Twelve-Week Daily Consumption of ad hoc Fortified Milk with ω-3, D, and Group B Vitamins Has a Positive Impact on Inflammaging Parameters: A Randomized Cross-Over Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Study Population
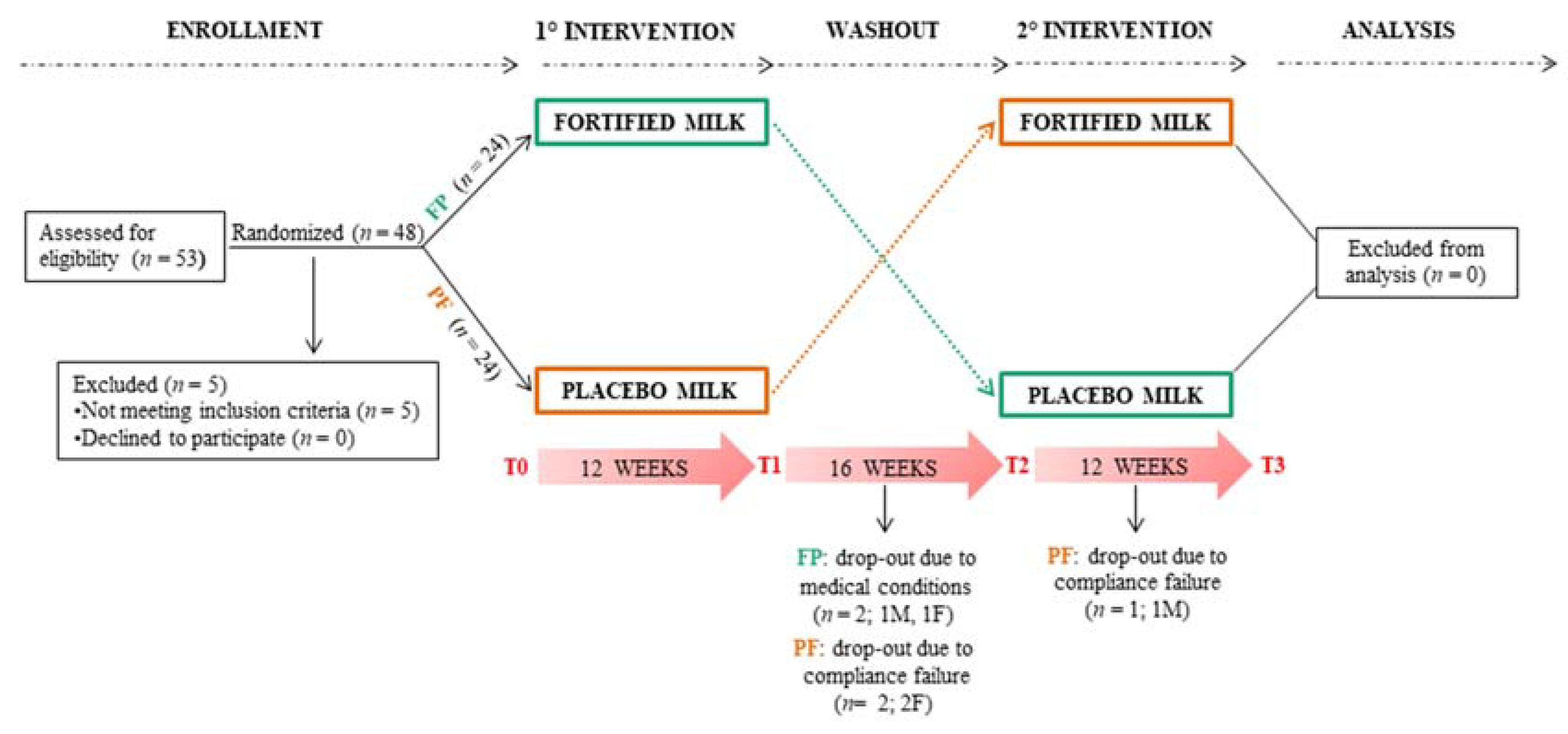
2.3. Study Design
2.4. Intervention
2.5. Data Collection
2.6. Blood Sampling and Hematological–Biochemical Measurements
2.7. Lipidomic Assay
2.8. Cytokine Measurement
2.9. Statistical Analyses
