Iron Status is Associated with Mood, Cognition, and Functional Ability in Older Adults: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Laboratory Analyses
2.3. Neurocognitive and Physical Functional Assessment
2.4. Nutritional Status and Anthropometric Characterization
2.5. Statistical Analysis
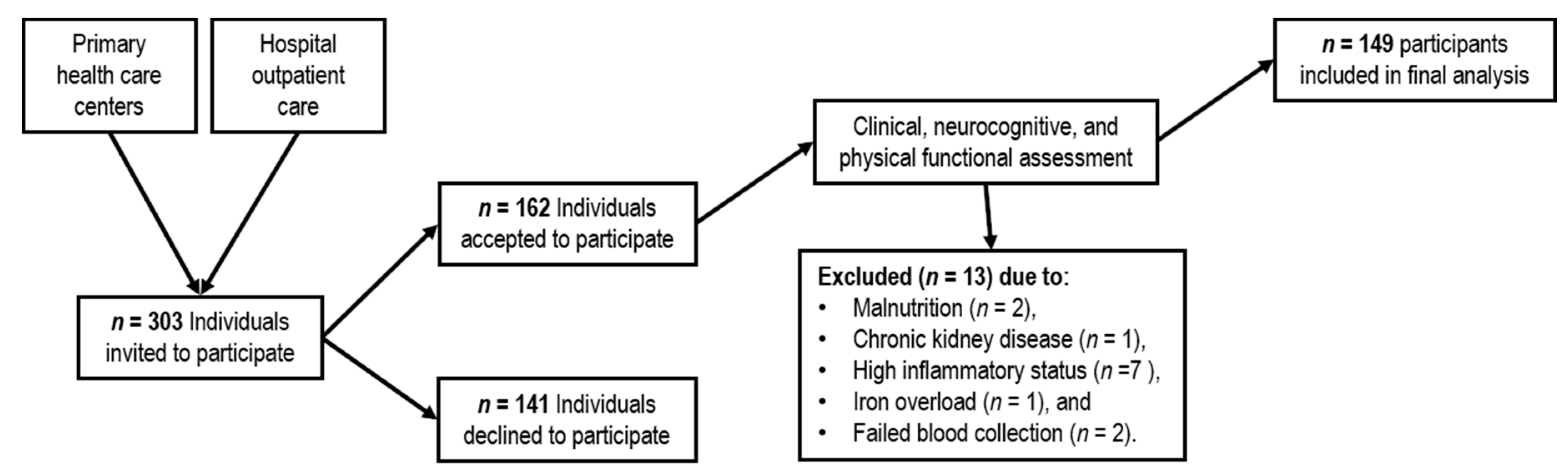
3. Results
3.1. Sample Characterization
3.2. Mood, Cognitive and Functional Characterization
3.3. Hematological Characterization
3.4. Nutritional Status is a Predictor of ID
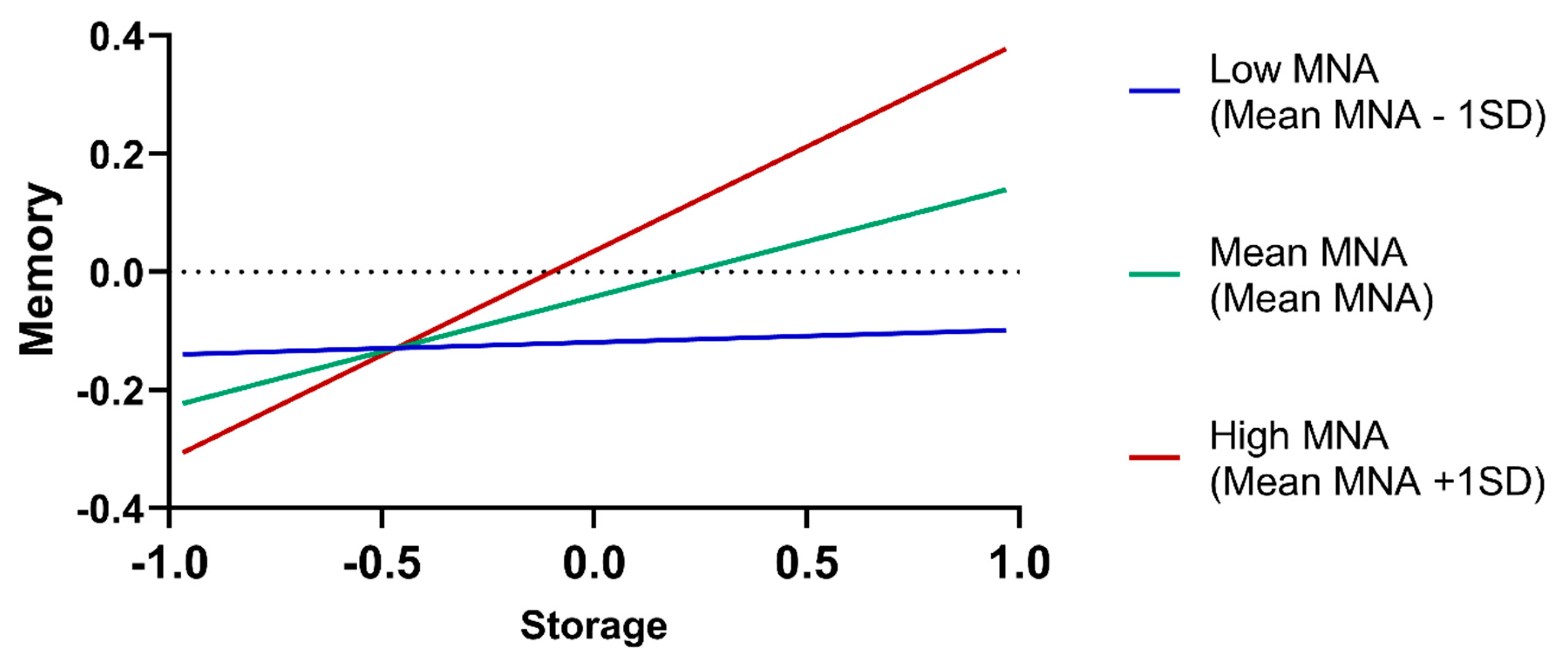
3.5. Iron Hematological Dimensions Predict Memory, Mood and Functional Ability
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variables (Mean; SD) | Iron Sufficiency | Iron Deficiency | |
|---|---|---|---|
| Psychological | t(df); p; Cohen’s d | ||
| 10.47; 5.89 | 13.20; 6.97 | −2.449(147); 0.016; 0.443 | |
| Cognitive | t(df); p; Cohen’s d | ||
| DS—Forward | 7.70; 2.24 | 6.7; 2.13 | 0.714(147); 0.476; 0.129 |
| DS—Backward | 4.43; 2.50 | 3.48; 1.66 | 0.711(147); 0.478; 0.129 |
| DS—Total | 12.13; 4.37 | 1.18; 3.34 | 0.779(147); 0.437; 0.141 |
| StroopVW a | 65.85; 21.27 | 63.51; 22.39 | 0.586(141); 0.559; 0.109 |
| Stroop—C b | 48.30; 15.10 | 47.70; 13.81 | 0.225(144); 0.822; 0.041 |
| Stroop—W&C b | 3.12; 12.98 | 28.56; 13.10 | 0.660(144); 0.511; 0.121 |
| SRT—LTS | 26.26; 13.73 | 22.7; 13.61 | 1.444(147); 0.151; 0.261 |
| SRT—CLTR | 16.04; 12.90 | 14.39; 12.91 | 0.713(147); 0.477; 0.129 |
| SRT—DR c | 5.75; 2.85 | 5.39; 2.87 | 0.705(139); 0.482; 0.129 |
| SRT—Intrusions | 2.70; 4.24 | 3.34; 3.49 | −0.879(147); 0.381; 0.159 |
| 17.66; 4.79 | 17.35; 5.31 | 0.335(138); 0.738; 0.063 | |
| CERAD—DR hits d | 5.76; 2.47 | 5.70; 2.28 | 0.133(138); 0.895; 0.025 |
| MMSE | 26.53; 3.45 | 26.52; 2.72 | 0.018(147); 0.986; 0.003 |
| Cognitive dimensions | t(df); p; Cohen’s d | ||
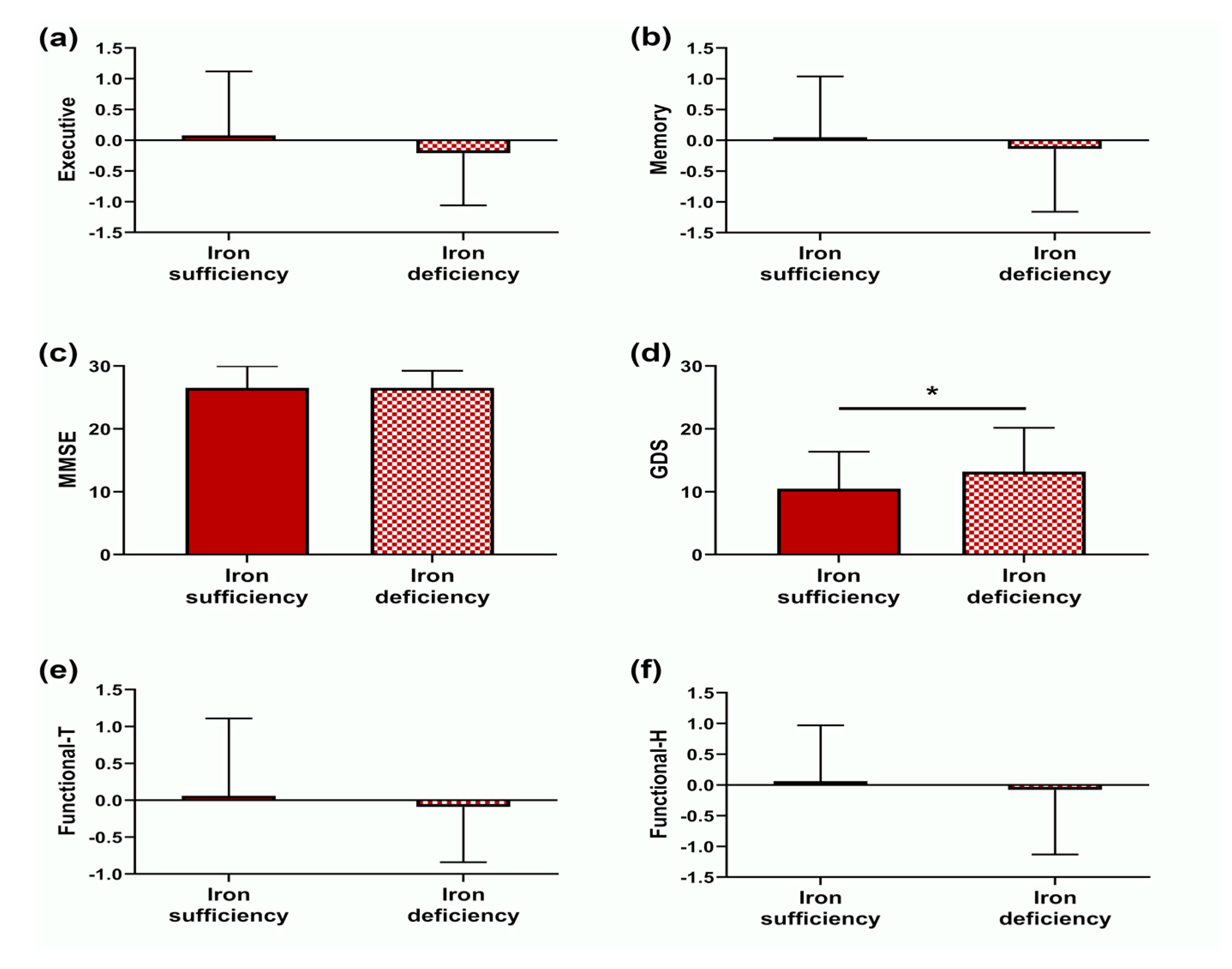
| Executive e | 0.08; 1.04 | −0.21; 0.85 | 0.852(146); 0.396; 0.291 |
| Memory f | 0.05; 0.99 | −0.14; 1.02 | 0.754(146); 0.452; 0.183 |
| Variables (Mean; SD) | Iron Sufficiency | Iron Deficiency | |
|---|---|---|---|
| QoFA a | t(df); p; Cohen’s d | ||
| Mobility tiredness | 4.55; 1.98 | 4.30; 1.76 | 0.732(146); 0.465; 0.133 |
| Lower limb tiredness | 4.35; 1.15 | 4.02; 1.05 | 1.608(146); 0.110; 0.291 |
| Upper limb tiredness | 3.76; 0.81 | 3.82; 0.54 | −0.441(146); 0.660; 0.080 |
| Mobility help | 5.53; 0.995 | 5.57; 0.998 | −0.220(146); 0.826; 0.040 |
| PADL help | 8.46; 1.16 | 8.07; 1.63 | 1.450(62.01); 0.152; 0.275 |
| Functional components a | t(df); p; Cohen’s d | ||
| Functional tiredness | 0.06; 1.05 | −0.09; 0.75 | 0.852(146); 0.396; 0.154 |
| Functional help | 0.06; 0.91 | −0.08; 1.05 | 0.832(146); 0.407; 0.142 |
| Iron Status a | F(df1; df2) | p | η2 partial | R2; R2adjusted |
|---|---|---|---|---|
| Executive b | 2.971(1; 134) | 0.087 | 0.022 | 0.398; 0.371 |
| Memory b | 0.781(1; 125) | 0.378 | 0.006 | 0.296; 0.262 |
| MMSE b | 0.409(1; 142) | 0.523 | 0.003 | 0.271; 0.240 |
| GDS c | 3.792(1; 149) | 0.053 | 0.026 | 0.148; 0.118 |
| Functional-T d | 0.006(1; 139) | 0.940 | 0.000 | 0.211; 0.182 |
| Functional-H d | 0.018(1; 139) | 0.894 | 0.000 | 0.182; 0.152 |
| Variables (Mean; SD) | Iron Sufficiency | Iron Deficiency | |
|---|---|---|---|
| Red Cells Indices a | t(df); p; Cohen’s d | ||
| RBC (1012/L) | 4.60; 0.43 | 4.45; 0.43 | 1.911(144); 0.058; 0.352 |
| Hemoglobin (mg/dL) | 14.17; 1.48 | 12.88; 1.56 | 4.702(144); <0.001; 0.866 |
| Hematocrit (%) | 41.23; 3.83 | 38.23; 4.05 | 4.205(144); <0.001; 0.774 |
