What Model of Nutrition Can Be Recommended to People Ending Their Professional Sports Career? An Analysis of the Mediterranean Diet and the CRON Diet in the Context of Former Athletes
Abstract
1. Introduction
2. Mediterranean Diet
3. Calorie Restriction with Optimal Nutrition Diet (CRON Diet)
4. Application of the Mediterranean and the CRON Diet in a Group of Former Athletes
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CRON | Calorie Restriction with Optimal Nutrition diet |
| NAFLD | Non-alcoholic fatty liver disease |
| HOMA | Homeostatic model assessment |
| HOMA-%β-cell | HOMA β-cell function |
| HOMA-IR | HOMA insulin resistance |
| DHA | docosahexaenoic acid |
| TXA2 | Thromboxane A2 |
| PGE2 | Prostaglandin E2 |
| COX-1 | cyclooxygenase 1 |
| COX-2 | cyclooxygenase 2 |
| CALERIE | Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy |
| IGF-1 | insulin-like growth factor |
| IR | insulin resistance |
| FOX | forkhead box |
| Bcl-XL protein | B-cell lymphoma-extra large |
| PNC-1 | pyrazinamidase/nicotinamidase-1 |
| PPARγ | peroxisome proliferator-activated receptor γ |
| CREBP | cAMP-response element-binding protein |
| PKA | protein kinase A |
| ChREBP | carbohydrate-responsive element-binding protein |
References
- Pilis, T.; Pilis, K.; Pluta, R.; Wroński, Z.; Pilis, W.; Garbrys, T.; Michalik, A. Recreational physical activity of former athletes and its consequences. Sci. Works Jan Długosz Acad. Częstochowa Phys. Cult. 2005, 6, 223–232. (In Polish) [Google Scholar]
- Arliani, G.G.; Lara, P.S.; Astur, D.C.; Cohen, M.; Gonçalves, J.P.P.; Ferretti, M. Impact of sports on health of former professional soccer players in Brazil. Acta Ortop. Bras. 2014, 22, 188–190. [Google Scholar] [CrossRef][Green Version]
- Hulbert, A.J.; Else, P.L. Basal Metabolic Rate: History, Composition, Regulation, and Usefulness. Physiol. Biochem. Zool. 2004, 77, 869–876. [Google Scholar] [CrossRef] [PubMed]
- King, N.A.; Caudwell, P.; Hopkins, M.; Byrne, N.M.; Colley, R.; Hills, A.P.; Stubbs, J.R.; Blundell, J.E. Metabolic and behavioral compensatory responses to exercise interventions: Barriers to weight loss. Obesity 2007, 15, 1373–1383. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, F.N.; Kuniyoshi, F.H.S.; Calvin, A.D.; Sierra-Johnson, J.; Romero-Corral, A.; Lopez-Jimenez, F.; George, C.F.; Rapoport, D.M.; Vogel, R.A.; Khandheria, B.; et al. Sleep-disordered breathing, Hypertension and Obesity in Retired National Football League Players. J. Am. Coll. Cardiol. 2010, 56, 1432–1433. [Google Scholar] [CrossRef]
- Marquet, L.A.; Brown, M.; Tafflet, M.; Nassif, H.; Mouraby, R.; Bourhaleb, S.; Toussaint, J.F.; Desgorces, F.D. No effect of weight cycling on the post-career BMI of weight class elite athletes. BMC Public Health 2013, 13, 510. [Google Scholar] [CrossRef]
- Hyman, M.H.; Dang, D.L.; Liu, Y. Differences in Obesity Measures and Selected Comorbidities in Former National Football League Professional Athletes. J. Occup. Environ. Med. 2012, 54, 816–819. [Google Scholar] [CrossRef]
- Abbey, E.L.; Wright, C.J.; Kirkpatrick, C.M. Nutrition practices and knowledge among NCAA Division III football players. J. Int. Soc. Sports Nutr. 2017, 14, 13. [Google Scholar] [CrossRef]
- Michalczyk, M.M.; Zając, A.; Mikołajec, K.; Zydek, G.; Langfort, J. No Modification in Blood Lipoprotein Concentration but Changes in Body Composition after 4 Weeks of Low Carbohydrate Diet (LCD) Followed by 7 Days of Carbohydrate Loading in Basketball Players. J. Hum. Kinet. 2018, 65, 125–137. [Google Scholar] [CrossRef]
- Michalczyk, M.M.; Chycki, J.; Zajac, A.; Maszczyk, A.; Zydek, G.; Langfort, J. Anaerobic Performance after a Low-Carbohydrate Diet (LCD) Followed by 7 Days of Carbohydrate Loading in Male Basketball Players. Nutrients 2019, 11, 778. [Google Scholar] [CrossRef]
- Borchers, J.R.; Clem, K.L.; Habash, D.L.; Nagaraja, H.N.; Stokley, L.M.; Best, T.M. Metabolic syndrome and insulin resistance in Division 1 collegiate football players. Med. Sci. Sports Exerc. 2009, 41, 2105–2110. [Google Scholar] [CrossRef] [PubMed]
- Kujala, U.M.; Kaprio, J.; Taimela, S.; Sarna, S. Prevalence of diabetes, hypertension, and ischemic heart Obesity in College Rowers 455 disease in former elite athletes. Metabolism 1994, 43, 1255–1260. [Google Scholar] [CrossRef]
- Czerwińska, M.; Hołowko, J.; Maciejewska, D.; Wysokiński, P.; Ficek, K.; Wilk, P.; Stachowska, E. Caloric Restriction Diet (CR Diet) or Mediterranean Diet (MD)—Which is the Best Choice for Former Athletes? Cent. Eur. J. Sport Sci. Med. 2016, 13, 23–35. [Google Scholar] [CrossRef]
