The Association of Vitamin D Status with Lipid Profile and Inflammation Biomarkers in Healthy Adolescents
Abstract
1. Introduction
2. Material and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hill, T.R.; Aspray, T.J. The role of vitamin D in maintaining bone health in older people. Ther. Adv. Musculoskelet. Dis. 2017, 9, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Li, N.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and Chronic Diseases. Aging Dis. 2017, 8, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Rolf, L.; Muris, A.-H.; Hupperts, R.; Damoiseaux, J. Illuminating vitamin D effects on B-cells—The multiple sclerosis perspective. Immunology 2016, 147, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Hoe, E.; Nathanielsz, J.; Toh, Z.Q.; Spry, L.; Marimla, R.; Balloch, A.; Mulholland, E.K.; Licciardi, P. Anti-Inflammatory Effects of Vitamin D on Human Immune Cells in the Context of Bacterial Infection. Nutrients 2016, 8, 806. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Kohler, L.N.; Kunihiro, A.G.; Jurutka, P. Vitamin D and Colorectal, Breast, and Prostate Cancers: A Review of the Epidemiological Evidence. J. Cancer 2016, 7, 232–240. [Google Scholar] [CrossRef]
- Christodoulou, S.; Goula, T.; Ververidis, A.; Drosos, G. Vitamin D and Bone Disease. BioMed Res. Int. 2012, 2013, 1–6. [Google Scholar] [CrossRef]
- Kota, S.; Jammula, S.; Kota, S.; Meher, L.; Modi, K. Correlation of vitamin D, bone mineral density and parathyroid hormone levels in adults with low bone density. Indian J. Orthop. 2013, 47, 402–407. [Google Scholar] [CrossRef]
- Lips, P.; Goldsmith, D.; De Jongh, R. Vitamin D and osteoporosis in chronic kidney disease. J. Nephrol. 2017, 30, 671–675. [Google Scholar] [CrossRef]
- Welsh, P.; Sattar, N. Vitamin D and chronic disease prevention. BMJ 2014, 348, g2280. [Google Scholar] [CrossRef]
- Ashtari, F.; Toghianifar, N.; Esfahani, S.H.Z.; Mansourian, M. Short-Term Effect of High-Dose Vitamin D on the Level of Interleukin 10 in Patients with Multiple Sclerosis: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Neuroimmunomodulation 2015, 22, 400–404. [Google Scholar] [CrossRef]
- Heine, G.; Niesner, U.; Chang, H.-D.; Steinmeyer, A.; Zügel, U.; Zuberbier, T.; Radbruch, A.; Worm, M. 1,25-dihydroxyvitamin D3promotes IL-10 production in human B cells. Eur. J. Immunol. 2008, 38, 2210–2218. [Google Scholar] [CrossRef] [PubMed]
