The Metabolic Effects of Cynara Supplementation in Overweight and Obese Class I Subjects with Newly Detected Impaired Fasting Glycemia: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
2.2. Dietary Supplement
2.3. Adverse Events
2.4. Glycemic and Lipidic Parameters
2.5. Anthropometric Mesaurements and Dietary Counseling
2.6. Body Composition
2.7. Primary and Secondary Endpoints
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Unwin, N.; Shaw, J.; Zimmet, P.; Alberti, K.G.M.M. Impaired glucose tolerance and impaired fasting glycaemia: The current status on definition and intervention. Diabet. Med. 2002, 19, 708–723. [Google Scholar] [PubMed]
- Glümer, C.; Jørgensen, T.; Borch-Johnsen, K. Prevalences of diabetes and impaired glucose regulation in a Danish population: The Inter99 study. Diabetes Care 2003, 26, 2335–2340. [Google Scholar] [CrossRef] [PubMed]
- Hui, H.; Tang, G.; Go, V.L.W. Hypoglycemic herbs and their action mechanisms. Chin. Med. 2009, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Neustadt, J.; Pieczenik, S.R. Medication-induced mitochondrial damage and disease. Mol. Nutr. Food Res. 2008, 52, 780–788. [Google Scholar] [CrossRef]
- Arion, W.J.; Canfield, W.K.; Ramos, F.C.; Su, M.L.; Burger, H.J.; Hemmerle, H.; Schubert, G.; Below, P.; Herling, A.W. Chlorogenic acid analogue S 3483: A potent competitive inhibitor of the hepatic and renal glucose-6-phosphatase systems. Arch. Biochem. Biophys. 1998, 351, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Matsui, T.; Ogunwande, I.; Abesundara, K.; Matsumoto, K. Anti-hyperglycemic Potential of Natural Products. Mini-Rev. Med. Chem. 2006, 6, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Fantini, N.; Colombo, G.; Giori, A.; Riva, A.; Morazzoni, P.; Bombardelli, E.; Carai, M.A.M. Evidence of glycemia-lowering effect by a Cynara scolymus L. extract in normal and obese rats. Phyther. Res. 2011, 25, 463–466. [Google Scholar] [CrossRef]
- Karthikesan, K.; Pari, L.; Menon, V.P. Combined treatment of tetrahydrocurcumin and chlorogenic acid exerts potential antihyperglycemic effect on streptozotocin-nicotinamide-induced diabetic rats. Gen. Physiol. Biophys. 2010, 29, 23–30. [Google Scholar] [CrossRef]
- Rondanelli, M.; Opizzi, A.; Faliva, M.; Sala, P.; Perna, S.; Riva, A.; Morazzoni, P.; Bombardelli, E.; Giacosa, A. Metabolic management in overweight subjects with naive impaired fasting glycaemia by means of a highly standardized extract from cynara scolymus: A double-blind, placebo-controlled, randomized clinical trial. Phyther. Res. 2014, 28, 33–41. [Google Scholar] [CrossRef]