| MCV (fL) | 89.61; 3.64 | 85.9; 4.78 | 5.076(144); <0.001; 0.934 |
| MCH (pg) | 30.78; 1.44 | 28.92; 2.06 | 5.354(57.8); <0.001; 1.144 |
| MCHC (g/dL) | 34.35; 0.92 | 33.65; 0.97 | 4.075(144); <0.001; 0.750 |
| RDW (%) | 13.14; 0.64 | 13.95; 1.11 | −4.439(52.49); <0.001; 1.015 |
| Iron biomarkers b | t(df); p; Cohen’s d | ||
| Fe (µg/dL) | 102.83; 24.85 | 70.64; 27.12 | 6.986(144); <0.001; 1.269 |
| TF (mg/dL) | 232.34; 28.96 | 285.43; 46.29 | −7.036(58.04); <0.001; 1.525 |
| FT (mg/mL) ¥ £ | 173.00; 209.25 | 35.50; 41.25 | −7.190(55.75); <0.001; 0.595 |
| TF sat. (%) | 31.84; 8.06 | 18.87; 8.12 | 8.906(144); <0.001; 1.617 |
| TIBC (µg/dL) | 326.30; 40,32 | 388.34; 61.23 | −6.168(59.7); <0.001; 1.314 |
| sTFr (mg/L) c ¥ £ | 1.02; 0.34 | 1.475; 0.80 | −6.170(81.8); <0.001; 0.507 |
| sTFr-logFT index ¥ £ | 0.483; 0.19 | 0.888; 0.77 | −7.810(41.3); <0.001; 0.646 |
| Body iron (mg/kg) | 17.02; 2.66 | 10.02; 4.70 | 9.249(55.2); <0.001; 2.072 |
| Iron dimensions | t(df); p; Cohen’s d | ||
| Storage b | 0.43; 0.49 | −1.01; 1.15 | 7.997(49.73); <0.001; 1.936 |
| Transport b | 0.37; 0.81 | −0.85; 0.87 | 8.156(144); <0.001; 1.481 |
| Transport S. b | 0.36; 0.70 | −0.84; 1.10 | 6.666(58.55); <0.001; 1.437 |
| Red cells C. a | 0.31; 0.72 | −0.77; 1.17 | 5.575(54.05); <0.001; 1.247 |
| Erythropoiesis a | 0.19; 0.95 | −0.46; 0.98 | 3.707(144); <0.001; 0.682 |
| Iron Status a | B | SE | Wald(df); p | OR (CI 95%) |
|---|---|---|---|---|
| Age | −0.028 | 0.027 | 1.141(1); 0.285 | 0.972 (0.923; 1.024) |
| Gender b | −0.920 | 0.435 | 4.478(1); 0.034 | 0.398 (0.170; 0.934) |
| hsCRP (log) | 1.817 | 0.598 | 9.236(1); 0.002 | 6.152 (1.906; 19.854) |
| MNA | −0.191 | 0.082 | 5.389(1); 0.020 | 0.826 (0.704; 0.971) |
| χ2(df); p; R2 Nagelkerke | 5.533(1); p = 0.019; 0.217 | |||
| Age | −0.031 | 0.026 | 1.365(1); 0.243 | 0.970 (0.921; 1.021) |
| Gender b | −1.211 | 0.435 | 7.746(1); 0.005 | 0.298 (0.127; 0.699) |
| hsCRP (log) | 1.966 | 0.634 | 9.625(1); 0.002 | 7.145 (2.063; 24.749) |
| BMI | −0.074 | 0.058 | 1.624(1); 0.203 | 0.929 (0.830; 1.040) |
| χ2(df); p; R2 Nagelkerke | 1.679(1); p = 0.195; 0.184 | |||
| Age | −0.026 | 0.026 | 0.940(1); 0.332 | 0.975 (0.926; 1.027) |
| Gender b | −1.625 | 0.602 | 7.294(1); 0.007 | 0.197 (0.061; 0.640) |
| hsCRP (log) | 1.742 | 0.623 | 7.834(1); 0.005 | 5.711 (1.686; 19.347) |
| %BF | −0.048 | 0.036 | 1.775(1); 0.183 | 0.953 (0.887; 1.023) |
| χ2(df); p; R2 Nagelkerke | 1.786(1); p=.181; 0.166 | |||
| Executive a | Memory a | MMSE a | GDS b | Functional-T c | Functional-H c | |
|---|---|---|---|---|---|---|
| MNA (β; p) | 0.172; 0.038 | 0.076; 0.381 | 0.140; 0.111 | 0.427; <0.001 | 0.271; 0.001 | 0.194; 0.021 |
| Storage (β; p) | 0.074; 0.332 | 0.179; 0.025 | 0.048; 0.544 | −0.068; 0.397 | 0.036; 0.652 | −0.025; 0.766 |
| Storage x MNA (β; p) | 0.056; 0.439 | 0.199; 0.010 | 0.114; 0.140 | −.079; 0.308 | −0.030; 0.704 | 0.095; 0.242 |
| R2adjusted; F; p | 0.381; 10.993; <0.001 | 0.360; 8.371; <0.001 | 0.263; 7.058; <0.001 | 0.260; 7.831; <0.001 | 0.249; 7.441; <0.001 | −0.189; 5.536; <0.001 |
| MNA (β; p) | 0.163; 0.046 | 0.113; 0.242 | 0.129; 0.137 | −0.411; <0.001 | 0.266; 0.001 | 0.200; 0.017 |
| Transport (β; p) | −0.037; 0.632 | 0.093; 0.275 | 0.111; 0.175 | −0.095; 0.250 | 0.036; 0.664 | −0.092; 0.287 |
| Transport x MNA (β; p) | −0.047; 0.521 | 0.169; 0.053 | 0.112; 0.146 | −0.022; 0.778 | −0.060; 0.445 | 0.080; 0.329 |
| R2adjusted; F; p | 0.377; 10.823; <0.001 | 0.287; 7.392; <0.001 | 0.271; 7.326; <0.001 | 0.259; 7.783; <0.001 | 0.252; 7.520; <0.001 | 0.193; 5.639; <0.001 |
