Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review
Abstract
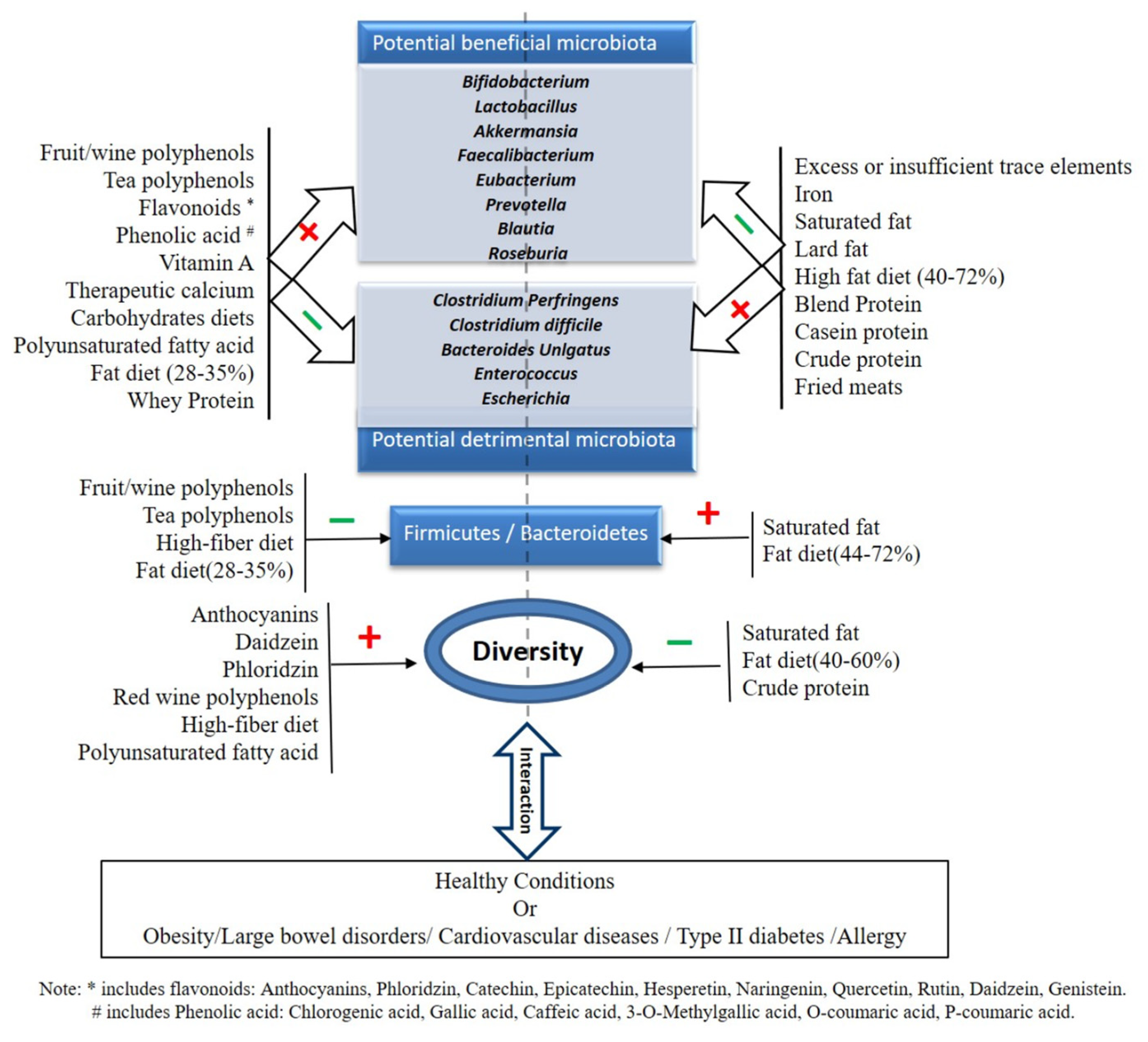
1. Introduction
2. Role of Dietary Micronutrients in Modulating Gut Microbiota
2.1. Polyphenols
2.2. Vitamins
2.3. Minerals and Trace Elements
3. The role of Dietary Macronutrients in Modulating Gut Microbiota
3.1. Carbohydrates
3.2. Fat
3.3. Protein
4. Summary and Future Perspective
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schmidt, T.S.; Raes, J.; Bork, P. The human gut microbiome: From association to modulation. Cell 2018, 172, 1198–1215. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.A.; Verdi, S.; Maxan, M.-E.; Shin, C.M.; Zierer, J.; Bowyer, R.C.; Martin, T.; Williams, F.M.; Menni, C.; Bell, J.T. Gut microbiota associations with common Dis. and prescription medications in a population-based cohort. Nat. Commun. 2018, 9, 2655. [Google Scholar] [CrossRef] [PubMed]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the gut microbiota in Nutr. and health. Br. Med J. 2018, 361, k2179. [Google Scholar] [CrossRef] [PubMed]
- Berg, R.D. The indigenous gastrointestinal microflora. Trends Microbiol. 1996, 4, 430–435. [Google Scholar] [CrossRef]
- Savage, D.C. Microbial ecology of the gastrointestinal tract. Annu. Rev. Microbiol. 1977, 31, 107–133. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 2016, 164, 337–340. [Google Scholar] [CrossRef]
- Rosner, J. Ten times more microbial cells than body cells in humans. Microbe 2014, 9, 47. [Google Scholar] [CrossRef]
- McFall-Ngai, M.; Hadfield, M.G.; Bosch, T.C.; Carey, H.V.; Domazet-Lošo, T.; Douglas, A.E.; Dubilier, N.; Eberl, G.; Fukami, T.; Gilbert, S.F. Animals in a bacterial world, a new imperative for the life Sci.s. Proc. Natl. Acad. Sci. 2013, 110, 3229–3236. [Google Scholar] [CrossRef]
- Abreu, C.; Lopez, A.O.; Gore, J. Pairing off: A bottom-up approach to the human gut microbiome. Mol. Syst. Biol. 2018, 14, e8425. [Google Scholar] [CrossRef]
- Consortium, H.M.J.R.S. A catalog of reference genomes from the human microbiome. Science 2010, 328, 994–999. [Google Scholar]
- Methé, B.A.; Nelson, K.E.; Pop, M.; Creasy, H.H.; Giglio, M.G.; Huttenhower, C.; Gevers, D.; Petrosino, J.F.; Abubucker, S.; Badger, J.H. A framework for human microbiome research. Nature 2012, 486, 215. [Google Scholar]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Jia, H.; Cai, X.; Zhong, H.; Feng, Q.; Sunagawa, S.; Arumugam, M.; Kultima, J.R.; Prifti, E.; Nielsen, T. An integrated catalog of reference genes in the human gut microbiome. Nat. Biotechnol. 2014, 32, 834. [Google Scholar] [CrossRef] [PubMed]
- Odamaki, T.; Kato, K.; Sugahara, H.; Hashikura, N.; Takahashi, S.; Xiao, J.Z.; Abe, F.; Osawa, R. Age-related changes in gut microbiota composition from newborn to centenarian: A cross-sectional study. BMC Microbiol. 2016, 16, 90. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, E.D.; Smits, S.A.; Tikhonov, M.; Higginbottom, S.K.; Wingreen, N.S.; Sonnenburg, J.L. Diet-induced extinctions in the gut microbiota compound over generations. Nature 2016, 529, 212. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Prince, A.L.; Bader, D.; Hu, M.; Ganu, R.; Baquero, K.; Blundell, P.; Harris, R.A.; Frias, A.E.; Grove, K.L. High-fat maternal diet during pregnancy persistently alters the offspring microbiome in a primate model. Nat. Commun. 2014, 5, 3889. [Google Scholar] [CrossRef]
- Paul, H.A.; Bomhof, M.R.; Vogel, H.J.; Reimer, R.A. Diet-induced changes in maternal gut microbiota and metabolomic profiles influence programming of offspring obesity risk in rats. Sci. Rep. 2016, 6, 20683. [Google Scholar] [CrossRef]
- Biasucci, G.; Benenati, B.; Morelli, L.; Bessi, E.; Boehm, G. Cesarean delivery may affect the early biodiversity of intestinal bacteria. J. Nutr. 2008, 138, 1796S–1800S. [Google Scholar] [CrossRef]
- Neu, J.; Rushing, J. Cesarean versus vaginal delivery: Long-term infant outcomes and the hygiene hypothesis. Clin. Perinatol. 2011, 38, 321–331. [Google Scholar] [CrossRef]
- Friedman, N.J.; Zeiger, R.S. The role of breast-feeding in the development of allergies and asthma. J. Allergy Clin. Immunol. 2005, 115, 1238–1248. [Google Scholar] [CrossRef]
- Harmsen, H.J.; Wildeboer–Veloo, A.C.; Raangs, G.C.; Wagendorp, A.A.; Klijn, N.; Bindels, J.G.; Welling, G.W. Analysis of intestinal flora development in breast-fed and formula-fed infants by using Mol. identification and detection methods. J. Pediatric Gastroenterol. Nutr. 2000, 30, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Aronoff, D.M. The influence of non-steroidal anti-inflammatory drugs on the gut microbiome. Clin. Microbiol. Infect. 2016, 22, e171–e178. [Google Scholar] [CrossRef] [PubMed]
- Jernberg, C.; Löfmark, S.; Edlund, C.; Jansson, J.K. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiol. 2010, 156, 3216–3223. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.A.; Worobey, M. Geographical variation of human gut microbial composition. Biol. Lett. 2014, 10, 20131037. [Google Scholar] [CrossRef] [PubMed]
- Heiman, M.L.; Greenway, F.L. A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 2016, 5, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Zhernakova, A.; Kurilshikov, A.; Bonder, M.J.; Tigchelaar, E.F.; Schirmer, M.; Vatanen, T.; Mujagic, Z.; Vila, A.V.; Falony, G.; Vieira-Silva, S. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 2016, 352, 565–569. [Google Scholar] [CrossRef]
- Zhang, N.; Ju, Z.; Zuo, T. Time for food: The impact of diet on gut microbiota and human health. Nutrition 2018, 51–52, 80–85. [Google Scholar] [CrossRef]
- Hiippala, K.; Jouhten, H.; Ronkainen, A.; Hartikainen, A.; Kainulainen, V.; Jalanka, J.; Satokari, R. The potential of gut commensals in reinforcing intestinal barrier Funct. and alleviating inflammation. Nutrition 2018, 10, 988. [Google Scholar]
- Cancello, R.; Turroni, S.; Rampelli, S.; Cattaldo, S.; Candela, M.; Cattani, L.; Mai, S.; Vietti, R.; Scacchi, M.; Brigidi, P. Effect of short-term dietary intervention and probiotic mix supplementation on the gut microbiota of elderly obese women. Nutrition 2019, 11, 3011. [Google Scholar] [CrossRef]
- O’Callaghan, A.A.; Corr, S.C. Establishing boundaries: The relationship that exists between intestinal epithelial cells and gut-dwelling bacteria. Microorg. 2019, 7, 663. [Google Scholar] [CrossRef]
- Gagniere, J.; Raisch, J.; Veziant, J.; Barnich, N.; Bonnet, R.; Buc, E.; Bringer, M.A.; Pezet, D.; Bonnet, M. Gut microbiota imbalance and colorectal cancer. World J. Gastroenterol. 2016, 22, 501–518. [Google Scholar] [CrossRef] [PubMed]
- Kowalska-Duplaga, K.; Gosiewski, T.; Kapusta, P.; Sroka-Oleksiak, A.; Wedrychowicz, A.; Pieczarkowski, S.; Ludwig-Slomczynska, A.H.; Wolkow, P.P.; Fyderek, K. Differences in the intestinal microbiome of healthy children and patients with newly diagnosed crohn’s disease. Sci. Rep. 2019, 9, 18880. [Google Scholar] [CrossRef] [PubMed]
- Alhinai, E.A.; Walton, G.E.; Commane, D.M. The role of the gut microbiota in colorectal cancer causation. Int. J. Mol. Sci. 2019, 20, 5295. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Nat. Acad. Sci. 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022. [Google Scholar] [CrossRef]
- Williamson, G. The role of polyphenols in modern. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Puupponen-Pimiä, R.; Aura, A.-M.; Oksman-Caldentey, K.-M.; Myllärinen, P.; Saarela, M.; Mattila-Sandholm, T.; Poutanen, K. Development of Functional ingredients for gut health. Trends Food Sci.Technol. 2002, 13, 3–11. [Google Scholar] [CrossRef]
- Han, X.; Shen, T.; Lou, H. Dietary polyphenols and their biological significance. Int. J. Mol. Sci.s 2007, 8, 950–988. [Google Scholar] [CrossRef]
- Selma, M.V.; Espin, J.C.; Tomas-Barberan, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef]
- Etxeberria, U.; Arias, N.; Boqué, N.; Macarulla, M.; Portillo, M.; Martínez, J.; Milagro, F. Reshaping faecal gut microbiota composition by the intake of trans-resveratrol and quercetin in high-fat sucrose diet-fed rats. J. Nutr.Biochem. 2015, 26, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Taira, T.; Yamaguchi, S.; Takahashi, A.; Okazaki, Y.; Yamaguchi, A.; Sakaguchi, H.; Chiji, H. Dietary polyphenols increase fecal mucin and immunoglobulin a and ameliorate the disturbance in gut microbiota caused by a high fat diet. J. Clin. Biochem.Nutr. 2015, 57, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zeng, B.; Liu, Z.; Liao, Z.; Zhong, Q.; Gu, L.; Wei, H.; Fang, X. Green tea polyphenols modulate colonic microbiota diversity and lipid Metab. in high-fat diet treated hfa mice. J. Food Sci. 2018, 83, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.-B.; Jeong, H.W.; Cho, D.; Lee, B.J.; Lee, J.H.; Choi, J.Y.; Bae, I.-H.; Lee, S.-J. Fermented green tea extract alleviates obesity and related complications and alters gut microbiota composition in diet-induced obese mice. J. Med. Food 2015, 18, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Santino, A.; Scarano, A.; De Santis, S.; De Benedictis, M.; Giovinazzo, G.; Chieppa, M. Gut microbiota modulation and anti-inflammatory properties of dietary polyphenols in ibd: New and consolidated perspectives. Curr. Pharm. Des. 2017, 23, 2344–2351. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-L.; Cesario, T.; Wang, Y.; Shanbrom, E.; Thrupp, L. Antibacterial activity of vegetables and juices. Nutrition 2003, 19, 994–996. [Google Scholar] [CrossRef]
- Tabasco, R.; Sánchez-Patán, F.; Monagas, M.; Bartolomé, B.; Moreno-Arribas, M.V.; Peláez, C.; Requena, T. Effect of grape polyphenols on lactic acid bacteria and bifidobacteria growth: Resistance and Metab. Food Microbiol. 2011, 28, 1345–1352. [Google Scholar] [CrossRef]
- Tzounis, X.; Vulevic, J.; Kuhnle, G.G.; George, T.; Leonczak, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2008, 99, 782–792. [Google Scholar] [CrossRef]
- Parkar, S.G.; Trower, T.M.; Stevenson, D.E. Fecal microbial Metab. of polyphenols and its effects on human gut microbiota. Anaerobe 2013, 23, 12–19. [Google Scholar] [CrossRef]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic Funct.: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev. 2001, 81, 1031–1064. [Google Scholar] [CrossRef]
- Firrman, J.; Liu, L.; Argoty, G.A.; Zhang, L.; Tomasula, P.; Wang, M.; Pontious, S.; Kobori, M.; Xiao, W. Analysis of temporal changes in growth and gene expression for commensal gut microbes in response to the polyphenol naringenin. Microbiol. Insights 2018, 11, 1178636118775100. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Collins, B.; Hoffman, J.; Martinez, K.; Grace, M.; Lila, M.A.; Cockrell, C.; Nadimpalli, A.; Chang, E.; Chuang, C.-C.; Zhong, W. A polyphenol-rich fraction obtained from table grapes decreases adiposity, insulin resistance and markers of inflammation and impacts gut microbiota in high-fat-fed mice. J. Nutr.Biochem. 2016, 31, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; Van Hée, V.F.; Bindels, L.B.; De Backer, F.; Cani, P.D.; Delzenne, N.M. Polyphenol-rich extract of pomegranate peel alleviates tissue inflammation and hypercholesterolaemia in high-fat diet-induced obese mice: Potential implication of the gut microbiota. Br. J. Nutr. 2013, 109, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Dolara, P.; Luceri, C.; De Filippo, C.; Femia, A.P.; Giovannelli, L.; Caderni, G.; Cecchini, C.; Silvi, S.; Orpianesi, C.; Cresci, A. Red wine polyphenols influence carcinogenesis, intestinal microflora, oxidative damage and gene expression profiles of colonic mucosa in f344 rats. Mutat. Res./Fundam. Mol. Mech. Mutagenesis 2005, 591, 237–246. [Google Scholar] [CrossRef]
- Axling, U.; Olsson, C.; Xu, J.; Fernandez, C.; Larsson, S.; Ström, K.; Ahrné, S.; Holm, C.; Molin, G.; Berger, K. Green tea powder and lactobacillus plantarum affect gut microbiota, lipid Metab. and inflammation in high-fat fed c57bl/6j mice. Nutr. Metab. 2012, 9, 105. [Google Scholar]
- Marques, C.; Fernandes, I.; Meireles, M.; Faria, A.; Spencer, J.P.; Mateus, N.; Calhau, C. Gut microbiota modulation accounts for the neuroprotective properties of anthocyanins. Sci. Rep. 2018, 8, 11341. [Google Scholar] [CrossRef]
- Mei, X.; Zhang, X.; Wang, Z.; Gao, Z.; Liu, G.; Hu, H.; Zou, L.; Li, X. Insulin sensitivity-enhancing activity of phlorizin is associated with lipopolysaccharide decrease and gut microbiota changes in obese and type 2 diabetes (db/db) mice. J. Agric. Food Chem. 2016, 64, 7502–7511. [Google Scholar] [CrossRef]
- Chacar, S.; Itani, T.; Hajal, J.; Saliba, Y.; Louka, N.; Faivre, J.F.; Maroun, R.; Fares, N. The impact of long-term intake of phenolic compounds-rich grape pomace on rat gut microbiota. J. Food Sci. 2018, 83, 246–251. [Google Scholar] [CrossRef]
- Viveros, A.; Chamorro, S.; Pizarro, M.; Arija, I.; Centeno, C.; Brenes, A. Effects of dietary polyphenol-rich grape products on intestinal microflora and gut morphology in broiler chicks. Poult. Sci. 2011, 90, 566–578. [Google Scholar] [CrossRef]
- Ohno, M.; Nishida, A.; Sugitani, Y.; Nishino, K.; Inatomi, O.; Sugimoto, M.; Kawahara, M.; Andoh, A. Nanoparticle curcumin ameliorates experimental colitis via modulation of gut microbiota and induction of regulatory t cells. PLoS ONE 2017, 12, e0185999. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.; Royston, K.J.; Li, Y.; Stoll, M.L.; Skibola, C.F.; Wilson, L.S.; Barnes, S.; Morrow, C.D.; Tollefsbol, T.O. Impact of genistein on the gut microbiome of humanized mice and its role in breast tumor inhibition. PLoS ONE 2017, 12, e0189756. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. The Am. J. Clin. Nutr. 2010, 93, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Sánchez-Alcoholado, L.; Pérez-Martínez, P.; Andrés-Lacueva, C.; Cardona, F.; Tinahones, F.; Queipo-Ortuño, M.I. Red wine polyphenols modulate fecal microbiota and reduce markers of the metabolic syndrome in obese patients. Food Funct. 2016, 7, 1775–1787. [Google Scholar]
- Queipo-Ortuño, M.I.; Boto-Ordóñez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Cardona Diaz, F.; Andres-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and Biochem.ical biomarkers. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Yang, J.; Shao, P.; Lee, R.-P.; Huang, J.; Ly, A.; Hsu, M.; Lu, Q.-Y.; Thames, G.; Heber, D. Health benefit of vegetable/fruit juice-based diet: Role of microbiome. Sci. Rep. 2017, 7, 2167. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.S.; Touyama, M.; Hisada, T.; Benno, Y. Effects of green tea consumption on human fecal microbiota with special reference to bifidobacterium species. Microbiol. Immunol. 2012, 56, 729–739. [Google Scholar] [CrossRef]