- Kuczmannová, A.; Balažová, A.; Račanská, E.; Kameníková, M.; Fialová, S.; Majerník, J.; Nagy, M.; Gál, P.; Mučaji, P.; Atanasov, A.G.; et al. Agrimonia eupatoria L. and Cynara cardunculus L. Water Infusions: Comparison of Anti-Diabetic Activities. Molecules 2016, 21, 564. [Google Scholar]
- Ostan, R.; Béné, M.C.; Spazzafumo, L.; Pinto, A.; Donini, L.M.; Pryen, F.; Charrouf, Z.; Valentini, L.; Lochs, H.; Bourdel-Marchasson, I.; et al. Impact of diet and nutraceutical supplementation on inflammation in elderly people. Results from the RISTOMED study, an open-label randomized control trial. Clin. Nutr. 2016, 35, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Colantuono, A.; Ferracane, R.; Vitaglione, P. Potential bioaccessibility and functionality of polyphenols and cynaropicrin from breads enriched with artichoke stem. Food Chem. 2018, 245, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Mare, R.; Mazza, E.; Ferro, Y.; Gliozzi, M.; Nucera, S.; Paone, S.; Aversa, I.; Pujia, R.; Marafioti, G.; Musolino, V.; et al. A new breakfast brioche containing bergamot fiber prevents insulin and glucose increase in healthy volunteers: A pilot study. Minerva Endocrinol. 2020. [Google Scholar] [CrossRef]
- Sherwin, R.S.; Anderson, R.M.; Buse, J.B.; Chin, M.H.; Eddy, D.; Fradkin, J.; Ganiats, T.G.; Ginsberg, H.N.; Kahn, R.; Nwankwo, R.; et al. The prevention or delay of type 2 diabetes: American Diabetes Association and National Institute of Diabetes, Digestive and Kidney Diseases. Diabetes Care 2002, 25, 742. [Google Scholar]
- Nathan, D.M.; Kuenen, J.; Borg, R.; Zheng, H.; Schoenfeld, D.; Heine, R.J. Translating the A1C assay into estimated average glucose values. Diabetes Care 2008, 31, 1473–1478. [Google Scholar] [CrossRef]
- Haffner, S.M.; Kennedy, E.; Gonzalez, C.; Stern, M.P.; Miettinen, H. A prospective analysis of the HOMA model. The Mexico City Diabetes Study. Diabetes Care 1996, 19, 1138–1141. [Google Scholar] [CrossRef] [PubMed]
- Frisancho, A.R. New standards of weight and body composition by frame size and height for assessment of nutritional status of adults and the elderly. Am. J. Clin. Nutr. 1984, 40, 808–819. [Google Scholar] [CrossRef]
- Mohammad, A.; De Lucia Rolfe, E.; Sleigh, A.; Kivisild, T.; Behbehani, K.; Wareham, N.J.; Brage, S.; Mohammad, T. Validity of visceral adiposity estimates from DXA against MRI in Kuwaiti men and women. Nutr. Diabetes 2017, 7, e238. [Google Scholar] [CrossRef]
