Antidiabetic Properties of Curcumin II: Evidence from In Vivo Studies
Abstract
1. Introduction
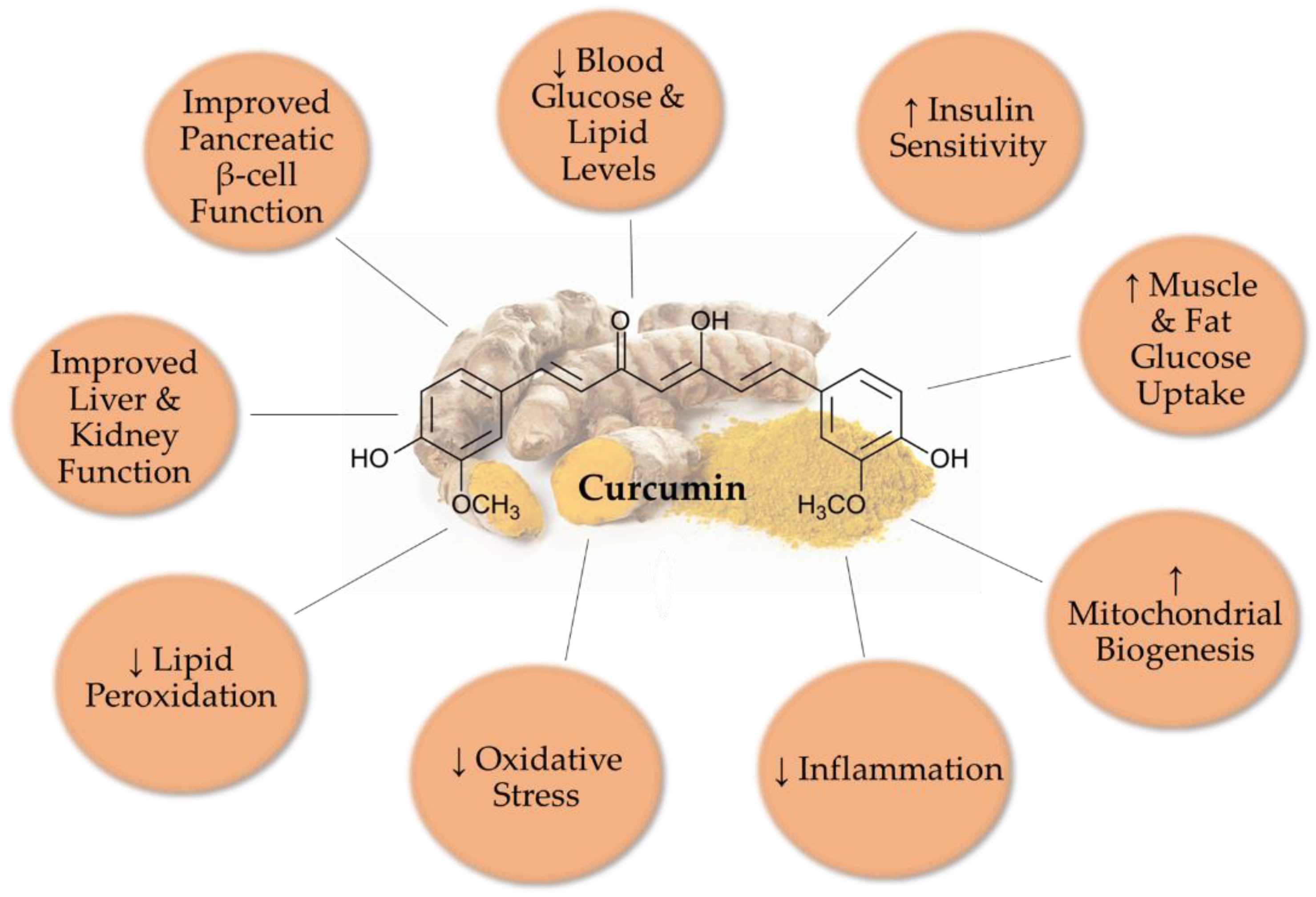
2. Antidiabetic Effects of Curcumin: In Vivo Animal Studies
2.1. Streptozotocin (STZ)-Induced Diabetes Model
2.2. Alloxan-Induced Diabetes Model
2.3. Genetic Diabetes Model
2.4. Diet-Induced Diabetes Model
3. Biological Effects of Curcumin: Human Studies
4. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Tripathy, D.; Chavez, A.O. Defects in insulin secretion and action in the pathogenesis of type 2 diabetes mellitus. Curr. Diab. Rep. 2010, 10, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, A.R. New perspectives into the molecular pathogenesis and treatment of type 2 diabetes. Cell 2001, 104, 517–529. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A. Dysfunctional fat cells, lipotoxicity and type 2 diabetes. Int. J. Clin. Pract. Suppl. 2004, 58, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Olson, P.; Hevener, A.; Mehl, I.; Chong, L.-W.; Olefsky, J.M.; Gonzalez, F.J.; Ham, J.; Kang, H.; Peters, J.M.; et al. PPAR regulates glucose metabolism and insulin sensitivity. Proc. Natl. Acad. Sci. USA 2006, 103, 3444–3449. [Google Scholar] [CrossRef]
- Defronzo, R.A. Banting Lecture. From the triumvirate to the ominous octet: A new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009, 58, 773–795. [Google Scholar] [CrossRef]
- Frigolet, M.E.; Torres, N.; Tovar, A.R. The renin-angiotensin system in adipose tissue and its metabolic consequences during obesity. J. Nutr. Biochem. 2013, 24, 2003–2015. [Google Scholar] [CrossRef]
- Vieira, A.R.; Abar, L.; Vingeliene, S.; Chan, D.S.M.; Aune, D.; Navarro-Rosenblatt, D.; Stevens, C.; Greenwood, D.; Norat, T. Fruits, vegetables and lung cancer risk: A systematic review and meta-analysis. Ann. Oncol. 2016, 27, 81–96. [Google Scholar] [CrossRef]
- Kuzma, J.N.; Schmidt, K.A.; Kratz, M. Prevention of metabolic diseases: Fruits (including fruit sugars) vs. vegetables. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 286–293. [Google Scholar] [CrossRef]
- Stefan, N.; Häring, H.-U.; Schulze, M.B. Metabolically healthy obesity: The low-hanging fruit in obesity treatment? Lancet Diabetes Endocrinol. 2018, 6, 249–258. [Google Scholar] [CrossRef]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Park, E.-J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113. [Google Scholar] [CrossRef]
- Serino, A.; Salazar, G. Protective Role of Polyphenols against Vascular Inflammation, Aging and Cardiovascular Disease. Nutrients 2018, 11, 53. [Google Scholar] [CrossRef]
- Moore, J.; Yousef, M.; Tsiani, E. Anticancer Effects of Rosemary (Rosmarinus officinalis L.) Extract and Rosemary Extract Polyphenols. Nutrients 2016, 8, 731. [Google Scholar] [CrossRef]
- Yousef, M.; Vlachogiannis, I.A.; Tsiani, E. Effects of Resveratrol against Lung Cancer: In Vitro and In Vivo Studies. Nutrients 2017, 9, 1231. [Google Scholar] [CrossRef] [PubMed]
- Naimi, M.; Vlavcheski, F.; Shamshoum, H.; Tsiani, E. Rosemary Extract as a Potential Anti-Hyperglycemic Agent: Current Evidence and Future Perspectives. Nutrients 2017, 9, 968. [Google Scholar] [CrossRef] [PubMed]
- Dreosti, I.E. Antioxidant polyphenols in tea, cocoa, and wine. Nutrition 2000, 16, 692–694. [Google Scholar] [CrossRef]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Amor, S.; Châlons, P.; Aires, V.; Delmas, D. Polyphenol Extracts from Red Wine and Grapevine: Potential Effects on Cancers. Diseases 2018, 6, 106. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef]
- Kocaadam, B.; Şanlier, N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit. Rev. Food Sci. Nutr. 2017, 57, 2889–2895. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, I.; Biswas, K.; Bandyopadhyay, U.; Banerjee, R.K. Turmeric and curcumin: Biological actions and medicinal applications. Cur. Sci. 2004, 87, 10. [Google Scholar]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Meng, B.; Li, J.; Cao, H. Antioxidant and antiinflammatory activities of curcumin on diabetes mellitus and its complications. Curr. Pharm. Des. 2013, 19, 2101–2113. [Google Scholar]
- Babu, P.S.; Srinivasan, K. Amelioration of renal lesions associated with diabetes by dietary curcumin in streptozotocin diabetic rats. Mol. Cell. Biochem. 1998, 181, 87–96. [Google Scholar] [CrossRef]
- Ali Hussain, H.E.M. Hypoglycemic, hypolipidemic and antioxidant properties of combination ofCurcumin fromCurcuma longa, Linn, and partially purified product fromAbroma augusta, Linn. in streptozotocin induced diabetes. Indian J. Clin. Biochem. 2002, 17, 33–43. [Google Scholar] [CrossRef]
- Murugan, P.; Pari, L. Antioxidant effect of tetrahydrocurcumin in streptozotocin–nicotinamide induced diabetic rats. Life Sci. 2006, 79, 1720–1728. [Google Scholar] [CrossRef]