- Iino, C.; Shimoyama, T.; Iino, K.; Yokoyama, Y.; Chinda, D.; Sakuraba, H.; Fukuda, S.; Nakaji, S. Daidzein intake is associated with equol producing status through an increase in the intestinal bacteria responsible for equol production. Nutrition 2019, 11, 433. [Google Scholar] [CrossRef]
- Mayta-Apaza, A.C. Evaluation of the impact of tart cherries polyphenols on the human gut microbiota and phenolic metabolites in vitro and in vivo. Master’s Thesis, Food Sci., University of Arkansas, Fayetteville, AR, USA, 2017. [Google Scholar]
- Mayta-Apaza, A.C.; Pottgen, E.; De Bodt, J.; Papp, N.; Marasini, D.; Howard, L.; Abranko, L.; Van de Wiele, T.; Lee, S.-O.; Carbonero, F. Impact of tart cherries polyphenols on the human gut microbiota and phenolic metabolites in vitro and in vivo. J. Nutr.Biochem.. 2018, 59, 160–172. [Google Scholar] [CrossRef]
- Li, Z.; Henning, S.M.; Lee, R.-P.; Lu, Q.-Y.; Summanen, P.H.; Thames, G.; Corbett, K.; Downes, J.; Tseng, C.-H.; Finegold, S.M. Pomegranate extract induces ellagitannin metabolite formation and changes stool microbiota in healthy volunteers. Food Funct. 2015, 6, 2487–2495. [Google Scholar]
- Most, J.; Penders, J.; Lucchesi, M.; Goossens, G.; Blaak, E. Gut microbiota composition in relation to the metabolic response to 12-week combined polyphenol supplementation in overweight men and women. Eur. J. Clin. Nutr. 2017, 71, 1040. [Google Scholar] [CrossRef] [PubMed]
- Haro, C.; Rangel-Zuniga, O.A.; Alcala-Diaz, J.F.; Gomez-Delgado, F.; Perez-Martinez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Landa, B.B.; Navas-Cortes, J.A.; Tena-Sempere, M.; et al. Intestinal microbiota is influenced by gender and body mass index. PLoS ONE 2016, 11, e0154090. [Google Scholar] [CrossRef] [PubMed]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota Funct.s: Metab. of Nutr. and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ko, G. Antiviral effect of vitamin A on norovirus Infect. via modulation of the gut microbiome. Sci. Rep. 2016, 6, 25835. [Google Scholar] [CrossRef]
- Lee, H.; Ko, G. New perspectives regarding the antiviral effect of vitamin A on norovirus using modulation of gut microbiota. Gut Microbes 2017, 8, 616–620. [Google Scholar] [CrossRef]
- Long, K.Z.; García, C.; Santos, J.I.; Rosado, J.L.; Hertzmark, E.; DuPont, H.L.; Ko, G. Vitamin A supplementation has divergent effects on norovirus Infect.s and Clin. symptoms among mexican children. J. Infect. Dis. 2007, 196, 978–985. [Google Scholar] [CrossRef]
- Thornton, K.A.; Mora-Plazas, M.; Marín, C.; Villamor, E. Vitamin A deficiency is associated with gastrointestinal and respiratory morbidity in school-age children. J. Nutr. 2014, 144, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, X.; Xiong, X.-Q.; Yang, T.; Cui, T.; Hou, N.-L.; Lai, X.; Liu, S.; Guo, M.; Liang, X.-H.; et al. Effect of vitamin A supplementation on gut microbiota in children with autism spectrum disorders - a pilot study. BMC Microbiol. 2017, 17, 204. [Google Scholar] [CrossRef]
- Lv, Z.; Wang, Y.; Yang, T.; Zhan, X.; Li, Z.; Hu, H.; Li, T.; Chen, J. Vitamin A deficiency impacts the structural segregation of gut microbiota in children with persistent diarrhea. J. Clin. Biochem. Nutr. 2016, 59, 113–121. [Google Scholar] [CrossRef]
- Huda, M.N.; Ahmad, S.M.; Kalanetra, K.M.; Taft, D.H.; Alam, M.J.; Khanam, A.; Raqib, R.; Underwood, M.A.; Mills, D.A.; Stephensen, C.B. Neonatal vitamin a supplementation and vitamin A status are associated with gut microbiome composition in bangladeshi infants in early infancy and at 2 years of age. J. Nutr. 2019, 149, 1075–1088. [Google Scholar] [CrossRef]
- Sperandio, V. Take your pick: Vitamins and microbiota facilitate pathogen clearance. Cell Host Microbe 2017, 21, 130–131. [Google Scholar]
- Degnan, P.H.; Barry, N.A.; Mok, K.C.; Taga, M.E.; Goodman, A.L. Human gut microbes use multiple transporters to distinguish vitamin B12 analogs and compete in the gut. Cell Host Microbe 2014, 15, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.J.; Clark, D.E.; Adli, M.; Kendall, M.M. Ethanolamine signaling promotes salmonella niche recognition and adaptation during Infect. Plos Pathog. 2015, 11, e1005278. [Google Scholar]
- Thiennimitr, P.; Winter, S.E.; Winter, M.G.; Xavier, M.N.; Tolstikov, V.; Huseby, D.L.; Sterzenbach, T.; Tsolis, R.M.; Roth, J.R.; Bäumler, A.J. Intestinal inflammation allows salmonella to use ethanolamine to compete with the microbiota. Proc. Natl. Acad. Sci. 2011, 108, 17480–17485. [Google Scholar] [CrossRef] [PubMed]
- Magnúsdóttir, S.; Ravcheev, D.; de Crécy-Lagard, V.; Thiele, I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 2015, 6, 148. [Google Scholar] [CrossRef] [PubMed]
- Harbige, L.S. Nutrition and immunity with emphasis on Infect. and autoimmune disease. Nutr. Health 1996, 10, 285–312. [Google Scholar] [CrossRef]
- Miki, T.; Goto, R.; Fujimoto, M.; Okada, N.; Hardt, W.D. The bactericidal lectin regiiibeta prolongs gut colonization and enteropathy in the streptomycin mouse model for salmonella diarrhea. Cell Host Microbe 2017, 21, 195–207. [Google Scholar]
- Subramanian, V.S.; Srinivasan, P.; Wildman, A.J.; Marchant, J.S.; Said, H.M. Mol. mechanism (s) involved in differential expression of vitamin c transporters along the intestinal tract. Am. J. Physiol. -Gastrointest. Liver Physiol. 2016, 312, G340–G347. [Google Scholar] [CrossRef]
- Li, L.; Krause, L.; Somerset, S. Associations between micronutrient intakes and gut microbiota in a group of adults with cystic fibrosis. Clin. Nutr. 2017, 36, 1097–1104. [Google Scholar] [CrossRef]
- Wilson, R.; Willis, J.; Gearry, R.; Hughes, A.; Lawley, B.; Skidmore, P.; Frampton, C.; Fleming, E.; Anderson, A.; Jones, L. Sungold kiwifruit supplementation of individuals with prediabetes alters gut microbiota and improves vitamin c status, anthropometric and Clinical markers. Nutrients 2018, 10, 895. [Google Scholar] [CrossRef]
- Xu, J.; Xu, C.; Chen, X.; Cai, X.; Yang, S.; Sheng, Y.; Wang, T. Regulation of an antioxidant blend on intestinal redox status and major microbiota in early weaned piglets. Nutrition 2014, 30, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and chronic Dis. Aging Dis. 2017, 8, 346. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Qureshi, O.S.; Gardner, D.; Hou, T.Z.; Briggs, Z.; Soskic, B.; Baker, J.; Raza, K.; Sansom, D.M. Vitamin D antagonises the suppressive effect of inflammatory cytokines on ctla-4 expression and regulatory Funct. PLoS ONE 2015, 10, e0131539. [Google Scholar] [CrossRef] [PubMed]
- Kesby, J.P.; Eyles, D.W.; Burne, T.H.; McGrath, J.J. The effects of vitamin D on brain development and adult brain Funct. Mol. Cell. Endocrinol. 2011, 347, 121–127. [Google Scholar] [CrossRef]
- Bertone-Johnson, E.R.; Powers, S.I.; Spangler, L.; Brunner, R.L.; Michael, Y.L.; Larson, J.C.; Millen, A.E.; Bueche, M.N.; Salmoirago-Blotcher, E.; Liu, S. Vitamin D intake from foods and supplements and depressive symptoms in a diverse population of older women. Am. J. of Clin. Nutr. 2011, 94, 1104–1112. [Google Scholar] [CrossRef]
- Ciubotaru, I.; Green, S.J.; Kukreja, S.; Barengolts, E. Significant differences in fecal microbiota are associated with various stages of glucose tolerance in african Am. male veterans. Transl. Res. 2015, 166, 401–411. [Google Scholar] [CrossRef]
- Kanhere, M.; He, J.; Hanfelt, J.; Gewirtz, A.T.; Chassaing, B.; Ivie, E.A.; Alvarez, J.A.; Hao, L.; Ziegler, T.R.; Tangpricha, V. Bolus weekly vitamin D3 supplementation impacts gut and airway microbiota in adults with cystic fibrosis: A double-blind, randomized, placebo-controlled Clin. trial. J. Clin. Endocrinol. Metab. 2017, 103, 564–574. [Google Scholar]
- Jacobs, C.; Hutton, B.; Ng, T.; Shorr, R.; Clemons, M. Is there a role for oral or intravenous ascorbate (vitamin C) in treating patients with cancer? A systematic review. Oncol. 2015, 20, 210–223. [Google Scholar] [CrossRef]
- Iniesta, R.R.; Rush, R.; Paciarotti, I.; Rhatigan, E.; Brougham, F.; McKenzie, J.; Wilson, D. Systematic review and meta-analysis: Prevalence and possible causes of vitamin D deficiency and insufficiency in pediatric cancer patients. Clin. Nutr. 2016, 35, 95–108. [Google Scholar] [CrossRef]
- Meeker, S.; Seamons, A.; Maggio-Price, L.; Paik, J. Protective links between vitamin D, inflammatory bowel disease and colon cancer. World J. Gastroenterol. 2016, 22, 933. [Google Scholar] [CrossRef]
- Ardesia, M.; Ferlazzo, G.; Fries, W. Vitamin D and inflammatory bowel disease. BioMed Res. Int. 2015, 2015, 470805. [Google Scholar] [CrossRef] [PubMed]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Högenauer, C.; Pieber, T.R. Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur. J. of Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaeizadeh, S.-A.; Tafazoli, N.; Ferns, G.A.; Avan, A.; Ghayour-Mobarhan, M. Vitamin D, the gut microbiome and inflammatory bowel disease. J. of Res. in Medical Sci. Off. J. Isfahan Univ. Med Sci. 2018, 23, 75. [Google Scholar]
- Sun, J. Dietary vitamin D, vitamin D receptor, and microbiome. Curr. Opinion in Clin. Nutr. Metab. Care 2018, 21, 471–474. [Google Scholar] [CrossRef]
- Clark, A.; Mach, N. Role of vitamin d in the hygiene hypothesis: The interplay between vitamin D, vitamin D receptors, gut microbiota, and immune response. Front. Immunol. 2016, 7, 627. [Google Scholar] [CrossRef]
- Garg, M.; Hendy, P.; Ding, J.N.; Shaw, S.; Hold, G.; Hart, A. The effect of vitamin D on intestinal inflammation and faecal microbiota in patients with ulcerative colitis. J. Crohn’s Colitis 2018, 12, 963–972. [Google Scholar] [CrossRef]
- Schäffler, H.; Herlemann, D.P.; Klinitzke, P.; Berlin, P.; Kreikemeyer, B.; Jaster, R.; Lamprecht, G. Vitamin D administration leads to a shift of the intestinal bacterial composition in crohn’s disease patients, but not in healthy controls. J. Dig. Dis. 2018, 19, 225–234. [Google Scholar] [CrossRef]
- Talsness, C.E.; Penders, J.; Jansen, E.H.J.M.; Damoiseaux, J.; Thijs, C.; Mommers, M. Influence of vitamin D on key bacterial taxa in infant microbiota in the koala birth cohort study. PLoS ONE 2017, 12, e0188011. [Google Scholar] [CrossRef]
- Luthold, R.V.; Fernandes, G.R.; Franco-de-Moraes, A.C.; Folchetti, L.G.; Ferreira, S.R.G. Gut microbiota interactions with the immunomodulatory role of vitamin D in normal individuals. Metabolism 2017, 69, 76–86. [Google Scholar] [CrossRef]
- Naderpoor, N.; Mousa, A.; Fernanda Gomez Arango, L.; Barrett, H.L.; Dekker Nitert, M.; de Courten, B. Effect of vitamin D supplementation on faecal microbiota: A randomised Clin. trial. Nutrition 2019, 11, 2888. [Google Scholar] [CrossRef]
- Zuo, K.; Li, J.; Xu, Q.; Hu, C.; Gao, Y.; Chen, M.; Hu, R.; Liu, Y.; Chi, H.; Yin, Q. Dysbiotic gut microbes may contribute to hypertension by limiting vitamin D production. Clin. Cardiol. 2019, 42, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Wang, H.; Zhang, X.; Li, X.; Yu, J. Ascorbic acid ameliorates oxidative stress and inflammation in dextran sulfate sodium-induced ulcerative colitis in mice. Int. J. Clin. Exp. Med. 2015, 8, 20245. [Google Scholar] [PubMed]
- Tahan, G.; Aytac, E.; Aytekin, H.; Gunduz, F.; Dogusoy, G.; Aydin, S.; Tahan, V.; Uzun, H. Vitamin e has a dual effect of anti-inflammatory and antioxidant activities in acetic acid–induced ulcerative colitis in rats. Can. J. Surg. 2011, 54, 333. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Wintergerst, E.S.; Beveridge, S.; Hornig, D.H. Selected vitamins and trace elements support immune Funct. by strengthening epithelial barriers and cellular and humoral immune responses. Br. J. Nutr. 2007, 98, S29–S35. [Google Scholar] [CrossRef]
- Pierre, J.F.; Hinterleitner, R.; Bouziat, R.; Hubert, N.A.; Leone, V.; Miyoshi, J.; Jabri, B.; Chang, E.B. Dietary antioxidant microNutr. alter mucosal inflammatory risk in a murine model of genetic and microbial susceptibility. J. Nutr. Biochem. 2018, 54, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Lee, S.; Kim, S.; Lee, J.; Ha, J.; Oh, H.; Lee, Y.; Kim, Y.; Yoon, Y. Vitamin e (α-tocopherol) consumption influences gut microbiota composition. Int. J. Food Sci.s Nutr. 2019, 1–5. [Google Scholar] [CrossRef]
- Mandal, S.; Godfrey, K.M.; McDonald, D.; Treuren, W.V.; Bjørnholt, J.V.; Midtvedt, T.; Moen, B.; Rudi, K.; Knight, R.; Brantsæter, A.L. Fat and vitamin intakes during pregnancy have stronger relations with a pro-inflammatory maternal microbiota than does carbohydrate intake. Microbiome 2016, 4, 55. [Google Scholar] [CrossRef]
- Tang, M.; Frank, D.N.; Sherlock, L.; Ir, D.; Robertson, C.E.; Krebs, N.F. Effect of vitamin e with therapeutic iron supplementation on iron repletion and gut microbiome in us iron deficient infants and toddlers. J. Pediatric Gastroenterol. Nutr. 2016, 63, 379–385. [Google Scholar]
- Skrypnik, K.; Suliburska, J. Association between the gut microbiota and mineral Metab. J. Sci. Food Agric. 2018, 98, 2449–2460. [Google Scholar] [CrossRef]
- Gîlcă-Blanariu, G.-E.; Diaconescu, S.; Ciocoiu, M.; Ștefănescu, G. New insights into the role of trace elements in IBD. BioMed. Res. Int. 2018, 2018, 1813047. [Google Scholar] [CrossRef]
- Zhang, F.; Ye, J.; Zhu, X.; Wang, L.; Gao, P.; Shu, G.; Jiang, Q.; Wang, S. Anti-obesity effects of dietary calcium: The evidence and possible mechanisms. Int. J. Mol. Sci.s 2019, 20, 3072. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.; Costa, J.; Alfenas, R. Could the beneficial effects of dietary calcium on obesity and diabetes control be mediated by changes in intestinal microbiota and integrity? Br. J. Nutr. 2015, 114, 1756–1765. [Google Scholar] [CrossRef] [PubMed]
- Trautvetter, U.; Camarinha-Silva, A.; Jahreis, G.; Lorkowski, S.; Glei, M. High phosphorus intake and gut-related parameters–results of a randomized placebo-controlled human intervention study. Nutr. J. 2018, 17, 23. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.N.; Bassis, C.M.; Zhang, L.; Zaidi, S.; Varani, J.; Bergin, I.L. Calcium reduces liver injury in mice on a high-fat diet: Alterations in microbial and bile acid profiles. PLoS ONE 2016, 11, e0166178. [Google Scholar] [CrossRef]
