Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits
Abstract
1. Introduction
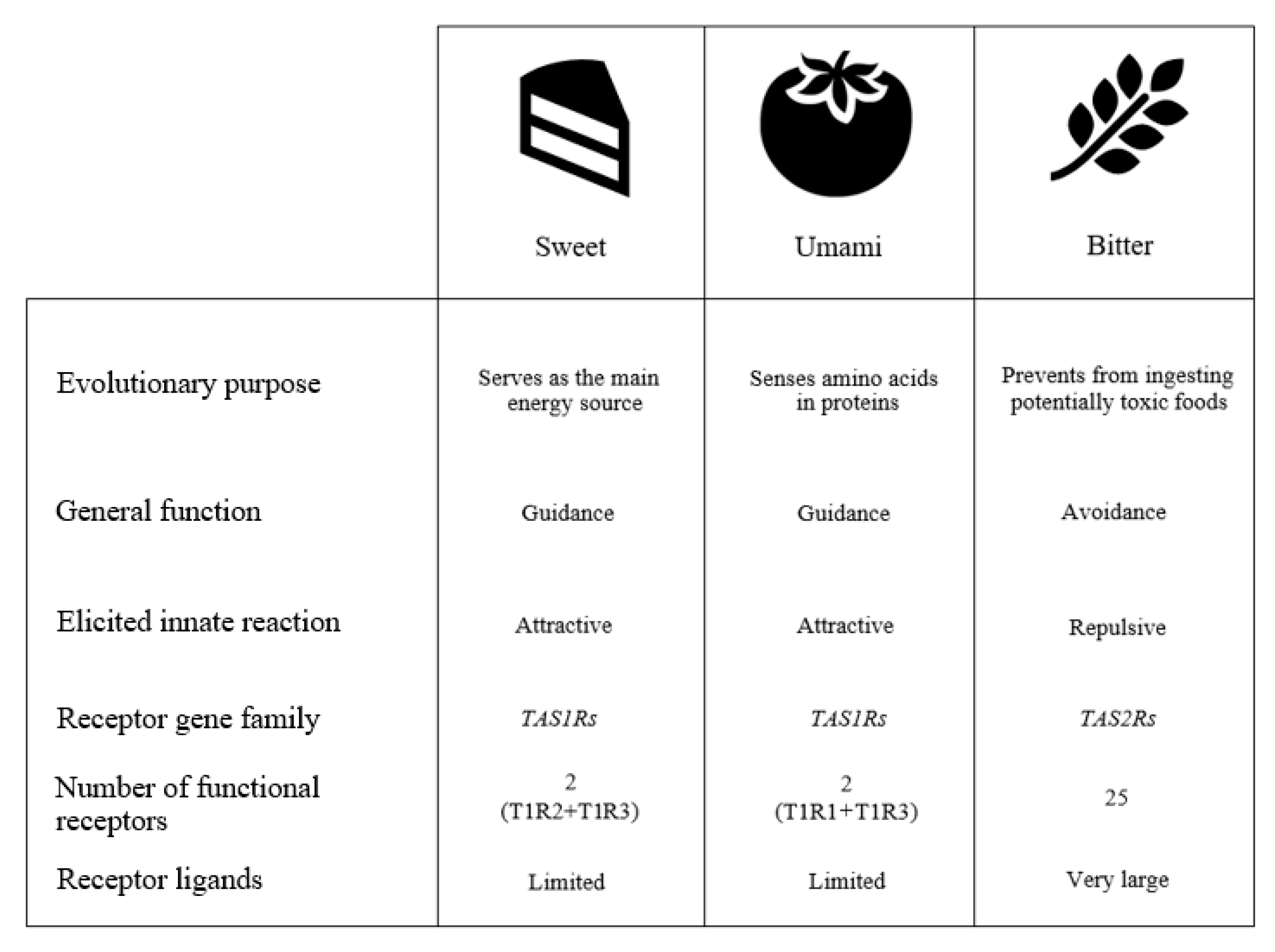
2. How We Taste
3. Taste Impairments
4. Determinants of Taste Alterations
4.1. Genetics
4.2. External Factors
4.3. Nutritional Factors
4.4. Biological Factors
4.5. Viral Illness
5. Potential Implications on Dietary Habits
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Green, B.G. Introduction: What is chemesthesis. In Chemestesis, Chemical Touch in Food and Eating; McDonald, S.T., Bolliet, D.A., Hayes, J.E., Eds.; Wiley-Blackwell Editor: Oxford, UK, 2016; pp. 1–7. ISBN 978-1-118-95173-6. [Google Scholar]
- Breslin, P.A.S. An Evolutionary Perspective on Food and Human Taste. Curr. Biol. 2013, 23, R409–R418. [Google Scholar] [CrossRef] [PubMed]
- Spence, C.; Auvray, M.; Smith, B. Confusing tastes with flavours. In Perception and Its Modalities; Stokes, D., Matthen, M., Briggs, S., Eds.; Oxford University Press: Oxford, UK, 2015; pp. 247–274. [Google Scholar] [CrossRef]
- ISO. Standard 5492: Terms Relating to Sensory Analysis; International Organization for Standardization: Geneva, Switzerland, 2008. [Google Scholar]
- Vincis, R.; Fontanini, A. Central taste anatomy and physiology. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 164, pp. 187–204. [Google Scholar] [CrossRef]
- De Araujo, I.E.; Simon, S.A. The gustatory cortex and multisensory integration. Int. J. Obes. 2009, 33, S34–S43. [Google Scholar] [CrossRef]
- Shepherd, G.M. Smell images and the flavour system in the human brain. Nat. Cell Biol. 2006, 444, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A. Ontogeny of taste preferences: Basic biology and implications for health. Am. J. Clin. Nutr. 2014, 99, 704S–711S. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, G.K. Basic Taste: A Perceptual Concept. J. Agric. Food Chem. 2019, 67, 13860–13869. [Google Scholar] [CrossRef]
- Witt, M. Anatomy and development of the human taste system. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 164, pp. 147–171. [Google Scholar] [CrossRef]
- Feeney, E.L.; Hayes, J.E. Regional Differences in Suprathreshold Intensity for Bitter and Umami Stimuli. Chemosens. Percept. 2014, 7, 147–157. [Google Scholar] [CrossRef]
