Relationship between Dispositional Mindfulness, Psychological Health, and Diet Quality among Healthy Midlife Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Assessments
2.2.1. Dispositional Mindfulness
2.2.2. Depressive Symptoms
2.2.3. Perceived Stress
2.2.4. Positive and Negative Affect
2.2.5. Dietary Intake and Quality
2.3. Statistical Approach
Exploratory Analyses
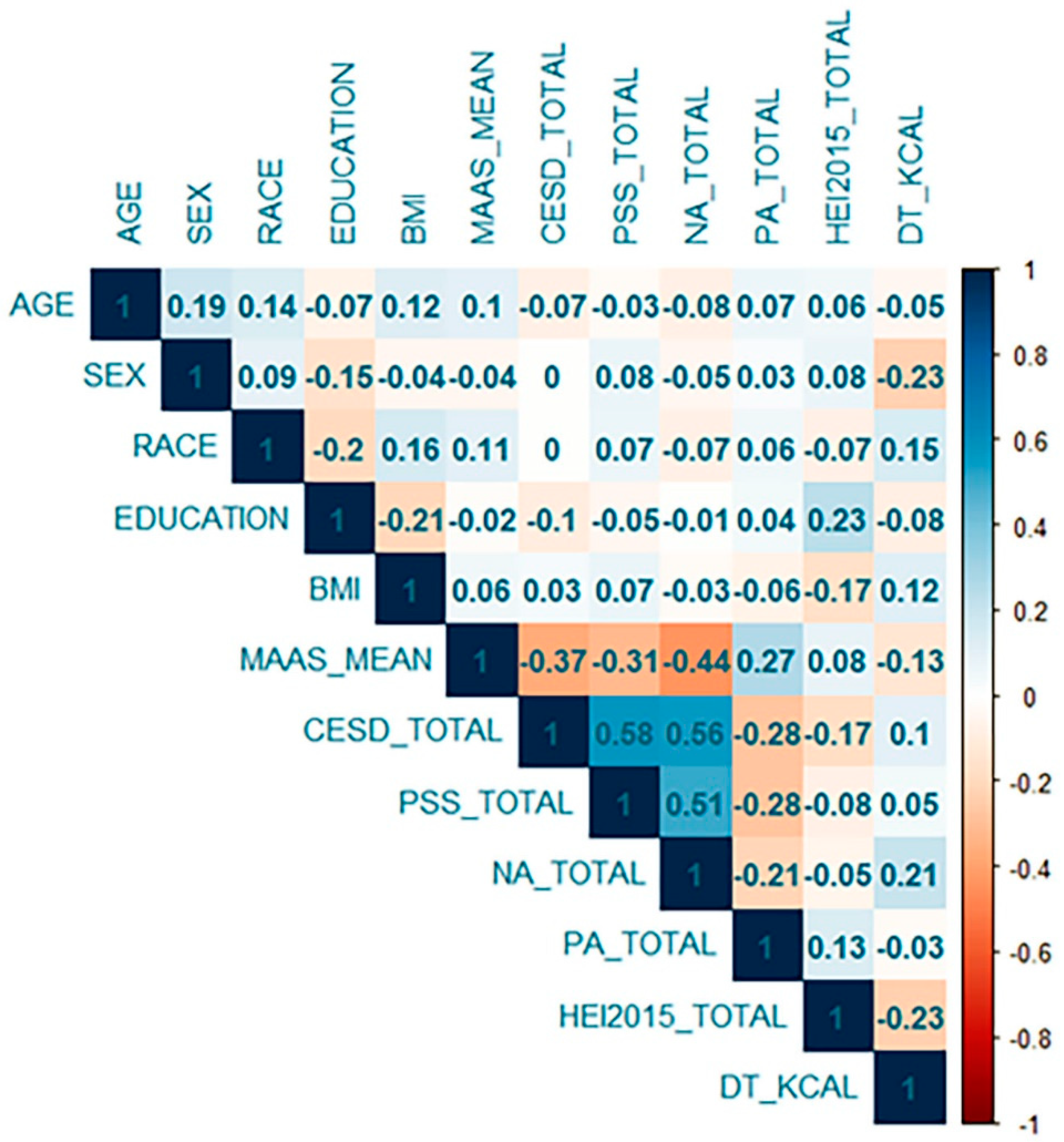
3. Results
3.1. Sample Characteristics
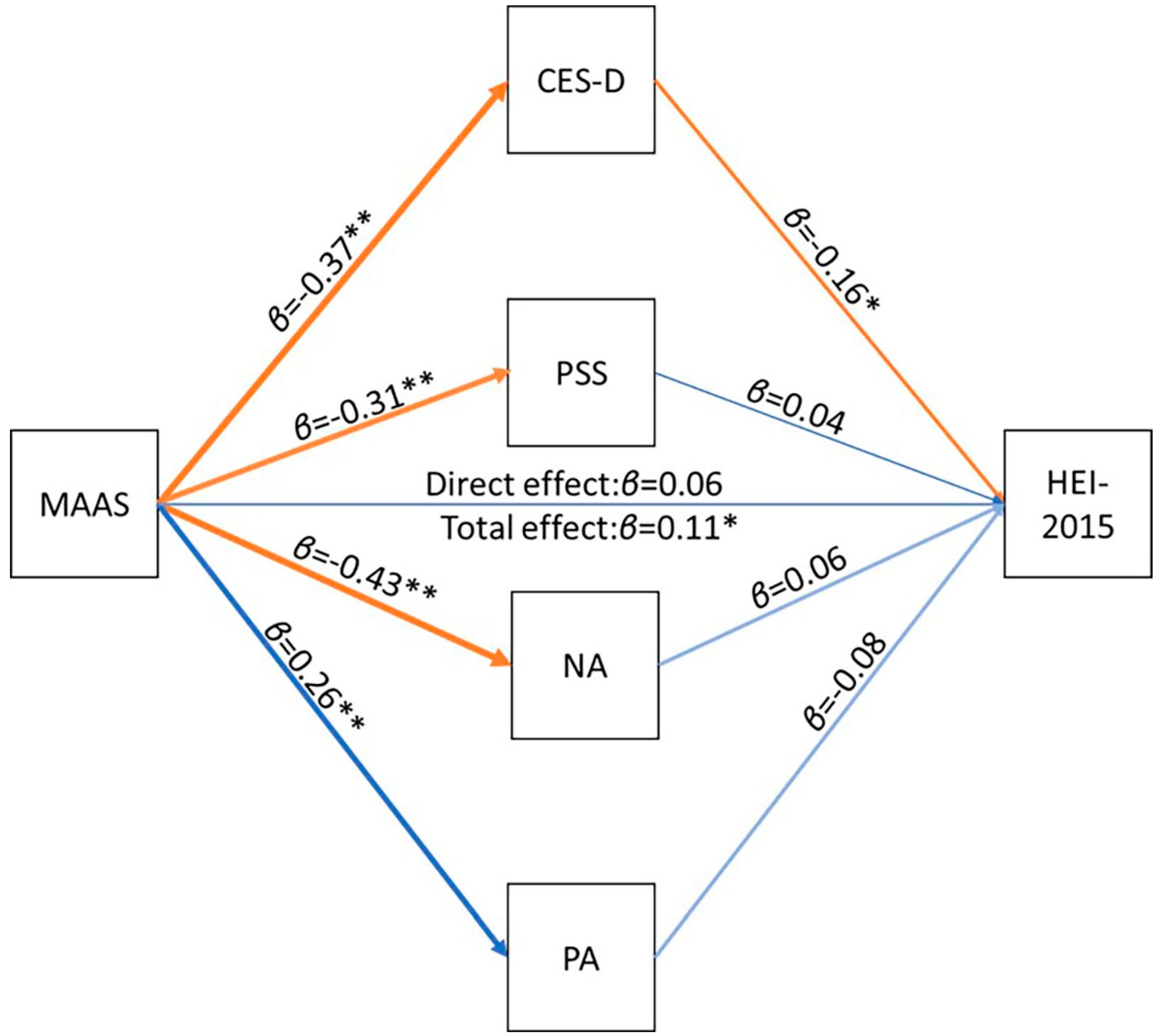
3.2. Total and Mediating Effects of Dispositional Mindfulness on Dietary Quality
4. Discussion
Limitations and Future Directions for Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mozaffarian, D.; Wilson, P.W.F.; Kannel, W.B. Beyond Established and Novel Risk Factors. Circulation 2008, 117, 3031–3038. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B.; Manson, J.E.; Stampfer, M.J.; Colditz, G.; Liu, S.; Solomon, C.G.; Willett, W.C. Diet, Lifestyle, and the Risk of Type 2 Diabetes Mellitus in Women. N. Engl. J. Med. 2001, 345, 790–797. [Google Scholar] [CrossRef] [PubMed]
- Lourida, I.; Hannon, E.; Littlejohns, T.J.; Langa, K.M.; Hyppönen, E.; Kuzma, E.; Llewellyn, D.J. Association of Lifestyle and Genetic Risk with Incidence of Dementia. JAMA 2019, 322, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Lourida, I.; Soni, M.; Thompson-Coon, J.; Purandare, N.; Lang, I.A.; Ukoumunne, O.C.; Llewellyn, D.J. Mediterranean Diet, Cognitive Function, and Dementia. Epidemiology 2013, 24, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Kohler, L.N.; Garcia, D.O.; Harris, R.B.; Oren, E.; Roe, D.J.; Jacobs, E.T. Adherence to Diet and Physical Activity Cancer Prevention Guidelines and Cancer Outcomes: A Systematic Review. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Cudhea, F.; Shan, Z.; Michaud, D.S.; Imamura, F.; Eom, H.; Ruan, M.; Rehm, C.D.; Liu, J.; Du, M.; et al. Preventable Cancer Burden Associated With Poor Diet in the United States. JNCI Cancer Spectr. 2019, 3, pkz034. [Google Scholar] [CrossRef] [PubMed]
- Quirk, S.E.; Williams, L.J.; O’Neil, A.; A Pasco, J.; Jacka, F.N.; Housden, S.; Berk, M.; Brennan, S.L. The association between diet quality, dietary patterns and depression in adults: A systematic review. BMC Psychiatry 2013, 13, 175. [Google Scholar] [CrossRef]
- Jardim, T.V.; Mozaffarian, D.; Abrahams-Gessel, S.; Sy, S.; Lee, Y.; Liu, J.; Huang, Y.; Rehm, C.; Wilde, P.; Micha, R.; et al. Cardiometabolic disease costs associated with suboptimal diet in the United States: A cost analysis based on a microsimulation model. PLoS Med. 2019, 16, e1002981. [Google Scholar] [CrossRef]
- United States Department of Agriculture. HEI Scores for Americans. Published 31 January 2019. Available online: https://www.fns.usda.gov/hei-scores-americans (accessed on 17 March 2020).
- Burke, L.E.; Dunbar-Jacob, J.; Orchard, T.J.; Sereika, S.M. Improving adherence to a cholesterol-lowering diet: A behavioral intervention study. Patient Educ. Couns. 2005, 57, 134–142. [Google Scholar] [CrossRef]