- Li, P.; Tang, T.; Chang, X.; Fan, X.; Chen, X.; Wang, R.; Fan, C.; Qi, K. Abnormality in maternal dietary calcium intake during pregnancy and lactation promotes body weight gain by affecting the gut microbiota in mouse offspring. Mol. Nutr. Food Res. 2018, 63, 1800399. [Google Scholar]
- Chaplin, A.; Parra, P.; Laraichi, S.; Serra, F.; Palou, A. Calcium supplementation modulates gut microbiota in a prebiotic manner in dietary obese mice. Mol. Nutr. Food Res. 2016, 60, 468–480. [Google Scholar]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in prevention and therapy. Nutrition 2015, 7, 8199–8226. [Google Scholar] [CrossRef]
- Pachikian, B.D.; Neyrinck, A.M.; Deldicque, L.; De Backer, F.C.; Catry, E.; Dewulf, E.M.; Sohet, F.M.; Bindels, L.B.; Everard, A.; Francaux, M. Changes in intestinal bifidobacteria levels are associated with the inflammatory response in magnesium-deficient mice. J. Nutr. 2010, 140, 509–514. [Google Scholar] [CrossRef]
- Jørgensen, B.P.; Winther, G.; Kihl, P.; Nielsen, D.S.; Wegener, G.; Hansen, A.K.; Sørensen, D.B. Dietary magnesium deficiency affects gut microbiota and anxiety-like behaviour in c57bl/6n mice. Acta Neuropsychiatr. 2015, 27, 307–311. [Google Scholar] [CrossRef]
- Winther, G.; Jørgensen, B.M.P.; Elfving, B.; Nielsen, D.S.; Kihl, P.; Lund, S.; Sørensen, D.B.; Wegener, G. Dietary magnesium deficiency alters gut microbiota and leads to depressive-like behaviour. Acta Neuropsychiatr. 2015, 27, 168–176. [Google Scholar] [CrossRef]
- Crowley, E.; Long-Smith, C.; Murphy, A.; Patterson, E.; Murphy, K.; O’Gorman, D.; Stanton, C.; Nolan, Y. Dietary supplementation with a magnesium-rich marine mineral blend enhances the diversity of gastrointestinal microbiota. Mar. Drugs 2018, 16, 216. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Excess iron intake as a factor in growth, Infect.s, and development of infants and young children. Am. J. Clin. Nutr. 2017, 106, 1681S–1687S. [Google Scholar] [CrossRef] [PubMed]
- Jaeggi, T.; Kortman, G.A.; Moretti, D.; Chassard, C.; Holding, P.; Dostal, A.; Boekhorst, J.; Timmerman, H.M.; Swinkels, D.W.; Tjalsma, H. Iron fortification adversely affects the gut microbiome, increases pathogen abundance and induces intestinal inflammation in kenyan infants. Gut 2015, 64, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Frank, D.N.; Hendricks, A.E.; Ir, D.; Esamai, F.; Liechty, E.; Hambidge, K.M.; Krebs, N.F. Iron in micronutrient powder promotes an unfavorable gut microbiota in kenyan infants. Nutrition 2017, 9, 776. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Chassard, C.; Rohner, F.; N’Goran, E.K.; Nindjin, C.; Dostal, A.; Utzinger, J.; Ghattas, H.; Lacroix, C.; Hurrell, R.F. The effects of iron fortification on the gut microbiota in african children: A randomized controlled trial in cote d’ivoire. Am. J. Clin. Nutr. 2010, 92, 1406–1415. [Google Scholar] [CrossRef]
- Qasem, W.; Azad, M.B.; Hossain, Z.; Azad, E.; Jorgensen, S.; San Juan, S.C.; Cai, C.; Khafipour, E.; Beta, T.; Roberts, L.J. Assessment of complementary feeding of canadian infants: Effects on microbiome oxidative stress, a randomized controlled trial. BMC Pediatrics 2017, 17, 54. [Google Scholar]
- Dostal, A.; Baumgartner, J.; Riesen, N.; Chassard, C.; Smuts, C.M.; Zimmermann, M.B.; Lacroix, C. Effects of iron supplementation on dominant bacterial groups in the gut, faecal scfa and gut inflammation: A randomised, placebo-controlled intervention trial in south african children. Br. J. Nutr. 2014, 112, 547–556. [Google Scholar] [CrossRef]
- Sjödin, K.S.; Domellöf, M.; Lagerqvist, C.; Hernell, O.; Lönnerdal, B.; Szymlek-Gay, E.A.; Sjödin, A.; West, C.E.; Lind, T. Administration of ferrous sulfate drops has significant effects on the gut microbiota of iron-sufficient infants: A randomised controlled study. Gut 2019, 68, 2095–2097. [Google Scholar]
- Fang, S.; Zhuo, Z.; Yu, X.; Wang, H.; Feng, J. Oral administration of liquid iron preparation containing excess iron induces intestine and liver injury, impairs intestinal barrier Funct. and alters the gut microbiota in rats. J. of Trace Elem. Med. Biol. 2018, 47, 12–20. [Google Scholar] [CrossRef]
- Kortman, G.A.; Dutilh, B.E.; Maathuis, A.J.; Engelke, U.F.; Boekhorst, J.; Keegan, K.P.; Nielsen, F.G.; Betley, J.; Weir, J.C.; Kingsbury, Z. Microbial Metab. shifts towards an adverse profile with supplementary iron in the tim-2 in vitro model of the human colon. Front. Microbiol. 2016, 6, 1481. [Google Scholar] [CrossRef]
- Vazquez-Gutierrez, P.; Lacroix, C.; Jaeggi, T.; Zeder, C.; Zimmerman, M.B.; Chassard, C. Bifidobacteria strains isolated from stools of iron deficient infants can efficiently sequester iron. BMC Microbiol. 2015, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Nitert, M.D.; Gomez-Arango, L.F.; Barrett, H.L.; McIntyre, H.D.; Anderson, G.J.; Frazer, D.M.; Callaway, L.K. Iron supplementation has minor effects on gut microbiota composition in overweight and obese women in early pregnancy. Br. J. Nutr. 2018, 120, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Seura, T.; Yoshino, Y.; Fukuwatari, T. The relationship between habitual dietary intake and gut microbiota in young japanese women. J. Nutr. Sci. Vitaminol. 2017, 63, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Constante, M.; Fragoso, G.; Lupien-Meilleur, J.; Calvé, A.; Santos, M.M. Iron supplements modulate colon microbiota composition and potentiate the protective effects of probiotics in dextran sodium sulfate-induced colitis. Inflamm. Bowel Dis. 2017, 23, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Ijssennagger, N.; Belzer, C.; Hooiveld, G.J.; Dekker, J.; van Mil, S.W.; Müller, M.; Kleerebezem, M.; van der Meer, R. Gut microbiota facilitates dietary heme-induced epithelial hyperproliferation by opening the mucus barrier in colon. Proc. Natl. Acad. Sci.s 2015, 112, 10038–10043. [Google Scholar] [CrossRef]
- IJssennagger, N.; Derrien, M.; van Doorn, G.M.; Rijnierse, A.; van den Bogert, B.; Müller, M.; Dekker, J.; Kleerebezem, M.; van der Meer, R. Dietary heme alters microbiota and mucosa of mouse colon without Functional changes in host-microbe cross-talk. PLoS ONE 2012, 7, e49868. [Google Scholar] [CrossRef]
- Martin, O.C.; Olier, M.; Ellero-Simatos, S.; Naud, N.; Dupuy, J.; Huc, L.; Taché, S.; Graillot, V.; Levêque, M.; Bézirard, V. Haem iron reshapes colonic luminal environment: Impact on mucosal homeostasis and microbiome through aldehyde formation. Microbiome 2019, 7, 72. [Google Scholar] [CrossRef]
- Lee, T.; Clavel, T.; Smirnov, K.; Schmidt, A.; Lagkouvardos, I.; Walker, A.; Lucio, M.; Michalke, B.; Schmitt-Kopplin, P.; Fedorak, R.; et al. Oral versus intravenous iron replacement therapy distinctly alters the gut microbiota and metabolome in patients with ibd. Gut 2017, 66, 863–871. [Google Scholar] [CrossRef]
- Capra, S. Nutrient Reference Values for Australia and New Zealand: Including Recommended Dietary Intakes. Available online: https://www.nhmrc.gov.au/about-us/publications/nutrient-reference-values-australia-and-new-zealand-including-recommended-dietary-intakes (accessed on 21 January 2020).
- Borda-Molina, D.; Vital, M.; Sommerfeld, V.; Rodehutscord, M.; Camarinha-Silva, A. Insights into broilers’ gut microbiota fed with phosphorus, calcium, and phytase supplemented diets. Front.Microbiol. 2016, 7, 2033. [Google Scholar] [CrossRef]
- Ohashi, W.; Fukada, T. Contribution of zinc and zinc transporters in the pathogenesis of inflammatory bowel Dis. J. Immunol. Res. 2019, 2019, 8396878. [Google Scholar] [CrossRef]
- Reed, S.; Neuman, H.; Moscovich, S.; Glahn, R.; Koren, O.; Tako, E. Chronic zinc deficiency alters chick gut microbiota composition and Funct. Nutrition 2015, 7, 9768–9784. [Google Scholar]
- Shao, Y.; Lei, Z.; Yuan, J.; Yang, Y.; Guo, Y.; Zhang, B. Effect of zinc on growth performance, gut morphometry, and cecal microbial community in broilers challenged with salmonella enterica serovar typhimurium. J. Microbiol. 2014, 52, 1002–1011. [Google Scholar] [CrossRef] [PubMed]
- Zackular, J.P.; Moore, J.L.; Jordan, A.T.; Juttukonda, L.J.; Noto, M.J.; Nicholson, M.R.; Crews, J.D.; Semler, M.W.; Zhang, Y.; Ware, L.B. Dietary zinc alters the microbiota and decreases resistance to clostridium difficile Infect. Nat. Med. 2016, 22, 1330. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium intake, status, and health: A complex relationship. Horm. 2019, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kasaikina, M.V.; Kravtsova, M.A.; Lee, B.C.; Seravalli, J.; Peterson, D.A.; Walter, J.; Legge, R.; Benson, A.K.; Hatfield, D.L.; Gladyshev, V.N. Dietary selenium affects host selenoproteome expression by influencing the gut microbiota. FASEB J. 2011, 25, 2492–2499. [Google Scholar] [CrossRef]
- Zhai, Q.; Cen, S.; Li, P.; Tian, F.; Zhao, J.; Zhang, H.; Chen, W. Effects of dietary selenium supplementation on intestinal barrier and immune responses associated with its modulation of gut microbiota. Environ. Sci. Technol. Lett. 2018, 5, 724–730. [Google Scholar] [CrossRef]
- Shen, H.; Han, J.; Li, Y.; Lu, C.; Zhou, J.; Li, Y.; Su, X. Different host-specific responses in thyroid Funct. and gut microbiota modulation between diet-induced obese and normal mice given the same dose of iodine. Appl. Microbiol. BioTechnol. 2019, 103, 3537–3547. [Google Scholar] [CrossRef]
- Schroeder, B.O.; Birchenough, G.M.; Ståhlman, M.; Arike, L.; Johansson, M.E.; Hansson, G.C.; Bäckhed, F. Bifidobacteria or fiber protects against diet-induced microbiota-mediated colonic mucus deterioration. Cell Host Microbe 2018, 23, 27–40. [Google Scholar]
- Prajapati, B.; Rajput, P.; Kumar Jena, P.; Seshadri, S. Investigation of chitosan for prevention of diabetic progression through gut microbiota alteration in sugar rich diet induced diabetic rats. Curr. Pharm. BioTechnol. 2016, 17, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Avila-Nava, A.; Noriega, L.G.; Tovar, A.R.; Granados, O.; Perez-Cruz, C.; Pedraza-Chaverri, J.; Torres, N. Food combination based on a pre-hispanic mexican diet decreases metabolic and cognitive abnormalities and gut microbiota dysbiosis caused by a sucrose-enriched high-fat diet in rats. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Martens, E.C. Microbiome: Fibre for the future. Nature 2016, 529, 158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Benus, R.F.; van der Werf, T.S.; Welling, G.W.; Judd, P.A.; Taylor, M.A.; Harmsen, H.J.; Whelan, K. Association between faecalibacterium prausnitzii and dietary fibre in colonic fermentation in healthy human subjects. Br. J. Nutr. 2010, 104, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Carvalho-Wells, A.L.; Helmolz, K.; Nodet, C.; Molzer, C.; Leonard, C.; McKevith, B.; Thielecke, F.; Jackson, K.G.; Tuohy, K.M. Determination of the in vivo prebiotic potential of a maize-based whole grain breakfast cereal: A human feeding study. Br. J. Nutr. 2010, 104, 1353–1356. [Google Scholar] [CrossRef] [PubMed]
- García-Peris, P.; Velasco, C.; Lozano, M.; Moreno, Y.; Paron, L.; De la Cuerda, C.; Bretón, I.; Camblor, M.; García-Hernández, J.; Guarner, F. Effect of a mixture of inulin and fructo-oligosaccharide on lactobacillus and bifidobacterium intestinal microbiota of patients receiving radiotherapy; a randomised, double-blind, placebo-controlled trial. Nutr. Hosp. 2012, 27, 1908–1915. [Google Scholar]
- Dao, M.C.; Everard, A.; Aron-Wisnewsky, J.; Sokolovska, N.; Prifti, E.; Verger, E.O.; Kayser, B.D.; Levenez, F.; Chilloux, J.; Hoyles, L. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: Relationship with gut microbiome richness and ecology. Gut 2016, 65, 426–436. [Google Scholar] [CrossRef]
- Candela, M.; Biagi, E.; Soverini, M.; Consolandi, C.; Quercia, S.; Severgnini, M.; Peano, C.; Turroni, S.; Rampelli, S.; Pozzilli, P. Modulation of gut microbiota dysbioses in type 2 diabetic patients by macrobiotic ma-pi 2 diet. Br. J. Nutr. 2016, 116, 80–93. [Google Scholar] [CrossRef]
- Holscher, H.D.; Caporaso, J.G.; Hooda, S.; Brulc, J.M.; Fahey Jr, G.C.; Swanson, K.S. Fiber supplementation influences phylogenetic structure and Functional capacity of the human intestinal microbiome: Follow-up of a randomized controlled trial. Am. J. Clin. Nutr. 2014, 101, 55–64. [Google Scholar] [CrossRef]
- Tap, J.; Furet, J.P.; Bensaada, M.; Philippe, C.; Roth, H.; Rabot, S.; Lakhdari, O.; Lombard, V.; Henrissat, B.; Corthier, G. Gut microbiota richness promotes its stability upon increased dietary fibre intake in healthy adults. Environ. Microbiol. 2015, 17, 4954–4964. [Google Scholar] [CrossRef]
- Kovatcheva-Datchary, P.; Nilsson, A.; Akrami, R.; Lee, Y.S.; De Vadder, F.; Arora, T.; Hallen, A.; Martens, E.; Björck, I.; Bäckhed, F. Dietary fiber-induced improvement in glucose Metab. is associated with increased abundance of prevotella. Cell Metab. 2015, 22, 971–982. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from europe and rural africa. Proc. Natl. Acad. Sci.s 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Haro, C.; Montes-Borrego, M.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Tinahones, F.J.; Landa, B.B. Two healthy diets modulate gut microbial community improving insulin sensitivity in a human obese population. J. Clin. Endocrinol. 2016, 101, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Hald, S.; Schioldan, A.G.; Moore, M.E.; Dige, A.; Lærke, H.N.; Agnholt, J.; Knudsen, K.E.B.; Hermansen, K.; Marco, M.L.; Gregersen, S. Effects of arabinoxylan and resistant starch on intestinal microbiota and short-chain fatty acids in subjects with metabolic syndrome: A randomised crossover study. PLoS ONE 2016, 11, e0159223. [Google Scholar] [CrossRef] [PubMed]
- Cloetens, L.; Broekaert, W.F.; Delaedt, Y.; Ollevier, F.; Courtin, C.M.; Delcour, J.A.; Rutgeerts, P.; Verbeke, K. Tolerance of arabinoxylan-oligosaccharides and their prebiotic activity in healthy subjects: A randomised, placebo-controlled cross-over study. Br. J. Nutr. 2010, 103, 703–713. [Google Scholar] [CrossRef] [PubMed]
- François, I.E.; Lescroart, O.; Veraverbeke, W.S.; Marzorati, M.; Possemiers, S.; Evenepoel, P.; Hamer, H.; Houben, E.; Windey, K.; Welling, G.W. Effects of a wheat bran extract containing arabinoxylan oligosaccharides on gastrointestinal health parameters in healthy adult human volunteers: A double-blind, randomised, placebo-controlled, cross-over trial. Br. J. Nutr. 2012, 108, 2229–2242. [Google Scholar] [CrossRef] [PubMed]