3. Results
3.1. Baseline Characteristics of the Study Population
3.2. Circulating Micronutrient Levels
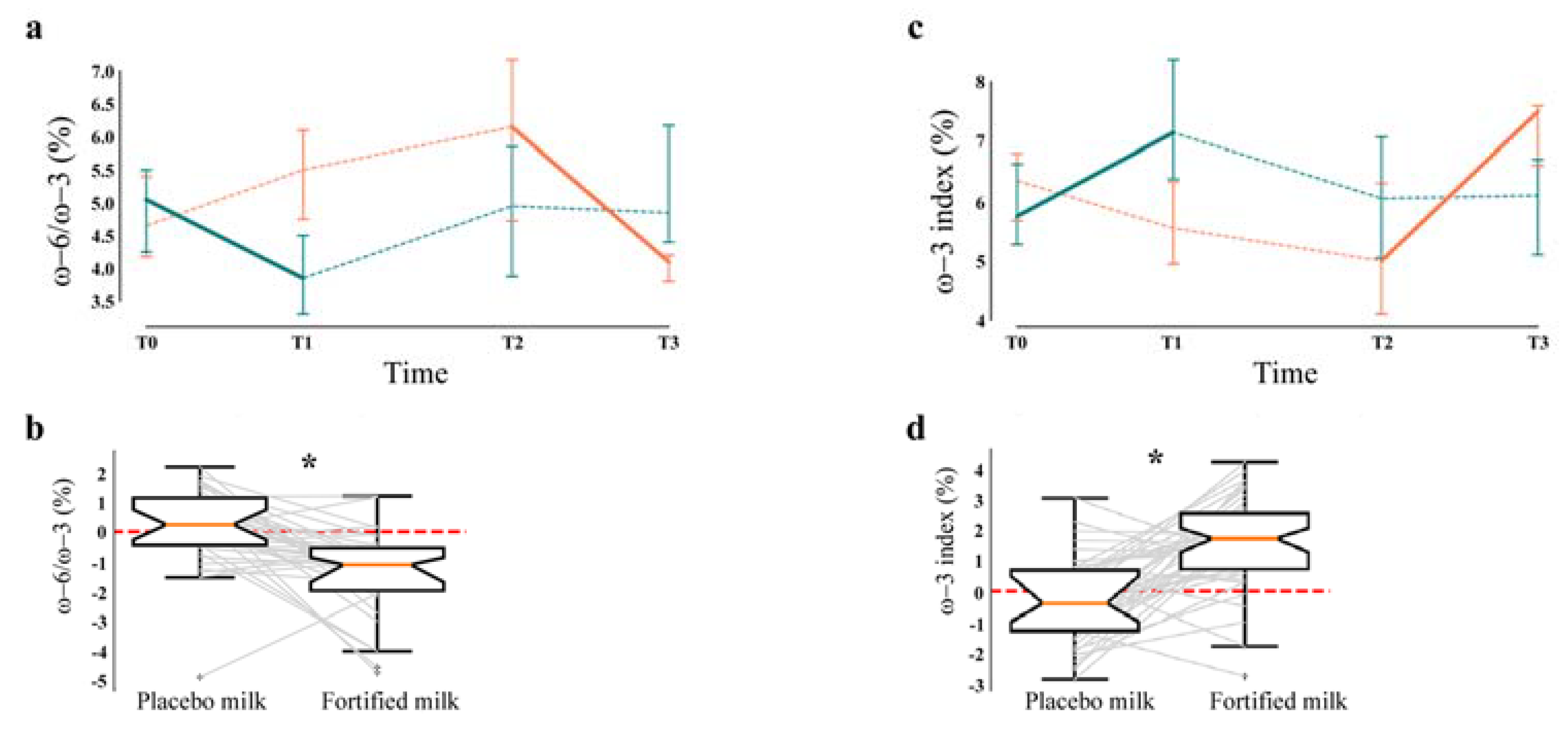
3.3. Lipidomics
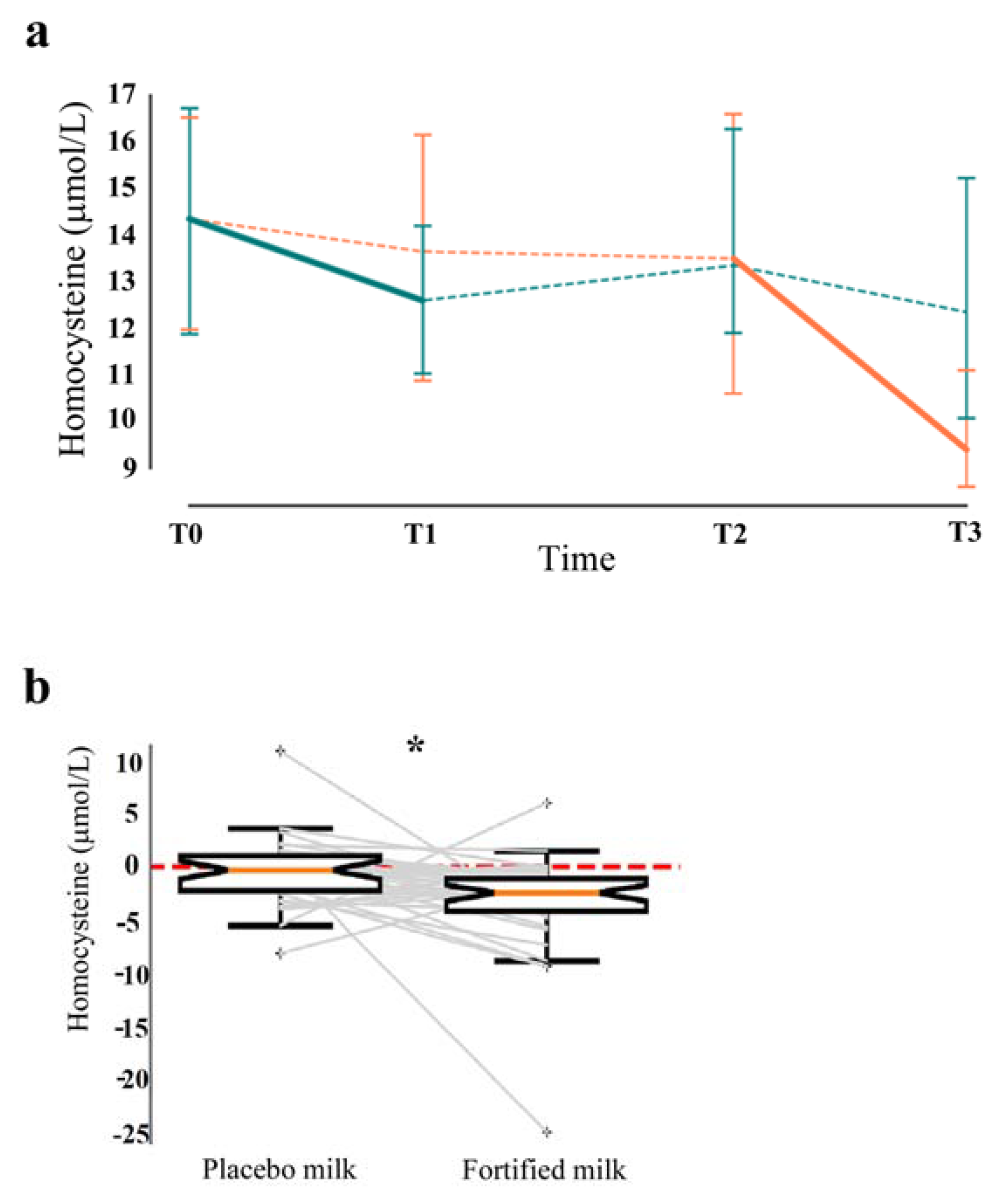
3.4. Hematological–Biochemical Assessment
3.5. Inflammatory Profile
3.6. Physical, Cognitive, and Mood Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Available online: https://www.istat.it/it/files//2019/02/Indicatoridemografici2018_EN.pdf (accessed on 31 March 2019).
