What Serum Sodium Concentration Is Suggestive for Underhydration in Geriatric Patients?
Abstract
1. Introduction
2. Materials and Methods
2.1. Statistical Analysis
2.2. Ethics Approval
3. Results
3.1. Hydration Status
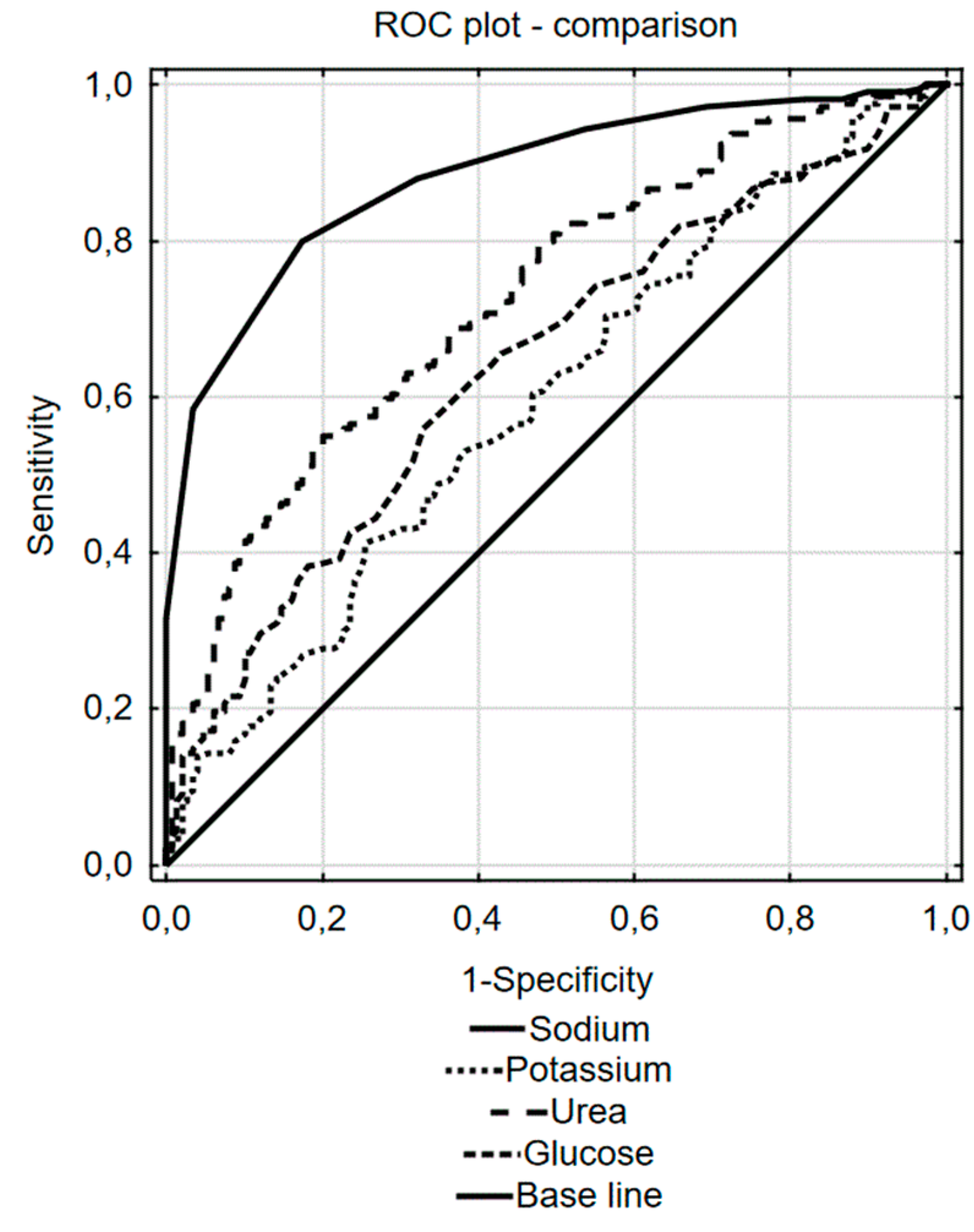
3.2. Predictive Discrimination of Underhydration with Serum Sodium, Urea, Glucose, and Potassium
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | under the curve area |
| ADL | activity of daily living |
| AMTS | Abbreviated Mental Test Score |
| CFS | Clinical Frailty Scale |
| CI | confidence interval |
| ESPEN | European Society for Clinical Nutrition and Metabolism |
| GDS | Geriatric Depression Scale |
| GFR | glomerular filtration rate |
| HOOP | Hydration and outcome in older patients admitted to hospital |
| IADL | instrumental activities of daily living |
| IQR | interquartile range |
| M | mean |
| Me | median |
| MNA-SF | Mini Nutritional Assessment- Short Form |
| n | number of cases |
| OARS | Older Americans Resources and Services |
| ROC | receiver-operating characteristic |
| SD | standard deviation |
References
- Begg, D.P. Disturbances of thirst and fluid balance associated with aging. Physiol. Behav. 2017, 178, 28–34. [Google Scholar] [CrossRef]
- Phillips, P.A.; Rolls, B.J.; Ledingham, J.G.; Forsling, M.L.; Morton, J.J.; Crowe, M.J.; Wollner, L. Reduced thirst after water deprivation in healthy elderly men. N. Engl. J. Med. 1984, 311, 753–759. [Google Scholar] [CrossRef]
- Luft, F.C.; Grim, C.E.; Fineberg, N.; Weinberger, M.C. Effects of volume expansion and contraction in normotensive whites, blacks, and subjects of different ages. Circulation 1979, 59, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E. Dehydration, hypernatremia, and hyponatremia. Clin. Geriatr. Med. 2015, 31, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Kavouras, S.A. Hydration, dehydration, underhydration, optimal hydration: Are we barking up the wrong tree? Eur. J. Nutr. 2019, 58, 471–473. [Google Scholar] [CrossRef] [PubMed]
- Warren, J.L.; Bacon, W.E.; Harris, T.; McBean, A.M.; Foley, D.J.; Phillips, C. The burden and outcomes associated with dehydration among US elderly, 1991. Am. J. Public Health 1994, 84, 1265–1269. [Google Scholar] [CrossRef]