- Colvin, J.L.; Pullicin, A.J.; Lim, J. Regional Differences in Taste Responsiveness: Effect of Stimulus and Tasting Mode. Chem. Senses 2018, 43, 645–653. [Google Scholar] [CrossRef]
- Higgins, M.J.; Hayes, J.E. Regional Variation of Bitter Taste and Aftertaste in Humans. Chem. Senses 2019, 44, 721–732. [Google Scholar] [CrossRef]
- Roper, S.D.; Chaudhari, N. Taste buds: Cells, signals and synapses. Nat. Rev. Neurosci. 2017, 18, 485–497. [Google Scholar] [CrossRef]
- Bachmanov, A.A.; Bosak, N.P.; Lin, C.; Matsumoto, I.; Ohmoto, M.; Reed, D.R.; Nelson, T.M. Genetics of Taste Receptors. Curr. Pharm. Des. 2014, 20, 2669–2683. [Google Scholar] [CrossRef]
- Tu, Y.-H.; Cooper, A.J.; Teng, B.; Chang, R.B.; Artiga, D.J.; Turner, H.N.; Mulhall, E.M.; Ye, W.; Smith, A.D.; Liman, E.R. An evolutionarily conserved gene family encodes proton-selective ion channels. Science 2018, 359, 1047–1050. [Google Scholar] [CrossRef]
- Leffler, A.; Mönter, B.; Koltzenburg, M. The role of the capsaicin receptor TRPV1 and acid-sensing ion channels (ASICS) in proton sensitivity of subpopulations of primary nociceptive neurons in rats and mice. Neuroscience 2006, 139, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Reeh, P.W.; Steen, K.H. Chapter 8. Tissue acidosis in nociception and pain. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 1996; Volume 113, pp. 143–151. [Google Scholar] [CrossRef]
- Bigiani, A. Does ENaC Work as Sodium Taste Receptor in Humans? Nutrients 2020, 12, 1195. [Google Scholar] [CrossRef] [PubMed]
- Diószegi, J.; Llanaj, E.; Adany, R. Genetic Background of Taste Perception, Taste Preferences, and Its Nutritional Implications: A Systematic Review. Front. Genet. 2019, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- Temussi, P.A. Natural sweet macromolecules: How sweet proteins work. Cell. Mol. Life Sci. 2006, 63, 1876–1888. [Google Scholar] [CrossRef] [PubMed]
- Dubois, G.E.; Prakash, I. Non-Caloric Sweeteners, Sweetness Modulators, and Sweetener Enhancers. Annu. Rev. Food Sci. Technol. 2012, 3, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Dubois, G.E. Molecular mechanism of sweetness sensation. Physiol. Behav. 2016, 164, 453–463. [Google Scholar] [CrossRef]
- Morini, G.; Bassoli, A.; Temussi, P.A. From Small Sweeteners to Sweet Proteins: Anatomy of the Binding Sites of the Human T1R2_T1R3 Receptor. J. Med. Chem. 2005, 48, 5520–5529. [Google Scholar] [CrossRef]
