Effects of a Fat-Rich Diet on the Spontaneous Release of Acetylcholine in the Neuromuscular Junction of Mice
Abstract
1. Introduction
2. Materials and Methods
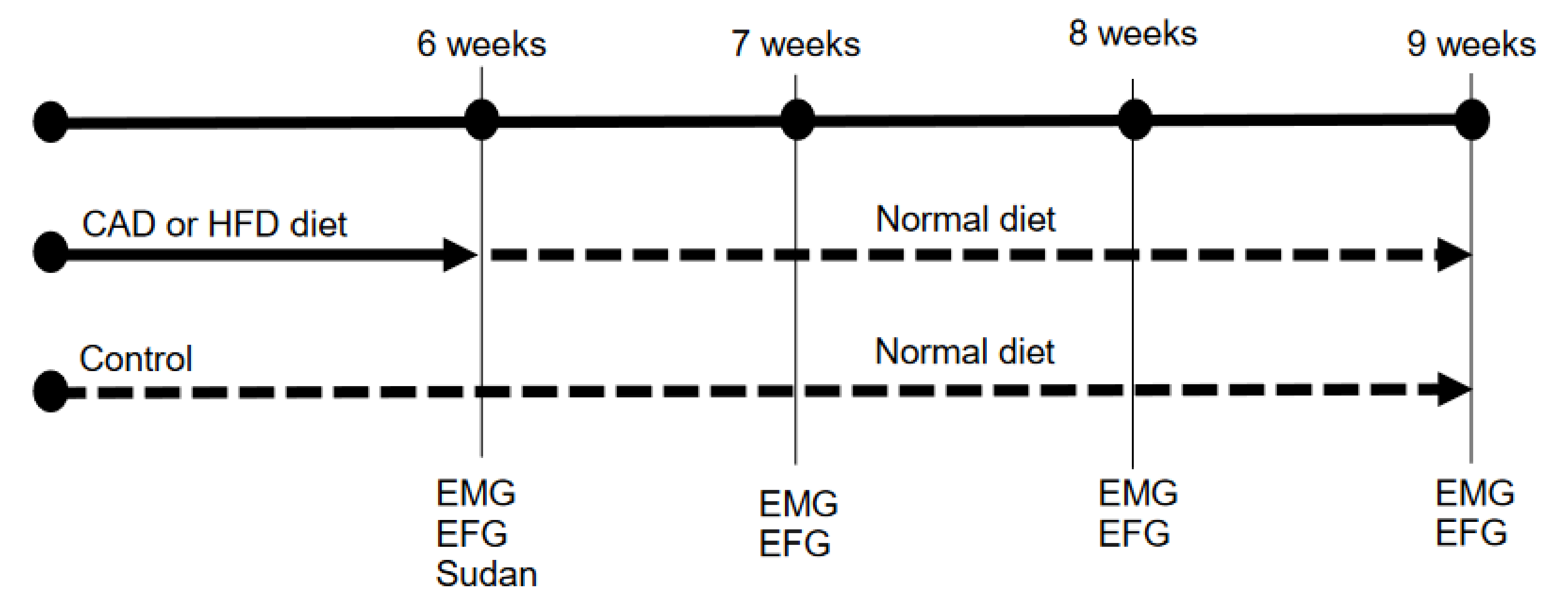
2.1. Animals and Dietary Protocol
2.2. Muscles
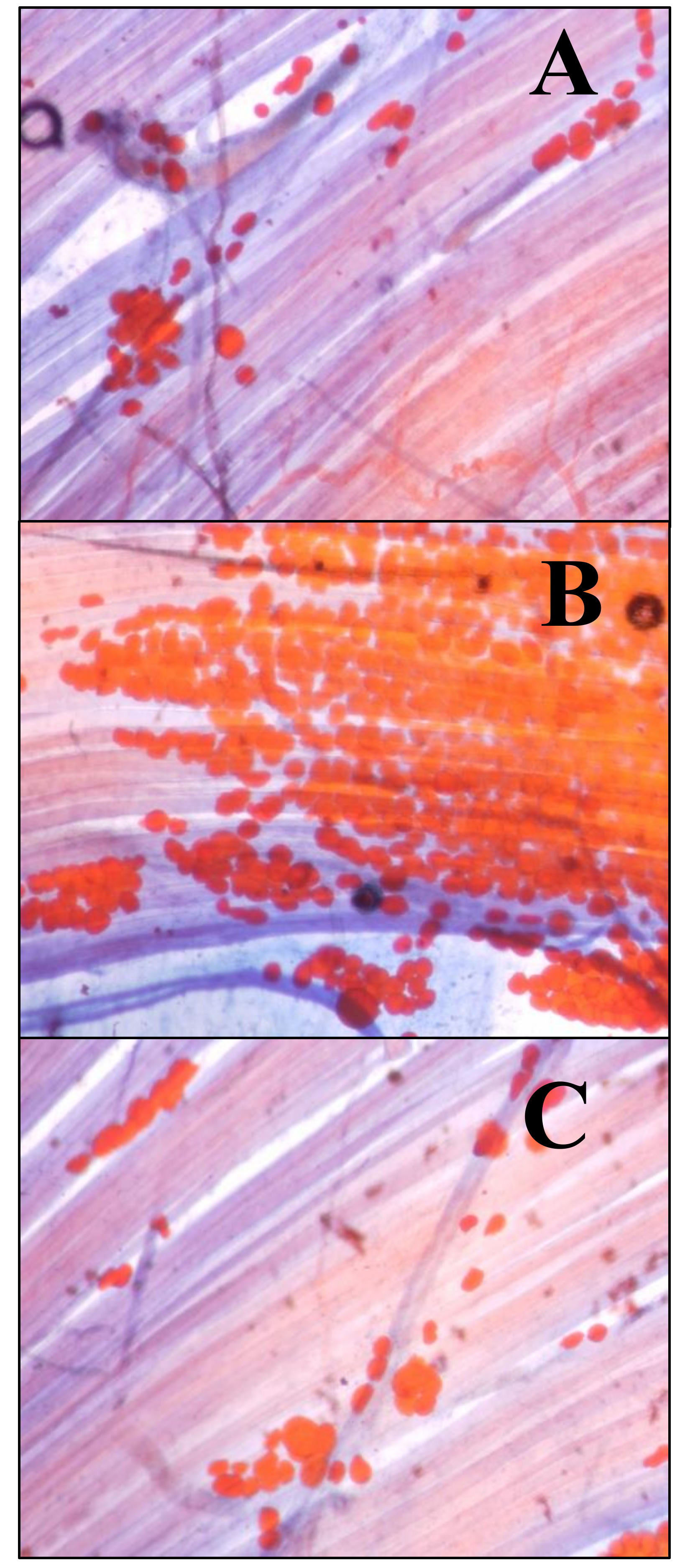
2.3. Sudan III
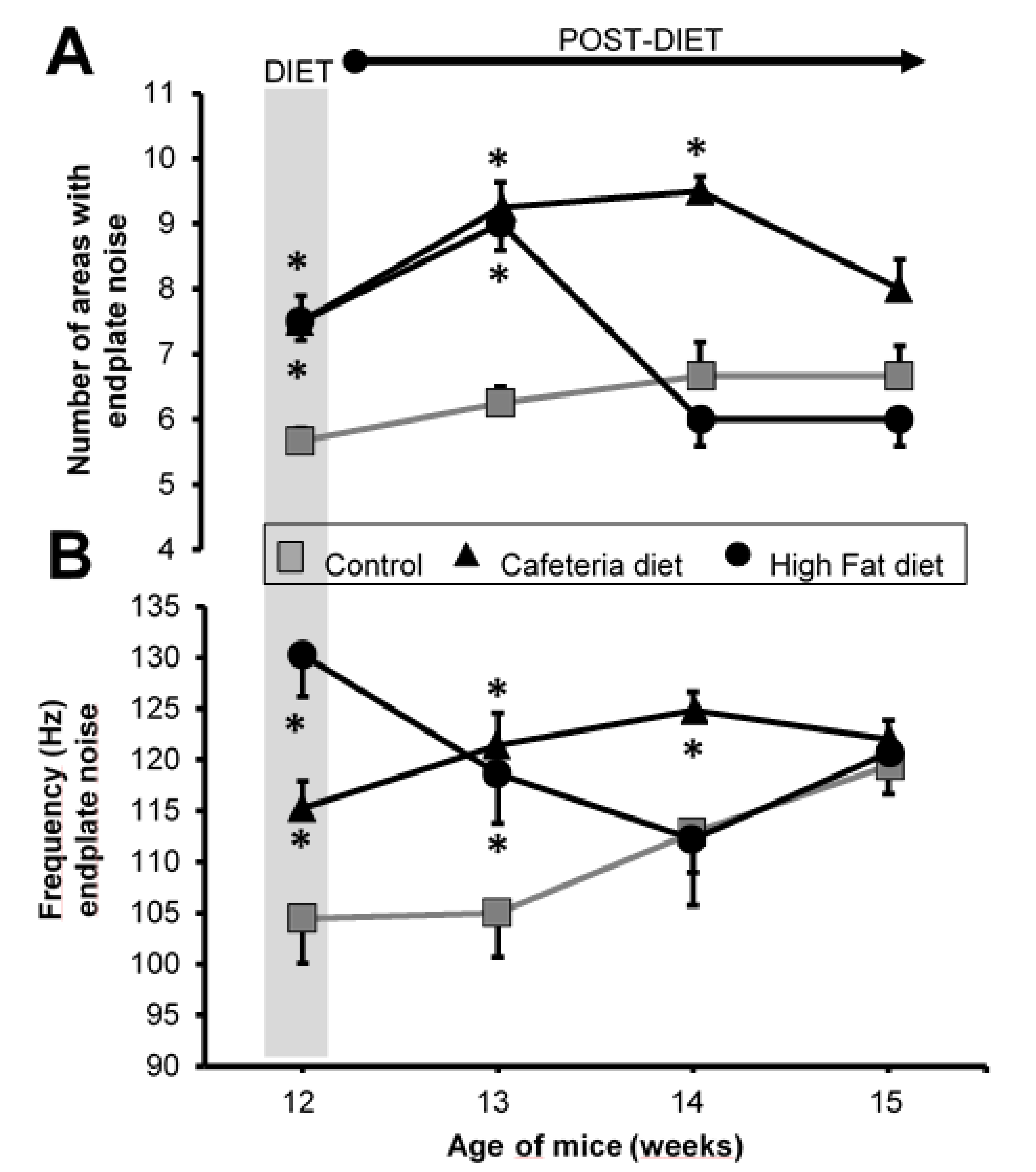
2.4. Endplate Noise Recordings
2.5. Electrophysiology: Intracellular Recordings
2.6. Statistical Procedure
3. Results
3.1. Body Weight Evolution
3.2. Muscle Fat
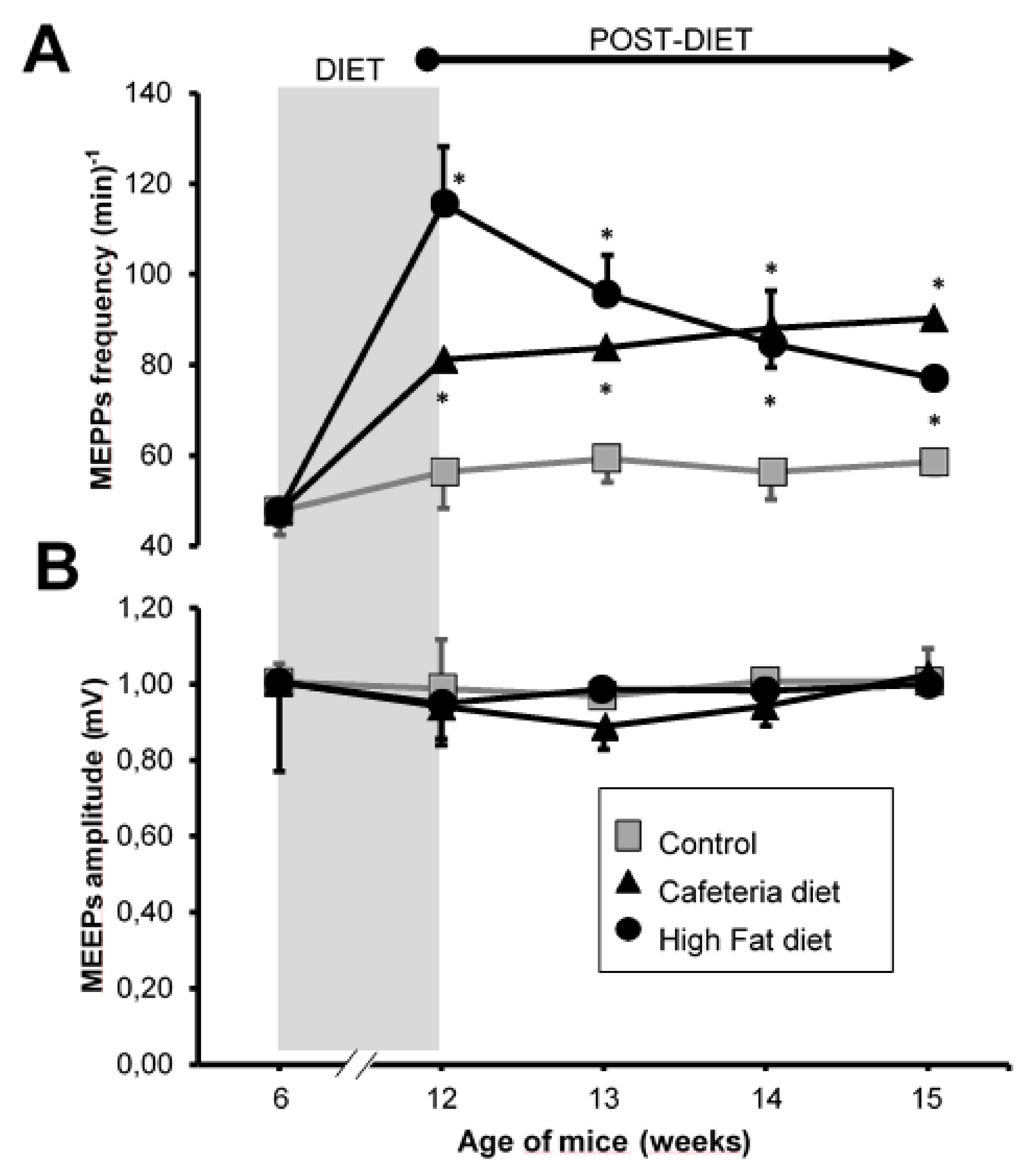
3.3. Electrophysiology: Intracellular Recording
3.4. Electromyography
4. Discussion
4.1. Overweight
4.2. Muscle Fat
4.3. Spontaneous Neurotransmission
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hruby, A.; Hu, F.B. The epidemiology of obesity: A Big Picture. Pharmacoeconomics 2015, 33, 673–689. [Google Scholar] [CrossRef] [PubMed]
- Obesidad y Sobrepeso. Available online: https://www.who.int/es/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 26 July 2020).
- Kelly, T.; Yang, W.; Chen, C.S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Okifuji, A.; Hare, B. The association between chronic pain and obesity. J. Pain Res. 2015, 8, 399. [Google Scholar] [CrossRef]
- Grotle, M.; Hagen, K.B.; Natvig, B.; Dahl, F.A.; Kvien, T.K. Obesity and osteoarthritis in knee, hipand/or hand: An epidemiological study in the general population with years follow-up. BMC Musculoskelet. Disord. 