- Slavin, A.J.; Maron, R.; Weiner, H.L. Mucosal administration of IL-10 enhances oral tolerance in autoimmune encephalomyelitis and diabetes. Int. Immunol. 2001, 13, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Mangin, M.; Sinha, R.; Fincher, K. Inflammation and vitamin D: The infection connection. Inflamm. Res. 2014, 63, 803–819. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, L.; Xu, H.-J.; Li, Y.; Hu, C.-M.; Yang, J.-Y.; Sun, M. The Anti-Inflammatory Effects of Vitamin D in Tumorigenesis. Int. J. Mol. Sci. 2018, 19, 2736. [Google Scholar] [CrossRef] [PubMed]
- Galler, J.R.; Koethe, J.R.; Yolken, R.H. Neurodevelopment: The Impact of Nutrition and Inflammation during Adolescence in Low-Resource Settings. Pediatrics 2017, 139, S72–S84. [Google Scholar] [CrossRef] [PubMed]
- Vohr, B.R.; Davis, E.P.; Wanke, C.A.; Krebs, N.F. Neurodevelopment: The Impact of Nutrition and Inflammation during Preconception and Pregnancy in Low-Resource Settings. Pediatrics 2017, 139, 139. [Google Scholar] [CrossRef]
- Wajant, H.; Siegmund, D. TNFR1 and TNFR2 in the Control of the Life and Death Balance of Macrophages. Front. Cell Dev. Biol. 2019, 7, 91. [Google Scholar] [CrossRef]
- Dobnig, H.; Pilz, S.; Scharnagl, H.; Renner, W.; Seelhorst, U.; Wellnitz, B.; Kinkeldei, J.; Boehm, B.O.; Weihrauch, G.; März, W. Independent Association of Low Serum 25-Hydroxyvitamin D and 1,25-Dihydroxyvitamin D Levels With All-Cause and Cardiovascular Mortality. Arch. Intern. Med. 2008, 168, 1340. [Google Scholar] [CrossRef]
- Li, Y.C.; Kong, J.; Wei, M.; Chen, Z.F.; Liu, S.Q.; Cao, L.P. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Investig. 2002, 110, 229–238. [Google Scholar] [CrossRef]
- Lee, J.H.; O’Keefe, J.H.; Bell, D.; Hensrud, D.D.; Holick, M.F. Vitamin D deficiency an important, common, and easily treatable cardiovascular risk factor? J. Am. Coll. Cardiol. 2008, 52, 1949–1956. [Google Scholar] [CrossRef]
- Wang, T.J.; Pencina, M.J.; Booth, S.L.; Jacques, P.F.; Ingelsson, E.; Lanier, K.; Benjamin, E.J.; D’Agostino, R.B.; Wolf, M.; Vasan, R.S.; et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Figenschau, Y.; Hutchinson, M.; Emaus, N.; Grimnes, G. High serum 25-hydroxyvitamin D concentrations are associated with a favorable serum lipid profile. Eur. J. Clin. Nutr. 2010, 64, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Si, S.; Liu, J.; Wang, Z.; Jia, H.; Feng, K.; Sun, L.; Song, S. The Associations of Serum Lipids with Vitamin D Status. PLOS ONE 2016, 11, e0165157. [Google Scholar] [CrossRef] [PubMed]