| MNA (β; p) | 0.163; 0.045 | 0.063; 0.475 | 0.130; 0.134 | −0.405; <0.001 | 0.288; <0.001 | 0.202; 0.015 |
| Transport S. (β; p) | 0.018; 0.811 | 0.060; 0.466 | 0.008; 0.919 | −0.109; 0.163 | −0.061; 0.432 | −0.076; 0.348 |
| Transport S. x MNA (β; p) | −0.020; 0.794 | 0.060; 0.459 | 0.074; 0.354 | 0.005; 0.946 | −0.035; 0.657 | 0.121; 0.138 |
| R2adjusted; F; p | 0.374; 10.714; <0.001 | 0.260; 6.576; <0.001 | 0.254; 6.782; <0.001 | 0.262; 7.895; <0.001 | 0.252; 7.541; <0.001 | 0.198; 5.789; <0.001 |
| MNA (β; p) | 0.187; 0.024 | 0.091; 0.312 | 0.127; 0.141 | −0.414; <0.001 | 0.271; 0.001 | 0.185; 0.026 |
| Red cells C. (β; p) | 0.044; 0.574 | 0.105; 0.215 | 0.062; 0.455 | 0.066; 0.942 | 0.098; 0.236 | −0.023; 0.790 |
| Red cells C. x MNA (β; p) | 0.055; 0.466 | 0.146; 0.077 | 0.119; 0.133 | 0.023; 0.777 | 0.004; 0.955 | 0.087; 0.298 |
| R2adjusted; F; p | 0.370; 10.534; <0.001 | 0.275; 7.017; <0.001 | 0.257; 6.891; <0.001 | 0.230; 6.794; <0.001 | 0.258; 7.764; <0.001 | 0.0176; 5.140; <0.001 |
| MNA (β; p) | 0.183; 0.025 | 0.086; 0.337 | 0.126; 0.145 | −0.406; <0.001 | 0.271; 0.001 | 0.181; 0.030 |
| Erythropoiesis (β; p) | 0.007; 0.939 | 0.229; 0.015 | 0.103; 0.262 | −0.222; 0.014 | 0.162; 0.066 | −0.029, 0.753 |
| Erythropoiesis x MNA (β; p) | −0.004; 0.954 | 0.75; 0.352 | 0.41; 0.594 | −0.053; 0.492 | −0.010; 0.896 | 0.064; 0.432 |
| R2adjusted; F; p | 0.366; 10.389; <0.001 | 0.293; 7.568; <0.001 | 0.252; 6.728; <0.001 | 0.267, 8.073; <0.001 | 0.269; 8.165; <0.001 | 0.172; 5.023; <0.001 |
References
- Hentze, M.W.; Muckenthaler, M.U.; Andrews, N.C. Balancing acts: Molecular control of mammalian iron metabolism. Cell 2004, 117, 285–297. [Google Scholar] [CrossRef]
- Zecca, L.; Youdim, M.B.; Riederer, P.; Connor, J.R.; Crichton, R.R. Iron, brain ageing and neurodegenerative disorders. Nat. Rev. Neurosci. 2004, 5, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Li, S.; Liu, G.; Yan, F.; Ma, X.; Huang, Z.; Tian, H. Body Iron Stores and Heme-Iron Intake in Relation to Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis. PLoS ONE 2012, 7, e41641. [Google Scholar] [CrossRef]
- Boccio, J.; Iyengar, V. Iron deficiency: Causes, consequences, and strategies to overcome this nutritional problem. Biol. Trace Elem. Res. 2003, 94, 1–31. [Google Scholar] [CrossRef]
- Mathers, C.; Fat, D.M.; Boerma, J.T.; World Health Organization. The Global Burden of Disease: 2004 Update; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Christensen, K.; Doblhammer, G.; Rau, R.; Vaupel, J.W. Ageing populations: The challenges ahead. Lancet 2009, 374, 1196–1208. [Google Scholar] [CrossRef]
- Rechel, B.; Grundy, E.; Robine, J.-M.; Cylus, J.; Mackenbach, J.P.; Knai, C.; McKee, M. Ageing in the European Union. Lancet 2013, 381, 1312–1322. [Google Scholar] [CrossRef]
- The, L. Ageing well: A global priority. Lancet 2012, 379, 1274. [Google Scholar]
- Qato, D.M.; Alexander, G.C.; Conti, R.M.; Johnson, M.; Schumm, P.; Lindau, S.T. Use of prescription and over-the-counter medications and dietary supplements among older adults in the United States. JAMA 2008, 300, 2867–2878. [Google Scholar]
- Clark, S.F. Iron Deficiency Anemia. Nutr. Clin. Pract. 2008, 23, 128–141. [Google Scholar] [CrossRef]
- De Benoist, B.; Cogswell, M.; Egli, I.; McLean, E. Worldwide Prevalence of Anaemia 1993–2005; WHO Global Database of Anaemia: Geneva, Switzerland, 2008. [Google Scholar]
- Hosking, D.E.; Ayton, S.; Beckett, N.; Booth, A.; Peters, R. More evidence is needed. Iron, incident cognitive decline and dementia: A systematic review. Ther. Adv. Chronic Dis. 2018, 9, 241–256. [Google Scholar] [CrossRef]