- Gay, H.C.; Rao, S.; Vaccarino, V.; Ali, M. Effects of Different Dietary Interventions on Blood Pressure. J. Hypertens. 2016, 67, 733–739. [Google Scholar] [CrossRef]
- Johns, D.J.; Hartmann-Boyce, J.; Jebb, S.A.; Aveyard, P. Diet or Exercise Interventions vs Combined Behavioral Weight Management Programs: A Systematic Review and Meta-Analysis of Direct Comparisons. J. Acad. Nutr. Diet. 2014, 114, 1557–1568. [Google Scholar] [CrossRef]
- Abbate, M.; Alfaro, L.G.; Bibiloni, M.D.M.; Tur, J.A. Efficacy of dietary intervention or in combination with exercise on primary prevention of cardiovascular disease: A systematic review. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1080–1093. [Google Scholar] [CrossRef]
- Mozaffarian, D. Dietary and Policy Priorities for Cardiovascular Disease, Diabetes, and Obesity. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef]
- Dohle, S.; Diel, K.; Hofmann, W. Executive functions and the self-regulation of eating behavior: A review. Appetite 2018, 124, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Wing, R.R.; Phelan, S. Long-term weight loss maintenance. Am. J. Clin. Nutr. 2005, 82, 222S–225S. [Google Scholar] [CrossRef]
- Niemeier, H.M.; Phelan, S.; Fava, J.L.; Wing, R.R. Internal Disinhibition Predicts Weight Regain Following Weight Loss and Weight Loss Maintenance. Obesity 2007, 15, 2485–2494. [Google Scholar] [CrossRef]
- Jansen, A.; Vanreyten, A.; Van Balveren, T.; Roefs, A.; Nederkoorn, C.; Havermans, R. Negative affect and cue-induced overeating in non-eating disordered obesity. Appetite 2008, 51, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Donofry, S.D.; Van Zoest, W.; Moonen, A.; Sacchetti, S.; Nederkoorn, C.; Roefs, A. Effect of dietary restraint and mood state on attentional processing of food cues. J. Behav. Ther. Exp. Psychiatry 2019, 62, 117–124. [Google Scholar] [CrossRef]
- Rudenga, K.J.; Sinha, R.; Small, D.M. Acute stress potentiates brain response to milkshake as a function of body weight and chronic stress. Int. J. Obes. 2012, 37, 309–316. [Google Scholar] [CrossRef]
- Meule, A.; Vögele, C.; Kübler, A. Restrained eating is related to accelerated reaction to high caloric foods and cardiac autonomic dysregulation. Appetite 2012, 58, 638–644. [Google Scholar] [CrossRef]
- Schepers, R.; Markus, C.R. Gene by cognition interaction on stress-induced attention bias for food: Effects of 5-HTTLPR and ruminative thinking. Biol. Psychol. 2017, 128, 21–28. [Google Scholar] [CrossRef]
- Cardi, V.; Leppanen, J.; Treasure, J. The effects of negative and positive mood induction on eating behaviour: A meta-analysis of laboratory studies in the healthy population and eating and weight disorders. Neurosci. Biobehav. Rev. 2015, 57, 299–309. [Google Scholar] [CrossRef]
- Epel, E.; Lapidus, R.; McEwen, B.; Brownell, K. Stress may add bite to appetite in women: A laboratory study of stress-induced cortisol and eating behavior. Psychoneuroendocrinology 2001, 26, 37–49. [Google Scholar] [CrossRef]
- Rutters, F.; Nieuwenhuizen, A.G.; Lemmens, S.G.T.; Born, J.M.; Westerterp-Plantenga, M.S. Acute Stress-related Changes in Eating in the Absence of Hunger. Obesity 2009, 17, 72–77. [Google Scholar] [CrossRef]
