Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABla-12 Improve Abdominal Pain Severity and Symptomology in Irritable Bowel Syndrome: Randomized Controlled Trial
Abstract
1. Introduction
2. Subjects and Methods
2.1. Study Population
2.2. Study Design
2.3. Study Product
2.4. Outcome Assessments
2.5. Statistical Analysis
3. Results
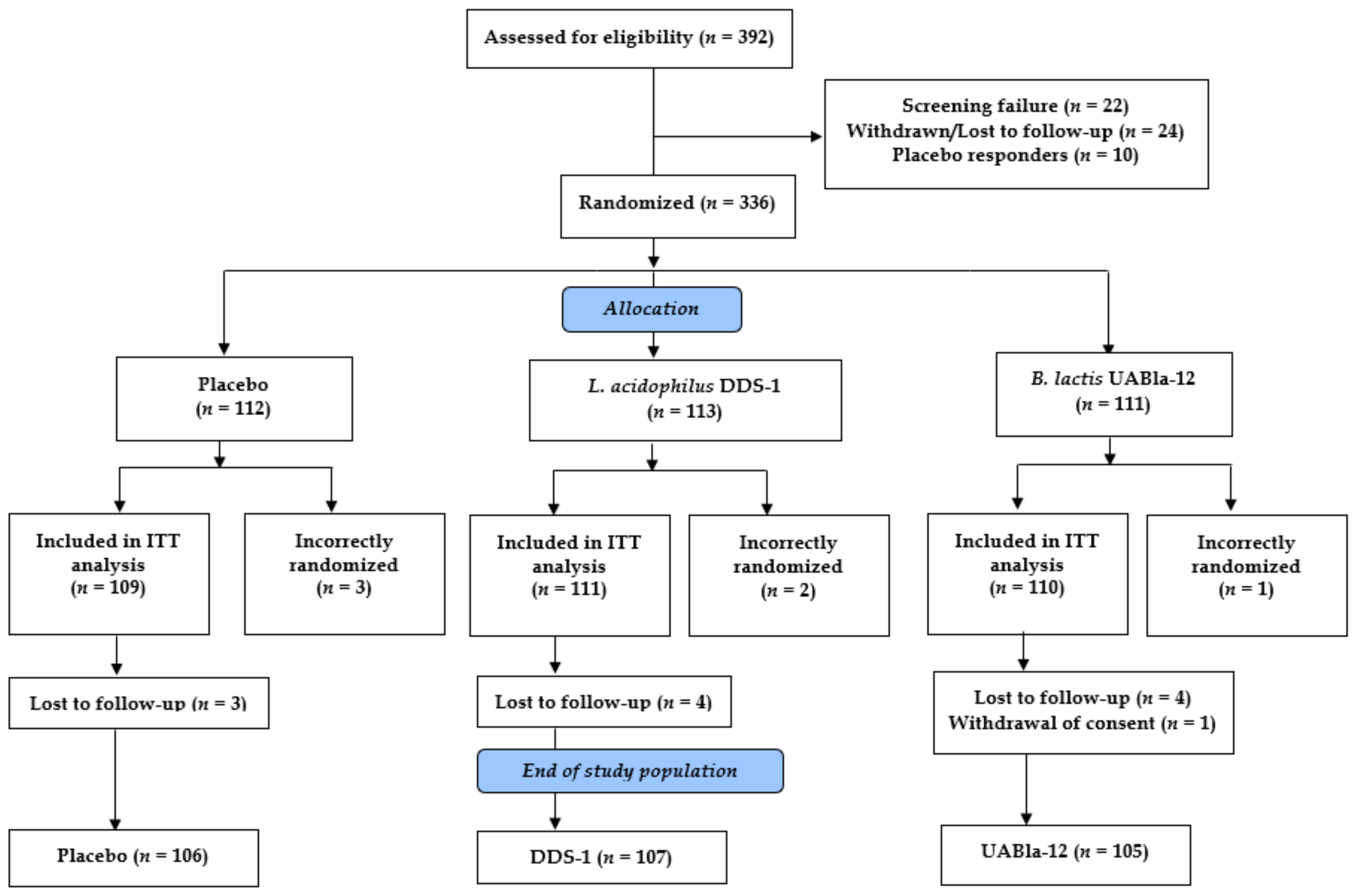
3.1. Study Parameters
3.2. Baseline Characteristics of Subjects
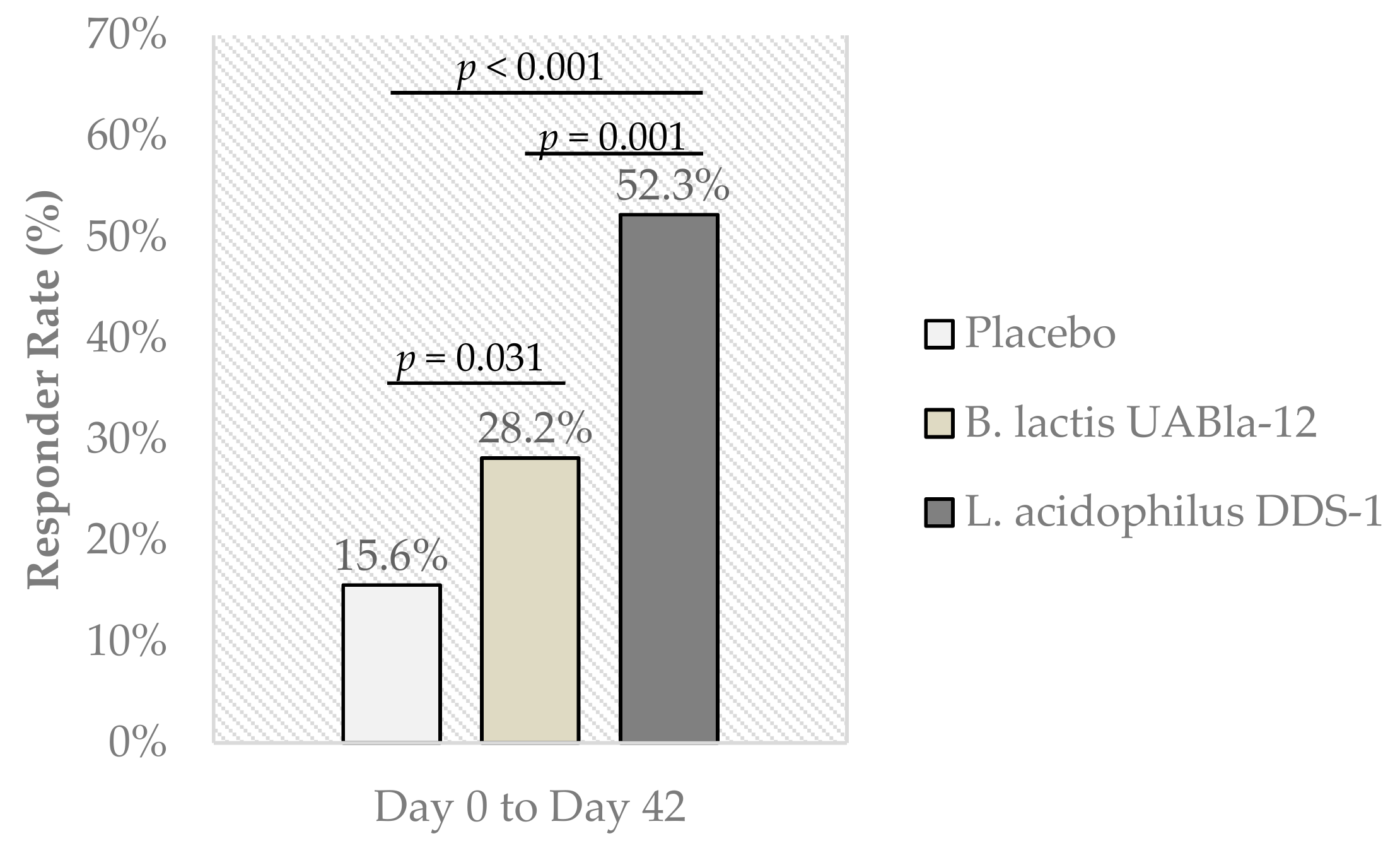
3.3. Abdominal Pain Severity
3.4. IBS Symptomology
3.5. Bowel Habits
3.6. Quality of Life, Perceived Stress and Product Tolerability
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holtmann, G.J.; Ford, A.C.; Talley, N.J. Pathophysiology of irritable bowel syndrome. Lancet Gastroenterol. Hepatol. 2016, 1, 133–146. [Google Scholar] [CrossRef]
- Barbara, G.; Feinle-Bisset, C.; Ghoshal, U.C.; Santos, J.; Vanner, S.J.; Vergnolle, N.; Zoetendal, E.G.; Quigley, E.M. The intestinal microenvironment and functional gastrointestinal disorders. Gastroenterology 2016, 150, 1305–1318. [Google Scholar] [CrossRef]
- Dale, H.F.; Rasmussen, S.H.; Asiller, Ö.Ö.; Lied, G.A. Probiotics in Irritable Bowel Syndrome: An Up-to-Date Systematic Review. Nutrients 2019, 11, 2048. [Google Scholar] [CrossRef]
- Lacy, B.; Patel, N. Rome Criteria and a Diagnostic Approach to Irritable Bowel Syndrome. J. Clin. Med. 2017, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Moayyedi, P.; Chey, W.D.; Harris, L.A.; Lacy, B.E.; Saito, Y.A.; Quigley, E.M.M. ACG Task Force on Management of Irritable Bowel Syndrome American College of Gastroenterology Monograph on Management of Irritable Bowel Syndrome. Am. J. Gastroenterol. 2018, 113, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Barbara, G.; Cremon, C.; Azpiroz, F. Probiotics in irritable bowel syndrome: Where are we? Neurogastroenterol. Motil. 2018, 30, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Harris, L.A.; Lacy, B.E.; Quigley, E.M.M.; Moayyedi, P. Systematic review with meta-analysis: The efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2018, 48, 1044–1060. [Google Scholar] [CrossRef] [PubMed]
