The Effect of Dietary Nitrate Supplementation on Isokinetic Torque in Adults: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
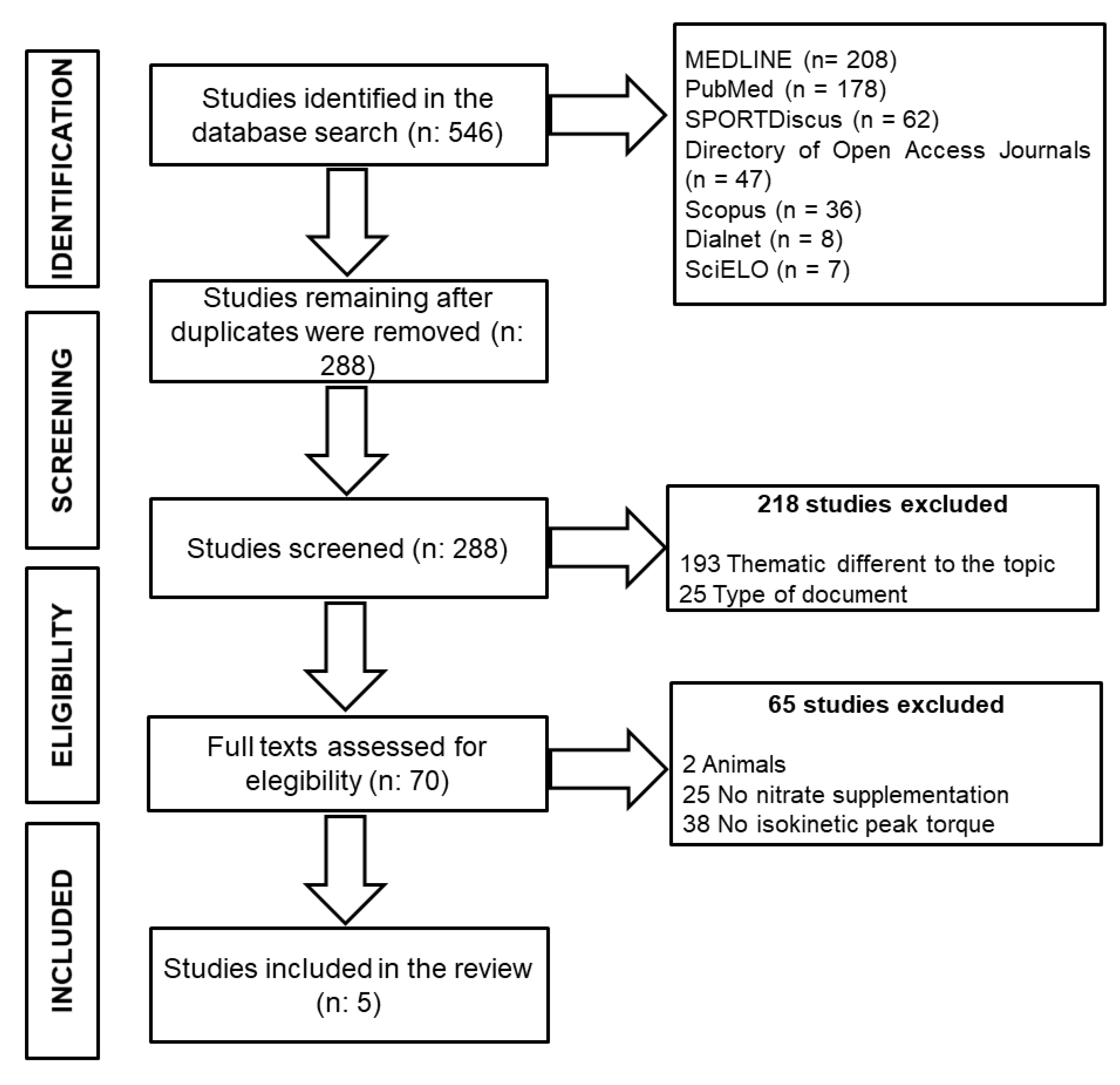
2. Methods
- -
- Studies that were published as a full article and performed in adults (age 18–80 years);
- -
- Studies that included a NO3− and placebo intervention;
- -
- Studies that assessed and reported isokinetic torque measures;
- -
- Studies that employed a randomized double-blind experimental design.
