Whole Goat Milk as a Source of Fat and Milk Fat Globule Membrane in Infant Formula
Abstract
1. Breast Milk and Breast Milk Substitutes
2. Infant Formula Made from Goat Milk
3. Whole Goat Milk Fat to Supply a Variety of Fatty Acids and sn-2 Palmitate
4. Whole Goat Milk as a Source of Cholesterol
5. The Secretion of Goat Milk
6. Whole Goat Milk as a Source of MFGM Polar Lipids
7. Whole Goat Milk as a Source of MFGM Proteins
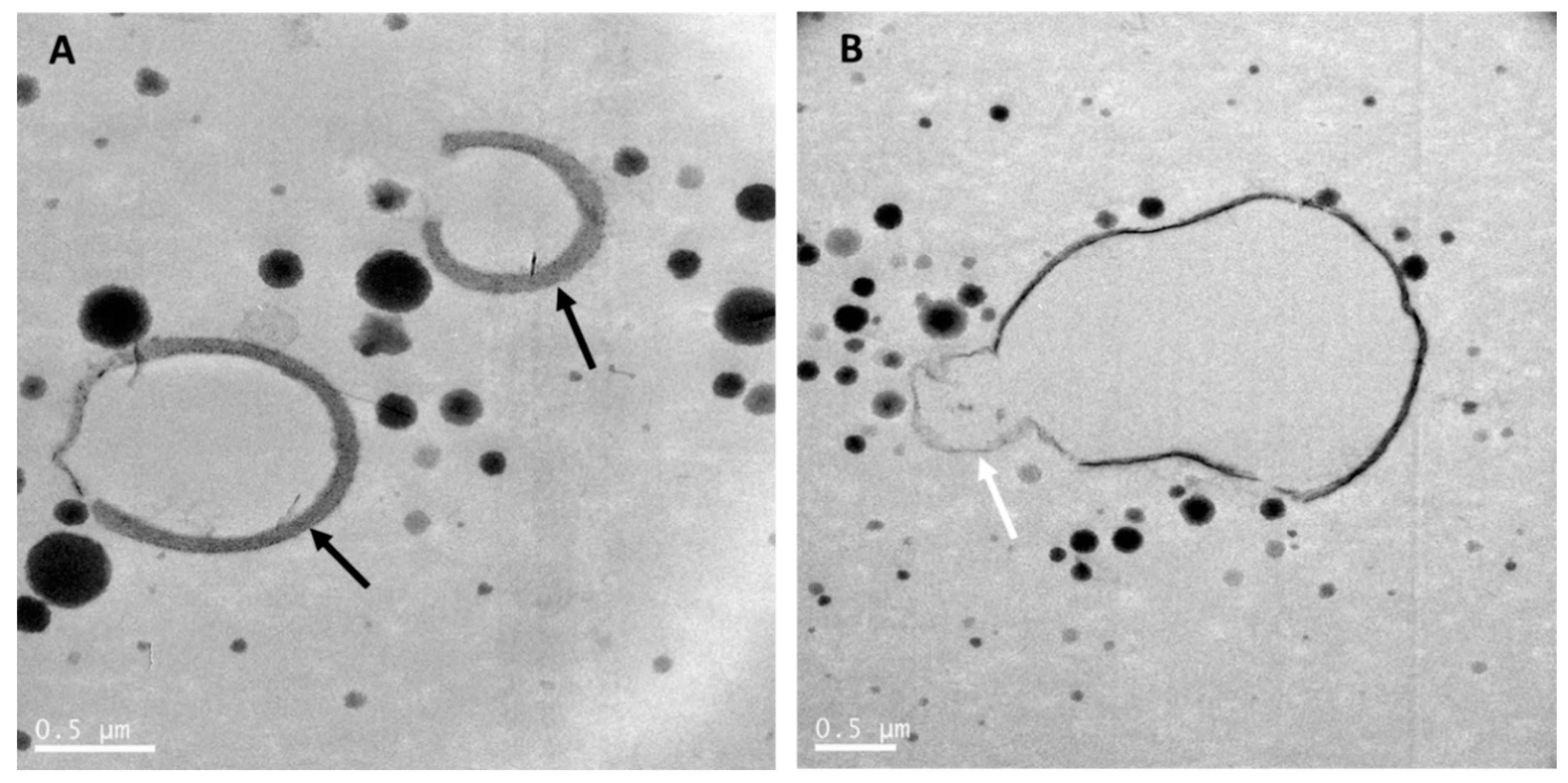
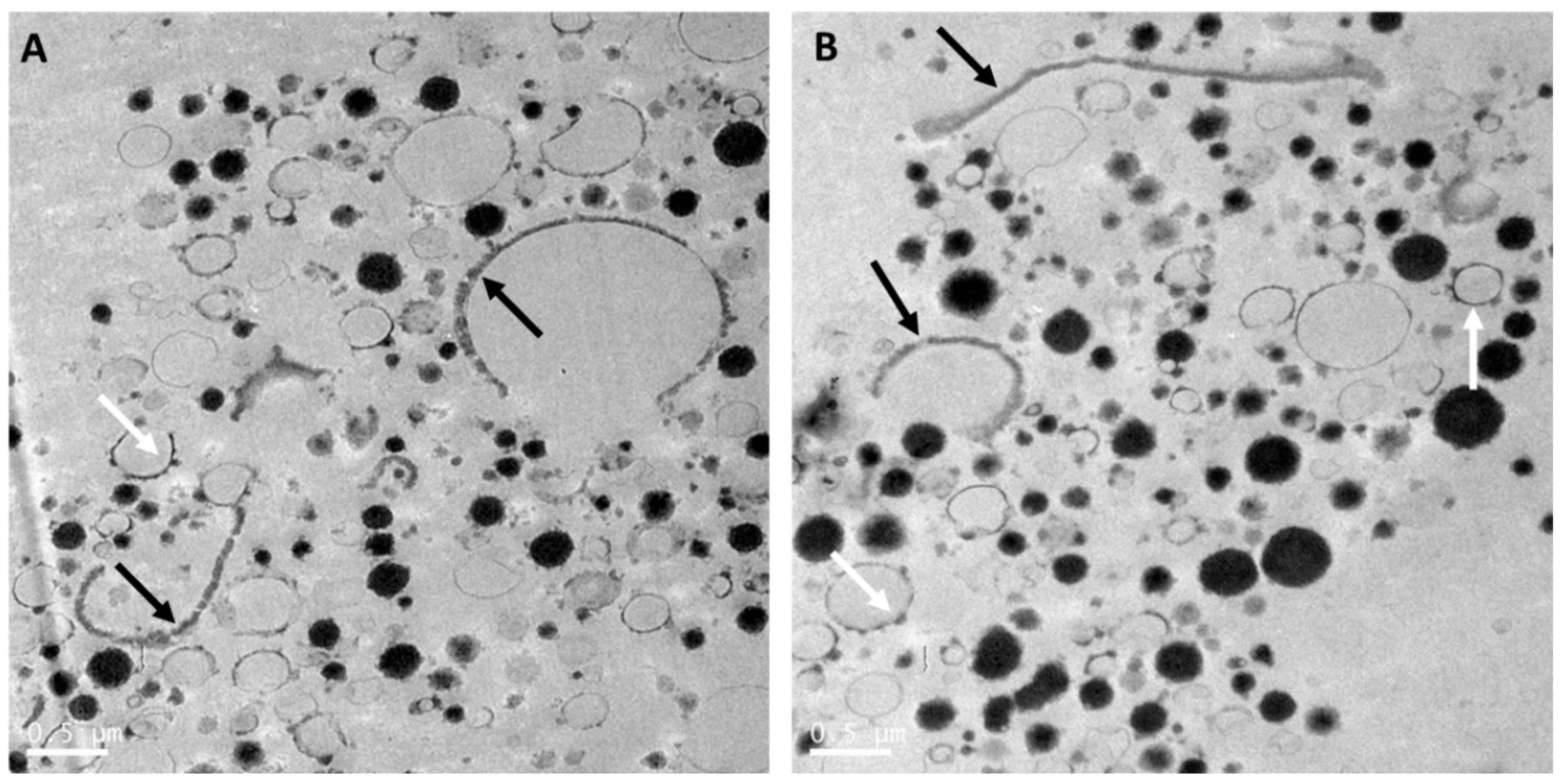
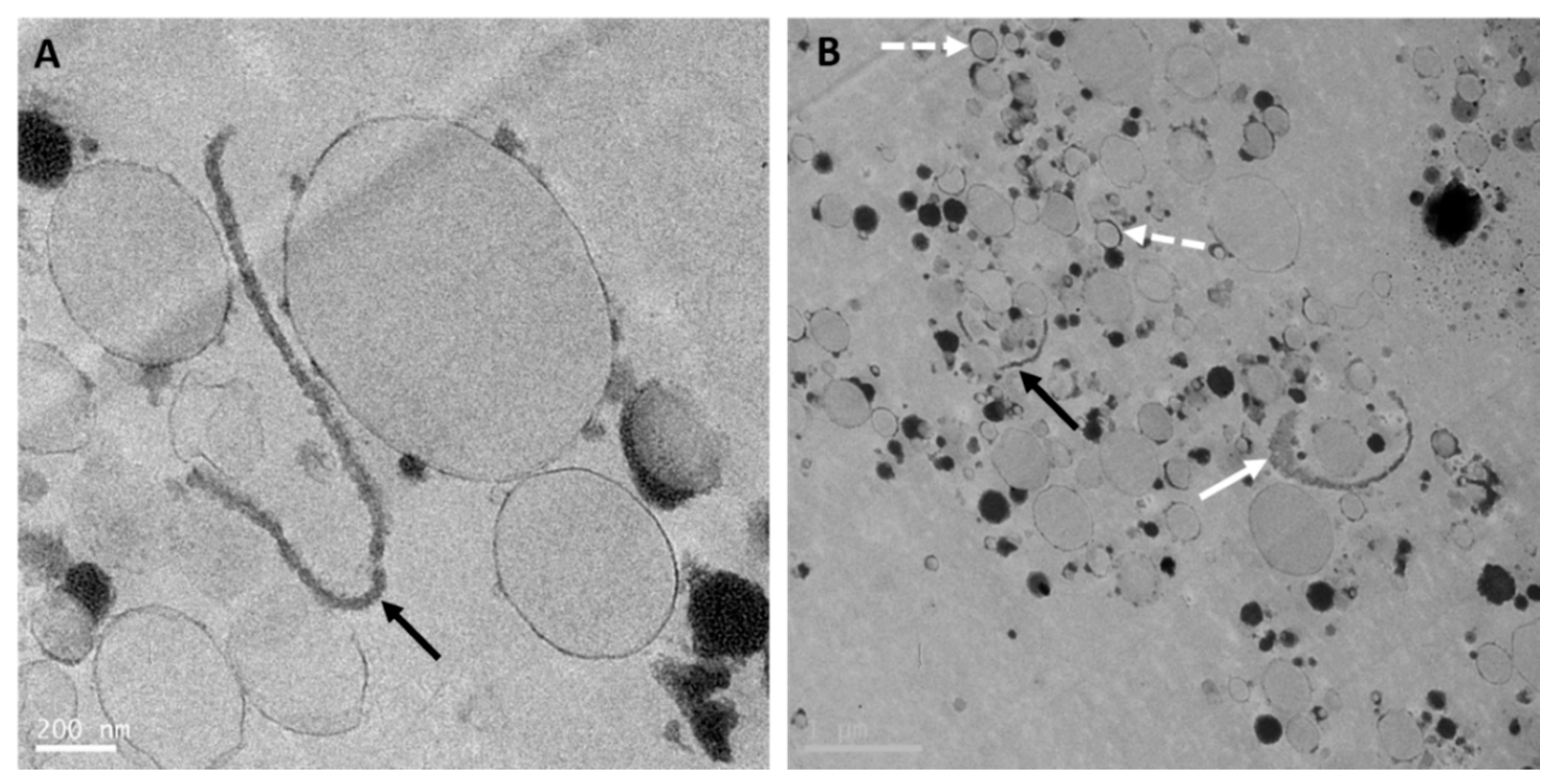
8. The Relevance of the Fat Microstructure in Infant Nutrition
9. Role of Milk Fat and MFGM in Infant Nutrition, Growth and Development
9.1. Influence of Lipid Composition and Structure on Lipid Digestion and Metabolism
9.1.1. Kinetics of Digestion
9.1.2. Triglyceride Structure
9.1.3. Cholesterol and Its Importance in Early Life
9.1.4. Size and Microstructure of the Milk Fat Globules
9.2. The Role of Milk Lipids in Cognition and Brain Development
9.2.1. The Role of Milk Lipids
9.2.2. The Role of MFGM Lipids
9.3. Development of the Immune System
9.3.1. Milk Lipids Have Antimicrobial Properties
9.3.2. The Role of MFGM
9.3.3. Modulation of the Gut Microbiota
9.3.4. Can Milk Lipids Mediate the Development of Allergies?
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- WHO (World Health Organisation). Report of the Expert Consultation on the Optimal Duration of Exclusive Breastfeeding; WHO: Geneva, Switzerland, 2001. [Google Scholar]
- Delplanque, B.; Gibson, R.; Koletzko, B.; Lapillonne, A.; Strandvik, B. Lipid quality in infant nutrition: Current knowledge and future opportunities. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 8–17. [Google Scholar] [CrossRef]
- Hageman, J.J.J.; Danielsen, M.; Nieuwenhuizen, A.G.; Feitsma, A.L.; Dalsgaard, T.K. Comparison of bovine milk fat and vegetable fat for infant formula: Implications for infant health. Int. Dairy J. 2019, 92, 37–49. [Google Scholar] [CrossRef]
- Lönnerdal, B. Infant formula and infant nutrition: Bioactive proteins of human milk and implications for composition of infant formulas. Am. J. Clin. Nutr. 2014, 99, 712S–717S. [Google Scholar] [CrossRef]
- Gallier, S.; Vocking, K.; Post, J.A.; Van De Heijning, B.; Acton, D.; Van Der Beek, E.M.; Van Baalen, T. A novel infant milk formula concept: Mimicking the human milk fat globule structure. Colloid Surf. B 2015, 136, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Dietary Triacylglycerol Structure and Its Role in Infant Nutrition. Adv. Nutr. 2011, 2, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Bronsky, J.; Campoy, C.; Embleton, N.; Fewtrell, M.; Mis, N.F.; Gerasimidis, K.; Hojsak, I.; Hulst, J.; Indrio, F.; Lapillonne, A.; et al. Palm Oil and Beta-palmitate in Infant Formula: A Position Paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 742–760. [Google Scholar] [CrossRef]
- Beekman, J.; MacMahon, S. The impact of infant formula production on the concentrations of 3-MCPD and glycidyl esters. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2020, 37, 48–60. [Google Scholar] [CrossRef]
- Verduci, E.; Eliose, S.; Cerrato, L.; Comberiati, P.; Calvani, M.; Palazzo, S.; Martelli, A.; Landi, M.; Trikamjee, T.; Peroni, D.G. Cow’s milk substitutes for children: Nutritional aspects of milk from different mammalian species, special formula and plant-based beverages. Nutrients 2019, 11, 1739. [Google Scholar] [CrossRef]
- Mennella, J.A.; Beauchamp, G.K. Flavor experiences during formula feeding are related to preferences during childhood. Early Hum. Dev. 2002, 68, 71–82. [Google Scholar] [CrossRef]
- Bahtia, J.; Greer, F.; Committee on Nutrition. Use of soy protein-based formulas in infant feeding. Pediatrics 2008, 121, 1062–1068. [Google Scholar] [CrossRef]
- Almaas, H.; Cases, A.L.; Devold, T.G.; Holm, H.; Langsrud, T.; Aabakken, L.; Aadnoey, T.; Vegarud, G.E. In vitro digestion of bovine and caprine milk by human gastric and duodenal enzymes. Int. Dairy J. 2006, 16, 961–968. [Google Scholar] [CrossRef]
