Prevention of Neural Tube Defects by Folic Acid Supplementation: A National Population-Based Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design of the Study
2.2. Source of Data and Population
2.3. Data Collected
2.4. Statistical Analysis
2.5. Details of Ethics Approval
3. Results
4. Discussion
4.1. Main Findings
4.2. Strengths and Limitations
4.3. Interpretation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2006–2016 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patients | ||||||||||||
| N | 13,361 | 13,761 | 14,352 | 14,355 | 14,920 | 14,774 | 14,875 | 14,682 | 14,540 | 14,931 | 14,509 | 159,060 |
| Age at conception | ||||||||||||
| Median (year) ± SD | 29 ± 5.42 | 29 ± 5.48 | 30 ± 5.50 | 30 ± 5.58 | 30 ± 5.60 | 30 ± 5.53 | 30 ± 5.50 | 30 ± 5.49 | 30 ± 5.46 | 30 ± 5.44 | 30 ± 5.44 | 30 ± 5.50 |
| Pregnancy counts | ||||||||||||
| First pregnancy (n) | 5.720 | 5.578 | 5.708 | 5.569 | 6.033 | 5.878 | 6.059 | 5.968 | 5.848 | 6.022 | 5.669 | 64.052 |
| First pregnancy (%) | 38% | 35% | 35% | 34% | 35% | 34% | 34% | 34% | 34% | 34% | 33% | 34% |
| Second or subsequent pregnancy (n) | 9419 | 10,151 | 10,709 | 10,956 | 11,295 | 11,467 | 11,634 | 11,598 | 11,494 | 11,715 | 11,571 | 122,009 |
| Second or subsequent pregnancy (%) | 62% | 65% | 65% | 66% | 65% | 66% | 66% | 66% | 66% | 66% | 67% | 66% |
| All pregnancy (n) | 15,139 | 15,729 | 16,417 | 16,525 | 17,328 | 17,345 | 17,693 | 17,566 | 17,342 | 17,737 | 17,240 | 186,061 |
| Health coverage | ||||||||||||
| Public universal health coverage | 8% | 8% | 8% | 8% | 8% | 8% | 8% | 9% | 9% | 9% | 9% | 8% |
| Mutual health coverage | 68% | 66% | 66% | 65% | 64% | 64% | 63% | 62% | 62% | 63% | 62% | 64% |
| Private health insurance coverage | 14% | 15% | 16% | 16% | 17% | 17% | 18% | 18% | 18% | 18% | 19% | 17% |
| Coverage by other institutions | 10% | 11% | 11% | 11% | 11% | 11% | 11% | 11% | 11% | 10% | 10% | 11% |
References
- European Commission. EU Science Hub, European Platform on Rare disease Restriction, EUROCAT DATA, EUROCAT, Prevalence per 10,000 Births. Neural Tube Defects, 2006 to 2016. All Full Registries. Available online: https://eu-rd-platform.jrc.ec.europa.eu/eurocat/eurocat-data/prevalence (accessed on 30 April 2020).
- Viswanathan, M.; Treiman, K.A.; Kish-Doto, J.; Middleton, J.C.; Coker-Schwimmer, E.J.L.; Nicholson, W.K. Folic Acid Supplementation: An Evidence Review for the US Preventive Services Task Force: Evidence Synthesis; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2017; Report No. 14-05214-EF-1. [Google Scholar]
- Yi, Y.; Lindemann, M.; Colligs, A.; Snowball, C. Economic burden of neural tube defects and impact of prevention with folic acid: A literature review. Eur. J. Pediatr. 2011, 170, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Molloy, A.M. Should vitamin B12 status be considered in assessing risk of neural tube defects? Ann. N. Y. Acad. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Molloy, A.M.; Pangilinan, F.; Brody, L.C. Genetic Risk Factors for Folate-Responsive Neural Tube Defects. Annu. Rev. Nutr. 2017, 37, 269–291. [Google Scholar] [CrossRef] [PubMed]
- MRC Vitamin Study Research Group. Prevention of neural tube defects, Results of the Medical Research Council Vitamin Study. Lancet 1991, 338, 131–137. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Fernández-Gaxiola, A.C.; Dowswell, T.; Peña-Rosas, J.P. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef]
- World Health Organization, Department of Making Pregnancy Safer. Prevention of neural tube defects, Chapter 1.6. In Standards for Maternal and Neonatal Care; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Castillo-Lancellotti, C.; Tur, J.A.; Uauy, R. Impact of folic acid fortification of flour on neural tube defects: A systematic review. Public Health Nutr. 2013, 16, 901–911. [Google Scholar] [CrossRef]
- Regulation N°1925/2006 of the European Parliament and of the Council of 20 December 2006 on the Addition of Vitamins and Minerals and of Certain Other Substances to Foods. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32006R1925 (accessed on 30 April 2020).