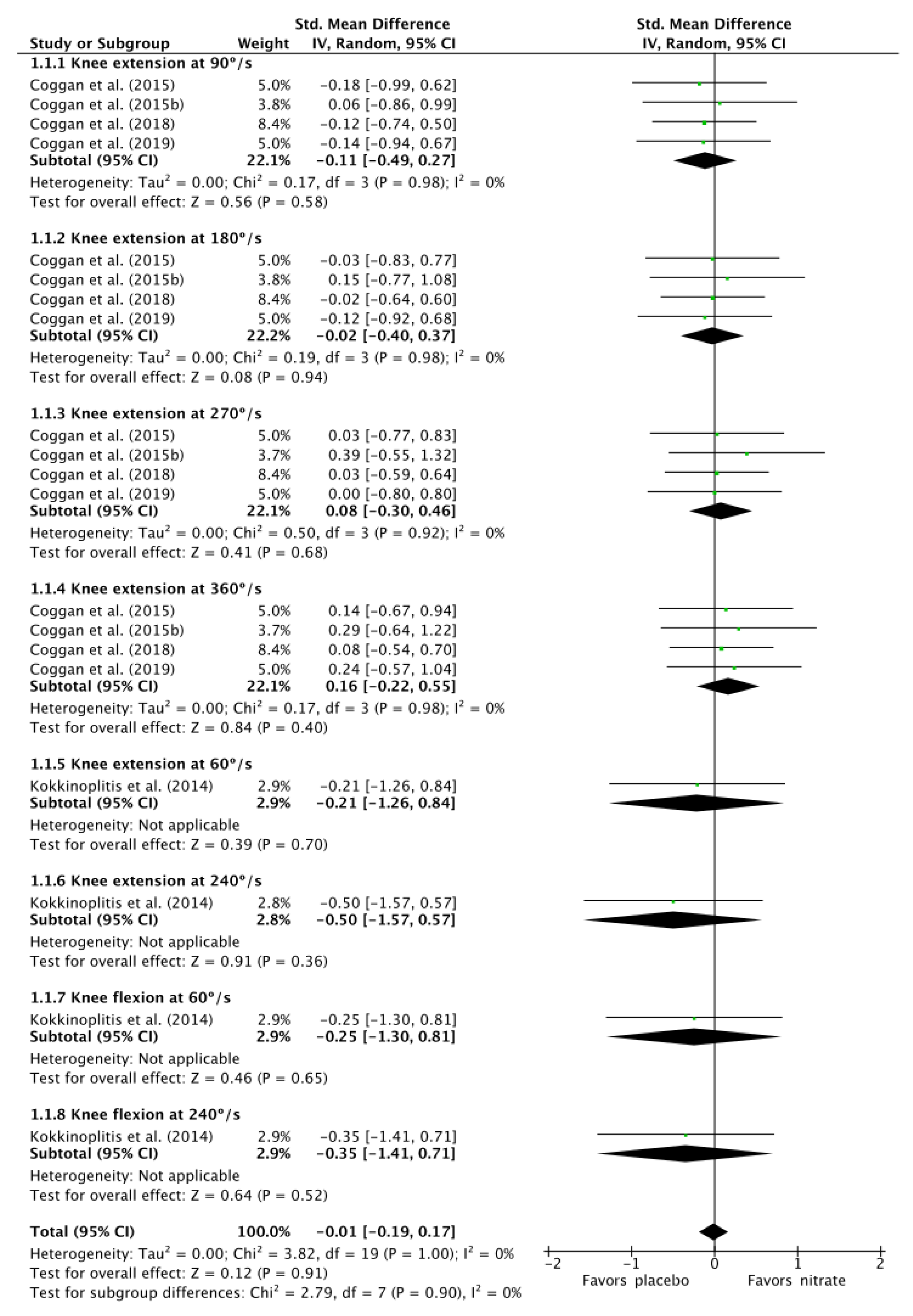
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Stone, M.H.; Moir, G.; Glaister, M.; Sanders, R. How much strength is necessary? Phys. Ther. Sport 2002, 3, 88–96. [Google Scholar] [CrossRef]
- Morrissey, M.C.; Harman, E.A.; Johnson, M.J. Resistance training modes: Specificity and effectiveness. Med. Sci. Sports Exerc. 1995, 27, 648–660. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W. Considerations and practical options for measuring muscle strength: A narrative review. Biomed Res. Int. 2019. [Google Scholar] [CrossRef] [PubMed]
- Gentil, P.; Del Vecchio, F.B.; Paoli, A.; Schoenfeld, B.J.; Bottaro, M. Isokinetic Dynamometry and 1RM Tests Produce Conflicting Results for Assessing Alterations in Muscle Strength. J. Hum. Kinet. 2017, 56, 19–27. [Google Scholar] [CrossRef]
- Paul, D.J.; Nassis, G.P. Testing strength and power in soccer players: The application of conventional and traditional methods of assessment. J. Strength Cond. Res. 2015, 29, 1748–1758. [Google Scholar] [CrossRef]
- DeLang, M.D.; Rouissi, M.; Bragazzi, N.L.; Chamari, K.; Salamh, P.A. Soccer footedness and between-limbs muscle strength: Systematic review and meta-analysis. Int. J. Sports Physiol. Perform. 2019, 14, 551–562. [Google Scholar] [CrossRef]
- Perrin, D.H. Isokinetic Exercise and Assessment; Human Kinetics Publishers: Champaign, IL, USA, 1993; ISBN 0873224647. [Google Scholar]
- Grgic, J.; Pickering, C. The effects of caffeine ingestion on isokinetic muscular strength: A meta-analysis. J. Sci. Med. Sport 2019, 22, 353–360. [Google Scholar] [CrossRef]
- Kues, J.M.; Rothstein, J.M.; Lamb, R.L. Obtaining Reliable Measurements of Knee Extensor Torque Produced During Maximal Voluntary Contractions: An Experimental Investigation. Phys. Ther. 1992, 72, 492–501. [Google Scholar] [CrossRef]
- Muñoz-Bermejo, L.; Pérez-Gómez, J.; Manzano, F.; Collado-Mateo, D.; Villafaina, S.; Adsuar, J.C. Reliability of isokinetic knee strength measurements in children: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0226274. [Google Scholar] [CrossRef]
- Abernethy, P.; Wilson, G.; Logan, P. Strength and Power Assessment: Issues, Controversies and Challenges. Sport Med. 1995, 19, 401–417. [Google Scholar] [CrossRef]
- Gleeson, N.P.; Mercer, T.H. The utility of isokinetic dynamometry in the assessment of human muscle function. Sport Med. 1996, 21, 18–34. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, O.H.; Stenager, E.; Dalgas, U. Muscle Strength and Poststroke Hemiplegia: A Systematic Review of Muscle Strength Assessment and Muscle Strength Impairment. Arch. Phys. Med. Rehabil. 2017, 98, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Pontes, S.S.; de Carvalho, A.L.R.; Almeida, K.D.O.; Neves, M.P.; Ribeiro Schindler, I.F.S.; Alves, I.G.N.; Arcanjo, F.L.; Gomes-Neto, M. Effects of isokinetic muscle strengthening on muscle strength, mobility, and gait in post-stroke patients: A systematic review and meta-analysis. Clin. Rehabil. 2019, 33, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Garthe, I.; Maughan, R.J. International Journal of Sport Nutrition and Exercise Metabolism. J. Danc. Med. Sci. 2004, 8, 123. [Google Scholar]