- Hodgkinson, A.J.; Wallace, O.A.M.; Boggs, I.; Broadhurst, M.; Prosser, C.G. Gastric digestion of cow and goat milk: Impact of infant and young child in vitro digestion conditions. Food Chem. 2017, 245, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Maathuis, A.; Havenaar, R.; He, T.; Bellmann, S. Protein digestion and quality of goat and cow milk infant formula and human milk under simulated infant conditions. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 661–666. [Google Scholar] [CrossRef]
- Hodgkinson, A.J.; McDonald, N.A.; Kivits, L.J.; Hurford, D.R.; Fahey, S.; Prosser, C.G. Allergic responses induced by goat milk αs1-casein in a murine model of gastrointestinal atopy. J. Dairy Sci. 2012, 95, 83–90. [Google Scholar] [CrossRef]
- Neveu, C.; Riaublanc, A.; Miranda, G.; Chich, J.F.; Martin, P. Is the apocrine milk secretion process observed in the goat species rooted in the perturbation of the intracellular transport mechanism induced by defective alleles at the αs1-Cn locus? Reprod. Nutr. Dev. 2002, 42, 163–172. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Y.; Eastwood, B.; Yang, Z.; de Campo, L.; Knott, R.; Prosser, C.; Carpenter, E.; Hemar, Y. Rheological and structural characterization of acidified skim milks and infant formulae made form cow and goat milk. Food Hydrocoll. 2019, 96, 161–170. [Google Scholar] [CrossRef]
- Jung, T.H.; Hwang, H.J.; Yun, S.S.; Lee, W.J.; Kim, J.W.; Ahn, J.Y.; Jeon, W.M.; Han, K.S. Hypoallergenic and Physicochemical Properties of the A2 β-Casein Fraction of Goat Milk. Korean J. Food Sci. Anim. Resour. 2017, 37, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Morales, F.A.; Castel Genis, J.M.; Guerrero, Y.M. Current status, challenges and the way forward for dairy goat production in Europe. Asian-Australas. J. Anim. Sci. 2019, 32, 1256–1265. [Google Scholar] [CrossRef]
- Sepe, L.; Argüello, A. Recent advances in dairy goat products. Asian Australas. J. Anim. Sci. 2019, 32, 1306–1320. [Google Scholar] [CrossRef]
- Grant, C.; Rotherham, B.; Sharpe, S.; Scragg, R.; Thompson, J.; Andrews, J.; Wall, C.; Murphy, J.; Lowry, D. Randomized, double-blind comparison of growth in infants receiving goat milk formula versus cow milk infant formula. J. Paediatr. Child Health 2005, 41, 564–568. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific opinion on the suitability of goat milk protein as a source of protein in infant formulae and in follow-on formulae. EFSA J. 2012, 10, 2603. [Google Scholar] [CrossRef][Green Version]
- Zhou, S.J.; Sullivan, T.; Gibson, R.A.; Lönnerdal, B.; Prosser, C.G.; Lowry, D.J.; Makrides, M. Nutritional adequacy of goat milk infant formulas for term infants: A double-blind randomised controlled trial. Br. J. Nutr. 2014, 111, 1641–1651. [Google Scholar] [CrossRef] [PubMed]
- Chilliard, Y.; Rouel, J.; Leroux, C. Goat’s alpha-s1 casein genotype influences its milk fatty acid composition and delta-9 desaturation ratios. Anim. Feed Sci. Technol. 2006, 131, 474–487. [Google Scholar] [CrossRef]
- Prosser, C.G.; Svetashev, V.I.; Vyssotski, M.V.; Lowry, D.J. Composition and distribution of fatty acids in triglycerides from goat infant formulas with milk fat. J. Dairy Sci. 2010, 93, 2857–2862. [Google Scholar] [CrossRef]
- Bernard, L.; Bonnet, M.; Delavaud, C.; Delosiere, M.; Ferlay, A.; Fougere, H.; Graulet, B. Milk fat globule in ruminant: Major and minor compounds, nutritional regulation and differences among species. Eur. J. Lipid Sci. Technol. 2018, 120, 1700039. [Google Scholar] [CrossRef]
- Prentice, P.M.; Schoemaker, M.H.; Vervoort, J.; Hettinga, K.; Lambers, T.T.; van Tol, E.A.F.; Acerini, C.L.; Olga, L.; Petry, C.J.; Hughes, I.A.; et al. Human milk short-chain fatty acid composition is associated with adiposity in infants. J. Nutr. 2019, 149, 716–722. [Google Scholar] [CrossRef]
- Stinson, L.F.; Gay, M.C.L.; Koleva, P.T.; Eggesbø, M.; Johnson, C.C.; Wegienka, G.; du Toit, E.; Shimojo, N.; Munblit, D.; Campbell, D.E.; et al. Human milk from atopic mothers has lower levels of short chain fatty acids. Front. Immunol. 2020, 11, 1427. [Google Scholar] [CrossRef]
