The Effect of DHA Supplementation on Cognition in Patients with Bipolar Disorder: An Exploratory Randomized Control Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design and Participants
2.2. Neuropsychological Battery
2.3. Statistical Analysis
3. Results
3.1. Socio Demographic and Clinical Variables
3.2. Neuropsychological Differences Between the Four Groups
3.3. Correlations Between Tests and Clinical or Sociodemographic Variables in BD Patients
4. Discussion
5. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Saunders, K.E.A.; Goodwin, G.M. The course of bipolar disorder. Adv. Psychiatr. Treat. 2010, 16, 318–330. [Google Scholar] [CrossRef]
- Dore, G.; Romans, S.E. Impact of bipolar affective disorder on family and partners. J. Affect. Disord. 2001, 67, 147–158. [Google Scholar] [CrossRef]
- Kurtz, M.M.; Gerraty, R.T. A meta-analytic investigation of neurocognitive deficits in bipolar illness: Profile and effects of clinical state. Neuropsychology 2009, 23, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Bora, E.; Yucel, M.; Pantelis, C. Cognitive endophenotypes of bipolar disorder: A meta-analysis of neuropsychological deficits in euthymic patients and their first-degree relatives. J. Affect. Disord. 2009, 113, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Arán, A.; Vieta, E.; Reinares, M.; Colom, F.; Torrent, C.; Sánchez-Moreno, J.; Benabarre, A.; Goikolea, J.M.; Comes, M.; Salamero, M. Cognitive function across manic or hypomanic, depressed, and euthymic states in bipolar disorder. Am. J. Psychiatry 2004, 161, 262–270. [Google Scholar] [CrossRef]
- Torres, I.J.; DeFreitas, V.G.; DeFreitas, C.M.; Kauer-Sant’Anna, M.; Bond, D.J.; Honer, W.G.; Lam, R.W.; Yatham, L.N. Neurocognitive functioning in patients with bipolar I disorder recently recovered from a first manic episode. J. Clin. Psychiatry 2010, 71, 1234–1242. [Google Scholar] [CrossRef]
- Balanzá-Martínez, V.; Rubio, C.; Selva-Vera, G.; Martinez-Aran, A.; Sánchez-Moreno, J.; Salazar-Fraile, J.; Tabarés-Seisdedosa, R. Neurocognitive endophenotypes (endophenocognitypes) from studies of relatives of bipolar disorder subjects: A systematic review. Neurosci. Biobehav. Rev. 2008, 32, 1426–1438. [Google Scholar] [CrossRef]
- Yatham, L.N.; Mackala, S.; Basivireddy, J.; Ahn, S.; Walji, N.; Hu, C.; Lam, R.W.; Torres, I.J. Lurasidone versus treatment as usual for cognitive impairment in euthymic patients with bipolar I disorder: A randomised, open-label, pilot study. Lancet Psychiatry 2017, 4, 208–217. [Google Scholar] [CrossRef]
- McIntyre, R.S.; Florea, I.; Tonnoir, B.; Loft, H.; Lam, R.W.; Christensen, M.C. Efficacy of vortioxetine on cognitive functioning in working patients with major depressive disorder. J. Clin. Psychiatry 2017, 78, 115–121. [Google Scholar] [CrossRef]
- Maud, C. Vortioxetine in bipolar depression induces a mixed/manic switch. Australas. Psychiatry 2016, 24, 206–207. [Google Scholar] [CrossRef]
