Dietary Short-Term Fiber Interventions in Arthritis Patients Increase Systemic SCFA Levels and Regulate Inflammation
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Intervention
2.3. Assessment of Safety (Side Effects, Accompanying Medication and Tolerability)
2.4. Data Analysis and Statistics
2.5. SCFA Measurements
2.6. Assessment of Human Cytokine Levels in the Serum
2.7. 16S rRNA Gene Sequencing-Based Microbiome Analysis
2.8. Statistical Analysis
2.9. Data Availability
3. Results
3.1. Patient and Healthy Control Characteristics
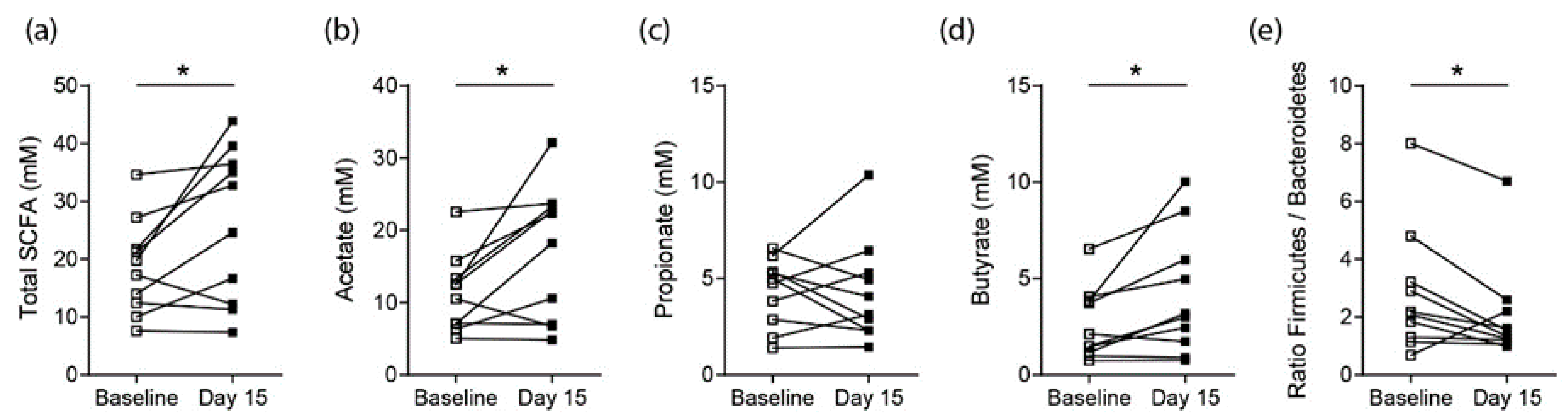
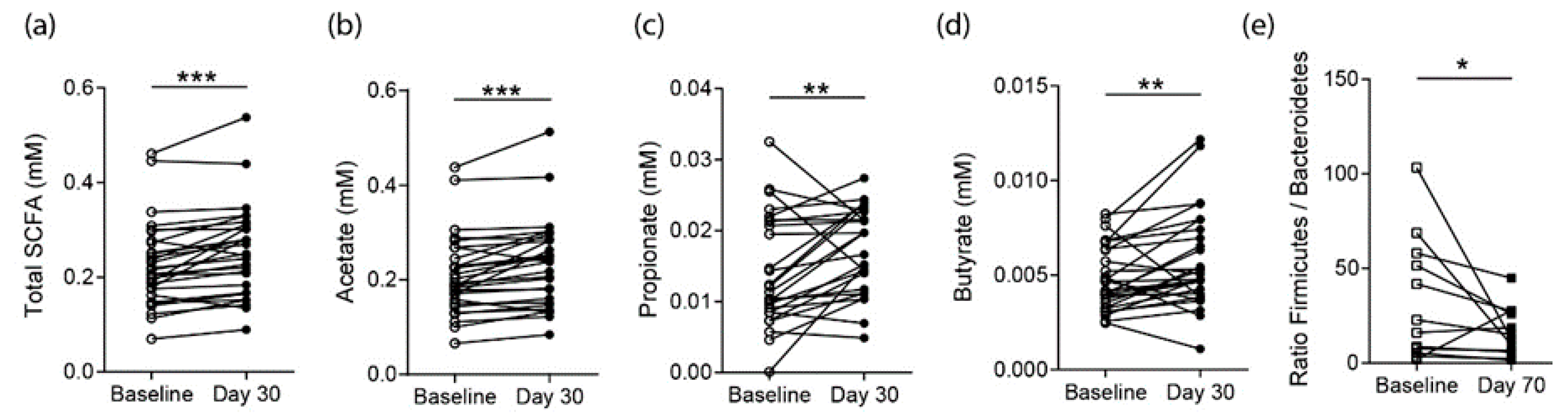
3.2. Effects on SCFA Concentrations in Healthy Controls
3.3. Effects on SCFA Concentrations in RA Patients
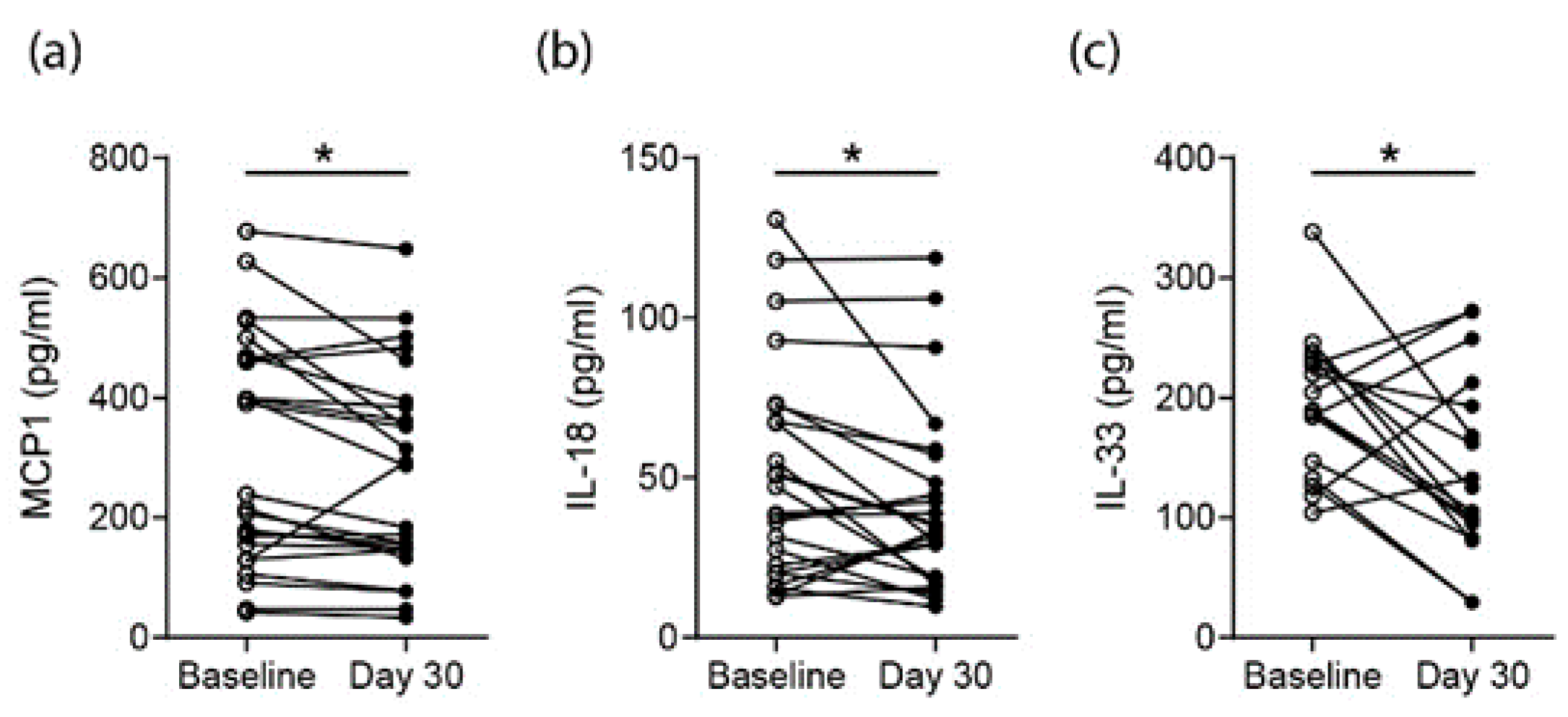
3.4. Effects on Proarthritogenic Cytokine Levels in RA Patients
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McInnes, I.B.; Schett, G. The Pathogenesis of rheumatoid arthritis. N. Engl. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef]
- McInnes, I.B.; Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat. Rev. Immunol. 2007, 7, 429–442. [Google Scholar] [CrossRef]
- Zaiss, M.M.; Jones, R.M.; Schett, G.; Pacifici, R. The gut-bone axis: How bacterial metabolites bridge the distance. J. Clin. Investig. 2019, 129, 3018–3028. [Google Scholar] [CrossRef]
- Vinolo, M.A.; Rodrigues, H.G.; Nachbar, R.T.; Curi, R. Regulation of inflammation by short chain fatty acids. Nutrients 2011, 3, 858–876. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van Der Veeken, J.; DeRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef] [PubMed]
- McInnes, I.B.; Liew, F.Y.; Gracie, J.A. Interleukin-18: A therapeutic target in rheumatoid arthritis? Arthritis Res. Ther. 2004, 7, 38–41. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Spoettl, T.; Hausmann, M.; Herlyn, M.; Gunckel, M.; Dirmeier, A.; Falk, W.; Herfarth, H.; Schoelmerich, J.; Rogler, G. Monocyte chemoattractant protein-1 (MCP-1) inhibits the intestinal-like differentiation of monocytes. Clin. Exp. Immunol. 2006, 145, 190–199. [Google Scholar] [CrossRef]
- Li, X.; Li, X.; Lu, W.-G.; Li, C.-W.; Xu, R.; Dong, J. IL-33: A promising therapeutic target for rheumatoid arthritis? Expert Opin. Ther. Targets 2011, 15, 529–534. [Google Scholar]