- Hartley, I.E.; Liem, D.G.; Keast, R.S.J. Umami as an ‘Alimentary’ Taste. A New Perspective on Taste Classification. Nutrients 2019, 11, 182. [Google Scholar] [CrossRef]
- Behrens, M.; Meyerhof, W. Vertebrate Bitter Taste Receptors: Keys for Survival in Changing Environments. J. Agric. Food Chem. 2018, 66, 2204–2213. [Google Scholar] [CrossRef]
- Finger, T.E.; Kinnamon, S.C. Taste isn’t just for taste buds anymore. F1000 Biol. Rep. 2011, 3, 20. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-J.; Depoortere, I.; Hatt, H. Therapeutic potential of ectopic olfactory and taste receptors. Nat. Rev. Drug Discov. 2018, 18, 116–138. [Google Scholar] [CrossRef] [PubMed]
- Chamoun, E.; Mutch, D.M.; Allen-Vercoe, E.; Buchholz, A.C.; Duncan, A.M.; Spriet, L.L.; Haines, J.; Ma, D.W. Guelph Family Health Study. A review of the associations between single nucleotide polymorphisms in taste receptors, eating behaviors, and health. Crit. Rev. Food Sci. Nutr. 2017, 58, 194–207. [Google Scholar] [CrossRef]
- Ekstrand, B.; Young, J.F.; Rasmussen, M.K. Taste receptors in the gut—A new target for health promoting properties in diet. Food Res. Int. 2017, 100, 1–8. [Google Scholar] [CrossRef]
- Rawal, S.; Hoffman, H.J.; Bainbridge, K.E.; Huedo-Medina, T.B.; Duffy, V.B. Prevalence and Risk Factors of Self-Reported Smell and Taste Alterations: Results from the 2011–2012 US National Health and Nutrition Examination Survey (NHANES). Chem. Senses 2015, 41, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Deems, D.A.; Doty, R.L.; Settle, R.G.; Moore-Gillon, V.; Shaman, P.; Mester, A.F.; Kimmelman, C.P.; Brightman, V.J.; Snow, J.B. Smell and Taste Disorders, A Study of 750 Patients From the University of Pennsylvania Smell and Taste Center. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 519–528. [Google Scholar] [CrossRef]
- Hummel, T.; Landis, B.N.; Hüttenbrink, K.-B. Smell and taste disorders. GMS Curr. Top. Otorhinolaryngol. Head Neck Surg. 2012, 20, 93–111. [Google Scholar]
- Davis, B.; Hoffman, H. What the Numbers Mean: An Epidemiological Perspective on Taste and Smell. Available online: https://www.nidcd.nih.gov/health/statistics/what-numbersmean-epidemiological-perspective-taste-smell (accessed on 8 June 2020).