- Pinheiro, S.J.; Bates, D.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 2013, 3, 111. [Google Scholar]
- Benjamin, Y.; Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 2001, 29, 1165–1188. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. Linear and Nonlinear Mixed Effects Models. R Package Version 2007, 3, 1–89. [Google Scholar]
- Dragan, S.; Andrica, F.; Serban, M.-C.; Timar, R. Polyphenols-Rich Natural Products for Treatment of Diabetes. Curr. Med. Chem. 2014, 22, 14–22. [Google Scholar] [CrossRef]
- Ben Salem, M.; Affes, H.; Ksouda, K.; Dhouibi, R.; Sahnoun, Z.; Hammami, S.; Zeghal, K.M. Pharmacological Studies of Artichoke Leaf Extract and Their Health Benefits. Plant Foods Hum. Nutr. 2015, 70, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, M.; Ben Abdallah Kolsi, R.; Dhouibi, R.; Ksouda, K.; Charfi, S.; Yaich, M.; Hammami, S.; Sahnoun, Z.; Zeghal, K.M.; Jamoussi, K.; et al. Protective effects of Cynara scolymus leaves extract on metabolic disorders and oxidative stress in alloxan-diabetic rats. BMC Complement. Altern. Med. 2017, 17, 328. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.F.; Cazarolli, L.H.; Lavado, C.; Mengatto, V.; Figueiredo, M.S.R.B.; Guedes, A.; Pizzolatti, M.G.; Silva, F.R.M.B. Effects of flavonoids on α-glucosidase activity: Potential targets for glucose homeostasis. Nutrition 2011, 27, 1161–1167. [Google Scholar] [CrossRef]
- Cazarolli, L.; Zanatta, L.; Alberton, E.; Figueiredo, M.R.B.; Folador, P.; Damazio, R.; Pizzolatti, M.; Silva, F.M.B. Flavonoids: Cellular and Molecular Mechanism of Action in Glucose Homeostasis. Mini-Rev. Med. Chem. 2008, 8, 1032–1038. [Google Scholar] [CrossRef]
- Natella, F.; Scaccini, C. Role of coffee in modulation of diabetes risk. Nutr. Rev. 2012, 70, 207–217. [Google Scholar] [CrossRef]
- Abdullah, H.-M.; Gardner, M.J.; Kurukulasuriya, L.R.; Sowers, J.R. Dysglycemia/prediabetes and cardiovascular risk factors. Rev. Cardiovasc. Med. 2009, 10, 202–208. [Google Scholar]
- Magied, M.M.A.; Din Hussien, S.E.L.; Zaki, S.M.; Said, R.M. EL Artichoke (Cynara scolymus L.) Leaves and Heads Extracts as Hypoglycemic and Hypocholesterolemic in Rats. J. Food Nutr. Res. 2016, 4, 60–68. [Google Scholar]
- Rezazadeh, K.; Aliashrafi, S.; Asghari-Jafarabadi, M.; Ebrahimi-Mameghani, M. Antioxidant response to artichoke leaf extract supplementation in metabolic syndrome: A double-blind placebo-controlled randomized clinical trial. Clin. Nutr. 2018, 37, 790–796. [Google Scholar] [CrossRef]