- Robert, C. Edgar, Henrik Flyvbjerg, Error filtering, pair assembly and error correction for next-generation sequencing reads. Bioinformatics 2015, 31, 3476–3482. [Google Scholar]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. S1), 4516–4522. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Chang, H.-W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Hao Zhu, T.; et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 1–17. [Google Scholar] [CrossRef]
- Koikkalainen, M.; Mykkãnen, H.; Erkkila, A.; Julkunen, J.; Saarinen, T.; Pyorala, K.; Uusitupa, M.; Lappalainen, R. Difficulties in changing the diet in relation to dietary fat intake among patients with coronary heart disease. Eur. J. Clin. Nutr. 1999, 53, 120–125. [Google Scholar] [CrossRef]
- Spring, B.; Schneider, K.; McFadden, H.G.; Vaughn, J.; Kozak, A.T.; Smith, M.; Moller, A.C.; Epstein, L.H.; DeMott, A.; Hedeker, D.; et al. Multiple behavior changes in diet and activity: A randomized controlled trial using mobile technology. Arch. Intern. Med. 2012, 172, 789–796. [Google Scholar] [CrossRef]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The firmicutes/bacteroidetes ratio: A relevant marker of gut dysbiosis in obese patients? Nutrients 2020, 12, 1474. [Google Scholar] [CrossRef]
- Astakhova, L.; Ngara, M.; Babich, O.; Prosekov, A.; Asyakina, L.; Dyshlyuk, L.; Midtvedt, T.; Zhou, X.; Ernberg, I.; Matskova, L. Short Chain Fatty Acids (SCFA) reprogram gene expression in human malignant epithelial and lymphoid cells. PLoS ONE 2016, 11, e0154102. [Google Scholar] [CrossRef]
- Nastasi, C.; Candela, M.; Bonefeld, C.M.; Geisler, C.; Hansen, M.; Krejsgaard, T.; Biagi, E.; Andersen, M.H.; Brigidi, P.; Ødum, N. The effect of short-chain fatty acids on human monocyte-derived dendritic cells. Sci. Rep. 2015, 5, 16148. [Google Scholar] [CrossRef]
- Peng, L.; Li, Z.-R.; Green, R.S.; Holzman, I.R.; Lin, J. Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers. J. Nutr. 2009, 139, 1619–1625. [Google Scholar] [CrossRef]
- Vinolo, M.A.R.; Ferguson, G.J.; Kulkarni, S.; Damoulakis, G.; Anderson, K.; Bohlooly-Y, M.; Stephens, L.; Hawkins, P.T.; Curi, R. SCFAs induce mouse neutrophil chemotaxis through the GPR43 receptor. PLoS ONE 2011, 6, e21205. [Google Scholar] [CrossRef]