- Goss, A.J.; Kaser, M.; Costafreda, S.G.; Sahakian, B.J.; Fu, C.H. Modafinil augmentation therapy in unipolar and bipolar depression: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Psychiatry 2013, 74, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Ciappolino, V.; Mazzocchi, A.; Botturi, A.; Turolo, S.; Delvecchio, G.; Agostoni, C.; Brambilla, P. The Role of Docosahexaenoic Acid (DHA) on Cognitive Functions in Psychiatric Disorders. Nutrients 2019, 11, 769. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Preacher, K.J.; MacCallum, R.C.; Atkinson, C.; Malarkey, W.; Glaser, R. Chronic stress and age-related increases in the proinflammatory cytokine IL-6. Proc. Natl. Acad. Sci. USA 2003, 100, 9090–9095. [Google Scholar] [CrossRef]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Perinatal biochemistry and physiology of long-chain polyunsaturated fatty acids. J. Pediatr. 2003, 143 (Suppl. 4), 1–8. [Google Scholar] [CrossRef]
- European Food Safety Authority the Panel on Dietetic Products, Nutrition and Allergies on a request from Mead Johnson Nutritionals. Scientific opinion on DHA and ARA and visual development. EFSA J. 2009, 941, 1–14. [Google Scholar]
- Bernard, J.Y.; DeAgostini, M.; Forhan, A.; de Lauzon-Guillain, B.; Charles, M.A.; Heude, B. EDEN Mother-Child Cohort Study Group The dietary n6:n3 fatty acid ratio during pregnancy is inversely associated with child neurodevelopment in the EDEN mother-childcohort. J. Nutr. 2013, 143, 1481–1488. [Google Scholar]
- Cardoso, C.; Afonso, C.; Bandarra, N.M. Dietary DHA and health: Cognitive function ageing. Nutr. Res. Rev. 2016, 29, 281–294. [Google Scholar] [CrossRef]
- Goustard-Langelier, B.; Guesnet, P.; Durand, G.; Antoine, J.M.; Alessandri, J.M. n-3 and n-6 fatty acid enrichment by dietary fish oil and phospholipid sources in brain cortical areas and nonneural tissues of formula-fed piglets. Lipids 1999, 34, 5–16. [Google Scholar] [CrossRef]
- Anderson, V.; Fenwick, T.; Manly, T.; Robertson, I. Attentional skills following traumatic brain injury in childhood: A componential analysis. Brain Inj. 1998, 12, 937–949. [Google Scholar] [CrossRef]
- Barkley, R.A. The executive functions and self-regulation: An evolutionary neuropsychological perspective. Neuropsychol. Rev. 2001, 11, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Joffre, C.; Nadjar, A.; Lebbadi, M.; Calon, F.; Laye, S. n-3 LCPUFA improves cognition: The young, the old and the sick. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Salem, N.; Vandal, M.; Calon, F. The benefit of docosahexaenoic acid for the adult brain in aging and dementia. Prostaglandins Leukot. Essent. Fat. Acids 2015, 92, 15–22. [Google Scholar] [CrossRef]
- Echeverría, F.; Valenzuela, R.; Catalina Hernandez-Roda, M.; Valenzuela, A. Docosahexaenoic acid (DHA), a fundamental fatty acid for the brain: New dietary sources. Prostaglandins Leukot. Essent. Fat. Acids 2017, 124, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Conklin, S.M. Dietary intake of the long-chain omega-3 fatty acids is associated with increased grey matter volume in the perigenual cingulate cortex. In Proceedings of the 65th Annual Scientific Meeting of the American Psychosomatic Society, Budapest, Hungary, 7–10 March 2007. [Google Scholar]