- Ju, S.Y.; Lee, J.Y.; Kim, D.H. Association of metabolic syndrome and its components with all-cause and cardiovascular mortality in the elderly: A meta-analysis of prospective cohort studies. Medicine (Baltimore) 2017, 96, e8491. [Google Scholar] [CrossRef] [PubMed]
- Farias, D.L.; Tibana, R.A.; Teixeira, T.G.; Vieira, D.C.; Tarja, V.; Nascimento Dda, C.; de Oliveira Silva, A.; Funghetto, S.S.; de Sousa Coura, M.A.; Valduga, R.; et al. Elderly women with metabolic syndrome present higher cardiovascular risk and lower relative muscle strength. Einstein (Sao Paulo) 2013, 11, 174–179. [Google Scholar] [CrossRef][Green Version]
- Emami, M.; Behforouz, A.; Jarahi, L.; Zarifian, A.; Rashidlamir, A.; Rashed, M.M. The Risk of Developing Obesity, Insulin Resistance, and Metabolic Syndrome in Former Power-sports Athletes-Does Sports Career Termination Increase the Risk. Indian J. Endocrinol. Metab. 2018, 22, 515–519. [Google Scholar]
- Maznev, I.; Grigorov, V.; Goldberg, L.; Ignatovic, G. Typical electrocardiographic deviations after cessation of physical activities in elite sportsmen. Cardiovasc. J. S. Afr. 2003, 14, 12–15. [Google Scholar]
- Apor, P.; Radi, A. Master sportsmen. Health status and life expectancies of physically active elderly. Orv. Hetil. 2010, 151, 110–113. [Google Scholar] [CrossRef]
- Sirotin, A.B.; Belozerova, L.M.; Sergeeva, I.G.; Zhukov, V.N.; Kolegova, N.G. The influence of different types of physical exertions on the mature males’ biological age. Adv. Gerontol. 2014, 27, 179–183. [Google Scholar]
- Melanson, E.L. The effect of exercise on non-exercise physical activity and sedentary behavior in adults. Obes. Rev. 2017, 18, 40–49. [Google Scholar] [CrossRef]
- Hansen, E.A.; Emanuelsen, A.; Gertsen, R.; Sørensen, S.R. Improved Marathon Performance by In-Race Nutritional Strategy Intervention. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 645–655. [Google Scholar] [CrossRef]
- O’Kane, J.W.; Teitz, C.C.; Fontana, S.M.; Lind, B.K. Prevalence of obesity in adult population of former college rowers. J. Am. Board Fam. Pract. 2002, 15, 451–456. [Google Scholar]
- Sarna, S.; Sahi, T.; Koskenvuo, M.; Kaprio, J. Increased life expectancy of world class male athletes. Med. Sci. Sports Exerc. 1993, 25, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Pihl, E.; Jurimae, T. Relationships between body weight change and cardiovascular disease risk factors in male former athletes. Int. J. Obes. 2001, 25, 1057–1062. [Google Scholar] [CrossRef] [PubMed]
- Hołowko, J.; Michalczyk, M.M.; Zając, A.; Czerwińska-Rogowska, M.; Ryterska, K.; Banaszczak, M.; Jakubczyk, K.; Stachowska, E. Six Weeks of Calorie Restriction Improves Body Composition and Lipid Profile in Obese and Overweight Former Athletes. Nutrients 2019, 11, 1461. [Google Scholar] [CrossRef] [PubMed]
- Altomare, R.; Cacciabaudo, F.; Damiano, G.; Palumbo, V.D.; Gioviale, M.C.; Bellavia, M.; Tomasello, G.; Lo Monte, A.I. The Mediterranean Diet: A History of Health. Iran. J. Public Health 2013, 42, 449–457. [Google Scholar]
- Malikowska, K.; Grabańska-Martyńska, K. History of the Mediterranean diet in the prevention of cardiovascular diseases. Acta Med. Pol. 2016, 6, 41–49. (In Polish) [Google Scholar]
- Keys, A. Mediterranean diet and public health: Personal reflections. Am. J. Clin. Nutr. 1995, 61, 1321–1323. [Google Scholar] [CrossRef]
- Bogacka, A.; Nawrot, B.; Gryza, M.; Balejko, E.; Kucharska, E. An assessment of the knowledge and compliance with the principles of the Mediterranean diet in patients of cardiology clinics. Żywn. Nauka Technol. Jakość 2008, 2, 128–139. (In Polish) [Google Scholar]
- Menotti, A.; Kromhout, D.; Blackburn, H.; Fidanza, F.; Buzina, R.; Nissinen, A. Food intake patterns and 25-year mortality from coronary heart disease: Cross-cultural correlations in the seven countries study. Eur. J. Epidemiol. 1999, 15, 507–515. [Google Scholar] [CrossRef]
- Kromhout, D.; Menotti, A.; Bloemberg, B.; Aravanis, C.; Blackburn, H.; Buzina, R.; Dontas, A.S.; Fidanza, F.; Giampaoli, S.; Jansen, A.; et al. Dietary saturated and trans fatty acids and cholesterol and 25-year mortality from coronary heart disease: The seven countries study. Prev. Med. 1995, 24, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- The European Society of Cardiology (ESC) Working Group on diabetes, pre-diabetes and cardiovascular diseases in collaboration with the European Society for the Study of Diabetes (EASD): European Society of Cardiology guidelines on diabetes and pre-diabetes coexisting with cardiovascular disease, developed in collaboration with the European Society for the Study of Diabetes. Eur. Heart J. 2020, 2, 255–323.