- Didari, T.; Mozaffari, S.; Nikfar, S.; Abdollahi, M. Effectiveness of probiotics in irritable bowel syndrome: Updated systematic review with meta-analysis. World J. Gastroenterol. 2015, 21, 3072–3084. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Guo, C.; Mu, D.; Feng, B.; Zuo, X.; Li, Y. Effects of probiotic type, dose and treatment duration on irritable bowel syndrome diagnosed by Rome III criteria: A meta-analysis. BMC Gastroenterol. 2016, 16, 1–11. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Ford, A.C.; Moayyedi, P.; Lacy, B.E.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.R.; Quigley, E.M.M. American college of gastroenterology monograph on the management of irritable bowel syndrome and chronic idiopathic constipation. Am. J. Gastroenterol. 2014, 109, S2–S26. [Google Scholar] [CrossRef]
- Martoni, C.J.; Evans, M.; Chow, C.T.; Chan, L.S.; Leyer, G. Impact of a probiotic product on bowel habits and microbial profile in participants with functional constipation: A randomized controlled trial. J. Dig. Dis. 2019, 20, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Pakdaman, M.N.; Udani, J.K.; Molina, J.P.; Shahani, M. The effects of the DDS-1 strain of lactobacillus on symptomatic relief for lactose intolerance - A randomized, double-blind, placebo-controlled, crossover clinical trial. Nutr. J. 2016, 15. [Google Scholar] [CrossRef] [PubMed]
- Gerasimov, S.V.; Ivantsiv, V.A.; Bobryk, L.M.; Tsitsura, O.O.; Dedyshin, L.P.; Guta, N.V.; Yandyo, B.V. Role of short-term use of L. acidophilus DDS-1 and B. lactis UABLA-12 in acute respiratory infections in children: A randomized controlled trial. Eur. J. Clin. Nutr. 2016, 70, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Gerasimov, S.V.; Vasjuta, V.V.; Myhovych, O.O.; Bondarchuk, L.I. Probiotic supplement reduces Atopic Dermatitis in preschool children: A randomized, double-blind, placebo-controlled, clinical trial. Am. J. Clin. Dermatol. 2010, 11, 351–361. [Google Scholar] [CrossRef]