- Martysiak-Żurowska, D.; Kiełbratowska, B.; Szlagatys-Sidorkiewicz, A. The content of conjugated linoleic acid and vaccenic acid in the breast milk of women from Gdansk and the surrounding district, as well as in, infant formulas and follow-up formulas. Nutritional recommendation for nursing women. Dev. Period Med. 2018, 22, 128–134. [Google Scholar]
- Jenness, R. Composition and Characteristics of Goat Milk: Review 1968−1979. J. Dairy Sci. 1980, 63, 1605–1630. [Google Scholar] [CrossRef]
- Innis, S.M. Palmitic acid in early human development. Crit. Rev. Food Sci. Nutr. 2015, 56, 1952–1959. [Google Scholar] [CrossRef]
- Ha, J.K.; Lindsay, R.C. Release of volatile branched-chain and other fatty acids from ruminant milk fats by various lipases. J. Dairy Sci. 1993, 76, 677–690. [Google Scholar] [CrossRef] [PubMed]
- Dingess, K.A.; Valentine, C.J.; Ollberding, N.J.; Davidson, B.S.; Woo, J.G.; Summer, S.; Peng, Y.M.; Guerrero, M.L.; Ruiz-Palacios, G.M.; Ran-Ressler, R.R.; et al. Branched-chain fatty acid composition of human milk and the impact of maternal diet: The Global Exploration of Human Milk (GEHM) Study. Am. J. Clin. Nutr. 2017, 105, 177–184. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.K.; Park, Y.; Behre, R.; Harrison, L.Y.; Shultz, T.D.; McGuire, M.A. Conjugated linoleic acid concentrations of human milk and infant formula. Nutr. Res. 1991, 17, 1277–1283. [Google Scholar] [CrossRef]
- Giuffrida, F.; Marmet, C.; Tavazzi, I.; Fontannaz, P.; Sauser, J.; Lee, L.Y.; Destaillats, F. Quantification of 1,3-olein-2-palmitin (OPO) and Palmitic Acid in sn-2 Position of Triacylglycerols in Human Milk by Liquid Chromatography Coupled with Mass Spectrometry. Molecules 2019, 24, 22. [Google Scholar] [CrossRef] [PubMed]
- Bourlieu, C.; Bouzerzour, K.; Ferret-Bernard, S.; Le Bourgot, C.; Chever, S.; Menard, O.; Deglaire, A.; Cuinet, I.; Le Ruyet, P.; Bonhomme, C.; et al. Infant formula interface and fat source impact on neonatal digestion and gut microbiota. Eur. J. Lipid Sci. Technol. 2015, 117, 1500–1512. [Google Scholar] [CrossRef]
- Timby, N.; Domellöf, E.; Hernell, O.; Lönnerdal, B.; Domellöf, M. Neurodevelopment, nutrition, and growth until 12 mo of age in infants fed a low-energy, low-protein formula supplemented with bovine milk fat globule membranes: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 99, 860–868. [Google Scholar] [CrossRef]
- Claumarchirant, L.; Matencio, E.; Sanchez-Siles, L.M.; Alegria, A.; Lagarda, M.J. Sterol composition in infant formulas and estimated intake. J. Agric. Food Chem. 2015, 63, 7245–7251. [Google Scholar] [CrossRef]
- Gallier, S.; MacGibbon, A.K.H.; McJarrow, P. Milk fat globule membrane (MFGM) supplementation and cognition. Agro FOOD Ind. Hi-Tech 2018, 29, 14–16. [Google Scholar]
- Lee, H.; Padhi, E.; Hasegawa, Y.; Larke, J.; Parenti, M.; Wang, A.; Hernell, O.; Lönnerdal, B.; Slupsky, C. Compositional dynamics of the milk fat globule and its role in infant development. Front. Pediatr. 2018, 6, 313. [Google Scholar] [CrossRef]
- Jensen, R.G.; Ferris, A.M.; Lammi-Keefe, C.J.; Henderson, R.A. Lipids of bovine and human milks: A comparison. J. Dairy Sci. 1990, 73, 223–240. [Google Scholar] [CrossRef]
- Wooding, F.B.P. The mechanism of secretion of the milk fat globule. J. Cell Sci. 1971, 9, 805–821. [Google Scholar] [PubMed]
- Wooding, F.B.P.; Peaker, M.; Linzell, J.L. Theories of milk secretion: Evidence from the electron microscopic examination of milk. Nature 1970, 226, 762–764. [Google Scholar] [CrossRef] [PubMed]
- Janssen, M.M.T.; Walstra, P. Cytoplasmic remnants in milk of certain species. Neth. Milk Dairy J. 1982, 36, 365. [Google Scholar]
- Huston, G.E.; Patton, S. Factors related to the formation of cytoplasmic crescents on milk fat globules. J. Dairy Sci. 1990, 73, 2061–2066. [Google Scholar] [CrossRef]
- Cebo, C.; Lopez, C.; Henry, C.; Beauvallet, C.; Menard, O.; Bevilacqua, C.; Bouvier, F.; Caillat, H.; Martin, P. Goat αs1-casein genotype affects milk fat globule physicochemical properties and the composition of the milk fat globule membrane. J. Dairy Sci. 2012, 95, 6215–6229. [Google Scholar] [CrossRef]