- Werthmann, J.; Field, M.; Roefs, A.; Nederkoorn, C.; Jansen, A. Attention bias for chocolate increases chocolate consumption–An attention bias modification study. J. Behav. Ther. Exp. Psychiatry 2014, 45, 136–143. [Google Scholar] [CrossRef]
- Zellner, D.A.; Loaiza, S.; Gonzalez, Z.; Pita, J.; Morales, J.; Pecora, D.; Wolf, A. Food selection changes under stress. Physiol. Behav. 2006, 87, 789–793. [Google Scholar] [CrossRef]
- Yokum, S.; Ng, J.; Stice, E. Attentional Bias to Food Images Associated with Elevated Weight and Future Weight Gain: An fMRI Study. Obesity 2011, 19, 1775–1783. [Google Scholar] [CrossRef]
- Mikolajczyk, R.; El Ansari, W.; Maxwell, A.E. Food consumption frequency and perceived stress and depressive symptoms among students in three European countries. Nutr. J. 2009, 8, 31. [Google Scholar] [CrossRef]
- Barrington, W.E.; Beresford, S.A.; McGregor, B.A.; White, E. Perceived Stress and Eating Behaviors by Sex, Obesity Status, and Stress Vulnerability: Findings from the Vitamins and Lifestyle (VITAL) Study. J. Acad. Nutr. Diet. 2014, 114, 1791–1799. [Google Scholar] [CrossRef]
- Short, M.M.; Mazmanian, D.; Oinonen, K.; Mushquash, C. Executive function and self-regulation mediate dispositional mindfulness and well-being. Pers. Individ. Differ. 2016, 93, 97–103. [Google Scholar] [CrossRef]
- Ludwig, D.S. Mindfulness in Medicine. JAMA 2008, 300, 1350–1352. [Google Scholar] [CrossRef]
- Keng, S.-L.; Smoski, M.J.; Robins, C.J. Effects of mindfulness on psychological health: A review of empirical studies. Clin. Psychol. Rev. 2011, 31, 1041–1056. [Google Scholar] [CrossRef] [PubMed]
- Loucks, E.B.; Schuman-Olivier, Z.; Britton, W.B.; Fresco, D.M.; Desbordes, G.; Brewer, J.A.; Fulwiler, C.E. Mindfulness and Cardiovascular Disease Risk: State of the Evidence, Plausible Mechanisms, and Theoretical Framework. Curr. Cardiol. Rep. 2015, 17, 1–11. [Google Scholar] [CrossRef]
- Rogers, J.M.; Ferrari, M.; Mosely, K.; Lang, C.P.; Brennan, L. Mindfulness-based interventions for adults who are overweight or obese: A meta-analysis of physical and psychological health outcomes. Obes. Rev. 2016, 18, 51–67. [Google Scholar] [CrossRef]
- Tomlinson, E.R.; Yousaf, O.; Vittersø, A.D.; Jones, L. Dispositional Mindfulness and Psychological Health: A Systematic Review. Mindfulness 2018, 9, 23–43. [Google Scholar] [CrossRef]
- Loucks, E.B.; Britton, W.B.; Howe, C.J.; Eaton, C.B.; Buka, S.L. Positive Associations of Dispositional Mindfulness with Cardiovascular Health: The New England Family Study. Int. J. Behav. Med. 2015, 22, 540–550. [Google Scholar] [CrossRef]
- Tomfohr, L.M.; Pung, M.A.; Mills, P.J.; Edwards, K. Trait mindfulness is associated with blood pressure and interleukin-6: Exploring interactions among subscales of the Five Facet Mindfulness Questionnaire to better understand relationships between mindfulness and health. J. Behav. Med. 2014, 38, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Fisher, N.R.; Mead, B.; Lattimore, P.; Emalinowski, P. Dispositional mindfulness and reward motivated eating: The role of emotion regulation and mental habit. Appetite 2017, 118, 41–48. [Google Scholar] [CrossRef]