- Kettaneh, A.; Fardet, L.; Mario, N.; Retbi, A.; Taright, N.; Tiev, K.; Reinhard, I.; Guidet, B.; Cabane, J. The 2003 heat wave in France: Hydratation status changes in older inpatients. Eur. J. Epidemiol. 2010, 25, 517–524. [Google Scholar] [CrossRef]
- El-Sharkawy, A.M.; Watson, P.; Neal, K.R.; Ljungqvist, O.; Maughan, R.J.; Sahota, O.; Lobo, D.N. Hydration and outcome in older patients admitted to hospital (the HOOP prospective cohort study). Age Ageing 2015, 44, 943–947. [Google Scholar] [CrossRef]
- Shimizu, M.; Kinoshita, K.; Hattori, K.; Ota, Y.; Kanai, T.; Kobayashi, H.; Tokuda, Y. Physical signs of dehydration in the elderly. Intern. Med. 2012, 51, 1207–1210. [Google Scholar] [CrossRef]
- Bunn, D.K.; Hooper, L. Signs and symptoms of low-intake dehydration do not work in older care home residents-DRIE diagnostic accuracy study. J. Am. Med. Dir. Assoc. 2019, 20, 963–970. [Google Scholar] [CrossRef]
- Thomas, D.R.; Tariq, S.H.; Makhdomm, S.; Haddad, R.; Moinuddin, A. Physician misdiagnosis of dehydration in older adults. J. Am. Med. Dir. Assoc. 2004, 5 (Suppl. 2), S30–S34. [Google Scholar] [CrossRef]
- Bennett, J.A.; Thomas, V.; Riegel, B. Unrecognized chronic dehydration in older adults: Examining prevalence rate and risk factors. J. Gerontol. Nurs. 2004, 30, 22–28. [Google Scholar] [CrossRef]
- Himmelstein, D.U.; Jones, A.A.; Woolhandler, S. Hypernatremic dehydration in nursing home patients: An indicator of neglect. J. Am. Geriatr. Soc. 1983, 31, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Breen, T.; Brueske, B.; Sidhu, M.S.; Murphree, D.H.; Kashani, K.B.; Barsness, G.W.; Jentzer, J.C. Abnormal serum sodium is associated with increased mortality among unselected cardiac intensive care unit patients. J. Am. Heart Assoc. 2020, 9, e014140. [Google Scholar] [CrossRef] [PubMed]
- Darmon, M.; Timsit, J.F.; Francais, A.; Nguile-Makao, M.; Adrie, C.; Cohen, Y.; Garrouste-Orgeas, M.; Goldgran-Toledano, D.; Dumenil, A.S.; Jamali, S.; et al. Association between hypernatraemia acquired in the ICU and mortality: A cohort study. Nephrol. Dial. Transpl. 2010, 25, 2510–2515. [Google Scholar] [CrossRef] [PubMed]
- Phillips, P.A.; Bretherton, M.; Johnston, C.I.; Gray, L. Reduced osmotic thirst in healthy elderly men. Am. J. Physiol. 1991, 261, R166–R171. [Google Scholar] [CrossRef]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef]