- Gajewska, D.; Pałkowska-Goździk, E.; Lange, E.; Niegowska, J.; Pasko, P.; Kościołek, A.; Fibich, K.; Gudej, S. Standards of dietary management in cardiology in adults. 2016 Opinion of the Polish Society of Dietetics. Dietetyka 2016, 9. (In Polish) [Google Scholar]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet: A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef]
- Food Pyramid Adapted to the Recommendations of the Mediterranean Diet. Available online: https://www.oldwayswebstore.org/make-every-day-mediterranean-an-oldways-4-week-menu-plan-book/ (accessed on 17 June 2020).
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef]
- Ministry of health and welfare, supreme scientific health council: Dietary guidelines for adults in Greece. Arch. Hell. Med. 1999, 16, 516–524.
- Trichopoulou, A.; Lagiou, P. Healthy traditional Mediterranean diet: An expression of culture, history, and lifestyle. Nutr. Rev. 1997, 55, 383–389. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.N.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- Chudzińska, M.; Nartowicz, M.; Zukow, W.; Sinkiewicz, W. Why the Mediterranean diet? J. Educ. Health Sport 2018, 8, 326–337. [Google Scholar]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean Diet, its Components, and Cardiovascular Disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Pintó, X.; Fanlo-Maresma, M.; Corbella, E.; Corbella, X.; Mitjavila, M.T.; Moreno, J.J. A Mediterranean Diet Rich in Extra-Virgin Olive Oil Is Associated with a Reduced Prevalence of Nonalcoholic Fatty Liver Disease in Older Individuals at High Cardiovascular Risk. J. Nutr. 2019, 149, 1920–1929. [Google Scholar] [CrossRef] [PubMed]
- Bower, A.; Marquez, S.; de Mejia, E.G. The Health Benefits of Selected Culinary Herbs and Spices Found in the Traditional Mediterranean Diet. Crit. Rev. Food Sci. Nutr. 2016, 56, 2728–2746. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Merino, J.; Sun, Q.; Fitó, M.; Salas-Salvadó, J. Dietary Polyphenols, Mediterranean Diet, Prediabetes, and Type 2 Diabetes: A Narrative Review of the Evidence. Oxid. Med. Cell. Longev. 2017, 2017, 6723931. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Ros, E.; Valderas-Martinez, P.; Casas, R.; Arranz, S.; Guillén, M.; Lamuela-Raventós, R.M.; Llorach, R.; Andres-Lacueva, C.; et al. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: A randomized clinical trial. Clin. Nutr. 2013, 32, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Chudzińska, M.; Wołowiec, Ł.; Zukow, W.; Sinkiewicz, W. Mediterranean diet recommended not only in cardiovascular diseases. J. Educ. Health Sport 2017, 7, 732–746. [Google Scholar]
- Szostak, W.B.; Cybulska, B.; Kłosiewicz-Latoszek, L.; Szostak-Węgierek, D. Prevention of cardiovascular diseases-progress in 2013. Med. Prakt. 2014, 7–8, 16–27. (In Polish) [Google Scholar]
- de Luis, D.A.; Primo, D.; Izaola, O.; Gomez Hoyos, E.; Lopez Gomez, J.J.; Ortola, A.; Aller, R. The role of the variant in adiponectin gene rs266729 on weight loss and cardiovascular risk factors after a hypocaloric diet with Mediterranean pattern. Nutrition 2019, 60, 1–5. [Google Scholar] [CrossRef]
- Esposito, K.; Maiorino, M.I.; Bellastella, G.; Panagiotakos, D.B.; Giugliano, D. Mediterranean diet for type 2 diabetes: Cardiometabolic benefits. Endocrine 2017, 56, 27–32. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for NAFLD; evidence and plausible mechanisms. Liver Int. 2017, 37, 936–949. [Google Scholar] [CrossRef]
