Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function
Abstract
1. Introduction
2. Determination of Protein Quality
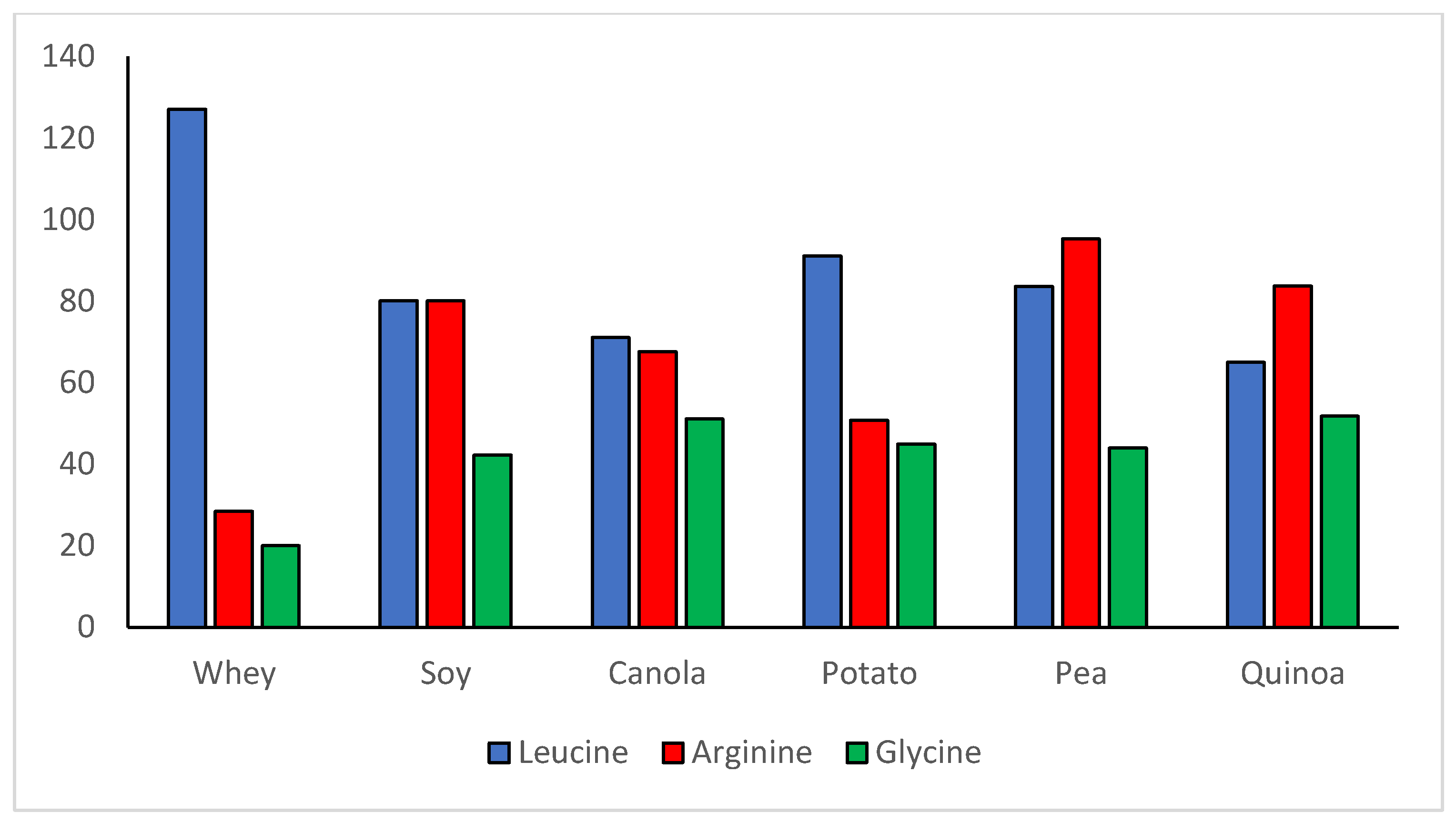
3. The Quality of Plant Proteins
4. Importance of Plant Proteins in Health
4.1. Plant Protein and Cardiovascular Disease and Metabolic Risk Factors
4.2. Plant Protein and Diabetes
4.3. Plant Protein Intake and Incidence of Cancer
4.4. Plant Proteins as Functional Foods
4.5. Plant Protein Intake and Its Relationship to Mortality
4.6. Renoprotective Effect of Plant Proteins
4.7. Plant Proteins for Lean Body Mass and Strength
5. Health Concerns Associated with Plant Proteins
5.1. Antinutrients
5.2. Soy Protein and Isoflavones
5.3. Plant-Based Protein and Allergenicity
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Henchion, M.; Hayes, M.; Mullen, A.M.; Fenelon, M.; Tiwari, B. Future Protein Supply and Demand: Strategies and Factors Influencing a Sustainable Equilibrium. Foods 2017, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Lee, J.S.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The Health, Aging, and Body Composition (Health ABC) study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.L.; Wang, Y.; Bergia Iii, R.E.; Campbell, W.W. Protein Intake Greater than the RDA Differentially Influences Whole-Body Lean Mass Responses to Purposeful Catabolic and Anabolic Stressors: A Systematic Review and Meta-analysis. Adv. Nutr. 2020, 11, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.J.; Milan, A.M.; Mitchell, S.M.; Zeng, N.; Ramzan, F.; Sharma, P.; Knowles, S.O.; Roy, N.C.; Sjodin, A.; Wagner, K.H.; et al. The effects of dietary protein intake on appendicular lean mass and muscle function in elderly men: A 10-wk randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, S.Y.; McGlory, C.; D’Souza, L.K.; Morgan, A.K.; Saddler, N.I.; Baker, S.K.; Parise, G.; Phillips, S.M. A randomized controlled trial of the impact of protein supplementation on leg lean mass and integrated muscle protein synthesis during inactivity and energy restriction in older persons. Am. J. Clin. Nutr. 2018, 108, 1060–1068. [Google Scholar] [CrossRef]
- Park, Y.; Choi, J.E.; Hwang, H.S. Protein supplementation improves muscle mass and physical performance in undernourished prefrail and frail elderly subjects: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2018, 108, 1026–1033. [Google Scholar] [CrossRef]
- Kerstetter, J.E.; Looker, A.C.; Insogna, K.L. Low dietary protein and low bone density. Calcif. Tissue Int. 2000, 66, 313. [Google Scholar] [CrossRef]
- Hannan, M.T.; Tucker, K.L.; Dawson-Hughes, B.; Cupples, L.A.; Felson, D.T.; Kiel, D.P. Effect of dietary protein on bone loss in elderly men and women: The Framingham Osteoporosis Study. J. Bone Min. Res. 2000, 15, 2504–2512. [Google Scholar] [CrossRef]
- Rapuri, P.B.; Gallagher, J.C.; Haynatzka, V. Protein intake: Effects on bone mineral density and the rate of bone loss in elderly women. Am. J. Clin. Nutr. 2003, 77, 1517–1525. [Google Scholar] [CrossRef]
- Pimentel, D.; Pimentel, M. Sustainability of meat-based and plant-based diets and the environment. Am. J. Clin. Nutr. 2003, 78, 660S–663S. [Google Scholar] [CrossRef]
- Institute of Medicine of the National Academies. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; National Academies Press: Washington, DC, USA, 2005. [Google Scholar]
- Boye, J.; Wijesinha-Bettoni, R.; Burlingame, B. Protein quality evaluation twenty years after the introduction of the protein digestibility corrected amino acid score method. Br. J. Nutr. 2012, 108 (Suppl. 2), S183–S211. [Google Scholar] [CrossRef] [PubMed]
- Food and Agricultural Organization of the United Nations. FAO Food and Nutrition Paper 51: Protein Quality Evaluation: Report of a Joint FAO/WHO Expert Consultation; FAO: Rome, Italy, 1991; pp. 1–66. [Google Scholar]
- Darragh, A.J.; Hodgkinson, S.M. Quantifying the digestibility of dietary protein. J. Nutr. 2000, 130, 1850S–1856S. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G. The protein digestibility-corrected amino acid score. J. Nutr. 2000, 130, 1865S–1867S. [Google Scholar] [CrossRef] [PubMed]
- Food and Agricultural Organization of the United Nations. FAO Food and Nutrition Paper 92: Dietary Protein Quality Evaluation in Human Nutrition: Report of an FAO Expert Consultation; FAO: Rome, Italy, 2013; pp. 1–66. [Google Scholar]
- Hoffman, J.R.; Falvo, M.J. Protein-which is best? J. Sports Sci. Med. 2004, 3, 118–130. [Google Scholar] [PubMed]
- U.S. Dairy Export Council. Reference Manual for U.S. Milk Powders 2005 Revised Edition. Available online: file:///C:/Users/hertzsr/Downloads/Milk_Powder_Reference_Manual_Complete1.pdf (accessed on 11 July 2020).
