Vitamin D Status Impacts Genital Mucosal Immunity and Markers of HIV-1 Susceptibility in Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Study
2.2. Measurement of Serum and Cervicovaginal 25(OH)D
2.3. Cervicovaginal Lavage Collection
2.4. Anti-HIV Activity of the Cervicovaginal Lavage
2.5. Soluble Mucosal Proteins in CV Secretions
2.6. Analysis of Vaginal Immune Cell Populations and Histology
2.7. HIV-1 p24 Antigen Production from Ectocervical Tissues after Ex Vivo HIV-1 Infection
2.8. RNA Isolation
2.9. Quantitative Real-Time PCR (qPCR)
2.10. Statistical Analysis
3. Results
3.1. Demographic Data
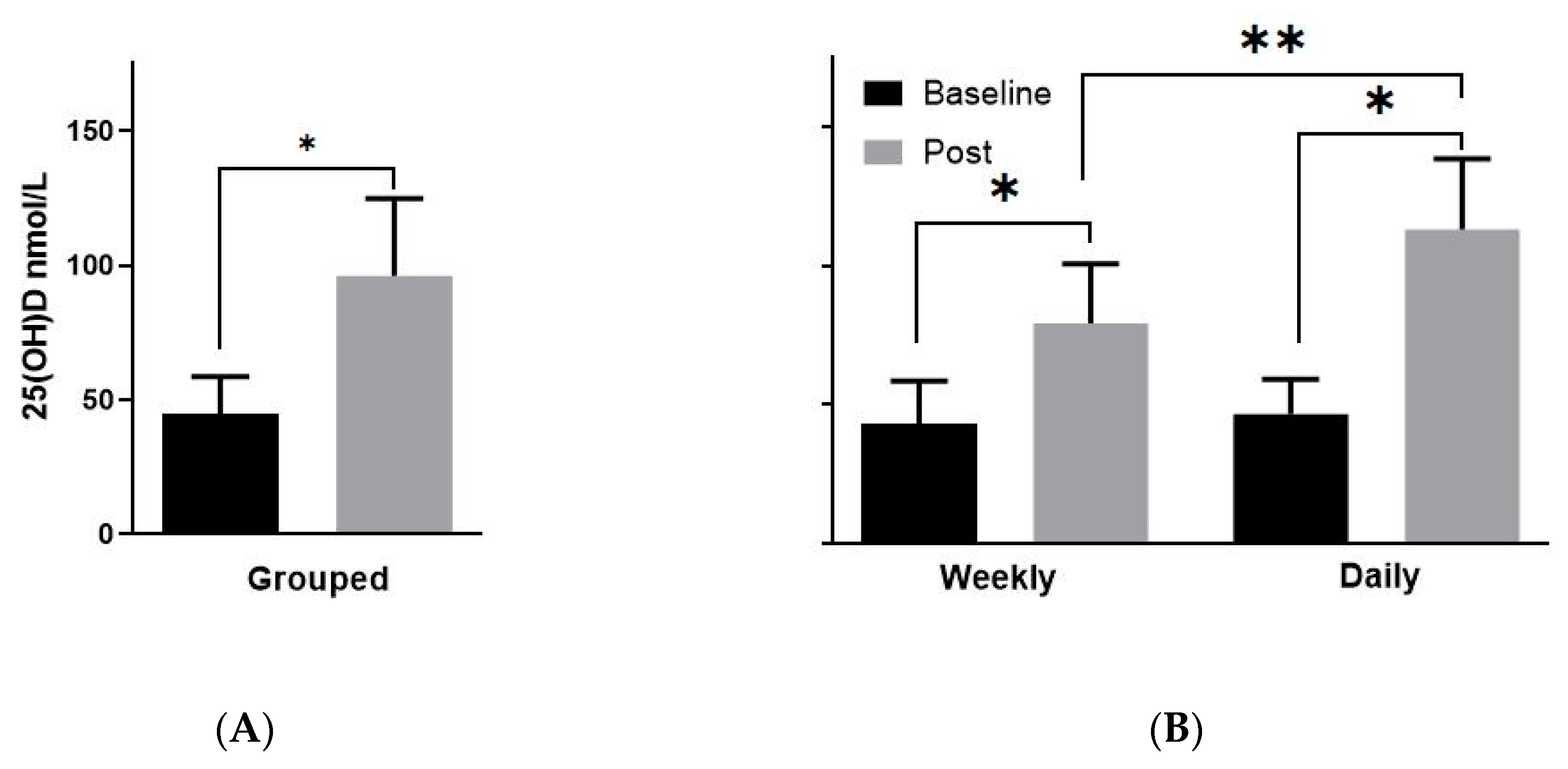
3.2. Vitamin D Levels Post-Supplementation
3.3. Vitamin D Supplementation Alters Cervicovaginal Immune Cell Expression
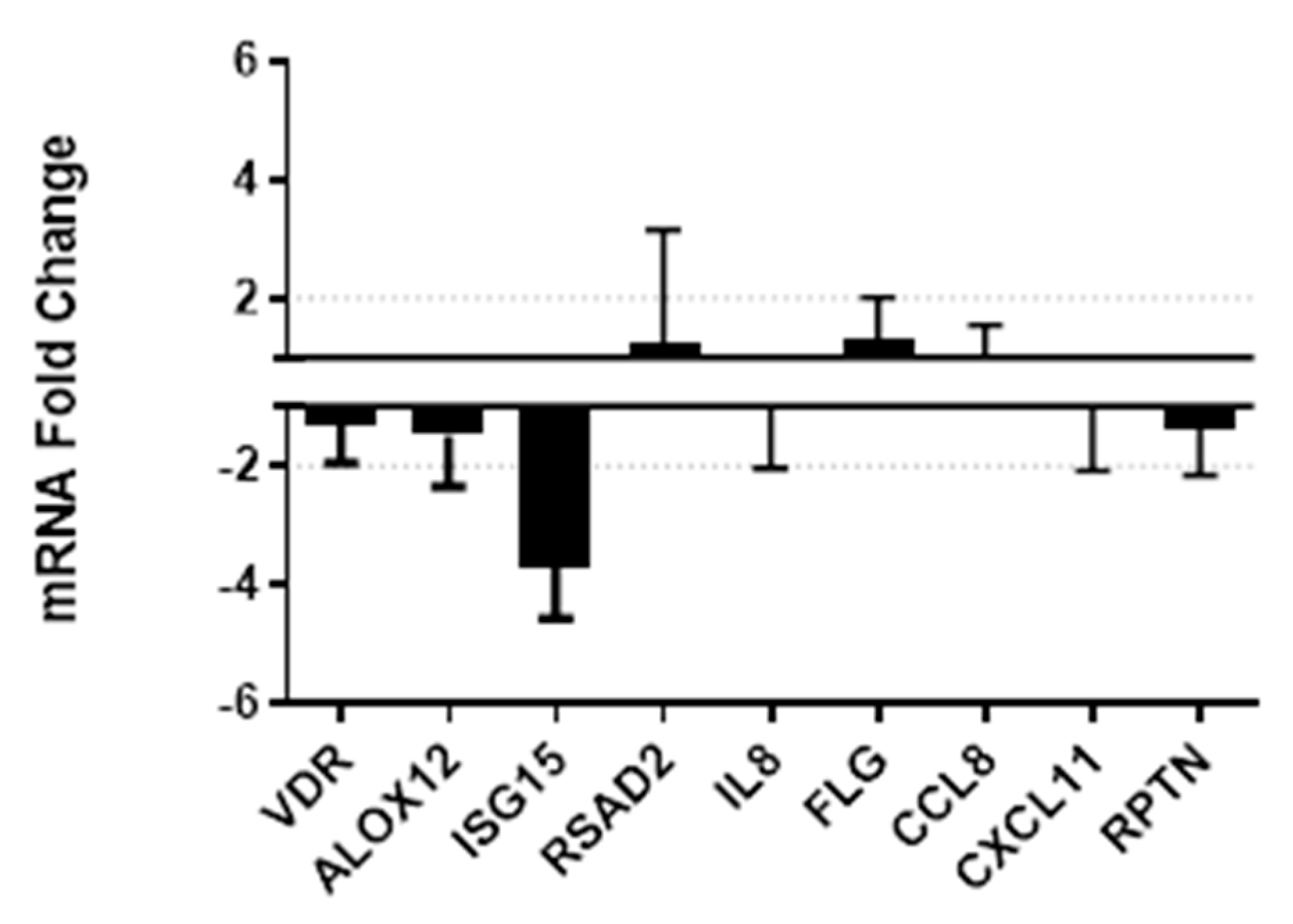
3.4. Vitamin D Supplementation Effects on Gene Expression and Secretion of Innate Immune Factors
3.5. Inhibition of HIV-1 by CVL Fluid Following Vitamin D Supplementation
3.6. Vitamin D Supplementation and Susceptibility of Cervical Tissues to Ex Vivo HIV Infection
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef]
- Grant, W.B. An estimate of the global reduction in mortality rates through doubling vitamin D levels. Eur. J. Clin. Nutr. 2011, 65, 1016–1026. [Google Scholar] [CrossRef]
- Bikle, D.D. Vitamin D regulation of immune function. Vitam. Horm. 2011, 86, 1–21. [Google Scholar] [CrossRef]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Aguilar-Jimenez, W.; Zapata, W.; Caruz, A.; Rugeles, M.T. High transcript levels of vitamin D receptor are correlated with higher mRNA expression of human beta defensins and IL-10 in mucosa of HIV-1-exposed seronegative individuals. PLoS ONE 2013, 8, e82717. [Google Scholar] [CrossRef]
- Bakdash, G.; Schneider, L.P.; Van Capel, T.M.; Kapsenberg, M.L.; Teunissen, M.B.; De Jong, E.C. Intradermal application of vitamin D3 increases migration of CD14+ dermal dendritic cells and promotes the development of Foxp3+ regulatory T cells. Hum. Vaccines Immunother. 2013, 9, 250–258. [Google Scholar] [CrossRef]
- Von Essen, M.R.; Kongsbak, M.; Schjerling, P.; Olgaard, K.; Odum, N.; Geisler, C. Vitamin D controls T cell antigen receptor signaling and activation of human T cells. Nat. Immunol. 2010, 11, 344–349. [Google Scholar] [CrossRef]
- Cantorna, M.T.; Waddell, A. The vitamin D receptor turns off chronically activated T cells. Ann. N. Y. Acad. Sci. 2014, 1317, 70–75. [Google Scholar] [CrossRef]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef]
- Beard, J.A.; Bearden, A.; Striker, R. Vitamin D and the anti-viral state. J. Clin. Virol 2011, 50, 194–200. [Google Scholar] [CrossRef]