- Lecerf, J.-M.; Dépeint, F.; Clerc, E.; Dugenet, Y.; Niamba, C.N.; Rhazi, L.; Cayzeele, A.; Abdelnour, G.; Jaruga, A.; Younes, H. Xylo-oligosaccharide (xos) in combination with inulin modulates both the intestinal environment and immune status in healthy subjects, while xos alone only shows prebiotic properties. Br. J. Nutr. 2012, 108, 1847–1858. [Google Scholar] [CrossRef]
- Walton, G.E.; Lu, C.; Trogh, I.; Arnaut, F.; Gibson, G.R. A randomised, double-blind, placebo controlled cross-over study to determine the gastrointestinal effects of consumption of arabinoxylan-oligosaccharides enriched bread in healthy volunteers. Nutr. J. 2012, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Childs, C.E.; Röytiö, H.; Alhoniemi, E.; Fekete, A.A.; Forssten, S.D.; Hudjec, N.; Lim, Y.N.; Steger, C.J.; Yaqoob, P.; Tuohy, K.M. Xylo-oligosaccharides alone or in synbiotic combination with bifidobacterium animalis subsp. Lactis induce bifidogenesis and modulate markers of immune Funct. in healthy adults: A double-blind, placebo-controlled, randomised, factorial cross-over study. Br. J. Nutr. 2014, 111, 1945–1956. [Google Scholar] [CrossRef]
- Finegold, S.M.; Li, Z.; Summanen, P.H.; Downes, J.; Thames, G.; Corbett, K.; Dowd, S.; Krak, M.; Heber, D. Xylooligosaccharide increases bifidobacteria but not lactobacilli in human gut microbiota. Food Funct. 2014, 5, 436–445. [Google Scholar] [CrossRef]
- François, I.E.; Lescroart, O.; Veraverbeke, W.S.; Marzorati, M.; Possemiers, S.; Hamer, H.; Windey, K.; Welling, G.W.; Delcour, J.A.; Courtin, C.M. Effects of wheat bran extract containing arabinoxylan oligosaccharides on gastrointestinal parameters in healthy preadolescent children. J. Pediatric Gastroenterol. Nutr. 2014, 58, 647–653. [Google Scholar] [CrossRef]
- Windey, K.; De Preter, V.; Huys, G.; Broekaert, W.F.; Delcour, J.A.; Louat, T.; Herman, J.; Verbeke, K. Wheat bran extract alters colonic fermentation and microbial composition, but does not affect faecal water toxicity: A randomised controlled trial in healthy subjects. Br. J. Nutr. 2015, 113, 225–238. [Google Scholar] [CrossRef]
- Broekaert, W.F.; Courtin, C.M.; Verbeke, K.; Van de Wiele, T.; Verstraete, W.; Delcour, J.A. Prebiotic and other health-related effects of cereal-derived arabinoxylans, arabinoxylan-oligosaccharides, and xylooligosaccharides. Crit. Rev.Food Sci. Nutr. 2011, 51, 178–194. [Google Scholar] [CrossRef] [PubMed]
- Giovannini, M.; Verduci, E.; Gregori, D.; Ballali, S.; Soldi, S.; Ghisleni, D.; Riva, E.; Group, P.T.S. Prebiotic effect of an infant formula supplemented with galacto-oligosaccharides: Randomized multicenter trial. J. Am. Coll. Nutr. 2014, 33, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Ladirat, S.E.; Schuren, F.H.; Schoterman, M.H.; Nauta, A.; Gruppen, H.; Schols, H.A. Impact of galacto-oligosaccharides on the gut microbiota composition and metabolic activity upon antibiotic treatment during in vitro fermentation. FEMS Microbiol. Ecol. 2014, 87, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Monteagudo-Mera, A.; Arthur, J.; Jobin, C.; Keku, T.; Bruno-Barcena, J.; Azcarate-Peril, M. High purity galacto-oligosaccharides enhance specific bifidobacterium species and their metabolic activity in the mouse gut microbiome. Benef. Microbes 2016, 7, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.; Martinez, I.; Walter, J.; Hutkins, R. A dose dependent impact of prebiotic galactooligosaccharides on the intestinal microbiota of healthy adults. Int. J. Food Microbiol. 2010, 144, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Davis, L.M.; Martínez, I.; Walter, J.; Goin, C.; Hutkins, R.W. Barcoded pyrosequencing reveals that consumption of galactooligosaccharides results in a highly specific bifidogenic response in humans. PLoS ONE 2011, 6, e25200. [Google Scholar] [CrossRef] [PubMed]
- Walton, G.E.; van den Heuvel, E.G.; Kosters, M.H.; Rastall, R.A.; Tuohy, K.M.; Gibson, G.R. A randomised crossover study investigating the effects of galacto-oligosaccharides on the faecal microbiota in men and women over 50 years of age. Br. J. Nutr. 2012, 107, 1466–1475. [Google Scholar] [CrossRef]
- Musilova, S.; Rada, V.; Marounek, M.; Nevoral, J.; Dušková, D.; Bunesova, V.; Vlkova, E.; Zelenka, R. Prebiotic effects of a novel combination of galactooligosaccharides and maltodextrins. J. Med. Food 2015, 18, 685–689. [Google Scholar] [CrossRef]
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P.; Tzortzis, G.; Toward, R.E.; Gibson, G.R. Influence of galacto-oligosaccharide mixture (b-gos) on gut microbiota, immune parameters and metabonomics in elderly persons. Br. J. Nutr. 2015, 114, 586–595. [Google Scholar] [CrossRef]
- Azcarate-Peril, M.A.; Ritter, A.J.; Savaiano, D.; Monteagudo-Mera, A.; Anderson, C.; Magness, S.T.; Klaenhammer, T.R. Impact of short-chain galactooligosaccharides on the gut microbiome of lactose-intolerant individuals. Proc. Natl. Acad. Sci.s 2017, 114, E367–E375. [Google Scholar] [CrossRef]
- Whisner, C.M.; Martin, B.R.; Schoterman, M.H.; Nakatsu, C.H.; McCabe, L.D.; McCabe, G.P.; Wastney, M.E.; Van Den Heuvel, E.G.; Weaver, C.M. Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: A double-blind cross-over trial. Br. J. Nutr. 2013, 110, 1292–1303. [Google Scholar] [CrossRef] [PubMed]
- Scalabrin, D.M.; Mitmesser, S.H.; Welling, G.W.; Harris, C.L.; Marunycz, J.D.; Walker, D.C.; Bos, N.A.; Tölkkö, S.; Salminen, S.; Vanderhoof, J.A. New prebiotic blend of polydextrose and galacto-oligosaccharides has a bifidogenic effect in young infants. J. Pediatric Gastroenterol. Nutr. 2012, 54, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Westerbeek, E.; Slump, R.; Lafeber, H.; Knol, J.; Georgi, G.; Fetter, W.; van Elburg, R. The effect of enteral supplementation of specific neutral and acidic oligosaccharides on the faecal microbiota and intestinal microenvironment in preterm infants. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Sierra, C.; Bernal, M.-J.; Blasco, J.; Martínez, R.; Dalmau, J.; Ortuno, I.; Espín, B.; Vasallo, M.-I.; Gil, D.; Vidal, M.-L. Prebiotic effect during the first year of life in healthy infants fed formula containing gos as the only prebiotic: A multicentre, randomised, double-blind and placebo-controlled trial. Eur. J. Nutr. 2015, 54, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Civardi, E.; Garofoli, F.; Longo, S.; Mongini, M.E.; Grenci, B.; Mazzucchelli, I.; Angelini, M.; Castellazzi, A.; Fasano, F.; Grinzato, A. Safety, growth, and support to healthy gut microbiota by an infant formula enriched with Functional compounds. Clin. Nutr. 2017, 36, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Paganini, D.; Uyoga, M.A.; Kortman, G.A.M.; Cercamondi, C.I.; Moretti, D.; Barth-Jaeggi, T.; Schwab, C.; Boekhorst, J.; Timmerman, H.M.; Lacroix, C.; et al. Prebiotic galacto-oligosaccharides mitigate the adverse effects of iron fortification on the gut microbiome: A randomised controlled study in kenyan infants. Gut 2017, 66, 1956–1967. [Google Scholar] [CrossRef]
- Francavilla, R.; Calasso, M.; Calace, L.; Siragusa, S.; Ndagijimana, M.; Vernocchi, P.; Brunetti, L.; Mancino, G.; Tedeschi, G.; Guerzoni, E. Effect of lactose on gut microbiota and metabolome of infants with cow’s milk allergy. Pediatric Allergy Immunol. 2012, 23, 420–427. [Google Scholar] [CrossRef]
- Fernando, W.; Hill, J.; Zello, G.; Tyler, R.; Dahl, W.; Van Kessel, A. Diets supplemented with chickpea or its main oligosaccharide component raffinose modify faecal microbial composition in healthy adults. Benef. Microbes 2010, 1, 197–207. [Google Scholar] [CrossRef]
- Licht, T.R.; Hansen, M.; Poulsen, M.; Dragsted, L.O. Dietary carbohydrate source influences Mol. fingerprints of the rat faecal microbiota. BMC Microbiol. 2006, 6, 98. [Google Scholar] [CrossRef]
- Catry, E.; Bindels, L.B.; Tailleux, A.; Lestavel, S.; Neyrinck, A.M.; Goossens, J.-F.; Lobysheva, I.; Plovier, H.; Essaghir, A.; Demoulin, J.-B. Targeting the gut microbiota with inulin-type fructans: PreClin. demonstration of a novel approach in the management of endothelial dysFunct. Gut 2018, 67, 271–283. [Google Scholar] [CrossRef]
- Ramirez-Farias, C.; Slezak, K.; Fuller, Z.; Duncan, A.; Holtrop, G.; Louis, P. Effect of inulin on the human gut microbiota: Stimulation of bifidobacterium adolescentis and faecalibacterium prausnitzii. Br. J. Nutr. 2008, 101, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P.; Fuentes, S.; Puylaert, P.G.; Neyrinck, A.M.; Bindels, L.B.; de Vos, W.M.; Gibson, G.R.; Thissen, J.-P. Insight into the prebiotic concept: Lessons from an exploratory, double blind intervention study with inulin-type fructans in obese women. Gut 2013, 62, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, D.; Falony, G.; Vieira-Silva, S.; Wang, J.; Sailer, M.; Theis, S.; Verbeke, K.; Raes, J. Prebiotic inulin-type fructans induce specific changes in the human gut microbiota. Gut 2017, 66, 1968–1974. [Google Scholar] [CrossRef] [PubMed]
- Drabińska, N.; Jarocka-Cyrta, E.; Markiewicz, L.H.; Krupa-Kozak, U. The effect of oligofructose-enriched inulin on faecal bacterial counts and microbiota-associated characteristics in celiac disease children following a gluten-free diet: Results of a randomized, placebo-controlled trial. Nutrition 2018, 10, 201. [Google Scholar] [CrossRef]
- Salazar, N.; Dewulf, E.M.; Neyrinck, A.M.; Bindels, L.B.; Cani, P.D.; Mahillon, J.; de Vos, W.M.; Thissen, J.-P.; Gueimonde, M.; Clara, G. Inulin-type fructans modulate intestinal bifidobacterium species populations and decrease fecal short-chain fatty acids in obese women. Clin. Nutr. 2015, 34, 501–507. [Google Scholar] [CrossRef]
- Nicolucci, A.C.; Hume, M.P.; Martinez, I.; Mayengbam, S.; Walter, J.; Reimer, R.A. Prebiotics reduce body fat and alter intestinal microbiota in children who are overweight or with obesity. Gastroenterol. 2017, 153, 711–722. [Google Scholar] [CrossRef]
- Yang, X.; Darko, K.O.; Huang, Y.; He, C.; Yang, H.; He, S.; Li, J.; Li, J.; Hocher, B.; Yin, Y. Resistant starch regulates gut microbiota: Structure, Biochem. and cell signalling. Cell. Physiol. Biochem. 2017, 42, 306–318. [Google Scholar] [CrossRef]
- Martínez, I.; Kim, J.; Duffy, P.R.; Schlegel, V.L.; Walter, J. Resistant starches types 2 and 4 have differential effects on the composition of the fecal microbiota in human subjects. PLoS ONE 2010, 5, e15046. [Google Scholar] [CrossRef]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011, 5, 220. [Google Scholar] [CrossRef]
- Le Leu, R.K.; Winter, J.M.; Christophersen, C.T.; Young, G.P.; Humphreys, K.J.; Hu, Y.; Gratz, S.W.; Miller, R.B.; Topping, D.L.; Bird, A.R. Butyrylated starch intake can prevent red meat-induced o 6-methyl-2-deoxyguanosine adducts in human rectal tissue: A randomised Clin. trial. Br. J. Nutr. 2015, 114, 220–230. [Google Scholar] [CrossRef]
- Cluny, N.L.; Eller, L.K.; Keenan, C.M.; Reimer, R.A.; Sharkey, K.A. Interactive effects of oligofructose and obesity predisposition on gut hormones and microbiota in diet-induced obese rats. Obes. 2015, 23, 769–778. [Google Scholar] [CrossRef]
- Wernimont, S.; Northington, R.; Kullen, M.J.; Yao, M.; Bettler, J. Effect of an α-lactalbumin-enriched infant formula supplemented with oligofructose on fecal microbiota, stool characteristics, and hydration status: A randomized, double-blind, controlled trial. Clin. Pediatrics 2015, 54, 359–370. [Google Scholar] [CrossRef]
- Raninen, K.; Lappi, J.; Mykkänen, H.; Poutanen, K. Dietary fiber type reflects Physiol. Functionality: Comparison of grain fiber, inulin, and polydextrose. Nutr. Rev. 2011, 69, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Costabile, A.; Fava, F.; Röytiö, H.; Forssten, S.D.; Olli, K.; Klievink, J.; Rowland, I.R.; Ouwehand, A.C.; Rastall, R.A.; Gibson, G.R. Impact of polydextrose on the faecal microbiota: A double-blind, crossover, placebo-controlled feeding study in healthy human subjects. Br. J. Nutr. 2012, 108, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Wolters, M.; Ahrens, J.; Perez, M.R.; Watkins, C.; Sanz, Y.; Benítez-Páez, A.; Stanton, C.; Günther, K. Dietary fat, the gut microbiota, and metabolic health–a systematic review conducted within the mynewgut project. Clin. Nutr. 2019, 38, 2504–2520. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.K.; Boudry, G.; Lemay, D.G.; Raybould, H.E. Changes in intestinal barrier Funct. and gut microbiota in high-fat diet-fed rats are dynamic and region dependent. Am. J. Physiol. -Gastrointest. Liver Physiol. 2015, 308, G840–G851. [Google Scholar] [CrossRef]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill–Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterol. 2009, 137, 1716–1724. [Google Scholar] [CrossRef]
- Chen, G.; Xie, M.; Wan, P.; Chen, D.; Dai, Z.; Ye, H.; Hu, B.; Zeng, X.; Liu, Z. Fuzhuan brick tea polysaccharides attenuate metabolic syndrome in high-fat diet induced mice in association with modulation in the gut microbiota. J. Agric. Food Chem. 2018, 66, 2783–2795. [Google Scholar] [CrossRef]
- Murphy, E.; Cotter, P.; Healy, S.; Marques, T.M.; O’sullivan, O.; Fouhy, F.; Clarke, S.; O’toole, P.; Quigley, E.M.; Stanton, C. Composition and energy harvesting capacity of the gut microbiota: Relationship to diet, obesity and time in mouse models. Gut 2010, 59, 1635–1642. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Ha, C.W.; Campbell, C.R.; Mitchell, A.J.; Dinudom, A.; Oscarsson, J.; Cook, D.I.; Hunt, N.H.; Caterson, I.D.; Holmes, A.J. Increased gut permeability and microbiota change associate with mesenteric fat inflammation and metabolic dysFunct. in diet-induced obese mice. PLoS ONE 2012, 7, e34233. [Google Scholar] [CrossRef]
- Kim, K.-A.; Gu, W.; Lee, I.-A.; Joh, E.-H.; Kim, D.-H. High fat diet-induced gut microbiota exacerbates inflammation and obesity in mice via the tlr4 signaling pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef] [PubMed]
- Cowan, T.E.; Palmnäs, M.S.; Yang, J.; Bomhof, M.R.; Ardell, K.L.; Reimer, R.A.; Vogel, H.J.; Shearer, J. Chronic coffee consumption in the diet-induced obese rat: Impact on gut microbiota and serum metabolomics. J. Nutr. Biochem. 2014, 25, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Mujico, J.R.; Baccan, G.C.; Gheorghe, A.; Díaz, L.E.; Marcos, A. Changes in gut microbiota due to supplemented fatty acids in diet-induced obese mice. Br. J. Nutr. 2013, 110, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet–induced obesity and diabetes in mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027. [Google Scholar] [CrossRef] [PubMed]