- Young, V.R.; Pellet, P.L. Plant proteins in relation to human protein and amino acid nutrition. Am. J. Clin. Nutr. 1994, 59, 1203S–1212S. [Google Scholar] [CrossRef]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef]
- Rutherfurd, S.M.; Fanning, A.C.; Miller, B.J.; Moughan, P.J. Protein digestibility-corrected amino acid scores and digestible indispensable amino acid scores differentially describe protein quality in growing male rats. J. Nutr. 2015, 145, 372–379. [Google Scholar] [CrossRef]
- Moughan, P.J.; Gilani, S.; Rutherfurd, S.M.; Tome, D. Report of a Sub-comittee of the 2011 FAO Consulation on "Protein Quality Evaluation in Human Nutrition" on: The Assessment of Amino Acid Digestibility in Foods for Humans and Including a Collation of Published Ileal Amino acid Digestibility Data for Human Foods; FAO: Rome, Italy, 2013. [Google Scholar]
- Etzel, M.R. Manufacture and use of dairy protein fractions. J. Nutr. 2004, 134, 996S–1002S. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Appendix B. True protein digestibility value of common foods. Fed. Regist. 1993, 58, 2193–2195. [Google Scholar]
- Hughes, G.J.; Ryan, D.J.; Mukherjea, R.; Schasteen, C.S. Protein digestibility-corrected amino acid scores (PDCAAS) for soy protein isolates and concentrate: Criteria for evaluation. J. Agric. Food Chem. 2011, 59, 12707–12712. [Google Scholar] [CrossRef]
- Fleddermann, M.; Fechner, A.; Rossler, A.; Bahr, M.; Pastor, A.; Liebert, F.; Jahreis, G. Nutritional evaluation of rapeseed protein compared to soy protein for quality, plasma amino acids, and nitrogen balance--a randomized cross-over intervention study in humans. Clin. Nutr. 2013, 32, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D. DSM CanolaPRO. Available online: https://www.globalfoodforums.com/wp-content/uploads/2017/04/DSM-CanolaPRO-Clean-Label-2017-POSTED-4.4.17.pdf (accessed on 14 July 2020).
- Oikawa, S.Y.; Bahniwal, R.; Holloway, T.M.; Lim, C.; McLeod, J.C.; McGlory, C.; Baker, S.K.; Phillips, S.M. Potato Protein Isolate Stimulates Muscle Protein Synthesis at Rest and with Resistance Exercise in Young Women. Nutrients 2020, 12, 1235. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Spelbrink, R.E.; Witteman, B.J.; Giuseppin, M.L. Digestion kinetics of potato protein isolates in vitro and in vivo. Int. J. Food Sci. Nutr. 2013, 64, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Hughes, B.P. The amino-acid composition of potato protein and of cooked potato. Br. J. Nutr. 1958, 12, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Kowalczewski, P.L.; Olejnik, A.; Bialas, W.; Rybicka, I.; Zielinska-Dawidziak, M.; Siger, A.; Kubiak, P.; Lewandowicz, G. The Nutritional Value and Biological Activity of Concentrated Protein Fraction of Potato Juice. Nutrients 2019, 11, 1523. [Google Scholar] [CrossRef] [PubMed]
- Kaldy, M.S.; Markakis, P. Amino acid composition of selected potato varieties. J. Food Sci. 1972, 37, 375–377. [Google Scholar] [CrossRef]
- Yang, H.; Guerin-Deremaux, L.; Zhou, L.; Fratus, A.; Wils, D.; Zhang, C.; Zhang, K.; Miller, L.E. Evaluation of nutritional quality of a novel pea protein. Food Anal. 2012, 23, 8–10. [Google Scholar]
- Nowak, V.; Du, J.; Charrondiere, U.R. Assessment of the nutritional composition of quinoa (Chenopodium quinoa Willd.). Food Chem. 2016, 193, 47–54. [Google Scholar] [CrossRef]
- Repo-Carrasco, R.; Espinoza, C.; Jacobsen, S.E. Nutritional Value and Use of the Andean Crops Quinoa (Chenopodium quinoa) and Kañiwa (Chenopodium pallidicaule). Food Rev. Int. 2003, 19, 179–189. [Google Scholar] [CrossRef]
- Mota, C.; Santos, M.; Mauro, R.; Samman, N.; Matos, A.S.; Torres, D.; Castanheira, I. Protein content and amino acids profile of pseudocereals. Food Chem. 2016, 193, 55–61. [Google Scholar] [CrossRef]
- US Department of Agriculture Agricultural Research Service. Food Data Central. Available online: https://fdc.nal.usda.gov/ (accessed on 6 September 2020).
- Schlick, G.; Bubenheim, D.L. Quinoa: An emerging “new” crop with potential for CELSS. In NASA Technical Paper 3422; NASA Ames Research Center: Moffet Field, CA, USA, 1993; pp. 1–6. [Google Scholar]
- Ruales, J.; Nair, B.M. Nutritional quality of the protein in quinoa (Chenopodium quinoa, Willd) seeds. Plant Foods Hum. Nutr. 1992, 42, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ranhotra, G.S.; Gelroth, J.A.; Glaser, B.K.; Lorenz, K.J.; Johnson, D.L. Composition and protein nutritional quality of quinoa. Cereal Chem. 1993, 70, 303–305. [Google Scholar]
- El-Adawy, T.A. Nutritional composition and antinutritional factors of chickpeas (Cicer arietinum L.) undergoing different cooking methods and germination. Plant Foods Hum. Nutr. 2002, 57, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Pulse Canada. Protein Quality of Cooked Pulses. Available online: http://www.pulsecanada.com/wp-content/uploads/2017/09/Pulses-and-Protein-Quality.pdf (accessed on 29 August 2020).
- AdvantaFava. AdvantaFava Resolve 90 Technical Specification Version 1.1. Available online: https://www.ulprospector.com/documents/1573514.pdf?bs=6673&b=750879&st=20&r=na&ind=food (accessed on 18 July 2020).
- Lisiewska, Z.; Kmiecik, W.; Słupski, J. Content of amino acids in raw and frozen broad beans (Vicia faba var. major) seeds at milk maturity stage, depending on the processing method. Food Chem. 2007, 105, 1468–1473. [Google Scholar] [CrossRef]
- Kaldy, M.S.; Kasting, R. Amino acid composition and protein quality of eight faba bean cultivars. Can. J. Plan. Sci. 1974, 54, 869–871. [Google Scholar] [CrossRef]
- Vogelsang-O’Dwyer, M.; Petersen, I.L.; Joehnke, M.S.; Sorensen, J.C.; Bez, J.; Detzel, A.; Busch, M.; Krueger, M.; O’Mahony, J.A.; Arendt, E.K.; et al. Comparison of Faba Bean Protein Ingredients Produced Using Dry Fractionation and Isoelectric Precipitation: Techno-Functional, Nutritional and Environmental Performance. Foods 2020, 9, 322. [Google Scholar] [CrossRef]
- Sarwar, G.; Peace, R.W. Comparisons between true digestibility of total nitrogen and limiting amino acids in vegetable proteins fed to rats. J. Nutr. 1986, 116, 1172–1184. [Google Scholar] [CrossRef]
- Fitzpatrick, K.; Adolphe, J. Barley-a Healthy Protein Source. Available online: https://gobarley.com/wp-content/uploads/2018/03/EN-GoBarley-Healthy-Protein-Source.pdf (accessed on 15 July 2020).