- Food Fortification Initiative. Wheat Flour Fortification Status in Eastern and Western Europe. 2020. Available online: http://ffinetwork.org/regional_activity/europe.php (accessed on 30 April 2020).
- HAS. Haute Autorité de Santé—Projet de Grossesse Informations, Messages de Prévention, Examens à Proposer, Recommandations. 2009. Available online: https://www.has-sante.fr/upload/docs/application/pdf/2010/01/projet_de_grossesse_informations_messages_de_prevention_examens_a_proposer_argumentaire.pdf (accessed on 30 April 2020).
- EUROCAT. Prevention of Neural Tube Defects by Periconceptional Folic Acid Supplementation in Europe. Special Report. 2009. Available online: https://eu-rd-platform.jrc.ec.europa.eu/sites/default/files/eurocat-Special-Report-NTD-3rdEd-2009.pdf (accessed on 30 April 2020).
- Finer, L.B.; Zolna, M.R. Unintended pregnancy in the United States: Incidence and disparities. Contraception 2011, 84, 478–485. [Google Scholar] [CrossRef]
- Tort, J.; Lelong, N.; Prunet, C.; Khoshnood, B.; Blondel, B. Maternal and health care determinants of preconceptional use of folic acid supplementation in France: Results from the 2010 National Perinatal Survey. BJOG 2013, 120, 1661–1667. [Google Scholar] [CrossRef]
- Camier, A.; Kadawathagedara, M.; Lioret, S.; Bois, C.; Cheminat, M.; Dufourg, M.-N.; Charles, M.-A.; De Lauzon, B. Social Inequalities in Prenatal Folic Acid Supplementation: Results from the ELFE Cohort. Nutrients 2019, 11, 1108. [Google Scholar] [CrossRef]
- Bezin, J.; Duong, M.; Lassalle, R.; Droz, C.; Pariente, A.; Blin, P.; Moore, N. The national healthcare system claims databases in France, SNIIRAM and EGB: Powerful tools for pharmacoepidemiology. Pharmacoepidemiol. Drug Saf. 2017, 26, 954–962. [Google Scholar] [CrossRef]
- Bérard, A.; Abbas-Chorfa, F.; Kassai-Koupai, B.; Vial, T.; Nguyen, K.A.; Sheehy, O.; Schott, A.-M. The French Pregnancy Cohort: Medication use during pregnancy in the French population. PLoS ONE 2019, 14, e0219095. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, S.; Jaddoe, V.W.V.; Mackenbach, J.P.; Hofman, A.; Steegers-Theunissen, R.P.M.; Steegers, E.A.P. Determinants of folic acid use in early pregnancy in a multi-ethnic urban population in The Netherlands: The Generation R study. Prev. Med. 2008, 47, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Zetstra-van der Woude, P.A.; de Walle, H.E.K.; de Jong-van den Berg, L.T.W. Periconceptional folic acid use: Still room to improve. Birth Defects Res. 2012, 94, 96–101. [Google Scholar] [CrossRef] [PubMed]
- McKeating, A.; Farren, M.; Cawley, S.; Daly, N.; McCartney, D.; Turner, M.J. Maternal folic acid supplementation trends 2009–2013. Acta Obstet. Gynecol. Scand. 2015, 94, 727–733. [Google Scholar] [CrossRef]
- Vandevijvere, S.; Amsalkhir, S.; Van Oyen, H.; Moreno-Reyes, R. Determinants of folate status in pregnant women: Results from a national cross-sectional survey in Belgium. Eur. J. Clin. Nutr. 2012, 66, 1172–1177. [Google Scholar] [CrossRef]
- WHO. Serum and Red Blood Cell Folate Concentrations for Assessing Folate Status in Populations. Vitamin and Mineral Nutrition Information System; World Health Organization: Geneva, Switzerland, 2015; Available online: https://apps.who.int/iris/bitstream/handle/10665/162114/WHO_NMH_NHD_EPG_15.01.pdf?ua=1 (accessed on 30 April 2020).
