The Dietary Inflammatory Index and Chronic Lymphocytic Leukaemia in the MCC Spain Study
Abstract
1. Introduction
2. Materials and Methods
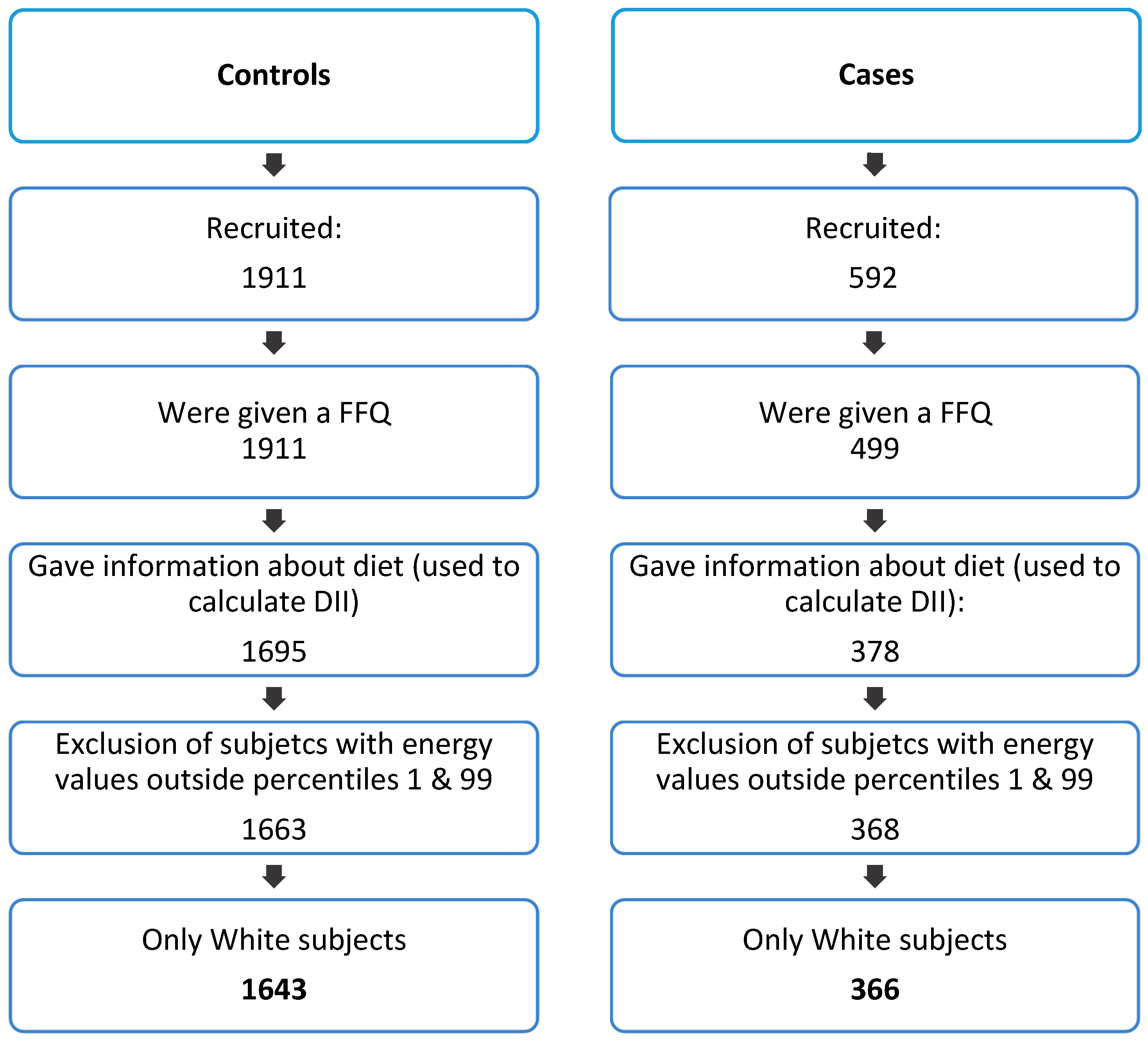
2.1. Study Population
2.2. Outcome Definition
2.3. Data Collection and Sources
2.4. Dietary Inflammatory Index
2.5. Statistical Analyses
3. Results
3.1. Characteristics of Cases and Controls in the MCC Spain Study
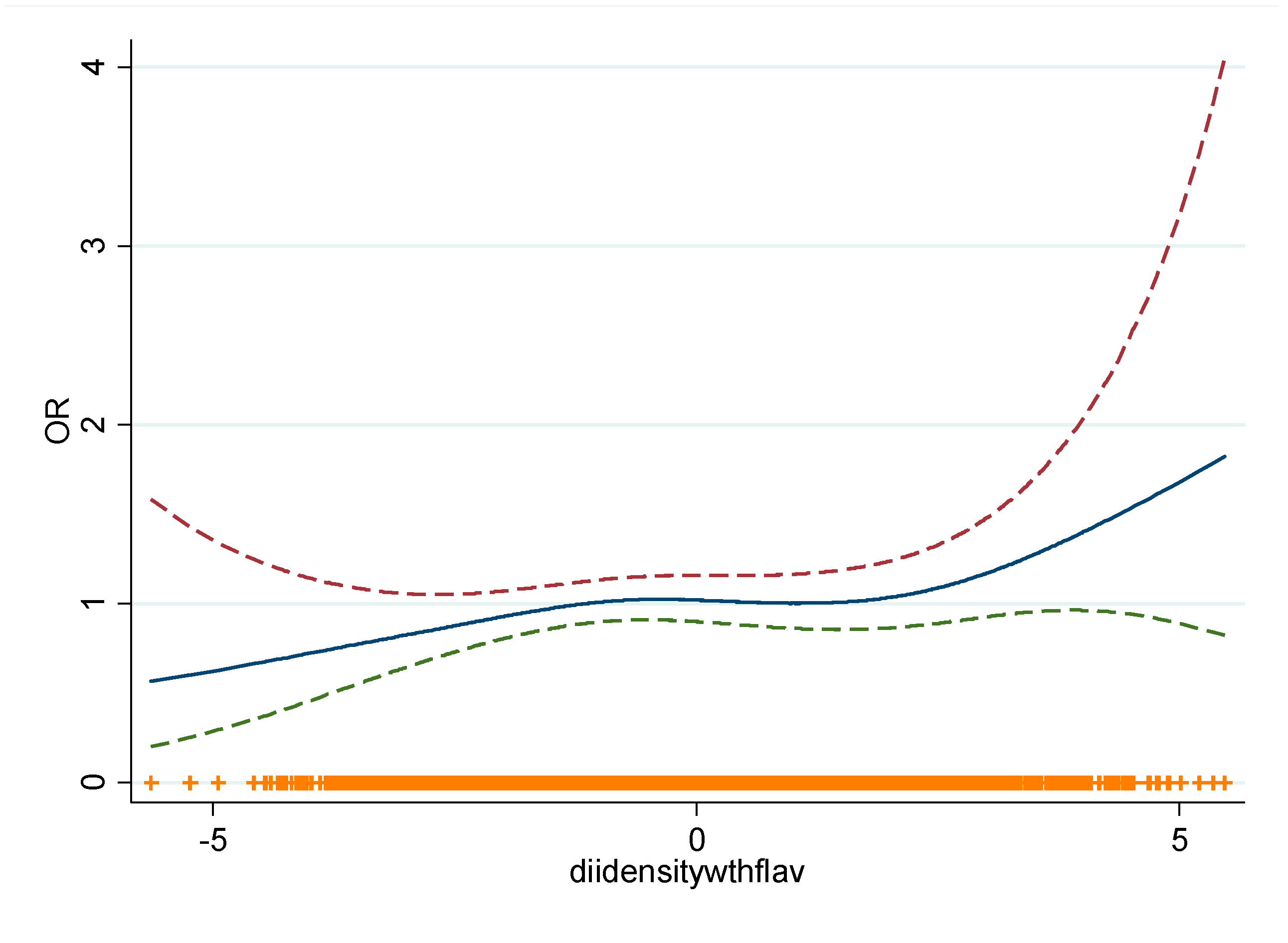
3.2. E-DII and CLL Risk
3.3. Sensitivity Analyses.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eichhorst, B.; Robak, T.; Montserrat, E.; Ghia, P.; Hillmen, P.; Hallek, M.; Buske, C. Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26, v78–v84. [Google Scholar] [CrossRef] [PubMed]
- Goldin, L.R.; Slager, S.L.; Caporaso, N.E. Familial chronic lymphocytic leukemia. Curr. Opin. Hematol. 2010, 17, 350–355. [Google Scholar] [CrossRef]
- Kipps, T.J.; Stevenson, F.K.; Wu, C.J.; Croce, C.M.; Packham, G.; Wierda, W.G.; O’Brien, S.; Gribben, J.; Rai, K. Chronic lymphocytic leukaemia. Nat. Rev. Dis. Prim. 2017, 3, 16096. [Google Scholar] [CrossRef] [PubMed]
- Noone, A.; Howlader, N.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.; et al. SEER Cancer Statistics Review 1975–2012; National Cancer Institute: Bethesda, MD, USA, 2015. [Google Scholar]
- Cerhan, J.R.; Kricker, A.; Paltiel, O.; Flowers, C.R.; Wang, S.S.; Monnereau, A.; Blair, A.; Maso, L.D.; Kane, E.V.; Nieters, A.; et al. Medical History, Lifestyle, Family History, and Occupational Risk Factors for Diffuse Large B-Cell Lymphoma: The InterLymph Non-Hodgkin Lymphoma Subtypes Project. JNCI Monogr. 2014, 2014, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Aschebrook-Kilfoy, B.; Ollberding, N.J.; Kolar, C.; Lawson, T.A.; Smith, S.M.; Weisenburger, D.D.; Chiu, B.C.-H. Meat intake and risk of non-Hodgkin lymphoma. Cancer Causes Control 2012, 23, 1681–1692. [Google Scholar] [CrossRef]
- Campagna, M.; Cocco, P.; Zucca, M.; Angelucci, E.; Gabbas, A.; Latte, G.C.; Uras, A.; Rais, M.; Sanna, S.; Ennas, M.G. Risk of lymphoma subtypes and dietary habits in a Mediterranean area. Cancer Epidemiol. 2015, 39, 1093–1098. [Google Scholar] [CrossRef]
- Tsai, H.T.; Cross, A.J.; Graubard, B.I.; Oken, M.; Schatzkin, A.; Caporaso, N.E. Dietary factors and risk of chronic lymphocytic leukemia and small lymphocytic lymphoma: A pooled analysis of two prospective studies. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2680–2684. [Google Scholar] [CrossRef][Green Version]