- Bodwell, C.E.; Satterlee, L.D.; Hackler, L.R. Protein digestibility of the same protein preparations by human and rat assays and by in vitro enzymatic digestion methods. Am. J. Clin. Nutr. 1980, 33, 677–686. [Google Scholar] [CrossRef]
- Amagliani, L.; O’Regan, J.; Kelly, A.L.; O’Mahony, J.A. Composition and protein profile analysis of rice protein ingredients. J. Food Compos. Anal. 2017, 59, 18–26. [Google Scholar] [CrossRef]
- Han, S.W.; Chee, K.M.; Cho, S.J. Nutritional quality of rice bran protein in comparison to animal and vegetable protein. Food Chem. 2015, 172, 766–769. [Google Scholar] [CrossRef]
- Kalman, D.S. Amino Acid Composition of an Organic Brown Rice Protein Concentrate and Isolate Compared to Soy and Whey Concentrates and Isolates. Foods 2014, 3, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Tate and Lyle. Oat Protein: Health Benefits and Product Applications: Innovating to Meet Nutrition, Health, and Wellness Needs Every Day. Available online: https://www.tateandlyle.com/sites/default/files/2018-04/tate-lyle-proatein-brochure-2017.pdf (accessed on 18 July 2020).
- Singh, B.; Singh, U. Peanut as a source of protein for human foods. Plant Foods Hum. Nutr. 1991, 41, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Hey, S.J. The contribution of wheat to human diet and health. Food Energy Secur. 2015, 4, 178–202. [Google Scholar] [CrossRef] [PubMed]
- Al-Gaby, A.M.A. Amino acid composition and biological effects of supplementing broad bean and corn proteins with Nigella sativa (black cumin) cake protein. Nahrung 1998, 42, 290–294. [Google Scholar] [CrossRef]
- Caire-Juvera, G.; Vazquez-Ortiz, F.A.; Grijalva-Haro, M.I. Amino acid composition, score and in vitro protein digestibility of foods commonly consumed in northwest Mexico. Nutr. Hosp. 2013, 28, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Health Canada. Elements within the Nutrition Facts Table: Protein. Available online: https://www.inspection.gc.ca/food-label-requirements/labelling/industry/nutrition-labelling/elements-within-the-nutrition-facts-table/eng/1389206763218/1389206811747?chap=7 (accessed on 11 July 2020).
- Kamei, Y.; Hatazawa, Y.; Uchitomi, R.; Yoshimura, R.; Miura, S. Regulation of Skeletal Muscle Function by Amino Acids. Nutrients 2020, 12, 261. [Google Scholar] [CrossRef]
- Breen, L.; Phillips, S.M. Skeletal muscle protein metabolism in the elderly: Interventions to counteract the ‘anabolic resistance’ of ageing. Nutr. Metab. 2011, 8, 68. [Google Scholar] [CrossRef]
- Wu, G. Amino acids: Metabolism, functions, and nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef]
- Wu, G.; Morris, S.M. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef]
- Ahmadi, A.R.; Rayyani, E.; Bahreini, M.; Mansoori, A. The effect of glutamine supplementation on athletic performance, body composition, and immune function: A systematic review and a meta-analysis of clinical trials. Clin. Nutr. 2019, 38, 1076–1091. [Google Scholar] [CrossRef]
- Li, P.; Wu, G. Roles of dietary glycine, proline, and hydroxyproline in collagen synthesis and animal growth. Amino Acids 2018, 50, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Hevia, E.; De Paz-Lugo, P.; Cornish-Bowden, A.; Cardenas, M.L. A weak link in metabolism: The metabolic capacity for glycine biosynthesis does not satisfy the need for collagen synthesis. J. Biosci. 2009, 34, 853–872. [Google Scholar] [CrossRef]
- Razak, M.A.; Begum, P.S.; Viswanath, B.; Rajagopal, S. Multifarious Beneficial Effect of Nonessential Amino Acid, Glycine: A Review. Oxid. Med. Cell Longev. 2017, 2017, 1716701. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wu, Z.; Dai, Z.; Yang, Y.; Wang, J.; Wu, G. Glycine metabolism in animals and humans: Implications for nutrition and health. Amino Acids 2013, 45, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Heine, W.E.; Klein, P.D.; Reeds, P.J. The importance of alpha-lactalbumin in infant nutrition. J. Nutr. 1991, 121, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Paul, G.L. The rationale for consuming protein blends in sports nutrition. J. Am. Coll Nutr. 2009, 28, 464S–472S. [Google Scholar] [CrossRef] [PubMed]
- Swaisgood, H.E. Protein and amino acid composition of bovine milk. In Handbook of Milk Composition; Jensen, R.G., Ed.; Elsevier: Amsterdam, The Netherlands, 1995; pp. 464–468. [Google Scholar]
- Rutherfurd, S.M.; Moughan, P.J. The digestible amino acid composition of several milk proteins: Application of a new bioassay. J. Dairy Sci. 1998, 81, 909–917. [Google Scholar] [CrossRef]
- Li, S.S.; Blanco Mejia, S.; Lytvyn, L.; Stewart, S.E.; Viguiliouk, E.; Ha, V.; de Souza, R.J.; Leiter, L.A.; Kendall, C.W.C.; Jenkins, D.J.A.; et al. Effect of Plant Protein on Blood Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2017, 6. [Google Scholar] [CrossRef]
- Zhao, H.; Song, A.; Zheng, C.; Wang, M.; Song, G. Effects of plant protein and animal protein on lipid profile, body weight and body mass index on patients with hypercholesterolemia: A systematic review and meta-analysis. Acta Diabetol. 2020. [Google Scholar] [CrossRef]
- Lin, Y.; Mouratidou, T.; Vereecken, C.; Kersting, M.; Bolca, S.; de Moraes, A.C.F.; Cuenca-Garcia, M.; Moreno, L.A.; Gonzalez-Gross, M.; Valtuena, J.; et al. Dietary animal and plant protein intakes and their associations with obesity and cardio-metabolic indicators in European adolescents: The HELENA cross-sectional study. Nutr. J. 2015, 14, 10. [Google Scholar] [CrossRef]
- Campbell, W.W. Animal-based and plant-based protein-rich foods and cardiovascular health: A complex conundrum. Am. J. Clin. Nutr. 2019, 110, 8–9. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.K.; Skulas-Ray, A.C.; Champagne, C.M.; Kris-Etherton, P.M. Plant protein and animal proteins: Do they differentially affect cardiovascular disease risk? Adv. Nutr. 2015, 6, 712–728. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, N.; Chiu, S.; Williams, P.T.; S, M.K.; Krauss, R.M. Effects of red meat, white meat, and nonmeat protein sources on atherogenic lipoprotein measures in the context of low compared with high saturated fat intake: A randomized controlled trial. Am. J. Clin. Nutr. 2019, 110, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Stewart, K.; Oda, K.; Batech, M.; Herring, R.P.; Fraser, G.E. Vegetarian diets and incidence of diabetes in the Adventist Health Study-2. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 292–299. [Google Scholar] [CrossRef]
- Malik, V.S.; Li, Y.; Tobias, D.K.; Pan, A.; Hu, F.B. Dietary Protein Intake and Risk of Type 2 Diabetes in US Men and Women. Am. J. Epidemiol. 2016, 183, 715–728. [Google Scholar] [CrossRef]
- Smith, C.E.; Mollard, R.C.; Luhovyy, B.L.; Anderson, G.H. The effect of yellow pea protein and fibre on short-term food intake, subjective appetite and glycaemic response in healthy young men. Br. J. Nutr. 2012, 108 (Suppl. 1), S74–S80. [Google Scholar] [CrossRef]
- Konig, D.; Muser, K.; Berg, A.; Deibert, P. Fuel selection and appetite-regulating hormones after intake of a soy protein-based meal replacement. Nutrition 2012, 28, 35–39. [Google Scholar] [CrossRef]