- Atella, V.; Piano Mortari, A.; Kopinska, J.; Belotti, F.; Lapi, F.; Cricelli, C.; Fontana, L. Trends in age-related disease burden and healthcare utilization. Aging Cell 2019. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.who.int/ageing/10-priorities/en (accessed on 30 April 2019).
- Martucci, M.; Ostan, R.; Biondi, F.; Bellavista, E.; Fabbri, C.; Bertarelli, C.; Salvioli, S.; Capri, M.; Franceschi, C.; Santoro, A. Mediterranean diet and inflammaging within the hormesis paradigm. Nutr. Rev. 2017, 75, 442–455. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, T.S.; Rampelli, S.; Jeffery, I.B.; Santoro, A.; Neto, M.C.; Capri, M.; Giampieri, E.; Jennings, A.; Candela, M.; Turroni, S.; et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: The NU-AGE 1-year dietary intervention across 5 European countries. Gut 2020. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 4–9. [Google Scholar] [CrossRef]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Kennedy, B.K.; Berger, S.L.; Brunet, A.; Campisi, J.; Cuervo, A.M.; Epel, E.S.; Franceschi, C.; Lithgow, G.J.; Morimoto, R.I.; Pessin, J.E.; et al. Geroscience: Linking aging to chronic disease. Cell 2014, 159, 709–713. [Google Scholar] [CrossRef]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative Western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef]
- Hoffman, R. Micronutrient deficiencies in the elderly—Could ready meals be part of the solution? J. Nutr. Sci. 2017, 6, e2. [Google Scholar] [CrossRef]
- Watson, J.; Lee, M.; Garcia-Casal, M.N. Consequences of Inadequate Intakes of Vitamin A, Vitamin B(12), Vitamin D, Calcium, Iron, and Folate in Older Persons. Curr. Geriatr. Rep. 2018, 7, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Baró, L.; Fonollá, J.; Peña, J.L.; Martínez-Férez, A.; Lucena, A.; Jiménez, J.; Boza, J.J.; López-Huertas, E. n-3 Fatty acids plus oleic acid and vitamin supplemented milk consumption reduces total and LDL cholesterol, homocysteine and levels of endothelial adhesion molecules in healthy humans. Clin. Nutr. 2003, 22, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; Baró, L.; Fonollá, J.; González-Santiago, M.; Martínez-Férez, A.; Castillo, R.; Jiménez, J.; Boza, J.J.; López-Huertas, E. Cardiovascular effects of milk enriched with omega-3 polyunsaturated fatty acids, oleic acid, folic acid, and vitamins E and B6 in volunteers with mild hyperlipidemia. Nutrition 2004, 20, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; Fonollá, J.; Marti, J.L.; Jiménez, J.; Boza, J.J.; López-Huertas, E. Intake of fish oil, oleic acid, folic acid, and vitamins B-6 and E for 1 year decreases plasma C-reactive protein and reduces coronary heart disease risk factors in male patients in a cardiac rehabilitation program. J. Nutr. Feb. 2007, 137, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Benito, P.; Caballero, J.; Moreno, J.; Gutiérrez-Alcántara, C.; Muñoz, C.; Rojo, G.; Garcia, S.; Soriguer, F.C. Effects of milk enriched with omega-3 fatty acid, oleic acid and folic acid in patients with metabolic syndrome. Clin. Nutr. 2006, 25, 581–587. [Google Scholar] [CrossRef]
- Molfino, A.; Gioia, G.; Rossi Fanelli, F.; Muscaritoli, M. The role for dietary omega-3 fatty acids supplementation in older adults. Nutrients 2014, 6, 4058–4073. [Google Scholar] [CrossRef]
- Tan, A.; Sullenbarger, B.; Prakash, R.; McDaniel, J.C. Supplementation with eicosapentaenoic acid and docosahexaenoic acid reduces high levels of circulating proinflammatory cytokines in aging adults: A randomized, controlled study. Prostaglandins Leukot. Essent. Fat. Acids 2018, 132, 23–29. [Google Scholar] [CrossRef]
- Davinelli, S.; Corbi, G.; Righetti, S.; Sears, B.; Olarte, H.H.; Grassi, D.; Scapagnini, G. Cardioprotection by Cocoa Polyphenols and ω-3 Fatty Acids: A Disease-Prevention Perspective on Aging-Associated Cardiovascular Risk. J. Med. Food 2018, 21, 1060–1069. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Lippi, G.; Nouvenne, A.; Ticinesi, A.; Bonelli, P.; Salvagno, G.L.; Cervellin, G.; Guidi, G.C. The burden of vitamin D deficiency in a mediterranean country without a policy of food fortification. Acta. Biomed. 2015, 86, 59–62. [Google Scholar]