- Devkota, S.; Wang, Y.; Musch, M.W.; Leone, V.; Fehlner-Peach, H.; Nadimpalli, A.; Antonopoulos, D.A.; Jabri, B.; Chang, E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in il10−/− mice. Nature 2012, 487, 104. [Google Scholar] [CrossRef]
- Lecomte, V.; Kaakoush, N.O.; Maloney, C.A.; Raipuria, M.; Huinao, K.D.; Mitchell, H.M.; Morris, M.J. Changes in gut microbiota in rats fed a high fat diet correlate with obesity-associated metabolic parameters. PLoS ONE 2015, 10, e0126931. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, F.; Yuan, J.; Li, J.; Jiang, D.; Zhang, J.; Li, H.; Wang, R.; Tang, J.; Huang, T.; et al. Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: A 6-month randomised controlled-feeding trial. Gut 2019, 68, 1417–1429. [Google Scholar] [CrossRef]
- Haro, C.; Garcia-Carpintero, S.; Rangel-Zuniga, O.A.; Alcala-Diaz, J.F.; Landa, B.B.; Clemente, J.C.; Perez-Martinez, P.; Lopez-Miranda, J.; Perez-Jimenez, F.; Camargo, A. Consumption of two healthy dietary patterns restored microbiota dysbiosis in obese patients with metabolic dysFunct. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Lee, S.; Keirsey, K.I.; Kirkland, R.; Grunewald, Z.I.; Fischer, J.G.; de La Serre, C.B. Blueberry supplementation influences the gut microbiota, inflammation, and insulin resistance in high-fat-diet–fed rats. J. Nutr. 2018, 148, 209–219. [Google Scholar] [CrossRef]
- Qiao, Y.; Sun, J.; Ding, Y.; Le, G.; Shi, Y. Alterations of the gut microbiota in high-fat diet mice is strongly linked to oxidative stress. Appl. Microbiol. BioTechnol. 2013, 97, 1689–1697. [Google Scholar] [CrossRef] [PubMed]
- Santos-Marcos, J.A.; Haro, C.; Vega-Rojas, A.; Alcala-Diaz, J.F.; Molina-Abril, H.; Leon-Acuña, A.; Lopez-Moreno, J.; Landa, B.B.; Tena-Sempere, M.; Perez-Martinez, P. Sex differences in the gut microbiota as potential determinants of gender predisposition to disease. Mol. Nutr. Food Res. 2019, 63, 1800870. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; O’Doherty, R.M.; Murphy, E.F.; Wall, R.; O’Sullivan, O.; Nilaweera, K.; Fitzgerald, G.F.; Cotter, P.D.; Ross, R.P.; Stanton, C. Impact of dietary fatty acids on metabolic activity and host intestinal microbiota composition in c57bl/6j mice. Br. J. Nutr. 2014, 111, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- De Wit, N.; Derrien, M.; Bosch-Vermeulen, H.; Oosterink, E.; Keshtkar, S.; Duval, C.; de Vogel-van den Bosch, J.; Kleerebezem, M.; Müller, M.; van der Meer, R. Saturated fat stimulates obesity and hepatic steatosis and affects gut microbiota composition by an enhanced overflow of dietary fat to the distal intestine. Am. J. Physiol. -Gastrointest. Liver Physiol. 2012, 303, G589–G599. [Google Scholar] [CrossRef] [PubMed]
- Zentek, J.; Buchheit-Renko, S.; Männer, K.; Pieper, R.; Vahjen, W. Intestinal concentrations of free and encapsulated dietary medium-chain fatty acids and effects on gastric microbial ecology and bacterial metabolic products in the digestive tract of piglets. Arch. Anim. Nutr. 2012, 66, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Ojo, B.; El-Rassi, G.D.; Payton, M.E.; Perkins-Veazie, P.; Clarke, S.; Smith, B.J.; Lucas, E.A. Mango supplementation modulates gut microbial dysbiosis and short-chain fatty acid production independent of body weight reduction in c57bl/6 mice fed a high-fat diet–3. J. Nutr. 2016, 146, 1483–1491. [Google Scholar] [CrossRef]
- Younge, N.; Yang, Q.; Seed, P.C. Enteral high fat-polyunsaturated fatty acid blend alters the pathogen composition of the intestinal microbiome in premature infants with an enterostomy. J. Pediatrics 2017, 181, 93–101. [Google Scholar] [CrossRef]
- Sprong, R.; Schonewille, A.; Van der Meer, R. Dietary cheese whey protein protects rats against mild dextran sulfate sodium–induced colitis: Role of mucin and microbiota. J. Dairy Sci. 2010, 93, 1364–1371. [Google Scholar] [CrossRef]
- McAllan, L.; Skuse, P.; Cotter, P.D.; O’Connor, P.; Cryan, J.F.; Ross, R.P.; Fitzgerald, G.; Roche, H.M.; Nilaweera, K.N. Protein quality and the protein to carbohydrate ratio within a high fat diet influences energy balance and the gut microbiota in c57bl/6j mice. PLoS ONE 2014, 9, e88904. [Google Scholar] [CrossRef]
- Nakatani, A.; Li, X.; Miyamoto, J.; Igarashi, M.; Watanabe, H.; Sutou, A.; Watanabe, K.; Motoyama, T.; Tachibana, N.; Kohno, M. Dietary mung bean protein reduces high-fat diet-induced weight gain by modulating host bile acid Metab. in a gut microbiota-dependent manner. Biochem. Biophys. Res. Commun. 2018, 501, 955–961. [Google Scholar] [CrossRef]
- Rist, V.T.; Weiss, E.; Sauer, N.; Mosenthin, R.; Eklund, M. Effect of dietary protein supply originating from soybean meal or casein on the intestinal microbiota of piglets. Anaerobe 2014, 25, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Holm, J.B.; Rønnevik, A.; Tastesen, H.S.; Fjære, E.; Fauske, K.R.; Liisberg, U.; Madsen, L.; Kristiansen, K.; Liaset, B. Diet-induced obesity, energy Metab. and gut microbiota in c57bl/6j mice fed western diets based on lean seafood or lean meat mixtures. J. Nutr. Biochem. 2016, 31, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Kostovcikova, K.; Coufal, S.; Galanova, N.; Fajstova, A.; Hudcovic, T.; Kostovcik, M.; Prochazkova, P.; Zakostelska, Z.J.; Cermakova, M.; Sediva, B. Diet rich in animal protein promotes pro-inflammatory macrophage response and exacerbates colitis in mice. Front. Immunol. 2019, 10, 919. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Cerrudo, V.; Audebert, M.; Dumont, F.; Mancano, G.; Khodorova, N.; Andriamihaja, M. Quantity and source of dietary protein influence metabolite production by gut microbiota and rectal mucosa gene expression: A randomized, parallel, double-blind trial in overweight humans. Am. J. Clin. Nutr. 2017, 106, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Pérez, D.; Bressa, C.; Bailén, M.; Hamed-Bousdar, S.; Naclerio, F.; Carmona, M.; Pérez, M.; González-Soltero, R.; Montalvo-Lominchar, M.G.; Carabaña, C. Effect of a protein supplement on the gut microbiota of endurance athletes: A randomized, controlled, double-blind pilot study. Nutrition 2018, 10, 337. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Chen, Y.A.; Tuohy, K.M. A comparative in vitro investigation into the effects of cooked meats on the human faecal microbiota. Anaerobe 2010, 16, 572–577. [Google Scholar] [CrossRef]
- Harris, V.C.; Haak, B.W.; Boele van Hensbroek, M.; Wiersinga, W.J. The intestinal microbiome in infectious Dis.: The Clin. relevance of a rapidly emerging field. Open Forum Infect. Dis. 2017, 4, ofx144. [Google Scholar] [CrossRef]
- Harsch, I.; Konturek, P. The role of gut microbiota in obesity and type 2 and type 1 diabetes mellitus: New insights into “old” Dis. Med Sci.s 2018, 6, 32. [Google Scholar] [CrossRef]
- Koropatkin, N.M.; Cameron, E.A.; Martens, E.C. How glycan Metab. shapes the human gut microbiota. Nat. Rev. Microbiol. 2012, 10, 323. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559. [Google Scholar] [CrossRef]
- Witzig, M.; da Silva, A.C.; Green-Engert, R.; Hoelzle, K.; Zeller, E.; Seifert, J.; Hoelzle, L.E.; Rodehutscord, M. Spatial variation of the gut microbiota in broiler chickens as affected by dietary available phosphorus and assessed by t-rflp analysis and 454 pyrosequencing. PLoS ONE 2015, 10, e0143442. [Google Scholar]
- Hang, I.; Rinttila, T.; Zentek, J.; Kettunen, A.; Alaja, S.; Apajalahti, J.; Harmoinen, J.; de Vos, W.M.; Spillmann, T. Effect of high contents of dietary animal-derived protein or carbohydrates on canine faecal microbiota. BMC Vet. Res. 2012, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Lubbs, D.; Vester, B.; Fastinger, N.; Swanson, K. Dietary protein concentration affects intestinal microbiota of adult cats: A study using dgge and qpcr to evaluate differences in microbial populations in the feline gastrointestinal tract. J. Anim. Physiol. Anim. Nutr. 2009, 93, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Lletjós, S.; Andriamihaja, M.; Blais, A.; Grauso, M.; Lepage, P.; Davila, A.-M.; Viel, R.; Gaudichon, C.; Leclerc, M.; Blachier, F. Dietary protein intake level modulates mucosal healing and mucosa-adherent microbiota in mouse model of colitis. Nutrition 2019, 11, 514. [Google Scholar] [CrossRef] [PubMed]
| Dose and Treatment Duration/Test Model | F/B | Potentially Beneficial Microbiota | Potentially Detrimental Microbiota | Species | Diversity | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bifidobacterium sp. | Lactobacillus sp. | Akkermansia sp. | Fecalibacterium sp. | Eubacterium sp. | Bacteroides sp. | Prevotella sp. | Blautia sp. | Clostridium sp. | Ruminococcus sp. | Eggerthella sp. | Roseburia sp. | Coprococcus sp. | Ruminococcus sp. | Clostridium sp. | Bacteroides sp. | Enterobacter sp. | Enterococcus sp. | |||||
| Anthocyanins | ||||||||||||||||||||||
| Marques et al. (2018) | Standard diet with Blackberry anthocyanin rich extract (25 mg/Kg body weight per day) for 17 weeks and standard diet as reference/rat model [57] | ↑ | ||||||||||||||||||||
| High-fat diet (HFD) with blackberry anthocyanin rich extract (25 mg/Kg body weight per day) for 17 weeks and HFD as reference/rat model [57] | ↑ | ↓ | ↑ | |||||||||||||||||||
| Catechin | ||||||||||||||||||||||
| Tzounis et al. (2010) | A high-cocoa flavanol group (21 mg catechin/day) for four weeks of treatment and a low-cocoa flavanol group (3 mg catechin/day) as reference/clinical trials (22 healthy human volunteers) [63] | ↑ | ↑ | ↓ | ||||||||||||||||||
| Tzounis et al. (2008) | Catechin (150 mg/L) inoculated in stirring batch-culture vessels containing fecal slurry (1:10, w/v), for treatment up to 48 h and incubating flavanol monomers in medium without fecal slurry inoculation as reference/in vitro [48] | ↑ | ↑ | ↓ | Clostridium coccoides–Eubacterium rectale group; C. histolyticum group | |||||||||||||||||
| Caffeic acid | ||||||||||||||||||||||
| Parkar et al. (2013) | Caffeic acid (10 μg/mL) in fermentation medium for 48 h and a control without polyphenol as reference/in vitro [49] | ↑ | ↑ | ↑ | ||||||||||||||||||
| Chlorogenic acid | ||||||||||||||||||||||
| Parkar et al. (2013) | Chlorogenic acid (30 and 100 μg/mL) in fermentation medium for 48 h and a control without polyphenol as reference/in vitro [49] | ↑ | ↑ | ↓ | ||||||||||||||||||
| Daidzein | ||||||||||||||||||||||
| Iino et al. (2019) | Diadzin intake (three traditional Japanese soybean products: natto, tofu, and fried tofu) was calculated based on brief self-administered diet history questionnaire/clinical trials (1044 healthy subjects: 411 men and 633 women) [68] | ↑ | ||||||||||||||||||||
| Epicatechin | ||||||||||||||||||||||
| Tzounis et al. (2008) | Epicatechin (1000 mg/L) inoculated in stirring batch-culture vessels containing fecal slurry (1:10, w/v), for treatment up to 48 h and incubating flavanol monomers in medium without fecal slurry inoculation as reference/in vitro [48] | ↑ | ↑ | E. rectale; C.coccoide | ||||||||||||||||||
| Genistein | ||||||||||||||||||||||
| Paul et al. (2017) | Genistein diet (0.25 g/kg) for 4 weeks and the control group without genistein as reference/mice model (humanized germ-free mice that received fecal transplantation from breast cancer patients) [62] | ↑ | ↑ | ↑ | ↑ | ↑ | ↓ | A. municiphila Ruminococcus torques Clostridium hatheway Bacteroides eggerthii Bacteroides ovatus Eubacterium dolichum | ||||||||||||||
| Phloridzin | ||||||||||||||||||||||
| Mei et al. (2016) | Phloridzin (20 mg/kg/day) for 10 weeks and the control (vehicle treated) as reference/mice model (type 2 diabetes mice model) [58] | ↑ | A. muciniphila | ↑ | ||||||||||||||||||
| Quercetin | ||||||||||||||||||||||
| Etxeberria et al. (2015) | Quercetin supplementation (30 mg/kg body weight/day) for 6 weeks and the control group as reference/rat model (rats fed HFD) [41] | ↓ | ||||||||||||||||||||
| Epigallocatechin-3-gallate and resveratrol (EGCG + RES) | ||||||||||||||||||||||
| Most et al. (2017) | EGCG + RES group (282 mg/d, 80 mg/day) for 12 weeks and baseline as reference/clinical trials (19 subjects: overweight, obese females, 37.8 ± 1.6 years, BMI 29.6 ± 0.5 kg/m2) [72] | ↓ | ↓ | F. prausnitizii | ||||||||||||||||||
| Fruit-derived polyphenols | ||||||||||||||||||||||
| Mayta-Apaza et al. (2018) | Tart cherry juice consumption (8 oz/day) for 5 days and baseline as reference/clinical trials (10 healthy participants with low-bacteroides: 5 males, 5 females, aged 23–30 years) [70] | ↑ | ↑ | ↑ | ↓ | |||||||||||||||||
| Tart cherry juice consumption (8 oz/day) for 5 days and baseline as reference/clinical trials (10 healthy participants with high-bacteroides: 5males, 5females, aged 23–30 years) [70] | ↓ | ↓ | ↑ | |||||||||||||||||||
| Henning et al. (2017) | Vegetable/fruit juice-based juices (consumed 6 bottles, 16 oz./bottle) for 3 days and baseline as reference/clinical trials (25 healthy subjects: 18–50 years of age with a custom diet including < 3 servings of fruits/vegetables per day) [66] | ↑ | No significant effect | |||||||||||||||||||
| Li et al. (2015) | Pomegranate (POM) extract (1000 m/d, total phenolic content expressed as gallic acid equivalents of 680 μg/g) for 4 weeks and baseline as reference/clinical trials (20 healthy adults: 9 women and 11 men) [71] | ↑ | ↑ | ↑ | A. muciniphila | |||||||||||||||||
| Chacar et al. (2018) | Different concentrations of grape phenolic compounds (2.5 and 5 mg/kg/d diluted in 0.1% Dimethyl Sulfoxide) for 14 months and the control group (0.1% Dimethyl Sulfoxide alone) as reference/rat model (30, 2-mo-old rats) [59] | ↑ | ||||||||||||||||||||
| Collins et al. (2016) | A diet of high fat (HF) with an equal combination of the extractable and non-extractable grape-derived polyphenols (EP and NEP) for 16 weeks and the HF diet group as reference/mice model [53] | ↑ | No significant effect | |||||||||||||||||||
| Taira et al. (2015) | A diet of high fat (HF) with polyphenols from aronia, haskap, and bilerry separately (anthocyanin content was 0.4%) for 4 weeks and the HF diet group as reference/rat model [42] | ↓ | ↓ | |||||||||||||||||||