- Boutinaud, M.; Jammes, H. Potential uses of milk epithelial cells: A review. Reprod. Nutr. Dev. 2002, 42, 133–147. [Google Scholar] [CrossRef]
- Chanat, E.; Martin, P.; Ollivier-Bousquet, M. Alpha(S1)-casein is required for the efficient transport of beta- and kappa-casein from the endoplasmic reticulum to the golgi apparatus of mammary epithelial cells. J. Cell Sci. 1999, 112, 3399–3412. [Google Scholar]
- Zheng, L.; Fleith, M.; Giuffrida, F.; O’Neill, B.V.; Schneider, N. Dietary polar lipids and cognitive development: A narrative review. Adv. Nutr. 2019, 10, 1163–1176. [Google Scholar] [CrossRef]
- MacKenzie, A.; Vyssotski, M.; Nekrasov, E. Quantitative analysis of dairy phospholipids by 31P NMR. J. Am. Oil Chem. Soc. 2009, 86, 757–763. [Google Scholar] [CrossRef]
- Pimentel, L.; Gomes, A.; Pintado, M.; Rodriguez-Alcala, L.M. Isolation and analysis of phospholipids in dairy foods. J. Anal. Methods Chem. 2016. [Google Scholar] [CrossRef]
- Garcia, C.; Lutz, N.W.; Confort-Gouny, S.; Cozzone, P.J.; Armand, M.; Bernard, M. Phospholipid fingerprints of milk from different mammalians determined by 31P NMR: Towards specific interest in human health. Food Chem. 2012, 135, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Puente, R.; Garcia-Pardo, L.A.; Rueda, R.; Gil, A.; Hueso, P. Changes in ganglioside and sialic acid contents of goat milk during lactation. J. Dairy Sci. 1994, 77, 39–44. [Google Scholar] [CrossRef]
- Puente, R.; Garcia-Pardo, L.A.; Rueda, R.; Gil, A.; Hueso, P. Seasonal variations in the concentration of gangliosides and sialic acids in milk from different mammalian species. Int. Dairy J. 1996, 6, 315–322. [Google Scholar] [CrossRef]
- De Sousa, Y.R.F.; Vasconcelos, M.A.; Costa, R.G.; Filho, C.A.; de Paiva, E.P.; Queiroga, R.C.R. Sialic acid content of goat milk during lactation. Livest. Sci. 2015, 177, 175–180. [Google Scholar] [CrossRef]
- Khor, G.L.; Shyam, S.; Misra, S.; Fong, B.; Chong, M.H.Z.; Sulaiman, N.; Lee, Y.L.; Cannan, R.; Rowan, A. Correlation between dietary intake and serum ganglioside concentrations: A cross-sectional study among Malaysian toddlers. BMC Nutr. 2016, 2, 74. [Google Scholar] [CrossRef]
- Altman, M.O.; Gagneux, P. Absence of Neu5Gc and Presence of Anti-Neu5Gc Antibodies in Humans—An Evolutionary Perspective. Front. Immunol. 2019, 10, 789. [Google Scholar] [CrossRef]
- Hernell, O.; Timby, N.; Domellöf, M.; Lönnerdal, B. Clinical benefits of milk fat globule membranes for infants and children. J. Pediatr. 2016, 173, S60–S65. [Google Scholar] [CrossRef]
- Cebo, C.; Caillat, H.; Bouvier, F.; Martin, P. Major proteins of the milk fat globule membrane. J. Dairy Sci. 2010, 93, 868–876. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, C.; Sun, X.; Jiang, S.; Guo, M. Characterization of the milk fat globule membrane proteome in colostrum and mature milk of Xinong Saanen goats. J. Dairy Sci. 2020, 103, 3017–3024. [Google Scholar] [CrossRef]
- Manoni, M.; Di Lorenzo, C.; Ottoboni, M.; Tretola, M.; Pinotti, L. Comparative proteomics of milk fat globule membrane (MFGM) proteome across species and lactation stages and the potentials of MFGM fractions in infant formula preparation. Foods 2020, 9, 1251. [Google Scholar] [CrossRef]
- Cebo, C.; Martin, P. Inter-species comparison of milk fat globule membrane proteins highlights the molecular diversity of lactadherin. Int. Dairy J. 2012, 24, 70–77. [Google Scholar] [CrossRef]
- Juvarajah, T.; Wan-Ibrahim, W.I.; Ashrafzadeh, A.; Othman, S.; Hashim, O.H.; Fung, S.Y.; Abdul-Rahman, P.S. Human milk fat globule membrane contains hundreds of abundantly expressed and nutritionally beneficial proteins that are generally lacking in caprine milk. Breastfeed. Med. 2018, 13, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Gallier, S. Nature’s complex emulsion: The fat globules of milk. Food Hydrocolloid 2017, 68, 81–89. [Google Scholar] [CrossRef]