- Welge-Lüssen, A.; Dörig, P.; Wolfensberger, M.; Krone, F.; Hummel, T. A study about the frequency of taste disorders. J. Neurol. 2010, 258, 386–392. [Google Scholar] [CrossRef]
- Syed, Q.; Hendler, K.T.; Koncilja, K. The Impact of Aging and Medical Status on Dysgeusia. Am. J. Med. 2016, 129, 753.e1–753.e6. [Google Scholar] [CrossRef]
- Smith, A.; Farbman, A.; Dancis, J. Absence of Taste-Bud Papillae in Familial Dysautonomia. Science 1965, 147, 1040–1041. [Google Scholar] [CrossRef]
- Kim, U.-K.; Jorgenson, E.; Coon, H.; Leppert, M.; Risch, N.; Drayna, D. Positional Cloning of the Human Quantitative Trait Locus Underlying Taste Sensitivity to Phenylthiocarbamide. Science 2003, 299, 1221–1225. [Google Scholar] [CrossRef]
- Risso, D.S.; Mezzavilla, M.; Pagani, L.; Robino, A.; Morini, G.; Tofanelli, S.; Carrai, M.; Campa, D.; Barale, R.; CaraDonna, F.; et al. Global diversity in the TAS2R38 bitter taste receptor: Revisiting a classic evolutionary PROPosal. Sci. Rep. 2016, 6, 25506. [Google Scholar] [CrossRef] [PubMed]
- Risso, D.S.; Morini, G.; Pagani, L.; Quagliariello, A.; Giuliani, C.; De Fanti, S.; Sazzini, M.; Luiselli, N.; Tofanelli, S. Genetic signature of differential sensitivity to stevioside in the Italian population. Genes Nutr. 2014, 9, 401. [Google Scholar] [CrossRef] [PubMed]
- Genter, M.B.; Doty, R.L. Toxic exposures and the senses of taste and smell. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 164, pp. 389–408. [Google Scholar] [CrossRef]
- Nolden, A.A.; Hwang, L.-D.; Boltong, A.; Reed, D.R. Chemosensory Changes from Cancer Treatment and Their Effects on Patients’ Food Behavior: A Scoping Review. Nutrients 2019, 11, 2285. [Google Scholar] [CrossRef] [PubMed]
- Van Oort, S.; Kramer, E.; De Groot, J.-W.; Visser, O. Taste alterations and cancer treatment. Curr. Opin. Support. Palliat. Care 2018, 12, 162–167. [Google Scholar] [CrossRef]
- Schofield, P.W.; Doty, R.L. The influence of head injury on olfactory and gustatory function. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2019; Volume 164, pp. 409–429. [Google Scholar] [CrossRef]
- Elterman, K.G.; Mallampati, S.R.; Kaye, A.D.; Urman, R.D. Postoperative Alterations in Taste and Smell. Anesthesiol. Pain Med. 2014, 4, e18527. [Google Scholar] [CrossRef]
- McLaughlin, L. Taste dysfunction and eating behaviors in survivors of head and neck cancer treatment. Medsurg Nurs. 2014, 23, 165. [Google Scholar]
- Bromley, S.M. Smell and taste disorders: A primary care approach. Am. Fam. Physician 2000, 61, 427–436. [Google Scholar]
- Klasser, G.D.; Utsman, R.; Epstein, J.B. Taste change associated with a dental procedure: Case report and review of the literature. J. Can. Dent. Assoc. 2008, 74, 455–461. [Google Scholar]
- Schneller, L.M.; McIntosh, S.; Li, N.; Rahman, I.; Ossip, D.; Goniewicz, M.; O’Connor, R.J. Tobacco use and chemosensory impairments among current adult tobacco users in the US: Data from NHANES 2013–2014. Tob. Induc. Dis. 2018, 16, 43. [Google Scholar] [CrossRef]
- Silva, C.S.; Dias, V.R.; Almeida, J.A.R.; Brazil, J.M.; Santos, R.A.; Milagres, M.P. Effect of Heavy Consumption of Alcoholic Beverages on the Perception of Sweet and Salty Taste. Alcohol Alcohol. 2015, 51, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, T.S.; Blanchard, P.; Wang, L.; Foote, R.L.; Zhang, X.; Frank, S.J. Radiation-Related Alterations of Taste Function in Patients With Head and Neck Cancer: A Systematic Review. Curr. Treat. Options Oncol. 2018, 19, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Yagi, T.; Asakawa, A.; Ueda, H.; Ikeda, S.; Miyawaki, S.; Inui, A. The Role of Zinc in the Treatment of Taste Disorders. Recent Patents Food Nutr. Agric. 2013, 5, 44–51. [Google Scholar] [CrossRef]