- Purdue, M.P.; Bassani, D.G.; Klar, N.S.; Sloan, M.; Kreiger, N. Canadian Cancer Registries Epidemiology Research Group Dietary factors and risk of non-Hodgkin lymphoma by histologic subtype: A case-control analysis. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1665–1676. [Google Scholar]
- Chang, E.T. Dietary Factors and Risk of Non-Hodgkin Lymphoma in Men and Women. Cancer Epidemiol. Biomark. Prev. 2005, 14, 512–520. [Google Scholar] [CrossRef]
- Casabonne, D.; Gracia, E.; Espinosa, A.; Bustamante, M.; Benavente, Y.; Robles, C.; Costas, L.; Alonso, E.; Gonzalez-Barca, E.; Tardón, A.; et al. Fruit and vegetable intake and vitamin C transporter gene (SLC23A2) polymorphisms in chronic lymphocytic leukaemia. Eur. J. Nutr. 2017, 56, 1123–1133. [Google Scholar] [CrossRef]
- Chang, E.T.; Balter, K.M.; Torrang, A.; Smedby, K.E.; Melbye, M.; Sundstrom, C.; Glimelius, B.; Adami, H.-O. Nutrient Intake and Risk of Non-Hodgkin’s Lymphoma. Am. J. Epidemiol. 2006, 164, 1222–1232. [Google Scholar] [CrossRef] [PubMed]
- Charbonneau, B.; O’Connor, H.M.; Wang, A.H.; Liebow, M.; Thompson, C.A.; Fredericksen, Z.S.; Macon, W.R.; Slager, S.L.; Call, T.G.; Habermann, T.M.; et al. Trans Fatty Acid Intake Is Associated with Increased Risk and n3 Fatty Acid Intake with Reduced Risk of Non-Hodgkin Lymphoma. J. Nutr. 2013, 143, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Chiu, B.C.-H.; Kwon, S.; Evens, A.M.; Surawicz, T.; Smith, S.M.; Weisenburger, D.D. Dietary intake of fruit and vegetables and risk of non-Hodgkin lymphoma. Cancer Causes Control. 2011, 22, 1183–1195. [Google Scholar] [CrossRef] [PubMed]
- Fritschi, L.; Ambrosini, G.L.; Kliewer, E.V.; Johnson, K.C.; Dryer, D.; Paulse, B.; Dewar, R.; Kreiger, N.; Kliewer, E.; Robson, D.; et al. Dietary fish intake and risk of leukaemia, multiple myeloma, and non-Hodgkin lymphoma. Cancer Epidemiol. Biomark. Prev. 2004, 13, 532–537. [Google Scholar]
- Koutros, S.; Zhang, Y.; Zhu, Y.; Mayne, S.T.; Zahm, S.H.; Holford, T.R.; Leaderer, B.P.; Boyle, P.; Zheng, T. Nutrients Contributing to One-Carbon Metabolism and Risk of Non-Hodgkin Lymphoma Subtypes. Am. J. Epidemiol. 2007, 167, 287–294. [Google Scholar] [CrossRef]
- Mikhak, B.; Bracci, P.M.; Gong, Z. Intake of Vitamins D and A and Calcium and Risk of Non-Hodgkin Lymphoma: San Francisco Bay Area Population-Based Case-Control Study. Nutr. Cancer 2012, 64, 674–684. [Google Scholar] [CrossRef]