- Sharma, S.; Kulkarni, S.K.; Chopra, K. Curcumin, the Active Principle of Turmeric (curcuma Longa), Ameliorates Diabetic Nephropathy in Rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 940–945. [Google Scholar] [CrossRef]
- Suryanarayana, P.; Satyanarayana, A.; Balakrishna, N.; Kumar, P.U.; Reddy, G.B. Effect of turmeric and curcumin on oxidative stress and antioxidant enzymes in streptozotocin-induced diabetic rat. Med. Sci. Monit. 2007, 13, BR286–BR292. [Google Scholar]
- Tikoo, K.; Meena, R.L.; Kabra, D.G.; Gaikwad, A.B. Change in post-translational modifications of histone H3, heat-shock protein-27 and MAP kinase p38 expression by curcumin in streptozotocin-induced type I diabetic nephropathy. Br. J. Pharm. 2008, 153, 1225–1231. [Google Scholar] [CrossRef]
- Kanitkar, M.; Gokhale, K.; Galande, S.; Bhonde, R.R. Novel role of curcumin in the prevention of cytokine-induced islet death in vitro and diabetogenesis in vivo. Br. J. Pharm. 2008, 155, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Chiu, J.; Khan, Z.A.; Farhangkhoee, H.; Chakrabarti, S. Curcumin prevents diabetes-associated abnormalities in the kidneys by inhibiting p300 and nuclear factor-κB. Nutrition 2009, 25, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Karthikesan, K.; Menon, V.P. Comparative and combined effect of chlorogenic acid and tetrahydrocurcumin on antioxidant disparities in chemical induced experimental diabetes. Mol. Cell. Biochem. 2010, 341, 109–117. [Google Scholar] [CrossRef] [PubMed]
- El-Azab, M.F.; Attia, F.M.; El-Mowafy, A.M. Novel role of curcumin combined with bone marrow transplantation in reversing experimental diabetes: Effects on pancreatic islet regeneration, oxidative stress, and inflammatory cytokines. Eur. J. Pharm. 2011, 658, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Soetikno, V.; Watanabe, K.; Sari, F.R.; Harima, M.; Thandavarayan, R.A.; Veeraveedu, P.T.; Arozal, W.; Sukumaran, V.; Lakshmanan, A.P.; Arumugam, S.; et al. Curcumin attenuates diabetic nephropathy by inhibiting PKC-α and PKC-β1 activity in streptozotocin-induced type I diabetic rats. Mol. Nutr. Food Res. 2011, 55, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Soetikno, V.; Sari, F.R.; Veeraveedu, P.T.; Thandavarayan, R.A.; Harima, M.; Sukumaran, V.; Lakshmanan, A.P.; Suzuki, K.; Kawachi, H.; Watanabe, K. Curcumin ameliorates macrophage infiltration by inhibiting NF-κB activation and proinflammatory cytokines in streptozotocin induced-diabetic nephropathy. Nutr. Metab. (Lond.) 2011, 8, 35. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, Y.; Cai, L.; Cai, Y.; Hu, J.; Yu, C.; Li, J.; Feng, Z.; Yang, S.; Li, X.; et al. Inhibition of high glucose-induced inflammatory response and macrophage infiltration by a novel curcumin derivative prevents renal injury in diabetic rats. Br. J. Pharm. 2012, 166, 1169–1182. [Google Scholar] [CrossRef]
- Gutierres, V.O.; Pinheiro, C.M.; Assis, R.P.; Vendramini, R.C.; Pepato, M.T.; Brunetti, I.L. Curcumin-supplemented yoghurt improves physiological and biochemical markers of experimental diabetes. Br. J. Nutr. 2012, 108, 440–448. [Google Scholar] [CrossRef]
- Abdel Aziz, M.T.; El-Asmar, M.F.; El-Ibrashy, I.N.; Rezq, A.M.; Al-Malki, A.L.; Wassef, M.A.; Fouad, H.H.; Ahmed, H.H.; Taha, F.M.; Hassouna, A.A.; et al. Effect of novel water soluble curcumin derivative on experimental type- 1 diabetes mellitus (short term study). Diabetol. Metab. Syndr. 2012, 4, 30. [Google Scholar] [CrossRef]
- Pan, Y.; Zhu, G.; Wang, Y.; Cai, L.; Cai, Y.; Hu, J.; Li, Y.; Yan, Y.; Wang, Z.; Li, X.; et al. Attenuation of high-glucose-induced inflammatory response by a novel curcumin derivative B06 contributes to its protection from diabetic pathogenic changes in rat kidney and heart. J. Nutr. Biochem. 2013, 24, 146–155. [Google Scholar] [CrossRef]
- Soetikno, V.; Sari, F.R.; Sukumaran, V.; Lakshmanan, A.P.; Harima, M.; Suzuki, K.; Kawachi, H.; Watanabe, K. Curcumin decreases renal triglyceride accumulation through AMPK–SREBP signaling pathway in streptozotocin-induced type 1 diabetic rats. J. Nutr. Biochem. 2013, 24, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Huang, Y.; Wang, Z.; Fang, Q.; Sun, Y.; Tong, C.; Peng, K.; Wang, Y.; Miao, L.; Cai, L.; et al. Inhibition of MAPK-mediated ACE expression by compound C66 prevents STZ-induced diabetic nephropathy. J. Cell. Mol. Med. 2014, 18, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.-N.; Yang, Z.-Y.; Lv, S.-S.; Liu, X.-C.; Guan, G.-J.; Liu, G. Curcumin prevents diabetic nephropathy against inflammatory response via reversing caveolin-1 Tyr14 phosphorylation influenced TLR4 activation. Int. Immunopharmacol. 2014, 23, 236–246. [Google Scholar] [CrossRef]