- Viguiliouk, E.; Stewart, S.E.; Jayalath, V.H.; Ng, A.P.; Mirrahimi, A.; de Souza, R.J.; Hanley, A.J.; Bazinet, R.P.; Blanco Mejia, S.; Leiter, L.A.; et al. Effect of Replacing Animal Protein with Plant Protein on Glycemic Control in Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2015, 7, 9804–9824. [Google Scholar] [CrossRef]
- Markova, M.; Koelman, L.; Hornemann, S.; Pivovarova, O.; Sucher, S.; Machann, J.; Rudovich, N.; Thomann, R.; Schneeweiss, R.; Rohn, S.; et al. Effects of plant and animal high protein diets on immune-inflammatory biomarkers: A 6-week intervention trial. Clin. Nutr. 2020, 39, 862–869. [Google Scholar] [CrossRef]
- Markova, M.; Pivovarova, O.; Hornemann, S.; Sucher, S.; Frahnow, T.; Wegner, K.; Machann, J.; Petzke, K.J.; Hierholzer, J.; Lichtinghagen, R.; et al. Isocaloric Diets High in Animal or Plant Protein Reduce Liver Fat and Inflammation in Individuals With Type 2 Diabetes. Gastroenterology 2017, 152, 571–585.e578. [Google Scholar] [CrossRef]
- Pivovarova-Ramich, O.; Markova, M.; Weber, D.; Sucher, S.; Hornemann, S.; Rudovich, N.; Raila, J.; Sunaga-Franze, D.; Sauer, S.; Rohn, S.; et al. Effects of diets high in animal or plant protein on oxidative stress in individuals with type 2 diabetes: A randomized clinical trial. Redox Biol. 2020, 29, 101397. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh-Rostami, Z.; Hemmatdar, Z.; Pishdad, G.R.; Faghih, S. Moderate Consumption of Red Meat, Compared to Soy or Non-Soy Legume, Has No Adverse Effect on Cardio-Metabolic Factors in Patients with Type 2 Diabetes. Exp. Clin. Endocrinol. Diabetes 2019. [Google Scholar] [CrossRef] [PubMed]
- Andersen, V.; Halekoh, U.; Tjonneland, A.; Vogel, U.; Kopp, T.I. Intake of Red and Processed Meat, Use of Non-Steroid Anti-Inflammatory Drugs, Genetic Variants and Risk of Colorectal Cancer: A Prospective Study of the Danish “Diet, Cancer and Health” Cohort. Int. J. Mol. Sci. 2019, 20, 1121. [Google Scholar] [CrossRef] [PubMed]
- Moller, G.; Andersen, J.R.; Jalo, E.; Ritz, C.; Brand-Miller, J.; Larsen, T.M.; Silvestre, M.P.; Fogelholm, M.; Poppitt, S.D.; Raben, A.; et al. The association of dietary animal and plant protein with putative risk markers of colorectal cancer in overweight pre-diabetic individuals during a weight-reducing programme: A PREVIEW sub-study. Eur. J. Nutr. 2020, 59, 1517–1527. [Google Scholar] [CrossRef]
- Chatterjee, C.; Gleddie, S.; Xiao, C.W. Soybean Bioactive Peptides and Their Functional Properties. Nutrients 2018, 10, 1211. [Google Scholar] [CrossRef]
- Lonnie, M.; Laurie, I.; Myers, M.; Horgan, G.; Russell, W.R.; Johnstone, A.M. Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies. Nutrients 2020, 12, 2291. [Google Scholar] [CrossRef]
- Huang, J.; Liao, L.M.; Weinstein, S.J.; Sinha, R.; Graubard, B.I.; Albanes, D. Association Between Plant and Animal Protein Intake and Overall and Cause-Specific Mortality. JAMA Intern. Med. 2020. [Google Scholar] [CrossRef]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef]
- Grotto, D.; Zied, E. The Standard American Diet and its relationship to the health status of Americans. Nutr. Clin. Pract. 2010, 25, 603–612. [Google Scholar] [CrossRef]
- Smyth, A.; Griffin, M.; Yusuf, S.; Mann, J.F.; Reddan, D.; Canavan, M.; Newell, J.; O’Donnell, M. Diet and Major Renal Outcomes: A Prospective Cohort Study. The NIH-AARP Diet and Health Study. J. Ren. Nutr. 2016, 26, 288–298. [Google Scholar] [CrossRef]
- Bernier-Jean, A.; Prince, R.L.; Lewis, J.R.; Craig, J.C.; Hodgson, J.M.; Lim, W.H.; Teixeira-Pinto, A.; Wong, G. Dietary plant and animal protein intake and decline in estimated glomerular filtration rate among elderly women: A 10-year longitudinal cohort study. Nephrol. Dial. Transplant. 2020. [Google Scholar] [CrossRef] [PubMed]
- Viberti, G.; Bognetti, E.; Wiseman, M.J.; Dodds, R.; Gross, J.L.; Keen, H. Effect of a protein-restricted diet on renal response to a meat meal in humans. Am. J. Physiol. 1987, 253, F388–F393. [Google Scholar] [CrossRef] [PubMed]
- Bosch, J.P.; Saccaggi, A.; Lauer, A.; Ronco, C.; Belledonne, M.; Glabman, S. Renal functional reserve in humans: Effect of protein intake on glomerular filtration rate. Am. J. Med. 1983, 75, 943–950. [Google Scholar] [CrossRef]
- Stephenson, T.J.; Setchel, K.D.R.; Kendall, C.W.; Jenkins, D.J.A.; Anderson, J.W.; Fanti, P. Effect of soy protein-rich diet on renal function in young adults with insulin-dependent diabetes mellitus. Clin. Nephrol. 2005, 64, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Adair, K.E.; Bowden, R.G. Ameliorating Chronic Kidney Disease Using a Whole Food Plant-Based Diet. Nutrients 2020, 12, 1007. [Google Scholar] [CrossRef] [PubMed]
- Rebholz, C.M.; Coresh, J.; Grams, M.E.; Steffen, L.M.; Anderson, C.A.; Appel, L.J.; Crews, D.C. Dietary Acid Load and Incident Chronic Kidney Disease: Results from the ARIC Study. Am. J. Nephrol. 2015, 42, 427–435. [Google Scholar] [CrossRef]
- Goraya, N.; Simoni, J.; Jo, C.H.; Wesson, D.E. A comparison of treating metabolic acidosis in CKD stage 4 hypertensive kidney disease with fruits and vegetables or sodium bicarbonate. Clin. J. Am. Soc. Nephrol. 2013, 8, 371–381. [Google Scholar] [CrossRef]
- Goraya, N.; Simoni, J.; Jo, C.H.; Wesson, D.E. Treatment of metabolic acidosis in patients with stage 3 chronic kidney disease with fruits and vegetables or oral bicarbonate reduces urine angiotensinogen and preserves glomerular filtration rate. Kidney Int. 2014, 86, 1031–1038. [Google Scholar] [CrossRef]
- Moe, S.M.; Zidehsarai, M.P.; Chambers, M.A.; Jackman, L.A.; Radcliffe, J.S.; Trevino, L.L.; Donahue, S.E.; Asplin, J.R. Vegetarian compared with meat dietary protein source and phosphorus homeostasis in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 257–264. [Google Scholar] [CrossRef]
- Scialla, J.J.; Appel, L.J.; Wolf, M.; Yang, W.; Zhang, X.; Sozio, S.M.; Miller, E.R., 3rd; Bazzano, L.A.; Cuevas, M.; Glenn, M.J.; et al. Plant protein intake is associated with fibroblast growth factor 23 and serum bicarbonate levels in patients with chronic kidney disease: The Chronic Renal Insufficiency Cohort study. J. Ren. Nutr. 2012, 22, 379–388.e371. [Google Scholar] [CrossRef]
- Kubota, M.; Watanabe, R.; Yamaguchi, M.; Hosojima, M.; Saito, A.; Fujii, M.; Fujimura, S.; Kadowaki, M. Rice endosperm protein slows progression of fatty liver and diabetic nephropathy in Zucker diabetic fatty rats. Br. J. Nutr. 2016, 116, 1326–1335. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.J.; Haynes, T.E.; Kohli, R.; Hu, J.; Shi, W.; Spencer, T.E.; Carroll, R.J.; Meininger, C.J.; Wu, G. Dietary L-arginine supplementation reduces fat mass in Zucker diabetic fatty rats. J. Nutr. 2005, 135, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.W. Beneficial effects of soy protein consumption for renal function. Asia Pac. J. Clin. Nutr. 2008, 17, 324–328. [Google Scholar] [PubMed]
- McGraw, N.J.; Krul, E.S.; Grunz-Borgmann, E.; Parrish, A.R. Soy-based renoprotection. World J. Nephrol. 2016, 5, 233–257. [Google Scholar] [CrossRef] [PubMed]
- National Kidney Foundation. Plant-Based Diet and Kidney Health. Available online: https://www.kidney.org/atoz/content/plant-based (accessed on 11 October 2020).