- Han, Y.-Y.; Forno, E.; Shivappa, N.; Wirth, M.D.; Hébert, J.R.; Celedón, J.C. The Dietary Inflammatory Index and Current Wheeze Among Children and Adults in the United States. J. Allergy Clin. Immunol. Pract. 2018, 6, 834–841.e2. [Google Scholar] [CrossRef]
- Lee, H.; Lee, I.S.; Choue, R. Obesity, Inflammation and Diet. Pediatr. Gastroenterol. Hepatol. Nutr. 2013, 16, 143. [Google Scholar] [CrossRef]
- Nguyen, X.-M.T.; Lane, J.; Smith, B.R.; Nguyen, N.T. Changes in Inflammatory Biomarkers Across Weight Classes in a Representative US Population: A Link Between Obesity and Inflammation. J. Gastrointest. Surg. 2009, 13, 1205–1212. [Google Scholar] [CrossRef]
- Gunter, M.J.; Stolzenberg-Solomon, R.; Cross, A.J.; Leitzmann, M.F.; Weinstein, S.; Wood, R.J.; Virtamo, J.; Taylor, P.R.; Albanes, D.; Sinha, R. A Prospective Study of Serum C-Reactive Protein and Colorectal Cancer Risk in Men. Cancer Res. 2006, 66, 2483–2487. [Google Scholar] [CrossRef]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O.; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Philip, M.; Rowley, D.A.; Schreiber, H. Inflammation as a tumor promoter in cancer induction. Semin. Cancer Biol. 2004, 14, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Smedby, K.E.; Ponzoni, M. The aetiology of B-cell lymphoid malignancies with a focus on chronic inflammation and infections. J. Intern. Med. 2017, 282, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Rozovski, U.; Keating, M.J.; Estrov, Z. Targeting inflammatory pathways in chronic lymphocytic leukemia. Crit. Rev. Oncol. Hematol. 2013, 88, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Aeberli, I.; Gerber, P.A.; Hochuli, M.; Kohler, S.; Haile, S.R.; Gouni-Berthold, I.; Berthold, H.K.; Spinas, G.A.; Berneis, K. Low to moderate sugar-sweetened beverage consumption impairs glucose and lipid metabolism and promotes inflammation in healthy young men: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 94, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Kimiagar, M.; Mehrabi, Y.; Azadbakht, L.; Hu, F.B.; Willett, W.C. Dietary Patterns and Markers of Systemic Inflammation among Iranian Women. J. Nutr. 2007, 137, 992–998. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Egan, B.M.; Geesey, M.E. Relation of dietary fat and fiber to elevation of C-reactive protein. Am. J. Cardiol. 2003, 92, 1335–1339. [Google Scholar] [CrossRef]