- Sun, L.; Chen, Z.; Liu, X.; Liu, H.; Guan, G.; Liu, G. Curcumin ameliorates epithelial-to-mesenchymal transition of podocytes in vivo and in vitro via regulating caveolin-1. Biomed. Pharmacother. 2014, 68, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Abd Allah, E.S.H.; Gomaa, A.M.S. Effects of curcumin and captopril on the functions of kidney and nerve in streptozotocin-induced diabetic rats: Role of angiotensin converting enzyme 1. Appl. Physiol. Nutr. Metab. 2015, 40, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.-N.; Liu, X.-C.; Chen, X.-J.; Guan, G.-J.; Liu, G. Curcumin attenuates high glucose-induced podocyte apoptosis by regulating functional connections between caveolin-1 phosphorylation and ROS. Acta Pharm. Sin. 2016, 37, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.S.; Alkafafy, M.E.; Ahmed, M.M.; Soliman, M.M. Renoprotective effect of curcumin against the combined oxidative stress of diabetes and nicotine in rats. Mol. Med. Rep. 2016, 13, 3017–3026. [Google Scholar] [CrossRef]
- Rashid, K.; Chowdhury, S.; Ghosh, S.; Sil, P.C. Curcumin attenuates oxidative stress induced NFκB mediated inflammation and endoplasmic reticulum dependent apoptosis of splenocytes in diabetes. Biochem. Pharmacol. 2017, 143, 140–155. [Google Scholar] [CrossRef]
- Ganugula, R.; Arora, M.; Jaisamut, P.; Wiwattanapatapee, R.; Jørgensen, H.G.; Venkatpurwar, V.P.; Zhou, B.; Rodrigues Hoffmann, A.; Basu, R.; Guo, S.; et al. Nano-curcumin safely prevents streptozotocin-induced inflammation and apoptosis in pancreatic beta cells for effective management of Type 1 diabetes mellitus. Br. J. Pharm. 2017, 174, 2074–2084. [Google Scholar] [CrossRef]
- Arun, N.; Nalini, N. Efficacy of turmeric on blood sugar and polyol pathway in diabetic albino rats. Plant. Foods Hum. Nutr. 2002, 57, 41–52. [Google Scholar] [CrossRef]
- Das, K.K.; Razzaghi-Asl, N.; Tikare, S.N.; Di Santo, R.; Costi, R.; Messore, A.; Pescatori, L.; Crucitti, G.C.; Jargar, J.G.; Dhundasi, S.A.; et al. Hypoglycemic activity of curcumin synthetic analogues in alloxan-induced diabetic rats. J. Enzym. Inhib. Med. Chem. 2016, 31, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, M.; Mimaki, Y.; Nishiyama, T.; Mae, T.; Kishida, H.; Tsukagawa, M.; Takahashi, K.; Kawada, T.; Nakagawa, K.; Kitahara, M. Hypoglycemic effects of turmeric (Curcuma longa L. rhizomes) on genetically diabetic KK-Ay mice. Biol. Pharm. Bull. 2005, 28, 937–939. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, T.; Mae, T.; Kishida, H.; Tsukagawa, M.; Mimaki, Y.; Kuroda, M.; Sashida, Y.; Takahashi, K.; Kawada, T.; Nakagawa, K.; et al. Curcuminoids and sesquiterpenoids in turmeric (Curcuma longa L.) suppress an increase in blood glucose level in type 2 diabetic KK-Ay mice. J. Agric. Food Chem. 2005, 53, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.-I.; Choi, M.-S.; Jung, U.J.; Kim, H.-J.; Yeo, J.; Jeon, S.-M.; Lee, M.-K. Effect of curcumin supplementation on blood glucose, plasma insulin, and glucose homeostasis related enzyme activities in diabetic db/db mice. Mol. Nutr. Food Res. 2008, 52, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Tao, L.; Mei, W.; Luo, R.; Fu, X.; Wang, L.; Yang, W.; Liu, C. Effect of curcumin on the expression of p-STAT3 and IκB in db/db mice. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2014, 39, 591–597. [Google Scholar] [PubMed]
- Soto-Urquieta, M.G.; López-Briones, S.; Pérez-Vázquez, V.; Saavedra-Molina, A.; González-Hernández, G.A.; Ramírez-Emiliano, J. Curcumin restores mitochondrial functions and decreases lipid peroxidation in liver and kidneys of diabetic db/db mice. Biol. Res. 2014, 47, 74. [Google Scholar] [CrossRef] [PubMed]
- Kawano, K.; Hirashima, T.; Mori, S.; Natori, T. OLETF (Otsuka Long-Evans Tokushima Fatty) rat: A new NIDDM rat strain. Diabetes Res. Clin. Pract. 1994, 24, S317–S320. [Google Scholar] [CrossRef]
- Kim, B.H.; Lee, E.S.; Choi, R.; Nawaboot, J.; Lee, M.Y.; Lee, E.Y.; Kim, H.S.; Chung, C.H. Protective Effects of Curcumin on Renal Oxidative Stress and Lipid Metabolism in a Rat Model of Type 2 Diabetic Nephropathy. Yonsei Med. J. 2016, 57, 664–673. [Google Scholar] [CrossRef]
- Weisberg, S.; Leibel, R.; Tortoriello, D.V. Proteasome inhibitors, including curcumin, improve pancreatic β-cell function and insulin sensitivity in diabetic mice. Nutr. Diabetes 2016, 6, e205. [Google Scholar] [CrossRef]
- Lu, M.; Yin, N.; Liu, W.; Cui, X.; Chen, S.; Wang, E. Curcumin Ameliorates Diabetic Nephropathy by Suppressing NLRP3 Inflammasome Signaling. Biomed. Res. Int. 2017. [Google Scholar] [CrossRef]
- Asai, A.; Miyazawa, T. Dietary curcuminoids prevent high-fat diet-induced lipid accumulation in rat liver and epididymal adipose tissue. J. Nutr. 2001, 131, 2932–2935. [Google Scholar] [CrossRef] [PubMed]
- Arafa, H.M.M. Curcumin attenuates diet-induced hypercholesterolemia in rats. Med. Sci. Monit. 2005, 11, BR228–BR234. [Google Scholar] [PubMed]