- Jimoh, O.F.; Brown, T.J.; Bunn, D.; Hooper, L. Beverage intake and drinking patterns-clues to support older people living in long-term care to drink well: DRIE and FISE studies. Nutrients 2019, 11, 447. [Google Scholar] [CrossRef]
- Wojszel, Z.B.; Magnuszewski, L.; Swietek, M.; Kasiukiewicz, A.; Swietek, M.L.; Zubrewicz, K. Frailty syndrome and functional correlates of atrial fibrillation in patients admitted to the geriatric ward. Gerontol. Pol. 2019, 27, 11–15. [Google Scholar]
- Wojszel, Z.B.; Kasiukiewicz, A.; Magnuszewski, L. Health and functional determinants of orthostatic hypotension in geriatric ward patients: A retrospective cross sectional cohort study. J. Nutr. Health Aging 2019, 23, 509–517. [Google Scholar] [CrossRef]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The barthel index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Fillenbaum, G.G.; Smyer, M.A. The development, validity, and reliability of the OARS multidimensional functional assessment questionnaire. J. Gerontol. 1981, 36, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Hodkinson, H.M. Evaluation of a mental test score for assessment of mental impairment in the elderly. Age Ageing 1972, 1, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982, 17, 37–49. [Google Scholar] [CrossRef]
- Kaiser, M.J.; Bauer, J.M.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.; Charlton, K.E.; Maggio, M.; et al. Validation of the mini nutritional assessment short-form (MNA-SF): A practical tool for identification of nutritional status. J. Nutr. Health Aging 2009, 13, 782–788. [Google Scholar] [CrossRef]
- Rockwood, K.; Song, X.; MacKnight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005, 173, 489–495. [Google Scholar] [CrossRef]
- Khajuria, A.; Krahn, J. Osmolality revisited-deriving and validating the best formula for calculated osmolality. Clin. Biochem. 2005, 38, 514–519. [Google Scholar] [CrossRef]
- Hooper, L.; Abdelhamid, A.; Ali, A.; Bunn, D.K.; Jennings, A.; John, W.G.; Kerry, S.; Lindner, G.; Pfortmueller, C.A.; Sjostrand, F.; et al. Diagnostic accuracy of calculated serum osmolarity to predict dehydration in older people: Adding value to pathology laboratory reports. BMJ Open 2015, 5, e008846. [Google Scholar] [CrossRef]
- Lavizzo-Mourey, R.; Johnson, J.; Stolley, P. Risk factors for dehydration among elderly nursing home residents. J. Am. Geriatr. Soc. 1988, 36, 213–218. [Google Scholar] [CrossRef]