- Wirth, M. The Dietary Inflammatory Index: A New Tool for Assessing Diet Quality Based on Inflammatory Potential. Digest 2014, 49, 1–9. [Google Scholar]
- Zinöcker, M.K.; Lindseth, I.A. The western diet–microbiome-host interaction and its role in metabolic disease. Nutrients 2018, 10, 365. [Google Scholar] [CrossRef]
- Vetizou, M.; Pitt, J.M.; Daillere, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.M.; et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 2015, 350, 1079–1084. [Google Scholar] [CrossRef]
- Singh, R.K.; Chang, H.W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of diet on the gut microbiome and implications for human health. J. Trans. Med. 2017, 15, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Cavicchia, P.P.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Ma, Y.; Ockene, I.S.; Hébert, J.R. A New Dietary Inflammatory Index Predicts Interval Changes in Serum High-Sensitivity C-Reactive Protein. J. Nutr. 2009, 139, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Shivappa, N.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Hébert, J.R. Designing and developing a literature-derived, population –based dietary inflammatory index. Public Health Nutr. 2014, 17, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Fowler, M.E.; Akinyemiju, T.F. Meta-analysis of the association between dietary inflammatory index (DII) and cancer outcomes. Int. J. Cancer 2017, 141, 2215–2227. [Google Scholar] [CrossRef] [PubMed]
- Solans, M.; Benavente, Y.; Saez, M.; Agudo, A.; Jakszyn, P.; Naudin, S.; Hosnijeh, F.S.; Gunter, M.; Huybrechts, I.; Ferrari, P.; et al. Inflammatory potential of diet and risk of lymphoma in the European Prospective Investigation into Cancer and Nutrition. Eur. J. Nutr. 2019, 107, 607–616. [Google Scholar] [CrossRef]
- Shivappa, N.; Hébert, J.R.; Taborelli, M.; Zucchetto, A.; Montella, M.; Libra, M.; La Vecchia, C.; Serraino, D.; Polesel, J. Association between dietary inflammatory index and Hodgkin’s lymphoma in an Italian case-control study. Nutrition 2018, 53, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Shivappa, N.; Hébert, J.R.; Taborelli, M.; Montella, M.; Libra, M.; Zucchetto, A.; Crispo, A.; Grimaldi, M.; La Vecchia, C.; Serraino, D.; et al. Dietary inflammatory index and non-Hodgkin lymphoma risk in an Italian case–control study. Cancer Causes Control. 2017, 28, 791–799. [Google Scholar] [CrossRef]