- Vemuri, R.; Shinde, T.; Shastri, M.D.; Perera, A.P.; Tristram, S.; Martoni, C.J.; Gundamaraju, R.; Ahuja, K.D.K.; Ball, M.; Eri, R. A human origin strain Lactobacillus acidophilus DDS-1 exhibits superior in vitro probiotic efficacy in comparison to plant or dairy origin probiotics. Int. J. Med. Sci. 2018, 15, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Vemuri, R.; Shinde, T.; Gundamaraju, R.; Gondalia, S.V.; Karpe, A.V.; Beale, D.J.; Martoni, C.J.; Eri, R. Lactobacillus acidophilus DDS-1 modulates the gut microbiota and improves metabolic profiles in aging mice. Nutrients 2018, 10, 1255. [Google Scholar] [CrossRef] [PubMed]
- Vemuri, R.; Gundamaraju, R.; Shinde, T.; Perera, A.P.; Basheer, W.; Southam, B.; Gondalia, S.V.; Karpe, A.V.; Beale, D.J.; Tristram, S.; et al. Lactobacillus acidophilus DDS-1 modulates intestinal-specific microbiota, short-chain fatty acid and immunological profiles in aging mice. Nutrients 2019, 11, 1297. [Google Scholar] [CrossRef] [PubMed]
- Nagala, R.; Routray, C. Clinical Case Study—Multispecies probiotic supplement minimizes symptoms of irritable bowel syndrome. US Gastroenterol. Hepatol. Rev. 2011, 7, 1–2. [Google Scholar]
- Spiegel, B.M.R.; Bolus, R.; Harris, L.A.; Lucak, S.; Chey, W.D.; Sayuk, G.; Esrailian, E.; Lembo, A.; Karsan, H.; Tillisch, K.; et al. Characterizing abdominal pain in IBS: Guidance for study inclusion criteria, outcome measurement and clinical practice. Aliment. Pharmacol. Ther. 2010, 32, 1192–1202. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, B.; Bolus, R.; Harris, L.A.; Lucak, S.; Naliboff, B.; Esrailian, E.; Chey, W.D.; Lembo, A.; Karsan, H.; Tillisch, K.; et al. Measuring IBS patient reported outcomes with an abdominal pain numeric rating scale: Results from the proof cohort. Aliment. Pharmacol. Ther. 2009, 30, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, B.M.R.; Bolus, R.; Agarwal, N.; Sayuk, G.; Harris, L.A.; Lucak, S.; Esrailian, E.; Chey, W.D.; Lembo, A.; Karsan, H.; et al. Measuring symptoms in the irritable bowel syndrome: Development of a framework for clinical trials. Aliment. Pharmacol. Ther. 2010, 32, 1275–1291. [Google Scholar] [CrossRef] [PubMed]
- Francis, C.Y.; Morris, J.; Whorwell, P.J. The irritable bowel severity scoring system: A simple method of monitoring irritable bowel syndrome and its progress. Aliment. Pharmacol. Ther. 1997, 11, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Blake, M.R.; Raker, J.M.; Whelan, K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment. Pharmacol. Ther. 2016, 44, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Patrick, D.L.; Drossman, D.A.; Frederick, I.O.; Dicesare, J.; Puder, K.L. Quality of life in persons with irritable bowel syndrome: Development and validation of a new measure. Dig. Dis. Sci. 1998, 43, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Williamson, G. Perceived stress in a probability sample of the United States. Soc. Psychol. Heal. 1988, 31–67. [Google Scholar]
- Ghoshal, U.C.; Abraham, P.; Bhatt, C.; Choudhuri, G.; Bhatia, S.J.; Shenoy, K.T.; Banka, N.H.; Bose, K.; Bohidar, N.P.; Chakravartty, K.; et al. Epidemiological and clinical profile of irritable bowel syndrome in India: Report of the Indian Society of Gastroenterology Task Force. Indian J. Gastroenterol. 2008, 27, 22–28. [Google Scholar]
- Masudur Rahman, M.; Mahadeva, S.; Ghoshal, U.C. Epidemiological and clinical perspectives on irritable bowel syndrome in India, Bangladesh and Malaysia: A review. World J. Gastroenterol. 2017, 23, 6788–6801. [Google Scholar] [CrossRef]
- Food and Drug Administration. Guidance for Industry: Irritable Bowel Syndrome-Clinical Evaluation of Drugs for Treatment; Docket No. FDA-2012-D-0146; Food and Drug Administration: Silver Spring, MD, USA, 2012; pp. 32124–32125.