- Neidlein, S.; Wirth, R.; Pourhassan, M. Iron deficiency, fatigue and muscle strength and function in older hospitalized patients. Eur. J. Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, K.; Konomi, A. Iron deficiency without anaemia is a potential cause of fatigue: Meta-analyses of randomised controlled trials and cross-sectional studies. Br. J. Nutr. 2017, 117, 1422–1431. [Google Scholar] [CrossRef]
- Haas, J.D.; Brownlie, T. Iron Deficiency and Reduced Work Capacity: A Critical Review of the Research to Determine a Causal Relationship. J. Nutr. 2001, 131, 676S–690S. [Google Scholar] [CrossRef] [PubMed]
- Beard, J.L.; Borel, M.J.; Derr, J. Impaired thermoregulation and thyroid function in iron-deficiency anemia. Am. J. Clin. Nutr. 1990, 52, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, N.; Sun, J.; Krause, D.; Mastro, A.; Handte, G. Immune function is impaired in iron-deficient, homebound, older women. Am. J. Clin. Nutr. 2004, 79, 516–521. [Google Scholar] [CrossRef]
- Peters, R.; Burch, L.; Warner, J.; Beckett, N.; Poulter, R.; Bulpitt, C. Haemoglobin, anaemia, dementia and cognitive decline in the elderly, a systematic review. BMC Geriatr. 2008, 8, 18. [Google Scholar] [CrossRef]
- WHO. Iron Deficiency Anaemia: Assessment, Prevention and Control. A Guide for Programme Managers; WHO/NHD/01.3: 2001; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Reynolds, E. Vitamin B12, folic acid, and the nervous system. Lancet Neurol. 2006, 5, 949–960. [Google Scholar] [CrossRef]
- Chaves, P.H. Functional outcomes of anemia in older adults. In Seminars in Hematology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 255–260. [Google Scholar]
- Penninx, B.W.; Guralnik, J.M.; Onder, G.; Ferrucci, L.; Wallace, R.B.; Pahor, M. Anemia and decline in physical performance among older persons. Am. J. Med. 2003, 115, 104–110. [Google Scholar] [CrossRef]
- Penninx, B.W.; Pahor, M.; Cesari, M.; Corsi, A.M.; Woodman, R.C.; Bandinelli, S.; Guralnik, J.M.; Ferrucci, L. Anemia is associated with disability and decreased physical performance and muscle strength in the elderly. J. Am. Geriatr. Soc. 2004, 52, 719–724. [Google Scholar] [CrossRef]
- Thein, M.; Ershler, W.B.; Artz, A.S.; Tecson, J.; Robinson, B.E.; Rothstein, G.; Liede, A.; Gylys-Colwell, I.; Lu, Z.J.; Robbins, S. Diminished quality of life and physical function in community-dwelling elderly with anemia. Medicine 2009, 88, 107–114. [Google Scholar] [CrossRef]
- Cook, J.D.; Flowers, C.H.; Skikne, B.S. The quantitative assessment of body iron. Blood 2003, 101, 3359–3364. [Google Scholar] [CrossRef] [PubMed]
- Murray-Kolb, L.E.; Beard, J.L. Iron treatment normalizes cognitive functioning in young women. Am. J. Clin. Nutr. 2007, 85, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Rimon, E.; Kagansky, N.; Kagansky, M.; Mechnick, L.; Mashiah, T.; Namir, M.; Levy, S. Are we giving too much iron? Low-dose iron therapy is effective in octogenarians. Am. J. Med. 2005, 118, 1142–1147. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, B.B.; Cankurtaran, M.; Haznedaroglu, I.C.; Halil, M.; Ulger, Z.; Altun, B.; Ariogul, S. Iron deficiency can cause cognitive impairment in geriatric patients. J. Nutr. Health Aging 2012, 16, 220–224. [Google Scholar] [CrossRef]
- Santos, N.C.; Costa, P.S.; Cunha, P.; Cotter, J.; Sampaio, A.; Zihl, J.; Almeida, O.F.; Cerqueira, J.J.; Palha, J.A.; Sousa, N. Mood is a key determinant of cognitive performance in community-dwelling older adults: A cross-sectional analysis. Age 2013, 35, 1983–1993. [Google Scholar] [CrossRef]