- Domínguez, R.; Maté-Muñoz, J.L.; Cuenca, E.; García-Fernández, P.; Mata-Ordoñez, F.; Lozano-Estevan, M.C.; Veiga-Herreros, P.; da Silva, S.F.; Garnacho-Castaño, M.V. Effects of beetroot juice supplementation on intermittent high-intensity exercise efforts. J. Int. Soc. Sports Nutr. 2018, 15, 1–12. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167. [Google Scholar] [CrossRef]
- Jones, A.M. Dietary nitrate supplementation and exercise performance. Sport Med. 2014, 44, 35–45. [Google Scholar] [CrossRef]
- Bailey, S.J.; Fulford, J.; Vanhatalo, A.; Winyard, P.G.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation enhances muscle contractile efficiency during knee-extensor exercise in humans. J. Appl. Physiol. 2010, 109, 135–148. [Google Scholar] [CrossRef]
- Ferguson, S.K.; Hirai, D.M.; Copp, S.W.; Holdsworth, C.T.; Allen, J.D.; Jones, A.M.; Musch, T.I.; Poole, D.C. Impact of dietary nitrate supplementation via beetroot juice on exercising muscle vascular control in rats. J. Physiol. 2013, 591, 547–557. [Google Scholar] [CrossRef]
- Hernández, A.; Schiffer, T.A.; Ivarsson, N.; Cheng, A.J.; Bruton, J.D.; Lundberg, J.O.; Weitzberg, E.; Westerblad, H. Dietary nitrate increases tetanic [Ca 2+] i and contractile force in mouse fast-twitch muscle. J. Physiol. 2012, 590, 3575–3583. [Google Scholar] [CrossRef]
- Whitfield, J.; Gamu, D.; Heigenhauser, G.J.F.; Van Loon, L.J.C.; Spriet, L.L.; Tupling, A.R.; Holloway, G.P. Beetroot juice increases human muscle force without changing Ca2+-handling proteins. Med. Sci. Sports Exerc. 2017, 49, 2016–2024. [Google Scholar] [CrossRef] [PubMed]
- Haider, G.; Folland, J.P. Nitrate supplementation enhances the contractile properties of human skeletal muscle. Med. Sci. Sports Exerc. 2014, 46, 2234–2243. [Google Scholar] [CrossRef] [PubMed]
- Wickham, K.A.; McCarthy, D.G.; Pereira, J.M.; Cervone, D.T.; Verdijk, L.B.; van Loon, L.J.C.; Power, G.A.; Spriet, L.L. No effect of beetroot juice supplementation on exercise economy and performance in recreationally active females despite increased torque production. Physiol. Rep. 2019, 7, e13982. [Google Scholar] [CrossRef] [PubMed]
- Hoon, M.W.; Fornusek, C.; Chapman, P.G.; Johnson, N.A. The effect of nitrate supplementation on muscle contraction in healthy adults. Eur. J. Sport Sci. 2015, 15, 712–719. [Google Scholar] [CrossRef]
- Tillin, N.A.; Moudy, S.; Nourse, K.M.; Tyler, C.J. Nitrate Supplement Benefits Contractile Forces in Fatigued but Not Unfatigued Muscle. Med. Sci. Sports Exerc. 2018, 50, 2122–2131. [Google Scholar] [CrossRef]