- Fabri, M.; Stenger, S.; Shin, D.M.; Yuk, J.M.; Liu, P.T.; Realegeno, S.; Lee, H.M.; Krutzik, S.R.; Schenk, M.; Sieling, P.A.; et al. Vitamin D is required for IFN-gamma-mediated antimicrobial activity of human macrophages. Sci. Transl. Med. 2011, 3, 104ra102. [Google Scholar] [CrossRef]
- Wang, S. Epidemiology of vitamin D in health and disease. Nutr. Res. Rev. 2009, 22, 188–203. [Google Scholar] [CrossRef]
- Grant, W.B. Epidemiology of disease risks in relation to vitamin D insufficiency. Prog. Biophys. Mol. Biol. 2006, 92, 65–79. [Google Scholar] [CrossRef]
- Adeyemi, O.M.; Agniel, D.; French, A.L.; Tien, P.C.; Weber, K.; Glesby, M.J.; Villacres, M.C.; Sharma, A.; Merenstein, D.; Golub, E.T.; et al. Vitamin D deficiency in HIV-infected and HIV-uninfected women in the United States. J. Acquir. Immune Defic. Syndr. 2011, 57, 197–204. [Google Scholar] [CrossRef]
- Eckard, A.R.; Leong, T.; Avery, A.; Castillo, M.D.; Bonilla, H.; Storer, N.; Labbato, D.; Khaitan, A.; Tangpricha, V.; McComsey, G.A. Short communication: High prevalence of vitamin D deficiency in HIV-infected and HIV-uninfected pregnant women. AIDS Res. Hum. Retrovir. 2013, 29, 1224–1228. [Google Scholar] [CrossRef]
- Pinzone, M.R.; Di Rosa, M.; Malaguarnera, M.; Madeddu, G.; Foca, E.; Ceccarelli, G.; d’Ettorre, G.; Vullo, V.; Fisichella, R.; Cacopardo, B.; et al. Vitamin D deficiency in HIV infection: An underestimated and undertreated epidemic. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1218–1232. [Google Scholar]
- Lachmann, R.; Bevan, M.A.; Kim, S.; Patel, N.; Hawrylowicz, C.; Vyakarnam, A.; Peters, B.S. A comparative phase 1 clinical trial to identify anti-infective mechanisms of vitamin D in people with HIV infection. AIDS 2015, 29, 1127–1135. [Google Scholar] [CrossRef]
- Alvarez, N.; Aguilar-Jimenez, W.; Rugeles, M.T. The Potential Protective Role of Vitamin D Supplementation on HIV-1 Infection. Front. Immunol. 2019, 10, 2291. [Google Scholar] [CrossRef]
- Hewison, M. An update on vitamin D and human immunity. Clin. Endocrinol. 2012, 76, 315–325. [Google Scholar] [CrossRef]
- Diaz, L.; Noyola-Martinez, N.; Barrera, D.; Hernandez, G.; Avila, E.; Halhali, A.; Larrea, F. Calcitriol inhibits TNF-alpha-induced inflammatory cytokines in human trophoblasts. J. Reprod. Immunol. 2009, 81, 17–24. [Google Scholar] [CrossRef]
- Akoh, C.C.; Pressman, E.K.; Cooper, E.; Queenan, R.A.; Pillittere, J.; O’Brien, K.O. Low Vitamin D is Associated with Infections and Proinflammatory Cytokines During Pregnancy. Reprod. Sci. 2018, 25, 414–423. [Google Scholar] [CrossRef]