- Santos, N.C.; Costa, P.S.; Cunha, P.; Portugal-Nunes, C.; Amorim, L.; Cotter, J.; Cerqueira, J.J.; Palha, J.A.; Sousa, N. Clinical, physical and lifestyle variables and relationship with cognition and mood in aging: A cross-sectional analysis of distinct educational groups. Front. Aging Neurosci. 2014, 6, 21. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Strauss, E. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary; Oxford University Press: New York, NY, USA, 2006. [Google Scholar]
- Buschke, H.; Sliwinski, M.; Kuslansky, G.; Lipton, R.B. Aging, encoding specificity, and memory change in the Double Memory Test. J. Int. Neuropsychol. Soc. 1995, 1, 483–493. [Google Scholar] [CrossRef]
- Morris, J.C.; Heyman, A.; Mohs, R.C.; Hughes, J.P.; van Belle, G.; Fillenbaum, G.; Mellits, E.D.; Clark, C. The Consortium to Establish a Registry for Alzheimer’s Disease (CERAD). Part I. Clinical and neuropsychological assessment of Alzheimer’s disease. Neurology 1989, 39, 1159–1165. [Google Scholar]
- Yesavage, J.A.; Brink, T.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1983, 17, 37–49. [Google Scholar] [CrossRef]
- Avlund, K.; Kreiner, S.; Schultz-Larsen, K. Functional ability scales for the elderly: A validation study. Eur. J. Public Health 1996, 6, 35–42. [Google Scholar] [CrossRef]
- Kondrup, J.; Allison, S.P.; Elia, M.; Vellas, B.; Plauth, M. ESPEN guidelines for nutrition screening 2002. Clin. Nutr. 2003, 22, 415–421. [Google Scholar] [CrossRef]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.-L. The mini nutritional assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Kline, R.B. Principles and Practice of Structural Equation Modeling; Guilford Press: New York, NY, USA, 2011. [Google Scholar]
- DeVellis, R.F. Scale Development: Theory and Applications; Sage Publications: Thousand Oaks, CA, USA, 2011; Volume 26. [Google Scholar]
- Hayes, A.; Matthes, J. Computational procedures for probing interactions in OLS and logistic regression: SPSS and SAS implementations. Behav. Res. Methods 2009, 41, 924–936. [Google Scholar] [CrossRef]
- Beard, J.L.; Hendricks, M.K.; Perez, E.M.; Murray-Kolb, L.E.; Berg, A.; Vernon-Feagans, L.; Irlam, J.; Isaacs, W.; Sive, A.; Tomlinson, M. Maternal Iron Deficiency Anemia Affects Postpartum Emotions and Cognition. J. Nutr. 2005, 135, 267–272. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Cogswell, M.E.; McDonald, T. Have we forgotten the significance of postpartum iron deficiency? Am. J. Obstet. Gynecol. 2005, 193, 36–44. [Google Scholar] [CrossRef]
- Grantham-McGregor, S.; Ani, C. A Review of Studies on the Effect of Iron Deficiency on Cognitive Development in Children. J. Nutr. 2001, 131, 649S–668S. [Google Scholar] [CrossRef]
- Halterman, J.S.; Kaczorowski, J.M.; Aligne, C.A.; Auinger, P.; Szilagyi, P.G. Iron Deficiency and Cognitive Achievement Among School-Aged Children and Adolescents in the United States. Pediatrics 2001, 107, 1381–1386. [Google Scholar] [CrossRef]
- Deal, J.A.; Carlson, M.C.; Xue, Q.L.; Fried, L.P.; Chaves, P.H. Anemia and 9-year domain-specific cognitive decline in community-dwelling older women: The Women’s Health and Aging Study II. J. Am. Geriatr. Soc. 2009, 57, 1604–1611. [Google Scholar] [CrossRef]
- Denny, S.D.; Kuchibhatla, M.N.; Cohen, H.J. Impact of anemia on mortality, cognition, and function in community-dwelling elderly. Am. J. Med. 2006, 119, 327–334. [Google Scholar] [CrossRef]