- Rondanelli, M.; Giacosa, A.; Opizzi, A.; Faliva, M.A.; Sala, P.; Perna, S.; Riva, A.; Morazzoni, P.; Bombardelli, E. Beneficial effects of artichoke leaf extract supplementation on increasing HDL-cholesterol in subjects with primary mild hypercholesterolaemia: A double-blind, randomized, placebo-controlled trial. Int. J. Food Sci. Nutr. 2013, 64, 7–15. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Fogacci, F.; Bove, M.; Giovannini, M.; Veronesi, M.; Borghi, C. Short-Term Effects of Dry Extracts of Artichokeand Berberis in Hypercholesterolemic Patients Without Cardiovascular Disease. Am. J. Cardiol. 2019, 123, 588–591. [Google Scholar] [CrossRef] [PubMed]
- Barrat, E.; Zaïr, Y.; Ogier, N.; Housez, B.; Vergara, C.; Maudet, C.; Lescuyer, J.F.; Bard, J.M.; Carpentier, Y.A.; Cazaubiel, M.; et al. A combined natural supplement lowers LDL cholesterol in subjects with moderate untreated hypercholesterolemia: A randomized placebo-controlled trial. Int. J. Food Sci. Nutr. 2013, 64, 882–889. [Google Scholar] [CrossRef]
- Ogier, N.; Amiot, M.J.; Georgé, S.; Maillot, M.; Mallmann, C.; Maraninchi, M.; Morange, S.; Lescuyer, J.F.; Peltier, S.L.; Cardinault, N. LDL-cholesterol-lowering effect of a dietary supplement with plant extracts in subjects with moderate hypercholesterolemia. Eur. J. Nutr. 2013, 52, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Björntorp, P. “Portal” adipose tissue as a generator of risk factors for cardiovascular disease and diabetes. Arteriosclerosis 1990, 10, 493–496. [Google Scholar] [CrossRef]
- Attallah, H.; Friedlander, A.L.; Hoffman, A.R. Visceral obesity, impaired glucose tolerance, metabolic syndrome, and growth hormone therapy. Growth Horm. IGF Res. 2006, 16, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi-Mameghani, M.; Asghari-Jafarabadi, M.; Rezazadeh, K. TCF7L2-rs7903146 polymorphism modulates the effect of artichoke leaf extract supplementation on insulin resistance in metabolic syndrome: A randomized, double-blind, placebo-controlled trial. J. Integr. Med. 2018, 16, 329–334. [Google Scholar] [CrossRef]
- Rezazadeh, K.; Rahmati-Yamchi, M.; Mohammadnejad, L.; Ebrahimi-Mameghani, M.; Delazar, A. Effects of artichoke leaf extract supplementation on metabolic parameters in women with metabolic syndrome: Influence of TCF7L2-rs7903146 and FTO-rs9939609 polymorphisms. Phyther. Res. 2018, 32, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Rodgers, K.; Lai, C.-T.; Sutherland, S. Challenges in natural health product research: The importance of standardization. Proc. West. Pharmacol. Soc. 2007, 50, 24–30. [Google Scholar]