- Khazai, N.; Judd, S.E.; Tangpricha, V. Calcium and vitamin D: Skeletal and extraskeletal health. Curr. Rheumatol. Rep. 2008, 10, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Santoro, D.; Sebekova, K.; Teta, D.; De Nicola, L. Extraskeletal Functions of Vitamin D. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mariani, E.; Mangialasche, F.; Feliziani, F.T.; Cecchetti, R.; Malavolta, M.; Bastiani, P.; Baglioni, M.; Dedoussis, G.; Fulop, T.; Herbein, G.; et al. Effects of zinc supplementation on antioxidant enzyme activities in healthy old subjects. Exp. Gerontol. 2008, 43, 445. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef]
- Mocchegiani, E.; Romeo, J.; Malavolta, M.; Costarelli, L.; Giacconi, R.; Diaz, L.E.; Marcos, A. Zinc: Dietary intake and impact of supplementation on immune function in elderly. AGE 2013, 35, 839–860. [Google Scholar] [CrossRef]
- Rita Cardoso, B.; Silva Bandeira, V.; Jacob-Filho, W.; Franciscato Cozzolino, S.M. Selenium status in elderly: Relation to cognitive decline. J. Trace Elem. Med. Biol. 2014, 28, 422–426. [Google Scholar] [CrossRef]
- Li, W.F.; Zhang, D.D.; Xia, J.T.; Wen, S.F.; Guo, J.; Li, Z.C. The association between B vitamins supplementation and adverse cardiovascular events: A meta-analysis. Int. J. Clin. Exp. Med. 2014, 7, 1923–1930. [Google Scholar]
- Available online: https://www.sinu.it/html/pag/tabelle_larn_2014_rev.asp (accessed on 30 December 2018).
- Biesalski, H.K.; Tinz, J. Multivitamin/mineral supplements: Rationale and safety—A systematic review. Nutrition 2017, 33, 76–82. [Google Scholar] [CrossRef]
- Katz, S.; Downs, T.D.; Cash, H.R.; Grotz, R.C. Progress in development of the index of ADL. Gerontologist 1970, 10, 20–30. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Stroop, J.R. Studies of interference in serial verbal reactions. J. Exp. Psychol. 1935, 18, 643. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Magni, E.; Binetti, G.; Bianchetti, A.; Rozzini, R.; Trabucchi, M. Mini-Mental State Examination: A normative study in Italian elderly population. Eur. J. Neurol. 1996, 3, 198–202. [Google Scholar] [CrossRef]
- Stijntjes, M.; de Craen, A.J.M.; van Heemst, D.; Meskers, C.G.M.; van Buchem, M.A.; Westendorp, R.G.J.; Slagboom, P.E.; Maier, A.B. Familial Longevity Is Marked by Better Cognitive Performance at Middle Age: The Leiden Longevity Study. PLoS ONE 2013, 8, 5796. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982, 17, 37–49. [Google Scholar] [CrossRef]
- Rabin, R.; de Charro, F. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef]
- Puca, A.A.; Andrew, P.; Novelli, V.; Anselmi, C.V.; Somalvico, F.; Cirillo, N.A.; Chatgilialoglu, C.; Ferreri, C. Fatty acid profile of erythrocyte membranes as possible biomarker of longevity. Rejuvenation Res. 2008, 11, 63–72. [Google Scholar] [CrossRef]
- Pes, G.M.; Lio, D.; Carru, C.; Deiana, L.; Baggio, G.; Franceschi, C.; Ferrucci, L.; Oliveri, F.; Scola, L.; Crivello, A.; et al. Association between longevity and cytokine gene polymorphisms. A study in Sardinian centenarians. Aging Clin. Exp. Res. 2004, 16, 244–248. [Google Scholar] [CrossRef]
- Viviani Anselmi, C.; Ferreri, C.; Novelli, V.; Roncarati, R.; Bronzini, R.; Marchese, G.; Somalvico, F.; Condorelli, G.; Montenero, A.S.; Puca, A.A. Fatty acid percentage in erythrocyte membranes of atrial flutter/fibrillation patients and controls. J. Interv. Card Electrophysiol. 2010, 27, 95–99. [Google Scholar] [CrossRef]
- Sierra, F. The Emergence of Geroscience as an Interdisciplinary Approach to the Enhancement of Health Span and Life Span. Cold Spring Harb. Perspect. Med. 2016, 6, a025163. [Google Scholar] [CrossRef] [PubMed]