- Fontecha, J.; Brink, L.; Wu, S.; Pouliot, Y.; Visioli, F.; Jimenez-Flores, R. Sources, production, and clinical treatments of milk fat globule membrane for infant nutrition and well-being. Nutrients 2020, 12, 1607. [Google Scholar] [CrossRef]
- Armand, M.; Hamosh, M.; Mehta, N.R.; Angelus, P.A.; Philpott, J.R.; Henderson, T.R.; Dwyer, N.K.; Lairon, D.; Hamosh, P. Effect of human milk or formula on gastric function and fat digestion in the premature infant. Pediatr Res. 1996, 40, 429–437. [Google Scholar] [CrossRef]
- Hamosh, M.; Peterson, J.A.; Henderson, T.R.; Scallan, C.D.; Kiwan, R.; Ceriani, R.L.; Armand, M.; Mehta, N.R.; Hamosh, P. Protective function of human milk: The milk fat globule. Semin. Perinatol. 1999, 23, 242–249. [Google Scholar] [CrossRef]
- Bar-Yoseph, F.; Lifshitz, Y.; Cohen, T. Review of sn-2 palmitate oil implications for infant health. Prostaglandins Leukot. Essent. Fatty Acids 2013, 89, 139–143. [Google Scholar] [CrossRef]
- Han, Y.; Chang, E.Y.; Kim, J.; Ahn, K.; Kim, H.Y.; Hwang, E.M.; Lowry, D.; Prosser, C.; Lee, S.I. Association of infant feeding practices in the general population with infant growth and stool characteristics. Nutr. Res. Pract. 2011, 5, 308–312. [Google Scholar] [CrossRef][Green Version]
- Infante, D.D.; Prosser, C.G.; Tormo, R. Constipated patients fed goat milk protein formula: A case series study. J. Nutr. Health Sci. 2018, 5, 203. [Google Scholar]
- Xu, M.; Wang, Y.; Dai, Z.; Zhang, Y.; Li, Y.; Wang, J. Comparison of growth and nutritional status I ninfants receiving goat milk-based formula or cow milk-based formula: A randomized, double-blind study. Food Nutr. Res. 2015, 59, 28613. [Google Scholar] [CrossRef]
- Breij, L.M.; Abrahamse-Berkeveld, M.; Vandenplas, Y.; Jespers, S.N.J.; de Mol, A.C.; Khoo, P.C.; Kalenga, M.; Peeters, S.; van Beek, R.H.T.; Norbruis, O.F.; et al. An infant formula with large, milk phospholipid–coated lipid droplets containing a mixture of dairy and vegetable lipids supports adequate growth and is well tolerated in healthy, term infants. Am. J. Clin. Nutr. 2019, 109, 586–596. [Google Scholar] [CrossRef] [PubMed]
- Kriaa, A.; Bourgin, M.; Potiron, A.; Mkaouar, H.; Jablaoui, A.; Gerard, P.; Maguin, E.; Rhimi, M. Microbial impact on cholesterol and bile acid metabolism: Current status and future prospects. J. Lipid Res. 2019, 60, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Molinero, N.; Ruiz, L.; Sanchez, B.; Margolles, A.; Delgado, S. Intestinal bacteria interplay with bile and cholesterol metabolism: Implications on host physiology. Front. Physiol. 2019, 10, 185. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A. Role of sphingolipids in infant gut health and immunity. J. Pediatr. 2016, 173, S53–S59. [Google Scholar] [CrossRef]
- Wong, W.W.; Hachey, D.L.; Insull, W.; Opekun, A.R.; Klein, P.D. Effect of dietary cholesterol synthesis in breast-fed or formula-fed infants. J. Lipid Res. 1993, 34, 1403–1411. [Google Scholar]
- Shamir, R.; Nganga, A.; Berkowitz, D.; Diamond, E.; Lischinsky, S.; Lombardo, D.; Shehadeh, N. Serum levels of bile-salt stimulated lipase and breast feeding. J. Pediatr. Endocrinol. Metab. 2003, 16, 1289–1294. [Google Scholar] [CrossRef]
- Bayley, T.M.; Alasmi, M.; Thorkelson, T.; Jones, P.J.H.; Corcoran, J.; Krug-Wispe, S.; Tsang, R.C. Longer term effects of early dietary cholesterol level on synthesis and circulating cholesterol concentrations in human infants. Metabolism 2002, 51, 25–33. [Google Scholar] [CrossRef]
- Timby, N.; Lönnerdal, B.; Hernell, O.; Domellöf, M. Cardiovascular risk markers until 12 mo of age in infants fed a formula supplemented with bovine milk fat globule membranes. Pediatr. Res. 2014, 76, 394–400. [Google Scholar] [CrossRef]
- Le Huërou-Luron, I.; Bouzerzour, K.; Ferret-Bernard, S.; Menard, O.; Le Normand, L.; Perrier, C.; Le Bourgot, C.; Jardin, J.; Bourlieu, C.; Carton, T.; et al. A Mixture of milk and vegetable lipids in infant formula changes gut digestion, mucosal immunity and microbiota composition in neonatal piglets. Eur. J. Nutr. 2018, 57, 463–476. [Google Scholar] [CrossRef]