- Wakefield, B.J.; Mentes, J.; Holman, J.E.; Culp, K. Risk factors and outcomes associated with hospital admission for dehydration. Rehabil. Nurs. 2008, 33, 233–241. [Google Scholar] [CrossRef]
- Hooper, L.; Abdelhamid, A.; Attreed, N.J.; Campbell, W.W.; Channell, A.M.; Chassagne, P.; Culp, K.R.; Fletcher, S.J.; Fortes, M.B.; Fuller, N.; et al. Clinical symptoms, signs and tests for identification of impending and current water-loss dehydration in older people. Cochrane Database Syst. Rev. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
| All n = 358 (100%) | Underhydrated n = 209 (58.4%) | Euhydrated n = 149 (41.6%) | p value1 | |
|---|---|---|---|---|
| Age, years, Me (IQR) | 82 (78–86) | 82 (78–86) | 82 (77–86) | 0.33 |
| Gender | ||||
| Male, n (%) | 84 (23.5) | 48 (23.0) | 36 (24.2) | 0.80 |
| Female, n (%) | 274 (76.5) | 161 (77.0) | 113 (75.8) | |
| Living in long term care, n (%) | 11 (3.1) | 7 (3.3) | 4 (2.7) | 1.0 |
| Living alone, n (%) | 105 (30.3), n = 347 | 64 (31.7), n = 202 | 41 (28.3), n = 145 | 0.55 |
| Hospitalization in the last year, n (%) | 98 (27.6), n = 355 | 55 (26.6), n = 207 | 43 (29.1), n = 148 | 0.63 |
| Number of chronic diseases, Me (IQR) | 5.0 (3.0–6.0) | 5.0 (4.0–6.0) | 4.0 (3.0–6.0) | 0.001 |
| Multimorbidity, n (%) | 210 (58.7) | 134 (64.1) | 76 (51.0) | 0.02 |
| Number of medications, Me (IQR) | 7.0 (5.0–9.0), n = 350 | 7.0 (5.0–10.0), n = 206 | 7.0 (4.0–9.0), n = 144 | 0.01 |
| Polypharmacy, n (%) | 275 (78.6) | 172 (83.5) | 103 (71.5) | 0.008 |
| Barthel Index, Me (IQR) | 90 (70–100), n = 356 | 90 (70–100), n = 208 | 90 (65–100), n = 148 | 0.48 |
| IADL, Me (IQR) | 7.0 (2.0–11.0), n = 352 | 7.0 (2.0–11.0), n = 206 | 7.0 (1.75–11.0), n = 146 | 0.55 |
| AMTS, Me (IQR) | 8.0 (6.0–9.0), n = 332 | 8.0 (6.0–9.0), n = 196 | 8.0 (6.0–9.0), n = 136 | 0.88 |
| GDS, Me (IQR) | 6.5 (3.0–10.0), n = 320 | 6.0 (3.0–10.0), n = 188 | 7.0 (4.0–10.0), n = 132 | 0.32 |
| MNA-SF, Me (IQR) | 11.0 (9.0–13.0), n = 349 | 12.0 (9.0–13.0), n = 203 | 11.0 (9.0–13.0), n = 146 | 0.26 |
| CFS, Me (IQR) | 5.0 (4.0–6.0) | 5.0 (4.0–6.0) | 5.0 (4.0–5.5) | 0.69 |
| Osmolarity, mMol/ L, Me (IQR) | 292.9 (288.4–296.0) | 295.4 (293.6–298.4) | 287.7 (283.8–289.6) | <0.001 |
| Serum sodium, mmol/L, Me (IQR) | 140.0 (138.0–141.0) | 141.0 (140.0–142.0) | 138.0 (136.0–139.0) | <0.001 |