- Kancherla, V.; Wagh, K.; Johnson, Q.; Oakley, G.P. A 2017 global update on folic acid-preventable spina bifida and anencephaly. Birth Defects Res. 2018, 110, 1139–1147. [Google Scholar] [CrossRef]
- Force, U.P.S.T.; Bibbins-Domingo, K.; Grossman, D.C.; Curry, S.J.; Davidson, K.W.; Epling, J.W.; García, F.A.; Kemper, A.R.; Krist, A.H.; Kurth, A.E.; et al. Folic Acid Supplementation for the Prevention of Neural Tube Defects: US Preventive Services Task Force Recommendation Statement. JAMA 2017, 317, 183. [Google Scholar] [CrossRef]
- Wals, P.D.; Uh, S.-H.; Lowry, R.B.; Evans, J.A.; Crowley, M.; Niyonsenga, T. Reduction in Neural-Tube Defects after Folic Acid Fortification in Canada. N. Engl. J. Med. 2007. [Google Scholar] [CrossRef]
- Lindzon, G.; O’Connor, D.L. Folate during reproduction: The Canadian experience with folic acid fortification. Nutr. Res. Pract. 2007, 1, 163. [Google Scholar] [CrossRef]
- Kancherla, V.; Pachón, H.; Blencowe, H.; Martinez, H.; Oakley, G.P.; Berry, R.J. The Fallacy of Using Administrative Data in Assessing the Effectiveness of Food Fortification. Comment on: “Folic Acid Fortification and Neural Tube Defect Risk: Analysis of the Food Fortification Initiative Dataset. Nutrients 2020, 12, 247”. Nutrients 2020, 12, 1352. [Google Scholar] [CrossRef]
- Westmark, C.J.; Murphy, M.E. Reply to “The Fallacy of Using Administrative Data in Assessing the Effectiveness of Food Fortification. Comment on: Folic Acid Fortification and Neural Tube Defect Risk: Analysis of the Food Fortification Initiative Dataset. Nutrients 2020, 12, 247”. Nutrients 2020, 12, 1335. [Google Scholar] [CrossRef]
- Murphy, M.E.; Westmark, C.J. Folic Acid Fortification and Neural Tube Defect Risk: Analysis of the Food Fortification Initiative Dataset. Nutrients 2020, 12, 247. [Google Scholar] [CrossRef]
- Grosse, S.D.; Berry, R.J.; Mick Tilford, J.; Kucik, J.E.; Waitzman, N.J. Retrospective Assessment of Cost Savings From Prevention. Am. J. Prev. Med. 2016, 50, S74–S80. [Google Scholar] [CrossRef]
- Ami, N.; Bernstein, M.; Boucher, F.; Rieder, M.; Parker, L. Folate and neural tube defects: The role of supplements and food fortification. Paediatr. Child Health 2016, 21, 5. [Google Scholar] [CrossRef]
- UK Government (GOV.UK). Proposal to Add Folic Acid to Flour: Consultation Document, Updated 27 September 2019. Available online: https://www.gov.uk/government/consultations/adding-folic-acid-to-flour/proposal-to-add-folic-acid-to-flour-consultation-document#fn:2 (accessed on 30 April 2020).