- Ford, A.C.; Moayyedi, P. Meta-analysis: Factors affecting placebo response rate in the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2010, 32, 144–158. [Google Scholar] [CrossRef] [PubMed]
- Spiller, R.C. Problems and challenges in the design of irritable bowel syndrome clinical trials: Experience from published trials. Am. J. Med. 1999, 107, 91S–97S. [Google Scholar] [CrossRef]
- Mangel, A.W. Study design issues in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2004, 19, 141–142. [Google Scholar] [CrossRef]
- Miller, L.E. Study design considerations for irritable bowel syndrome clinical trials. Ann. Gastroenterol. Q. Publ. Hell. Soc. Gastroenterol. 2014, 27, 338–345. [Google Scholar]
- Dorn, S.D.; Kaptchuk, T.J.; Park, J.B.; Nguyen, L.T.; Canenguez, K.; Nam, B.H.; Woods, K.B.; Conboy, L.A.; Stason, W.B.; Lembo, A.J. A meta-analysis of the placebo response in complementary and alternative medicine trials of irritable bowel syndrome. Neurogastroenterol. Motil. 2007, 19, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Snapinn, S.M.; Jiang, Q. Responder analyses and the assessment of a clinically relevant treatment effect. Trials 2007, 8, 31. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Guidance on the scientific requirements for health claims related to gut and immune function. EFSA J. 2011, 9, 1984. [Google Scholar] [CrossRef]
- Upadhyay, R.; Singh, A. Irritable Bowel Syndrome: The Indian Scenario. Med. Updat. 2013, 255–258. [Google Scholar]
- Chong, P.P.; Chin, V.K.; Looi, C.Y.; Wong, W.F.; Madhavan, P.; Yong, V.C. The microbiome and irritable bowel syndrome - A review on the pathophysiology, current research and future therapy. Front. Microbiol. 2019, 10, 1–23. [Google Scholar]
- Distrutti, E.; Monaldi, L.; Ricci, P.; Fiorucci, S. Gut microbiota role in irritable bowel syndrome: New therapeutic strategies. World J. Gastroenterol. 2016, 22, 2219–2241. [Google Scholar] [CrossRef]
- Bellini, M.; Gambaccini, D.; Stasi, C.; Urbano, M.T.; Marchi, S.; Usai-Satta, P. Irritable bowel syndrome: A disease still searching for pathogenesis, diagnosis and therapy. World J. Gastroenterol. 2014, 20, 8807–8820. [Google Scholar]
- Simren, M.; Palsson, O.S.; Whitehead, W.E. Update on Rome IV Criteria for Colorectal Disorders: Implications for Clinical Practice. Curr. Gastroenterol. Rep. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, V.S.; Chen, W.; Shu, D.; Paulus, B.; Bethwaite, P.; Tie, A.; Wilson, I. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology 2002, 122, 1778–1783. [Google Scholar] [CrossRef]
- Guilarte, M.; Santos, J.; De Torres, I.; Alonso, C.; Vicario, M.; Ramos, L.; Martinez, C.; Casellas, F.; Saperas, E.; Malagelada, J.R. Diarrhoea-predominant IBS patients show mast cell activation and hyperplasia in the jejunum. Gut 2007, 56, 203–209. [Google Scholar] [CrossRef]