- Bender, D.; Townsend, J.R.; Vantrease, W.C.; Marshall, A.C.; Henry, R.N.; Heffington, S.H.; Johnson, K.D. Acute beetroot juice administration improves peak isometric force production in adolescent males. Appl. Physiol. Nutr. Metab. 2018, 43, 816–821. [Google Scholar] [CrossRef]
- Flanagan, S.D.; Looney, D.P.; Miller, M.J.S.; DuPont, W.H.; Pryor, L.; Creighton, B.C.; Sterczala, A.J.; Szivak, T.K.; Hooper, D.R.; Maresh, C.M.; et al. The Effects of Nitrate-Rich Supplementation on Neuromuscular Efficiency during Heavy Resistance Exercise. J. Am. Coll. Nutr. 2016, 35, 100–107. [Google Scholar] [CrossRef]
- Fulford, J.; Winyard, P.G.; Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; Jones, A.M. Influence of dietary nitrate supplementation on human skeletal muscle metabolism and force production during maximum voluntary contractions. Pflug. Arch. Eur. J. Physiol. 2013, 465, 517–528. [Google Scholar] [CrossRef]
- Coggan, A.R.; Leibowitz, J.L.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Spearie, C.A.; Waller, S.; Farmer, M.; Peterson, L.R. Effect of acute dietary nitrate intake on maximal knee extensor speed and power in healthy men and women. Nitric Oxide-Biol. Chem. 2015, 48, 16–21. [Google Scholar] [CrossRef]
- Coggan, A.R.; Leibowitz, J.L.; Spearie, C.A.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Mahmood, K.; Park, S.; Waller, S.; Farmer, M.; et al. Acute Dietary Nitrate Intake Improves Muscle Contractile Function in Patients with Heart Failure: A Double-Blind, Placebo-Controlled, Randomized Trial. Circ. Hear. Fail. 2015, 8, 914–920. [Google Scholar] [CrossRef]
- Coggan, A.R.; Hoffman, R.L.; Gray, D.A.; Moorthi, R.N.; Thomas, D.P.; Leibowitz, J.L.; Thies, D.; Peterson, L.R. A Single Dose of Dietary Nitrate Increases Maximal Knee Extensor Angular Velocity and Power in Healthy Older Men and Women. J. Gerontol. Ser. A 2020, 75, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Henneman, E.; Somjen, G.; Carpenter, D.O. Excitability and inhibitibility of motoneurons of different sizes. J. Neurophysiol. 1965, 28, 599–620. [Google Scholar] [CrossRef] [PubMed]
- Sargeant, A. Neuromuscular determinants of human performance. In Physiological Determinants of Exercise Tolerance in Humans; Whipp, B., Sargeant, A., Eds.; Portland Press: London, UK, 1999; pp. 13–28. [Google Scholar]
- Domínguez, R.; Garnacho-Castaño, M.V.; Cuenca, E.; García-Fernández, P.; Muñoz-González, A.; de Jesús, F.; Lozano-Estevan, M.D.C.; Fernandes da Silva, S.; Veiga-Herreros, P.; Maté-Muñoz, J.L. Effects of beetroot juice supplementation on a 30-s high-intensity inertial cycle ergometer test. Nutrients 2017, 9, 1360. [Google Scholar] [CrossRef] [PubMed]
- Cuenca, E.; Jodra, P.; Pérez-López, A.; González-Rodríguez, L.G.; da Silva, S.F.; Veiga-Herreros, P.; Domínguez, R. Effects of beetroot juice supplementation on performance and fatigue in a 30-s all-out sprint exercise: A randomized, double-blind cross-over study. Nutrients 2018, 10, 1222. [Google Scholar] [CrossRef]