- Castaño-Vinyals, G.; Aragonés, N.; Pérez-Gómez, B.; Martín, V.; Llorca, J.; Moreno, V.; Altzibar, J.M.; Ardanaz, E.; de Sanjosé, S.; Jiménez-Moleón, J.J.; et al. Population-based multicase-control study in common tumors in Spain (MCC-Spain): Rationale and study design. Gac. Sanit. 2015, 29, 308–315. [Google Scholar] [CrossRef]
- Solans, M.; Castelló, A.; Benavente, Y.; Marcos-Gragera, R.; Amiano, P.; Gracia-Lavedan, E.; Costas, L.; Robles, C.; Gonzalez Barca, E.; de la Banda, E.; et al. Adherence to the Western, Prudent, and Mediterranean dietary patterns and chronic lymphocytic leukemia in the MCC-Spain study. Haematologica 2018, 103. [Google Scholar] [CrossRef]
- García-Closas, R.; García-Closas, M.; Kogevinas, M.; Malats, N.; Silverman, D.; Serra, C.; Tardón, A.; Carrato, A.; Castaño-Vinyals, G.; Dosemeci, M.; et al. Food, nutrient and heterocyclic amine intake and the risk of bladder cancer. Eur. J. Cancer 2007, 43, 1731–1740. [Google Scholar] [CrossRef]
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation; WHO: Geneva, Switzerland, 2011; pp. 1–39. [Google Scholar]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, S.; Kaaks, R.; Jakszyn, P.; Bergmann, M.; Molina, E.; Boutron-Ruault, M.C.; Rohrmann, S.; Jakobsen, M.U.; Vineis, P.; Boeing, H.; et al. Consumption of meat and dairy and lymphoma risk in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 2010, 128, 623–634. [Google Scholar]
- Bertrand, K.A.; Giovannucci, E.; Rosner, B.A.; Zhang, S.M.; Laden, F.; Birmann, B.M. Dietary fat intake and risk of non-Hodgkin lymphoma in 2 large prospective cohorts. Am. J. Clin. Nutr. 2017, 106, 650–656. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ross, J.A.; Kasum, C.M.; Davies, S.M.; Jacobs, D.R.; Folsom, A.R.; Potter, J.D. Diet and risk of leukemia in the Iowa Women’s Health Study. Cancer Epidemiol. Biomark. Prev. 2002, 11, 777–781. [Google Scholar]
- Chang, E.T.; Canchola, A.J.; Clarke, C.A.; Lu, Y.; West, D.W.; Bernstein, L.; Wang, S.S.; Horn-Ross, P.L. Dietary phytocompounds and risk of lymphoid malignancies in the California Teachers Study cohort. Cancer Causes Control. 2011, 22, 237–249. [Google Scholar] [CrossRef]
- Smedby, K.E.; Baecklund, E.; Askling, J. Malignant Lymphomas in Autoimmunity and Inflammation: A Review of Risks, Risk Factors, and Lymphoma Characteristics. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2069–2077. [Google Scholar] [CrossRef]