- Shao, W.; Yu, Z.; Chiang, Y.; Yang, Y.; Chai, T.; Foltz, W.; Lu, H.; Fantus, I.G.; Jin, T. Curcumin prevents high fat diet induced insulin resistance and obesity via attenuating lipogenesis in liver and inflammatory pathway in adipocytes. PLoS ONE 2012, 7, e28784. [Google Scholar] [CrossRef] [PubMed]
- He, H.-J.; Wang, G.-Y.; Gao, Y.; Ling, W.-H.; Yu, Z.-W.; Jin, T.-R. Curcumin attenuates Nrf2 signaling defect, oxidative stress in muscle and glucose intolerance in high fat diet-fed mice. World J. Diabetes 2012, 3, 94–104. [Google Scholar] [CrossRef]
- Martínez-Morúa, A.; Soto-Urquieta, M.G.; Franco-Robles, E.; Zúñiga-Trujillo, I.; Campos-Cervantes, A.; Pérez-Vázquez, V.; Ramírez-Emiliano, J. Curcumin decreases oxidative stress in mitochondria isolated from liver and kidneys of high-fat diet-induced obese mice. J. Asian Nat. Prod. Res. 2013, 15, 905–915. [Google Scholar] [CrossRef]
- Maithili Karpaga Selvi, N.; Sridhar, M.G.; Swaminathan, R.P.; Sripradha, R. Curcumin Attenuates Oxidative Stress and Activation of Redox-Sensitive Kinases in High Fructose-and High-Fat-Fed Male Wistar Rats. Sci. Pharm. 2015, 83, 159–175. [Google Scholar] [CrossRef]
- Kelany, M.E.; Hakami, T.M.; Omar, A.H. Curcumin improves the metabolic syndrome in high-fructose-diet-fed rats: Role of TNF-α, NF-κB, and oxidative stress. Can. J. Physiol. Pharm. 2016, 95, 140–150. [Google Scholar] [CrossRef]
- Ding, L.; Li, J.; Song, B.; Xiao, X.; Zhang, B.; Qi, M.; Huang, W.; Yang, L.; Wang, Z. Curcumin rescues high fat diet-induced obesity and insulin sensitivity in mice through regulating SREBP pathway. Toxicol. Appl. Pharm. 2016, 304, 99–109. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, B.; Huang, F.; Liu, B.; Xie, Y. Curcumin inhibits lipolysis via suppression of ER stress in adipose tissue and prevents hepatic insulin resistance. J. Lipid Res. 2016, 57, 1243–1255. [Google Scholar] [CrossRef]
- Wickenberg, J.; Ingemansson, S.L.; Hlebowicz, J. Effects of Curcuma longa (turmeric) on postprandial plasma glucose and insulin in healthy subjects. Nutr. J. 2010, 9, 43. [Google Scholar] [CrossRef]
- Sukandar, E.Y.; Permana, H.; Adnyana, I.K.; Sigit, J.I.; Llyas, R.A.; Hasimun, P.; Mardiyah, D. Clinical Study of Turmeric (Curcuma longa L.) and Garlic (Allium sativum L.) Extracts as Antihyperglycemic and Antihyperlipidemic Agent in Type-2 Diabetes-Dyslipidemia Patients. Int. J. Pharmacol. 2010, 6, 456–463. [Google Scholar] [CrossRef]
- Sukandar, E.Y.; Sudjana, P.; Adnyana, I.K.; Setiawan, A.S.; Yuniarni, U. Recent Study of Turmeric in Combination with Garlic as Antidiabetic Agent. Procedia Chem. 2014, 13, 44–56. [Google Scholar] [CrossRef]
- Khajehdehi, P.; Pakfetrat, M.; Javidnia, K.; Azad, F.; Malekmakan, L.; Nasab, M.H.; Dehghanzadeh, G. Oral supplementation of turmeric attenuates proteinuria, transforming growth factor-β and interleukin-8 levels in patients with overt type 2 diabetic nephropathy: A randomized, double-blind and placebo-controlled study. Scand. J. Urol. Nephrol. 2011, 45, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Appendino, G.; Belcaro, G.; Cornelli, U.; Luzzi, R.; Togni, S.; Dugall, M.; Cesarone, M.R.; Feragalli, B.; Ippolito, E.; Errichi, B.M.; et al. Potential role of curcumin phytosome (Meriva) in controlling the evolution of diabetic microangiopathy. A pilot study. Panminerva Med. 2011, 53, 43–49. [Google Scholar] [PubMed]
- Steigerwalt, R.; Nebbioso, M.; Appendino, G.; Belcaro, G.; Ciammaichella, G.; Cornelli, U.; Luzzi, R.; Togni, S.; Dugall, M.; Cesarone, M.R.; et al. Meriva®, a lecithinized curcumin delivery system, in diabetic microangiopathy and retinopathy. Panminerva Med. 2012, 54, 11–16. [Google Scholar] [PubMed]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Luechapudiporn, R.; Phisalaphong, C.; Jirawatnotai, S. Curcumin Extract for Prevention of Type 2 Diabetes. Diabetes Care 2012, 35, 2121–2127. [Google Scholar] [CrossRef] [PubMed]
- Na, L.-X.; Li, Y.; Pan, H.-Z.; Zhou, X.-L.; Sun, D.-J.; Meng, M.; Li, X.-X.; Sun, C.-H. Curcuminoids exert glucose-lowering effect in type 2 diabetes by decreasing serum free fatty acids: A double-blind, placebo-controlled trial. Mol. Nutr. Food Res. 2013, 57, 1569–1577. [Google Scholar] [CrossRef]
- Grant, S.J.; Chang, D.H.-T.; Liu, J.; Wong, V.; Kiat, H.; Bensoussan, A. Chinese herbal medicine for impaired glucose tolerance: A randomized placebo controlled trial. BMC Complement. Altern Med. 2013, 13, 104. [Google Scholar] [CrossRef]
- Mohammadi, A.; Sahebkar, A.; Iranshahi, M.; Amini, M.; Khojasteh, R.; Ghayour-Mobarhan, M.; Ferns, G.A. Effects of supplementation with curcuminoids on dyslipidemia in obese patients: A randomized crossover trial. Phytother Res. 2013, 27, 374–379. [Google Scholar] [CrossRef]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Phonrat, B.; Tungtrongchitr, R.; Jirawatnotai, S. Reduction of atherogenic risk in patients with type 2 diabetes by curcuminoid extract: A randomized controlled trial. J. Nutr. Biochem. 2014, 25, 144–150. [Google Scholar] [CrossRef]