- Haghikia, A.; Jörg, S.; Duscha, A.; Kaisler, J.; Manzel, A.; Waschbisch, A.; Hammer, A.; Lee, H.; May, C.; Wilck, N.; et al. Dietary fatty acids directly impact central nervous system autoimmunity via the small intestine. Immunity 2015, 43, 817–829. [Google Scholar] [CrossRef]
- Lucas, S.; Omata, Y.; Hofmann, J.; Böttcher, M.; Iljazovic, A.; Sarter, K.; Albrecht, O.; Schulz, O.; Krishnacoumar, B.; Krönke, G.; et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Porporato, P.; Dewulf, E.; Verrax, J.; Neyrinck, A.M.; Martin, J.C.; Scott, K.P.; Calderon, P.B.; Feron, O.; Muccioli, G.G.; et al. Gut microbiota-derived propionate reduces cancer cell proliferation in the liver. Br. J. Cancer 2012, 107, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Blottiere, H.M.; Buecher, B.; Galmiche, J.-P.; Cherbut, C. Molecular analysis of the effect of short-chain fatty acids on intestinal cell proliferation. Proc. Nutr. Soc. 2003, 62, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Siavoshian, S.; Segain, J.-P.; Kornprobst, M.; Bonnet, C.; Cherbut, C.; Galmiche, J.-P.; Blottiere, H. Butyrate and trichostatin A effects on the proliferation/differentiation of human intestinal epithelial cells: Induction of cyclin D3 and p21 expression. Gut 2000, 46, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Matthews, G.M.; Howarth, G.S.; Butler, R.N. Short-chain fatty acids induce apoptosis in colon cancer cells associated with changes to intracellular redox state and glucose metabolism. Chemotherapy 2012, 58, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Chen, Y.; Jiang, H.; Nie, D. Short-chain fatty acids induced autophagy serves as an adaptive strategy for retarding mitochondria-mediated apoptotic cell death. Cell Death Differ. 2011, 18, 602–618. [Google Scholar] [CrossRef]
- Fellows, R.; Denizot, J.; Stellato, C.; Cuomo, A.; Jain, P.; Stoyanova, E.; Balázi, S.; Hajnády, Z.; Liebert, A.; Kazakevych, J.; et al. Microbiota derived short chain fatty acids promote histone crotonylation in the colon through histone deacetylases. Nat. Commun. 2018, 9, 105. [Google Scholar] [CrossRef]
- Waldecker, M.; Kautenburger, T.; Daumann, H.; Busch, C.; Schrenk, D. Inhibition of histone-deacetylase activity by short-chain fatty acids and some polyphenol metabolites formed in the colon. J. Nutr. Biochem. 2008, 19, 587–593. [Google Scholar] [CrossRef]