- Bikle, D.; Teichert, A.; Hawker, N.; Xie, Z.; Oda, Y. Sequential regulation of keratinocyte differentiation by 1,25(OH)2D3, VDR, and its coregulators. J. Steroid Biochem. Mol. Biol. 2007, 103, 396–404. [Google Scholar] [CrossRef]
- Yildirim, B.; Abban, G.; Erdogan, B.S. Immunohistochemical detection of 1,25-dihydroxyvitamin D receptor in rat vaginal epithelium. Fertil. Steril. 2004, 82, 1602–1608. [Google Scholar] [CrossRef]
- Abban, G.; Yildirim, N.B.; Jetten, A.M. Regulation of the vitamin D receptor and cornifin beta expression in vaginal epithelium of the rats through vitamin D3. Eur. J. Histochem. 2008, 52, 107–114. [Google Scholar] [CrossRef]
- Lee, A.; Lee, M.R.; Lee, H.H.; Kim, Y.S.; Kim, J.M.; Enkhbold, T.; Kim, T.H. Vitamin D Proliferates Vaginal Epithelium through RhoA Expression in Postmenopausal Atrophic Vagina tissue. Mol. Cells 2017, 40, 677–684. [Google Scholar] [CrossRef]
- Kim, T.H.; Lee, H.H.; Park, J. Immunohistochemical detection of the 1,25-dihydroxy vitamin D receptor in the human vagina. Iran. J. Reprod. Med. 2014, 12, 805–810. [Google Scholar]
- Lahey, T.; Ghosh, M.; Fahey, J.V.; Shen, Z.; Mukura, L.R.; Song, Y.; Cu-Uvin, S.; Mayer, K.H.; Wright, P.F.; Kappes, J.C.; et al. Selective impact of HIV disease progression on the innate immune system in the human female reproductive tract. PLoS ONE 2012, 7, e38100. [Google Scholar] [CrossRef]
- Chandra, N.; Thurman, A.R.; Anderson, S.; Cunningham, T.D.; Yousefieh, N.; Mauck, C.; Doncel, G.F. Depot Medroxyprogesterone Acetate Increases Immune Cell Numbers and Activation Markers in Human Vaginal Mucosal Tissues. AIDS Res. Hum. Retrovir. 2012, 29, 592–601. [Google Scholar] [CrossRef]
- Thurman, A.R.; Chandra, N.; Yousefieh, N.; Zalenskaya, I.; Kimble, T.; Asin, S.; Rollenhagen, C.; Anderson, S.M.; Herold, B.; Mesquita, P.M.; et al. Comparison of Follicular and Luteal Phase Mucosal Markers of HIV Susceptibility in Healthy Women. AIDS Res. Hum. Retrovir. 2016, 32, 547–560. [Google Scholar] [CrossRef]
- Zalenskaya, I.A.; Joseph, T.; Bavarva, J.; Yousefieh, N.; Jackson, S.S.; Fashemi, T.; Yamamoto, H.S.; Settlage, R.; Fichorova, R.N.; Doncel, G.F. Gene Expression Profiling of Human Vaginal Cells In Vitro Discriminates Compounds with Pro-Inflammatory and Mucosa-Altering Properties: Novel Biomarkers for Preclinical Testing of HIV Microbicide Candidates. PLoS ONE 2015, 10, e0128557. [Google Scholar] [CrossRef]
- Richardson-Harman, N.; Mauck, C.; McGowan, I.; Anton, P. Dose-response relationship between tissue concentrations of UC781 and explant infectibility with HIV type 1 in the RMP-01 rectal safety study. AIDS Res. Hum. Retrovir. 2012, 28, 1422–1433. [Google Scholar] [CrossRef]