- Jodra, P.; Domínguez, R.; Sánchez-Oliver, A.J.; Veiga-Herreros, P.; Bailey, S.J. Effect of beetroot juice supplementation on mood, perceived exertion, and performance during a 30-second wingate test. Int. J. Sports Physiol. Perform. 2020, 15, 243–248. [Google Scholar] [CrossRef]
- Rimer, E.G.; Peterson, L.R.; Coggan, A.R.; Martin, J.C. Increase in maximal cycling power with acute dietary nitrate supplementation. Int. J. Sports Physiol. Perform. 2016, 11, 715–720. [Google Scholar] [CrossRef]
- Bailey, S.J.; Varnham, R.L.; DiMenna, F.J.; Breese, B.C.; Wylie, L.J.; Jones, A.M. Inorganic nitrate supplementation improves muscle oxygenation, O2 uptake kinetics, and exercise tolerance at high but not low pedal rates. J. Appl. Physiol. 2015, 118, 1396–1405. [Google Scholar] [CrossRef]
- Kokkinoplitis, K.; Chester, N. The effect of beetroot juice on repeated sprint performance and muscle force production. J. Phys. Educ. Sport 2014, 14, 242. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Altman, D.G.; Booth, A.; et al. Preferred reporting items for systematic review and meta-analysis protocols (prisma-p) 2015: Elaboration and explanation. BMJ 2015, 349, 7647. [Google Scholar] [CrossRef]
- Brown, P.; Brunnhuber, K.; Chalkidou, K.; Chalmers, I.; Clarke, M.; Fenton, M.; Forbes, C.; Glanville, J.; Hicks, N.J.; Moody, J.; et al. How to formulate research recommendations. Br. Med. J. 2006, 333, 804–806. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro Scale for Rating Quality of Randomized Controlled Trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.V. Distribution Theory for Glass’s Estimator of Effect size and Related Estimators. J. Educ. Stat. 1981, 6, 107–128. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Hillsdale, N.J., Ed.; Lawrence Earlbaum Associates: New York, NY, USA, 1988; ISBN 0805802835. [Google Scholar]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J. Cochrane Handbook for Systematic Reviews of Interventions Version 6.0; Cochrane: London, UK, 2019. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Br. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Coggan, A.R.; Broadstreet, S.R.; Mikhalkova, D.; Bole, I.; Leibowitz, J.L.; Kadkhodayan, A.; Park, S.; Thomas, D.P.; Thies, D.; Peterson, L.R. Dietary nitrate-induced increases in human muscle power: High versus low responders. Physiol. Rep. 2018, 6, e13575. [Google Scholar] [CrossRef]
- Wylie, L.J.; Kelly, J.; Bailey, S.J.; Blackwell, J.R.; Skiba, P.F.; Winyard, P.G.; Jeukendrup, A.E.; Vanhatalo, A.; Jones, A.M. Beetroot juice and exercise: Pharmacodynamic and dose-response relationships. J. Appl. Physiol. 2013, 115, 325–336. [Google Scholar] [CrossRef]