2008, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Luime, J.J.; Kuiper, J.I.; Koes, B.W.; Verhaar, J.A.; Miedema, H.S.; Burdorf, A. Work-related risk factorsfor the incidence and recurrence of shoulder and neck complaints among nursing-home andelderly-care workers. Scand. J. Work Environ. Health 2004, 30, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Bigal, M.E.; Lipton, R.B. Obesity is a risk factor for transformed migraine but not chronictension-type headache. Neurology 2006, 67, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Bigal, M.E. Body Mass Index and Episodic Headaches: A Population-Based Study. Arch. Intern. Med. 2007, 167, 1964–1970. [Google Scholar] [CrossRef]
- Hozumi, J.; Sumitani, M.; Matsubayashi, Y.; Abe, H.; Oshima, Y.; Chikuda, H.; Takeshita, K.; Yamada, Y. Relationship between Neuropathic Pain and Obesity. Pain Res. Manag. 2016, 2016, 2487924. [Google Scholar] [CrossRef]
- Bastard, J.P.; Maachi, M.; Lagathu, C.; Kim, M.J.; Caron, M.; Vidal, H.; Capeau, J.; Feve, B. Recent advances in therelationship between obesity, inflammation, and insulin resistance. Eur. Cytokine Netw. 2006, 17, 4–12. [Google Scholar]
- Gandhi, R.; Perruccio, A.V.; Rizek, R.; Dessouki, O.; Evans, H.M.K.; Mahomed, N.N. Obesity-Related Adipokines Predict Patient-Reported Shoulder Pain. Obes. Facts 2013, 6, 536–541. [Google Scholar] [CrossRef]
- Wu, H.; Ballantyne, C.M. Skeletal muscle inflammation and insulin resistance in obesity. J. Clin. Investig. 2017, 127, 43–54. [Google Scholar] [CrossRef]
- Simons, D.G.; Travell, J.G.; Fernández de las Peñas, C.; Finnegan, M.; Freeman, J.L.; Donnelly, J.M. Travell, Simons & Simons’ Myofascial Pain and Dysfunction: The Trigger Point Manual, 3rd ed.; Wolters Kluwer: Philadelphia, PA, USA, 2019. [Google Scholar]
- Mayoral, O.; Salvat, I. Fisioterapia Invasiva del Síndrome de Dolor Myofascial, 1st ed.; Médica Panamericana: Madrid, Spain, 2017. [Google Scholar]