- Tak, S.R.; Hendrieckx, C.; Nefs, G.; Nyklíček, I.; Speight, J.; Pouwer, F. The association between types of eating behaviour and dispositional mindfulness in adults with diabetes. Results from Diabetes MILES. The Netherlands. Appetite 2015, 87, 288–295. [Google Scholar] [CrossRef]
- Godfrey, K.M.; Gallo, L.C.; Afari, N. Mindfulness-based interventions for binge eating: A systematic review and meta-analysis. J. Behav. Med. 2014, 38, 348–362. [Google Scholar] [CrossRef]
- Fisher, N.; Lattimore, P.; Emalinowski, P. Attention with a mindful attitude attenuates subjective appetitive reactions and food intake following food-cue exposure. Appetite 2016, 99, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Forman, E.M.; Butryn, M.L. A new look at the science of weight control: How acceptance and commitment strategies can address the challenge of self-regulation. Appetite 2015, 84, 171–180. [Google Scholar] [CrossRef]
- Forman, E.M.; Butryn, M.L.; Manasse, S.M.; Crosby, R.D.; Goldstein, S.P.; Wyckoff, E.P.; Thomas, J.G. Acceptance-based versus standard behavioral treatment for obesity: Results from the mind your health randomized controlled trial. Obesity 2016, 24, 2050–2056. [Google Scholar] [CrossRef]
- Forman, E.M.; Manasse, S.M.; Butryn, M.L.; Crosby, R.D.; Dallal, D.H.; Crochiere, R.J. Long-Term Follow-up of the Mind Your Health Project: Acceptance-Based versus Standard Behavioral Treatment for Obesity. Obesity 2019, 27, 565–571. [Google Scholar] [CrossRef]
- Kristeller, J.L.; Wolever, R.Q. Mindfulness-Based Eating Awareness Training for Treating Binge Eating Disorder: The Conceptual Foundation. Eat. Disord. 2010, 19, 49–61. [Google Scholar] [CrossRef]
- Kristeller, J.; Wolever, R.Q.; Sheets, V. Mindfulness-Based Eating Awareness Training (MB-EAT) for Binge Eating: A Randomized Clinical Trial. Mindfulness 2013, 5, 282–297. [Google Scholar] [CrossRef]
- Wnuk, S.; Du, C.T.; Van Exan, J.; Wallwork, A.; Warwick, K.; Tremblay, L.; Kowgier, M.; Sockalingam, S. Mindfulness-Based Eating and Awareness Training for Post-Bariatric Surgery Patients: A Feasibility Pilot Study. Mindfulness 2017, 9, 949–960. [Google Scholar] [CrossRef]
- Beshara, M.; Hutchinson, A.D.; Wilson, C. Does mindfulness matter? Everyday mindfulness, mindful eating and self-reported serving size of energy dense foods among a sample of South Australian adults. Appetite 2013, 67, 25–29. [Google Scholar] [CrossRef]
- Fanning, J.; Osborn, C.Y.; Lagotte, A.E.; Mayberry, L.S. Relationships between dispositional mindfulness, health behaviors, and hemoglobin A1c among adults with type 2 diabetes. J. Behav. Med. 2018, 41, 798–805. [Google Scholar] [CrossRef]
- Gilbert, D.; Waltz, J. Mindfulness and Health Behaviors. Mindfulness 2010, 1, 227–234. [Google Scholar] [CrossRef]
- Salmoirago-Blotcher, E.; Hunsinger, M.; Morgan, L.; Fischer, D.; Carmody, J. Mindfulness-Based Stress Reduction and Change in Health-Related Behaviors. J. Evid. Based Integr. Med. 2013, 18, 243–247. [Google Scholar] [CrossRef]