- Obeid, R.; Herrmann, W. Mechanisms of homocysteine neurotoxicity in neurodegenerative diseases with special reference to dementia. FEBS Lett. 2006, 580, 2994–3005. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R. Lowering blood homocysteine with folic acid-based supplements: Meta-analysis of randomised trials. Indian Heart J. 2000, 52, 59–64. [Google Scholar]
- McCaddon, A.; Davies, G.; Hudson, P.; Tandy, S.; Cattell, H. Total serum homocysteine in senile dementia of Alzheimer type. Int. J. Geriatr. Psychiatry 1998, 13, 235–239. [Google Scholar] [CrossRef]
- Ford, E.S.; Smith, S.J.; Stroup, D.F.; Steinberg, K.K.; Mueller, P.W.; Thacker, S.B. Homocyst(e)ine and cardiovascular disease: A systematic review of the evidence with special emphasis on case-control studies and nested case-control studies. Int. J. Epidemiol. 2002, 31, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, S.; Beiser, A.; Selhub, J.; Jacques, P.F.; Rosenberg, I.H.; D’Agostino, R.B.; Wilson, P.W.; Wolf, P.A. Plasma homocysteine as a risk factor for dementia and Alzheimer’s disease. N. Engl. J. Med. 2002, 346, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, P.S.; Valenzuela, M.J.; Brodaty, H.; Wang, X.L.; Looi, J.; Lorentz, L.; Howard, L.; Jones, M.; Zagami, A.S.; Gillies, D.; et al. Homocysteine as a risk factor for cognitive impairment in stroke patients. Dement. Geriatr. Cogn. Disord. 2003, 15, 155–162. [Google Scholar] [CrossRef]
- Vidal, J.S.; Dufouil, C.; Ducros, V.; Tzourio, C. Homocysteine, folate and cognition in a large community-based sample of elderly people--the 3C Dijon Study. Neuroepidemiology 2008, 30, 207–214. [Google Scholar] [CrossRef]
- Hooshmand, B.; Polvikoski, T.; Kivipelto, M.; Tanskanen, M.; Myllykangas, L.; Erkinjuntti, T.; Mäkelä, M.; Oinas, M.; Paetau, A.; Scheltens, P.; et al. Plasma homocysteine, Alzheimer and cerebrovascular pathology: A population-based autopsy study. Brain 2013, 136, 2707–2716. [Google Scholar] [CrossRef]
- Kalita, J.; Misra, U.K. Benefit of vitamin B-12 supplementation in asymptomatic elderly: A matter of endpoints. Am. J. Clin. Nutr. 2015, 102, 529–530. [Google Scholar] [CrossRef][Green Version]
- Vogiatzoglou, A.; Refsum, H.; Johnston, C.; Smith, S.M.; Bradley, K.M.; de Jager, C.; Budge, M.M.; Smith, A.D. Vitamin B12 status and rate of brain volume loss in community-dwelling elderly. Neurology 2008, 71, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Hooshmand, B.; Mangialasche, F.; Kalpouzos, G.; Solomon, A.; Kåreholt, I.; Smith, A.D.; Refsum, H.; Wang, R.; Mühlmann, M.; Ertl-Wagner, B.; et al. Association of Vitamin B12, Folate, and Sulfur Amino Acids With Brain Magnetic Resonance Imaging Measures in Older Adults: A Longitudinal Population-Based Study. JAMA Psychiatry 2016, 73, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Narayan, S.K.; Firbank, M.J.; Saxby, B.K.; Stansby, G.; Hansrani, M.; O’Brien, J.T.; Ford, G.A. Elevated plasma homocysteine is associated with increased brain atrophy rates in older subjects with mild hypertension. Dement. Geriatr. Cogn. Disord. 2011, 31, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Leishear, K.; Ferrucci, L.; Lauretani, F.; Boudreau, R.M.; Studenski, S.A.; Rosano, C.; Abbate, R.; Gori, A.M.; Corsi, A.M.; Di Iorio, A.; et al. Vitamin B12 and homocysteine levels and 6-year change in peripheral nerve function and neurological signs. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 537–543. [Google Scholar] [CrossRef]
- Clarke, R.; Harrison, G.; Richards, S.; Vital Trial Collaborative Group. Effect of vitamins and aspirin on markers of platelet activation, oxidative stress and homocysteine in people at high risk of dementia. J. Intern. Med. 2003, 254, 67–75. [Google Scholar]
- Valkovic, P.; Benetin, J.; Blazícek, P.; Valkovicová, L.; Gmitterová, K.; Kukumberg, P. Reduced plasma homocysteine levels in levodopa/entacapone treated Parkinson patients. Parkinsonism Relat. Disord. 2005, 11, 253–256. [Google Scholar] [CrossRef]