- Gerwin, R.D.; Dommerholt, J.; Shah, J.P. An expansion of Simons’ integrated hypothesis of trigger point formation. Curr. Pain Headache Rep. 2004, 8, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.P.; Phillips, T.M.; Danoff, J.V.; Gerber, L.H. An in vivo microanalytical technique for measuring the local biochemical milieu of human skeletal muscle. J. Appl. Physiol. 2005, 99, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Margalef, R.; Sisquella, M.; Bosque, M.; Romeu, C.; Mayoral, O.; Monterde, S.; Priego, M.; Guerra-Perez, R.; Ortiz, N.; Tomàs, J.; et al. Experimental myofascial trigger point creation in rodents. J. Appl. Physiol. 2019, 126, 160–169. [Google Scholar] [CrossRef]
- Simons, D.G.; Hong, C.Z.; Simons, L.S. Endplate Potentials Are Common to Midfiber Myofacial Trigger Points. Am. J. Phys. Med. Rehabil. 2002, 81, 212–222. [Google Scholar] [CrossRef]
- Sampey, B.P.; Vanhoose, A.M.; Winfield, H.M.; Freemerman, A.J.; Muehlbauer, M.J.; Fueger, P.T.; Newgard, C.B.; Makowski, L. Cafeteria Diet Is a Robust Model of Human Metabolic Syndrome With Liver and Adipose Inflammation: Comparison to High-Fat Diet. Obesity 2011, 19, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Zeeni, N.; Dagher-Hamalian, C.; Dimassi, H.; Faour, W.H. Cafeteria diet-fed mice is a pertinent model of obesity-induced organ damage: A potential role of inflammation. Inflamm. Res. 2015, 64, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Reynés, B.; García-Ruiz, E.; Díaz-Rúa, R.; Palou, A.; Oliver, P. Reversion to a control balanced diet is able to restore body weight and to recover altered metabolic parameters in adult rats long-term fed on a cafeteria diet. Food Res. Int. 2014, 64, 839–848. [Google Scholar] [CrossRef]