- Risso, D.S.; Howard, L.; VanWaes, C.; Drayna, D. A potential trigger for pine mouth: A case of a homozygous phenylthiocarbamide taster. Nutr. Res. 2015, 35, 1122–1125. [Google Scholar] [CrossRef]
- Martin, L.J.; Sollars, S.I. Contributory role of sex differences in the variations of gustatory function. J. Neurosci. Res. 2016, 95, 594–603. [Google Scholar] [CrossRef]
- Toffanello, E.; Inelmen, E.; Imoscopi, A.; Perissinotto, E.; Coin, A.; Miotto, F.; Donini, L.M.; Cucinotta, D.; Barbagallo, M.; Manzato, E.; et al. Taste loss in hospitalized multimorbid elderly subjects. Clin. Interv. Aging 2013, 8, 167–174. [Google Scholar] [CrossRef]
- Shah, M.; Deeb, J.; Fernando, M.; Noyce, A.; Visentin, E.; Findley, L.J.; Hawkes, C.H. Abnormality of taste and smell in Parkinson’s disease. Park. Relat. Disord. 2009, 15, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Lang, C.; Leuschner, T.; Ulrich, K.; Stößel, C.; Heckmann, J.; Hummel, T. Taste in dementing diseases and Parkinsonism. J. Neurol. Sci. 2006, 248, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Akerlund, A.; Bende, M.; Murphy, C. Olfactory Threshold and Nasal Mucosal Changes in Experimentally Induced Common Cold. Acta Oto-Laryngol. 1995, 115, 88–92. [Google Scholar] [CrossRef]
- Damm, M.; Vent, J.; Schmidt, M.; Theissen, P.; Eckel, H.E.; Lötsch, J.; Hummel, T. Intranasal Volume and Olfactory Function. Chem. Senses 2002, 27, 831–839. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, M.; Brand, J.; Huang, L. Inflammation and Taste Disorders. Ann. N. Y. Acad. Sci. 2009, 1170, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.W.; Brann, D.H.; Farruggia, M.C.; Bhutani, S.; Pellegrino, R.; Tsukahara, T.; Weinreb, C.; Joseph, P.V.; Larson, E.D.; Parma, V.; et al. COVID-19 and the Chemical Senses: Supporting Players Take Center Stage. Neuron 2020, 107, 219–233. [Google Scholar] [CrossRef]
- Pellegrino, R.; Cooper, K.W.; Di Pizio, A.; Joseph, P.V.; Bhutani, S.; Parma, V. Coronaviruses and the Chemical Senses: Past, Present, and Future. Chem. Senses 2020, 45, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Huart, C.; Philpott, C.; Konstantinidis, I.; Altundag, A.; Trecca, E.M.C.; Cassano, M.; Rombaux, P.; Hummel, T. Comparison of COVID-19 and common cold chemosensory dysfunction. Rhinology 2020, 58. [Google Scholar] [CrossRef]
- Aziz, M.; Perisetti, A.; Lee-Smith, W.M.; Gajendran, M.; Bansal, P.; Goyal, H. Taste Changes (Dysgeusia) in COVID-19: A Systematic Review and Meta-analysis. Gastroenterology 2020, 159, 1132–1133. [Google Scholar] [CrossRef] [PubMed]
- Lozada-Nur, F.; Chainani-Wu, N.; Fortuna, G.; Sroussi, H. Dysgeusia in COVID-19: Possible Mechanisms and Implications. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, C.; Bufe, B.; Winnig, M.; Hofmann, T.; Frank, O.; Behrens, M.; Lewtschenko, T.; Slack, J.P.; Ward, C.D.; Meyerhof, W. Bitter Taste Receptors for Saccharin and Acesulfame K. J. Neurosci. 2004, 24, 10260–10265. [Google Scholar] [CrossRef] [PubMed]
- Antinucci, M.; Risso, D.S. A Matter of Taste: Lineage-Specific Loss of Function of Taste Receptor Genes in Vertebrates. Front. Mol. Biosci. 2017, 4, 81. [Google Scholar] [CrossRef]
- Li, X.; Glaser, D.; Li, W.; Johnson, W.E.; O’Brien, S.J.; Beauchamp, G.K.; Brand, J.G. Analyses of Sweet Receptor Gene (Tas1r2) and Preference for Sweet Stimuli in Species of Carnivora. J. Hered. 2009, 100, S90–S100. [Google Scholar] [CrossRef]
- Risso, D.S.; Kozlitina, J.; Sainz, E.; Gutierrez, J.; Wooding, S.; Getachew, B.; Luiselli, D.; Berg, C.J.; Drayna, D. Genetic Variation in the TAS2R38 Bitter Taste Receptor and Smoking Behaviors. PLoS ONE 2016, 11, e0164157. [Google Scholar] [CrossRef]