- Smedby, K.E.; Askling, J.; Mariette, X.; Baecklund, E. Autoimmune and inflammatory disorders and risk of malignant lymphomas—An update. J. Intern. Med. 2008, 264, 514–527. [Google Scholar] [CrossRef]
- Morton, L.M.; Slager, S.L.; Cerhan, J.R.; Wang, S.S.; Vajdic, C.M.; Skibola, C.F.; Bracci, P.M.; de Sanjose, S.; Smedby, K.E.; Chiu, B.C.H.; et al. Etiologic Heterogeneity Among Non-Hodgkin Lymphoma Subtypes: The InterLymph Non-Hodgkin Lymphoma Subtypes Project. JNCI Monogr. 2014, 2014, 130–144. [Google Scholar] [CrossRef]
- Fan, Y.; Jin, X.; Man, C.; Gao, Z.; Wang, X.; Fan, Y.; Jin, X.; Man, C.; Gao, Z.; Wang, X.; et al. Meta-analysis of the association between the inflammatory potential of diet and colorectal cancer risk. Oncotarget 2017, 8, 59592–59600. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, W.; Zhang, D. Association Between Dietary Inflammation Index and The Risk of Colorectal Cancer: A Meta-Analysis. Nutr. Cancer 2018, 70, 14–22. [Google Scholar] [CrossRef]
- Agudo, A.; Cayssials, V.; Bonet, C.; Tjønneland, A.; Overvad, K.; Boutron-Ruault, M.C.; Affret, A.; Fagherazzi, G.; Katzke, V.; Schübel, R.; et al. Inflammatory potential of the diet and risk of gastric cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am. J. Clin. Nutr. 2018, 107, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, C.; Zhou, C.; Zhuang, J.; Tang, S.; Yu, J.; Tian, J.; Feng, F.; Liu, L.; Zhang, T.; et al. Meta-analysis of the association between the dietary inflammatory index (DII) and breast cancer risk. Eur. J. Clin. Nutr. 2019, 73, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Moradi, S.; Issah, A.; Mohammadi, H.; Mirzaei, K. Associations between dietary inflammatory index and incidence of breast and prostate cancer: A systematic review and meta-analysis. Nutrition 2018, 55–56, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Jayedi, A.; Emadi, A.; Shab-Bidar, S. Dietary Inflammatory Index and Site-Specific Cancer Risk: A Systematic Review and Dose-Response Meta-Analysis. Adv. Nutr. 2018, 9, 388–403. [Google Scholar] [CrossRef] [PubMed]
- The Selection and Use of Essential Medicines. Chronic Lymphocytic Leukemia. WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2015. Available online: https://apps.who.int/iris/bitstream/handle/10665/189763/9789241209946_eng.pdf;jsessionid=7DDAD42597A0228084BAA548B4797C33?sequence=1 (accessed on 13 December 2019).