- Richardson-Harman, N.; Lackman-Smith, C.; Fletcher, P.S.; Anton, P.A.; Bremer, J.W.; Dezzutti, C.S.; Elliott, J.; Grivel, J.C.; Guenthner, P.; Gupta, P.; et al. Multisite comparison of anti-human immunodeficiency virus microbicide activity in explant assays using a novel endpoint analysis. J. Clin. Microbiol. 2009, 47, 3530–3539. [Google Scholar] [CrossRef]
- Livak, K.J.; Wills, Q.F.; Tipping, A.J.; Datta, K.; Mittal, R.; Goldson, A.J.; Sexton, D.W.; Holmes, C.C. Methods for qPCR gene expression profiling applied to 1440 lymphoblastoid single cells. Methods 2013, 59, 71–79. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Fryar, C.D.; Kruszon-Moran, D.; Gu, Q.; Ogden, C.L. Mean Body Weight, Height, Waist Circumference, and Body Mass Index Among Adults: United States, 1999–2000 Through 2015–2016. Natl. Health Stat. Rep. 2018, 122, 1–16. [Google Scholar]
- Ghosh, M. Secreted mucosal antimicrobials in the female reproductive tract that are important to consider for HIV prevention. Am. J. Reprod. Immunol. 2014, 71, 575–588. [Google Scholar] [CrossRef]
- Zalenskaya, I.A. Use of Injectable Depot Medroxyprogesterone Acetate (DMPA) Downregulates Cervicovaginal Epithelial Barrier Genes—Similarly to Mucosal Inflammation. In Proceedings of the HIV Research for Prevention 2016, Chicago, IL, USA, 17–21 October 2016. [Google Scholar]
- Guo, L.; Xu, X.Q.; Zhou, L.; Zhou, R.H.; Wang, X.; Li, J.L.; Liu, J.B.; Liu, H.; Zhang, B.; Ho, W.Z. Human Intestinal Epithelial Cells Release Antiviral Factors That Inhibit HIV Infection of Macrophages. Front. Immunol. 2018, 9, 247. [Google Scholar] [CrossRef]
- Okumura, A.; Lu, G.; Pitha-Rowe, I.; Pitha, P.M. Innate antiviral response targets HIV-1 release by the induction of ubiquitin-like protein ISG15. Proc. Natl. Acad. Sci. USA 2006, 103, 1440–1445. [Google Scholar] [CrossRef]
- Scagnolari, C.; Monteleone, K.; Selvaggi, C.; Pierangeli, A.; D’Ettorre, G.; Mezzaroma, I.; Turriziani, O.; Gentile, M.; Vullo, V.; Antonelli, G. ISG15 expression correlates with HIV-1 viral load and with factors regulating T cell response. Immunobiology 2016, 221, 282–290. [Google Scholar] [CrossRef]
- Aleyasin, A.; Hosseini, M.A.; Mahdavi, A.; Safdarian, L.; Fallahi, P.; Mohajeri, M.R.; Abbasi, M.; Esfahani, F. Predictive value of the level of vitamin D in follicular fluid on the outcome of assisted reproductive technology. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 159, 132–137. [Google Scholar] [CrossRef]