- Macnaughton, L.S.; Wardle, S.L.; Witard, O.C.; McGlory, C.; Hamilton, D.L.; Jeromson, S.; Lawrence, C.E.; Wallis, G.A.; Tipton, K.D. The response of muscle protein synthesis following whole-body resistance exercise is greater following 40 g than 20 g of ingested whey protein. Physiol. Rep. 2016, 4. [Google Scholar] [CrossRef]
- Rieu, I.; Balage, M.; Sornet, C.; Giraudet, C.; Pujos, E.; Grizard, J.; Mosoni, L.; Dardevet, D. Leucine supplementation improves muscle protein synthesis in elderly men independently of hyperaminoacidaemia. J. Physiol. 2006, 575, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Wardle, S.L.; Macnaughton, L.S.; Hodgson, A.B.; Tipton, K.D. Protein Considerations for Optimising Skeletal Muscle Mass in Healthy Young and Older Adults. Nutrients 2016, 8, 181. [Google Scholar] [CrossRef]
- Yang, Y.; Breen, L.; Burd, N.A.; Hector, A.J.; Churchward-Venne, T.A.; Josse, A.R.; Tarnopolsky, M.A.; Phillips, S.M. Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br. J. Nutr. 2012, 108, 1780–1788. [Google Scholar] [CrossRef]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Crombag, J.J.; Langer, H.; Bierau, J.; Respondek, F.; van Loon, L.J. Ingestion of Wheat Protein Increases In Vivo Muscle Protein Synthesis Rates in Healthy Older Men in a Randomized Trial. J. Nutr. 2016, 146, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.J.; Churchward-Venne, T.A.; Parise, G.; Bellamy, L.; Baker, S.K.; Smith, K.; Atherton, P.J.; Phillips, S.M. Acute post-exercise myofibrillar protein synthesis is not correlated with resistance training-induced muscle hypertrophy in young men. PLoS ONE 2014, 9, e89431. [Google Scholar] [CrossRef] [PubMed]
- Hartman, J.W.; Tang, J.E.; Wilkinson, S.B.; Tarnopolsky, M.A.; Lawrence, R.L.; Fullerton, A.V.; Phillips, S.M. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am. J. Clin. Nutr. 2007, 86, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Volek, J.S.; Volk, B.M.; Gomez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.D.; et al. Whey protein supplementation during resistance training augments lean body mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Messina, M.; Lynch, H.; Dickinson, J.M.; Reed, K.E. No Difference Between the Effects of Supplementing With Soy Protein Versus Animal Protein on Gains in Muscle Mass and Strength in Response to Resistance Exercise. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 674–685. [Google Scholar] [CrossRef]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Wilson, S.M.C.; Kalman, D.S.; Dudeck, J.E.; Jager, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12, 86. [Google Scholar] [CrossRef]
- Kalman, D.; Escalante, A.; Hewlings, S.J.; Willoughby, D.S. The body composition effects of extra protein in elite mixed martial artists undergoing frequent training over a six-week period. EC Nutr. 2018, 13, 6. [Google Scholar]
- Babault, N.; Paizis, C.; Deley, G.; Guerin-Deremaux, L.; Saniez, M.H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, Placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12, 3. [Google Scholar] [CrossRef]
- Banaszek, A.; Townsend, J.R.; Bender, D.; Vantrease, W.C.; Marshall, A.C.; Johnson, K.D. The Effects of Whey vs. Pea Protein on Physical Adaptations Following 8-Weeks of High-Intensity Functional Training (HIFT): A Pilot Study. Sports (Basel) 2019, 7, 12. [Google Scholar] [CrossRef]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef]
- Hernandez, M.; Montalvo, I.; Sousa, V.; Sotelo, A. The protein efficiency ratios of 30:70 mixtures of animal:vegetable protein are similar or higher than those of the animal foods alone. J. Nutr. 1996, 126, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Cope, M.B.; Mukherjea, R.; Jennings, K.; Volpi, E.; et al. Soy-dairy protein blend and whey protein ingestion after resistance exercise increases amino acid transport and transporter expression in human skeletal muscle. J. Appl. Physiol. 2014, 116, 1353–1364. [Google Scholar] [CrossRef]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Fry, C.S.; Borack, M.S.; Cope, M.B.; Mukherjea, R.; et al. Protein blend ingestion following resistance exercise promotes human muscle protein synthesis. J. Nutr. 2013, 143, 410–416. [Google Scholar] [CrossRef]
- Popova, A.; Mihaylova, D. Antinutrients in Plant-based Foods: A Review. Open Biotechnol. J. 2019, 13, 68–76. [Google Scholar] [CrossRef]
- Messina, M. Soy and Health Update: Evaluation of the Clinical and Epidemiologic Literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.S.; Setchell, K.D.R.; Stocco, D.M.; Lephart, E.D. Dietary soy-phytoestrogens decrease testosterone levels and prostate weight without altering LH, 5a-reductase or testicular steroidogenic acute regulatory peptide levels in adult male Sprague-Dawley rats. J. Endocrinol. 2001, 170, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, S.; Shen, X.-Y.; Yang, N.-Y.; Liu, Y.; Tsuji, I.; Yamamura, T.; Li, J.; Li, X.-M. Phytoestrogens inhibiting androgen receptor signal and prostate cancer cell proliferation. Chem. Res. Chin. Univ. 2013, 29, 911–916. [Google Scholar] [CrossRef]
- Bektic, J.; Berger, A.P.; Pfeil, K.; Dobler, G.; Bartsch, G.; Klocker, H. Androgen receptor regulation by physiological concentrations of the isoflavonoid genistein in androgen-dependent LNCaP cells is mediated by estrogen receptor beta. Eur. Urol. 2004, 45, 245–251. [Google Scholar] [CrossRef]
- Genovese, M.I.; Barbosa, A.C.; Pinto Mda, S.; Lajolo, F.M. Commercial soy protein ingredients as isoflavone sources for functional foods. Plant Foods Hum. Nutr. 2007, 62, 53–58. [Google Scholar] [CrossRef]
- European Food Safety Authority. Risk assessment for peri- and post-menopausal women taking food supplements containing isolated isoflavones. EFSA J. 2015, 13. [Google Scholar] [CrossRef]
- Hamilton-Reeves, J.M.; Vazquez, G.; Duval, S.J.; Phipps, W.R.; Kurzer, M.S.; Messina, M.J. Clinical studies show no effects of soy protein or isoflavones on reproductive hormones in men: Results of a meta-analysis. Fertil. Steril. 2010, 94, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Dillingham, B.L.; McVeigh, B.L.; Lampe, J.W.; Duncan, A.M. Soy protein isolates of varying isoflavone content exert minor effects on serum reproductive hormones in healthy young men. J. Nutr. 2005, 135, 584–591. [Google Scholar] [CrossRef] [PubMed]