- Lalanza, J.F.; Caimari, A.; del Bas, J.M.; Torregrosa, D.; Cigarroa, I.; Pallàs, M. Effects Of A Post-Weaning Cafeteria Diet In Young Rats: Metabolic Syndrome, Reduced Activity And Low Anxiety-Like Behaviour. PLoS ONE 2014, 9, e85049. [Google Scholar] [CrossRef]
- Rogers, P.J. Returning “cafeteria-fed” rats to a chow diet: Negative contrast and effects of obesity on feeding behaviour. Physiol. Behav. 1985, 35, 493–499. [Google Scholar] [CrossRef]
- Winzell, M.S.; Ahren, B. The High-Fat Diet-Fed Mouse: A Model for Studying Mechanisms and Treatment of Impaired Glucose Tolerance and Type 2 Diabetes. Diabetes 2004, 53, S215–S219. [Google Scholar] [CrossRef] [PubMed]
- Fink, L.N.; Costford, S.R.; Lee, Y.S.; Jensen, T.E.; Bilan, P.J.; Oberbach, A.; Blüher, M.; Olefsky, J.M.; Sams, A.; Klip, A. Pro-Inflammatory macrophages increase in skeletal muscle of high fat-Fed mice and correlate with metabolic risk markers in humans: Muscle Macrophages in Obesity and Diabetes. Obesity 2014, 22, 747–757. [Google Scholar] [CrossRef]
- Song, Z.; Xie, W.; Chen, S.; Strong, J.A.; Print, M.S.; Wang, J.I.; Shareef, A.F.; Ulrich-Lai, Y.M.; Zhang, J.M. High-fat diet increases pain behaviors in rats with or without obesity. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Gasparin, F.R.S.; Carreño, F.O.; Mewes, J.M.; Gilglioni, E.H.; Pagadigorria, C.L.S.; Natali, M.R.M.; Utsunomiya, K.S.; Constantin, R.P.; Ouchida, A.T.; Curti, C.; et al. Sex differences in the development of hepatic steatosis in cafeteria diet-induced obesity in young mice. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 2495–2509. [Google Scholar] [CrossRef] [PubMed]