- Ciappolino, V.; Delvecchio, G.; Agostoni, C.; Mazzocchi, A.; Altamura, A.C.; Brambilla, P. The role of n-3 polyunsaturated fatty acids (n-3PUFAs) in affective disorders. J. Affect. Disord. 2017, 224, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Pajak, A.; Marventano, S.; Castellano, S.; Galvano, F.; Bucolo, C.; Drago, F.; Caraci, F. Role of omega-3 fatty acids in the treatment of depressive disorders: A comprehensive meta-analysis of randomized clinical trials. PLoS ONE 2014, 9, e96905. [Google Scholar] [CrossRef]
- Osher, Y.; Bersudsky, Y.; Belmaker, R.H. Omega-3 eicosapentaenoic acid in bipolar depression: Report of a small open-label study. J. Clin. Psychiatry 2005, 66, 726–729. [Google Scholar] [CrossRef]
- Frangou, S.; Lewis, M.; McCrone, P. Efficacy of ethyl-eicosapentaenoic acid in bipolar depression: Randomized double-blind placebo-controlled study. Br. J. Psychiatry 2006, 188, 46–50. [Google Scholar] [CrossRef]
- Stoll, A.L.; Severus, W.E.; Freeman, M.P.; Rueter, S.; Zboyan, H.A.; Diamond, E.; Cress, K.K.; Marangell, L.B. MD Omega 3 fatty acids in bipolar disorder: A preliminary double-blind, placebo-controlled trial. Arch. Gen. Psychiatry 1999, 56, 407–412. [Google Scholar] [CrossRef]
- Agostoni, C.; Massetto, N.; Biasucci, G.; Rottoli, A.; Bonvissuto, M.; Bruzzese, M.G.; Giovannini, M.; Riva, E. Effects of long-chain polyunsaturated fatty acid supplementation on fatty acid status and visual function in treated children with hyperphenylalaninemia. J. Pediatr. 2000, 137, 504–509. [Google Scholar] [CrossRef] [PubMed]
- First, M.B.; Spitzer, R.L.; Gibbon, M.; Williams, J.B.W. Structured Clinical Interview for DSM-IV Axis I Disorders, Clinician Version (SCID-CV); Biometrics Research, New York State Psychiatric Institute: New York, NY, USA, 2002. [Google Scholar]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Young, R.C.; Biggs, J.T.; Ziegler, V.E.; Meyer, D.A. A rating scale for mania: Reliability, validity and sensitivity. Br. J. Psychiatry 1978, 133, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Caletti, E.; Paoli, R.A.; Fiorentini, A.; Cigliobianco, M.; Zugno, E.; Serati, M.; Orsenigo, G.; Grillo, P.; Zago, S.; Caldiroli, A.; et al. Neuropsychology, social cognition and global functioning among bipolar, schizophrenic patients and healthy controls: Preliminary data. Front. Hum. Neurosci. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- First, M.B.; Spitzer, R.L.; Gibbon, M.L.; Williams, J.B.W. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Non-Patient Edition; (SCID-I/NP) Biometrics Research, New York State Psychiatric Institute: New York, NY, USA, 2002. [Google Scholar]