- He, X.; Parenti, M.; Grip, T.; Domellöf, M.; Lönnerdal, B.; Hernell, O.; Timby, N.; Slupsky, C.M. Metabolic phenotype of breast-fed infants, and infants fed standard formula or bovine supplemented formula: A randomized controlled trial. Sci. Rep. 2019, 9, 339. [Google Scholar] [CrossRef]
- Gianni, M.L.; Roggero, P.; Baudry, C.; Fressange-Mazda, C.; Galli, C.; Agostoni, C.; Le Ruyet, P.; Mosca, F. An infant formula containing dairy lipids increased red blood cell membrane omega 3 fatty acids in 4 month-old healthy newborns: A randomized controlled trial. BMC Pediatr. 2018, 18, 53. [Google Scholar] [CrossRef] [PubMed]
- Uhl, O.; Hellmuth, C.; Demmelmair, H.; Zhou, S.J.; Makrides, M.; Prosser, C.; Lowry, C.; Gibson, R.A.; Koletzko, B. Dietary effects on plasma glycerophospholipids. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Gardner, A.S.; Rahman, I.A.; Lai, C.T.; Hepworth, A.; Trengove, N.; Hartmann, P.E.; Geddes, D.T. Changes in fatty acid composition of human milk in response to cold-like symptoms in the lactating mother and infant. Nutrients 2017, 9, 1034. [Google Scholar] [CrossRef] [PubMed]
- Novak, E.M.; Innis, S.M. Impact of maternal dietary n-3 and n-6 fatty acids on milk medium-chain fatty acids and the implications for neonatal liver metabolism. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E807–E817. [Google Scholar] [CrossRef]
- Demmelmair, H.; Uhl, O.; Zhou, S.J.; Makrides, M.; Gibson, R.; Prosser, C.; Koletzko, B. Plasma sphingomyelins of infants differ according to source of milk fat. In Abstracts of the 51th Annual Meeting of the European Society for Paediatric Gastroenterology, Hepatology and Nutrition; Geneva. 2018. J. Pediatr. Gastroenterol. Nutr. 2018, 66, 941. [Google Scholar]
- Li, F.; Wu, S.S.; Berseth, C.L.; Harris, C.L.; Richards, J.D.; Wampler, J.L.; Zhuang, W.; Cleghorn, G.; Rudolph, C.D.; Liu, B.; et al. Improved neurodevelopmental outcomes associated with bovine milk fat globule membrane and lactoferrin in infant formula: A randomized, controlled trial. J. Pediatr. 2019, 215, 24–31. [Google Scholar] [CrossRef]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Imran, A.; Qasim, M.; Zafar, S.; Kamran, S.K.S.; Razzaq, A.; Aziz, N.; et al. Role of cholesterol and sphingolipids in brain development and neurological diseases. Lipids Health Dis. 2019, 18, 26. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Xu, J.; Xue, C. Sphingolipids in food and their critical roles in human health. Crit. Rev. Food Sci. Nutr. 2020. [Google Scholar] [CrossRef]
- Koletzko, B. Human milk lipids. Ann. Nutr. Metab. 2016, 69, 28–40. [Google Scholar] [CrossRef]
- Gurnida, D.A.; Rowan, A.M.; Idjradinata, P.; Muchtadi, D.; Sekarwana, N. Association of complex lipids containing gangliosides with cognitive development of 6-month-old infants. Early Hum. Dev. 2012, 88, 595–601. [Google Scholar] [CrossRef]
- Lee, H.; Zavaleta, N.; Chen, S.Y.; Lönnerdal, B.; Slupsky, C. Effect of bovine milk fat globule membranes as a complementary food on the serum metabolome and immune markers of 6-11 month-old infants. NPJ Sci. Food 2018, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Grip, T.; Dyrlund, T.S.; Ahonen, L.; Domellöf, M.; Hernell, O.; Hyöyläinen, T.; Knip, M.; Lönnerdal, B.; Orešič, M.; Timby, N. Serum, plasma and erythrocyte membrane lipidomes in infants fed formula supplemented with bovine milk fat globule membranes. Pediatr. Res. 2018, 84, 726–732. [Google Scholar] [CrossRef] [PubMed]
- Rueda, R.; Sabatel, J.L.; Maldonado, J.; Molina-Font, J.A.; Gil, A. Addition of gangliosides to an adapted milk formula modifies levels of fecal Escherichia coli in preterm newborn infants. J. Pediatr. 1998, 133, 90–94. [Google Scholar] [CrossRef]
- Zavaleta, N.; Kvistgaard, A.S.; Graverholt, G.; Respicio, G.; Guija, H.; Valencia, N.; Lönnerdal, B. Efficacy of an MFGM-enriched complementary food in diarrhea, anemia, and micronutrient status in infants. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 561–568. [Google Scholar] [CrossRef]