- Johansson, P.; Almqvist, E.G.; Johansson, J.O.; Mattsson, N.; Andreasson, U.; Hansson, O.; Wallin, A.; Blennow, K.; Zetterberg, H.; Svensson, J. Cerebrospinal fluid (CSF) 25-hydroxyvitamin D concentration and CSF acetylcholinesterase activity are reduced in patients with Alzheimer’s disease. PLoS ONE 2013, 8, e81989. [Google Scholar] [CrossRef]
- Jadhav, N.J.; Gokhale, S.; Seervi, M.; Patil, P.S.; Alagarasu, K. Immunomodulatory effect of 1, 25 dihydroxy vitamin D3 on the expression of RNA sensing pattern recognition receptor genes and cytokine response in dengue virus infected U937-DC-SIGN cells and THP-1 macrophages. Int. Immunopharmacol. 2018, 62, 237–243. [Google Scholar] [CrossRef]
- Perng, Y.C.; Lenschow, D.J. ISG15 in antiviral immunity and beyond. Nat. Rev. Microbiol. 2018, 16, 423–439. [Google Scholar] [CrossRef]
- Gao, L.; Wang, Y.; Li, Y.; Dong, Y.; Yang, A.; Zhang, J.; Li, F.; Zhang, R. Genome-wide expression profiling analysis to identify key genes in the anti-HIV mechanism of CD4(+) and CD8(+) T cells. J. Med. Virol. 2018, 90, 1199–1209. [Google Scholar] [CrossRef]
- Khare, D.; Godbole, N.M.; Pawar, S.D.; Mohan, V.; Pandey, G.; Gupta, S.; Kumar, D.; Dhole, T.N.; Godbole, M.M. Calcitriol [1, 25[OH]2 D3] pre—And post-treatment suppresses inflammatory response to influenza A (H1N1) infection in human lung A549 epithelial cells. Eur. J. Nutr. 2013, 52, 1405–1415. [Google Scholar] [CrossRef]
- Padovan, E.; Terracciano, L.; Certa, U.; Jacobs, B.; Reschner, A.; Bolli, M.; Spagnoli, G.C.; Borden, E.C.; Heberer, M. Interferon stimulated gene 15 constitutively produced by melanoma cells induces e-cadherin expression on human dendritic cells. Cancer Res. 2002, 62, 3453–3458. [Google Scholar]
- Recht, M.; Borden, E.C.; Knight, E., Jr. A human 15-kDa IFN-induced protein induces the secretion of IFN-gamma. J. Immunol. 1991, 147, 2617–2623. [Google Scholar]
- Sun, L.; Wang, X.; Zhou, Y.; Zhou, R.H.; Ho, W.Z.; Li, J.L. Exosomes contribute to the transmission of anti-HIV activity from TLR3-activated brain microvascular endothelial cells to macrophages. Antivir. Res. 2016, 134, 167–171. [Google Scholar] [CrossRef]
- Owhashi, M.; Taoka, Y.; Ishii, K.; Nakazawa, S.; Uemura, H.; Kambara, H. Identification of a ubiquitin family protein as a novel neutrophil chemotactic factor. Biochem. Biophys. Res. Commun. 2003, 309, 533–539. [Google Scholar] [CrossRef]
- Aguilar-Jimenez, W.; Villegas-Ospina, S.; Gonzalez, S.; Zapata, W.; Saulle, I.; Garziano, M.; Biasin, M.; Clerici, M.; Rugeles, M.T. Active and precursor forms of Vitamin D reduce HIV-1 infection in vitro. J. Acquir. Immune Defic. Syndr. 2016. [Google Scholar] [CrossRef]