| First Pregnancy n (%) [95% CI] | Second or Subsequent Pregnancy n (%) [95% CI] | All Patients n (%) [95% CI] | |
|---|---|---|---|
| Rate of dispensing of folic acid 0.4 mg | 4231 (6.61%) [6.41–6.80%] | 4971 (4.07%) [3.96–4.19%] | 9202 (4.95%) [4.85–5.04%] |
| Rate of dispensing of folic acid 5 mg | 384 (0.60%) [0.54–0.66%] | 2233 (1.83%) [1.75–1.91%] | 2617 (1.41%) [1.35–1.46%] |
| Rate of dispensing of folic acid 0.4 mg and 5 mg | 4615 (7.21%) [7.00–3.83%] | 7204 (5.90%) [5.77–6.04%] | 11,819 (6.35%) [6.24–6.46%] |
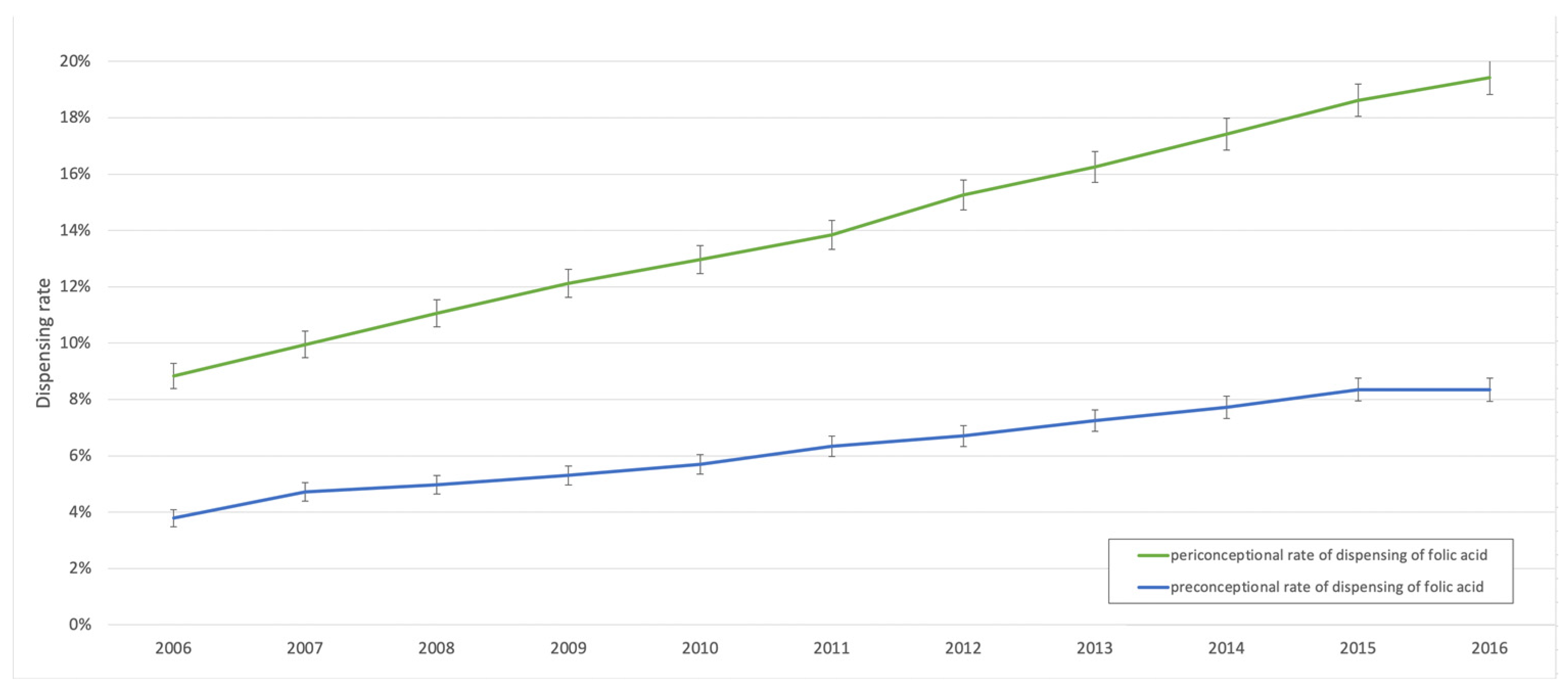
| Year | Pregnancies | Pregnancies with Preconceptional Dispensing of Folic Acid | Rate of Preconceptional Dispensing of Folic Acid | |
|---|---|---|---|---|
| n | n | % | 95% CI | |
| 2006 | 15,139 | 574 | 3.79% | [3.49–4.10%] |
| 2007 | 15,729 | 743 | 4.72% | [4.39–5.06%] |
| 2008 | 16,417 | 816 | 4.97% | [4.64–5.30%] |
| 2009 | 16,525 | 877 | 5.31% | [4.97–5.65%] |
| 2010 | 17,328 | 989 | 5.71% | [5.36–6.05%] |
| 2011 | 17,345 | 1101 | 6.35% | [5.98–6.71%] |
| 2012 | 17,693 | 1187 | 6.71% | [6.34–7.08%] |
| 2013 | 17,566 | 1274 | 7.25% | [6.87–7.64%] |
| 2014 | 17,342 | 1339 | 7.72% | [7.32–8.12%] |
| 2015 | 17,737 | 1481 | 8.35% | [7.94–8.76%] |
| 2016 | 17,240 | 1438 | 8.34% | [7.93–8.75%] |
| 2006–2016 | 186,061 | 11,819 | 6.35% | [6.24–6.46%] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Fournière, B.; Dhombres, F.; Maurice, P.; de Foucaud, S.; Lallemant, P.; Zérah, M.; Guilbaud, L.; Jouannic, J.-M. Prevention of Neural Tube Defects by Folic Acid Supplementation: A National Population-Based Study. Nutrients 2020, 12, 3170. https://doi.org/10.3390/nu12103170
de la Fournière B, Dhombres F, Maurice P, de Foucaud S, Lallemant P, Zérah M, Guilbaud L, Jouannic J-M. Prevention of Neural Tube Defects by Folic Acid Supplementation: A National Population-Based Study. Nutrients. 2020; 12(10):3170. https://doi.org/10.3390/nu12103170
Chicago/Turabian Stylede la Fournière, Benoît, Ferdinand Dhombres, Paul Maurice, Sabine de Foucaud, Pauline Lallemant, Michel Zérah, Lucie Guilbaud, and Jean-Marie Jouannic. 2020. "Prevention of Neural Tube Defects by Folic Acid Supplementation: A National Population-Based Study" Nutrients 12, no. 10: 3170. https://doi.org/10.3390/nu12103170
APA Stylede la Fournière, B., Dhombres, F., Maurice, P., de Foucaud, S., Lallemant, P., Zérah, M., Guilbaud, L., & Jouannic, J.-M. (2020). Prevention of Neural Tube Defects by Folic Acid Supplementation: A National Population-Based Study. Nutrients, 12(10), 3170. https://doi.org/10.3390/nu12103170