- Petersson, S.D.; Philippou, E. Mediterranean diet, cognitive function, and dementia: A systematic review of the evidence. Adv Nutr. 2016, 7, 889–904. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Stubbs, B.; Noale, M.; Solmi, M.; Luchini, C.; Maggi, S. Adherence to the Mediterranean diet is associated with better quality of life: Data from the Osteoarthritis Initiative. Am. J. Clin. Nutr. 2016, 104, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Sugioka, Y.; Tada, M.; Okano, T.; Mamoto, K.; Inui, K.; Habu, D.; Koike, T. Monounsaturated fatty acids might be key factors in the Mediterranean diet that suppress rheumatoid arthritis disease activity: The TOMORROW study. Clin. Nutr. 2017, 37, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Zappalà, G.; Bernardini, S.; Giambini, I.; Bes-Rastrollo, M.; Martinez-Gonzalez, M. Adherence to the Mediterranean diet is inversely associated with metabolic syndrome occurrence: A metaanalysis of observational studies. Int. J. Food Sci. Nutr. 2017, 68, 138–148. [Google Scholar] [CrossRef]
- Berrino, F. Mediterranean Diet and its association with reduced invasive breast cancer risk. JAMA Oncol. 2016, 2, 535–536. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Does a Mediterranean-type diet reduce cancer risk? Curr. Nutr. Rep. 2016, 5, 9–17. [Google Scholar] [CrossRef]
- Rosato, V.; Guercio, V.; Bosetti, C.; Negri, E.; Serraino, D.; Giacosa, A.; Montella, M.; La Vecchia, C.; Tavani, A. Mediterranean diet and colorectal cancer risk: A pooled analysis of three Italian case-control studies. Br. J. Cancer 2016, 115, 862–865. [Google Scholar] [CrossRef]
- Giraldi, L.; Panic, N.; Cadoni, G.; Boccia, S.; Leoncini, E. Association between Mediterranean diet and head and neck cancer: Results of a large case-control study in Italy. Eur. J. Cancer Prev. 2016, 26, 418–423. [Google Scholar] [CrossRef]
- Bifulco, M. How does the Mediterranean diet decrease the risk of a variety of cancers? Eur. J. Clin. Nutr. 2015, 69, 1372. [Google Scholar] [CrossRef]
- Rienks, J.; Dobson, A.J.; Mishra, G.D. Mediterranean dietary pattern and prevalence and incidence of depressive symptoms in mid-aged women: Results from a large community-based prospective study. Eur. J. Clin. Nutr. 2013, 67, 75–82. [Google Scholar] [CrossRef]
- Jacka, F.N.; O’Neil, A.; Opie, R.; Itsiopoulos, C.; Cotton, S.; Mohebbi, M.; Castle, D.; Dash, S.; Mihalopoulos, C.; Chatterton, M.L.; et al. A randomised controlled trial of dietary improvement for adults with major depression (the ‘SMILES’ trial). BMC Med. 2017, 15, 23. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Díaz-López, A.; Ruiz-Canela, M.; Basora, J.; Fitó, M.; Corella, D. Effect of a Lifestyle Intervention Program with Energy-Restricted Mediterranean Diet and Exercise on Weight Loss and Cardiovascular Risk Factors: One-Year Results of the PREDIMED-Plus Trial. Diabetes Care 2019, 42, 777–788. [Google Scholar] [CrossRef]
- Maciejewska, D.; Michalczyk, M.; Czerwińska-Rogowska, M.; Banaszczak, M.; Ryterska, K.; Jakubczyk, K.; Piotrowski, J.; Hołowko, J.; Drozd, A.; Wysokiński, P.; et al. Seeking Optimal Nutrition for Healthy Body Mass Reduction among Former Athletes. J. Hum. Kinet. 2017, 60, 63–75. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C. Omega-3 fatty acids in cardiovascular disease—An uphill battle. Prostaglandins Leukot. Essent. Fat. Acids 2015, 92, 41–47. [Google Scholar] [CrossRef]
- Burke, M.F.; Burke, F.M.; Soffer, D.E. Review of Cardiometabolic Effects of Prescription Omega-3 Fatty Acids. Curr. Atheroscler. Rep. 2017, 19, 60. [Google Scholar] [CrossRef]