- Durga, J.; van Boxtel, M.P.; Schouten, E.G.; Kok, F.J.; Jolles, J.; Katan, M.B.; Verhoef, P. Effect of 3-year folic acid supplementation on cognitive function in older adults in the FACIT trial: A randomised, double blind, controlled trial. Lancet 2007, 369, 208–216. [Google Scholar] [CrossRef]
- Aisen, P.S.; Schneider, L.S.; Sano, M.; Diaz-Arrastia, R.; van Dyck, C.H.; Weiner, M.F.; Bottiglieri, T.; Jin, S.; Stokes, K.T.; Thomas, R.G.; et al. High-dose B vitamin supplementation and cognitive decline in Alzheimer disease: A randomized controlled trial. JAMA. 2008, 300, 177483. [Google Scholar] [CrossRef]
- Passeri, G.; Pini, G.; Troiano, L.; Vescovini, R.; Sansoni, P.; Passeri, M.; Gueresi, P.; Delsignore, R.; Pedrazzoni, M.; Franceschi, C. Low vitamin D status, high bone turnover, and bone fractures in centenarians. J. Clin. Endocrinol. Metab. 2003, 88, 5109–5115. [Google Scholar] [CrossRef]
- Heaney, R.P. Vitamin D, nutritional deficiency, and the medical paradigm. J. Clin. Endocrinol. Metab. 2003, 88, 5107–5108. [Google Scholar] [CrossRef]
- Boucher, B.J. The problems of vitamin d insufficiency in older people. Aging Dis. 2012, 3, 313–329. [Google Scholar] [PubMed]
- Kweder, H.; Eidi, H. Vitamin D deficiency in elderly: Risk factors and drugs impact on vitamin D status. Avicenna J. Med. 2018, 8, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.R.; Aspray, T.J. The role of vitamin D in maintaining bone health in older people. Ther. Adv. Musculoskelet Dis. 2017, 9, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Jennings, A.; Cashman, K.D.; Gillings, R.; Cassidy, A.; Tang, J.; Fraser, W.; Dowling, K.G.; Hull, G.L.J.; Berendsen, A.A.M.; De Groot, L.C.P.G.M.; et al. A Mediterranean-like dietary pattern with vitamin D3 (10 µg/d) supplements reduced the rate of bone loss in older Europeans with osteoporosis at baseline: Results of a 1-y randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Marseglia, A.; Xu, W.; Fratiglioni, L.; Fabbri, C.; Berendsen, A.A.M.; Bialecka-Debek, A.; Jennings, A.; Gillings, R.; Meunier, N.; Caumon, E.; et al. Effect of the NU-AGE Diet on Cognitive Functioning in Older Adults: A Randomized Controlled Trial. Front. Physiol. 2018, 9, 349. [Google Scholar] [CrossRef]
- Bełz, A.; Stolecki, M.; Kudła, M.; Głogowska-Szeląg, J. Vitamin D—A geriatric point of view. Wiad. Lek. 2018, 71, 1628–1631. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A. Optimal serum 25-hydroxyvitamin D levels for multiple health outcomes. Adv. Exp. Med. Biol. 2014, 810, 500–525. [Google Scholar]
- Traber, M.G.; Sies, H. Vitamin E in humans: Demand and delivery. Annu. Rev. Nutr. 1996, 16, 321–347. [Google Scholar] [CrossRef]
- Cohn, W. Bioavailability of vitamin E. Eur. J. Clin. Nutr. 1997, 51, 80–85. [Google Scholar]
- Reboul, E. Vitamin E Bioavailability: Mechanisms of Intestinal Absorption in the Spotlight. Antioxidants 2017, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Roi, S.; Nowicki, M.; Dhaussy, A.; Huertas, A.; Amiot, M.J.; Reboul, E. Fat-soluble vitamin intestinal absorption: Absorption sites in the intestine and interactions for absorption. Food Chem. 2015, 172, 155–160. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.E.; Hurst, R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Dietary factors influencing zinc absorption. J. Nutr. 2000, 130, 1378–1383. [Google Scholar] [CrossRef] [PubMed]
- Krebs, N.F. Overview of zinc absorption and excretion in the human gastrointestinal tract. J. Nutr. 2000, 130, 1374–1377. [Google Scholar] [CrossRef] [PubMed]
- Dupont, J.; Dedeyne, L.; Dalle, S.; Koppo, K.; Gielen, E. The role of omega-3 in the prevention and treatment of sarcopenia. Aging Clin. Exp. Res. 2019, 31, 825–836. [Google Scholar] [CrossRef] [PubMed]
- von Schacky, C. Omega-3 index and cardiovascular health. Nutrients. 2014, 6, 799–814. [Google Scholar] [CrossRef]
- Block, R.C.; Harris, W.S.; Reid, K.J.; Sands, S.A.; Spertus, J.A. EPA and DHA in blood cell membranes from acute coronary syndrome patients and controls. Atherosclerosis 2008, 197, 821–828. [Google Scholar] [CrossRef]