- Gonzalez, S.M.; Aguilar-Jimenez, W.; Trujillo-Gil, E.; Zapata, W.; Su, R.C.; Ball, T.B.; Rugeles, M.T. Vitamin D treatment of peripheral blood mononuclear cells modulated immune activation and reduced susceptibility to HIV-1 infection of CD4+ T lymphocytes. PLoS ONE 2019, 14, e0222878. [Google Scholar] [CrossRef]
- Assa, A.; Vong, L.; Pinnell, L.J.; Avitzur, N.; Johnson-Henry, K.C.; Sherman, P.M. Vitamin D deficiency promotes epithelial barrier dysfunction and intestinal inflammation. J. Infect. Dis. 2014, 210, 1296–1305. [Google Scholar] [CrossRef]
- Thurman, A.; Chandra, N.; Schwartz, J.L.; Brache, V.; Chen, B.A.; Asin, S.; Rollenhagen, C.; Herold, B.C.; Fichorova, R.N.; Hillier, S.L.; et al. The Effect of Hormonal Contraception on Cervicovaginal Mucosal End Points Associated with HIV Acquisition. AIDS Res. Hum. Retrovir. 2019, 35, 853–864. [Google Scholar] [CrossRef]
- Thurman, A.R.; Yousefieh, N.; Chandra, N.; Kimble, T.; Asin, S.; Rollenhagen, C.; Anderson, S.M.; Herold, B.C.; Freiermuth, J.L.; Starkman, B.S.; et al. Comparison of mucosal markers of human immunodeficiency virus susceptibility in healthy premenopausal versus postmenopausal women. AIDS Res. Hum. Retrovir. 2017, 33, 807–819. [Google Scholar] [CrossRef]
- Campbell, G.R.; Spector, S.A. Hormonally active vitamin D3 (1alpha,25-dihydroxycholecalciferol) triggers autophagy in human macrophages that inhibits HIV-1 infection. J. Biol. Chem. 2011, 286, 18890–18902. [Google Scholar] [CrossRef]
- Pauza, C.D.; Kornbluth, R.; Emau, P.; Richman, D.D.; Deftos, L.J. Vitamin D3 compounds regulate human immunodeficiency virus type 1 replication in U937 monoblastoid cells and in monocyte-derived macrophages. J. Leukoc. Biol. 1993, 53, 157–164. [Google Scholar] [CrossRef]
- Skolnik, P.R.; Jahn, B.; Wang, M.Z.; Rota, T.R.; Hirsch, M.S.; Krane, S.M. Enhancement of human immunodeficiency virus 1 replication in monocytes by 1,25-dihydroxycholecalciferol. Proc. Natl. Acad. Sci. USA 1991, 88, 6632–6636. [Google Scholar] [CrossRef]
- Coussens, A.K.; Naude, C.E.; Goliath, R.; Chaplin, G.; Wilkinson, R.J.; Jablonski, N.G. High-dose vitamin D3 reduces deficiency caused by low UVB exposure and limits HIV-1 replication in urban Southern Africans. Proc. Natl. Acad. Sci. USA 2015, 112, 8052–8057. [Google Scholar] [CrossRef]
- Cole, A.M.; Cole, A.L. Antimicrobial polypeptides are key anti-HIV-1 effector molecules of cervicovaginal host defense. Am. J. Reprod. Immunol. 2008, 59, 27–34. [Google Scholar] [CrossRef]
- Ghosh, M.; Jais, M.; Biswas, R.; Jarin, J.; Daniels, J.; Joy, C.; Juzumaite, M.; Emmanuel, V.; Gomez-Lobo, V. Immune biomarkers and anti-HIV activity in the reproductive tract of sexually active and sexually inactive adolescent girls. Am. J. Reprod. Immunol. 2018, 79, e12846. [Google Scholar] [CrossRef]