- Beaton, L.K.; McVeigh, B.L.; Dillingham, B.L.; Lampe, J.W.; Duncan, A.M. Soy protein isolates of varying isoflavone content do not adversely affect semen quality in healthy young men. Fertil. Steril. 2010, 94, 1717–1722. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zeng, R.; Huang, J.; Li, X.; Zhang, J.; Ho, J.C.; Zheng, Y. Dietary Protein Sources and Incidence of Breast Cancer: A Dose-Response Meta-Analysis of Prospective Studies. Nutrients 2016, 8, 730. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.T.; Jin, F.; Li, J.G.; Xu, Y.Y.; Dong, H.T.; Liu, Q.; Xing, P.; Zhu, G.L.; Xu, H.; Miao, Z.F. Dietary isoflavones or isoflavone-rich food intake and breast cancer risk: A meta-analysis of prospective cohort studies. Clin. Nutr. 2019, 38, 136–145. [Google Scholar] [CrossRef]
- Qiu, S.; Jiang, C. Soy and isoflavones consumption and breast cancer survival and recurrence: A systematic review and meta-analysis. Eur. J. Nutr. 2019, 58, 3079–3090. [Google Scholar] [CrossRef] [PubMed]
- Eakin, A.; Kelsberg, G.; Safranek, S.; Neher, J.O. Does high dietary soy intake affect a woman’s risk of primary or recurrent breast cancer? J. Fam. Pract. 2015, 64, 660–662. [Google Scholar]
- Chi, F.; Wu, R.; Zeng, Y.C.; Xing, R.; Liu, Y.; Xu, Z.G. Post-diagnosis soy food intake and breast cancer survival: A meta-analysis of cohort studies. Asian Pac. J. Cancer Prev. 2013, 14, 2407–2412. [Google Scholar] [CrossRef]
- Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J. Clin. 2012, 62, 242–274. [Google Scholar] [CrossRef]
- Van Wyk, J.J.; Arnold, M.B.; Wynn, J.; Pepper, F. The effects of a soybean product on thyroid function in humans. Pediatrics 1959, 24, 752–760. [Google Scholar]
- Hydovitz, J.D. Occurrence of goiter in an infant on a soy diet. N. Engl. J. Med. 1960, 262, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Doerge, D.R. Dietary genistein inactivates rat thyroid peroxidase in vivo without an apparent hypothyroid effect. Toxicol. Appl. Pharmacol. 2000, 168, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Divi, R.L.; Doerge, D.R. Inhibition of thyroid peroxidase by dietary flavonoids. Chem. Res. Toxicol. 1996, 9, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Otun, J.; Sahebkar, A.; Ostlundh, L.; Atkin, S.L.; Sathyapalan, T. Systematic Review and Meta-analysis on the Effect of Soy on Thyroid Function. Sci. Rep. 2019, 9, 3964. [Google Scholar] [CrossRef]
- Messina, M.; Redmond, G. Effects of soy protein and soybean isoflavones on thyroid function in healthy adults and hypothyroid patients: A review of the relevant literature. Thyroid 2006, 16, 249–258. [Google Scholar] [CrossRef]
- Bell, D.S.H.; Ovalle, F. Use of soy protein supplement and resultant need for increased dose of levothryroxine. Endocr. Pract. 2001, 7, 193–194. [Google Scholar] [CrossRef]
- Palmer, S. Plant Proteins. Today’s Dietit. 2017, 19, 26–33. [Google Scholar]
- Mintel Press Team. US Non-Dairy Milk Sales Grow 61% over the Last 5 Years. Available online: https://www.mintel.com/press-centre/food-and-drink/us-non-dairy-milk-sales-grow-61-over-the-last-five-years#:~:text=New%20research%20from%20Mintel%20reveals,reach%20%242.11%20billion%20in%202017 (accessed on 20 August 2020).
- Bridges, M. Moo-over, cow’s milk: The rise of plant-based dairy alternatives. Pract. Gastroenterol. 2018, 171, 20–27. [Google Scholar]
- McCarthy, K.S.; Parker, M.; Ameerally, A.; Drake, S.L.; Drake, M.A. Drivers of choice for fluid milk versus plant-based alternatives: What are consumer perceptions of fluid milk? J. Dairy Sci. 2017, 100, 6125–6138. [Google Scholar] [CrossRef]
- Makinen, O.E.; Wanhalinna, V.; Zannini, E.; Arendt, E.K. Foods for Special Dietary Needs: Non-dairy Plant-based Milk Substitutes and Fermented Dairy-type Products. Crit. Rev. Food Sci. Nutr. 2016, 56, 339–349. [Google Scholar] [CrossRef]
- O’Connor, A. Got Almond Milk? Dairy Farms Protest Milk Label on Nondairy Drinks. Available online: https://www.nytimes.com/2017/02/13/well/eat/got-almond-milk-dairy-farms-protest-milk-label-on-nondairy-drinks.html (accessed on 20 August 2020).
- NIAID-Sponsored Expert Panel; Boyce, J.A.; Assa’ad, A.; Burks, A.W.; Jones, S.M.; Sampson, H.A.; Wood, R.A.; Plaut, M.; Cooper, S.F.; Fenton, M.J.; et al. Guidelines for the diagnosis and management of food allergy in the United States: Report of the NIAID-sponsored expert panel. J. Allergy Clin. Immunol. 2010, 126, S1–S58. [Google Scholar] [CrossRef] [PubMed]
- Sampson, H.A.; Aceves, S.; Bock, S.A.; James, J.; Jones, S.; Lang, D.; Nadeau, K.; Nowak-Wegrzyn, A.; Oppenheimer, J.; Perry, T.T.; et al. Food allergy: A practice parameter update-2014. J. Allergy Clin. Immunol. 2014, 134, 1016–1025.e1043. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.S.; Warren, C.M.; Smith, B.M.; Blumenstock, J.A.; Jiang, J.; Davis, M.M.; Nadeau, K.C. The public health impact of parent-reported childhood food allergies in the United States. Pediatrics 2018, 142, e20181235. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.S.; Warren, C.M.; Smith, B.M.; Jiang, J.; Blumenstock, J.A.; Davis, M.M.; Schleimer, R.P.; Nadeau, K.C. Prevalence and Severity of Food Allergies Among US Adults. JAMA Netw. Open 2019, 2, e185630. [Google Scholar] [CrossRef]
- Taylor, S.L. Food Allergies-a Challenge for cuRrent and Emerging Proteins. Available online: https://www.globalfoodforums.com/wp-content/uploads/2018/05/Protein-Food-Allergies-Steve-Taylor.pdf (accessed on 20 August 2020).
- Koeberl, M.; Sharp, M.F.; Tian, R.; Buddhadasa, S.; Clarke, D.; Roberts, J. Lupine allergen detecting capability and cross-reactivity of related legumes by ELISA. Food Chem. 2018, 256, 105–112. [Google Scholar] [CrossRef]
- Jappe, U.; Vieths, S. Lupine, a source of new as well as hidden food allergens. Mol. Nutr. Food Res. 2010, 54, 113–126. [Google Scholar] [CrossRef]
- NC Soybean Producers Association. History of Soybeans. Available online: https://ncsoy.org/media-resources/history-of-soybeans/#:~:text=Soybeans%20originated%20in%20Southeast%20Asia,colony%20of%20Georgia%20in%201765 (accessed on 20 August 2020).
