2.9 (2.7–3.0) | 0.230 |
| ARFS-Alternate Protein (score/6) | 1.7 (1.5–1.8) | 1.9 (1.7–2.0) | 0.071 |
| ARFS-Dairy (score/11) | 3.6 (3.4–3.8) | 4.0 (3.7–4.2) | 0.011 |
| ARFS-Spreads-Sauces (score/2) | 0.8 (0.7–0.9) | 0.8 (0.7–0.9) | 0.736 |
| ARFS-Water (score/1) | 0.3 (0.3–0.4) | 0.5 (0.4–0.6) | 0.001 |
| Carbohydrate (% of total energy) | 45.4 (44.5–46.3) | 42.7 (41.7–43.7) | <0.001 |
| Protein (% of total energy) | 18.7 (18.3–19.1) | 20.5 (20.0–21.0) | <0.001 |
| Fat (% of total energy) | 32.5 (31.8–33.2) | 34.3 (33.5–35.0) | 0.001 |
| Saturated fat (% of total energy) | 14.1 (13.7–14.5) | 15.0 (14.5–15.4) | 0.008 |
| Polyunsaturated fat (% of total energy) | 3.8 (3.7–3.9) | 4.0 (3.8–4.1) | 0.032 |
| Confectionery (% total energy) | 5.6 (4.9–6.2) | 4.6 (3.8–5.3) | 0.047 |
| Baked sweet products (% total energy) | 6.1 (5.5–6.7) | 5.9 (5.2–6.5) | 0.294 |
| Takeaway foods (% total energy) | 7.2 (6.7–7.8) | 5.9 (5.3–6.5) | 0.002 |
| Alcoholic beverage (% of total energy) | 5.0 (4.0–5.9) | 4.0 (3.0–4.0) | 0.177 |
| Sugar-sweetened drinks (% total energy) | 3.6 (3.0–4.2) | 1.7 (1.1–2.4) | <0.001 |
| Soft drinks (% consuming ≥ 1/week) | 48 (18.8) | 22 (10.9) | 0.252 |
| Sugar-sweetened drinks (%consuming≥ 1/week) | 76 (29.9) | 38 (18.8) | 0.008 |
| Fresh fish (% consuming ≥ 1/week) | 55 (21.5) | 71 (35.2) | 0.01 |
| Canned fish (% consuming ≥ 1/week) | 78 (30.5) | 75 (37.2) | 0.379 |
| Crumbed/battered fish (% consuming ≥ 1/week) | 50 (19.6) | 50 (24.7) | 0.036 |
| Sodium (mg/day) | 2329 (2218–2440) | 1984 (1857–2109) | <0.001 |
| Potassium (mg/day) | 3927 (3757–4097) | 3526 (3334–3718) | 0.003 |
| Men (n = 256) | Women (n = 202) | |
|---|---|---|
| Protein | 21 (8.2) | 8 (4.0) |
| Fibre | 148 (57.8) | 101 (50.2) |
| Folate | 104 (40.6) | 100 (49.8) |
| Vitamin A | 31 (12.1) | 17 (8.5) |
| Vitamin C | 5 (2) | 1 (0.5) |
| Thiamine | 29 (11.3) | 25 (12.4) |
| Riboflavin | 16 (6.3) | 12 (6) |
| Sodium | 155 (60.5) | 85 (42.3) |
| Potassium | 128 (50) | 57 (28.4) |
| Magnesium | 88 (34.4) | 40 (19.9) |
| Calcium | 103 (40.2) | 106 (52.7) |
| Iron | 10 (3.9) | 5 (2.5) |
| Zinc | 92 (35.9) | 12 (6) |
| Age- and Gender-adjusted (Model 1) a | Multivariable-Adjusted b (Model 2) | Model 2 excl. Potential Misreporting c | |
|---|---|---|---|
| Waist:hip | |||
| ARFS total | −0.001 (−0.001, 0.000) ** | −0.001 (−0.001, 0.000) * | −0.001 (−0.001, 0.000) * |
| ARFS vegetable | −0.001 (−0.003, 0.000) * | n.s. | n.s. |
| ARFS fruit | −0.003 (−0.005, −0.001) ** | −0.002 (−0.004, 0.000) * | −0.003(−0.005, −0.001) * |
| ARFS grain | −0.006 (−0.009, −0.003) *** | −0.004 (−0.007, −0.001) * | −0.004(−0.007,−0.001) ** |
| ARFS alt. protein | −0.005 (−0.009, 0.000)* | n.s. | n.s. |
| HDL cholesterol | |||
| ARFS vegetable | 0.008 (0.002, 0.015) ** | n.s. | 0.007 (0.000, 0.013) * |
| ARFS meat | 0.045 (0.023, 0.068) *** | 0.037 (0.015, 0.059) ** | 0.044 (0.021, 0.067) *** |
| eGFR | |||
| ARFS meat | 1.210 (0.002, 2.418) | 1.365 (0.164, 2.566) * | 1.305 (0.022, 2.587) * |
| ARFS alt. protein d | 1.339 (0.001, 2.677) | 1.342 (0.021, 2.664) * | n.s. |
| Total cholesterol | |||
| ARFS dairy | −0.080 (−0.129, −0.030) ** | −0.067 (−0.109, −0.024) ** | −0.073 (−0.118,−0.028) *** |
| Age- and Gender-Adjusted (Model 1) a | Multivariable Adjusted b (Model 2) | Model 2 excl. Potential Misreporting c | |
|---|---|---|---|
| Waist:hip | |||