- Keefe, R.S.; Fox, K.H.; Davis, V.G.; Kennel, C.; Walker, T.M.; Burdick, K.E.; Harvey, P.D. The Brief Assessment of Cognition in Affective Disorders (BAC-A): Performance of patients with bipolar depression and healthy controls. J. Affect. Disord. 2014, 166, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Bauer, I.E.; Keefe, R.S.; Sanches, M.; Suchting, R.; Green, C.E.; Soares, J.C. Evaluation of cognitive function in bipolar disorder using the Brief Assessment of Cognition in Affective Disorders (BAC-A). J. Psychiatr. Res. 2015, 60, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, M.G.; Bonivento, C.; Garzitto, M.; Caletti, E.; Perlini, C.; Piccin, S.; Lazzaretti, M.; Marinelli, V.; Sala, M.; Abbiati, V.; et al. The brief assessment of cognition in affective disorders: Normative data for the Italian population. J. Affect. Disord. 2019, 252, 245–252. [Google Scholar] [CrossRef]
- The R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Goeman, J.J.; Finos, L. The inheritance procedure: Multiple testing of tree structured hypotheses. Stat. Appl. Genet. Mol. Biol. 2012, 11, 11. [Google Scholar] [CrossRef]
- Muldoon, M.F.; Ryan, C.M.; Yao, J.K.; Conklin, S.M.; Manuck, S.B. Long-chain Omega-3 Fatty Acids and Optimization of Cognitive Performance. Mil. Med. 2014, 179 (Suppl. 11), 95–105. [Google Scholar] [CrossRef]
- Fontani, G.; Corradeschi, F.; Felici, A.; Alfatti, F.; Migliorini, S.; Lodi, L. Cognitive and physiological effects of Omega-3 polyunsaturated fatty acid supplementation in healthy subjects. Eur. J. Clin. Investig. 2005, 35, 691–699. [Google Scholar] [CrossRef]
- Stonehouse, W.; Conlon, C.A.; Podd, J.; Hill, S.R.; Minihane, A.M.; Haskell, C.; Kennedy, D. DHA supplementation improved both memory and reaction time in healthy young adults: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 1134–1143. [Google Scholar] [CrossRef]
- Conklin, S.M.; Gianaros, P.J.; Brown, S.M.; Yao, J.K.; Hariri, A.R.; Manuck, S.B.; Muldon, M.F. Long-chain omega-3 fatty acid intake is associated positively with corticolimbic gray matter volume in healthy adults. Neurosci. Lett. 2007, 421, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.Y.; Huang, S.Y.; Su, K.P. A meta-analytic review of polyunsaturated fatty acid compositions in patients with depression. Biol. Psychiatry 2010, 68, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Peet, M.; Murphy, B.; Shay, J.; Horrobin, D. Depletion of omega-3 fatty acid levels in red blood cell membranes of depressive patients. Biol. Psychiatry 1998, 43, 315–319. [Google Scholar] [CrossRef]
- McNamara, R.K. Evaluation of docosahexaenoic acid deficiency as a preventable risk factor for recurrent affective disorders: Current status, future directions, and dietary recommendations. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Sylvia, L.G.; Nierenberg, A.A.; Stange, J.P.; Peckham, A.D.; Deckersbach, T. Development of an integrated psychosocial treatment to address the medical burden associated with bipolar disorder. J. Psychiatr. Pract. 2011, 17, 224–232. [Google Scholar] [CrossRef]