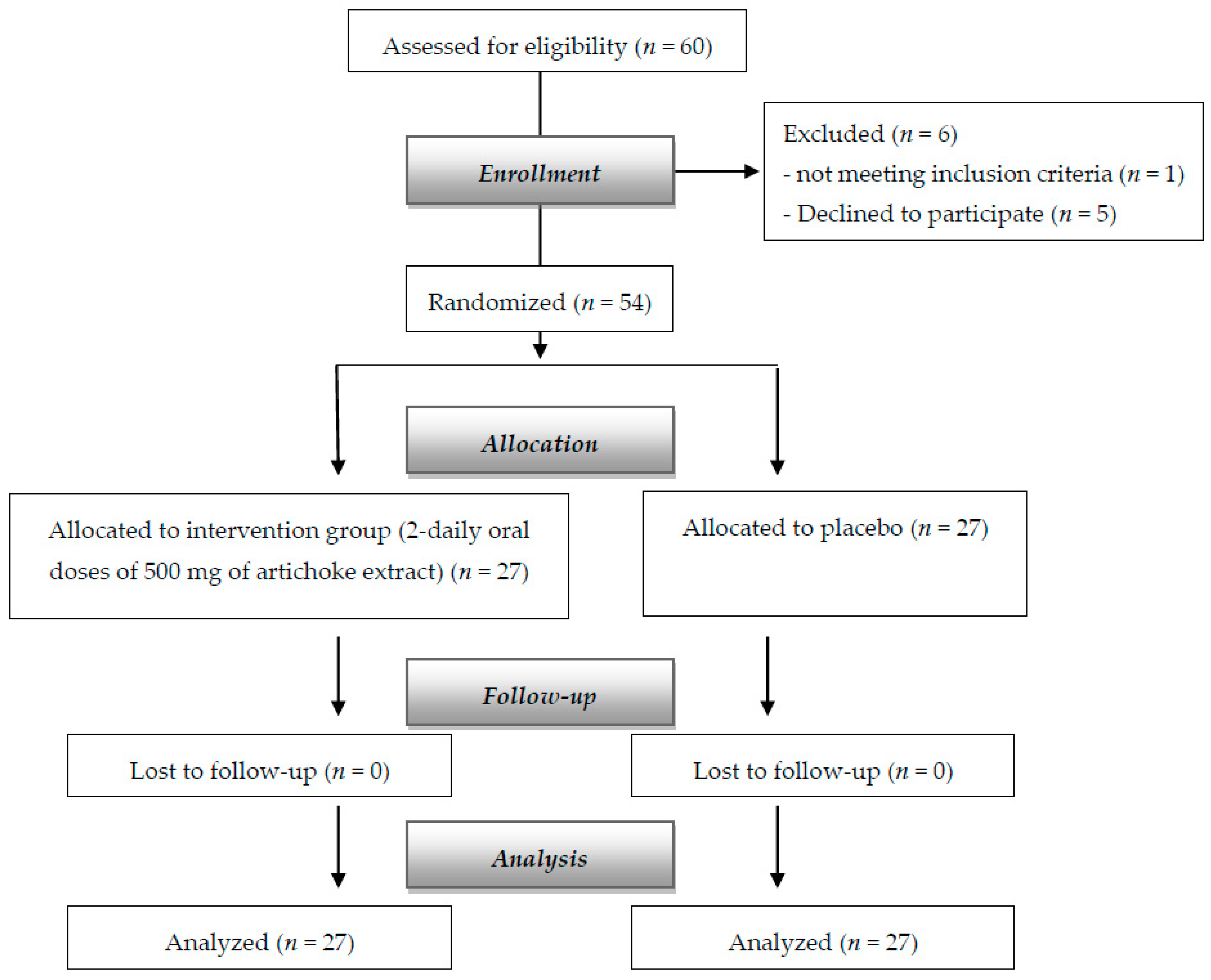
| Supplemented (n = 27) Mean (SD) | Placebo (n = 27) Mean (SD) | p-Value | |
|---|---|---|---|
| Age (years) | 51.44 (6.64) | 51.55 (6.01) | 0.95 |
| BMI (kg/m2) | 28.95 (3.59) | 29.72 (2.53) | 0.36 |
| Primary endpoint | |||
| Fasting Glycemia (mmol/L) | 6.15 (0.53) | 6.23 (0.51) | 0.59 |
| Secondary endpoints | |||
| Total Cholesterol (mg/dL) | 5.43 (0.53) | 5.35 (0.56) | 0.59 |
| HDL (mg/dL) | 1.51 (0.48) | 1.49 (0.55) | 0.89 |
| Total Cholesterol/HDL | 3.90 (1.12) | 4.08 (1.52) | 0.62 |
| LDL (mg/dL) | 3.29 (0.57) | 3.23 (0.86) | 0.75 |
| LDL/HDL | 2.45 (0.99) | 2.62 (1.38) | 0.61 |
| Triglycerides (mg/dL) | 1.35 (0.49) | 1.36 (0.36) | 0.93 |
| Insulin (mcU/mL) | 14.87 (7.19) | 12.69 (2.68) | 0.15 |
| Glycated Hemoglobin (%) | 6.10 (0.56) | 6.30 (0.64) | 0.22 |
| A1c-Derived Average Glucose (mmol/L) | 7.12 (0.89) | 7.45 (1.03) | 0.22 |
| HOMA | 4.08 (1.96) | 3.26 (1.01) | 0.06 |
| ApoB (mg/dL) | 119.78 (18.27) | 117.89 (13.77) | 0.67 |
| ApoA (mg/dL) | 123.04 (16.95) | 120.11 (22.63) | 0.59 |
| ApoB/ApoA | 0.99 (0.21) | 1.01 (0.21) | 0.73 |
| Systolic Blood Pressure (mmHg) | 124.42 (6.68) | 123.52 (7.31) | 0.64 |
| Diastolic Blood Pressure (mmHg) | 81.54 (5.62) | 80.37 (6.19) | 0.47 |
| AST (UI/L) | 18.48 (5.14) | 18.37 (3.94) | 0.93 |
| ALT (UI/L) | 19.48 (5.06) | 20.22 (3.84) | 0.55 |
| GGT (U/L) | 18.19 (7.20) | 19.44 (6.42) | 0.50 |
| Creatinine (mg/dL) | 0.83 (0.07) | 0.86 (0.12) | 0.41 |
| Waist Circumference (cm) | 108.68 (10.79) | 106.89 (12.63) | 0.58 |
| VAT (g) | 1693.37 (871.50) | 1849.41 (851.68) | 0.51 |
| Fat Mass (g) | 32,620.00 (12,068.42) | 36,380.78 (10,132.98) | 0.22 |
| Lean Mass (g) | 43,296.41 (8917.54) | 45,429.04 (6867.50) | 0.33 |
| Supplemented Group (n = 27) | Placebo Group (n = 27) | |||||
|---|---|---|---|---|---|---|
| t0 | t1 | t2 | t0 | t1 | t2 | |
| Primary endpoint | ||||||
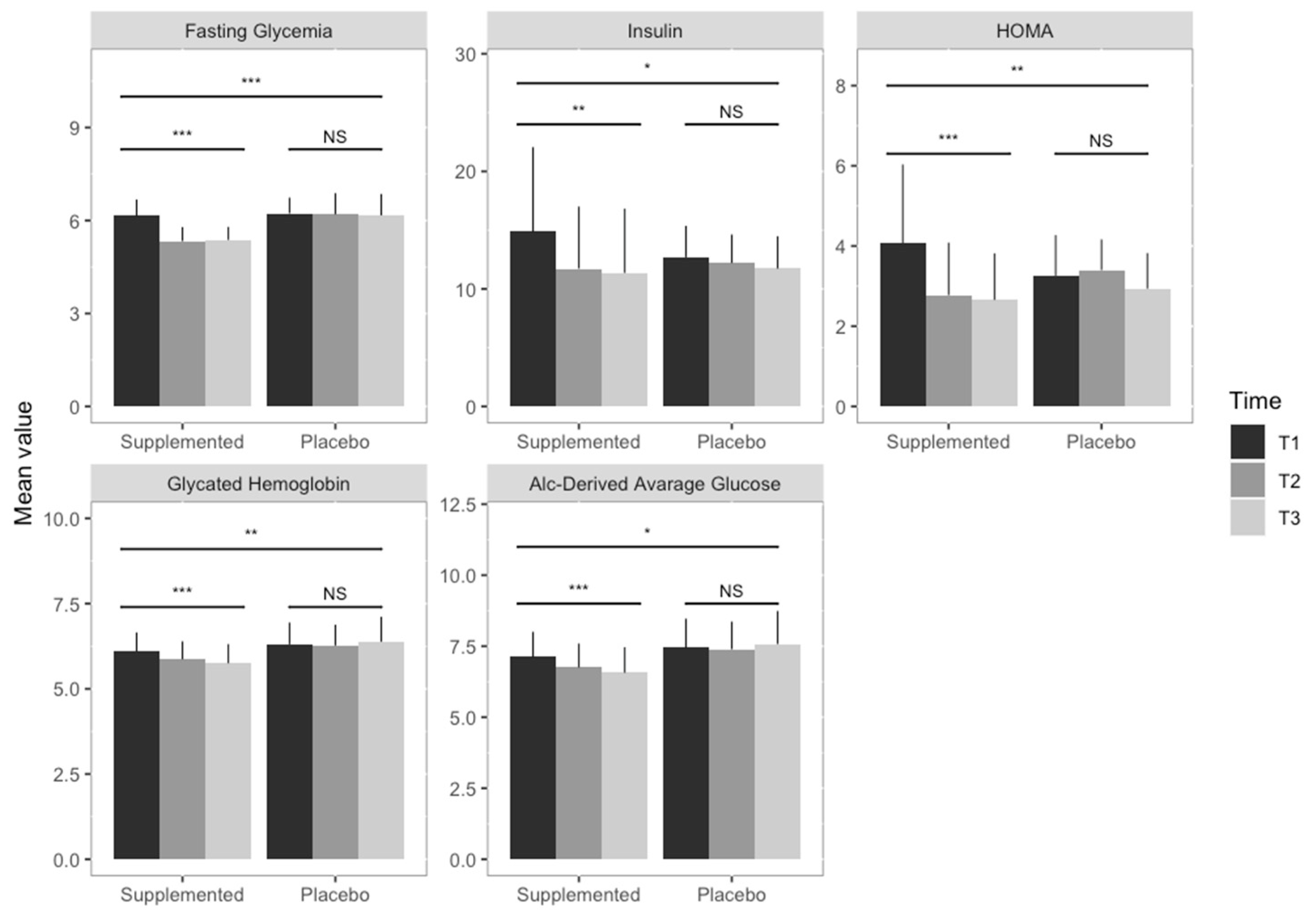
| Fasting Glycemia (mmol/L) | 6.15 (0.53) | 5.33 (0.46) | 5.37 (0.43) | 6.23 (0.51) | 6.20 (0.68) | 6.17 (0.69) |
| Secondary endpoints | ||||||
| Total Cholesterol (mg/dL) | 5.43 (0.53) | 5.29 (0.68) | 5.37 (0.55) | 5.35 (0.56) | 5.41 (0.63) | 5.42 (0.63) |
| HDL (mg/dL) | 1.51 (0.48) | 1.61 (0.52) | 1.68 (0.50) | 1.49 (0.55) | 1.45 (0.54) | 1.46 (0.53) |