- Yuille, S.; Reichardt, N.; Panda, S.; Dunbar, H.; Mulder, I.E. Human gut bacteria as potent class I histone deacetylase inhibitors in vitro through production of butyric acid and valeric acid. PLoS ONE 2018, 13, e0201073. [Google Scholar] [CrossRef]
- Tajik, N.; Frech, M.; Schulz, O.; Schälter, F.; Lucas, S.; Azizov, V.; Dürholz, K.; Steffen, F.; Omata, Y.; Rings, A.; et al. Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Tyagi, A.M.; Yu, M.; Darby, T.M.; Vaccaro, C.; Li, J.-Y.; Owens, J.A.; Hsu, E.; Adams, J.; Weitzmann, M.N.; Jones, R.M.; et al. The microbial metabolite butyrate stimulates bone formation via t regulatory cell-mediated regulation of WNT10B expression. Immunity 2018, 49, 1116–1131. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, M.; Noto, D.; Kaga, N.; Chiba, A.; Miyake, S. The dual role of short fatty acid chains in the pathogenesis of autoimmune disease models. PLoS ONE 2017, 12, e0173032. [Google Scholar] [CrossRef] [PubMed]
- Rosser, E.C.; Piper, C.J.; Matei, D.E.; Blair, P.A.; Rendeiro, A.F.; Orford, M.; Alber, D.G.; Krausgruber, T.; Catalan, D.; Klein, N.; et al. Microbiota-derived metabolites suppress arthritis by amplifying aryl-hydrocarbon receptor activation in regulatory B Cells. Cell Metab. 2020, 31, 837–851. [Google Scholar] [CrossRef]
- Häger, J.; Bang, H.; Hagen, M.; Frech, M.; Träger, P.; Sokolova, M.V.; Steffen, U.; Tascilar, K.; Sarter, K.; Schett, G.; et al. The role of dietary fiber in rheumatoid arthritis patients: A Feasibility study. Nutrients 2019, 11, 2392. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef]
- Ip, W.K.; Wong, C.K.; Lam, C.W.K. Interleukin (IL)-4 and IL-13 up-regulate monocyte chemoattractant protein-1 expression in human bronchial epithelial cells: Involvement of p38 mitogen-activated protein kinase, extracellular signal-regulated kinase 1/2 and Janus kinase-2 but not c-Jun NH2-terminal kinase 1/2 signalling pathways. Clin. Exp. Immunol. 2006, 145, 162–172. [Google Scholar]
- Ellingsen, T.; Elling, P.; Olson, A.; Elling, H.; Baandrup, U.; Matsushima, K.; Deleuran, B.; Stengaard-Pedersen, K. Monocyte chemoattractant protein 1 (MCP-1) in temporal arteritis and polymyalgia rheumatica. Ann. Rheum. Dis. 2000, 59, 775–780. [Google Scholar] [CrossRef]