- Yang, Y.-S.; Su, Y.-F.; Yang, H.-W.; Lee, Y.-H.; Chou, J.I.; Ueng, K.-C. Lipid-lowering effects of curcumin in patients with metabolic syndrome: A randomized, double-blind, placebo-controlled trial. Phytother Res. 2014, 28, 1770–1777. [Google Scholar] [CrossRef] [PubMed]
- Neerati, P.; Devde, R.; Gangi, A.K. Evaluation of the effect of curcumin capsules on glyburide therapy in patients with type-2 diabetes mellitus. Phytother Res. 2014, 28, 1796–1800. [Google Scholar] [CrossRef] [PubMed]
- Na, L.X.; Yan, B.L.; Jiang, S.; Cui, H.L.; Li, Y.; Sun, C.H. Curcuminoids Target Decreasing Serum Adipocyte-fatty Acid Binding Protein Levels in Their Glucose-lowering Effect in Patients with Type 2 Diabetes. Biomed. Environ. Sci. 2014, 27, 902–906. [Google Scholar] [PubMed]
- Di Pierro, F.; Bressan, A.; Ranaldi, D.; Rapacioli, G.; Giacomelli, L.; Bertuccioli, A. Potential role of bioavailable curcumin in weight loss and omental adipose tissue decrease: Preliminary data of a randomized, controlled trial in overweight people with metabolic syndrome. Preliminary study. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 4195–4202. [Google Scholar] [PubMed]
- Yang, H.; Xu, W.; Zhou, Z.; Liu, J.; Li, X.; Chen, L.; Weng, J.; Yu, Z. Curcumin attenuates urinary excretion of albumin in type II diabetic patients with enhancing nuclear factor erythroid-derived 2-like 2 (Nrf2) system and repressing inflammatory signaling efficacies. Exp. Clin. Endocrinol. Diabetes 2015, 123, 360–367. [Google Scholar] [CrossRef]
- Rahimi, H.R.; Mohammadpour, A.H.; Dastani, M.; Jaafari, M.R.; Abnous, K.; Ghayour Mobarhan, M.; Kazemi Oskuee, R. The effect of nano-curcumin on HbA1c, fasting blood glucose, and lipid profile in diabetic subjects: A randomized clinical trial. Avicenna J. Phytomed. 2016, 6, 567–577. [Google Scholar]
- Jiménez-Osorio, A.S.; García-Niño, W.R.; González-Reyes, S.; Álvarez-Mejía, A.E.; Guerra-León, S.; Salazar-Segovia, J.; Falcón, I.; Montes de Oca-Solano, H.; Madero, M.; Pedraza-Chaverri, J. The Effect of Dietary Supplementation with Curcumin on Redox Status and Nrf2 Activation in Patients with Nondiabetic or Diabetic Proteinuric Chronic Kidney Disease: A Pilot Study. J. Ren. Nutr. 2016, 26, 237–244. [Google Scholar] [CrossRef]
- Panahi, Y.; Khalili, N.; Sahebi, E.; Namazi, S.; Reiner, Ž.; Majeed, M.; Sahebkar, A. Curcuminoids modify lipid profile in type 2 diabetes mellitus: A randomized controlled trial. Complement. Ther. Med. 2017, 33, 1–5. [Google Scholar] [CrossRef]
- Panahi, Y.; Khalili, N.; Sahebi, E.; Namazi, S.; Simental-Mendía, L.E.; Majeed, M.; Sahebkar, A. Effects of Curcuminoids Plus Piperine on Glycemic, Hepatic and Inflammatory Biomarkers in Patients with Type 2 Diabetes Mellitus: A Randomized Double-Blind Placebo-Controlled Trial. Drug Res. (Stuttg.) 2018, 68, 403–409. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Fogacci, F.; Morbini, M.; Colletti, A.; Bove, M.; Veronesi, M.; Giovannini, M.; Borghi, C. Nutraceutical Effects on Glucose and Lipid Metabolism in Patients with Impaired Fasting Glucose: A Pilot, Double-Blind, Placebo-Controlled, Randomized Clinical Trial on a Combined Product. High Blood Press. Cardiovasc. Prev. 2017, 24, 283–288. [Google Scholar] [CrossRef][Green Version]
- Hodaei, H.; Adibian, M.; Nikpayam, O.; Hedayati, M.; Sohrab, G. The effect of curcumin supplementation on anthropometric indices, insulin resistance and oxidative stress in patients with type 2 diabetes: A randomized, double-blind clinical trial. Diabetol. Metab. Syndr. 2019, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Tan, M.; Zhong, Z.; Chen, M.; Wang, Y. Nanotechnologies for Curcumin: An Ancient Puzzler Meets Modern Solutions. Available online: https://www.hindawi.com/journals/jnm/2011/723178/ (accessed on 17 December 2019).
- Flora, G.; Gupta, D.; Tiwari, A. Nanocurcumin: A promising therapeutic advancement over native curcumin. Crit. Rev. Ther. Drug Carrier Syst. 2013, 30, 331–368. [Google Scholar] [CrossRef] [PubMed]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Bolignano, D.; Cernaro, V.; Gembillo, G.; Baggetta, R.; Buemi, M.; D’Arrigo, G. Antioxidant agents for delaying diabetic kidney disease progression: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0178699. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: Systematic review and meta-analysis. JAMA 2007, 297, 842–857. [Google Scholar] [CrossRef] [PubMed]
- Katsiki, N.; Manes, C. Is there a role for supplemented antioxidants in the prevention of atherosclerosis? Clin. Nutr. 2009, 28, 3–9. [Google Scholar] [CrossRef]
- Halliwell, B. The antioxidant paradox: Less paradoxical now? Br. J. Clin. Pharm. 2013, 75, 637–644. [Google Scholar] [CrossRef]
- Liu, Z.-Q. Antioxidants may not always be beneficial to health. Nutrition 2014, 30, 131–133. [Google Scholar] [CrossRef]