- Bunyavanich, S.; Berin, M.C. Food allergy and the microbiome: Current understandings and future directions. J. Allergy Clin. Immun. 2019, 144, 1468–1477. [Google Scholar] [CrossRef] [PubMed]
- Pascal, M.; Perez-Gordo, M.; Caballero, T.; Escribese, M.M.; Longo, M.N.L.; Luengo, O.; Manso, L.; Matheu, V.; Seoane, E.; Zamorano, M.; et al. Microbiome and allergic diseases. Front. Immunol. 2018, 9, 1584. [Google Scholar] [CrossRef]
- Tannock, G.W.; Lawley, B.; Munro, K.; Pathmanathan, S.G.; Zhou, S.J.; Makrides, M.; Gibson, R.A.; Sullivan, T.; Prosser, C.G.; Lowry, D.; et al. Comparison of the compositions of the stool microbiotas of infants fed goat milk formula, cow milk-based formula, or breast milk. Appl. Environ. Microbiol. 2013, 79, 3040–3048. [Google Scholar] [CrossRef]
- Infante, D.P.; Tormo, R.C.; Conde, M.Z. Use of goat’s milk in patients with cow’s milk allergy. An. Pediatr. 2003, 59, 138–142. [Google Scholar] [CrossRef]
- Lara-Villoslada, F.; Olivares, M.; Jimenez, J.; Boza, J.; Xaus, J. Goat milk is less immunogenic than cow milk in a murine model of atopy. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 354–360. [Google Scholar] [CrossRef]
- Kao, H.F.; Wang, Y.C.; Tseng, H.Y.; Wu, L.S.H.; Tsai, H.J.; Hsieh, M.H.; Chen, P.C.; Kuo, W.S.; Liu, L.F.; Liu, Z.G.; et al. Goat milk consumption enhances innate and adaptive immunities and alleviates allergen-induced airway inflammation in offspring mice. Front. Immunol. 2020, 11, 184. [Google Scholar] [CrossRef]
- Bellioni-Businco, B.; Paganelli, R.; Lucenti, P.; Giampietro, P.G.; Perborn, H.; Businco, L. Allergenicity of goat’s milk in children with cow’s milk allergy. J. Allergy Clin. Immun. 1999, 103, 1191–1194. [Google Scholar] [CrossRef]
- Mazzocchi, A.; D’Oria, V.; De Cosmi, V.; Bettocchi, S.; Milani, G.P.; Silano, M.; Agostoni, C. The role of lipids in human milk and infant formulae. Nutrients 2018, 10, 567. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Meyer, R.W.; Nwaru, B.I.; Roduit, C.; Untersmayr, E.; Adel-Patient, K.; Agache, I.; Agostoni, C.; Akdis, C.A.; Bischoff, S.C.; et al. EAACI paper: Influence of dietary fatty acids on asthma, food allergy, and atopic dermatitis. Allergy 2019, 74, 1429–1444. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y.M.; Berdyshev, E.; Goleva, E. Cutaneous barrier dysfunction in allergic diseases. J. Allergy Clin. Immun. 2020, 145, 1485–1497. [Google Scholar] [CrossRef] [PubMed]
- Haruta, Y.; Kato, K.; Yshioka, T. Dietary phospholipid concentrate from bovine milk improves epidermal function in hairless mice. Biosci. Biotechnol. Biochem. 2014, 72, 2151–2157. [Google Scholar] [CrossRef][Green Version]
- Higurashi, S.; Haruta-Ono, Y.; Urazono, H.; Kobayashi, T.; Kadooka, Y. Improvement of skin condition by oral supplementation with sphingomyelin-containing milk phospholipids in a double-blind, placebo-controlled, randomized trial. J. Dairy Sci. 2015, 98, 6706–6712. [Google Scholar] [CrossRef]
- Morifuji, M.; Oba, C.; Ichikawa, S.; Ito, K.; Kawahata, K.; Asami, Y.; Ikegami, S.; Itoh, H.; Sugawara, T. A novel mechanism for improvement of dry skin by dietary milk phospholipids: Effect on epidermal covalently bound ceramides and skin inflammation in hairless mice. J. Dermatol. Sci. 2014, 78, 224–231. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Liu, L.; Zhang, H.; Zhang, Y.; Chang, Y.H.; Zhu, Q.P. Comparative lipidomics analysis of human, bovine and caprine milk by UHPLC-Q-TOF-MS. Food Chem. 2020, 310, 125865. [Google Scholar] [CrossRef]
| Human Milk 1 Europe | Human Milk 1 Asia | Cow Milk 2 | Goat Milk 2 | Whole Goat Milk-Based IF 3 48% MF | Whole Goat Milk-Based IF 4 55% MF | Cow Milk-Based IF Vegetable Oil Only 6 | Cow Milk-Based IF MF 1 | |
|---|---|---|---|---|---|---|---|---|
| Butyric acid C4:0 | ND | ND | 3.2–3.3 | 2.0–2.6 | 1.17 | 3.1 | ND | 2.4 |
| Caproic acid C6:0 | 0.39 | 0.07 | 1.6–2.1 | 2.4–2.9 | 1.06 | 2.5 | ND/0.2 | 1.3 |
| Caprylic acid C8:0 | 0.19 (0.09–0.24) | 0.17 (0.11–0.28) | 1.2–1.3 | 2.7–2.7 | 1.11 | 2.0 | 1.2/2.5 | 1.7 |