- Gammone, M.A.; Riccioni, G.; Parrinello, G.; D’Orazio, N. Omega-3 Polyunsaturated Fatty Acids: Benefits and Endpoints in Sport. Nutrients 2018, 11, 46. [Google Scholar] [CrossRef]
- Shei, R.J.; Lindley, M.R.; Mickleborough, T.D. Omega-3 polyunsaturated fatty acids in the optimization of physical performance. Mil. Med. 2014, 179, 144–156. [Google Scholar] [CrossRef]
- Goel, A.; Pothineni, N.V.; Singhal, M.; Paydak, H.; Saldeen, T.; Mehta, J.L. Fish, Fish Oils and Cardioprotection: Promise or Fish Tale? Int. J. Mol. Sci. 2018, 19, 3703. [Google Scholar] [CrossRef]
- Mashima, R.; Okuyama, T. The role of lipoxygenases in pathophysiology; new insights and future perspectives. Redox Biol. 2015, 6, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Funk, C.D. Lipoxygenase pathways as mediators of early inflammatory events in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1204–1206. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Bulló, M.; Babio, N.; Martínez-González, M.A.; Estruch, R.; Covas, M.I.; Wärnberg, J.; Arós, F.; Lapetra, J.; Serra-Majem, L.; et al. Mediterranean diet and risk of hyperuricemia in elderly participants at high cardiovascular risk. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 1263–1270. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, K.E.; Parma, D.L.; Muñoz, E.; Spaniol, M.; Wargovich, M.; Ramirez, A.G. Dietary intervention among breast cancer survivors increased adherence to a Mediterranean-style, anti-inflammatory dietary pattern: The Rx for Better Breast Health Randomized Controlled Trial. Breast Cancer Res. Treat. 2019, 173, 145–154. [Google Scholar] [CrossRef]
- Yubero-Serrano, E.M.; Gonzalez-Guardia, L.; Rangel-Zuñiga, O.; Delgado-Lista, J.; Gutierrez-Mariscal, F.M.; Perez-Martinez, P.; Delgado-Casado, N.; Cruz-Teno, C.; Tinahones, F.J.; Villalba, J.M.; et al. Mediterranean diet supplemented with coenzyme Q10 modifies the expression of proinflammatory and endoplasmic reticulum stress-related genes in elderly men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 3–10. [Google Scholar] [CrossRef]
- Itsiopoulos, C.; Brazionis, L.; Kaimakamis, M.; Cameron, M.; Best, J.D.; O’Dea, K.; Rowley, K. Can the Mediterranean diet lower HbA1c in type 2 diabetes? Results from a randomized cross-over study. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 740–747. [Google Scholar] [CrossRef]
- Sharples, A.P.; Hughes, D.C.; Deane, C.S.; Saini, A.; Selman, C.; Stewart, C.E. Longevity and skeletal muscle mass: The role of IGF signalling, the sirtuins, dietary restriction and protein intake. Aging Cell 2015, 14, 511–523. [Google Scholar] [CrossRef]
- Holloszy, J.O.; Fontana, L. Caloric Restriction in Humans. Exp. Gerontol. 2007, 42, 709–712. [Google Scholar] [CrossRef]
- Fontana, L.; Meyer, T.E.; Klein, S.; Holloszy, J.O. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 6659–6663. [Google Scholar] [CrossRef]
- Larson-Meyer, D.E.; Heilbronn, L.K.; Redman, L.M.; Newcomer, B.R.; Frisard, M.I.; Anton, S.; Smith, S.R.; Alfonso, A.; Ravussin, E. Effect of calorie restriction with or without exercise on insulin sensitivity, beta-cell function, fat cell size, and ectopic lipid in overweight subjects. Diabetes Care 2006, 29, 1337–1344. [Google Scholar] [CrossRef]
- Wątroba, M.; Szukiewicz, D. The role of sirtuins in aging and age-related diseases. Adv. Med. Sci. 2016, 61, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Ungvari, Z.; Minor, R.K.; Le Couteur, D.G.; de Cabo, R. Are sirtuins viable targets for improving healthspan and lifespan? Nat. Rev. Drug Discov. 2012, 11, 443–461. [Google Scholar] [CrossRef]