| Animal | Curcumin Concentration/Duration | Serum Effects | Other Effects | Reference |
|---|---|---|---|---|
| Albino Wistar rats | 0.5% of diet; 8 weeks | ↓ Phospholipid ↓ Triglyceride ↑ PUFA/SFA ratio | ↓ Kidney weight ↓ Renal lesion progression ↓ Renal damage ↓ Urine ALT and AST ↓ Kidney alkaline and acid phosphatase ↓ Glucose-6- phosphatase ↓ Lactate dehydrogenase ↑ ATPase activity | [25] |
| Albino Wistar rats | 300 mg/kg b.w./day; 8 weeks | ↓ Glucose ↓ Cholesterol ↓ Triglyceride ↓ Urea ↑ Creatinine | ↓ Body weight ↓ Kidney lipid peroxidation ↑ Kidney Creatinine excreted ↑ Kidney SOD activity ↑ Kidney Catalase activity | [26] |
| Wistar Rats | 80 mg/kg b.w./day; 45 days | ↓ Glucose ↑ Insulin | ↓ Lipid peroxidation Kidney: ↑ SOD ↑ Catalase ↑ GPx activity ↑ Glutathione-S-transferase Kidney and liver: ↓ TBARS ↓ H2O2 | [27] |
| Sprague–Dawley rats | 15 and 30 mg/kg b.w./day; 2 weeks | ↓ Glucose ↓ Creatinine | ↓ Renal changes ↓ Oxidative stress ↓ Urine albumin ↓ Proteinuria ↑ Creatinine clearance ↓ Lipid peroxidation Kidney: ↓ MDA ↑ SOD activity ↑ Catalase activity | [28] |
| Wistar-NIN rats | 0.01% curcumin; 8 weeks | ↓ Glucose ↓ Insulin ↑ SOD activity | ↓ TBARS ↑ Pancreas catalase activity ↓ Pancreas SOD activity ↓ Glutathione-S-transferase activity | [29] |
| Sprague–Dawley rats | 50 mg/kg b.w./day; 6 weeks | ↓ Urea ↓ Creatinine | ↑ Albumin ↓ HSP-27 protein ↓ p38 protein ↑ Acetyl-histone H3 ↑ Phospho-histone H3 | [30] |
| C57/BL6J mice | 7.5 mg/kg b.w./day; 10 h prior to STZ | ↓ Glucose ↑ Insulin ↓ IL-16 ↓ TNF-α | ↑ Glucose clearance ↑ GLUT2 mRNA ↓ Pancreatic IL-6 and TNF-α | [31] |
| Sprague-Dawley rats | 150 mg/kg b.w./day; 1 month | ↓ Glucose | ↓ Body weight ↓ eNOS ↓ TGF-β1 mRNA ↓ Endothelin-1 mRNA ↓ Fibronectin mRNA ↓ EBF mRNA ↓ Oxidative stress ↓ HO-1 mRNA ↓ Nuclear 8-OHdG ↓ Cytosolic NT ↓ Mesangial expansion ↓ p300 activity ↓ NF-κB activity | [32] |
| Wistar rats | 80 mg/kg b.w./day; 45 days | ↓ Glucose ↑ Insulin | Kidney and liver: ↓ Morphological changes ↓ Oxidative stress ↓ TBARS ↓ HP ↑ SOD activity ↑ CAT activity ↑ GPx activity ↑ Glutathione activity | [33] |
| Swiss albino mice | 10 mM; 10 µL/mouse i.p.; 28 days and 106 BMCs, single injection | ↓ Glucose ↑ Insulin ↓ TNF-α ↓ IL-1β | ↑ Islet regeneration Pancreas: ↓ MDA levels ↑ SOD activity ↑ Catalase activity ↑ GPx activity | [34] |
| Sprague–Dawley rats | 100 mg/kg b.w./day; 8 weeks | ↓ Glucose ↓ Urea ↓ Creatinine | Kidney: ↓ Histological changes ↓ PKC-α total and membrane protein ↓ PKC-β1 total and membrane protein ↓ P300 mRNA levels ↓ NOX4 and p67phox protein ↓ MDA levels ↑ GPx levels ↓ TGF-β1, VEGF, CTGF and OPN protein ↓ Type IV collagen, fibronectin and Flk-1 | [35] |
| Sprague–Dawley rats | 100 mg/kg b.w./day; 8 weeks | ↓ Glucose ↓ Urea ↓ Creatinine | Kidney: ↓ Glomerular and tubular histological changes ↓ Segmental sclerosis ↓ Macrophage infiltration ↓ TNF-α and IL-1β mRNA and protein ↑ IκBα protein ↓ ICAM-1, MCP-1 and TGF-β1 protein ↓ Phospho-NF-κB | [36] |
| Sprague–Dawley rats | 1 and 5 mg/kg b.w./day; 6 weeks | ↓ TNF-α ↓ Creatinine | Kidney: ↓ Fibrosis ↓ TNF-α mRNA ↓ iNOS mRNA ↓ COX-2 mRNA ↓ TGF-β mRNA ↓ MCP-1 mRNA ↓ Glycogen levels ↓ Type IV collagen expression | [37] |
| Wistar rats | 60 and 90 mg/kg b.w./day; 31 days | ↓ Glucose ↓ Triglyceride | ↓ Proteinuria ↓ Hepatic AST activity ↓ Hepatic ALT activity ↓ Hepatic ALP activity | [38] |
| Curl:HEL1 rats | 10 mg/kg b.w./day; 45 days | ↓ Glucose ↑ Insulin ↓ Cholesterol ↓ Triglyceride ↓ LDL ↑ HDL | ↓ Pancreas, aorta and liver MDA levels | [39] |
| Sprague–Dawley rats | 0.2 mg/kg b.w./day; 6 weeks | ↓ TNF-α ↓ Creatinine ↓ Nitrite | ↓ Renal macrophage infiltration Kidney and cardiac: ↓ TNF-α, COX-2, TGF-β and MCP-1 ↓ Glycogen ↓ Collagen IV expression | [40] |
| Sprague–Dawley rats | 100 mg/kg b.w./day; 8 weeks | ↓ Triglyceride | Kidney: ↓ Renal triglyceride ↑ Phospho-AMPK ↓ SREBP-1c protein ↓ TGF-β protein ↓ EGF protein ↓ Type IV collagen protein ↓ Fibronectin protein ↑ Nephrin protein | [41] |
| C57BL/6 mice | 5 mg/kg b.w./second day; 67 days | ↓ Creatinine ↓ Albumin ↓ Total protein ↓ Ang II | Kidney: ↓ Fibrosis ↓ Macrophage infiltration ↓ Glycogen levels ↓ Collagen IV ↓ ACE mRNA ↓ Phospho-ERK ↓ Phospho-p38 ↓ Phospho-JNK | [42] |
| Wistar rats | 100 mg/kg b.w./day; 12 weeks | No measured effects | ↓ Renal fibrosis ↓ Kidney weight ↓ IL-6, IL-1β and TNF-α protein ↓ Urinary protein ↓ Glomerulosclerosis index ↓ Fibronectin and collagen IV protein ↓ Glycogen ↓ Phospho-Cav-1 ↓ TLR4 mRNA | [43] |
| Wistar rats | 100 mg/kg b.w./day; 12 weeks | ↓ Glucose | ↓ Renal fibrosis ↓ Kidney weight ↓ Urinary protein ↓ Glomerulosclerosis index ↑ P-cadherin and synaptopodin ↓ FSP-1, α-SMA and snail ↓ Phospho-Cav-1 | [44] |