| Na < 135 mmol/ L | 24 (6.7) | 4 (1.9) | 20 (13.4) | <0.001 |
| Serum potassium, mmol/L, M (SD) | 4.43 (0.46) | 4.50 (0.46) | 4.33 (0.44) | 0.001 |
| Serum urea, mmol/ L, Me (IQR) | 6.9 (5.5–8.8) | 7.8 (6.2–9.9) | 5.8 (4.9–7.3) | <0.001 |
| Serum glucose, mmol/L, Me (IQR) | 5.5 (5.0–6.3) | 5.6 (5.2–6.7) | 5.3 (4.9–5.8) | <0.001 |
| Serum creatinine, mmol/L, Me (IQR) | 86.2 (74.3–107.2), n = 354 | 91.05 (77.8–115.8), n = 207 | 78.7 (69.8–96.4), n = 147 | <0.001 |
| eGFR, ml/min/1.73m2, M (SD) | 57.8 (17.1), n = 354 | 58.2 (16.8), n = 207 | 58.4 (16.7), n = 147 | 0.91 |
| ROC Index | Sodium | Urea | Glucose | Potassium |
|---|---|---|---|---|
| AUC | 0.88 | 0.730 | 0.64 | 0.60 |
| 95% CI of AUC | 0.85–0.92 | 0.68–0.78 | 0.59–0.70 | 0.54–0.65 |
| p value | < 0.0001 | < 0.0001 | < 0.0001 | 0.002 |
| Youden Index J | 0.625 | 0.349 | 0.231 | 0.156 |
| Cut-off criterion | 140 mmol/L | 7.52 | 5.61 | 4.57 |
| Sensitivity (%) | 0.80 | 0.75 | 0.56 | 0.411 |
| 95% CI of sensitivity | 0.74–0.86 | 0.68–0.80 | 0.48–0.62 | 0.34–0.48 |
| Specificity | 0.83 | 0.72 | 0.67 | 0.745 |
| 95% CI of specificity | 0.75–0.88 | 0.65–0.79 | 0.59–0.75 | 0.67–0.81 |
| Positive likelihood ratio | 4.58 | 2.73 | 1.70 | 1.61 |
| Negative likelihood ratio | 0.24 | 0.35 | 0.66 | 0.79 |
| Difference between areas | ||||
| Sodium | 0.15 (0.10–0.21) | 0.24 (0.18–0.30) | 0.29 (0.23–0.35) | |
| p value | < 0.001 | < 0.001 | < 0.001 | |
| Urea | 0.09 (0.01–0.16) | 0.14 (-0.06–0.21) | ||
| p value | 0.02 | < 0.001 | ||
| Glucose | 0.05 (-0.03–0.13) | |||
| p value | 0.25 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wojszel, Z.B. What Serum Sodium Concentration Is Suggestive for Underhydration in Geriatric Patients? Nutrients 2020, 12, 496. https://doi.org/10.3390/nu12020496
Wojszel ZB. What Serum Sodium Concentration Is Suggestive for Underhydration in Geriatric Patients? Nutrients. 2020; 12(2):496. https://doi.org/10.3390/nu12020496
Chicago/Turabian StyleWojszel, Zyta Beata. 2020. "What Serum Sodium Concentration Is Suggestive for Underhydration in Geriatric Patients?" Nutrients 12, no. 2: 496. https://doi.org/10.3390/nu12020496
APA StyleWojszel, Z. B. (2020). What Serum Sodium Concentration Is Suggestive for Underhydration in Geriatric Patients? Nutrients, 12(2), 496. https://doi.org/10.3390/nu12020496