- McLachlan, E.M.; Martin, A.R. Non-linear summation of end-plate potentials in the frog and mouse. J. Physiol. 1981, 311, 307–324. [Google Scholar] [CrossRef]
- Khan, I.M.; Perrard, X.Y.; Brunner, G.; Lui, H.; Sparks, L.M.; Smith, S.R.; Wang, X.; Shi, Z.Z.; Lewis, D.E.; Wu, H.; et al. Intermuscular and perimuscular fat expansion in obesity correlates with skeletal muscle T cell and macrophage infiltration and insulin resistance. Int. J. Obes. 2015, 39, 1607–1618. [Google Scholar] [CrossRef] [PubMed]
- García, J.; Engelhardt, J.I.; Appel, S.H.; Stefani, E. Increased MEPP frequency as an early sign of experimental immune-mediated motoneuron disease: Early Sign of Experimental MND. Ann. Neurol. 1990, 28, 329–334. [Google Scholar] [CrossRef]
- Smith, M.M.; Minson, C.T. Obesity and adipokines: Effects on sympathetic overactivity: Adipokines and sympathetic outflow. J. Physiol. 2012, 590, 1787–1801. [Google Scholar] [CrossRef]
- Scherrer, U.; Randin, D.; Tappy, L.; Vollenweider, P.; Jéquier, E.; Nicod, P. Body fat and sympathetic nerve activity in healthy subjects. Circulation 1994, 89, 2634–2640. [Google Scholar] [CrossRef]
- Chen, J.T.; Chen, S.M.; Kuan, T.S.; Chung, K.C.; Hong, C.Z. Phentolamine effect on the spontaneous electrical activity of active loci in a myofascial trigger spot of rabbit skeletal muscle. Arch. Phys. Med. Rehabil. 1998, 79, 790–794. [Google Scholar] [CrossRef]
- McNulty, W.H.; Gevirtz, R.N.; Hubbard, D.R.; Berkoff, G.M. Needle electromyographic evaluation of trigger point response to a psychological stressor. Psychophysiology 1994, 31, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Lustrino, D.; Silveira, W.A.; Wild, F.; Straka, T.; Issop, Y.; O’Connor, E.; Cox, D.; Reischl, M.; Marquardt, T.; et al. Sympathetic innervation controls homeostasis of neuromuscular junctions in health and disease. Proc. Natl. Acad. Sci. USA 2016, 113, 746–750. [Google Scholar] [CrossRef] [PubMed]
- Coccurello, R.; Maccarrone, M. Hedonic Eating and the “Delicious Circle”: From Lipid-Derived Mediators to Brain Dopamine and Back. Front. Neurosci. 2018, 12, 271. [Google Scholar] [CrossRef]
- Stice, E.; Figlewicz, D.P.; Gosnell, B.A.; Levine, A.S.; Pratt, W.E. The contribution of brain reward circuits to the obesity epidemic. Neurosci. Biobehav. Rev. 2013, 37, 2047–2058. [Google Scholar] [CrossRef]
- Totsch, S.K.; Waite, M.E.; Sorge, R.E. Dietary Influence on Pain via the Immune System. Prog. Mol. Biol. Transl. Sci. 2015, 131, 435–469. [Google Scholar] [PubMed]
- Totsch, S.K.; Quinn, T.L.; Strath, L.J.; McMeekin, L.J.; Cowell, R.M.; Gower, B.A.; Sorge, R.E. The impact of the Standard American Diet in rats: Effects on behavior, physiology and recovery from inflammatory injury. Scand. J. Pain 2017, 17, 316–324. [Google Scholar] [CrossRef]
- Matikainen-Ankney, B.A.; Kravitz, A.V. Persistent effects of obesity: A neuroplasticity hypothesis: Neuroplasticity hypothesis of obesity. Ann. N. Y. Acad. Sci. 2018, 1428, 221–239. [Google Scholar] [CrossRef]
| CAD | HFD | Regular Chow Diet | |
|---|---|---|---|
| Calories (kcal) | 459 | 532 | 397 |
| Total Fat | 23 | 60.6 | 6.9 |
| Saturated Fat | 11 | 21.7 | - |
| Total Carbohydrate | 56 | 26.3 | 68 |
| Sugars | 24 | 9.7 | - |
| Dietary Fiber | 2.5 | - | - |
| Protein | 5.3 | 13.1 | 25 |
| Sodium | 0.65 | 0.23 | 0.3 |
| Procedure | Age | Control | CAD | HFD |