| Neyrinck et al. (2013) | A diet of high fat (HF) with pomegranate peel-derived polyphenols (6 mg/d per mouse) for 4 weeks and the HF diet group as reference/mice model [54] | ↑ | ||||||||||||||||||||
| Viveros et al. (2011) | Antibiotic-free diets containing grape pomace concentrate (60 g/kg) or grape seed extract (7.2 g/kg) for 21 days and the antibiotic-free diets group as reference/broiler chicks model [60] | ↑ | ↑ | ↓ | ↑ | ↑ | ||||||||||||||||
| Mayta-Apaza et al. (2017) | Microbial suspensions (25 mL) of human distal colon compartments cultured with apricots/cherries (5 mL or g), 37 °C for 48 h and stool samples with a low diversity microbiota (dominated by Verrumicrobia and Synergistes) as reference/in vitro (68 human stool samples) [69] | ↑ | ||||||||||||||||||||
| Red wine-derived polyphenols | ||||||||||||||||||||||
| Moreno-Indias et al. (2016) | Participants drank red wine (272 mL/d) for 30 days and baseline as reference/clinical trials (metabolic syndrome patients) [64] | ↓ | ↑ | ↑ | ↑ | ↑ | ↑ | ↓ | F. prausnitzii E. cloacae | ↑ | ||||||||||||
| Queipo-Ortuño et al. (2012) | Red wine polyphenol (272 mL per day, containing polyphenol of 733–797 mg) for 4 weeks and the baseline as reference/clinical trial (healthy men) [65] | ↑ | ↑ | ↑ | ↑ | ↑ | ↑ | ↑ | Bacteroides uniformis Eggerthella lenta Blautia coccoides Eubacterium rectale | ↑ | ||||||||||||
| Dolara et al. (2005) | Red wine polyphenols (50 mg/kg) for 16 weeks and the HFD group as reference/rat model [55] | ↑ | ↑ | ↓ | No significant effect | |||||||||||||||||
| Tea-derived polyphenols | ||||||||||||||||||||||
| Jin et al. (2012) | Green tea (1000 mL/day) for 10 days and baseline as reference/clinical trials [67] | ↑ | ||||||||||||||||||||
| Wang et al. (2018) | TP-HFD groups (TP Low- HFD group with 0.05% TP, TP Middle- HFD group with 0.25% TP, TP High- HFD group with 0.8% TP) for 8 weeks and HFD group as reference/mice model [43] | ↓ | ↑ | |||||||||||||||||||
| Seo et al. (2015) | HFD with fermented green tea group for 8 weeks and HFD group as reference/mice model [44] | ↓ | ||||||||||||||||||||
| Does and Treatment Duration/Test Model | F/B | Potentially Beneficial Microbiota | Potentially Detrimental Microbiota | Species | Diversity | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bifidobacterium sp. | Lactobacillus sp. | Akkermansia sp. | Ruminococcus sp. | Fecalibacterium sp. | Dialister sp. | Allobaculum sp. | Blautia sp. | Clostridium sp. | Roseburia sp. | Lactococcus sp. | Enterobacteria sp. | Escherichia sp. | Salmonella sp. | Bacteroides sp. | Ruminococcus sp. | Veillonella sp. | Clostridium sp. | Coprococcus sp. | Shigella sp. | Oscillibacter sp. | Enterococcus sp. | Pseudomonas sp. | |||||
| Vitamin A | |||||||||||||||||||||||||||
| Huda et al. (2019) | One dose of 50,000 IU vitamin A within 48 h of birth and placebo as reference/clinical trial (306 infants in early (6–15 week) or late (2 y) infancy) [81] | ↑ | ↑ | ||||||||||||||||||||||||
| Liu et al. (2017) | A dose of 200,000 IU vitamin A once orally for a 6-month follow-up intervention and baseline as reference/cinical trial (64 children with autism spectrum disorder, aged 1 to 8 years old) [79] | ↓ | ↑ | ||||||||||||||||||||||||
| Lv et al. (2016) | Vitamin A—deficient and vitamin A—normal as reference/clinical trial (59 pediatric patients with persistent diarrhea aged 1–12 months) [80] | ↓ | ↑ | Clostridium butyricum | ↓ | ||||||||||||||||||||||
| Lee and Ko (2017) | 1 mg/kg/day of retinoic acid, the metabolite of dietary vitamin A on murine norovirus infection mice for 8 days and the murine norovirus infection mice as reference/mice model [76] | ↑ | ↑ | ↑ | ↑ | ||||||||||||||||||||||
| Lee et al. (2016) | 1 mg/kg/day of retinoic acid, the metabolite of dietary vitamin A for 8 days and the control group as reference/mice model [75] | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| 1 mg/kg/day of retinoic acid, the metabolite of dietary vitamin A on murine norovirus infection mice for 8 days, and the murine norovirus infection mice as reference/mice model [75] | ↑ | ||||||||||||||||||||||||||
| Vitamin B | |||||||||||||||||||||||||||
| Miki et al. (2017) | Oral gavage with pyridoxine hydrochloride (vitamin B6) at 100 μg/mouse (Sigma) at 1–7 days after infection and baseline as reference/mice model (streptomycin-treated and Salmonella enterica serovar Typhimuirum-infected mice) [88] | ↓ | |||||||||||||||||||||||||
| Vitamin C | |||||||||||||||||||||||||||
| Li et al. (2017) | Mean 294.9 mg/d for men and mean 189.8 mg/day for women (3-day food dairy using household measures) and baseline as reference/clinical trial (16 free-living adults with cystic fibrosis) [90] | ↓ | |||||||||||||||||||||||||
| Vitamin D | |||||||||||||||||||||||||||
| Naderpoor et al. (2019) | > 75 nmol/L 25-hydroxyvitamin D (25(OH)D) and vitamin D-deficient (≤ 50 nmol/L) as reference/clinical trial (25 overweight or obese (BMI ≥25 kg/m2) healthy adults) [111] | ↓ | ↓ | ↑ | No effect | ||||||||||||||||||||||
| Garg et al. (2018) | A dose of 40,000 IU vitamin D once weekly for 8 weeks using two capsules of 20,000 IU (Plenachol, Encap) and baseline as reference/clinical trial (patients with vitamin D deficiency: 25[OH]D < 50 nmol/L) [107] | ↑ | No effect | ||||||||||||||||||||||||
| Talsness et al. (2017) | Maternal supplementation of vitamin D was carried out at 0μg/day, <10 μg/day, or ≥ 10 μg/day during pregnancy and the fecal samples from their one-month old babies were tested for gut microbiota/clinical trial [109] | ↓ | ↓ | ↑ | ↓ | Bacteroides fragilis; Clostridium difficile | |||||||||||||||||||||
| Kanhere et al. (2017) | 50,000 IU of oral vitamin D3 supplemented for 12 weeks and placebo as reference/clinical trial (Cystic Fibrosis patients as model) [98] | ↑ | ↓ | ||||||||||||||||||||||||
| Luthold et al. (2017) | vitamin D intake ≥ 10 μg/day in three tertiles (tertile 1: 4.21–18.93 ng/mL, tertile 2: 18.93–26.48 ng/mL, tertile 3: 26.48–61.30 ng/mL) and the highest vitamin D intake as reference/clinical trial (150 young healthy adults) [110] | ↑ | ↑ | ||||||||||||||||||||||||
| Bashir et al. (2016) | Vitamin D3 supplementation with weekly dose of 980 IU/kg bodyweight for 4 weeks, and 490 IU/kg bodyweight for the remaining 4 weeks, the baseline as reference/clinical trial (healthy volunteer) [103] | ↓ | ↓ | ↑ | |||||||||||||||||||||||
| Ciubotaru et al. (2015) | Vitamin D supplementation with weekly ergocalciferol (50,000 IU) on stable normal glucose tolerance for 12 months and the same treatment on stable prediabetes as reference/clinical trial [97] | ↓ | ↓ | ↓ | ↓ | ||||||||||||||||||||||
| Vitamin E | |||||||||||||||||||||||||||
| Tang et al. (2016) | Vitamin E (18 mg/day) added iron therapy (6 mg/kg/d) for 8 weeks and placebo added iron therapy (6 mg/kg/day) as reference/clinical trial (infants and toddlers who were at risk of iron deficiency) [119] | ↑ | |||||||||||||||||||||||||
| Choi et al. (2019) | High vitamin E group (0.18 mg/20 g of bw per day) treated by oral gavage for 34 days, the control group (0.2 mL of corn oil) and low vitamin E group (0.06 mg/20 g of bw per day) as reference/mice model [117] | ↓ | |||||||||||||||||||||||||
| Calcium | |||||||||||||||||||||||||||
| Trautvetter et al. (2018) | Supplementation of 1000 mg calcium +1000 mg phosphorus/day for 8 weeks and the supplementation of 1000 mg phosphorus/day as reference/clinical trials (healthy men) [124] | ↑ | Clostridium XVIII | ||||||||||||||||||||||||
| Supplementation of 1000 mg calcium +1000 mg phosphorus/day for 8 weeks and the supplementation of 500 mg calcium + 1000 mg phosphorus/day as reference/clinical trials (healthy men) [124] | ↓ | ↑ | Clostridium XVIII | ||||||||||||||||||||||||
| Li et al. (2018) | Maternal insufficient calcium intake (2.5 g/kg) / Maternal excess calcium intake (12 g/kg) for 8 weeks will influence the gut microbiota in the offspring/mice model [126] | ↑* | |||||||||||||||||||||||||
| Chaplin et al. (2016) | HFD enriched with calcium supplementation (12 g/kg) for 54 days and HFD group (4 g/kg) as reference/mice model [127] | ↑ | ↑ | ||||||||||||||||||||||||
| Borda-Molina et al. (2016) | Diet mixed with the supplementation of calcium (3 g/kg) for 10 days and diet without calcium group as reference/broiler chickens model [151] | ↓ | |||||||||||||||||||||||||
| Aslam et al. (2016) | HFD enriched with calcium supplementation (5.25 g/kg) for 18 months and HFD group (0.41 g/kg) as reference/mice model [125] | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| Magnesium** | |||||||||||||||||||||||||||
| Jørgensen et al. (2015) | Dietary magnesium deficiency (0.02% magnesium) for 6 weeks and standard diet (0.2% magnesium) group as reference/mice model [130] | ↓ | |||||||||||||||||||||||||
| Winther et al. (2015) | Dietary magnesium deficiency (0.02% magnesium) for 6 weeks and standard diet (0.2% magnesium) group as reference / Mice model [131] | No significant effect | |||||||||||||||||||||||||
| Pachikian et al. (2010) | Magnesium-deficient diet (70 mg/kg) for 4 days and control diet (500 mg/kg) group as reference/mice model [129] | ↓ | |||||||||||||||||||||||||
| Magnesium-deficient diet (70 mg/kg) for 21 days and control diet (500 mg/kg) group as reference/mice model [129] | ↑ | ↑ | |||||||||||||||||||||||||
| Iron | |||||||||||||||||||||||||||
| Kotryna Simonyté Sjodin et al. (2019) | High-iron-fortified formula (6.4 mg Fe/day) for 45 days and the baseline as reference/clinical trials (6-month-old healthy Swedish infants) [139] | ↓ | |||||||||||||||||||||||||
| High-iron-fortified formula (6.4 mg Fe/day) for 45 days and low-iron-fortified formula (1.2 mg Fe/day) as reference/clinical trials (6-month-old healthy Swedish infants) [139] | ↓ | ↑ | |||||||||||||||||||||||||
| High-iron-fortified formula (6.4 mg Fe/day) for 45 days and iron drops (no-added-iron formula with liquid ferrous sulfate supplementation (5.7 mg Fe/day) as reference/clinical trials (6-month-old healthy Swedish infants) [139] | ↑ | ↓ | |||||||||||||||||||||||||
| Tang et al. (2017) | Multiple micronutrient powder containing 12.5 mg iron daily for 3 months and multiple micronutrient powder without the iron as reference/clinical trials (6-month-old Kenyan infants) [135] | ↓ | ↑ | ↑ | |||||||||||||||||||||||
| Lee et al. (2017) | Iron therapy via Per Oral over 3 months and via Intravenous as reference/clinical trials (IBD patients with anemia) [149] | ↓ | ↓ | Fecalibacterium prausnitzii; Ruminococcus bromii | |||||||||||||||||||||||
| Jaeggi et al. (2015) | Home-fortified maize porridge containing 2.5 mg iron daily for 4 months and porridge without the iron as reference/clinical trials (6-month-old Kenyan infants) [134] | ↑ | ↑ | ↑ | Escherichia coli | ||||||||||||||||||||||
| Jaeggi et al. (2015) | Home-fortified maize porridge containing 12.5 mg iron daily for 4 months and porridge without the iron as reference/clinical trials (6-month-old Kenyan infants) [134] | ↓ | ↑ | ↑ | ↑ | ↑ | Escherichia coli | ||||||||||||||||||||
| Dostal et al. (2014) | Oral tablets containing 50 mh Fe for 4 d/week, last for 38 weeks, and the placebo as reference/clinical trial (6–11 rural South African children with Fe deficiency) [138] | ||||||||||||||||||||||||||
| Zimmermann et al. (2010) | Iron-fortified biscuits containing 20 mg Fe/d, and 4 times/week, last for 6 months, and the nonfortified biscuits as reference/clinical trial (6–14 Ivorian children) [136] | ↓ | ↑ | ↑ | ↑ | ||||||||||||||||||||||
| Fang et al. (2018) | 1 mL liquid iron (Liquid iron preparations were prepared by dissolving FeSO4 salt in 1 mL of 0.01 mol/L HCL) preparation containing 8 mg, 16 mg, or 24 mg of iron for 30 days and control group (0.01 mol/L HCI) as reference/rat model [140] | ↓ | ↑ | ↑ | |||||||||||||||||||||||
| Constante et al. (2017) | Iron-sufficient diet (50 mg/kg) for 4 weeks and iron-deficient diet (5 mg/kg) as reference/mice model [145] | ↓ | ↑ | ↑ | Bacteroides caccae | ||||||||||||||||||||||
| Iron-supplemented diet (500 mg/kg) for 4 weeks and iron-sufficient diet (50 mg/kg) as reference/mice model [145] | ↑ | ||||||||||||||||||||||||||
| Ferrous bisglycinate (FBG) (50 mg/kg) for 4 weeks and ferrous sulfate (FS) (50 mg/kg) as reference/mice model [145] | |||||||||||||||||||||||||||
| Ferric ethylenediaminetetraacetic acid (FEDTA) for 4 weeks and ferrous sulfate (FS) (50 mg/kg) as reference/mice model [145] | ↓ | ↓ | ↓ | ||||||||||||||||||||||||
| Kortman et al. (2016) | Medium with iron (50 or 250 µmol/l ferrous sulfate, 50 or 250 µmol/l ferric citrate, or 50 umol/L hemin) incubated for 44 h at 37 ℃ and medium without supplementary iron as reference/in vitro (Human fecal sample) [141] | ||||||||||||||||||||||||||
| Phosphorus | |||||||||||||||||||||||||||
| Borda-Molina et al. (2016) | Diet mixed with the supplementation of Phosphorus (3 g/kg) for 10 days and diet without calcium group as reference/broiler chickens model [151] | ↑↓# | ↑ | Lactobacillus taiwanensis | ↑↓ | ||||||||||||||||||||||
| Witzig et al. (2015) | Diet mixed with the supplementation of phosphorus for 10 days and diet without phosphorus group as reference/broiler chickens model [253] | ↑ ↓# | Lactobacillus taiwanensis; Lactobacillus salivarius; Lactobacillus crispatus; Lactobacillus reuteri | ↑↓ | |||||||||||||||||||||||
| Zinc | |||||||||||||||||||||||||||
| Zackular et al. (2016) | High Zn diet (1000 mg/kg) for 5 weeks and control diet (29 mg/kg) as reference/mice model [155] | ↑ | ↑ | Clostridium XI | ↓ | ||||||||||||||||||||||
| Reed et al. (2015) | Zn diet (42 ug/g) for 28 days and Zn deficiency diet (2.5 ug/g) as reference/chicks model [153] | ↓ | ↓ | ↑ | |||||||||||||||||||||||
| Shao et al. (2014) | Supplemental Zn (120 mg/kg) diet for 42 days and diet without Zn as reference/Salmonella Typhimurium-challenged Broiler chicken model [154] | ↑ | ↓ | ↑ | |||||||||||||||||||||||
| Selenium | |||||||||||||||||||||||||||
| Zhai et al. (2018) | Selenium diet (added amounts of Se 0.4 mg/kg) for 8 weeks and Se-deficient diet (Se level < 0.01 mg /kg) as reference/mice model [158] | ↑ | Not significant effect | ||||||||||||||||||||||||
| Kasaikina et al. (2011) | Selenium diet (added amounts of Se 0, 0.1, 0.4, and 2.25 ppm) for 10 weeks and Se-deficient diet (0 ppm Se diet) as reference/mice model [157] | ↑ | |||||||||||||||||||||||||