- Lucca, U.; Tettamanti, M.; Mosconi, P.; Apolone, G.; Gandini, F.; Nobili, A.; Tallone, M.V.; Detoma, P.; Giacomin, A.; Clerico, M.; et al. Association of Mild Anemia with Cognitive, Functional, Mood and Quality of Life Outcomes in the Elderly: The “Health and Anemia” Study. PLoS ONE 2008, 3, e1920. [Google Scholar] [CrossRef]
- Terekeci, H.M.; Kucukardali, Y.; Onem, Y.; Erikci, A.A.; Kucukardali, B.; Sahan, B.; Sayan, O.; Celik, S.; Gulec, M.; Sanisoglu, Y.S.; et al. Relationship between anaemia and cognitive functions in elderly people. Eur. J. Intern. Med. 2010, 21, 87–90. [Google Scholar] [CrossRef]
- Fretham, S.J.B.; Carlson, E.S.; Georgieff, M.K. The Role of Iron in Learning and Memory. Adv. Nutr. 2011, 2, 112–121. [Google Scholar] [CrossRef]
- Beard, J.L. Iron Biology in Immune Function, Muscle Metabolism and Neuronal Functioning. J. Nutr. 2001, 131, 568S–580S. [Google Scholar] [CrossRef]
- Shah, R.C.; Wilson, R.S.; Tang, Y.; Dong, X.; Murray, A.; Bennett, D.A. Relation of hemoglobin to level of cognitive function in older persons. Neuroepidemiology 2009, 32, 40–46. [Google Scholar] [CrossRef]
- Lukowski, A.F.; Koss, M.; Burden, M.J.; Jonides, J.; Nelson, C.A.; Kaciroti, N.; Jimenez, E.; Lozoff, B. Iron deficiency in infancy and neurocognitive functioning at 19 years: Evidence of long-term deficits in executive function and recognition memory. Nutr. Neurosci. 2010, 13, 54–70. [Google Scholar] [CrossRef]
- Lozoff, B.; Beard, J.; Connor, J.; Barbara, F.; Georgieff, M.; Schallert, T. Long-lasting neural and behavioral effects of iron deficiency in infancy. Nutr. Rev. 2006, 64, S34–S91. [Google Scholar] [CrossRef]
- Blanton, C.A.; Green, M.W.; Kretsch, M.J. Body iron is associated with cognitive executive planning function in college women. Br. J. Nutr. 2013, 109, 906–913. [Google Scholar] [CrossRef]
- Rouault, T.A.; Cooperman, S. Brain Iron Metabolism. Semin. Pediatr. Neurol. 2006, 13, 142–148. [Google Scholar] [CrossRef]
- Squire, L.R. Memory systems of the brain: A brief history and current perspective. Neurobiol. Learn. Mem. 2004, 82, 171–177. [Google Scholar] [CrossRef]
- Rao, R.; Tkac, I.; Townsend, E.L.; Gruetter, R.; Georgieff, M.K. Perinatal Iron Deficiency Alters the Neurochemical Profile of the Developing Rat Hippocampus. J. Nutr. 2003, 133, 3215–3221. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.V.; Fretham, S.J.B.; Carlson, E.S.; Georgieff, M.K. Long-term reduction of hippocampal brain-derived neurotrophic factor activity after fetal-neonatal iron deficiency in adult rats. Pediatr. Res. 2009, 65, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Onder, G.; Penninx, B.W.; Cesari, M.; Bandinelli, S.; Lauretani, F.; Bartali, B.; Gori, A.M.; Pahor, M.; Ferrucci, L. Anemia is associated with depression in older adults: Results from the InCHIANTI study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2005, 60, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Molloy, G.J. Cross-sectional and longitudinal associations between anemia and depressive symptoms in the English Longitudinal Study of Ageing. J. Am. Geriatr. Soc. 2009, 57, 948–949. [Google Scholar] [CrossRef]
- Stewart, R.; Hirani, V. Relationship between depressive symptoms, anemia, and iron status in older residents from a national survey population. Psychosom. Med. 2012, 74, 208–213. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef]
- Brutsaert, T.D.; Hernandez-Cordero, S.; Rivera, J.; Viola, T.; Hughes, G.; Haas, J.D. Iron supplementation improves progressive fatigue resistance during dynamic knee extensor exercise in iron-depleted, nonanemic women. Am. J. Clin. Nutr. 2003, 77, 441–448. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Wawer, A.A.; Gillings, R.; Jennings, A.; Myint, P.K. Iron status in the elderly. Mech. Ageing Dev. 2013. [Google Scholar] [CrossRef]