- Reedy, J.; Lerman, J.L.; Krebs-Smith, S.M.; Kirkpatrick, S.I.; Pannucci, T.E.; Wilson, M.M.; Subar, A.F.; Kahle, L.L.; Tooze, J.A. Evaluation of the Healthy Eating Index-2015. J. Acad. Nutr. Diet. 2018, 118, 1622–1633. [Google Scholar] [CrossRef]
- Brown, K.W.; Ryan, R.M. The benefits of being present: Mindfulness and its role in psychological well-being. J. Pers. Soc. Psychol. 2003, 84, 822–848. [Google Scholar] [CrossRef]
- MacKillop, J.; Anderson, E.J. Further Psychometric Validation of the Mindful Attention Awareness Scale (MAAS). J. Psychopathol. Behav. Assess. 2007, 29, 289–293. [Google Scholar] [CrossRef]
- Osman, A.; Lamis, D.A.; Bagge, C.L.; Freedenthal, S.; Barnes, S.M. The Mindful Attention Awareness Scale: Further Examination of Dimensionality, Reliability, and Concurrent Validity Estimates. J. Pers. Assess. 2015, 98, 189–199. [Google Scholar] [CrossRef]
- Van Dam, N.T.; Earleywine, M.; Borders, A. Measuring mindfulness? An Item Response Theory analysis of the Mindful Attention Awareness Scale. Pers. Individ. Differ. 2010, 49, 805–810. [Google Scholar] [CrossRef]
- Carlson, L.E.; Brown, K.W. Validation of the Mindful Attention Awareness Scale in a cancer population. J. Psychosom. Res. 2005, 58, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Cebolla, A.; Luciano, J.V.; DeMarzo, M.; Navarro-Gil, M.; Campayo, J.G. Psychometric properties of the Spanish version of the mindful attention awareness scale (MAAS) in patients with fibromyalgia. Heal. Qual. Life Outcomes 2013, 11, 6. [Google Scholar] [CrossRef]
- Kilpatrick, L.A.; Suyenobu, B.Y.; Smith, S.R.; Bueller, J.A.; Goodman, T.; Creswell, J.D.; Tillisch, K.; Mayer, E.A.; Naliboff, B.D. Impact of mindfulness-based stress reduction training on intrinsic brain connectivity. Neuro Image 2011, 56, 290–298. [Google Scholar] [CrossRef]
- Lyvers, M.; Makin, C.; Toms, E.; Thorberg, F.A.; Samios, C. Trait Mindfulness in Relation to Emotional Self-Regulation and Executive Function. Mindfulness 2013, 5, 619–625. [Google Scholar] [CrossRef]
- Lu, J.; Huffman, K. A Meta-Analysis of Correlations between Trait Mindfulness and Impulsivity: Implications for Counseling. Int. J. Adv. Couns. 2017, 39, 345–359. [Google Scholar] [CrossRef]
- Radloff, L.S. The CES-D Scale. Appl. Psychol. Meas. 1977, 1, 385–401. [Google Scholar] [CrossRef]
- Orme, J.G.; Reis, J.; Herz, E.J. Factorial and discriminant validity of the center for epidemiological studies depression (CES-D) scale. J. Clin. Psychol. 1986, 42, 28–33. [Google Scholar] [CrossRef]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A Global Measure of Perceived Stress. J. Heal. Soc. Behav. 1983, 24, 385. [Google Scholar] [CrossRef]
- Taylor, J.M. Psychometric analysis of the Ten-Item Perceived Stress Scale. Psychol. Assess. 2015, 27, 90–101. [Google Scholar] [CrossRef]
- Watson, D.; Clark, A.L. The PANAS-X: Manual for the Positive and Negative Affect Schedule-Expanded Form; University of Iowa: Iowa City, IA, USA, 1994. [Google Scholar] [CrossRef]