- Kagawa, Y. Impact of Westernization on the nutrition of Japanese: Changes in physique, cancer, longevity and centenarians. Prev. Med. 1978, 7, 205–217. [Google Scholar] [CrossRef]
- Redman, L.M.; Ravussin, E. Caloric restriction in humans: Impact on physiological, psychological, and behavioral outcomes. Antioxid. Redox Signal. 2011, 14, 275–287. [Google Scholar] [CrossRef] [PubMed]
- Most, J.; Tosti, V.; Redman, L.M.; Fontana, L. Calorie restriction in humans: An Update. Ageing Res. Rev. 2017, 39, 36–45. [Google Scholar] [CrossRef]
- Weiss, E.P.; Racette, S.B.; Villareal, D.T.; Fontana, L.; Steger-May, K.; Schechtman, K.B.; Klein, S.; Holloszy, J.O.; Washington University School of Medicine CALERIE Group. Improvements in glucose tolerance and insulin action induced by increasing energy expenditure or decreasing energy intake: A randomized controlled trial. Am. J. Clin. Nutr. 2006, 84, 1033–1042. [Google Scholar] [PubMed]
- Larson-Meyer, D.E.; Redman, L.; Heilbronn, L.K.; Martin, C.K.; Ravussin, E. Caloric Restriction with or without Exercise. Med. Sci. Sport Exerc. 2010, 42, 152–159. [Google Scholar] [CrossRef]
- Ros, M.; Carrascosa, J.M. Current Nutritional and Pharmacological Anti-Aging Interventions. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165612. [Google Scholar] [CrossRef]
- Escobar, K.A.; Cole, N.H.; Mermier, C.M.; Van Dusseldorp, T.A. Autophagy and aging: Maintaining the proteome through exercise and caloric restriction. Aging Cell 2019, 18, e12876. [Google Scholar] [CrossRef]
- Madeo, F.; Zimmermann, A.; Maiuri, M.C.; Kroemer, G. Essential role for autophagy in life span extension. J. Clin. Investig. 2015, 125, 85–93. [Google Scholar] [CrossRef]
- Trexler, E.T.; Smith-Ryan, A.E.; Norton, L.E. Metabolic adaptation to weight loss: Implications for the athlete. J. Int. Soc. Sports Nutr. 2014, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, K.; Habboush, Y.; Sultan, S. Intermittent Fasting: The Choice for a Healthier Lifestyle. Cureus 2018, 10, e2947. [Google Scholar] [CrossRef] [PubMed]
- Moro, T.; Tinsley, G.; Bianco, A.; Marcolin, G.; Pacelli, Q.F.; Battaglia, G.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. J. Transl. Med. 2016, 14, 290. [Google Scholar] [CrossRef] [PubMed]
- Arnason, T.G.; Bowen, M.W.; Mansell, K.D. Effects of intermittent fasting on health markers in those with type 2 diabetes: A pilot study. World J. Diabetes 2017, 8, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Larson-Meyer, D.E.; Newcomer, B.R.; Heilbronn, L.K.; Volaufova, J.; Smith, S.R.; Alfonso, A.J.; Lefevre, M.; Rood, J.C.; Williamson, D.A.; Ravussin, E.; et al. Effect of 6-month calorie restriction and exercise on serum and liver lipids and markers of liver function. Obesity 2008, 16, 1355–1362. [Google Scholar] [CrossRef] [PubMed]
- Manore, M.M. Effect of physical activity on thiamine, riboflavin and vitamin B-6 requirements. Am. J. Clin. Nutr. 2000, 72, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Pons, V.; Riera, J.; Capó, X.; Martorell, M.; Sureda, A.; Tur, J.A.; Drobnic, F.; Pons, A. Calorie restriction regime enhances physical performance of trained athletes. J. Int. Soc. Sports Nutr. 2018, 15, 12. [Google Scholar] [CrossRef]
- van der Beek, E.J.; van Dokkum, W.; Wedel, M.; Schrijver, J.; van den Berg, H. Thiamin, riboflavin and vitamin B6: Impact of restricted intake on physical performance in man. J. Am. Coll. Nutr. 1994, 13, 629–640. [Google Scholar] [CrossRef]