- Hsu, H.-S.; Li, C.-I.; Liu, C.-S.; Lin, C.-C.; Huang, K.-C.; Li, T.-C.; Huang, H.-Y.; Lin, W.-Y. Iron deficiency is associated with increased risk for cardiovascular disease and all-cause mortality in the elderly living in long-term care facilities. Nutrition 2013, 29, 737–743. [Google Scholar] [CrossRef]
- Guigoz, Y. The Mini Nutritional Assessment (MNA®) review of the literature-what does it tell us? J. Nutr. Health Aging 2006, 10, 466. [Google Scholar]
- Murphy, M.; Brooks, C.; New, S.; Lumbers, M. The use of the Mini-Nutritional Assessment (MNA) tool in elderly orthopaedic patients. Eur. J. Clin. Nutr. 2000, 54, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Selvi Öztorun, H.; Çınar, E.; Turgut, T.; Mut Sürmeli, D.; Bahşi, R.; Atmış, V.; Varlı, M.; Aras, S. The impact of treatment for iron deficiency and iron deficiency anemia on nutritional status, physical performance, and cognitive function in geriatric patients. Eur. Geriatr. Med. 2018, 9, 493–500. [Google Scholar] [CrossRef]
- Goodwin, J.S.; Goodwin, J.M.; Garry, P.J. ASsociation between nutritional status and cognitive functioning in a healthy elderly population. JAMA 1983, 249, 2917–2921. [Google Scholar] [CrossRef] [PubMed]
- Boult, C.; Krinke, U.B.; Urdangarin, C.F.; Skarin, V. The validity of nutritional status as a marker for future disability and depressive symptoms among high-risk older adults. J. Am. Geriatr. Soc. 1999, 47, 995–999. [Google Scholar] [CrossRef] [PubMed]
- Olin, A.Ö.; Koochek, A.; Ljungqvist, O.; Cederholm, T. Nutritional status, well-being and functional ability in frail elderly service flat residents. Eur. J. Clin. Nutr. 2004, 59, 263–270. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thurnham, D.I.; McCabe, G.P. Influence of infection and inflammation on biomarkers of nutritional status with an emphasis on vitamin A and iron. In Proceedings of the Report: Priorities in the Assessment of Vitamin A and Iron stAtus in Populations, Panama City, Panama, 15–17 September 2010. [Google Scholar]
- Ganz, T.; Nemeth, E. Iron imports. IV. Hepcidin and regulation of body iron metabolism. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G199–G203. [Google Scholar] [CrossRef] [PubMed]
- Hentze, M.W.; Muckenthaler, M.U.; Galy, B.; Camaschella, C. Two to tango: Regulation of Mammalian iron metabolism. Cell 2010, 142, 24–38. [Google Scholar] [CrossRef]
- Thomas, D.W.; Hinchliffe, R.F.; Briggs, C.; Macdougall, I.C.; Littlewood, T.; Cavill, I.; British Committee for Standards in Haematology. Guideline for the laboratory diagnosis of functional iron deficiency. Br. J. Haematol. 2013, 161, 639–648. [Google Scholar] [CrossRef]
- Musallam, K.M.; Taher, A.T. Iron deficiency beyond erythropoiesis: Should we be concerned? Curr. Med. Res. Opin. 2018, 34, 81–93. [Google Scholar] [CrossRef]
| Variables | Iron Sufficiency | Iron Deficiency | |
|---|---|---|---|
| Socio-Demographic (Mean; SD) | t(df); p; Cohen’s d | ||
| Age (years) | 66; 8 | 66; 7 | 0.197(147); 0.844; 0.036 |
| Education (school years) | 5; 4 | 5; 4 | −0.043(147); 0.966; 0.008 |
| Anthropometric (mean; SD) | t(df); p; Cohen’s d | ||
| Weight (kg) a | 74; 2 | 71; 2 | 1.426(138); 0.156; 0.267 |
| Height (m) a | 1.60; 0.08 | 1.56; 0.08 | 2.218(138); 0.028; 0.415 |
| BMI (kg/m2) a | 29; 4 | 29; 4 | −0.236(138); 0.814; 0.044 |
| %BF (%) b | 32; 8 | 34; 8 | −1.394(134); 0.166; 0.266 |
| Gender (n; %) | χ2(df); p; φ | ||