| Capric acid C10:0 | 1.29 (0.83–1.63) | 1.31 (0.52–2.48) | 3.0–3.1 | 8.4–9.7 | 3.43 | 7.3 | 1.1/1.8 | 2.2 |
| Lauric acid C12:0 | 5.98 (4.15–8.33) | 5.56 (2.97–13.82) | 3.1–3.3 | 3.3–4.3 | 1.54 | 4.2 | 5.4/13.4 | 6.3 |
| Myristic acid C14:0 | 6.44 (4.98–9.38) | 5.70 (3.50–12.12) | 9.5–12.1 | 9.6–10.3 | 3.68 | 7.0 | 4.6/5.2 | 7.2 |
| Myristoleic acid C14:1 | 0.18 | 0.26 (0.03–1.11) | 0.7–1.1 | 0.09–0.16 | 0.12 | ND | ND/ND | 0.8 |
| Pentadecanoic acid C15:0 | 0.25 (0.16–0.32) | 0.20 (0.08–0.50) | ND | ND | 0.35 | 0.6 | ND/ND | 0.6 |
| Palmitic acid C16:0 | 21.93 (15.43–25.62) | 21.78 (17.55–29.00) | 26.5–32.2 | 24.6–27.7 | 12.30 | 17.0 | 26.3/7.7 | 18.9 |
| Palmitoleic acid C16:1 n-7 | 1.98 (1.65–2.31) | 2.44 (1.29–4.59) | ND | ND | 0.39 | ND | 0.6/0.1 | 1.1 |
| Heptadecanoic acid C17:0 | 0.29 (0.22–0.33) | 0.28 (0.19–0.41) | ND | ND | 0.29 | 0.4 | ND/ND | 0.3 |
| Stearic acid C18:0 | 7.37 (5.58–9.52) | 5.58 (3.90–6.79) | 8.9–14.6 | 9.7–12.5 | 5.89 | 6.3 | 5.3/3.2 | 6.7 |
| Oleic acid C18:1 n-9 | 36.30 (28.93–41.69) | 30.80 (21.85–36.96) | 19.3–24.1 | 19.4–24.0 | 40.65 | 31.0 | 37.6/43.3 | 28.1 |
| Linoleic acid C18:2 n-6 | 13.99 (10.16–16.59) | 16.90 (7.53–24.29) | ND | ND | 10.79 | 14.0 | 14.0/20.5 | 16.7 |
| Conjugated linoleic acid C18:2 c9, t11 | 0.27–0.49 5 | ND | 0.1–1.9 | 0.4–3.7 | 0.33 | ND | ND/ND | ND |
| α-linolenic acid C18:3 n-3 | 0.76 (0.49–1.05) | 1.47 (0.35–4.06) | ND | ND | 1.58 | 1.2 | 1.6/1.8 | 1.5 |
| Arachidic acid C20:0 | 0.21 (0.14–0.31) | 0.32 (0.03–2.97) | ND | ND | 0.24 | 0.3 | ND/0.3 | 0.3 |
| Arachidonic acid C20:4 n-6 | 0.47 (0.37–0.64) | 0.64 (0.30–2.57) | ND | ND | 0.45 | ND | 0.3/0.3 | ND |
| Eicosapentaenoic acid C20:5 n-3 | 0.09 (0.05–0.13) | 0.31 (0.07–1.59) | ND | ND | 0.12 | ND | ND/0.0 | ND |
| Behenic acid C22:0 | 0.09 (0.05–0.13) | 0.08 (0.05–0.14) | ND | ND | 0.33 | ND | ND/0.4 | 0.1 |
| Docosahexaenoic acid C22:6 n-3 | 0.28 (0.18–0.42) | 0.55 (0.19–1.13) | ND | ND | 0.44 | ND | 0.2/0.2 | ND |
| Tetracosanoic acid C24:0 | 0.07 (0.03–0.16) | 0.07 (0.01–0.14) | ND | ND | 0.21 | ND | ND/0.1 | ND |
| Human Milk 1,2 | Cow Milk 2,3 | Goat Milk 3 | Whole Goat Milk-Based IF 4 48% MF | Whole Goat Milk-Based IF 3 55% MF | Cow Milk-Based IF Vegetable Oils Only 2 | |
|---|---|---|---|---|---|---|
| % sn-2 palmitic acid | 51–88 | 37–45 | 35 | 29.5 | 31 | 10–20/39–47 5 |
| Human Milk 1 | Cow Milk 2 | Goat Milk 3 | Whole Moat Milk-Based IF 4 | Cow Milk-Based IF 5 | |
|---|---|---|---|---|---|
| Cholesterol (mg/L) | 90–200 | 100–300 | 100–200 | 58 | 14.6–51 |
| Human Milk 1 | Cow Milk 3 | Goat Milk 3 | Whole Moat Milk-Based IF 4 | |
|---|---|---|---|---|
| Total PL (mg/L) | 286.6 (98–474 2) | 294–400 5 | 276 5 | 169.2 |
| PL (mg/L) | ||||
| PI | 2.2–21 | 0.5–26.7 6 | 21.1 4 | 17.5 |
| PC | 32–124 | 18.2–128.0 6 | 93.7 4 | 51.6 |
| PS | 11–45 | 2.5–56.5 6 | 38.7 4 | 18.3 |
| PE | 26–103 | 19.2–143.0 6 | 91.6 4 | 40.2 |
| SM | 25–177 | 11.9–98.9 6 | 91.2 4,7 | 38.7 7 |
| % of total phospholipids | ||||
| PI | 1.1–10 | 0.1–9.0 | 2.21–9.4 | 11 |
| PC | 19–38 | 25.9–33.2 | 27.4–31.6 | 31 |
| PS | 3.7–17 | 0.12–9.1 | 2.41–14.0 | 11 |
| PE | 5.9–36 | 23.4–46.7 | 26.9–46.1 | 24 |
| SM | 29–43 | 19.8–25.4 | 16.1–27.3 | 23 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallier, S.; Tolenaars, L.; Prosser, C. Whole Goat Milk as a Source of Fat and Milk Fat Globule Membrane in Infant Formula. Nutrients 2020, 12, 3486. https://doi.org/10.3390/nu12113486
Gallier S, Tolenaars L, Prosser C. Whole Goat Milk as a Source of Fat and Milk Fat Globule Membrane in Infant Formula. Nutrients. 2020; 12(11):3486. https://doi.org/10.3390/nu12113486
Chicago/Turabian StyleGallier, Sophie, Louise Tolenaars, and Colin Prosser. 2020. "Whole Goat Milk as a Source of Fat and Milk Fat Globule Membrane in Infant Formula" Nutrients 12, no. 11: 3486. https://doi.org/10.3390/nu12113486
APA StyleGallier, S., Tolenaars, L., & Prosser, C. (2020). Whole Goat Milk as a Source of Fat and Milk Fat Globule Membrane in Infant Formula. Nutrients, 12(11), 3486. https://doi.org/10.3390/nu12113486