| Total Cholesterol/HDL | 3.90 (1.12) | 3.57 (1.04) | 3.44 (0.95) | 4.08 (1.52) | 4.18 (1.38) | 4.17 (1.36) |
| LDL (mg/dL) | 3.29 (0.57) | 3.11 (0.67) | 3.11 (0.54) | 3.23 (0.86) | 3.34 (0.84) | 3.34 (0.87) |
| LDL/HDL | 2.45 (0.99) | 2.18 (0.95) | 2.07 (0.88) | 2.62 (1.38) | 2.70 (1.24) | 2.69 (1.23) |
| Triglycerides (mg/dL) | 1.35 (0.49) | 1.26 (0.43) | 1.28 (0.41) | 1.36 (0.36) | 1.33 (0.34) | 1.36 (0.37) |
| Insulin (mcU/mL) | 14.87 (7.19) | 11.72 (5.29) | 11.36 (5.48) | 12.69 (2.68) | 12.20 (2.44) | 11.73 (2.76) |
| Glycated Hemoglobin (%) | 6.10 (0.56) | 5.87 (0.53) | 5.75 (0.57) | 6.30 (0.64) | 6.27 (0.62) | 6.38 (0.74) |
| A1c-Derived Average Glucose (mmol/L) | 7.12 (0.89) | 6.75 (0.84) | 6.56 (0.90) | 7.45 (1.03) | 7.39 (0.98) | 7.57 (1.17) |
| HOMA | 4.08 (1.96) | 2.77 (1.31) | 2.66 (1.16) | 3.26 (1.01) | 3.40 (0.77) | 2.94 (0.89) |
| ApoB (mg/dL) | 119.78 (18.27) | 115.11 (17.41) | 112.33 (22.06) | 117.89 (13.77) | 121.44 (15.82) | 122.44 (15.63) |
| ApoA (mg/dL) | 123.04 (16.95) | 124.74 (18.61) | 130.52 (15.26) | 120.11 (22.63) | 115.78 (22.94) | 118.59 (18.92) |
| ApoB/ApoA | 0.99 (0.21) | 0.94 (0.18) | 0.87 (0.21) | 1.01 (0.21) | 1.08 (0.24) | 1.06 (0.20) |
| Systolic Blood Pressure (mmHg) | 124.42 (6.68) | 122.88 (7.77) | - | 123.52 (7.31) | 122.59 (5.61) | - |
| Diastolic Blood Pressure (mmHg) | 81.54 (5.62) | 80.38 (5.99)) | - | 80.37 (6.19) | 79.44 (5.60) | - |
| AST (UI/L) | 18.48 (5.14) | 18.59 (3.96) | 18.81 (3.29) | 18.37 (3.94) | 18.78 (3.88) | 17.59 (3.46) |
| ALT (UI/L) | 19.48 (5.06) | 19.19 (4.35) | 17.52 (3.75) | 20.22 (3.84) | 20.33 (3.23) | 20.78 2.69 |
| GGT (U/L) | 18.19 (7.20) | 18.30 (6.82) | 19.30 (4.05) | 19.44 (6.42) | 19.22 (6.17) | 19.00 (6.69) |
| Creatinine (mg/dL) | 0.83 (0.07) | 0.83 (0.08) | 0.82 (0.07) | 0.86 (0.12) | 0.84 (0.12) | 0.84 (0.12) |
| Waist Circumference (cm) | 108.68 (10.79) | 105.35 (9.81) | - | 106.89 (12.63) | 105.45 (12.19) | - |
| VAT (g) | 1693.37 (871.50) | 1482.63 (823.19) | - | 1849.41 (851.68) | 1861.04 (847.59) | - |
| Fat Mass (g) | 32,620 (12,068.42) | 30,847.37 (10,892.80) | - | 36,380.78 (10,132.98) | 34,940.78 (13,435.03) | - |
| Lean Mass (g) | 43,296.41 (8917.54) | 43,900.81 (9097.43) | - | 45,429.04 (6867.50) | 45,601.11 (7039.09) | - |
| Time * Group β (95% CI) | p-Value | |
|---|---|---|
| Primary endpoint | ||
| Fasting Glycemia (mmol/L) | 0.36 (0.24; 0.48) | <0.0001 |
| Secondary endpoints | ||
| Total Cholesterol (mg/dL) | 0.06 (−0.07; 0.14) | 0.12 |
| HDL (mg/dL) | −0.10 (−0.13; 0.06) | <0.0001 |