- National Institutes of Health U.S. Department of Health & Human Services. Vitamin B6. Available online: http://ods.od.nih.gov/factsheets/VitaminB6-HealthProfessional/ (accessed on 11 December 2019).
- Linus Pauling Institute. Micronutrient Research for Optimum Health. Vitamin B6. Available online: http://lpi.oregonstate.edu/infocenter/vitamins/vitaminB6/ (accessed on 11 December 2019).
- National Institutes of Health U.S. National Library of Medicine. Vitamin B6 [online]. Available online: http://www.nlm.nih.gov/medlineplus/ency/article/002402.htm (accessed on 11 December 2019).
- Kimura, K.D.; Tissenbaum, H.A.; Liu, Y.; Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 1997, 277, 942–946. [Google Scholar] [CrossRef]
- Pierce, S.B.; Costa, M.; Wisotzkey, R.; Devadhar, S.; Homburger, S.A.; Buchman, A.R.; Ferguson, K.C.; Heller, J.; Platt, D.M.; Pasquinelli, A.A.; et al. Regulation of DAF-2 receptor signaling by human insulin and ins-1, a member of the unusually large and diverse C. elegans insulin gene family. Genes Dev. 2001, 15, 672–686. [Google Scholar] [CrossRef]
- Murphy, C.T.; McCarroll, S.A.; Bargmann, C.I.; Fraser, A.; Kamath, R.S.; Ahringer, J. Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 2003, 424, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Calnan, D.R.; Brunet, A. The FoxO code. Oncogene 2008, 27, 2276–2288. [Google Scholar] [CrossRef]
- Maiese, K.; Chong, Z.Z.; Shang, Y.C. OutFOXOing disease and disability: The therapeutic potential of targeting FoxO proteins. Trends Mol. Med. 2008, 14, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Barthel, A.; Schmoll, D.; Unterman, T.G. FoxO proteins in insulin action and metabolism. Trends Endocrinol. Metab. 2005, 16, 183–189. [Google Scholar] [CrossRef]
- Hedrick, S.M. The cunning little vixen: Foxo and the cycle of life and death. Nat. Immunol. 2009, 10, 1057–1063. [Google Scholar] [CrossRef]
- Cheng, Z.; White, M.F. Targeting Forkhead Box O1 from the Concept to Metabolic Diseases: Lessons from Mouse Models. Antioxid. Redox Signal. 2011, 14, 649–661. [Google Scholar] [CrossRef]
- Birkenkamp, K.U.; Coffer, P.J. Regulation of cell survival and proliferation by the FOXO (Forkhead box, class 0) subfamily of forkhead transcription factors. Biochem. Soc. Trans. 2003, 31, 292–297. [Google Scholar] [CrossRef]
- Dai, H.; Sinclair, D.A.; Ellis, J.L.; Steegborn, C. Sirtuin Activators and Inhibitors: Promises, Achievements, and Challenges. Pharmacol. Ther. 2018, 188, 140–154. [Google Scholar] [CrossRef]
- Wiercińska, M.; Rosołowska-Huszcz, D. Natural and synthetic modulators of sirtuin activity. Kosmos 2017, 66, 365–377. (In Polish) [Google Scholar]
- Kaeberlein, M.; Hu, D.; Kerr, E.O.; Tsuchiya, M.; Westman, E.A.; Dang, N.; Fields, S.; Kennedy, B.K. Increased life span due to calorie restriction in respiratory-deficient yeast. PLoS Genet. 2005, 1, e69. [Google Scholar] [CrossRef]
- Gawęcka, A.; Stachowska, E. Molecular mechanism of caloric restriction. Ann. Acad. Med. Stetin. 2014, 60, 25–28. (In Polish) [Google Scholar] [PubMed]
- Guarente, L. Calorie restriction and sirtuins revisited. Genes Dev. 2013, 27, 2072–2085. [Google Scholar] [CrossRef] [PubMed]
- Favero, G.; Franceschetti, L.; Rodella, L.F.; Rezzani, R. Sirtuins, aging, and cardiovascular risks. Age (Dordr.) 2015, 37, 65. [Google Scholar] [CrossRef]
- North, B.J.; Verdin, E. Sirtuins: Sir2-related NAD-dependent protein deacetylases of FOXO transcription factors by the SIRT1 deacetylase. Science 2004, 303, 2011–2015. [Google Scholar]