- Kemp, D.E.; Gao, K.; Chan, P.K.; Ganocy, S.J.; Findling, R.L.; Calabrese, J.R. Medical comorbidity in bipolar disorder: Relationship between illnesses of the endocrine/metabolic system and treatment outcome. Bipolar Disord. 2010, 12, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Hibbeln, J.R.; Palmer, J.W.; Davis, J.M. Are disturbances in lipid-protein interactions by phospholipase-A2 a predisposing factor in affective illness? Biol. Psychiatry 1989, 25, 945–961. [Google Scholar] [CrossRef]
- Noaghiul, S.; Hibbeln, J.R. Cross-national comparisons of seafood consumption and rates of bipolar disorders. Am. J. Psychiatry 2003, 160, 2222–2227. [Google Scholar] [CrossRef]
- Sublette, M.E.; Bosetti, F.; DeMar, J.C.; Ma, K.; Bell, J.M.; Fagin-Jones, S.; Russ, M.J.; Rapoport, S.I. Plasma free polyunsaturated fatty acid levels are associated with symptom severity in acute mania. Bipolar Disord. 2007, 9, 759–765. [Google Scholar] [CrossRef]
- McNamara, R.K.; Jandacek, R.; Rider, T.; Tso, P.; Dwivedi, Y.; Pandey, G.N. Selective deficits in erythrocyte docosahexaenoic acid composition in adult patients with bipolar disorder and major depressive disorder. J. Affect. Disord. 2010, 126, 303–311. [Google Scholar] [CrossRef]
- Evans, S.J.; Kamali, M.; Prossin, A.R.; Harrington, G.J.; Ellingrod, V.L.; McInnis, M.G.; Burant, C.F. Association of plasma omega-3 and omega-6 lipids with burden of disease measures in bipolar subjects. J. Psychiatr. Res. 2012, 46, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.J.; Prossin, A.R.; Harrington, G.J.; Kamali, M.; Ellingrod, V.L.; Burant, C.F.; McInnis, M.G. Fats and factors: Lipid profiles associate with personality factors and suicidal history in bipolar subjects. PLoS ONE 2012, 7, e29297. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Jandacek, R.; Rider, T.; Tso, P.; Cole-Strauss, A.; Lipton, J.W. Differential effects of antipsychotic medications on polyunsaturated fatty acid biosynthesis in rats: Relationship with liver delta6-desaturase expression. Schizophr. Res. 2011, 129, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Tournikioti, K.; Ferentinos, P.; Michopoulos, I.; Alevizaki, M.; Soldatos, C.R.; Dikeos, D.; Douzenis, A. Clinical and treatment-related predictors of cognition in bipolar disorder: Focus on visual paired associative learning. Eur. Arch. Psychiatry Clin. Neurosci. 2017, 267, 661–669. [Google Scholar] [CrossRef]
- Altamura, A.C.; Buoli, M.; Caldiroli, A.; Caron, L.; Cumerlato Melter, C.; Dobrea, C.; Cigliobianco, M.; Zanelli Quarantini, F. Misdiagnosis, duration of untreated illness (DUI) and outcome in bipolar patients with psychotic symptoms: A naturalistic study. J. Affect. Disord. 2015, 182, 70–75. [Google Scholar] [CrossRef]
- Green, M.F. Cognitive impairment and functional outcome in schizophrenia and bipolar disorder. J. Clin. Psychiatry 2006, 67, e12. [Google Scholar] [CrossRef]