- Kapil, V.; Milsom, A.B.; Okorie, M.; Maleki-Toyserkani, S.; Akram, F.; Rehman, F.; Arghandawi, S.; Pearl, V.; Benjamin, N.; Loukogeorgakis, S.; et al. Inorganic nitrate supplementation lowers blood pressure in humans: Role for nitrite-derived no. Hypertension 2010, 56, 274–281. [Google Scholar] [CrossRef]
- Webb, A.J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; et al. Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension 2008, 51, 784–790. [Google Scholar] [CrossRef]
- Jo, E.; Fischer, M.; Auslander, A.T.; Beigarten, A.; Daggy, B.; Hansen, K.; Kessler, L.; Osmond, A.; Wang, H.; Wes, R. The effects of multi-day vs. Single pre-exercise nitrate supplement dosing on simulated cycling time trial performance and skeletal muscle oxygenation. J. Strength Cond. Res. 2019, 33, 217–224. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Pavey, T.G.; Wilkerson, D.P.; Benjamin, N.; Winyard, P.G.; Jones, A.M. Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2010, 299, R1121–R1131. [Google Scholar] [CrossRef]
- Nyakayiru, J.; van Loon, L.J.C.; Verdijk, L.B. Could intramuscular storage of dietary nitrate contribute to its ergogenic effect? A mini-review. Free Radic. Biol. Med. 2020, 152, 295–300. [Google Scholar] [CrossRef]
- Van De Walle, G.P.; Vukovich, M.D. The effect of nitrate supplementation on exercise tolerance and performance: A systematic review and meta-analysis. J. Strength Cond. Res. 2018, 32, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Taddei, S.; Virdis, A.; Ghiadoni, L.; Salvetti, G.; Bernini, G.; Magagna, A.; Salvetti, A. Age-related reduction of NO availability and oxidative stress in humans. Hypertension 2001, 38, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Ivy, J.L. Inorganic Nitrate Supplementation for Cardiovascular Health. Methodist Debakey Cardiovasc. J. 2019, 15, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.D.; Khan, T.; Zeballos, G.A.; Mathew, L.; Potharlanka, P.; Knecht, M.; Whelan, J. Decreased activity of the L-arginine-nitric oxide metabolic pathway in patients with congestive heart failure. Circulation 1999, 99, 2113–2117. [Google Scholar] [CrossRef]
- Lauer, T.; Preik, M.; Rassaf, T.; Strauer, B.E.; Deussen, A.; Feelisch, M.; Kelm, M. Plasma nitrite rather than nitrate reflects regional endothelial nitric oxide synthase activity but lacks intrinsic vasodilator action. Proc. Natl. Acad. Sci. USA 2001, 98, 12814–12819. [Google Scholar] [CrossRef]
- San Juan, A.F.; Dominguez, R.; Lago-Rodríguez, Á.; Montoya, J.J.; Tan, R.; Bailey, S.J. Effects of dietary nitrate supplementation on weightlifting exercise performance in healthy adults: A systematic review. Nutrients 2020, 12, 2227. [Google Scholar] [CrossRef]
- Rodríguez-Fernández, A.; Castillo, D.; Raya-González, J.; Domínguez, R.; Bailey, S.J. Beetroot juice supplementation increases concentric and eccentric muscle power output. Original investigation. J. Sci. Med. Sport 2020. [Google Scholar] [CrossRef]