- Picca, A.; Pesce, V.; Lezza, A.M.S. Does eating less make you live longer and better? An update on calorie restriction. Clin. Interv. Aging 2017, 12, 1887–1902. [Google Scholar] [CrossRef]
- Hołowko, J. CRON Diet as an Alternative for Sportsmen That Are Finishing Their Sports Career. Ph.D. Thesis, Pomeranian Medical University in Szczecin, Szczecin, Poland, 2020. Available online: https://ppm.pum.edu.pl/info/phd/PUM549b68fef5934d93bf20818693fc87ba/ (accessed on 17 November 2020). (In Polish).
- Gawęcka, A.; Ryterska, K.; Wysokiński, P.; Ficek, K.; Wilk, K.; Banaszczak, M.; Jakubczyk, K.; Piotrowski, J.; Czerwińska, M.; Hołowko, J.; et al. Changes in body mass and blood lipid levels in patients undergoing caloric restriction. Pomeranian J. Life Sci. 2014, 60, 29–33. (In Polish) [Google Scholar]
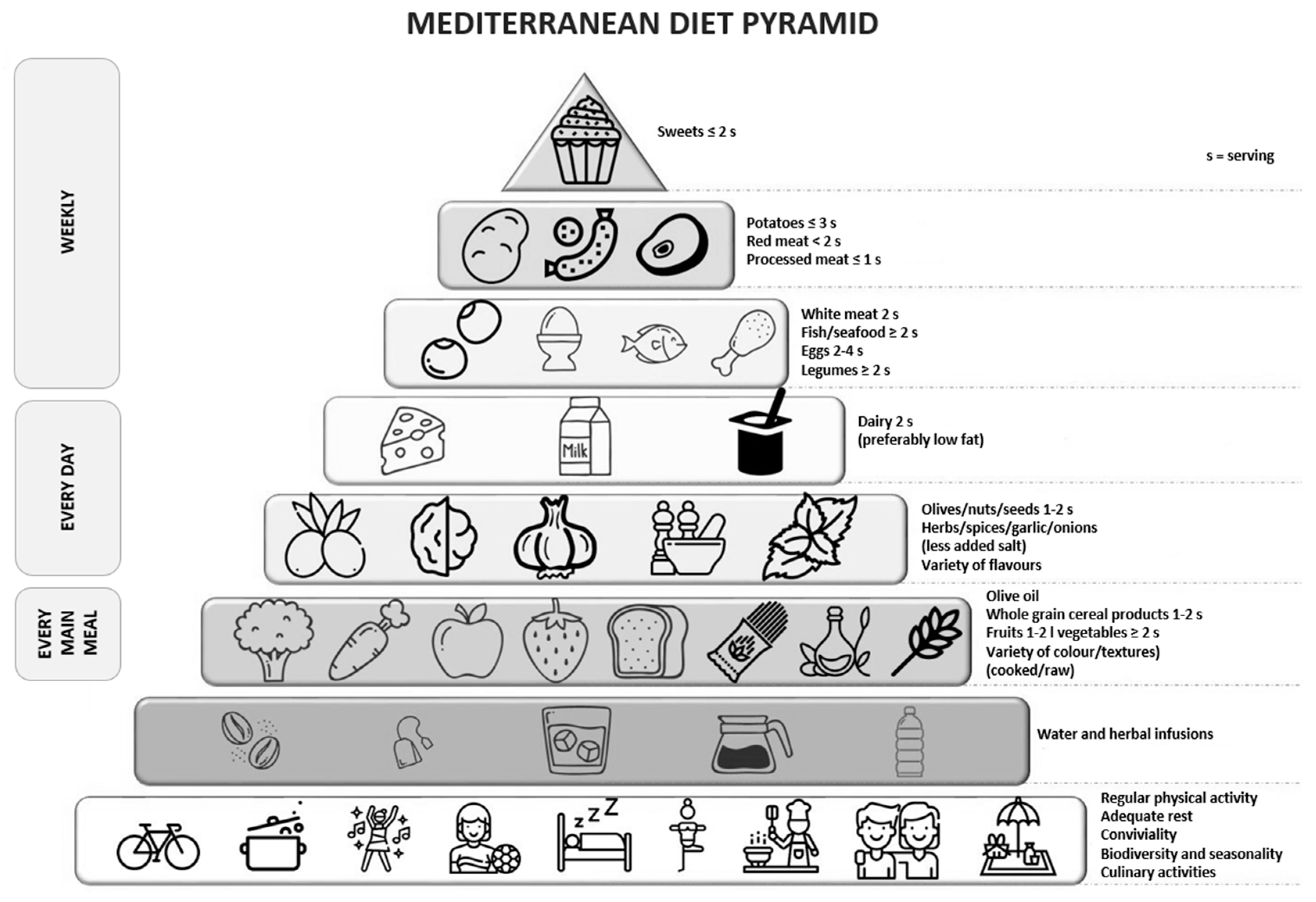
| Ingredients of the Diet | Make Every Day Mediterranean: An Oldways 4-Week Menu Plan Book (2019) | Mediterranean Diet Foundation (2011) | Greek Dietary Guidelines (1999) |
|---|---|---|---|
| Olive oil | in every meal | in every meal | main fat added |
| Vegetables | in every meal | ≥2 portions in each meal | 6 portions a day |
| Fruits | in every meal | 1–2 portions in each meal | 3 portions a day |
| Grains | in every meal | 1–2 portions in each meal | 8 portions a day |
| Legumes | in every meal | ≥2 portions in each meal | 3–4 portions per week |
| Nuts | in every meal | 1–2 portions every day | 3–4 portions per week |
| Fish/seafood | often, at least twice a week | ≥2 portions per week | 5–6 portions per week |
| Eggs | moderate portions, daily up to once a week | 2–4 portions per week | 3 portions per week |
| Poultry | moderate portions, daily to once a week | 2 portions per week | 4 portions per week |
| Dairy products | moderate portions, daily to once a week | 2 portions a day | 2 portions a day |
| Red meat | less often | <2 portions per week | 4 portions per month |
| Sweets | less often | <2 portions per week | 3 portions per week |
| Red wine | moderately | with moderation and respect to social beliefs | every day but in moderation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hołowko-Ziółek, J.; Cięszczyk, P.; Biliński, J.; Basak, G.W.; Stachowska, E. What Model of Nutrition Can Be Recommended to People Ending Their Professional Sports Career? An Analysis of the Mediterranean Diet and the CRON Diet in the Context of Former Athletes. Nutrients 2020, 12, 3604. https://doi.org/10.3390/nu12123604
Hołowko-Ziółek J, Cięszczyk P, Biliński J, Basak GW, Stachowska E. What Model of Nutrition Can Be Recommended to People Ending Their Professional Sports Career? An Analysis of the Mediterranean Diet and the CRON Diet in the Context of Former Athletes. Nutrients. 2020; 12(12):3604. https://doi.org/10.3390/nu12123604
Chicago/Turabian StyleHołowko-Ziółek, Joanna, Paweł Cięszczyk, Jarosław Biliński, Grzegorz W. Basak, and Ewa Stachowska. 2020. "What Model of Nutrition Can Be Recommended to People Ending Their Professional Sports Career? An Analysis of the Mediterranean Diet and the CRON Diet in the Context of Former Athletes" Nutrients 12, no. 12: 3604. https://doi.org/10.3390/nu12123604
APA StyleHołowko-Ziółek, J., Cięszczyk, P., Biliński, J., Basak, G. W., & Stachowska, E. (2020). What Model of Nutrition Can Be Recommended to People Ending Their Professional Sports Career? An Analysis of the Mediterranean Diet and the CRON Diet in the Context of Former Athletes. Nutrients, 12(12), 3604. https://doi.org/10.3390/nu12123604