- Harris, W.S. The omega-3 index: Clinical utility for therapeutic intervention. Curr. Cardiol. Rep. 2010, 12, 503–508. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients. 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Gómez-Pinilla, F. Brain foods: The effects of nutrients on brain function. Nat. Rev. Neurosci. 2008, 9, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Beauchet, O. Vitamin d in older adults: The need to specify standard values with respect to cognition. Front. Aging Neurosci. 2014, 15, 72. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A.; Wolters, M.; Hahn, A. Micronutrients at the interface between inflammation and infection--ascorbic acid and calciferol: Part 1, general overview with a focus on ascorbic acid. Inflamm. Allergy Drug Targets. 2011, 10, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Wimalawansa, S.J. Vitamin D Deficiency: Effects on Oxidative Stress, Epigenetics, Gene Regulation, and Aging. Biology 2019, 8, 30. [Google Scholar] [CrossRef]
| Diseases | Baseline FP (n = 24) PF (n = 24) (n, %) | q Value | |
|---|---|---|---|
| Neuropsychological disorders (anxiety/depression/insomnia) | 4 (17%) | 5 (21%) | 1 |
| Hypertension | 15 (63%) | 9 (38%) | 1 |
| Hypercholesterolemia | 2 (8%) | 6 (25%) | 1 |
| Cardiovascular disorders (rhythm disturbances/thrombosis) | 6 (25%) | 6 (25%) | 1 |
| Musculoskeletal system syndromes (arthrosis/hiatal hernia/osteoporosis/fibromyalgia/joint gout) | 3 (13%) | 4 (17%) | 1 |
| Chronic respiratory diseases (asthma/chronic obstructive pulmonary disease) | 0 (0%) | 2 (8%) | 1 |
| Digestive disorders (ulcerative esophagitis/gastroesophageal reflux/gastritis) | 5 (21%) | 4 (17%) | 1 |
| Type 2 diabetes | 1 (4%) | 1 (4%) | 1 |
| Prostatic hyperplasia/hypertrophy | 4 (17%) | 3 (13%) | 1 |
| Ocular hypertension/glaucoma | 2 (8%) | 1 (4%) | 1 |
| Hypothyroidism/hyperthyroidism | 3 (13%) | 3 (13%) | 1 |
| Medications (n) a | 3 ± 2 | 3 ± 2 | 1 |
| Placebo Milk | Fortified Milk | |||||
|---|---|---|---|---|---|---|
| 100 mL | 250 mL | %LARN (250 mL) | 100 mL | 250 mL | %LARN (250 mL) | |
| Energy (kcal) | 46 | 115 | - | 57 | 142 | - |
| Protein (g) | 3.10 | 7.75 | 12% | 4 | 10 | 15.5% |
| Total Carbohydrates (g) | 4.9 | 12.25 | 20% | 5.6 | 14 | 22.9% |
| Dietary fiber (g) | 0 | 0 | 0% | 0 | 0 | 0% |
| Sugars (g) | 4.9 | 12.25 | 20% | 5.6 | 14 | 22.9% |
| Lactose | <0.5 | <1.25 | - | <0.5 | <1.25 | - |
| Total fat (g) | 1.6 | 4 | 20% | 2.04 | 5.1 | 25.5% |
| Saturated fat (g) | 1.2 | 3 | 5% | 1.27 | 3.2 | 5.3% |
| DHA + EPA (mg) | - | - | - | 140 | 350 | 140% |
| Sodium (g) | 0.05 | 0.125 | 10% | 0.04 | 0.1 | 8.3% |
| Vitamin D3 (μg) | 0.05 | 0.125 | 0.70% | 7 | 17.5 | 100% |
| Vitamin E (mg) | 0.03 | 0.075 | 0.60% | 5 | 12.5 | 100% |
| Vitamin C (mg) | 2.1 | 5.25 | 5.50% | 19 | 47.5 | 50% |
| Vitamin B6 (mg) | 0.036 | 0.09 | 5.60% | 0.64 | 1.6 | 100% |
| Vitamin B12 (μg) | 0.35 | 0.875 | 35% | 1 | 2.5 | 100% |
| Vitamin B9 (μg) | 7 | 17.5 | 4.40% | 80 | 200 | 50% |
| Selenium (μg) | 0.5 | 1.25 | 2.30% | 22 | 55 | 100% |
| Zinc (mg) | 0.35 | 0.875 | 8.75% | 4 | 10 | 100% |
| Characteristics | Baseline FP vs. PF | q Value | |
|---|---|---|---|
| Age, years | 71 ± 4 | 70 ± 5 | 0.85 |
| Gender | |||
| Men, n (%) | 13 (54) | 13 (54) | - |
| Women, n (%) | 11 (46) | 11 (46) | |
| Height, cm | 165 ± 0.1 | 165 ± 0.1 | 0.99 |
| Weight, kg | 78± 16 | 72 ±11 | 0.65 |
| BMI, kg/m2 | 28 ± 5 | 26 ± 4 | 0.65 |
| Waist circumference, cm | 99 ± 16 | 93 ± 12 | 0.67 |
| Hip circumference, cm | 106 ± 9 | 101 ± 6 | 056 |
| Waist/hip ratio, cm | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.95 |
| Total cholesterol (mg/100 mL) | 205 ± 36 | 215 ± 43 | 0.85 |
| HDL cholesterol (mg/100 mL) | 56 ± 13 | 56 ± 11 | 0.95 |
| LDL cholesterol (mg/100 mL) | 125 ± 30 | 128 ± 26 | 0.99 |
| Triglycerides (mg/100 mL) | 111 ± 34 | 133 ± 85 | 0.77 |
| Glycaemia (mg/dL) | 100 ± 15 | 103 ± 32 | 0.92 |
| HOMA-IR | 2 ± 2 | 3 ± 2 | 0.92 |