- Egana, J.I.; Fuentes, A.; Steinke, F.H.; Uauy, R. Protein quality comparison of a new isolated soy protein and milk in Chilean preschool children. Nutr. Res. 1983, 3, 195–202. [Google Scholar] [CrossRef]
- Kay, T.; Ifeacho, C.L.; Onowu, G.; Udenze, V.; Nnamani, G.; Anazonwu-Bello, J.N.; Ifenu, F.; Modebe, V.; Emembolu, M. Use of soya bean to improve the protein content of the diet in West Africa and thus prevent kwashiorkor. J. Trop. Pediatr. Environ. Child. Health 1975, 21, 45–48. [Google Scholar]
- Mathew, A.; Raut, D.S. Effect of soyamilk on the growth of malnourished children admitted to hospital wards. Indian J. Nutr. Diet. 1981, 18, 260–267. [Google Scholar]
- Savage, J.H.; Kaeding, A.J.; Matsui, E.C.; Wood, R.A. The natural history of soy allergy. J. Allergy Clin. Immunol. 2010, 125, 683–686. [Google Scholar] [CrossRef]
- Cordle, C.T. Soy protein allergy: Incidence and relative severity. J. Nutr. 2004, 134, 1213S–1219S. [Google Scholar] [CrossRef] [PubMed]
- Vierk, K.A.; Koehler, K.M.; Fein, S.B.; Street, D.A. Prevalence of self-reported food allergy in American adults and use of food labels. J. Allergy Clin. Immunol. 2007, 119, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- FARE Food Allergy Research & Education. Soy Allergy. Available online: https://www.foodallergy.org/living-food-allergies/food-allergy-essentials/common-allergens/soy (accessed on 20 August 2020).
- Gupta, R.S.; Springston, E.E.; Warrier, M.R.; Smith, B.; Kumar, R.; Pongracic, J.; Holl, J.L. The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics 2011, 128, e9–e17. [Google Scholar] [CrossRef] [PubMed]
- Soller, L.; Ben-Shoshan, M.; Harrington, D.W.; Fragapane, J.; Joseph, L.; St Pierre, Y.; Godefroy, S.B.; La Vieille, S.; Elliott, S.J.; Clarke, A.E. Overall prevalence of self-reported food allergy in Canada. J. Allergy Clin. Immunol. 2012, 130, 986–988. [Google Scholar] [CrossRef] [PubMed]
- American College of Allergy, Asthma, and Immunology. Milk and Dairy Allergy. Available online: https://acaai.org/allergies/types-allergies/food-allergy/types-food-allergy/milk-dairy-allergy (accessed on 20 August 2020).
- Eggesbo, M.; Botten, G.; Halvorsen, R.; Magnus, P. The prevalence of allergy to egg: A population-based study in young children. Allergy 2001, 56, 403–411. [Google Scholar] [CrossRef]
- Sicherer, S.H.; Sampson, H.A. 9. Food allergy. J. Allergy Clin. Immunol. 2006, 117, S470–S475. [Google Scholar] [CrossRef]
- American College of Allergy, Asthma, and Immunology. Soy Allergy. Available online: https://acaai.org/allergies/types/food-allergies/types-food-allergy/soy-allergy (accessed on 20 August 2020).
- Nowak-Wegrzyn, A.; Conover-Walker, M.K.; Wood, R.A. Food-allergic reactions in schools and preschools. Arch. Pediatr. Adolesc. Med. 2001, 155, 790–795. [Google Scholar] [CrossRef]
- Soy Nutrition Institute. Soy Connection Fact Sheet: Soyfoods + Infants, Children, & Adolescents. Available online: https://thesoynutritioninstitute.com/wp-content/uploads/2019/04/childrens-health-fact-sheet.pdf (accessed on 20 August 2020).
- Shewry, P.R.; Beaudoin, F.; Jenkins, J.; Griffiths-Jones, S.; Mills, E.N.C. Plant protein families and their relationships to food allergy. Biochem Soc. Trans. 2002, 30, 906–910. [Google Scholar] [CrossRef]
- Shewry, P.R.; Pandya, M.J. The 2S albumin storage proteins. In Seed Proteins; Shewry, P.R., Casey, R., Eds.; Springer: New York, NY, USA, 1999. [Google Scholar]
- Carbonero, P.; Garcia-Olmedo, F. A multigene family of trypsin/a-amylase inhibitors from cereals. In Seed Proteins; Shewry, P.R., Casey, R., Eds.; Springer: New York, NY, USA, 1999; pp. 617–633. [Google Scholar]
- Douliez, J.P.; Michon, T.; Elmorjani, K.; Marion, D. Mini Review: Structure, Biological and Technological Functions of Lipid Transfer Proteins and Indolines, the Major Lipid Binding Proteins from Cereal Kernels. J. Cereal Sci. 2000, 32, 1–20. [Google Scholar] [CrossRef]
- Rahman, S.; Jolly, C.J.; Skerritt, J.H.; Wallosheck, A. Cloning of a wheat 15-kDa grain softness protein (GSP): GSP is a mixture of puroindoline-like polypeptides. Eur. J. Biochem. 1994, 223, 917–925. [Google Scholar] [CrossRef]
- Baud, F.; Pebay-Peyroula, E.; Cohen-Addad, C.; Odani, S.; Lehman, M.S. Crystal structure of hydrophobic protein from soybean; a member of a new cysteine-rich family. J. Mol. Biol. 1993, 231, 877–887. [Google Scholar] [CrossRef] [PubMed]
- University of Nebraska-Lincoln. Allergen Online: Home of the FARRP Allergen Database. Available online: http://www.allergenonline.com/ (accessed on 17 August 2020).