- Kozlitina, J.; Risso, D.; Lansu, K.; Olsen, R.H.J.; Sainz, E.; Luiselli, D.; Barik, A.; Frigerio-Domingues, C.; Pagani, L.; Wooding, S.; et al. An African-specific haplotype in MRGPRX4 is associated with menthol cigarette smoking. PLoS Genet. 2019, 15, e1007916. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, A.; Tsuchiya, A.; Nakajima, K.-I.; Ito, K.; Terada, T.; Shimizu-Ibuka, A.; Briand, L.; Asakura, T.; Misaka, T.; Abe, K. Human sweet taste receptor mediates acid-induced sweetness of miraculin. Proc. Natl. Acad. Sci. USA 2011, 108, 16819–16824. [Google Scholar] [CrossRef]
- Sanematsu, K.; Kusakabe, Y.; Shigemura, N.; Hirokawa, T.; Nakamura, S.; Imoto, T.; Ninomiya, Y. Molecular Mechanisms for Sweet-suppressing Effect of Gymnemic Acids. J. Biol. Chem. 2014, 289, 25711–25720. [Google Scholar] [CrossRef] [PubMed]
- Barragán, R.; Coltell, O.; Portolés, O.; Asensio, E.M.; Sorlí, J.V.; Ortega-Azorín, C.; González, J.I.; Saiz, C.; Fernández-Carrión, R.; Ordovás, J.M.; et al. Bitter, Sweet, Salty, Sour and Umami Taste Perception Decreases with Age: Sex-Specific Analysis, Modulation by Genetic Variants and Taste-Preference Associations in 18 to 80 Year-Old Subjects. Nutrients 2018, 10, 1539. [Google Scholar] [CrossRef]
- Cowart, B.J.; Young, I.; Feldman, R.S.; Lowry, L.D. Clinical disorders of smell and taste. In Tasting and Smelling; Academic Press: Cambridge, MA, USA, 1997; pp. 175–198. [Google Scholar]
- Imoscopi, A.; Inelmen, E.M.; Sergi, G.; Miotto, F.; Manzato, E. Taste loss in the elderly: Epidemiology, causes and consequences. Aging Clin. Exp. Res. 2012, 24, 570–579. [Google Scholar]
- Choo, E.; Dando, R. The Impact of Pregnancy on Taste Function. Chem. Senses 2017, 42, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Henkin, R.I.; Larson, A.L.; Powell, R.D. Hypogeusia, Dysgeusia, Hyposmia, and Dysosmia following Influenza-Like Infection. Ann. Otol. Rhinol. Laryngol. 1975, 84, 672–682. [Google Scholar] [CrossRef]
- Henkin, R.I.; Levy, L.M.; Fordyce, A. Taste and smell function in chronic disease: A Review of Clinical and Biochemical Evaluations of Taste and Smell Dysfunction in Over 5000 Patients at The Taste and Smell Clinic in Washington, DC. Am. J. Otolaryngol. 2013, 34, 477–489. [Google Scholar] [CrossRef]
- Mäkelä, M.J.; Puhakka, T.; Ruuskanen, O.; Leinonen, M.; Saikku, P.; Kimpimäki, M.; Blomqvist, S.; Hyypiä, T.; Arstila, P. Viruses and Bacteria in the Etiology of the Common Cold. J. Clin. Microbiol. 1998, 36, 539–542. [Google Scholar] [CrossRef]
- Giacomelli, A.; Pezzati, L.; Conti, F.; Bernacchia, D.; Siano, M.; Oreni, L.; Rusconi, S.; Gervasoni, C.; Ridolfo, A.L.; Rizzardini, G.; et al. Self-reported olfactory and taste disorders in SARS-CoV-2 patients: A cross-sectional study. Clin. Infect. Dis. 2020, 71, 889–890. [Google Scholar] [CrossRef]
- Parma, V.; Ohla, K.; Veldhuizen, M.G.; Niv, M.Y.; Kelly, C.E.; Bakke, A.J.; Cooper, K.W.; Bouysset, C.; Pirastu, N.; Dibattista, M.; et al. More Than Smell—COVID-19 Is Associated With Severe Impairment of Smell, Taste, and Chemesthesis. Chem. Senses 2020, 45, 609–622. [Google Scholar] [CrossRef] [PubMed]
- Vaira, L.A.; Deiana, G.; Fois, A.G.; Pirina, P.; Madeddu, G.; De Vito, A.; Babudieri, S.; Petrocelli, M.; Serra, A.; Bussu, F.; et al. Objective evaluation of anosmia and ageusia in COVID -19 patients: Single-center experience on 72 cases. Head Neck 2020, 42, 1252–1258. [Google Scholar] [CrossRef]
- Zang, R.; Castro, M.F.G.; McCune, B.T.; Zeng, Q.; Rothlauf, P.W.; Sonnek, N.M.; Liu, Z.; Brulois, K.F.; Wang, X.; Greenberg, H.B.; et al. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci. Immunol. 2020, 5, eabc3582. [Google Scholar] [CrossRef] [PubMed]