- Kelishadi, R.; Farajzadegan, Z.; Bahreynian, M. Association between vitamin D status and lipid profile in children and adolescents: A systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2014, 65, 404–410. [Google Scholar] [CrossRef]
- Mirmiran, P.; Hosseini-Esfahani, F.; Mehrabi, Y.; Hedayati, M.; Azizi, F. Reliability and relative validity of an FFQ for nutrients in the Tehran Lipid and Glucose Study. Public Health Nutr. 2009, 13, 654. [Google Scholar] [CrossRef]
- Kennel, K.A.; Drake, M.T.; Hurley, D.L. Vitamin D Deficiency in Adults: When to Test and How to Treat. Mayo Clin. Proc. 2010, 85, 752–758. [Google Scholar] [CrossRef]
- Pais, P.; Kamath, D.Y.; Xavier, D.; Sigamani, A. High sensitivity C-reactive protein (hsCRP) & cardiovascular disease: An Indian perspective. Indian J. Med. Res. 2015, 142, 261–268. [Google Scholar]
- Plichta, S.B.; Kelvin, E.A.; Munro, B.H. Munro’s Statistical Methods for Health Care Research; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012. [Google Scholar]
- Spotila, L.D.; Rodriguez, H.; Koch, M.; Adams, K.; Caminis, J.; Tenenhouse, H.S.; Tenenhouse, A. Association of a Polymorphism in the TNFR2 Gene with Low Bone Mineral Density. J. Bone Miner. Res. 2000, 15, 1376–1383. [Google Scholar] [CrossRef]
- Barker, T.; Brown, K.B.; E Rogers, V. Soluble TNF Receptors are Modulated by Vitamin D Status but not by Acute Perturbations in 25-Hydroxyvitamin D Following A Bolus of Supplemental Vitamin D. J. Cytokine Biol. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Bertolini, D.R.; Nedwin, G.E.; Bringman, T.S.; Smith, D.D.; Mundy, G.R. Stimulation of bone resorption and inhibition of bone formation in vitro by human tumour necrosis factors. Nature 1986, 319, 516–518. [Google Scholar] [CrossRef]
- Nguyen, L.; E Dewhirst, F.; Hauschka, P.V.; Stashenko, P. Interleukin-1 beta stimulates bone resorption and inhibits bone formation in vivo. Lymphokine Cytokine Res. 1991, 10, 15–21. [Google Scholar] [PubMed]
- Stashenko, P.; Dewhirst, F.E.; Rooney, M.L.; Desjardins, L.A.; Heeley, J.D. Interleukin-1β is a potent inhibitor of bone formation in vitro. J. Bone Miner. Res. 2009, 2, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Abu-Amer, Y. Tumor necrosis factor receptors types 1 and 2 differentially regulate osteoclastogenesis. J. Biol. Chem. 2000, 275, 275. [Google Scholar] [CrossRef] [PubMed]