| Female | 49; 33 | 32; 21 | 8.488(1); 0.004; −0.239 |
| Male | 56; 38 | 12; 8 | |
| Education, class (n; %) | χ2(df); p; φ | ||
| <4 years | 82; 55 | 29; 19 | 2.424(1); 0.120; 0.128 |
| ≥4 years | 23; 15 | 15; 10 | |
| BMI class (n; %) | χ2(df); p; φc | ||
| Normal | 17; 12 | 5; 3 | 0.756(2); 0.679; 0.073 |
| Overweight | 47; 34 | 19; 14 | |
| Obesity | 35; 25 | 17; 12 | |
| MNA score (mean; SD) | t(df); p; Cohen’s d | ||
| MNA score (points) | 27; 2 | 26; 3 | 2.731(138); 0.007; 0.490 |
| Nutritional status (n; %) | χ2(df); p; φ | ||
| Risk of malnutrition | 11; 8 | 8; 6 | 1.745(1); 0.277; −0.112 |
| Normal | 88; 63 | 33; 24 | |
| Inflammatory indices (mean; SD) | Z(U); p; r £ | ||
| hsCRP (mg/dL) ¥ £ | 1.56; 2.42 | 2.90; 2.27 | −2.880(1617.5); 0.004; 0.236 |
| Executive a | Memory a | MMSE a | GDS b | Functional-T c | Functional-H c | |
|---|---|---|---|---|---|---|
| Storage (β; p) | 0.112; 0.131 | 0.167; 0.037 | 0.051; 0.506 | −0.144; 0.085 | 0.094; 0.248 | 0.002; 0.976 |
| R2adjusted; F; p | 0.377; 14.804; <0.0001 | 0.289; 9.679; <0.001 | 0.248; 8.971; <0.001 | 0.115; 4.773; <0.001 | 0.188; 7.281; <0.001 | 0.165; 6.375; <0.001 |
| Transport (β; p) | 0.014; 0.857 | 0.124; 0.134 | 0.096; 0.221 | −0.176; 0.036 | 0.091; 0.283 | −0.055; 0.521 |
| R2adjusted; F; p | 0.366; 14.178; <0.001 | 0.277; 9.166; <0.001 | 0.254; 9.218; <0.001 | 0.124; 5.115; <0.001 | 0.186; 7.235; <0.001 | 0.168; 6.477; <0.001 |
| Transport S. (β; p) | 0.042; 0.556 | 0.070; 0.383 | 0.038; 0.618 | −0.181; 0.025 | −0.018; 0.827 | −0.039; 0.633 |
| R2adjusted; F; p | 0.367; 14.265; <0.001 | 0.268; 8.808; <0.001 | 0.247; 8.927; <0.001 | 0.128; 5.257; <0.001 | 0.180; 6.953; <0.001 | 0.166; 6.431; <0.001 |
| Red cells C. (β; p) | 0.094; 0.196 | 0.084; 0.300 | 0.051; 0.504 | −0.078; 0.354 | 0.148; 0.069 | −0.013; 0.879 |
| R2adjusted; F; p | 0.364; 14.040; <0.001 | 0.266; 8.731; <0.001 | 0.243; 8.758; <0.001 | 0.085; 3.690; 0.004 | 0.201; 7.827; <0.001 | 0.153; 5.905; <0.001 |
| Erythropoiesis (β; p) | 0.007; 0.930 | 0.227; 0.015 | 0.074; 0.412 | −0.301; 0.001 | 0.197; 0.032 | −0.012; 0.902 |
| R2adjusted; F; p | 0.355; 13.587; <0.001 | 0.295; 9.916; <0.001 | 0.244; 8.811; <0.001 | 0.144; 5.886; <0.001 | 0.209; 8.173; <0.001 | 0.153; 5.903; <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Portugal-Nunes, C.; Castanho, T.C.; Amorim, L.; Moreira, P.S.; Mariz, J.; Marques, F.; Sousa, N.; Santos, N.C.; Palha, J.A. Iron Status is Associated with Mood, Cognition, and Functional Ability in Older Adults: A Cross-Sectional Study. Nutrients 2020, 12, 3594. https://doi.org/10.3390/nu12113594
Portugal-Nunes C, Castanho TC, Amorim L, Moreira PS, Mariz J, Marques F, Sousa N, Santos NC, Palha JA. Iron Status is Associated with Mood, Cognition, and Functional Ability in Older Adults: A Cross-Sectional Study. Nutrients. 2020; 12(11):3594. https://doi.org/10.3390/nu12113594
Chicago/Turabian StylePortugal-Nunes, Carlos, Teresa Costa Castanho, Liliana Amorim, Pedro Silva Moreira, José Mariz, Fernanda Marques, Nuno Sousa, Nadine Correia Santos, and Joana Almeida Palha. 2020. "Iron Status is Associated with Mood, Cognition, and Functional Ability in Older Adults: A Cross-Sectional Study" Nutrients 12, no. 11: 3594. https://doi.org/10.3390/nu12113594
APA StylePortugal-Nunes, C., Castanho, T. C., Amorim, L., Moreira, P. S., Mariz, J., Marques, F., Sousa, N., Santos, N. C., & Palha, J. A. (2020). Iron Status is Associated with Mood, Cognition, and Functional Ability in Older Adults: A Cross-Sectional Study. Nutrients, 12(11), 3594. https://doi.org/10.3390/nu12113594