- Harigai, M.; Hara, M.; Yoshimura, T.; Leonard, E.J.; Inoue, K.; Kashiwazaki, S. Monocyte Chemoattractant Protein-1 (MCP-1) in inflammatory joint diseases and its involvement in the cytokine network of rheumatoid synovium. Clin. Immunol. Immunopathol. 1993, 69, 83–91. [Google Scholar] [CrossRef]
- Koch, A.; Kunkel, S.L.; A Harlow, L.; Johnson, B.; Evanoff, H.L.; Haines, G.K.; Burdick, M.D.; Pope, R.M.; Strieter, R.M. Enhanced production of monocyte chemoattractant protein-1 in rheumatoid arthritis. J. Clin. Investig. 1992, 90, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Stankovic, A.; Slavic, V.; Stamenkovic, B.; Kamenov, B.; Bojanovic, M.; Mitrovic, D.R. Serum and synovial fluid concentrations of CCL2 (MCP-1) chemokine in patients suffering rheumatoid arthritis and osteoarthritis reflect disease activity. Bratisl. Lek. List. 2009, 110, 641–646. [Google Scholar]
- Gong, J.-H.; Ratkay, L.G.; Waterfield, J.D.; Clark-Lewis, I. An antagonist of monocyte chemoattractant protein 1 (MCP-1) Inhibits arthritis in the MRL-lpr mouse model. J. Exp. Med. 1997, 186, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Cox, M.A.; Jackson, J.; Stanton, M.; Rojas-Triana, A.; Bober, L.; Laverty, M.; Yang, X.; Zhu, F.; Liu, J.; Wang, S.; et al. Short-chain fatty acids act as antiinflammatory mediators by regulating prostaglandin E(2) and cytokines. World J. Gastroenterol. 2009, 15, 5549–5557. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Mikami, D.; Kimura, H.; Kamiyama, K.; Morikawa, Y.; Yokoi, S.; Kasuno, K.; Takahashi, N.; Taniguchi, T.; Iwano, M. Short-chain fatty acids, GPR41 and GPR43 ligands, inhibit TNF-α-induced MCP-1 expression by modulating p38 and JNK signaling pathways in human renal cortical epithelial cells. Biochem. Biophys. Res. Commun. 2017, 486, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Dayer, J.-M. Interleukin-18, rheumatoid arthritis, and tissue destruction. J. Clin. Investig. 1999, 104, 1337–1339. [Google Scholar] [CrossRef] [PubMed]
- Dai, S.-M.; Shan, Z.-Z.; Xu, H.; Nishioka, K. Cellular targets of interleukin-18 in rheumatoid arthritis. Ann. Rheum. Dis. 2007, 66, 1411–1418. [Google Scholar] [CrossRef]