- Singh, Y.; Gupta, G.; Satija, S.; Pabreja, K.; Chellappan, D.K.; Dua, K. COVID -19 transmission through host cell directed network of GPCR. Drug Dev. Res. 2020, 81, 647–649. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Shen, W.; Rowan, N.R.; Kulaga, H.; Hillel, A.; Ramanathan, M., Jr.; Lane, A.P. Elevated ACE2 expression in the olfactory neuroepithelium: Implications for anosmia and upper respiratory SARS-CoV-2 entry and replication. Eur. Respir. J. 2020, 56, 2001948. [Google Scholar] [CrossRef] [PubMed]
- Sungnak, W.; Network, H.L.B.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat. Med. 2020, 26, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Zhao, J.; Martin, W.; Kallianpur, A.; Chung, M.K.; Jehi, L.; Sharifi, N.; Erzurum, S.; Eng, C.; Cheng, F. New insights into genetic susceptibility of COVID-19: An ACE2 and TMPRSS2 polymorphism analysis. BMC Med. 2020, 18, 1–8. [Google Scholar] [CrossRef]
- Mann, N.M. Management of smell and taste problems. Clevel. Clin. J. Med. 2002, 69, 329–336. [Google Scholar] [CrossRef]
- Bergdahl, M.; Bergdahl, J. Perceived taste disturbance in adults: Prevalence and association with oral and psychological factors and medication. Clin. Oral Investig. 2002, 6, 145–149. [Google Scholar] [CrossRef]
- Hur, K.; Choi, J.S.; Zheng, M.; Shen, J.; Wrobel, B. Association of alterations in smell and taste with depression in older adults. Laryngoscope 2018, 3, 94–99. [Google Scholar] [CrossRef]
- Sergi, G.; Bano, G.; Pizzato, S.; Veronese, N.; Manzato, E. Taste loss in the elderly: Possible implications for dietary habits. Crit. Rev. Food Sci. Nutr. 2017, 57, 3684–3689. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Wen, Q.; Xu, C.; Zhang, X.; Zeng, J.; Sha, A.M.; Lan, C.; Li, L.; Wang, H.; Yang, X.; et al. Elevated Salt Taste Threshold Is Associated with Increased Risk of Coronary Heart Disease. J. Cardiovasc. Transl. Res. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moemin, A.R.; Regenstein, J.; Abdel-Rahman, M.K. New Food Products for Sensory-Compromised Situations. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1625–1639. [Google Scholar] [CrossRef]
- Tan, M.; He, F.; MacGregor, G.A. Obesity and covid-19: The role of the food industry. BMJ 2020, 369, m2237. [Google Scholar] [CrossRef] [PubMed]
- Di Renzo, L.; Gualtieri, P.; Pivari, F.; Soldati, L.; Attinà, A.; Cinelli, G.; Leggeri, C.; Caparello, G.; Barrea, L.; Scerbo, F.; et al. Eating habits and lifestyle changes during COVID-19 lockdown: An Italian survey. J. Transl. Med. 2020, 18, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bakalis, S.; Valdramidis, V.P.; Argyropoulos, D.; Ahrné, L.; Chen, J.; Cullen, P.; Cummins, E.; Datta, A.; Emmanouilidis, C.; Foster, T.; et al. Perspectives from CO+RE: How COVID-19 changed our food systems and food security paradigms. Curr. Res. Food Sci. 2020, 3, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Brach, M.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M.; et al. Effects of COVID-19 Home Confinement on Eating Behaviour and Physical Activity: Results of the ECLB-COVID19 International Online Survey. Nutrients 2020, 12, 1583. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.J.; Barrientos, R.M. The impact of nutrition on COVID-19 susceptibility and long-term consequences. Brain Behav. Immun. 2020, 87, 53–54. [Google Scholar] [CrossRef]
- WHO. Nutrition Advice for Adults during the COVID-19 Outbreak. 2020. Available online: http://www.emro.who.int/nutrition/nutrition-infocus/nutrition-advice-for-adults-during-the-covid-19-outbreak.html (accessed on 26 October 2020).