- Opal, S.M.; DePalo, V.A. Anti-Inflammatory Cytokines. Chest 2000, 117, 1162–1172. [Google Scholar] [CrossRef]
- Takemura, M.; Matsumoto, H.; Niimi, A.; Ueda, T.; Matsuoka, H.; Yamaguchi, M.; Jinnai, M.; Muro, S.; Hirai, T.; Ito, Y.; et al. High sensitivity C-reactive protein in asthma. Eur. Respir. J. 2006, 27, 908–912. [Google Scholar] [CrossRef]
- Seyrek, N.; Karayaylali, I.; Balal, M.; Paydas, S.; Aikimbaev, K.; Çetíner, S.; Seydaoglu, G. Is there any relationship between serum levels of interleukin-10 and atherosclerosis in hemodialysis patients? Scand. J. Urol. Nephrol. 2005, 39, 405–409. [Google Scholar] [CrossRef]
- Ngo, D.T.; Sverdlov, A.; McNeil, J.J.; Horowitz, J.D. Does Vitamin D Modulate Asymmetric Dimethylarginine and C-Reactive Protein Concentrations? Am. J. Med. 2010, 123, 335–341. [Google Scholar] [CrossRef]
- Chen, N.; Wan, Z.; Han, S.; Li, B.; Zhang, Z.; Qin, L.-Q. Effect of Vitamin D Supplementation on the Level of Circulating High-Sensitivity C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials. Nutrients 2014, 6, 2206–2216. [Google Scholar] [CrossRef]
- Rahimi-Ardabili, H.; Gargari, B.P.; Farzadi, L. Effects of vitamin D on cardiovascular disease risk factors in polycystic ovary syndrome women with vitamin D deficiency. J. Endocrinol. Investig. 2012, 36, 28–32. [Google Scholar]
- Nam, G.E.; Kim, D.-H.; Cho, K.H.; Park, Y.G.; Han, K.D.; Kim, S.M.; Lee, S.H.; Ko, B.J.; Kim, M.J. 25-Hydroxyvitamin D insufficiency is associated with cardiometabolic risk in Korean adolescents: the 2008–2009 Korea National Health and Nutrition Examination Survey (KNHANES). Public Health Nutr. 2012, 17, 186–194. [Google Scholar] [CrossRef]
- Cho, H.-J.; Kang, H.-C.; Choi, S.-A.; Ju, Y.-C.; Lee, H.-S.; Park, H.-J. The possible role of Ca2+ on the activation of microsomal triglyceride transfer protein in rat hepatocytes. Biol. Pharm. Bull. 2005, 28, 1418–1423. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.; Lorenzen, J.K.; Svith, C.R.; Bartels, E.M.; Melanson, E.L.; Saris, W.H.; Tremblay, A.; Astrup, A. Effect of calcium from dairy and dietary supplements on faecal fat excretion: A meta-analysis of randomized controlled trials. Obes. Rev. 2009, 10, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Vaskonen, T.; Mervaala, E.; Sumuvuori, V.; Seppanen-Laakso, T.; Karppanen, H. Effects of calcium and plant sterols on serum lipids in obese Zucker rats on a low-fat diet. Br. J. Nutr. 2002, 87, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Song, S.J.; Si, S.; Liu, J.; Chen, X.; Zhou, L.; Jia, G.; Liu, G.; Niu, Y.; Wu, J.; Zhang, W.; et al. Vitamin D status in Chinese pregnant women and their newborns in Beijing and their relationships to birth size. Public Health Nutr. 2012, 16, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Frisch, S.; Berthold, H.; Götting, C.; Kuhn, J.; Kleesiek, K.; Stehle, P.; Koertke, H.; Koerfer, R. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am. J. Clin. Nutr. 2009, 89, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Karnchanasorn, R.; Ou, H.Y.; Chiu, K.C. Plasma 25-hydroxyvitamin D levels are favorably associated with beta-cell function. Pancreas 2012, 41, 863–868. [Google Scholar] [CrossRef]
- Howard, B.V. Insulin resistance and lipid metabolism. Am. J. Cardiol. 1999, 84, 28–32. [Google Scholar] [CrossRef]
- Tai, E.S.; Emmanuel, S.C.; Chew, S.K.; Tan, B.Y.; Tan, C. Isolated low HDL cholesterol: An insulin-resistant state only in the presence of fasting hypertriglyceridemia. Diabetes 1999, 48, 1088–1092. [Google Scholar] [CrossRef]
- Jiang, W.; Miyamoto, T.; Kakizawa, T.; Nishio, S.; Oiwa, A.; Takeda, T.; Suzuki, S.; Hashizume, K.; Miyamoto, T.; Kakizawa, T.; et al. Inhibition of LXRalpha signaling by vitamin D receptor: Possible role of VDR in bile acid synthesis. Biochem. Biophys. Res. Commun. 2006, 351, 176–184. [Google Scholar] [CrossRef]
- Fraunberger, P.; Schaefer, S.; Werdan, K.; Walli, A.K.; Seidel, D. Reduction of Circulating Cholesterol and Apolipoprotein Levels during Sepsis. Clin. Chem. Lab. Med. 1999, 37, 357–362. [Google Scholar] [CrossRef]
- Murch, O.; Collin, M.; Hinds, C.J.; Thiemermann, C. Lipoproteins in inflammation and sepsis. I. Basic science. Intensiv. Care Med. 2006, 33, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Shor, R.; Wainstein, J.; Oz, D.; Boaz, M.; Matas, Z.; Fux, A.; Halabe, A. Low HDL levels and the risk of death, sepsis and malignancy. Clin. Res. Cardiol. 2007, 97, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.A.; Berbée, J.F.; Rensen, P.C.; Kitchens, R.L. Apolipoprotein A-II augments monocyte responses to LPS by suppressing the inhibitory activity of LPS-binding protein. Innate Immun. 2008, 14, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-W.; Lee, H.-C. Vitamin D and health - The missing vitamin in humans. Pediatr. Neonatol. 2019, 60, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, D.; Lombardi, G.; Banfi, G. Concerning the vitamin D reference range: Pre-analytical and analytical variability of vitamin D measurement. Biochem. Med. 2017, 27, 030501. [Google Scholar] [CrossRef] [PubMed]