|---|---|---|---|---|
| 1: 6 weeks of age; 2: 6 weeks with diet | 12 weeks | 38.00 ± 2.85 (n = 20) | 58.03 ± 4.04 * (n = 20) | 55.15 ± 1.39 * (n = 20) |
| 1: 6 weeks of age; 2: 6 weeks with diet; 3: 1 week without diet | 13 weeks | 43.73 ± 0.88 (n = 12) | 45.53 ± 1.03 (n = 12) | 45.11 ± 2.11 (n = 12) |
| 1: 6 weeks of age; 2: 6 weeks with diet; 3: 2 weeks without diet | 14 weeks | 43.24 ± 0.86 (n = 8) | 45.50 ± 0.98 (n = 8) | 43.97 ± 3.09 (n = 8) |
| 1: 6 weeks of age; 2: 6 weeks with diet; 3: 3 weeks without diet | 15 weeks | 42.17 ± 2.85 (n = 4) | 44.67 ± 1.22 (n = 4) | 43.25 ± 2.57 (n = 4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gimenez-Donoso, C.; Bosque, M.; Vila, A.; Vilalta, G.; Santafe, M.M. Effects of a Fat-Rich Diet on the Spontaneous Release of Acetylcholine in the Neuromuscular Junction of Mice. Nutrients 2020, 12, 3216. https://doi.org/10.3390/nu12103216
Gimenez-Donoso C, Bosque M, Vila A, Vilalta G, Santafe MM. Effects of a Fat-Rich Diet on the Spontaneous Release of Acetylcholine in the Neuromuscular Junction of Mice. Nutrients. 2020; 12(10):3216. https://doi.org/10.3390/nu12103216
Chicago/Turabian StyleGimenez-Donoso, Carlos, Marc Bosque, Anna Vila, Gemma Vilalta, and Manel M Santafe. 2020. "Effects of a Fat-Rich Diet on the Spontaneous Release of Acetylcholine in the Neuromuscular Junction of Mice" Nutrients 12, no. 10: 3216. https://doi.org/10.3390/nu12103216
APA StyleGimenez-Donoso, C., Bosque, M., Vila, A., Vilalta, G., & Santafe, M. M. (2020). Effects of a Fat-Rich Diet on the Spontaneous Release of Acetylcholine in the Neuromuscular Junction of Mice. Nutrients, 12(10), 3216. https://doi.org/10.3390/nu12103216