| Total Cholesterol/HDL | 0.27 (0.15; 0.40) | 0.0002 |
| LDL (mg/dL) | 0.15 (0.05; 0.24) | 0.005 |
| LDL/HDL | 0.23 (0.11; 0.35) | 0.001 |
| Triglycerides (mg/dL) | 0.03 (−0.05; 0.12) | 0.55 |
| Insulin (mcU/mL) | 1.28 (0.23; 2.33) | 0.04 |
| Glycated Hemoglobin (%) | 0.21 (0.12; 0.31) | 0.0002 |
| A1c-Derived Average Glucose (mmol/L) | 0.34 (0.18; 0.50) | 0.0002 |
| HOMA | 0.55 (0.19; 0.91) | 0.007 |
| ApoB (mg/dL) | 6.00 (1.69; 10.31) | 0.01 |
| ApoA (mg/dL) | −4.50 (−8.25; −0.75) | 0.04 |
| ApoB/ApoA | 0.08 (0.03; 0.13) | 0.003 |
| Systolic Blood Pressure (mmHg) | 0.61 (−2.47; 3.69) | 0.83 |
| Diastolic Blood Pressure (mmHg) | 0.23 (−2.47; 2.92) | 0.90 |
| AST (UI/L) | −0.55 (−1.58; 0.47) | 0.37 |
| ALT (UI/L) | 1.26 (0.24; 2.27) | 0.03 |
| GGT (U/L) | −0.78 (−1.94; 0.38) | 0.28 |
| Creatinine (mg/dL) | −0.002 (−0.02; 0.02) | 0.90 |
| Waist Circumference (cm) | 1.89 (0.16; 3.61) | 0.05 |
| VAT (g) | 222.37 (83.54; 361.20) | 0.005 |
| Fat Mass (g) | 332.63 (−2754.20; 3419.46) | 0.90 |
| Lean Mass (g) | −432.33 (−1157.70; 293.03) | 0.34 |
| Supplemented Group | Placebo Group | |||
|---|---|---|---|---|
| Time β (95% CI) | p-Value | Time β (95% CI) | p-Value | |
| Primary endpoint | ||||
| Fasting Glycemia (mmol/L) | −0.39 (0.31; 0.45) | <0.0001 | −0.03 (−0.10; 0.04) | 0.53 |
| Secondary endpoints | ||||
| Total Cholesterol (mg/dL) | −0.03 (−0.09; 0.03) | 0.003 | 0.03 (−0.004; 0.07) | 0.38 |
| HDL (mg/dL) | 0.08 (0.06; 0.10) | <0.0001 | −0.02 (−0.05; 0.11) | 0.52 |
| Total Cholesterol/HDL | −0.23 (−0.31; −0.15) | <0.0001 | 0.04 (−0.06; 0.14) | 0.53 |
| LDL (mg/dL) | −0.09 (−0.17; −0.01) | 0.03 | 0.05 (−0.0004; 0.11) | 0.30 |
| LDL/HDL | −0.19 (−0.27; −0.11) | <0.0001 | 0.04 (−0.05; 0.13) | 0.53 |
| Triglycerides (mg/dL) | −0.04 (−0.12; 0.05) | 0.41 | −0.003 (−0.03; 0.03) | 0.86 |
| Insulin (mc/U/mL) | −1.76 (−2.73; −0.79) | 0.001 | −0.48 (−0.91; −0.05) | 0.24 |
| Glycated Hemoglobin (%) | −0.18 (−0.22; −0.13) | <0.0001 | 0.04 (−0.05; 0.13) | 0.53 |
| A1c-Derived Average Glucose (mmol/L) | −0.28 (−0.36; −0.20) | <0.0001 | 0.06 (−0.08; 0.20) | 0.53 |
| HOMA | −0.71 (−1.07; −0.41) | <0.0001 | −0.16 (−0.37; 0.05) | 0.48 |
| ApoB (mg/dL) | −3.72 (−7.15; −0.30) | 0.04 | 2.28 (−0.42; 4.97) | 0.40 |
| ApoA (mg/dL) | 3.74 (2.19; 5.29) | <0.0001 | −0.76 (−4.22; 2.70) | 0.72 |
| ApoB/ApoA | −0.06 (−0.09; −0.03) | 0.002 | 0.02 (−0.01; 0.06) | 0.52 |
| Systolic Blood Pressure (mmHg) | −1.54 (−3.73; 0.66) | 0.19 | −0.92 (−3.19; 1.34) | 0.53 |