| Wistar rats | 100 mg/kg b.w./day; 6 weeks | ↓ Glucose ↓ Cholesterol ↓ LDL ↓ Creatinine ↓ Urea | ↑ Body weight ↓ Kidney weight ↓ Nephropathy ↓ Renal ACE1 level Kidney and sciatic nerve: ↓ TNF-α ↓ IL-10 | [45] |
| Wistar rats | 100 and 200 mg/kg b.w./day; 8 weeks | No measured effects | ↓ Kidney weight ↓ Albumin excretion ↑ Creatinine clearance rate Kidney: ↑ SOD activity ↓ MDA content ↓ Bax and cleaved PARP protein ↓ Caspase-3 activity ↑ Bcl-2 protein ↑ Phospho-cav-1 | [46] |
| Wistar rats | 1.5 mg/kg b.w./day; 8 weeks | ↓ Cholesterol ↓ Triacylglycerol ↓ Phospholipid ↑ HDL ↓ Urea ↓ Creatinine | ↓ Oxidative stress ↓ MDA content ↓ γ-glutamyltranspeptidase level Kidney: ↓ NO ↑ SOD activity ↑ GPx activity ↓ Kidney morphological changes ↓ Vimentin mRNA ↓ Desmin mRNA ↓ SREBP-1 mRNA ↓ iNOS mRNA ↓ TGF-β1 mRNA ↑ Synaptopodin mRNA ↑ Connexin 43 mRNA ↑ Erythropoietin mRNA | [47] |
| Wistar rats | 100 mg/kg b.w./day; 8 weeks | ↓ Glucose ↑ Insulin | ↑ Splenic white pulp composition ↓ Splenic red pulp composition Spleen: ↓ ROS ↓ NO production ↓ Protein carboxylation ↑ SOD, catalase, glutathione, GPx and GSH activities ↓ TNF-α, IL-1β, IL-6, MCP-1, ICAM-1 and VCAM-1 ↓ TNF-α and IL-1β ↓ Nuclear NF-κB, COX-2 and iNOS ↑ Cytosolic NF-κB ↓ Caspase-12, calpain-1 and Bax ↑ Bcl-2 and mitochondrial cytochrome c ↓ Phospho-eIF2α, JNK, p38 and p53 | [48] |
| Sprague–Dawley rats | 50 mg/kg b.w./day; 28 days | ↓ Glucose ↑ Insulin | ↓ Islet death Pancreas: ↓ IL-1α, G-CSF, IL-10, IL-17A, IL-1β, IL-6, TNF-α, IL-4, GM-CSF, IFN-γ, IL-2, IL-5, IL-13 and IL-12p70 ↓ 8-oxo-dG | [49] |
| Animal | Curcumin Concentration/Duration | Serum Effects | Other Effects | Reference |
|---|---|---|---|---|
| Wistar rats | 0.08 mg/kg b.w./day; 21 days | ↓ Glucose ↓ HbA1c ↑ Hemoglobin ↓ TBARS ↑ Glutathione | Liver: ↓ Liver TBARS ↓ SDH activity ↑ Liver glutathione ↑ GPx activity | [50] |
| Wistar rats | 0.1 mg/kg b.w.; 2 h | ↓ Glucose | No measured effects | [51] |
| Animal | Curcumin Concentration/Duration | Serum Effects | Other Effects | Reference |
|---|---|---|---|---|
| KK-Ay mice | 1500 mg/kg b.w./day; 4 weeks | ↓ Glucose | No measured effects | [52] |
| KK-Ay mice | 137, 620 and 1500 mg/kg b.w./day; 4 weeks | ↓ Glucose | No measured effects | [53] |
| C57BL/KsJ-db/db Mice | 0.02%, wt/wt; 6 weeks | ↓ Glucose ↓ HbA1C ↑ Insulin ↓ Fatty acid ↓ Cholesterol ↓ Triglyceride | Liver: ↑ Glucokinase activity ↓ G6Pase activity ↓ PEPCK activity ↓ FAS activity ↓ β-oxidation activity ↓ CPT activity ↓ HMG-CoA reductase activity ↓ ACAT activity ↓ CAT activity ↓ GSH-Px activity ↓ MDA level | [54] |
| db/db mice | 200 mg/kg/day; 18 weeks | ↓ Glucose | ↓ Body weight Kidney: ↓ Albuminuria ↓ Glomerular sclerosis ↓ Mesangial area expansion ↓ Thickening of membrane ↓ ECM deposition ↓ Phospho-STAT3 ↑ IκB protein | [55] |
| db/db mice | 60 mg/kg/day; 4 weeks | ↓ Glucose | Kidney and liver: ↓ Mitochondrial dysfunction ↓ State 3 O2 consumption ↓ State 4 O2 consumption ↓ TBARS level Liver: ↑ ATPase activity ↑ NO synthesis | [56] |
| OLETF rats | 100 mg/kg/day; 45 weeks | ↓ Glucose ↓ Triglyceride ↓ Cholesterol ↑ Insulin | ↑ β cell function ↓ Glomerular hypertrophy ↓ GMB thickness ↓ Albuminuria Kidney: ↓ MDA ↑ SOD ↑ Nrf2 and HO-1 protein ↑ Phospho-AMPK and ACC ↓ SREBP-1, -2 and ADRP | [58] |
| Leprdb/db mice | 1500 mg/kg b.w./day | ↓ Glucose ↓ HbA1c ↑ Insulin | ↓ β-cell loss ↑ Ki-67-positive ↑ Insulin production ↑ Lean body mass ↑ Lifespan | [59] |
| C57BL/KsJ db/db mice | 200 mg/kg/day; 16 weeks | ↓ Glucose ↓ Creatinine ↓ Urea | ↓ Renal hypertrophy ↓ Glomerular matrix ↓ Kidney weight ↓ NLRP3 protein ↓ Collagen IV and fibronectin ↓ IL-1β protein ↓ Caspase-1 protein | [60] |
| Animal | Curcumin Concentration/Duration | Serum Effects | Other Effects | Reference |
|---|---|---|---|---|
| Sprague–Dawley rats | 1 g/100 g diet; 2 weeks | ↓ Triacylglycerol | ↓ Hepatic triacylglycerol levels ↑ Hepatic acyl-CoA oxidase ↓ Epididymal adipose tissue wt | [61] |
| Male swiss albino rats | 0.5% wt/wt; 7 days | ↓ Triglyceride ↓ Cholesterol ↓ LDL ↑ HDL ↓ AST activity ↓ ALT activity | No measured effects | [62] |
| C57BL/6J Mice | 0.4% dietary intake; 2 days/week for 28 weeks | ↓ Glucose ↓ Insulin ↑ Adiponectin | ↓ Body weight ↓ Epididymal fat mass ↓ Liver lipogenesis ↑ Liver and adipose phospho-Akt ↓ Liver and adipose phospho-GSKα/β ↓ Macrophage infiltration ↓ NF-κB protein ↓ Phospho-JNK ↓ ChREBP, SREBP-1 and TxNIP | [63] |
| C57BL/6J mice | 50 mg/kg b.w./day; 15 days | ↓ Glucose ↓ Insulin ↓ MDA | ↓ HOMA-IR Skeletal muscle: ↓ Mitochondrial MDA levels ↓ Mitochondrial ROS levels ↑ Nrf2 protein levels ↑ Nuclear Nrf2 protein levels | [64] |
| C57BL/6 mice | 0.5% and 1% wt/wt; 8 weeks | ↓ Triglyceride | ↓ Oxidative stress ↓ Mitochondrial dysfunction ↓ Oxygen consumption Kidney: ↑ State 3 O2 consumption ↑ State 4 O2 consumption ↑ mitochondrial NO synthesis ↓ TBARS levels Liver: ↓ Protein carbonyl ↓ TBARS levels | [65] |