- Napoli, N.; Strollo, R.; Sprini, D.; Maddaloni, E.; Rini, G.B.; Carmina, E. Serum 25-OH Vitamin D in relation to Bone Mineral Density and Bone Turnover. Int. J. Endocrinol. 2014, 2014, 1–5. [Google Scholar] [CrossRef]
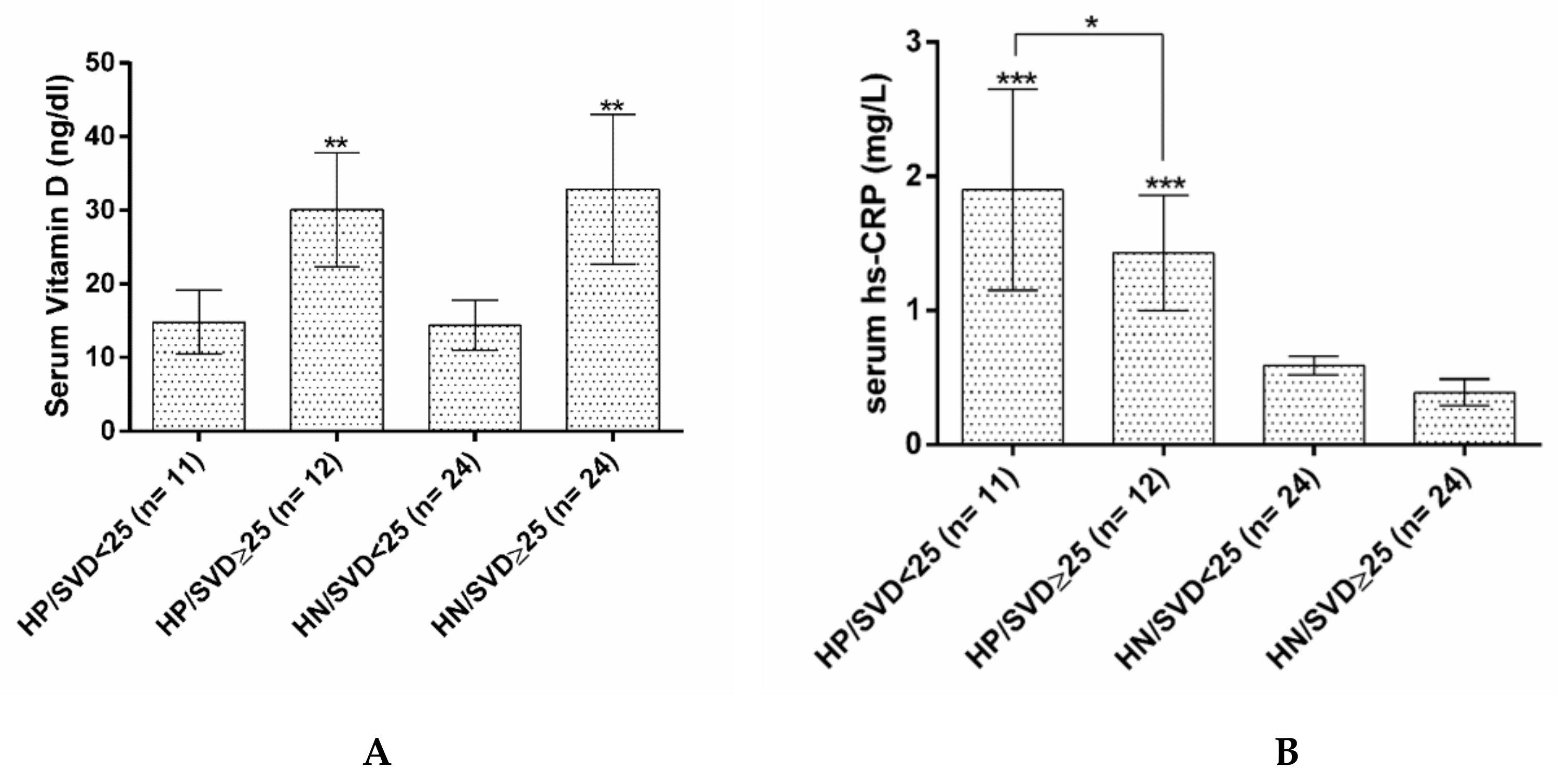
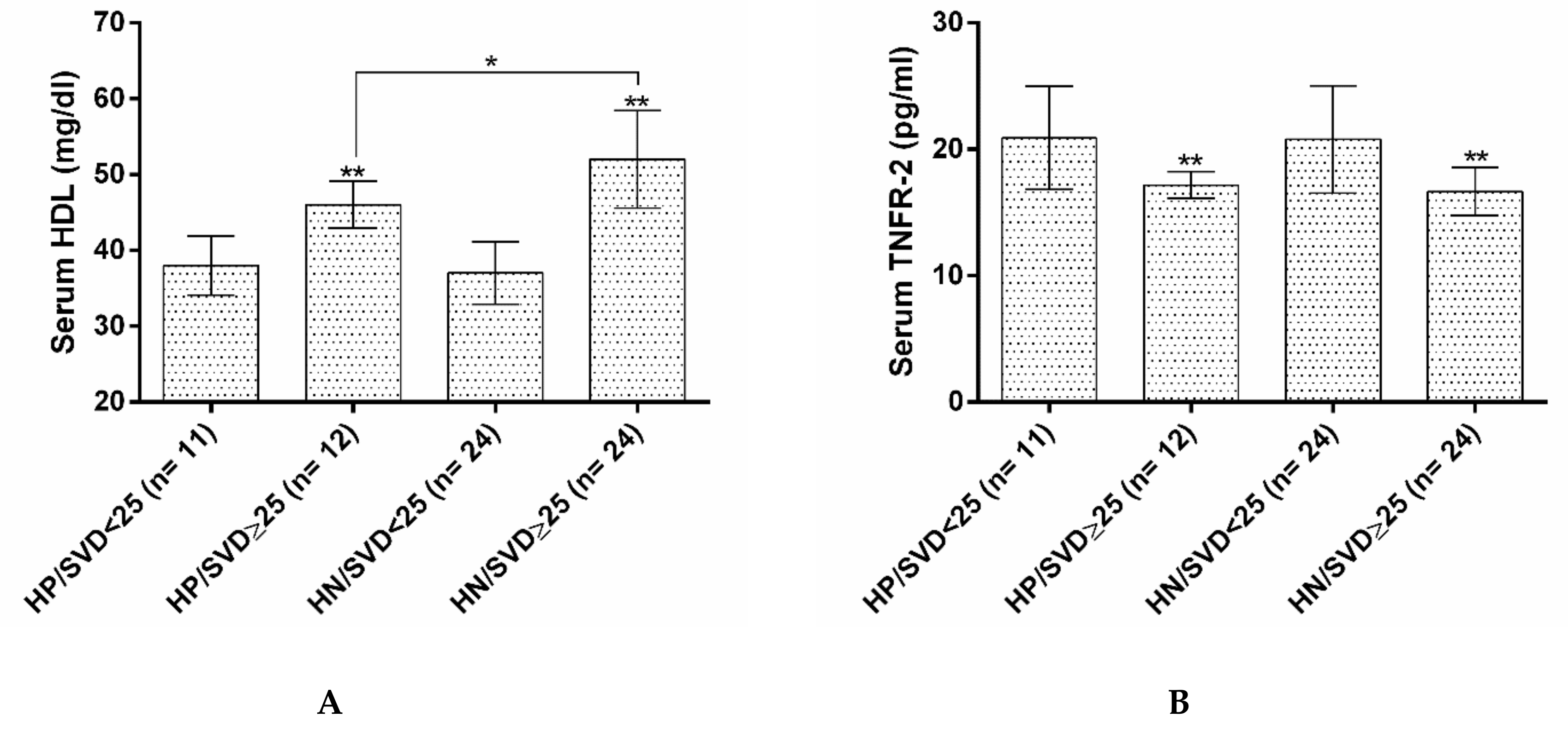
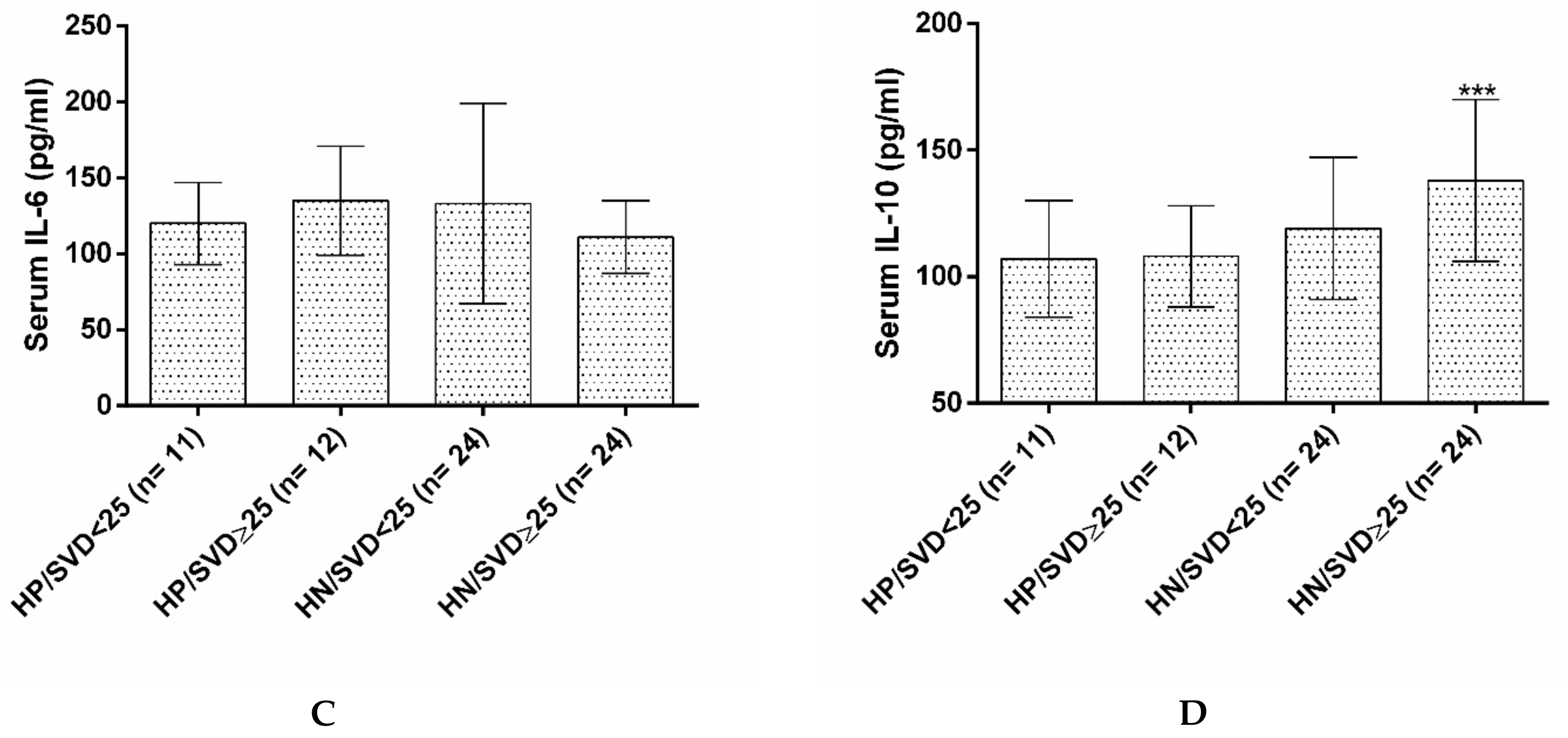
| Variable | Value | ||
|---|---|---|---|
| Anthropometric variables | Height (cm) | 165.67 ± 3.48 | |
| Weight (kg) | 63.19 ± 5.16 | ||
| BMI (kg/m2) | 23.00 ± 1.61 | ||
| BMI | Normal | 61 (85.9) | |
| Over-weight | 10 (14.1) | ||
| Dietary variables | Energy (Kcal) | 1908.00 ± 171.42 | |
| Carbohydrate (gr) | 245.94 ± 41.94 | ||
| Fat (gr) | 79.68 ± 16.64 | ||
| Protein (gr) | 58.31 ± 12.62 | ||
| Biochemical variables | Vitamin D (ng/mL) | 23.12 ± 11.31 | |
| TNFR-2 (pg/mL) | 18.84 ± 3.74 | ||
| IL-10 (pg/mL) | 114.82± 26.92 | ||
| IL-6 (pg/mL) | 124.08 ± 45.36 | ||
| hs-CRP (mg/L) | 1.28 ± 0.786 | ||
| Lipid profile | VLDL (mg/dL) | 24.36 ± 10.96 | |
| LDL (mg/dL) | 86.46 ± 18.32 | ||
| TC (mg/dL) | 166.97 ± 28.22 | ||
| TG (mg/dL) | 123.29 ± 54.01 | ||
| HDL (mg/dL) | 43.88 ± 8.21 | ||
| Variable | Vitamin D Status (<25 ng/mL vs. 25 ng/m L≤) | p-Value | ||
|---|---|---|---|---|
| Group with Serum Vitamin D below 25 (ng/mL) (SVD < 25) (n = 36) | Group with Serum Vitamin D 25 (ng/mL) and above (SVD ≥ 25) (n = 35) | |||
| Height (cm) | 165.40 ± 3.74 | 165.92 ± 4.43 | 0.647 * | |
| Weight (kg) | 61.76 ± 4.66 | 64.51 ± 6.93 | 0.101 * | |
| BMI (kg/m2) | 22.57 ± 1.54 | 22.41 ± 2.12 | 0.112 * | |
| BMI | Normal | 34 (94.4) | 27 (77.1) | 0.037 ** |
| Over-weight | 2 (5.6) | 8 (22.9) | ||
| Vitamin D (ng/mL) | 14.50 ± 3.70 | 31.98 ± 9.47 | <0.001 * | |
| TNFR-2 (pg/mL) | 20.82 ± 4.13 | 16.81 ± 1.67 | <0.001 * | |
| IL-10 (pg/mL) | 111.73 ± 25.42 | 118.00 ± 28.41 | 0.330 * | |
| IL-6 (pg/mL) | 128.93 ± 56.25 | 119.09 ± 30.48 | 0.365 * | |
| hs-CRP (mg/L) | 1.46 ± 0.880 | 1.10 ± 0.636 | 0.052 * | |
| VLDL (mg/dL) | 24.77 ± 11.78 | 23.94 ± 10.19 | 0.751 * | |
| LDL (mg/dL) | 88.61 ± 19.05 | 84.25 ± 17.54 | 0.320 * | |
| TC (mg/dL) | 169.33 ± 32.61 | 164.54 ± 23.08 | 0.479 * | |
| TG (mg/dL) | 126.30 ± 58.27 | 120.20 ± 49.91 | 0.637 * | |