- Gracie, J.A. Interleukin-18 as a potential target in inflammatory arthritis. Clin. Exp. Immunol. 2004, 136, 402–404. [Google Scholar] [CrossRef]
- Yasin, S.; Fall, N.; Brown, R.; Henderlight, M.; Canna, S.W.; Girard-Guyonvarc’H, C.; Gabay, C.; Grom, A.A.; Schulert, G.S. IL-18 as a biomarker linking systemic juvenile idiopathic arthritis and macrophage activation syndrome. Rheumatology 2020, 59, 361–366. [Google Scholar] [CrossRef]
- Leung, B.P.; McInnes, I.B.; Esfandiari, E.; Wei, X.-Q.; Liew, F.Y. Combined effects of IL-12 and IL-18 on the induction of collagen-induced arthritis. J. Immunol. 2000, 164, 6495–6502. [Google Scholar] [CrossRef]
- Wei, X.-Q.; Leung, B.P.; Arthur, H.M.L.; McInnes, I.B.; Liew, F.Y. Reduced incidence and severity of collagen-induced arthritis in mice lacking IL-18. J. Immunol. 2001, 166, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Jiang, H.-R.; Kewin, P.; Li, Y.; Mu, R.; Fraser, A.R.; Pitman, N.; Kurowska-Stolarska, M.; McKenzie, A.N.J.; McInnes, I.B.; et al. IL-33 exacerbates antigen-induced arthritis by activating mast cells. Proc. Natl. Acad. Sci. USA 2008, 105, 10913–10918. [Google Scholar] [CrossRef]
- Xu, D.; Jiang, H.-R.; Li, Y.; Pushparaj, P.N.; Kurowska-Stolarska, M.; Leung, B.P.; Mu, R.; Tay, H.K.; McKenzie, A.N.J.; McInnes, I.B.; et al. IL-33 exacerbates autoantibody-induced arthritis. J. Immunol. 2010, 184, 2620–2626. [Google Scholar] [CrossRef] [PubMed]
- Schulze, J.; Bickert, T.; Beil, F.T.; Zaiss, M.M.; Albers, J.; Wintges, K.; Streichert, T.; Klaetschke, K.; Keller, J.; Hissnauer, T.N.; et al. Interleukin-33 is expressed in differentiated osteoblasts and blocks osteoclast formation from bone marrow precursor cells. J. Bone Miner. Res. 2011, 26, 704–717. [Google Scholar] [CrossRef]
- Zaiss, M.M.; Kurowska-Stolarska, M.; Böhm, C.; Gary, R.; Scholtysek, C.; Stolarski, B.; Reilly, J.; Kerr, S.; Millar, N.L.; Kamradt, T.; et al. IL-33 shifts the balance from osteoclast to alternatively activated macrophage differentiation and protects from TNF-α-mediated bone loss. J. Immunol. 2011, 186, 6097–6105. [Google Scholar] [CrossRef]
| Demographic Characteristics (N = 10) | ||
|---|---|---|
| Age, years (mean ± SD) | 28 ± 4.87 | |
| BMI, units (mean ± SD) | 22.81 ± 1.66 | |
| Females, N | 0 | 0% |