| Iodine | |||||||||||||||||||||||||||
| Shen et al. (2019) | 18μg/kg/d iodine for 8 weeks and un-treated obese mice as reference/HFD-induced obesity mice model [159] | ↑ | ↓ | ↑ | ↑ | ↑ | |||||||||||||||||||||
| 18μg/kg/d iodine for 8 weeks and control group as reference/mice model [159] | ↑ | ↑ | ↑ | ↑ | ↓ | ↑ | Fecalibacterium prausnizii | ||||||||||||||||||||
| Dose and Treatment Duration/Test Model | F/B | Potentially Beneficial Microbiota | Potentially Detrimental Microbiota | Species | Diversity | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bifidobacterium sp. | Lactobacillus sp. | Akkermansia sp. | Fecalibacterium sp. | Roseburia sp. | Eubacterium sp. | Bacteroides sp. | Prevotella sp. | Clostridium sp. | Ruminococcus sp. | Eggerthella sp. | Ruminococcus sp. | Clostridium sp. | Bacteroides sp. | Enterococcus sp. | |||||
| Arabinoxylan | |||||||||||||||||||
| Hald et al. (2016) | Arabinoxylan (AX)-enriched diet (whole-grain rye and enzyme-treated wheat bran) for 4 weeks and baseline as reference/clinical trials (24 subjects: 39–75 years with metabolic syndrome) [174] | ↑ | ↓ | ||||||||||||||||
| Arabinoxylan (AX)-enriched diet (whole-grain rye and enzyme-treated wheat bran) for 4 weeks and a low-fiber western-style diet (refined grains and a minimal concentration dietary fiber) as reference/clinical trials (24 subjects: 39–75 years with metabolic syndrome) [174] | ↑ | ↓ | |||||||||||||||||
| Arabinoxylan-oligosaccharides | |||||||||||||||||||
| Windey et al. (2015) | Wheat bran extract (10 g/day) containing arabinoxylan-oligosaccharides for 3 weeks and placebo (maltodextrin, 10 g/day) as reference/clinical trials (20 healthy subjects: 17 women and 3 men; age range 19–44 years; BMI range 18.7–24.3 kg/m2) [182] | ↑ | Bifidobacterium adolescentis | ||||||||||||||||
| François et al. (2014) | WBE (enriched AXOS) at 5 g/day for 3 weeks and placebo as reference/clinical trials (29 healthy children, age range of 8–12 years) [181] | ↑ | |||||||||||||||||
| Walton et al. (2012) | AXOS treatment (2.2 g) for 21 days and placebo as reference/clinical trials (40 healthy adults: 20 males and 20 females, mean age 31.4 years (± 8.9), average BMI 23.3 kg/m2 (± 2.8)) [178] | ↑ | |||||||||||||||||
| François et al. (2012) | Wheat bran extract (WBE, 10 g/day)-enriched AXOS for 3 weeks; both WBE (3 g/day) group and placebo group as references/clinical trials (63 healthy adults) [176] | ↑ | |||||||||||||||||
| Cloetens et al. (2010) | Arabinoxylan-oligosaccharides (AXOS, 10 g/d) for 3 weeks and placebo as reference/clinical trials (20 healthy subjects: 14 women, 6 men; mean age 24 (sd 5) years, mean BMI 20.9 (sd 2.3) kg/m2) [175] | ↑ | |||||||||||||||||
| Xylo-oligosaccharide | |||||||||||||||||||
| Childs et al. (2014) | XOS (8 g/day) for 21 days and placebo as reference/clinical trials (44 healthy adults, 25–65 years) [179] | ↑ | |||||||||||||||||
| Finegold et al. (2014) | XOS (1.4 g/day or 2.8 g/day) for 8 weeks and the placebo as reference/clinical trials (32 healthy adults) [180] | ↑ | |||||||||||||||||
| Lecerf et al. (2012) | Xylo-oligosaccharide (XOS, 5 g/d) for 4 weeks and placebo (wheat maltodextrin, 5 g/day) as reference/clinical trials (60 healthy volunteers) [177] | ↑ | |||||||||||||||||
| Galacto-oligosaccharides | |||||||||||||||||||
| Azcarate-Peril et al. (2017) | GOS ( > 95% purity) treatment in 5 d increments according to a fixed schedule from 1.5 g/day to 15 g/day up to 36 days and placebo (Sweetose) as reference/clinical trials (52 lactose-intolerant individuals, mean age of 41 year, mean BMI of 27.1) [192] | ↑ | ↑ | ↑ | ↓ | ||||||||||||||
| Civardi et al. (2017) | GOS (7 g/L) for 60 ± 5 days and standard formula without GOS as reference/clinical trials (117 healthy infants: formula milk supplemented with functional ingredients (n = 55), or a standard formula (n = 62)) [197] | ↑ | |||||||||||||||||
| Paganini et al. (2017) | Iron-containing micronutrient powders (MNPs) with GOS (7.5 g/day) for 4 months; MNPs without iron and MNPs with 5 mg iron as references/clinical trials (Kenyan infants aged 6.5–9.5 months (n = 155)) [198] | ↑ | ↑ | ↓ | |||||||||||||||
| Musilova et al. (2015) | GOS (9 g/day) and maltodextrins (1 g/d) for 5 days and baseline as reference/clinical trials (fecal samples of 11 healthy adults: 6 women and 5 men; mean age, 35.18 ± 10.91 years) [190] | ↑ | |||||||||||||||||
| Vulevic et al. (2015) | GOS (5.5 g/day) for 10 weeks and placebo (maltodextrin) as reference/clinical trials (40 elderly volunteers: 25 women and 15 men; range age of 65–80 years) [191] | ↑ | ↑ | ||||||||||||||||
| Sierra et al. (2015) | GOS (0.44 g/day) for 6 months with additional feeding of GOS (0.5 g/dl) for 6 months and placebo as reference/clinical trials (365 healthy infants had a gestational age of 37–42 weeks and a birth weight greater than 2500 g) [196] | ↑ | |||||||||||||||||
| Giovannini et al. (2014) | GOS-supplemented formula (0.4 g/100 mL) for up to 70 days and the identical formula without GOS as reference/clinical trials (160 healthy infants: gestational age from 37 to 42 completed weeks; birth weight ≥ 2500 g) [184] | ↑ | ↑ | ↓ | |||||||||||||||
| Whisner et al. (2013) | Smoothie drinks with daily GOS intake 0 g, 5 g, and 10 g for three 3-week periods in a random order and baseline as reference/clinical trials (31 healthy adolescent girls: age 10–13 years) [193] | ↑ | |||||||||||||||||
| Westerbeek et al. (2013) | Mixture of neutral and acidic oligosaccharides (scGOS/lcFOS/pAOS) in increasing doses between days 3 and 30 of life to 1.5 g/kg/day, and the placebo as reference/clinical trials (113 healthy infants) [195] | ↑ | ↓ | ||||||||||||||||
| Scalabrin et al. (2012) | Formula with polydextrose (PDX) and galacto-oligosaccharides (GOS) (4 g/L, 1:1 ratio) for 60 days and control formula (Enfamil Lipil) as reference/clinical trials (230 healthy infants: 21- to 30-days-old with 37 to 42 weeks of gestational age, birth weight ≥2500 g) [194] | ↑ | |||||||||||||||||
| Walton et al. (2012 b) | GOS (4 g) twice/d for 3 weeks and placebo as reference/clinical trials (39 volunteers: age 50–81 years, BMI of 19.7–38.4 kg/m2) [189] | ↑ | |||||||||||||||||
| Davis et al. (2011) | GOS with four dosages (0, 2.5, 5, and 10 g/d) for 3 eewks each (12 weeks in total) and baseline as reference/clinical trials (18 healthy volunteers) [188] | ↑ | |||||||||||||||||
| Davis et al. (2010) | GOS-containing chocolate chews with dosage levels (5.0 g and 10.0 g) for 3 weeks and baseline as reference/clinical trials (18 subjects: 13 males and 5 females, ages of 19 to 50 years old) [187] | ↑ | |||||||||||||||||
| Monteagudo-Mera et al. (2016) | GOS (90% purity, 40 mL/day equivalent of 0.26 g/kg bodyweight of GOS) for 14 days and baseline as reference/mice model (four pathogen-free mice) [186] | ↑ | ↑ | ↑ | ↑ | ↓ | |||||||||||||
| Ladirat et al. (2014) | GOS (4.2 mg/mL) and 4 antibiotics (1 or 10 μg/mL) in the medium for healthy adult fecal sample fermentation for up to 48 h and antibiotic-treated samples as reference/in vitro (8 healthy adult fecal samples) [185] | ↑ | ↑ | ||||||||||||||||
| Raffinose-oligosaccharide | |||||||||||||||||||
| Fernando et al. (2010) | Diet fortified with canned chickpeas (200 g/day) or raffinose oligosaccharide (5 g/day) for 3 weeks and control diet as reference/clinical trials (12 healthy adults: 18–65 years) [200] | ↑ | ↓ | Clostridium clusters I/II and XI; Clostridium histolyticum; Clostridium lituseburense group | No significant effect | ||||||||||||||
| Lactose | |||||||||||||||||||
| Francavilla et al. (2012) | Formula with no lactose for 2 months followed by an identical lactose-containing (3.8% lactose) formula for an additional 2 months to infants with cow’s milk protein allergy (CMA), and the formula with no-lactose as reference/clinical trials (28 infants with CMA) [199] | ↑ | ↑ | ||||||||||||||||
| Inulin/inulin-type fructans | |||||||||||||||||||
| Drabinska et al. (2018) | Oligofructose-enriched inulin (Synergy 1) (10 g/d) for 3 months and placebo (maltodextrin; 7 g/day) as reference/clinical trials (34 pediatric celiac disease patients, mean age 10 years, 62% females, on a gluten-free diet) [206] | ↑ | |||||||||||||||||
| Vandeputte et al. (2017) | Chicory-derived Orafti inulin (12 g/day) for 4 weeks and placebo as reference/clinical trials (42 healthy adults with mild constipation) [205] | ↑ | No significant effect | ||||||||||||||||
| Nicolucci et al. (2017) | Oligofructose-enriched inulin (8 g/day) for 16 wks and placebo (maltodextrin) as reference/clinical trials (42 healthy children, 7–12 years, overweight or obese >85 th percentile of body mass index) [208] | ↑ | ↓ | Bacteroides vulgatus | |||||||||||||||
| Salazar et al. (2014) | ITF (16 g/day) for 3 months and the placebo maltodextrin group as reference/clinical trials (30 obese women, BMI > 30 kg/m2, age range of 18 to 65 years) [207] | ↑ | Bifidobacterium longum, B. pseudocatenulatum; B. adolescentis | ||||||||||||||||
| Dewulf et al. (2013) | ITF (Synergy 1, inulin/ oligofructose 50/50 mix, 16 g/day) for 3 months and placebo (maltodextrin, 16 g/d) as reference/clinical trials (30 obese women, BMI > 30 kg/m2, age range of 18 to 65 years) [204] | ↑ | ↑ | ↓ | Fecalibacterium prausnitzii; Bacteroides intestinalis; B. vulgatus | ||||||||||||||
| Ramirez-Farias et al. (2008) | Inulin (10 g/day) for 16 days and a control period without any supplement intake as reference/clinical trials (12 human volunteers) [203] | ↑ | ↑ | ||||||||||||||||
| Catry et al. (2017) | n-3 polyunsaturated fatty acid-depleted diet for 12 weeks with inulin-type fructans (ITF) (250 mg/mouse/d) for the last 15 days and group without ITF as reference/mice model (n-3 PUFA-depleted Apoe−/− mice) [202] | ↑ | No significant effect | ||||||||||||||||
| Licht et al. (2006) | Inulin (150 g/kg) in diet for 5 weeks and baseline as reference/Rats model (8 rats with western type diet) [201] | ↓ | |||||||||||||||||
| High-fiber diet | |||||||||||||||||||
| Dao et al. (2016) | Calorie-restricted diet (enriched with fiber and protein) for 6 weeks and baseline as reference/clinical trials (49 overweight and obese adults) [167] | ↑ | Akkermansia muciniphila | ||||||||||||||||
| Candela et al. (2016) | Fiber-rich macrobiotic Ma-Pi 2 diet and a diet recommended by Italian professional societies for type 2 diabetes (T2 D) treatment, baseline as reference/clinical trials (40 overweight T2 D patients, aged 50–77 years) [168] | ↑ | ↑ | ↑ | ↑ | ↑ | |||||||||||||
| Holscher et al. (2014) | Soluble corn fiber (21 g/day) for 21 days and no supplemental fiber placebo as reference/clinical trials (14 healthy adult men) [169] | ↓ | ↑ | ||||||||||||||||
| Tap et al. (2015) | A basal diet supplemented with dietary fiber (40 g/day) for 5 days and diet with dietary fiber (10 g/day) as reference/clinical trials (19 healthy adults: 9 males and 10 females, aged 19–25 years) [170] | ↑ | ↑ | ||||||||||||||||
| Kovatcheva-Datchary et al. (2015) | Barley kernel-based bread consumption for 3 days and white wheat flour bread group as reference/clinical trials (39 healthy subjects: 6 men and 33 women, age 50–70 years, BMI 18–28 kg/m2) [171] | ↑ | Prevotella copri | ||||||||||||||||
| García-Peris et al. (2012) | Mixture of fiber (6 g twice daily, 50% inulin and 50% fructo-oligosaccharide) from one week before to three weeks after radiotherapy and the placebo (maltodextrin, 6 g twice daily) as reference/clinical trials (≥ 18-year-old female patients with gynecological cancer who received radiotherapy after surgery) [166] | ↑ | ↑ | ||||||||||||||||
| De Filippo et al. (2010) | High-fiber diet represented by the one of children in a rural African village of Burkina Faso (BF) and a diet represented by the one of European (EU) children as reference/clinical trials (15 healthy children in BF, additional 15 healthy children in EU, age range of 1–6 years) [172] | ↓ | ↑ | ||||||||||||||||
| Benus et al. (2010) | Dietary fiber formula (dietary fiber 19.6 and 18.0 g/day) for 14 days and the fiber-free diet as reference/clinical trials (10 healthy subjects: 6 women and 4 men, age range of 21–34 years) [164] | ↑ | |||||||||||||||||
| Carvalho-Wells et al. (2010) | Maize-derived whole grain cereal (48 g/day) for 21 days and the same dose of placebo cereal as reference/clinical trials (28 healthy volunteers: 7 males and 21 females, age range 20–51 years, BMI 20–30 kg/m2) [165] | ↑ | |||||||||||||||||
| Avila-Nava et al. (2017) | Pre-hispanic Mexican diet (PMD: containing corn flour, black beans concentrate, nopal, chia and pumpkin seed, with 8 g fiber/100 g diet) for 3 months and control diet (AIN-93) as reference/rats model (18 rats sucrose-enriched high-fat diet caused gut microbiota dysbiosis) [162] | ↓ | ↑ | ||||||||||||||||
| Resistant Starch | |||||||||||||||||||
| Martínez et al. (2010) | Crackers (100 g/day) containing native starch for 3 weeks and the baseline as references/clinical trials (10 subjects: age range of 23–38 years) [210] | ↑ | Fecalibacterium prausnitzii | ||||||||||||||||
| Crackers (100 g/day) containing 33 g type 2 resistant starch (RS) for 3 wks and RS type 4 as references /Clinical trials (10 subjects: age range of 23–38 years) [210] | ↑ | ↑ | Ruminococcus bromii Eubacterium rectale | ||||||||||||||||
| Crackers (100 g/d) containing 33 g type 4 RS for 3 weeks and the baseline as references/clinical trials (10 subjects: age range of 23–38 years) [210] | ↓ | ↑ | Bifidobacterium adolescentis | ||||||||||||||||
| Butyrylated Starch | |||||||||||||||||||
| Hald et al. (2016) | Resistant starch (type 2) and arabinoxylan enriched diet for 4 weeks and the baseline as reference/clinical trials (19 subjects: 39–75 years, with metabolic syndrome) [174] | ↑ | ↓ | ||||||||||||||||
| Resistant starch (type 2) and arabinoxylan enriched diet for 4 weeks and a low-fiber western-style diet group as reference/clinical trials (19 subjects: 39–75 years, with metabolic syndrome) [174] | ↑ | ↓ | |||||||||||||||||
| Le Leu et al. (2015) | Butyrylated high-amylose maize starch (HAMSB, 40 g/d) with a high red meat diet (HRM, cooked red meat 300 g/d) for 4 weeks and baseline as reference/clinical trials (23 individuals with red meat-increased O6-methyl-2-deoxyguanosine adduct level) [212] | ↑ | ↑ | ↑ | ↓ | Clostridium coccoides C. leptum group Ruminococcus bromii; R. torques; R. gnavus | |||||||||||||