| Factor | Sub-Factor | Proposed Mechanisms of Action | References |
|---|---|---|---|
| Genetics | Genetic disorders (e.g., Familial dysautonomia) | Complete or severe depletion of taste papillae | [37] |
| Single nucleotide polymorphisms on taste receptor genes | Influence individuals’ ligand-receptor binding, generating different tasting phenotypes | [38,39,40] | |
| External | Chemicals (both from external and internal sources) | Interfere with the chemical composition or quantity of saliva; change the oral mucus membranes; impair taste receptor cells and efficacy of transduction; directly affect taste nerves | [41] |
| Drugs (e.g., antimycotics, antibiotics, anti-inflammatories, immunosuppressants, neurologic medications) | Changes in saliva production, secretion, quantity, and diffusion; damage to cranial nerves; modification of afferent pathways from the central nervous system | [41,42,43] | |
| Trauma (e.g., brain contusion, hemorrhage) | Direct injury to the tongue and to taste buds; damage taste nerves | [44] | |
| Surgeries (e.g., removal of third molars, middle ear, head and neck surgeries, general anesthesia) | Partial or complete nerve transection, traction, or stretching | [45,46] | |
| Oral appliances and conditions | Damage to central or peripheral nerves; decreased salivary flow rate | [47,48] | |
| Smoking | Increases respiratory infections and dental problems | [49] | |
| Alcohol consumption | Changes the sensitivity of taste receptors and interferes with the absorption of micronutrients, leading to functional changes in saliva and morphological changes in the taste buds | [50] | |
| Radiation therapies (e.g., in patients with head and neck cancers) | Impair intensity responsiveness, taste recognition and detection thresholds | [51] | |
| Nutrition | Zinc deficiency | Is a component of proteins involved in taste transduction | [52] |
| Specific foods (e.g., Pinus armandii pine nuts) | No potential triggers or common underlying medical causes have been identified yet | [53] | |
| Biology | Sex | Sex steroid hormones may modulate taste processing in the brain | [54] |
| Aging | Reduction in taste buds and papillae density; reduced neural responsiveness to tastes | [55] | |
| Neurological diseases (e.g., dementia, Parkinson syndrome) | Involvement of the frontal cortex; changes in salivary constitution | [56,57] | |
| Viral illness | Common cold (Rhinoviruses), Influenza (Orthomyxoviridae), MERS (MERS-CoV) Hepatitis (Hepatoviruses) | Mostly due to the nasal blockage, obstruction and swelling of the mucosa generated by increased mucus production, and changes in mucus composition; generate inflammation responses in taste tissues | [58,59,60,61] |
| COVID-19 (SARS-CoV-2) | Appears to impair more sweet and bitter tastes; may result from direct infection of cells in the tongue; secondary consequences of obstruction due to inflammation; damage to cranial nerves following the release of inflammatory cytokines; and/or lead to changes in localized cellular zinc homeostasis in oral gustatory cells | [61,62,63,64,65] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Risso, D.; Drayna, D.; Morini, G. Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits. Nutrients 2020, 12, 3284. https://doi.org/10.3390/nu12113284
Risso D, Drayna D, Morini G. Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits. Nutrients. 2020; 12(11):3284. https://doi.org/10.3390/nu12113284
Chicago/Turabian StyleRisso, Davide, Dennis Drayna, and Gabriella Morini. 2020. "Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits" Nutrients 12, no. 11: 3284. https://doi.org/10.3390/nu12113284
APA StyleRisso, D., Drayna, D., & Morini, G. (2020). Alteration, Reduction and Taste Loss: Main Causes and Potential Implications on Dietary Habits. Nutrients, 12(11), 3284. https://doi.org/10.3390/nu12113284