- Lomax, A.E.; Pradhananga, S.; Sessenwein, J.L.; O’Malley, D. Bacterial modulation of visceral sensation: Mediators and mechanisms. Am. J. Physiol. Liver Physiol. 2019, 317, G363–G372. [Google Scholar] [CrossRef] [PubMed]
- Rousseaux, C.; Thuru, X.; Gelot, A.; Barnich, N.; Neut, C.; Dubuquoy, L.; Dubuquoy, C.; Merour, E.; Geboes, K.; Chamaillard, M.; et al. Lactobacillus acidophilus modulates intestinal pain and induces opioid and cannabinoid receptors. Nat. Med. 2007, 13, 35–37. [Google Scholar] [CrossRef] [PubMed]
- Ringel-Kulka, T.; Palsson, O.S.; Maier, D.; Carroll, I.; Galanko, J.A.; Leyer, G.; Ringel, Y. Probiotic bacteria Lactobacillus acidophilus NCFM and Bifidobacterium lactis Bi-07 versus placebo for the symptoms of bloating in patients with functional bowel disorders: A double-blind study. J. Clin. Gastroenterol. 2011, 45, 518–525. [Google Scholar] [CrossRef]
- Ringel-Kulka, T.; Goldsmith, J.R.; Carroll, I.M.; Barros, S.P.; Palsson, O.; Jobin, C.; Ringel, Y. Lactobacillus acidophilus NCFM affects colonic mucosal opioid receptor expression in patients with functional abdominal pain - A randomised clinical study. Aliment. Pharmacol. Ther. 2014, 40, 200–207. [Google Scholar] [CrossRef]
- Ranuh, R.; Athiyyah, A.F.; Darma, A.; Risky, V.P.; Riawan, W.; Surono, I.S.; Sudarmo, S.M. Effect of the probiotic lactobacillus plantarum is-10506 on bdnf and 5ht stimulation: Role of intestinal microbiota on the gut-brain axis. Iran. J. Microbiol. 2019, 11, 145–150. [Google Scholar] [CrossRef]
- Qin, H.Y.; Cheng, C.W.; Tang, X.D.; Bian, Z.X. Impact of psychological stress on irritable bowel syndrome. World J. Gastroenterol. 2014, 20, 14126–14131. [Google Scholar] [CrossRef]
- Dhakan, D.B.; Maji, A.; Sharma, A.K.; Saxena, R.; Pulikkan, J.; Grace, T.; Gomez, A.; Scaria, J.; Amato, K.R.; Sharma, V.K. The unique composition of Indian gut microbiome, gene catalogue, and associated fecal metabolome deciphered using multi-omics approaches. Gigascience 2019, 8, 1–20. [Google Scholar] [CrossRef] [PubMed]
| Placebo (n = 109) | L. acidophilus DDS-1 (n = 111) | B. lactis UABla-12 (n = 110) | p Value | |
|---|---|---|---|---|
| Mean (SD) or n (%) | Mean (SD) or n (%) | Mean (SD) or n (%) | ||
| Age (years) | 37.61 (10.12) | 39.41 (11.80) | 41.60 (11.11) | 0.028 ‡ |
| Female gender, n (%) | 54 (49.5) | 58 (52.3) | 51 (46.6) | 0.681 † |
| BMI (kg/m2) | 24.10 (4.06) | 24.09 (4.34) | 23.78 (3.98) | 0.811 ‡ |
| Systolic BP (kPa) | 15.89 (1.13) | 15.96 (1.08) | 16.03 (1.11) | 0.660 § |
| Diastolic BP (kPa) | 10.09 (0.94) | 10.33 (0.86) | 10.25 (1.00) | 0.116 § |
| Pulse Rate (bpm) | 78.23 (8.63) | 76.96 (7.69) | 78.18 (8.10) | 0.426 ‡ |
| IBS duration (months) | 20.81 (10.92) | 20.85 (12.29) | 22.07 (12.80) | 0.674 ‡ |
| GAD score | 3.88 (1.81) | 4.04 (1.62) | 4.16 (1.64) | 0.378 § |
| PHQ-9 score | 5.38 (3.15) | 5.61 (3.05) | 5.32 (2.96) | 0.749 ‡ |
| Alcohol consumption | ||||
| None, n (%) | 96 (88.1%) | 100 (90.1%) | 97 (88.2%) | |