| Wistar rats | 200 mg/kg b.w./day; 10 weeks | ↓ Glucose ↑ Glutathione ↑ GPx ↑ Catalase ↓ MDA ↓ Total oxidant | ↓ Body weight ↓ Oxidative stress Liver: ↓ MDA levels ↓ Total oxidant levels ↑ Total antioxidant levels ↓ Phospho-ERK ↓ Phospho-p38 | [66] |
| Wistar rats | 200 mg/kg b.w./day; 8 weeks | ↓ Glucose ↓ Insulin ↓ TNF-α ↓ Leptin ↓ Triglyceride ↓ Cholesterol ↓ Uric acid ↓ MDA ↑ Catalase | ↓ HOMA-IR ↓ Oxidative stress ↓ Liver inflammation | [67] |
| C57BL/6 mice | 80 mg/kg b.w./day; 12 weeks | ↓ Insulin ↓ Triglyceride ↓ Cholesterol ↓ LDL | ↓ Body weight gain Liver: ↑ Insulin sensitivity ↓ Fat accumulation ↑ Adiponectin, LPL and UCP-1 ↓ HMGCR, FAS and SCD-1 ↑ Phospho-IRS-1 ↑ Phospho-IRS-2 ↑ Phospho-AKT | [68] |
| C57BL/6 mice | 50 mg/kg b.w./day; 12 weeks | ↓ Glucose | ↓ Adipose glycerol and FFA ↓ Liver lipid deposits Adipose: ↓ ER stress ↓ Phospho-IRE1α ↓ Phospho-eIF2α ↑ PDE3B protein ↑ Phospho-PKA ↑ Phospho-Akt ↓ G6Pase-α protein ↓ SREBP-1c protein | [69] |
| Condition | Curcumin Concentration/Duration | Serum Effects | Other Effects | Reference |
|---|---|---|---|---|
| Healthy individuals | 6 g; 30 and 60 mins | ↑ Insulin | No additional effects | [70] |
| T2DM dyslipidemia patients | 200 mg/capsule/day; 12 weeks | ↓ Glucose ↓ Triglyceride ↓ LDL ↑ HDL | ↓ BMI | [71] |
| T2DM patients | 200 mg/day; 14 weeks | ↓ Glucose ↓ HbA1c | No additional effects | [72] |
| Diabetic Nephropathy | 22.1 mg/day; 2 months | ↓ Glucose ↓ Triglyceride ↓ Cholesterol ↓ LDL ↓ TGF-β ↓ IL-8 | ↓ Proteinuria ↓ Urinary IL-8 | [73] |
| Diabetic Patients | 200 mg/day; 4 weeks | No measured effects | ↓ Microangiopathy ↑ Venoarteriolar response ↑ PO2 ↓ Skin flux ↓ Edema | [74] |
| Diabetic Patients | 200 mg/day; 4 weeks | No measured effects | ↓ Microangiopathy ↑ Venoarteriolar response ↑ PO2 ↓ Skin flux ↓ Edema | [75] |
| Pre-Diabetic Patients | 1500 mg/day; 9 months | ↓ Glucose ↓ HbA1c ↓ Insulin ↓ C-peptide ↑ Adiponectin | ↓ Diabetes ↑ β-cells function ↑ HOMA-β | [76] |
| Overweight Diabetic Patients | 300 mg/day; 3 months | ↓ Glucose ↓ HbA1c ↓ Triglycerides ↓ Total cholesterol ↓ Total fatty acid ↓ Saturated FFA ↓ Unsaturated FFA ↑ LPL activity | ↓ HOMA-IR | [77] |
| Pre-diabetic and controlled diabetic patients | 3.17 g Jiangtang Xiaozhi (13% curcumin); 16 weeks | ↓ Insulin ↑ HDL | ↓ HOMA-IR | [78] |
| Obese patients | 1 g/day; 30 days | ↓ Triglyceride | No additional effects | [79] |
| Diabetic patients | 1500 mg/day; 6 months | ↓ Insulin ↓ Triglyceride ↓ Leptin ↑ Adiponectin | ↓ HOMA-IR ↓ Visceral fat ↓ Total body fat ↓ Waist ↓ PWV Rt ↓ PWV Lt | [80] |
| Metabolic syndrome patients | 630 mg/thrice/day; 12 weeks | ↑ HDL ↓ LDL ↓ Total triglyceride | No additional effects | [81] |
| T2DM patients | 475 mg/once; 24 h | ↓ Glucose ↓ Triglyceride ↓ LDL ↓ VLDL ↑ HDL | No additional effects | [82] |
| Overweight Diabetic patients | 300mg/day; 3 months | ↓ A-FABP ↓ CRP ↓ TNF-α ↓ IL-6 ↑ SOD activity | No measured effects | [83] |
| Overweight patients with metabolic syndrome | 1600 mg/day; 30 days | No measurements | ↑ Weight loss ↓ Body fat % ↓ Waist ↓ Hip circumference ↓ BMI | [84] |
| Diabetic patients | 500 mg/day; 15-30 days | ↓ MDA ↓ LPS content ↓ U-mAlb ↑ IκB ↑ NQO-1 ↑ Nrf2 ↑ SOD1/2 ↓ Caspase-3 ↑ Bacteroides ↑ Bifidobacterium ↑ Lactobacillus | No measured effects | [85] |
| T2DM patients | 80 mg/day; 3 months | ↓ Fasting glucose ↓ HbA1c ↓ Triglyceride ↓ LDL | ↓ BMI | [86] |
| Diabetic proteinuric CKD patients | 320 mg/day; 8 weeks | ↑ Gpx activity ↑ SOD activity | ↓ Lipid peroxidation | [87] |
| Diabetic patients | 1000mg/day; 12 weeks | ↓ Triglyceride ↓ Non-HDL ↓ Lipoprotein ↑ HDL | No measured effects | [88] |
| Diabetic patients | 500 mg/day; 3 months | ↓ Glucose ↓ HbA1c ↓ Insulin ↓ C-peptide ↓ Creatinine ↓ ALT and AST | ↑ HOMA-IR ↓ Hepatic damage | [89] |
| Fasting glucose impaired patients | 125 mg/twice/day; 8 weeks | ↓ Fasting insulin ↓ Triglycerides ↑ HDL | ↓ HOMA-IR | [90] |
| T2DM patients | 1500 mg/thrice/day; 10 weeks | ↓ Glucose | ↓ Mean weight ↓ BMI ↓ Waist | [91] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Den Hartogh, D.J.; Gabriel, A.; Tsiani, E. Antidiabetic Properties of Curcumin II: Evidence from In Vivo Studies. Nutrients 2020, 12, 58. https://doi.org/10.3390/nu12010058
Den Hartogh DJ, Gabriel A, Tsiani E. Antidiabetic Properties of Curcumin II: Evidence from In Vivo Studies. Nutrients. 2020; 12(1):58. https://doi.org/10.3390/nu12010058
Chicago/Turabian StyleDen Hartogh, Danja J., Alessandra Gabriel, and Evangelia Tsiani. 2020. "Antidiabetic Properties of Curcumin II: Evidence from In Vivo Studies" Nutrients 12, no. 1: 58. https://doi.org/10.3390/nu12010058
APA StyleDen Hartogh, D. J., Gabriel, A., & Tsiani, E. (2020). Antidiabetic Properties of Curcumin II: Evidence from In Vivo Studies. Nutrients, 12(1), 58. https://doi.org/10.3390/nu12010058