- Crawford, J.R.; Henry, J.D. The Positive and Negative Affect Schedule (PANAS): Construct validity, measurement properties and normative data in a large non-clinical sample. Br. J. Clin. Psychol. 2004, 43, 245–265. [Google Scholar] [CrossRef]
- Merz, E.L.; Malcarne, V.L.; Roesch, S.C.; Ko, C.M.; Emerson, M.; Roma, V.G.; Sadler, G.R. Psychometric properties of Positive and Negative Affect Schedule (PANAS) original and short forms in an African American community sample. J. Affect. Disord. 2013, 151, 942–949. [Google Scholar] [CrossRef]
- Block, G.; Woods, M.; Potosky, A.; Clifford, C. Validation of a self-administered diet history questionnaire using multiple diet records. J. Clin. Epidemiol. 1990, 43, 1327–1335. [Google Scholar] [CrossRef]
- Boucher, B.; Cotterchio, M.; Kreiger, N.; Nadalin, V.; Block, T.; Block, G. Validity and reliability of the Block98 food-frequency questionnaire in a sample of Canadian women. Public Heal. Nutr. 2006, 9, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Subar, A.F.; Thompson, F.E.; Kipnis, V.; Midthune, D.; Hurwitz, P.; McNutt, S.; McIntosh, A.; Rosenfeld, S. Comparative Validation of the Block, Willett, and National Cancer Institute Food Frequency Questionnaires. Am. J. Epidemiol. 2001, 154, 1089–1099. [Google Scholar] [CrossRef]
- Krebs-Smith, S.M.; Pannucci, T.E.; Subar, A.F.; Kirkpatrick, S.I.; Lerman, J.L.; Tooze, J.A.; Wilson, M.M.; Reedy, J. Update of the Healthy Eating Index: HEI-2015. J. Acad. Nutr. Diet. 2018, 118, 1591–1602. [Google Scholar] [CrossRef] [PubMed]
- Panizza, C.E.; Shvetsov, Y.B.; Harmon, B.E.; Wilkens, L.R.; Le Marchand, L.; Haiman, C.; Reedy, J.; Boushey, C.J. Testing the Predictive Validity of the Healthy Eating Index-2015 in the Multiethnic Cohort: Is the Score Associated with a Reduced Risk of All-Cause and Cause-Specific Mortality? Nutrition 2018, 10, 452. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Diet Quality as Assessed by the Healthy Eating Index, the Alternate Healthy Eating Index, the Dietary Approaches to Stop Hypertension Score, and Health Outcomes: A Systematic Review and Meta-Analysis of Cohort Studies. J. Acad. Nutr. Diet. 2015, 115, 780–800. [Google Scholar] [CrossRef]
- Rosseel, Y. lavaan: An R package for structural equation modeling and more Version 0.5-12 (BETA). J. Stat. Softw. 2012, 48, 37. [Google Scholar] [CrossRef]
- Dunn, B.D.; Stefanovitch, I.; Evans, D.; Oliver, C.; Hawkins, A.; Dalgleish, T. Can you feel the beat? Interoceptive awareness is an interactive function of anxiety- and depression-specific symptom dimensions. Behav. Res. Ther. 2010, 48, 1133–1138. [Google Scholar] [CrossRef]
- Furman, D.J.; Waugh, C.E.; Bhattacharjee, K.; Thompson, R.J.; Gotlib, I.H. Interoceptive awareness, positive affect, and decision making in major depressive disorder. J. Affect. Disord. 2013, 151, 780–785. [Google Scholar] [CrossRef]
- Critchley, H.D.; Garfinkel, S.N. Interoception and emotion. Curr. Opin. Psychol. 2017, 17, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Bornemann, B.; Herbert, B.M.; Mehling, W.E.; Singer, T. Differential changes in self-reported aspects of interoceptive awareness through 3 months of contemplative training. Front. Psychol. 2015, 5, 1504. [Google Scholar] [CrossRef]