- Pietras, S.M.; Obayan, B.K.; Cai, M.H.; Holick, M.F. Vitamin D2 treatment for vitamin D deficiency and insufficiency for up to 6 years. Arch. Intern. Med. 2009, 169, 1806–1808. [Google Scholar] [CrossRef]
- Malabanan, A.; Veronikis, I.E.; Holick, M.F. Redefining vitamin D insufficiency. Lancet 1998, 351, 805–806. [Google Scholar] [CrossRef]
- Pepper, K.J.; Judd, S.E.; Nanes, M.S.; Tangpricha, V. Evaluation of vitamin D repletion regimens to correct vitamin D status in adults. Endocr. Pract. 2009, 15, 95–103. [Google Scholar] [CrossRef]
- Heaney, R.P.; Davies, K.M.; Chen, T.C.; Holick, M.F.; Barger-Lux, M.J. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am. J. Clin. Nutr. 2003, 77, 204–210. [Google Scholar] [CrossRef]
- Vieth, R.; Chan, P.C.; MacFarlane, G.D. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am. J. Clin. Nutr. 2001, 73, 288–294. [Google Scholar] [CrossRef]
- Bischof, M.G.; Heinze, G.; Vierhapper, H. Vitamin D status and its relation to age and body mass index. Horm. Res. 2006, 66, 211–215. [Google Scholar] [CrossRef]
- Lagunova, Z.; Porojnicu, A.C.; Lindberg, F.; Hexeberg, S.; Moan, J. The dependency of vitamin D status on body mass index, gender, age and season. Anticancer Res. 2009, 29, 3713–3720. [Google Scholar] [CrossRef]
| Variables | Total | Daily | Weekly | p-Value (Daily Versus Weekly) |
|---|---|---|---|---|
| Analysis Population | 20 | 10 | 10 | |
| Average Age (years) 1 | 37.05 ± 6.33 | 37.20 ± 1.97 | 36.90 ± 2.14 | 0.92 |
| Average BMI (kg/m2) 1 | 27.41 ± 4.57 | 24.99 ± 1.25 | 29.83 ± 1.24 | 0.01 |
| Education (years) 1 | 14.93 ± 2.21 | 14.80 ± 2.35 | 15.05 ± 2.19 | 0.80 |
| Gravidity 1 | 2.45 ± 1.50 | 2.30 ± 1.49 | 2.60 ± 1.58 | 0.67 |
| Parity 1 | 1.90 ± 1.29 | 2.0 ± 1.25 | 1.8 ± 1.4 | 0.74 |
| Vaginal pH at V1 1 | 4.12 ± 0.31 | 4.10 ± 0.32 | 4.14 ± 0.33 | 0.78 |
| Vaginal Nugent Score at V1 1 | 1.80 ± 1.70 | 1.40 ± 1.65 | 2.20 ± 1.75 | 0.31 |
| Race and Ethnicity | ||||
| Hispanic Ethnicity | 4 | 3 | 1 | - |
| Non-Hispanic Black | 7 | 1 | 6 | - |
| Non-Hispanic White | 8 | 5 | 3 | - |
| Asian | 1 | 1 | 0 | - |
| Variables | Pre-Supplementation 1 | Post-Supplementation 1 | p-Value |
|---|---|---|---|
| Epithelial Thickness (µm) | 225.5 ± 55.1 | 265.7 ± 88.0 | 0.05 |
| Number of Cell Layers | 20.8 ± 3.0 | 21.3 ± 3.1 | 0.5 |
| Vaginal immune cell populations/phenotype (cells/mm2) | |||
| CD45 Epi | 120.5 ± 43.0 | 117.1 ± 52.9 | 0.8 |
| CD45 LP | 95.9 ± 37.1 | 109.0 ± 44.7 | 0.2 |
| CD3 Epi | 93.5 ± 41.4 | 93.1 ± 47.0 | 0.9 |
| CD3 LP | 68.9 ± 32.7 | 79.6 ± 38.0 | 0.3 |
| CD8 Epi | 78.3 ± 37.6 | 68.8 ± 43.9 | 0.08 |