| HDL (mg/dL) | 37.61 ± 4.01 | 50.34 ± 6.14 | <0.001 * | |
| Dietary energy (Kcal) | 1853.98 ± 154.15 | 1953.90 ± 204.12 | 0.471 | |
| Dietary carbohydrate (gr) | 223.01 ± 51.32 | 254.11 ± 71.45 | 0.362 | |
| Dietary fat (gr) | 83.43 ± 17.13 | 79.12 ± 20.11 | 0.231 | |
| Dietary protein (gr) | 53.46 ± 10.07 | 56.12 ± 13.45 | 0.193 | |
| Variable | hs-CRP Status | p-Value * | ||
|---|---|---|---|---|
| Positive (n = 23) | Negative (n = 48) | |||
| Height (cm) | 165.18 ± 4.23 | 166.03 ± 4.00 | 0.463 * | |
| Weight (kg) | 61.68 ± 4.56 | 64.30 ± 6.81 | 0.125 * | |
| BMI (kg/m2) | 22.62 ± 1.73 | 23.29 ± 1.98 | 0.211 * | |
| BMI | Normal | 29 (96.7) | 32 (78) | 0.040 ** |
| Over-weight | 1 (3.3) | 9 (22) | ||
| Vitamin D (ng/mL) | 23.60 ± 12.00 | 22.11 ± 9.87 | 0.609 * | |
| TNFR-2 (pg/mL) | 18.71 ± 3.86 | 19.11 ± 3.54 | 0.676 * | |
| IL-10 (pg/mL) | 108.20 ± 22.00 | 128.62 ± 31.27 | 0.002 * | |
| IL-6 (pg/mL) | 122.42 ± 50.69 | 127.53 ± 32.24 | 0.661 * | |
| hs-CRP (mg/L) | 1.66 ± 0.653 | 0.500 ± 0.313 | <0.001 * | |
| VLDL (mg/dL) | 24.95 ± 10.75 | 23.13 ± 11.51 | 0.515 * | |
| LDL (mg/dL) | 87.12 ± 18.74 | 85.08 ± 17.74 | 0.664 * | |
| TC (mg/dL) | 169.75 ± 27.82 | 161.17 ± 28.77 | 0.233 * | |
| TG (mg/dL) | 127.02 ± 52.62 | 115.52 ± 57.21 | 0.405 * | |
| HDL (mg/dL) | 44.72 ± 9.15 | 42.13 ± 5.57 | 0.215 * | |
| Dietary energy (Kcal) | 1976.11 ± 181.06 | 2010.21 ± 190.41 | 0.312 | |
| Dietary carbohydrate (gr) | 276.32 ± 62.11 | 290.03 ± 82,13 | 0.231 | |
| Dietary fat (gr) | 77.34 ± 12.11 | 79.33 ± 19.34 | 0.194 | |
| Dietary protein (gr) | 61.06 ± 11.09 | 59.34 ± 12.34 | 0.209 | |
| Variable | (SVD < 25) (n = 36) | (SVD ≥ 25) (n = 35) | Total (n = 71) | |
|---|---|---|---|---|
| Height (cm) | p-value | 0.001 | 0.646 | 0.854 |
| r | −0.615 | 0.093 | 0.026 | |
| Weight (kg) | p-value | 0.072 | 0.398 | 0.101 |
| r | −0.366 | 0.169 | 0.230 | |
| BMI (kg/m2) | p-value | 0.992 | 0.458 | 0.077 |
| r | 0.002 | 0.149 | 0.247 | |
| TNFR-2 (pg/mL) | p-value | 0.875 | 0.061 | 0.002 |
| r | −0.027 | 0.319 | −0.367 | |
| IL-10 (pg/mL) | p-value | 0.091 | 0.102 | 0.541 |
| r | −0.286 | −0.281 | −0.074 | |
| IL-6 (pg/mL) | p-value | 0.222 | 0.167 | 0.108 |
| r | −0.206 | −0.239 | −0.193 | |
| hs-CRP (mg/L) | p-value | 0.401 | 0.823 | 0.106 |
| r | −0.144 | 0.039 | −0.193 | |
| VLDL (mg/dL) | p-value | 0.947 | 0.885 | 0.731 |
| r | −0.011 | −0.025 | −0.041 | |
| LDL (mg/dL) | p-value | 0.716 | 0.827 | 0.576 |
| r | 0.063 | 0.038 | −0.067 | |