| Creatinine (mg/dL) | 1 ± 0.2 | 1 ± 0.2 | 0.99 |
| Fibrinogen (mg/dL) | 294 ± 46 | 299 ± 42 | 0.99 |
| ALT (U/L) | 18 ± 7 | 18 ± 8 | 0.99 |
| ASP (U/L) | 19 ± 4 | 20 ± 5 | 0.99 |
| Micronutrients | Baseline FP vs. PF | q Value | After Washout FP vs. PF | q Value | ||
|---|---|---|---|---|---|---|
| Vitamin B9 (ng/mL) | 7 ± 4 | 7 ± 3 | 0.96 | 7.6 ± 3 | 6 ± 2 | 0.44 |
| Vitamin B6 (µg/L) | 14 ± 9 | 15 ± 6 | 0.70 | 18 ± 5 | 20 ± 25 | 0.96 |
| Vitamin B12 (pg/mL) | 376 ± 174 | 385 ± 125 | 0.96 | 339 ± 109 | 352 ± 154 | 0.96 |
| Vitamin 25 (OH)D (ng/mL) | 16 ± 11 | 15 ± 6 | 0.96 | 23 ± 9 | 20 ± 5 | 0.70 |
| Vitamin E (µg/mL) | 11 ± 3 | 13 ± 3 | 0.68 | 14 ± 3 | 12 ± 2 | 0.40 |
| Zinc (µg/L) | 809 ± 89 | 791 ± 172 | 0.96 | 799 ± 87 | 801 ± 97 | 0.96 |
| Selenium (µg/L) | 96 ± 11 | 97 ± 9 | 0.96 | 108 ± 14 | 108 ± 11 | 0.96 |
| DHA (% in e.m.) | 5 ± 1 | 5 ± 1 | 0.96 | 5.4 ± 1 | 4.4 ± 1 | 0.289 |
| EPA (% in e.m.) | 0.9 ± 0.4 | 0.9 ± 0.1 | 0.96 | 0.7 ± 0.3 | 0.8 ± 0.4 | 0.96 |
| Micronutrients | Reference Range | Baseline | After Fortified Milk | 95% CI | q Value |
|---|---|---|---|---|---|
| Vitamin B9 (ng/mL) | 2.5–20 | 7 ± 3 | 13 ± 5 | 5 (4–6) | <0.0001 * |
| Vitamin B6 (µg/L) | 3.6–18 | 16 ± 18 | 22 ± 10 | 4 (1–7) | 0.015 * |
| Vitamin B12 (pg/mL) | 200–910 | 378 ± 158 | 395 ± 153 | 52 (33–71) | 0.0001 * |
| Vitamin 25(OH)D (ng/mL) | 30–100 | 18 ± 10 | 23 ± 6 | 7 (5–9) | 0.0002 * |
| Vitamin E (µg/mL) | 3–12 | 12 ± 3 | 13 ± 3 | 1 (0–2) | 0.110 |
| Zinc (µg/L) | 600–1200 | 805 ± 89 | 798 ± 102 | −10 (−52–32) | 0.786 |
| Selenium (µg/L) | 0–150 | 102 ± 14 | 108 ± 15 | 4 (0.3–7) | 0.106 |
| Fatty Acids | Reference Range | Baseline | After Fortified Milk | 95% CI | q Value |
|---|---|---|---|---|---|
| Linoleic acid (%) | 9–16 | 11 ± 1 | 11 ± 1 | 0 (−1–0) | 0.058 |
| DGLA (%) | 1.9–2.4 | 2 ± 0.6 | 2 ± 0.3 | −0.1 (−0.3–0) | 0.190 |
| Arachidonic acid (%) | 13–17 | 16.3 ± 1 | 15.9 ± 2 | −1 (−1–0) | 0.001 * |
| TFA (%) | 0–0.4 | 0.2 ± 0.1 | 0.3 ± 0.1 | 0.02 (−0.03–0.07) | 0.559 |
| SFA (%) | 30–45 | 45 ± 2 | 45 ± 2 | −0.1 (−0.8–0.6) | 0.870 |
| MUFA (%) | 13–23 | 19 ± 1 | 19 ± 2 | −0.1 (−0.5–0.3) | 0.766 |
| PUFA (%) | 28–39 | 35 ± 2 | 36 ± 2 | 0.1 (−0.7–0.9) | |
| DHA (% in e. m.) | 5–7 | 5 ± 1 | 6 ± 1 | 1 (1–2) | <0.0001 * |
| EPA (% in e. m.) | 0.5–0.9 | 0.9 ± 0.4 | 1.1 ± 0.3 | 0.2 (0–0.3) | 0.040 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martucci, M.; Conte, M.; Bucci, L.; Giampieri, E.; Fabbri, C.; Palmas, M.G.; Izzi, M.; Salvioli, S.; Zambrini, A.V.; Orsi, C.; et al. Twelve-Week Daily Consumption of ad hoc Fortified Milk with ω-3, D, and Group B Vitamins Has a Positive Impact on Inflammaging Parameters: A Randomized Cross-Over Trial. Nutrients 2020, 12, 3580. https://doi.org/10.3390/nu12113580
Martucci M, Conte M, Bucci L, Giampieri E, Fabbri C, Palmas MG, Izzi M, Salvioli S, Zambrini AV, Orsi C, et al. Twelve-Week Daily Consumption of ad hoc Fortified Milk with ω-3, D, and Group B Vitamins Has a Positive Impact on Inflammaging Parameters: A Randomized Cross-Over Trial. Nutrients. 2020; 12(11):3580. https://doi.org/10.3390/nu12113580
Chicago/Turabian StyleMartucci, Morena, Maria Conte, Laura Bucci, Enrico Giampieri, Cristina Fabbri, Maria Giustina Palmas, Massimo Izzi, Stefano Salvioli, Angelo Vittorio Zambrini, Carla Orsi, and et al. 2020. "Twelve-Week Daily Consumption of ad hoc Fortified Milk with ω-3, D, and Group B Vitamins Has a Positive Impact on Inflammaging Parameters: A Randomized Cross-Over Trial" Nutrients 12, no. 11: 3580. https://doi.org/10.3390/nu12113580
APA StyleMartucci, M., Conte, M., Bucci, L., Giampieri, E., Fabbri, C., Palmas, M. G., Izzi, M., Salvioli, S., Zambrini, A. V., Orsi, C., Brigidi, P., Santoro, A., Capri, M., Monti, D., & Franceschi, C. (2020). Twelve-Week Daily Consumption of ad hoc Fortified Milk with ω-3, D, and Group B Vitamins Has a Positive Impact on Inflammaging Parameters: A Randomized Cross-Over Trial. Nutrients, 12(11), 3580. https://doi.org/10.3390/nu12113580