- De Jong, M.; Lazar, S.W.; Hug, K.; Mehling, W.E.; Hölzel, B.K.; Sack, A.T.; Peeters, F.; Ashih, H.; Mischoulon, D.; Gard, T. Effects of Mindfulness-Based Cognitive Therapy on Body Awareness in Patients with Chronic Pain and Comorbid Depression. Front. Psychol. 2016, 7. [Google Scholar] [CrossRef]
- Hanley, A.W.; Mehling, W.E.; Garland, E.L. Holding the body in mind: Interoceptive awareness, dispositional mindfulness and psychological well-being. J. Psychosom. Res. 2017, 99, 13–20. [Google Scholar] [CrossRef]
- Price, C.J.; Thompson, E.A.; Crowell, S.E.; Pike, K.; Cheng, S.C.; Parent, S.; Hooven, C. Immediate effects of interoceptive awareness training through Mindful Awareness in Body-oriented Therapy (MABT) for women in substance use disorder treatment. Subst. Abus. 2018, 40, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Britton, W.B.; Shahar, B.; Szepsenwol, O.; Jacobs, W.J. Mindfulness-Based Cognitive Therapy Improves Emotional Reactivity to Social Stress: Results from a Randomized Controlled Trial. Behav. Ther. 2012, 43, 365–380. [Google Scholar] [CrossRef] [PubMed]
- Farb, N.A.S.; Anderson, A.K.; Mayberg, H.; Bean, J.; McKeon, D.; Segal, Z.V. Minding one’s emotions: Mindfulness training alters the neural expression of sadness. Emotion 2010, 10, 25–33. [Google Scholar] [CrossRef]
- Spijkerman, M.; Pots, W.; Bohlmeijer, E. Effectiveness of online mindfulness-based interventions in improving mental health: A review and meta-analysis of randomised controlled trials. Clin. Psychol. Rev. 2016, 45, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Strauss, C.; Cavanagh, K.; Oliver, A.; Pettman, D. Mindfulness-Based Interventions for People Diagnosed with a Current Episode of an Anxiety or Depressive Disorder: A Meta-Analysis of Randomised Controlled Trials. PLoS ONE 2014, 9, e96110. [Google Scholar] [CrossRef]
- Ford, P.A.; Jaceldo-Siegl, K.; Lee, J.W.; Youngberg, W.; Tonstad, S. Intake of Mediterranean foods associated with positive affect and low negative affect. J. Psychosom. Res. 2013, 74, 142–148. [Google Scholar] [CrossRef]
- Richardson, A.S.; Arsenault, J.E.; Cates, S.C.; Muth, M.K. Perceived stress, unhealthy eating behaviors, and severe obesity in low-income women. Nutr. J. 2015, 14, 1–10. [Google Scholar] [CrossRef]
- Bonaccio, M.; Bonanni, A.E.; Di Castelnuovo, A.; De Lucia, F.; Donati, M.B.; De Gaetano, G.; Iacoviello, L.; on behalf of the Moli-sani Project Investigators. Low income is associated with poor adherence to a Mediterranean diet and a higher prevalence of obesity: Cross-sectional results from the Moli-sani study. BMJ Open 2012, 2, e001685. [Google Scholar] [CrossRef]
- Klesges, R.C. Who Underreports Dietary Intake in a Dietary Recall? Evidence from the Second National Health and Nutrition Examination Survey. J. Consult. Clin. Psychol. 1995, 63, 438–444. [Google Scholar]
- Lallukka, T.; Laaksonen, M.; Rahkonen, O.; Roos, E.; Lahelma, E. Multiple socio-economic circumstances and healthy food habits. Eur. J. Clin. Nutr. 2006, 61, 701–710. [Google Scholar] [CrossRef]