- Lu, Z.X.; He, J.F.; Zhang, Y.C.; Bing, D.J. Composition, physicochemical properties of pea protein and its application in functional foods. Crit. Rev. Food Sci. Nutr. 2020, 60, 2593–2605. [Google Scholar] [CrossRef] [PubMed]
- Lavine, E.; Ben-Shoshan, M. Anaphylaxis to hidden pea protein: A Canadian pediatric case series. J. Allergy Clin. Immunol. Pract. 2019, 7, 2070–2071. [Google Scholar] [CrossRef] [PubMed]
- Wensing, M.; Knulst, A.C.; Piersma, S.; O’Kane, F.; Knol, E.F.; Koppelman, S.J. Patients with anaphylaxis to pea can have peanut allergy caused by cross-reactive IgE to vicilin (Ara h 1). J. Allergy Clin. Immunol. 2003, 111, 420–424. [Google Scholar] [CrossRef]
- Messina, M.; Venter, C. Recent surveys on food allergy prevalence. Nutr. Today 2020, 55, 22–29. [Google Scholar] [CrossRef]
- Skypala, I.J. Food-Induced Anaphylaxis: Role of Hidden Allergens and Cofactors. Front. Immunol. 2019, 10, 673. [Google Scholar] [CrossRef]
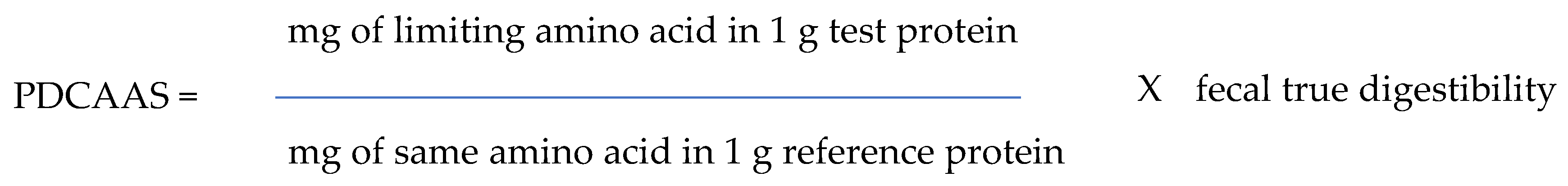
| Indispensable | Dispensable | Conditionally Indispensable |
|---|---|---|
| Histidine | Alanine | Arginine |
| Isoleucine | Aspartic acid | Cysteine |
| Leucine | Asparagine | Glutamine |
| Lysine | Glutamic acid | Glycine |
| Methionine | Serine | Proline |
| Phenylalanine | Tyrosine | |
| Threonine | ||
| Tryptophan | ||
| Valine |
| Protein | PDCAAS 1 | PDCAAS 2 | PDCAAS 3 | PDCAAS 4 | DIAAS 3 | Limiting Amino Acid(s), When Present | AA Profile: Materials Analyzed and References | Fractional Digestibility and References |
|---|---|---|---|---|---|---|---|---|
| Milk | 1.00 | 1.00 | 1.00 | 1.00 | 1.08 | None | Milk PC [20,21] | Fecal true protein: mean 0.96 [13,20] Ileal AA: range for individual AA 0.84–0.94 [22] |
| Whey | 1.00 | 1.00 | 0.97 | 1.00 | 0.90 | His | Whey PI [20,21,23] Whey PC [20,21] | Fecal true protein: mean 0.96 [20,24] Ileal AA: range for individual AA 0.89–1.00 [22] |
| Soy | 0.99 | 1.00 | 0.93 | 1.00 | 0.92 | SAA | Soy PI, Soy PC [25] Soy PI [20] Soy PI [21] | Fecal true protein: mean 0.97 [13,20,21,25] Ileal AA: range for individual AA 0.95–0.99 [22] |
| Canola | 0.88 | 1.00 | 0.93 | 1.00 | NA | AAA | Canola PI [26]; Canola PI [27] | Fecal true protein: mean 0.95 [26,27] |
| Potato | 0.87 | 1.00 | 0.87 | 1.00 | 0.85 | His | Solanic 100F Potato PI [28] Solanic 206P HMW and LMW [29] Potato protein [30] Potato juice protein concentrate [31] Avg of 6 potato varieties [32] | Fecal true protein: 0.89 [24] Ileal AA: range for individual AA 0.73–0.90 [22] |
| Pea | 0.83 | 0.84 | 0.78 | 0.91 | 0.66 | SAA *, Trp | Pea PC [33] Pea PC [21] Pea PC [20] | Fecal true protein: mean 0.97 [20,21,33] Ileal AA: range for individual AA 0.83–0.90 [20] |
| Quinoa | 0.78 | 0.89 | 0.77 | 0.84 | NA | Ile, Leu, Lys *, Thr, Val | Quinoa, raw [34] Quinoa [35] Quinoa from Salta [36] Uncooked quinoa [37] Field grown quinoa [38] Raw and unwashed quinoa [39] | Fecal true protein: mean 0.89 [39,40] |
| Chickpea | 0.77 | 0.85 | 0.71 | 0.71 | 0.69 | Leu, Lys, SAA *, Thr, Trp, Val | Boiled chickpeas [41] | Fecal true protein: 0.85 [42] Ilea AA: range for individual AA 0.72–0.9 [22] |
| Lentils | 0.73 | 0.73 | 0.68 | 0.80 | 0.75 | Leu, SAA *, Thr, Trp, Val | Lentils, mature seeds, ckd, bld without salt [37] | Fecal true protein: 0.85 [24] Ileal AA: range for individual AA 0.82–0.98 [22] |
| Red Kidney beans | 0.68 | 0.68 | 0.63 | 0.74 | 0.61 | Leu, Lys, SAA *, AAA, Thr, Trp, Val | Red kidney beans, cnd, drnd solids [37] | Fecal true protein: 0.81 [24] Ileal AA: range for individual AA 0.72–0.94 [22] |
| Fava/faba | 0.63 | 0.65 | 0.60 | 0..67 | NA | Lys, SAA *, Thr, Trp, Val | Fava bean PI [43] Cooked fava beans [44] Broadbeans, ckd [37] 8 faba cultivars [45] Faba bean PI [46] | Fecal true protein: 0.86 [47] |
| Barley | 0.63 | 0.71 | 0.64 | 0.76 | 0.50 | Lys * | Barley, pearled [48] | Fecal true protein: 0.98 [49] Ileal AA: range for individual AA 0.76–0.83 [22] |
| Pinto beans | 0.61 | 0.61 | 0.57 | 0.66 | NA | His, Ile, Leu, Lys, SAA *, AAA, Thr, Trp, Val | Pinto beans, cnd, drnd solids [37] | Fecal true protein: 0.73 [24] |
| Rice | 0.53 | 0.60 | 0.54 | 0.64 | 0.52 | Lys *, Thr | Rice PC [50] Rice endosperm protein [51] Oryzatein 90 and 80 Rice protein [52] Rice PC [21] | Fecal true protein: mean 0.90 [21,24,51] Ileal AA: mean ranges for individual AA 0.81–0.87 [21,22] |
| Oat | 0.51 | 0.59 | 0.52 | 0.62 | 0.44 | Lys *, Thr | Oat PC [53]; Rolled oats [21] | Fecal true protein: 0.91 [13] Ileal AA: range for individual AA 0.70–0.85 [22] |
| Peanut | 0.46 | 0.52 | 0.47 | 0.55 | 0.47 | Ile, Leu, Lys *, SAA, Thr, Trp, Val | Peanut PC and PI [54] Roasted peanuts [21] | Fecal true protein: 0.93 [24] Ileal AA: mean ranges for individual AA 0.82–0.96 [21,22] |
| Wheat | 0.45 | 0.51 | 0.46 | 0.54 | 0.39 | Ile, Leu *, Lys *, AAA, Thr *, Val | Whole meal and white flour [55] Wheat bran [21] | Fecal true protein: mean 0.94 [24] Ileal AA: mean ranges for individual AA 0.81–0.91 [22] (wheat bran, wheat flour, wheat gluten, wheat) |
| Corn | 0.41 | 0.47 | 0.42 | 0.50 | 0.38 | Ile, Lys *, SAA, Thr *, Trp*, Val | Corn meal [56] Corn tortillas [57] | Fecal true protein: 0.84 [24] Ileal AA: ranges for individual AA 0.75–0.88 [22] |
| Protein | Glutamine Concentration (mg/g Protein, Mean) | Glutamine Concentration (mg/g Protein, Range) |
|---|---|---|
| Wheat protein hydrolysate (n = 15) | 296 | 184–402 |
| Wheat protein isolate (n = 2) | 208 | 184–232 |
| Corn protein (n = 1) | 196 | -- |
| Rice protein (n = 1) | 130 | -- |
| Casein (n = 2) | 102 | 100–104 |
| Soy protein isolate (n = 2) | 100 | 94–106 |
| Soy protein concentrate (n = 1) | 94 | -- |
| Milk protein concentrate (n = 1) | 94 | -- |
| Whey protein concentrate (n = 2) | 57 | 50–63 |
| Ion exchange whey protein isolate (n = 1) | 34 | -- |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hertzler, S.R.; Lieblein-Boff, J.C.; Weiler, M.; Allgeier, C. Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients 2020, 12, 3704. https://doi.org/10.3390/nu12123704
Hertzler SR, Lieblein-Boff JC, Weiler M, Allgeier C. Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients. 2020; 12(12):3704. https://doi.org/10.3390/nu12123704
Chicago/Turabian StyleHertzler, Steven R., Jacqueline C. Lieblein-Boff, Mary Weiler, and Courtney Allgeier. 2020. "Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function" Nutrients 12, no. 12: 3704. https://doi.org/10.3390/nu12123704
APA StyleHertzler, S. R., Lieblein-Boff, J. C., Weiler, M., & Allgeier, C. (2020). Plant Proteins: Assessing Their Nutritional Quality and Effects on Health and Physical Function. Nutrients, 12(12), 3704. https://doi.org/10.3390/nu12123704