- Harris, W.S.; Masson, S.; Barlera, S.; Milani, V.; Pileggi, S.; Franzosi, M.G.; Tognoni, G.; Latini, R.; Marchioli, R.; Tavazzi, L. Red blood cell oleic acid levels reflect olive oil intake while omega-3 levels reflect fish intake and the use of omega-3 acid ethyl esters: The Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico-Heart Failure trial. Nutr. Res. 2016, 36, 989–994. [Google Scholar] [CrossRef]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.; Madden, J.; Gambell, J.; Young, S.; Wang, L.; Jebb, S.; Calder, P. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef]
- Meyer, B.J.; Groot, R.H.M. Effects of Omega-3 Long Chain Polyunsaturated Fatty Acid Supplementation on Cardiovascular Mortality: The Importance of the Dose of DHA. Nutrients 2017, 9, 1305. [Google Scholar] [CrossRef]
- Rogers, P.J.; Appleton, K.M.; Kessler, D.; Peters, T.J.; Gunnell, D.; Hayward, R.C.; Heatherley, S.V.; Christian, L.M.; McNaughton, S.A.; Ness, A. No effect of n-3 long-chain polyunsaturated fatty acid (EPA and DHA) supplementation on depressed mood and cognitive function: A randomised controlled trial. Br. J. Nutr. 2008, 99, 421–431. [Google Scholar] [CrossRef]
- Antypa, N.; Smelt, A.H.; Strengholt, A.; Van der Does, A.J. Effects of omega-3 fatty acid supplementation on mood and emotional information processing in recovered depressed individuals. J. Psychopharmacol. 2012, 26, 738–743. [Google Scholar] [CrossRef] [PubMed]
| BD-Omega 3 (n = 13) | BD-Placebo (n = 18) | HCs-Omega 3 (n = 7) | HCs Placebo (n = 8) | Statistics * | p-Value, Bonferroni Corrected | Post-hoc Results (After Correction for Multiple Comparisons with Holm Method) | |
|---|---|---|---|---|---|---|---|
| Age (years), mean ± SD | 36 ± 12 | 50.4 ± 11.3 | 33.1 ± 12.4 | 39.4 ± 13.9 | F =5.337, | p = 0.024 | HCs with Omega 3 < BD with placebo BD with placebo > BD with Omega 3 |
| Gender (males/females) | 3/10 | 6/12 | 4/3 | 2/6 | χ2 = 2.67 | p = 1 | |
| Educational level (years), mean ± SD | 14.5 ± 2.93 | 14.6 ± 3.36 | 15.6 ± 2.44 | 14.6 ± 2.26 | F = 0.24 | p = 1 | |
| Race | Caucasian | Caucasian | Caucasian | Caucasian | - | - | - |
| Age on onset, mean ± SD | 26.2 ± 9.68 | 29.2 ± 11.1 | - | - | t = 0.75 | p = 1 | |
| DUI (months), mean ± SD | 43.7 ± 59.9 | 67.9 ± 99.8 | - | - | t = 0.75 | p = 1 | |
| No. Hospitalization, mean ± SD | 2.38 ± 2.18 | 1.44 ± 1.69 | - | - | t = 1.31 | p = 1 | |
| GAF Total scores, mean ± SD | 79.2 ± 9.74 | 76.7 ± 8.51 | 91.6 ± 6.48 | 92.6 ± 4.9 | F = 10.78 | p = 0.008 | HCs with Omega 3 = HCs with placebo > BD with Omega 3 = BD with placebo |
| DHA plasma levels, mean ± SD | 2.13 ± 0.803 | 1.68 ± 0.392 | 2.59 ± 0.688 | 1.98 ± 0.039 | F = 4.78 | p = 0.047 | HCs with Omega 3 = HCs with placebo > BD with Omega 3 = BD with placebo |
| Delta DHA plasma levels, mean ± SD | 0.578 ± 0.661 | 0.243 ± 0.371 | 0.256 ± 0.604 | 0.270 ± 0.02 | F = 1.422 | p = 1 |
| BD Patients Omega 3 (n = 13) | BD Patients Placebo (n = 18) | HCs Omega 3 (n = 7) | HCs Placebo (n = 8) | Statistics* | p-Value, Bonferroni Corrected | |
|---|---|---|---|---|---|---|
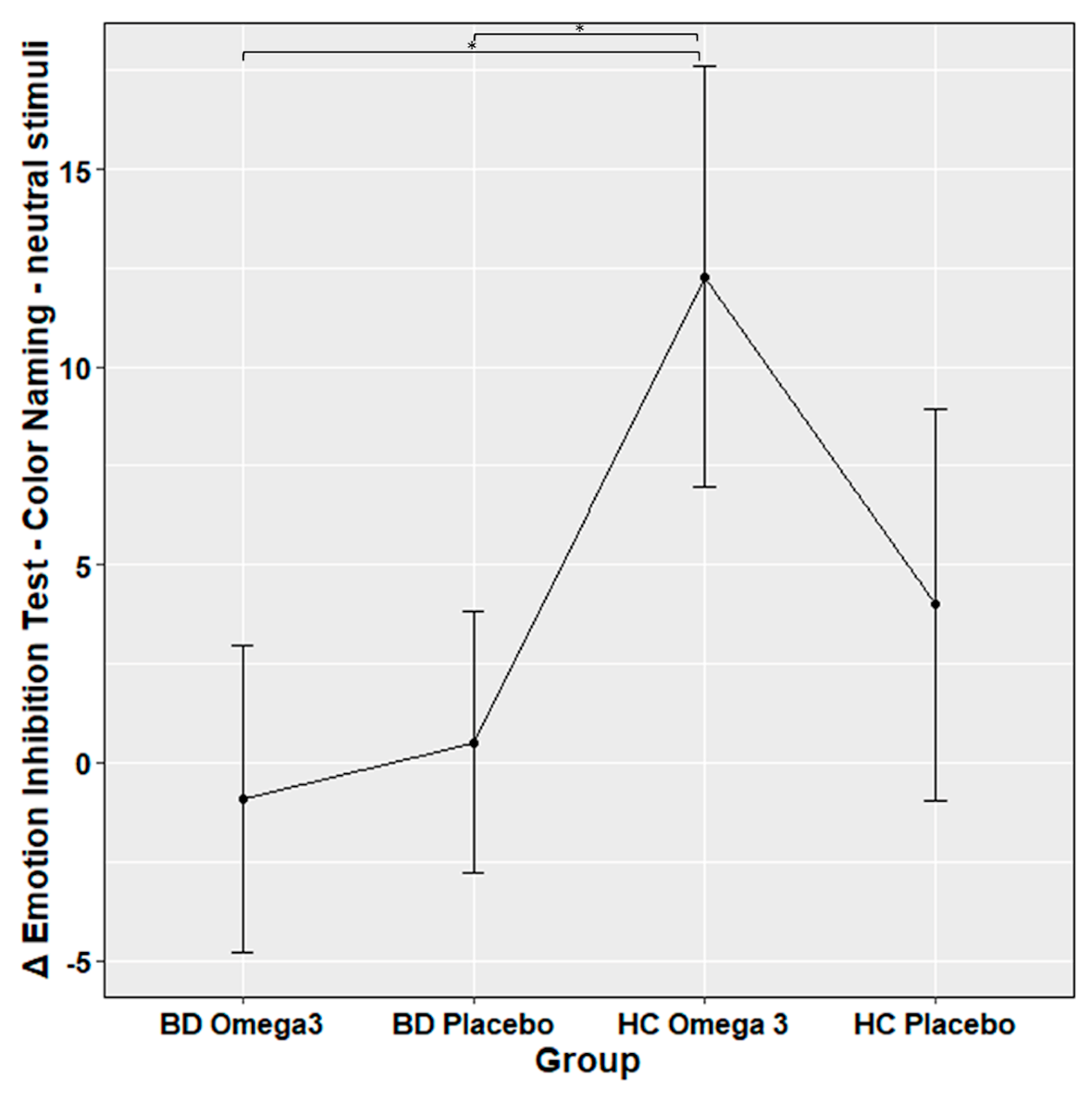