| CD8 LP | 44.5 ± 26.1 | 49.1 ± 26.6 | 0.7 |
| CD1a Epi | 45.0 ± 17.9 | 43.5 ± 20.4 | 0.7 |
| CD1a LP | 0.1 ± 0.6 | 1.4 ± 2.7 | 0.09 |
| CD4 Epi | 61.9 ± 40.3 | 44.2 ± 24.2 | 0.04 |
| CD4 LP | 121.9 ± 42.9 | 92.2 ± 33.1 | 0.02 |
| CCR5 Epi | 0.0 ± 0.0 | 0.3 ± 0.9 | 0.2 |
| CCR5 LP | 25.1 ± 11.2 | 24.2 ± 9.7 | 0.7 |
| Mediator (pg/mL) | Pre-Supplementation 1 | Post-Supplementation 1 | p-Value |
|---|---|---|---|
| Elafin | 2401.2 ± 2168.8 | 1899.3 ± 1765.8 | 0.20 |
| HBD-2 | 39.3 ± 69.7 | 23.0 ± 28.4 | 0.26 |
| LL-37 | 29,459.0 ± 49,726.2 | 18,184.4 ± 3340.2 | 0.18 |
| SLPI | 581.7 ± 700.0 | 453.5 ± 481.1 | 0.54 |
| Treatment Group | % Inhibition Pre-Supplementation | % Inhibition Post-Supplementation | p-Value |
|---|---|---|---|
| Overall (n = 19) | 69.68 ± 40.67 | 59.60 ± 33.67 | 0.15 |
| Weekly (n = 9) | 83.81 ± 21.14 | 66.86 ± 26.61 | 0.05 |
| Daily (n = 10) | 56.97 ± 50.32 | 53.06 ± 39.22 | 0.66 |
| Variable 1 | Pre-Supplementation 2 | Post-Supplementation 2 | p-Value |
|---|---|---|---|
| P24_Day 21 | 192.03 ± 305.88 | 88.71 ± 159.39 | 0.12 |
| P24_Soft | 194.4 ± 304.65 | 98.43 ± 159.44 | 0.36 |
| P24_Maximum | 198.32 ± 302.3 | 109.93 ± 162.47 | 0.22 |
| P24_Cumulative | 240.9 ± 336.49 | 168.69 ± 204.94 | 0.30 |
| P24_AUC | 1012.47 ± 1294.33 | 868.64 ± 971.37 | 0.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anderson, S.M.; Thurman, A.R.; Chandra, N.; Jackson, S.S.; Asin, S.; Rollenhagen, C.; Ghosh, M.; Daniels, J.; Vann, N.C.; Clark, M.R.; et al. Vitamin D Status Impacts Genital Mucosal Immunity and Markers of HIV-1 Susceptibility in Women. Nutrients 2020, 12, 3176. https://doi.org/10.3390/nu12103176
Anderson SM, Thurman AR, Chandra N, Jackson SS, Asin S, Rollenhagen C, Ghosh M, Daniels J, Vann NC, Clark MR, et al. Vitamin D Status Impacts Genital Mucosal Immunity and Markers of HIV-1 Susceptibility in Women. Nutrients. 2020; 12(10):3176. https://doi.org/10.3390/nu12103176
Chicago/Turabian StyleAnderson, Sharon M., Andrea R. Thurman, Neelima Chandra, Suzanne S. Jackson, Susana Asin, Christiane Rollenhagen, Mimi Ghosh, Jason Daniels, Nikolas C. Vann, Meredith R. Clark, and et al. 2020. "Vitamin D Status Impacts Genital Mucosal Immunity and Markers of HIV-1 Susceptibility in Women" Nutrients 12, no. 10: 3176. https://doi.org/10.3390/nu12103176
APA StyleAnderson, S. M., Thurman, A. R., Chandra, N., Jackson, S. S., Asin, S., Rollenhagen, C., Ghosh, M., Daniels, J., Vann, N. C., Clark, M. R., & Doncel, G. F. (2020). Vitamin D Status Impacts Genital Mucosal Immunity and Markers of HIV-1 Susceptibility in Women. Nutrients, 12(10), 3176. https://doi.org/10.3390/nu12103176