| TC (mg/dL) | p-value | 0.864 | 0.249 | 0.964 |
| r | −0.029 | 0.200 | −0.005 | |
| TG (mg/dL) | p-value | 0.916 | 0.934 | 0.700 |
| r | 0.018 | −0.014 | −0.047 | |
| HDL (mg/dL) | p-value | 0.165 | 0.561 | <0.001 |
| r | 0.236 | 0.102 | 0.657 | |
| Variable | hs-CRP Negative (n = 23) | hs-CRP Positive (n = 48) | Total (n = 71) | |
|---|---|---|---|---|
| Height (cm) | p-value | 0.150 | 0.823 | 0.588 |
| r | 0.269 | −0.051 | −0.077 | |
| Weight (kg) | p-value | 0.375 | 0.568 | 0.197 |
| r | 0.168 | −0.129 | −0.182 | |
| BMI (kg/m2) | p-value | 0.783 | 0.697 | 0.267 |
| r | 0.052 | −0.088 | −0.157 | |
| TNFR-2 (pg/mL) | p-value | 0.206 | 0.138 | 0.310 |
| r | 0.274 | 0.217 | 0.122 | |
| IL-10 (pg/mL) | p-value | 0.315 | 0.981 | 0.070 |
| r | 0.219 | 0.003 | −0.216 | |
| IL-6 (pg/mL) | p-value | 0.485 | 0.137 | 0.351 |
| r | 0.153 | 0.218 | 0.112 | |
| Vitamin D (ng/mL) | p-value | 0.020 | 0.031 | 0.106 |
| r | −0.481 | −0.311 | −0.193 | |
| VLDL (mg/dL) | p-value | 0.952 | 0.749 | 0.800 |
| r | 0.013 | -0.047 | 0.031 | |
| LDL (mg/dL) | p-value | 0.194 | 0.884 | 0.492 |
| r | 0.281 | 0.022 | 0.083 | |
| TC (mg/dL) | p-value | 0.219 | 0.872 | 0.315 |
| r | 0.267 | −0.024 | 0.121 | |
| TG (mg/dL) | p-value | 0.959 | 0.605 | 0.804 |
| r | 0.011 | −0.076 | 0.030 | |
| HDL (mg/dL) | p-value | 0.128 | 0.020 | 0.273 |
| r | −0.327 | −0.335 | −0.132 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yarparvar, A.; Elmadfa, I.; Djazayery, A.; Abdollahi, Z.; Salehi, F. The Association of Vitamin D Status with Lipid Profile and Inflammation Biomarkers in Healthy Adolescents. Nutrients 2020, 12, 590. https://doi.org/10.3390/nu12020590
Yarparvar A, Elmadfa I, Djazayery A, Abdollahi Z, Salehi F. The Association of Vitamin D Status with Lipid Profile and Inflammation Biomarkers in Healthy Adolescents. Nutrients. 2020; 12(2):590. https://doi.org/10.3390/nu12020590
Chicago/Turabian StyleYarparvar, Amirhossein, Ibrahim Elmadfa, Abolghassem Djazayery, Zahra Abdollahi, and Forouzan Salehi. 2020. "The Association of Vitamin D Status with Lipid Profile and Inflammation Biomarkers in Healthy Adolescents" Nutrients 12, no. 2: 590. https://doi.org/10.3390/nu12020590
APA StyleYarparvar, A., Elmadfa, I., Djazayery, A., Abdollahi, Z., & Salehi, F. (2020). The Association of Vitamin D Status with Lipid Profile and Inflammation Biomarkers in Healthy Adolescents. Nutrients, 12(2), 590. https://doi.org/10.3390/nu12020590