| Occasional, n (%) | 3 (2.8%) | 2 (1.8%) | 3 (2.7%) | 0.983 † |
| Not available, n (%) | 10 (9.2%) | 9 (8.1%) | 10 (9.1%) |
| Mean (SD) | p Value † | |||||
|---|---|---|---|---|---|---|
| Placebo (n = 109) | L. acidophilus DDS-1 (n = 111) | B. lactis UABla-12 (n = 110) | DDS-1 vs. Placebo | UABla-12 vs. Placebo | DDS-1 vs. UABla-12 | |
| APS-NRS Score | ||||||
| Day 0 | 6.94 (1.02) | 7.03 (0.99) | 6.84 (1.04) | 0.499 | 0.412 | 0.155 |
| AbsΔ (Day 21) | −0.71 (0.92) | −1.24 (1.10) | −0.88 (0.99) | < 0.001 | 0.178 | 0.012 |
| AbsΔ (Day 42) | −0.85 (1.45) | −2.59 (2.07) | −1.56 (1.83) | 0.001 | 0.001 | < 0.001 |
| Mean (SD) | p Value † | |||||
|---|---|---|---|---|---|---|
| Placebo (n = 109) | L. acidophilus DDS-1 (n = 111) | B. lactis UABla-12 (n = 110) | DDS-1 vs. Placebo | UABla-12 vs. Placebo | DDS-1 vs. UABla-12 | |
| IBS-SSS Total Score | ||||||
| Day 0 | 298.07 (55.68) | 310.90 (52.47) | 305.45 (48.82) | 0.108 | 0.324 | 0.504 |
| AbsΔ (Day 21) | −30.09 (57.76) | −56.06 (57.64) | −50.00 (63.72) | < 0.001 | 0.020 | 0.185 |
| AbsΔ (Day 42) | −55.70 (86.42) | −133.4 (95.19) | −104.5 (96.08) | < 0.001 | < 0.001 | 0.039 |
| Abdominal Pain Severity | ||||||
| Day 0 | 66.74 (11.39) | 68.24 (11.73) | 65.82 (11.30) | 0.213 | 0.644 | 0.075 |
| AbsΔ (Day 21) | −9.25 (15.09) | −15.50 (15.23) | −12.86 (16.36) | 0.001 | 0.077 | 0.121 |
| AbsΔ (Day 42) | −13.41 (20.17) | −32.94 (21.78) | −24.52 (24.35) | < 0.001 | < 0.001 | 0.009 |
| Abdominal Pain Duration | ||||||
| Day 0 | 53.21 (15.69) | 59.73 (18.85) | 57.55 (17.57) | 0.016 | 0.139 | 0.344 |
| AbsΔ (Day 21) | −4.77 (17.72) | −13.49 (21.79) | −10.95 (19.83) | 0.002 | 0.023 | 0.420 |
| AbsΔ (Day 42) | −8.13 (19.53) | −28.44 (24.80) | −20.95 (23.19) | < 0.001 | < 0.001 | 0.018 |
| Abdominal Distension | ||||||
| Day 0 | 58.39 (17.81) | 60.14 (16.96) | 58.23 (17.79) | 0.428 | 0.822 | 0.247 |
| AbsΔ (Day 21) | −7.66 (20.39) | −11.47 (16.26) | −9.67 (18.92) | 0.110 | 0.433 | 0.396 |
| AbsΔ (Day 42) | −15.47 (23.81) | −27.20 (25.15) | −21.90 (25.85) | < 0.001 | 0.034 | 0.098 |
| Bowel Habits | ||||||
| Day 0 | 60.05 (14.38) | 63.06 (13.79) | 63.09 (12.06) | 0.122 | 0.119 | 0.974 |
| AbsΔ (Day 21) | −4.11 (13.95) | −8.90 (13.82) | −8.86 (14.99) | 0.007 | 0.010 | 0.999 |
| AbsΔ (Day 42) | −7.99 (23.46) | −22.89 (22.46) | −17.86 (22.06) | < 0.001 | 0.001 | 0.067 |
| Effect on Quality of Life | ||||||
| Day 0 | 59.68 (14.57) | 59.73 (13.70) | 60.77 (11.62) | 0.985 | 0.657 | 0.496 |
| AbsΔ (Day 21) | −4.30 (15.76) | −6.70 (14.58) | −7.67 (15.61) | 0.043 | 0.112 | 0.663 |
| AbsΔ (Day 42) | −10.70 (21.98) | −21.88 (22.27) | −19.29 (21.66) | < 0.001 | 0.001 | 0.316 |
| Subjects (n, %) | ||||
|---|---|---|---|---|
| Group | Stool type (BSS) | Day 0 | Day 21 | Day 42 |
| Placebo (n = 109) | Constipation (1–2) | 25 (22.9) | 19 (17.4) | 28 (25.7) |
| Normal (3–5) | 70 (64.2) | 74 (67.9) | 68 (62.4) | |