- Wardle, J.; Haase, A.M.; Steptoe, A.; Nillapun, M.; Jonwutiwes, K.; Bellisie, F. Gender differences in food choice: The contribution of health beliefs and dieting. Ann. Behav. Med. 2004, 27, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.-S.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Heal. 2014, 36, e2014009. [Google Scholar] [CrossRef]
- Moynihan, J.A.; Chapman, B.P.; Klorman, R.; Krasner, M.S.; Duberstein, P.R.; Brown, K.W.; Talbot, N.L. Mindfulness-Based Stress Reduction for Older Adults: Effects on Executive Function, Frontal Alpha Asymmetry and Immune Function. Neuropsychobiology 2013, 68, 34–43. [Google Scholar] [CrossRef]
- Gallant, S.N. Mindfulness meditation practice and executive functioning: Breaking down the benefit. Conscious. Cogn. 2016, 40, 116–130. [Google Scholar] [CrossRef]
- Bergomi, C.; Tschacher, W.; Kupper, Z. The Assessment of Mindfulness with Self-Report Measures: Existing Scales and Open Issues. Mindfulness 2012, 4, 191–202. [Google Scholar] [CrossRef]
| Mean (SD) | |
|---|---|
| Age (years) | 43.19 (7.26) |
| BMI (kg/m2) | 27.08 (5.79) |
| MAAS average score | 4.28 (0.74) |
| PSS score | 15.89 (3.96) |
| CES-D score | 8.93 (8.25) |
| PANAS NA score | 15.57 (5.17) |
| PANAS PA score | 34.1 (6.69) |
| Total caloric intake (kilocalories) | 1901 (894.5) |
| HEI 2015 Total Score | 67.58 (10.59) |
| N (%) | |
| Sex (F) | 211 (51.97) |
| Education Level | |
| No High School diploma | 1 (0.20) |
| GED | 4 (0.98) |
| High School diploma | 23 (5.66) |
| Technical training | 19 (4.68) |
| Some college | 42 (10.34) |
| Associates degree | 39 (9.61) |
| Bachelor’s degree | 148 (36.45) |
| Master’s degree | 90 (22.17) |
| Doctoral degree | 40 (9.85) |
| Race | |
| White | 328 (80.79) |
| Black or African American | 72 (17.73) |
| Asian | 2 (0.49) |
| Multi-racial | 2 (0.49) |
| Other | 2 (0.49) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donofry, S.D.; Erickson, K.I.; Levine, M.D.; Gianaros, P.J.; Muldoon, M.F.; Manuck, S.B. Relationship between Dispositional Mindfulness, Psychological Health, and Diet Quality among Healthy Midlife Adults. Nutrients 2020, 12, 3414. https://doi.org/10.3390/nu12113414
Donofry SD, Erickson KI, Levine MD, Gianaros PJ, Muldoon MF, Manuck SB. Relationship between Dispositional Mindfulness, Psychological Health, and Diet Quality among Healthy Midlife Adults. Nutrients. 2020; 12(11):3414. https://doi.org/10.3390/nu12113414
Chicago/Turabian StyleDonofry, Shannon D., Kirk I. Erickson, Michele D. Levine, Peter J. Gianaros, Matthew F. Muldoon, and Stephen B. Manuck. 2020. "Relationship between Dispositional Mindfulness, Psychological Health, and Diet Quality among Healthy Midlife Adults" Nutrients 12, no. 11: 3414. https://doi.org/10.3390/nu12113414
APA StyleDonofry, S. D., Erickson, K. I., Levine, M. D., Gianaros, P. J., Muldoon, M. F., & Manuck, S. B. (2020). Relationship between Dispositional Mindfulness, Psychological Health, and Diet Quality among Healthy Midlife Adults. Nutrients, 12(11), 3414. https://doi.org/10.3390/nu12113414