| Reference | Subjects | Supplementation | Exercise Protocol | Findings (BR vs. PL) |
|---|---|---|---|---|
| Coggan et al. [30] | 7 M/5 F healthy adults | 120 min prior to exercise ingestion of 2 × 70 mL BR shots (~11.2 mmol NO3−) | Isokinetic knee extension peak torque at: 90°/s, 180°/s, 270°/s, and 360°/s | −1.86% at 90°/s −1.72% at 180°/s +0% at 270°/s +4.27% at 360°/s |
| Coggan et al. [31] | 5 M/4 F heart failure patients | 120 min prior to exercise ingestion of 2 × 70 mL BR shots (~11.2 mmol NO3−) | Isokinetic knee extension peak torque at: 90°/s, 180°/s, 270°/s, and 360°/s | +1.47% at 90°/s −3.81% at 180°/s +9.41% at 270°/s +10.94% at 360°/s * |
| Coggan et al. [48] | 13 M/ 7 F healthy young and older adults | 120 min prior to exercise ingestion of 2 × 70 mL BR shots (~12.3 mmol NO3−) | Isokinetic knee extension peak torque at: 90°/s, 180°/s, 270°/s, and 360°/s | −3.11% at 90°/s −0.67% at 180°/s +0.8% at 270°/s +3.06% at 360°/s |
| Coggan et al. [32] | 6 M/6 F healthy older adults | 120 min prior to exercise ingestion of 2 × 70 mL BR shots (~13.4 mmol NO3−) | Isokinetic knee extension peak torque relative to body mass at: 0°/s, 90°/s, 180°/s, 270°/s, and 360°/s | −2.06 at 0°/s −2.82% at 90°/s −2.94% at 180°/s +0% at 270°/s +8.33% at 360°/s |
| Kokkinoplitis and Chester [40] | 7 M healthy adults | 180 min prior to exercise ingestion of 1 × 70 mL BR shot (~6.4 mmol NO3−) | Isokinetic knee extension and flexion peak torque at: 60°/s and 240°/s | Knee extension −3.47% at 60°/s −5.56% at 240°/s Knee flexion −6.85% at 60°/s −13.83% at 240°/s |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lago-Rodríguez, Á.; Domínguez, R.; Ramos-Álvarez, J.J.; Tobal, F.M.; Jodra, P.; Tan, R.; Bailey, S.J. The Effect of Dietary Nitrate Supplementation on Isokinetic Torque in Adults: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 3022. https://doi.org/10.3390/nu12103022
Lago-Rodríguez Á, Domínguez R, Ramos-Álvarez JJ, Tobal FM, Jodra P, Tan R, Bailey SJ. The Effect of Dietary Nitrate Supplementation on Isokinetic Torque in Adults: A Systematic Review and Meta-Analysis. Nutrients. 2020; 12(10):3022. https://doi.org/10.3390/nu12103022
Chicago/Turabian StyleLago-Rodríguez, Ángel, Raúl Domínguez, Juan José Ramos-Álvarez, Francisco Miguel Tobal, Pablo Jodra, Rachel Tan, and Stephen J. Bailey. 2020. "The Effect of Dietary Nitrate Supplementation on Isokinetic Torque in Adults: A Systematic Review and Meta-Analysis" Nutrients 12, no. 10: 3022. https://doi.org/10.3390/nu12103022
APA StyleLago-Rodríguez, Á., Domínguez, R., Ramos-Álvarez, J. J., Tobal, F. M., Jodra, P., Tan, R., & Bailey, S. J. (2020). The Effect of Dietary Nitrate Supplementation on Isokinetic Torque in Adults: A Systematic Review and Meta-Analysis. Nutrients, 12(10), 3022. https://doi.org/10.3390/nu12103022