| Diarrhea (6–7) | 14 (12.8) | 14 (12.8) | 11 (10.1) | |
| Data not available | 0 | 2 (1.8) | 2 (1.8) | |
| L. acidophilus DDS-1 (n = 111) | Constipation (1–2) | 24 (21.6) | 14 (12.6) | 9 (8.1) |
| Normal (3–5) | 63 (56.8) | 82 (73.9) | 93 (83.8) | |
| Diarrhea (6–7) | 24 (21.6) | 13 (11.7) | 7 (6.3) | |
| Data not available | 0 | 2 (1.8) | 2 (1.8) | |
| B. lactis UABla-12 (n = 110) | Constipation (1–2) | 22 (20.0) | 18 (16.4) | 10 (9.1) |
| Normal (3–5) | 67 (60.9) | 63 (57.3) | 83 (75.5) | |
| Diarrhea (6–7) | 21 (19.1) | 24 (21.8) | 12 (10.9) | |
| Data not available | 0 | 5 (4.5) | 5 (4.5) | |
| p Value † | DDS-1 vs. Placebo | 0.234 | 0.733 | 0.002 |
| UABla-12 vs. Placebo | 0.438 | 0.444 | 0.022 | |
| DDS-1 vs. UABla-12 | 0.139 | 0.234 | 0.435 | |
| Subjects (n, %) | p (Within Group) † | ||||
|---|---|---|---|---|---|
| Group | Change in Stool Type | Day 0 to 21 | Day 0 to 42 | Day 0 to 21 | Day 0 to 42 |
| Placebo (n = 107) | Non-normal to Normal | 21 (19.6) | 21 (19.6) | 0.511 | 1.000 |
| Non-normal to Non-normal | 17 (15.9) | 17 (15.9) | |||
| Normal to Non-Normal | 16 (15.0) | 22 (20.6) | |||
| Normal to Normal | 53 (49.5) | 47 (43.9) | |||
| L. acidophilus DDS-1 (n = 109) | Non-normal to Normal | 31 (28.4) | 38 (34.9) | 0.006 | < 0.001 |
| Non-normal to Non-normal | 15 (13.8) | 8 (7.3) | |||
| Normal to Non-Normal | 12 (11.0) | 8 (7.3) | |||
| Normal to Normal | 51 (46.8) | 55 (50.5) | |||
| B. lactis UABla-12 (n = 105) | Non-normal to Normal | 16 (15.2) | 28 (26.7) | 1.000 | 0.002 |
| Non-normal to Non-normal | 26 (24.8) | 14 (13.3) | |||
| Normal to Non-Normal | 16 (15.2) | 8 (7.6) | |||
| Normal to Normal | 47 (44.8) | 55 (52.4) | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martoni, C.J.; Srivastava, S.; Leyer, G.J. Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABla-12 Improve Abdominal Pain Severity and Symptomology in Irritable Bowel Syndrome: Randomized Controlled Trial. Nutrients 2020, 12, 363. https://doi.org/10.3390/nu12020363
Martoni CJ, Srivastava S, Leyer GJ. Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABla-12 Improve Abdominal Pain Severity and Symptomology in Irritable Bowel Syndrome: Randomized Controlled Trial. Nutrients. 2020; 12(2):363. https://doi.org/10.3390/nu12020363
Chicago/Turabian StyleMartoni, Christopher J., Shalini Srivastava, and Gregory J. Leyer. 2020. "Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABla-12 Improve Abdominal Pain Severity and Symptomology in Irritable Bowel Syndrome: Randomized Controlled Trial" Nutrients 12, no. 2: 363. https://doi.org/10.3390/nu12020363
APA StyleMartoni, C. J., Srivastava, S., & Leyer, G. J. (2020). Lactobacillus acidophilus DDS-1 and Bifidobacterium lactis UABla-12 Improve Abdominal Pain Severity and Symptomology in Irritable Bowel Syndrome: Randomized Controlled Trial. Nutrients, 12(2), 363. https://doi.org/10.3390/nu12020363