| Ever smoker, N | 2 | 20% |
| Patient characteristics (N = 36) | ||
| Age, years (mean ± SD) | 56.19 ± 7.7 | |
| BMI, units (mean ± SD) | 26.63 ± 6.4 | |
| Females, N | 20 | 64.51% |
| Ever smoker, N | 6 | 19.35% |
| RA-specific data (N = 36) | ||
| Time since disease onset, years (mean ± SD) | 11.64 ± 9.39 | |
| Disease activity score (DAS) 28, units (mean ± SD) | 2.54 ± 0.28 | |
| Anti-CCP-IgG antibody positive, N | 15 | 48.38% |
| Rheumatoid Factor IgM positive, N | 12 | 38.7% |
| Accompanying treatment (N = 36) | ||
| Biological disease-modifying anti-rheumatic drugs, N | 23 | 74.10% |
| Abatacept, N | 3 | 9.67% |
| Rituximab, N | 7 | 22.58% |
| Tocilizumab, N | 8 | 25.8% |
| Tumor Necrosis Factor Inhibitors, N | 5 | 16.12% |
| Glucocorticoids, N | 5 | 16.12% |
| JAK-inhibitors, N | 4 | 12.9% |
| Methotrexate, N | 16 | 51.61% |
| Other conventional synthetic disease-modifying anti-rheumatic drugs, N | 1 | 3.22% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dürholz, K.; Hofmann, J.; Iljazovic, A.; Häger, J.; Lucas, S.; Sarter, K.; Strowig, T.; Bang, H.; Rech, J.; Schett, G.; et al. Dietary Short-Term Fiber Interventions in Arthritis Patients Increase Systemic SCFA Levels and Regulate Inflammation. Nutrients 2020, 12, 3207. https://doi.org/10.3390/nu12103207
Dürholz K, Hofmann J, Iljazovic A, Häger J, Lucas S, Sarter K, Strowig T, Bang H, Rech J, Schett G, et al. Dietary Short-Term Fiber Interventions in Arthritis Patients Increase Systemic SCFA Levels and Regulate Inflammation. Nutrients. 2020; 12(10):3207. https://doi.org/10.3390/nu12103207
Chicago/Turabian StyleDürholz, Kerstin, Jörg Hofmann, Aida Iljazovic, Julian Häger, Sébastien Lucas, Kerstin Sarter, Till Strowig, Holger Bang, Jürgen Rech, Georg Schett, and et al. 2020. "Dietary Short-Term Fiber Interventions in Arthritis Patients Increase Systemic SCFA Levels and Regulate Inflammation" Nutrients 12, no. 10: 3207. https://doi.org/10.3390/nu12103207
APA StyleDürholz, K., Hofmann, J., Iljazovic, A., Häger, J., Lucas, S., Sarter, K., Strowig, T., Bang, H., Rech, J., Schett, G., & Zaiss, M. M. (2020). Dietary Short-Term Fiber Interventions in Arthritis Patients Increase Systemic SCFA Levels and Regulate Inflammation. Nutrients, 12(10), 3207. https://doi.org/10.3390/nu12103207