| Crumbed/battered fish | 0.003 (0.000, 0.006) | 0.003 (0.000, 0.006) * | 0.004 (0.001, 0.007) ** |
| HDL cholesterol | |||
| Canned fish | 0.022 (0.009, 0.035) *** | 0.023 (0.010, 0.036) *** | 0.026 (0.013, 0.040) *** |
| eGFR | |||
| Fresh fish | 0.966 (0.239, 1.692) ** | 1.001 (0.273, 1.730) ** | 1.128 (0.373, 1.184) ** |
| HbA1c | |||
| Canned fish | −0.038 (−0.076,−0.001) * | −0.032 (−0.064,−0.001) * | −0.030 ( −0.63, 0.002) |
| Age- and Sex-Adjusted (Model 1) a | Multivariable Adjusted b (Model 2) | Multivariable Adjusted c (Model 3) | Model 3 Excluding Potential Misreporting d | |
|---|---|---|---|---|
| Waist:hip | ||||
| Sugar-sweetened beverages (%E) | 0.002 (0.001, 0.003) ** | 0.002 (0.001, 0.003) ** | 0.002 (0.001, 0.003) ** | 0.002 (0.001, 0.003) ** |
| Soft drink (number consumed/day) | 0.010 (0.002, 0.017) * | n.s. | n.s. | n.s. |
| HbA1c | ||||
| Sugar-sweetened beverage (%E) | 0.032 (0.014–0.051) *** | 0.035 (0.019–0.050) *** | 0.035 (0.019–0.050) *** | 0.029 (0.013–0.045) *** |
| Soft drink (number consumed/day)e | 0.262 (0.143, 0.380) *** | 0.174 (0.076, 0.272) *** | 0.178 (0.078, 0.277) *** | 0.159 (0.054, 0.264) ** |
| Diet soft drink (times consumed) | 0.058 (0.033, 0.083) *** | 0.029 (0.008, 0.050) ** | 0.029 (0.009, 0.050) ** | 0.030 (0.009, 0.051) ** |
| HDL cholesterol | ||||
| Sugar-sweetened beverage (%E) | −0.011 (−0.017,−0.004) *** | −0.009 (−0.015,−0.002) *** | −0.008 (−0.015,−0.001) * | −0.008 (−0.015, −0.001) * |
| Soft drink (number consumed/day) | −0.093 (−0.135,−0.051) *** | −0.062 (−0.104,−0.021) ** | −0.061(−0.103,−0.019) ** | −0.071 (−0.115, −0.027) ** |
| Diet soft drink intake | −0.011 (−0.020, −0.002) * | n.s. | n.s. | n.s. |
| eGFR | ||||
| Sugar-sweetened beverage (%E) | −0.419 (−0.748, −0.091) * | −0.400 (−0.744, −0.055) * | −0.363 (−0.709, −0.017) * | −0.396 (−0.759, −0.032) * |
| Soft drink (number consumed/day) | −3.802 (−6.099,−1.504) *** | −3.110 (−5.464, −0.756) * | −2.794 (−5.184, −0.404) * | −3.351 (−5.909, −0.792) * |
| Diet soft drink intake | −0.484 (−0.941, −0.028) * | n.s. | n.s. | n.s. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Owen, A.J.; Abramson, M.J.; Ikin, J.F.; McCaffrey, T.A.; Pomeroy, S.; Borg, B.M.; Gao, C.X.; Brown, D.; Liew, D. Recommended Intake of Key Food Groups and Cardiovascular Risk Factors in Australian Older, Rural-Dwelling Adults. Nutrients 2020, 12, 860. https://doi.org/10.3390/nu12030860
Owen AJ, Abramson MJ, Ikin JF, McCaffrey TA, Pomeroy S, Borg BM, Gao CX, Brown D, Liew D. Recommended Intake of Key Food Groups and Cardiovascular Risk Factors in Australian Older, Rural-Dwelling Adults. Nutrients. 2020; 12(3):860. https://doi.org/10.3390/nu12030860
Chicago/Turabian StyleOwen, Alice J., Michael J. Abramson, Jill F. Ikin, Tracy A. McCaffrey, Sylvia Pomeroy, Brigitte M. Borg, Caroline X. Gao, David Brown, and Danny Liew. 2020. "Recommended Intake of Key Food Groups and Cardiovascular Risk Factors in Australian Older, Rural-Dwelling Adults" Nutrients 12, no. 3: 860. https://doi.org/10.3390/nu12030860
APA StyleOwen, A. J., Abramson, M. J., Ikin, J. F., McCaffrey, T. A., Pomeroy, S., Borg, B. M., Gao, C. X., Brown, D., & Liew, D. (2020). Recommended Intake of Key Food Groups and Cardiovascular Risk Factors in Australian Older, Rural-Dwelling Adults. Nutrients, 12(3), 860. https://doi.org/10.3390/nu12030860