| Emotion Inhibition Test – Color Naming - neutral stimuli, mean accuracy ± SD | 0.0008 ± 0.14 | 0.024 ± 0.13 | 0.32 ± 0.25 | 0.121 ± 0.28 | HCs-Omega 3 vs. HCs-Placebo; z = 2.3 HCs-Omega 3 vs. BD-Omega 3; z = 3.9 HCs-Omega 3 vs. BD-Placebo; z = 3.7 HCs-Placebo vs. BD-Placebo; z = 1.5 HCs-Placebo vs. BD-Omega 3; z = 1.5 BD-Omega 3 vs. BD- Placebo; z = −0.05 | p = 0.4 p = 0.003 p = 0.005 p = 1 p = 1 p = 1 |
| Emotion Inhibition Test – Word Naming - neutral words, mean accuracy ± SD | 0.13 ± 0.27 | 0.86 ± 3.78 | 0.03 ± 0.23 | −0.05 ± 0.096 | HCs-Omega 3 vs. HCs-Placebo; z = −0.5 HCs-Omega 3 vs. BD-Omega 3; z = −0.7 HCs-Omega 3 vs. BD-Placebo; z = −1.6 HCs-Placebo vs. BD-Placebo; z = −1.2 HCs-Placebo vs. BD-Omega 3; z = −0.1 BD-Omega 3 vs. BD-Placebo; z = −1.1 | p = 1 p = 1 p = 1 p = 1 p = 1 p = 1 |
| Emotion Inhibition Test – Color Naming - neutral words, mean accuracy ± SD | 0.013 ± 0.16 | −0.026 ± 0.13 | 0.017 ± 0.09 | −0.028 ± 0.07 | HCs-Omega 3 vs. HCs-Placebo; z = 0.5 HCs-Omega 3 vs. BD-Omega 3; z = 0.1 HCs-Omega 3 vs. BD-Placebo; z = 0.6 HCs-Placebo vs. BD-Placebo; z = 0.05 HCs-Placebo vs. BD-Omega 3; z = -0.5 BD-Omega 3vs BD-Placebo; z = 0.6 | p = 1 p = 1 p = 1 p = 1 p = 1 p = 1 |
| Emotion Inhibition Test – Color Naming - affective words, mean accuracy ± SD | 0.0068 ± 0.11 | 0.018 ± 0.12 | 0.095 ± 0.27 | −0.0013 ± 0.12 | HCs-Omega 3 vs. HCs-Placebo; z = 1.5 HCs-Omega 3 vs. BD-OMEGA 3; z = 1.9 HCs-Omega 3 vs. BD-Placebo; z = 1.1 HCs-Placebo vs. BD-Placebo; z = −0.6 HCs-Placebo vs. BD-Omega 3; z = 0.2 BD-Omega 3 vs. BD-Placebo; z = −0.9 | p = 1 p = 1 p = 1 p = 1 p = 1 p = 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciappolino, V.; DelVecchio, G.; Prunas, C.; Andreella, A.; Finos, L.; Caletti, E.; Siri, F.; Mazzocchi, A.; Botturi, A.; Turolo, S.; et al. The Effect of DHA Supplementation on Cognition in Patients with Bipolar Disorder: An Exploratory Randomized Control Trial. Nutrients 2020, 12, 708. https://doi.org/10.3390/nu12030708
Ciappolino V, DelVecchio G, Prunas C, Andreella A, Finos L, Caletti E, Siri F, Mazzocchi A, Botturi A, Turolo S, et al. The Effect of DHA Supplementation on Cognition in Patients with Bipolar Disorder: An Exploratory Randomized Control Trial. Nutrients. 2020; 12(3):708. https://doi.org/10.3390/nu12030708
Chicago/Turabian StyleCiappolino, Valentina, Giuseppe DelVecchio, Cecilia Prunas, Angela Andreella, Livio Finos, Elisabetta Caletti, Francesca Siri, Alessandra Mazzocchi, Andrea Botturi, Stefano Turolo, and et al. 2020. "The Effect of DHA Supplementation on Cognition in Patients with Bipolar Disorder: An Exploratory Randomized Control Trial" Nutrients 12, no. 3: 708. https://doi.org/10.3390/nu12030708
APA StyleCiappolino, V., DelVecchio, G., Prunas, C., Andreella, A., Finos, L., Caletti, E., Siri, F., Mazzocchi, A., Botturi, A., Turolo, S., Agostoni, C., & Brambilla, P. (2020). The Effect of DHA Supplementation on Cognition in Patients with Bipolar Disorder: An Exploratory Randomized Control Trial. Nutrients, 12(3), 708. https://doi.org/10.3390/nu12030708