| Diastolic Blood Pressure (mmHg) | −1.15 (−2.70; 0.39) | 0.18 | −0.93 (−3.19; 1.34) | 0.53 |
| AST (UI/L) | 0.17 (−0.65; 0.98) | 0.68 | −0.39 (−1.02; 0.25) | 0.52 |
| ALT (UI/L) | −0.98 (−1.79; −0.17) | 0.03 | 0.28 (−0.35; −0.91) | 0.53 |
| GGT (U/L) | 0.55 (−0.42; 1.54) | 0.30 | −0.22 (−0.87; 0.43) | 0.57 |
| Creatinine (mg/dL) | −0.006 (−0.02; 0.008) | 0.42 | −0.008 (−0.02; 0.006) | 0.52 |
| Waist Circumference (cm) | −3.33 (−4.67; −1.98) | <0.0001 | −1.44 (−2.58; −0.29) | 0.24 |
| VAT (g) | −210.74 (−337.26; −84.22) | 0.003 | 11.63 (−53.19; 76.55) | 0.75 |
| Fat Mass (g) | −1772.63 (−2857.07; −688.19) | 0.003 | −1440 (−4410.26; 1530.26) | 0.53 |
| Lean Mass (g) | 604.41 (20.35; 1188.46) | 0.05 | 172.07 (−287.25; 631.40) | 0.54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rondanelli, M.; Riva, A.; Petrangolini, G.; Allegrini, P.; Bernardinelli, L.; Fazia, T.; Peroni, G.; Gasparri, C.; Nichetti, M.; Faliva, M.A.; et al. The Metabolic Effects of Cynara Supplementation in Overweight and Obese Class I Subjects with Newly Detected Impaired Fasting Glycemia: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Nutrients 2020, 12, 3298. https://doi.org/10.3390/nu12113298
Rondanelli M, Riva A, Petrangolini G, Allegrini P, Bernardinelli L, Fazia T, Peroni G, Gasparri C, Nichetti M, Faliva MA, et al. The Metabolic Effects of Cynara Supplementation in Overweight and Obese Class I Subjects with Newly Detected Impaired Fasting Glycemia: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Nutrients. 2020; 12(11):3298. https://doi.org/10.3390/nu12113298
Chicago/Turabian StyleRondanelli, Mariangela, Antonella Riva, Giovanna Petrangolini, Pietro Allegrini, Luisa Bernardinelli, Teresa Fazia, Gabriella Peroni, Clara Gasparri, Mara Nichetti, Milena Anna Faliva, and et al. 2020. "The Metabolic Effects of Cynara Supplementation in Overweight and Obese Class I Subjects with Newly Detected Impaired Fasting Glycemia: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial" Nutrients 12, no. 11: 3298. https://doi.org/10.3390/nu12113298
APA StyleRondanelli, M., Riva, A., Petrangolini, G., Allegrini, P., Bernardinelli, L., Fazia, T., Peroni, G., Gasparri, C., Nichetti, M., Faliva, M. A., Naso, M., & Perna, S. (2020). The Metabolic Effects of Cynara Supplementation in Overweight and Obese Class I Subjects with Newly Detected Impaired Fasting Glycemia: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Nutrients, 12(11), 3298. https://doi.org/10.3390/nu12113298