| Oligofructose | |||||||||||||||||||
| Wernimont et al. (2015) | Formula with oligofructose (OF, 3.0 g/L) for 8 weeks and identical formula without OF but enriched with α-lactalbumin (3.0 g/L) as reference/clinical trials (48 healthy infants) [214] | ↑ | |||||||||||||||||
| Cluny et al. (2015) | High-fat/high-sucrose (HFS) diet with 10% oligofructose for 6 weeks and HFS diet group as reference/rats model (46 rats with diet-induced obesity (23) and diet-resistant (23)) [213] | ↑ | ↑ | ↑ | ↓ | Clostridium leptum | |||||||||||||
| Polydextrose | |||||||||||||||||||
| Costabile et al. (2012) | Polydextrose (PDX; 8 g/d) for 3 weeks and the equivalent placebo (maltodextrin powder, 8 g/d) as reference/clinical trials (31 healthy subjects: age 18–50 years, BMI 19–25 kg/m2) [216] | ↑ | ↑ | Ruminococcus intestinalis Clostridium clusters I, II and IV | |||||||||||||||
| High/low carbohydrate diet | |||||||||||||||||||
| Haro et al. (2016) | High-complex carbohydrate diet with low fat (28% fat, 12% monounsaturated) for 1 year and the baseline group as reference/clinical trials (20 obese patients (men) with coronary heart disease) [173] | ↑ | ↓ | ↑ | Fecalibacterium prausnitzii | ||||||||||||||
| Ley et al. (2006) | Carbohydrate-restricted low-calorie diet for 1 year with obese people and a lean people group as reference/clinical trials (12 obese people) [35] | ↓ | ↑ | ||||||||||||||||
| Dose and Treatment Duration/Test Model | F/B | Potentially Beneficial Microbiota | Potentially Detrimental Microbiota | Species | Diversity | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bifidobacterium sp. | Lactobacillus sp. | Akkermansia sp. | Fecalibacterium sp. | Roseburia sp. | Eubacterium sp. | Bacteroides sp. | Prevotella sp. | Blautia sp. | Parabacteroides sp. | Ruminococcus sp. | Oscillospira sp. | Allobaculum sp. | Clostridium sp. | Enterococcus sp. | Escherichia sp. | Bilophila sp. | Streptococcus sp. | Alistipes sp. | Bacteroides sp. | Serratia sp. | Pantoea sp. | Citrobacter sp. | |||||
| Saturated fat | |||||||||||||||||||||||||||
| Patterson et al. (2014) | High-palm oil (45% energy from fat) for 16 weeks and compared with low-fat diet (12% energy from fat)/mice model [235] | ↑ | |||||||||||||||||||||||||
| De Wit et al. (2012) | High-palm oil (45% energy from fat) for 8 weeks and low-palm oil diet (10% energy from fat) as reference/mice model [236] | ↑ | ↑ | Clostridium XI | ↓ | ||||||||||||||||||||||
| Devkota et al. (2012) | Lard-based fat (37% energy from fat) for 24 weeks and low-fat diet (5% energy from fat) as reference/mice model [228] | ↑ | |||||||||||||||||||||||||
| Milk-derived fat (37% energy from fat) for 24 weeks and low-fat diet (5% energy from fat) as reference/mice model [228] | ↓ | ↑ | B. wadsworthia | ↓ | |||||||||||||||||||||||
| Zentek et al. (2012) | Diet with medium-chain fatty acids uncoated (MCFA) for 4 weeks and the diet without MCFA as reference/piglets model [237] | ↑ | ↑ | L. johnsonii; L. amylovorus | |||||||||||||||||||||||
| Polyunsaturated fatty acid (PUFA) | |||||||||||||||||||||||||||
| Younge et al. (2017) | Early enteral supplementation with a high fat-PUFA blend of fish oil and safflower oil up to 10 weeks and the standard nutritional therapy as reference/clinical trial (premature infants with an enterostomy) [239] | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | ↑ | |||||||||||||||||||
| Patterson et al. (2014) | High flaxseed/fish oil (45% energy from fat) for 16 weeks and compared with a low-fat diet (12% energy from fat)/mice model [235] | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| Devkota et al. (2012) | High safflower oil (37% energy from fat) for 24 weeks and low-fat diet (5% energy from fat) as reference/mice model [228] | ↓ | ↓ | ||||||||||||||||||||||||
| Fat diet (17% lard) | |||||||||||||||||||||||||||
| Avila-Nava et al. (2017) | HFD (17% lard fat and 7% soy oil) for 6 months and control diet (7% soy oil) as reference/rat model [162] | ↑ | ↓ | ↓ | ↓ | ||||||||||||||||||||||
| Fat diet (20–21.45% fat) | |||||||||||||||||||||||||||
| Wan et al. (2019) | Fat diet (20%) for 6 months and the baseline as reference/clinical trial (healthy young adults) [230] | ↑ | ↑ | ||||||||||||||||||||||||
| Lecomte et al. (2015) | HFD (21.45% fat) for 16 weeks and normal diet (12% fat) as reference/rat model [229] | ↓ | ↓ | ↑ | |||||||||||||||||||||||
| Qiao et al. (2013) | HFD (21.45% mixed fat) for 8 weeks and normal diet (4.89% fat) as reference/mice model [233] | ↑ | ↑ | Escherichia coli | |||||||||||||||||||||||
| Fat diet (28–35%) | |||||||||||||||||||||||||||
| Wan et al. (2019) | Moderate fat diet (30%) for 6 months and the baseline as reference/clinical trial (healthy young adults) [230] | ↓ | |||||||||||||||||||||||||
| Haro et al. (2017) | Dietary intervention of < 30% total fat for 2 years and the baseline as reference/clinical trial (male patients with coronary heart disease, who are obese and also with severe metabolic disease) [231] | ↓ | ↑ | ↑ | ↑ | ↓ | ↓ | ||||||||||||||||||||
| Haro et al. (2017) | Dietary intervention of minimum 35% fat for 2 years and the baseline as reference/clinical trial (male patients with coronary heart disease) [231] | ↓ | ↑ | ↑ | ↑ | ↑ | ↑ | ↑ | Parabacteroides distasonis Fecalibacterium prausnitzii | ||||||||||||||||||
| Haro et al. (2016) | Dietary intervention of 28% fat for 1 years and the baseline as reference/clinical trial (male obese patients with coronary heart disease) [173] | ↓ | ↑ | Fecalibacterium prausnitzii | |||||||||||||||||||||||
| Dietary intervention of 35% fat for 1 years and the baseline as reference/clinical trial (male obese patients with coronary heart disease) [173] | ↑ | ↓ | ↑ | ↑ | Parabacteroides distasonis | ||||||||||||||||||||||
| Fat diet (40%) | |||||||||||||||||||||||||||
| Wan et al. (2019) | High-fat diet (40%) for 6 months and the baseline as reference/clinical trial (healthy young adults) [230] | ↓ | ↓ | ↑ | ↑ | ||||||||||||||||||||||
| High-fat diet (40%) for 6 months and the lower-fat diet (20%) as reference/clinical trial (healthy young adults) [230] | ↓ | ↓ | ↓ | ↑ | ↑ | ↓ | |||||||||||||||||||||
| HFD (44–45% fat) | |||||||||||||||||||||||||||
| Chen et al. (2018) | HFD (45% fat) for 8 weeks and control diet (10% fat) as reference/mice model [220] | ↑ | |||||||||||||||||||||||||
| Collins et al. (2016) | HFD (44% mixed fat) for 16 weeks and low-fat diet (10% fat) as reference/mice model [53] | ↓ | |||||||||||||||||||||||||
| Hamilton et al. (2015) | HFD (45% mixed fat) for 1, 3, 6 weeks and normal diet (13% fat) as reference/rat model [218] | ↑ | ↓ | ||||||||||||||||||||||||
| Murphy et al. (2010) | HFD (45% fat) for 8 weeks and low-fat diet (10% fat) as reference/mice model [221] | ↑ | |||||||||||||||||||||||||
| Hildebrandt et al. (2009) | HFD (45% fat) for 1 month and standard diet (12% fat) as reference/mice model [219] | ↑ | |||||||||||||||||||||||||
| HFD (60% fat) | |||||||||||||||||||||||||||
| Ojo et al. (2016) | HFD (60% fat) for 12 weeks and control diet (10% fat) as reference/mice model [238] | ↓ | ↓ | ||||||||||||||||||||||||
| Cowan et al. (2014) | HFD (60% fat) for 10 weeks and control diet (12% fat) as reference/rat model [224] | ↑ | ↑ | ↑ | Clostridium leptum | ||||||||||||||||||||||
| Mujico et al. (2013) | HFD (60% fat) for 19 weeks and maintenance diet (12% fat) as reference/mice model [225] | ↑ | ↓ | ||||||||||||||||||||||||
| Lam et al. (2012) | HFD (60% mixed fat) for 8, 12 weeks and control diet (10% fat) as reference/mice model [222] | ↑ | ↓ | ↓ | |||||||||||||||||||||||
| Kim et al. (2012) | HFD (60% fat) for 8 weeks and low-fat diet (10% fat) as reference/mice model [223] | ↑ | |||||||||||||||||||||||||
| HFD (72% fat) | |||||||||||||||||||||||||||
| Cani et al. (2008) | HFD (72% fat) for 4 weeks and standard A04 diet as reference/mice model [226] | ↑ | ↓ | ||||||||||||||||||||||||
| Ley et al. (2005) | HFD (72% fat) for 19 weeks and maintenance diet (12% fat) as reference/mice model [34] | ↑ | |||||||||||||||||||||||||
| Dose and Treatment Duration/Test Model | F/B | Potentially Beneficial Microbiota | Potentially Detrimental Microbiota | Species | Diversity | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bifidobacterium sp. | Lactobacillus sp. | Desulfovibrio sp. | Robinsoniella sp. | Barnesiella sp. | Allobaculum sp. | Akkermansia sp. | Roseburia sp. | Blautia sp. | Fecalibacterium sp. | Bacteroides sp. | Clostridium sp. | Enterorhabdus sp. | Clostridium sp. | Alloprevotella sp. | Haemophilus sp. | Ethanoligenens sp. | Klebsiella sp. | Porphyromonas sp. | Escherichia sp. | Turicibacter sp. | Staphylococcus sp. | Enterococcus sp. | |||||
| Blend protein | |||||||||||||||||||||||||||
| Moreno-Pérez et al. (2018) | Protein group (20 g/day) for 10 weeks and control group (no protein) as reference/clinical trials [247] | ↓ | ↓ | ↓ | No significant effect | ||||||||||||||||||||||
| Protein group (20 g//day) for 70 days and before protein treatment group as reference/clinical trials [247] | ↓ | ↓ | ↓ | No significant effect | |||||||||||||||||||||||
| Casein protein | |||||||||||||||||||||||||||
| Rist et al. (2014) | Casein-based group/CAS (85–335 g/kg) for 3 experimental periods (7 days/period) and Soybean meal-based group/SBM (85–335 g/kg) as reference/piglet model [243] | ↓ | ↓ | ↓ | |||||||||||||||||||||||
| Soybean protein | |||||||||||||||||||||||||||
| Rist et al. (2014) | SBM group (85–335 g/kg) for 3 experimental periods (7 days/period) and the CAS (85–335 g/kg) as reference/piglet model [243] | ↑ | |||||||||||||||||||||||||
| Crude protein | |||||||||||||||||||||||||||
| Kostovcikova et al. (2019) | High animal protein-based diet (514 g/kg) for 3 weeks and control group (176 g/kg) as reference/mice model [245] | ↑ | ↑ | ↑ | A. muciniphila | ↓ | |||||||||||||||||||||
| High animal protein-based diet (514 g/kg) for 3 weeks and baseline as reference/mice model [245] | ↑ | ↑ | ↑ | Escherichia coli | |||||||||||||||||||||||
| Animal protein-based control diet (176 g/kg) for 3 weeks and baseline as reference/mice model [245] | ↑ | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| High plant protein-based diet (500 g/kg) for 3 weeks and control group (173 g/kg) as reference/mice model [245] | ↑ | ↑ | Escherichia coli | ||||||||||||||||||||||||
| High plant protein-based diet (514 g/kg) for 3 weeks and baseline as reference/mice model [245] | ↑ | ↑ | Escherichia coli | ||||||||||||||||||||||||
| Plant protein-based control diet (176 g/kg) for 3 weeks and baseline as reference/mice model [245] | ↑ | ||||||||||||||||||||||||||
| Hang et al. (2012) | High protein group (crude protein 609 g/kg) for 28 days and dry commercial group (264 g/kg) as reference/dog model [254] | ↓ | |||||||||||||||||||||||||
| Lubbs et al. (2009) | High protein group (crude protein 50%) for 8 weeks and moderate-protein group (crude protein 30%) as reference/cat model [255] | ↓ | ↑ | C. perfringens | ↑ | ||||||||||||||||||||||
| Milk protein | |||||||||||||||||||||||||||
| Vidal-Lletjós et al. (2019) | Isocaloric diets with 53% protein for 3 days and the diets with 30% protein as reference/DDS-treated mice model [256] | ↓ | ↑ | ↑ | ↑ | ↑ | ↓ | ||||||||||||||||||||
| Isocaloric diets with 53% protein for 3 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↓ | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| Isocaloric diets with 30% protein for 3 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↓ | ↑ | |||||||||||||||||||||||||
| Isocaloric diets with 53% protein for 6 days and the diets with 30% protein as reference/DDS-treated mice model [256] | ↓ | ↓ | ↓ | Clostridium XI | ↓ | ||||||||||||||||||||||
| Isocaloric diets with 53% protein for 6 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↑ | ↑ | |||||||||||||||||||||||||
| Isocaloric diets with 30% protein for 6 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↑ | ||||||||||||||||||||||||||
| Isocaloric diets with 53% protein for 21 days and the diets with 30% protein as reference/DDS-treated mice model [256] | ↓ | ↓ | ↓ | ↑ | ↑ | ↑ | ↑ | ||||||||||||||||||||
| Isocaloric diets with 53% protein for 21 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↓ | ↑ | ↑ | ↑ | |||||||||||||||||||||||
| Isocaloric diets with 30% protein for 21 days and the diets with 14% protein as reference/DDS-treated mice model [256] | ↑ | ||||||||||||||||||||||||||
| Mung bean protein | |||||||||||||||||||||||||||
| Nakatani et al. (2018) | HFD-mung bean protein isolate (MPI) group (205.3 g MPI) for 4 weeks and HFD group (casein 200 g rather than MPI) as reference/mice model [242] | ↓ | ↓ | ||||||||||||||||||||||||
| Seafood protein | |||||||||||||||||||||||||||
| Holm et al. (2016) | Seafood western diet (exchange casein with seafood powder) for 12 weeks and meat western diet (exchange casein with lean meat powder) as reference/mice model [244] | ↓ | ↑ | ↑ | ↑ | ↓ | |||||||||||||||||||||
| Whey Protein | |||||||||||||||||||||||||||
| McAllan et al. (2014) | HFD-whey protein isolate (WPI) group (HFD with 20% WPI) for 21 weeks and HFD group as reference/mice model [241] | ↑ | ↑ | ↑ | ↓ | ||||||||||||||||||||||
| Fried meats | |||||||||||||||||||||||||||
| Shen et al. (2010) | Fried meats (fry without oil at 300℃ for 15 min, then ground, frozen, and freeze-dried) for fermentation over 48 h./in vitro, human fecal sample [248] | ↑ | Clostridium perfringens | ||||||||||||||||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Q.; Liang, Q.; Balakrishnan, B.; Belobrajdic, D.P.; Feng, Q.-J.; Zhang, W. Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients 2020, 12, 381. https://doi.org/10.3390/nu12020381
Yang Q, Liang Q, Balakrishnan B, Belobrajdic DP, Feng Q-J, Zhang W. Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients. 2020; 12(2):381. https://doi.org/10.3390/nu12020381
Chicago/Turabian StyleYang, Qi, Qi Liang, Biju Balakrishnan, Damien P Belobrajdic, Qian-Jin Feng, and Wei Zhang. 2020. "Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review" Nutrients 12, no. 2: 381. https://doi.org/10.3390/nu12020381
APA StyleYang, Q., Liang, Q., Balakrishnan, B., Belobrajdic, D. P., Feng, Q.-J., & Zhang, W. (2020). Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients, 12(2), 381. https://doi.org/10.3390/nu12020381