- Clarke, C.A.; Glaser, S.L.; Gomez, S.L.; Wang, S.S.; Keegan, T.H.; Yang, J.; Chang, E.T. Lymphoid malignancies in US Asians: Incidence rate differences by birthplace and acculturation. Cancer Epidemiol. Prev. Biomark. 2011, 20, 1064–1077. [Google Scholar] [CrossRef]
- Wu, S.J.; Huang, S.Y.; Lin, C.T.; Lin, Y.; Chang, C.J.; Tien, H.F. The incidence of chronic lymphocytic leukemia in Taiwan, 1986–2005: A distinct increasing trend with birth-cohort effect. Blood 2010, 116, 4430–4435. [Google Scholar] [CrossRef]
- Serafini, M.; Peluso, I.; Raguzzini, A. Flavonoids as anti-inflammatory agents. Proc. Nutr. Soc. 2010, 69, 273–278. [Google Scholar] [CrossRef]
- Calvert, C.; Cade, J.; Barrett, J.H.; Woodhouse, A. Using cross-check questions to address the problem of mis-reporting of specific food groups on Food Frequency Questionnaires. UKWCS Steering Group. United Kingdom Women’s Cohort Study Steering Group. Eur. J. Clin. Nutr. 1997, 51, 708–712. [Google Scholar] [CrossRef]
- Avellone, G.; Di Garbo, V.; Campisi, D.; De Simone, R.; Raneli, G.; Scaglione, R.; Licata, G. Effects of moderate Sicilian red wine consumption on inflammatory biomarkers of atherosclerosis. Eur. J. Clin. Nutr. 2006, 60, 41–47. [Google Scholar] [CrossRef]
- Sierksma, A.; van der Gaag, M.; Kluft, C.; Hendriks, H. Moderate alcohol consumption reduces plasma C-reactive protein and fibrinogen levels; a randomized, diet-controlled intervention study. Eur. J. Clin. Nutr. 2002, 56, 1130–1136. [Google Scholar] [CrossRef]
| Variables ** | Controls | Cases | p Value a |
|---|---|---|---|
| E-DII, mean (SD) | −0.20 (2.0) | −0.13 (2.0) | 0.51 |
| Age (years), mean (SD) | 63.5 (11.4) | 66.2 (10.2) | <0.001 |
| Male, N (%) | 939 (57.2) | 215 (58.7) | 0.58 |
| Education, N (%) | 0.81 | ||
| Primary | 861 (52.4) | 197 (53.8) | |
| Secondary | 481 (29.3) | 107 (29.2) | |
| University | 301 (18.3) | 62 (16.9) | |
| Region, N (%) | <0.001 | ||
| Barcelona | 889 (54.1) | 240 (65.6) | |
| Asturias | 208 (12.7) | 50 (13.7) | |
| Cantabria | 324 (19.7) | 21 (5.7) | |
| Granada | 148 (9.0) | 27 (7.4) | |
| Gerona | 74 (4.5) | 28 (7.7) | |
| BMI (kg/m2), mean (SD)b | 26.9 (4.5) | 27.3 (4.4) | 0.13 |
| Weight (kg), mean (SD) | 73.9 (13.7) | 75.0 (14.3) | 0.15 |
| Height (cm), mean (SD) | 165.7 (8.5) | 166.0 (9.1) | 0.61 |
| Waist-to-hip ratio, N (%) with higher risk of metabolic complications c | 1239 (76.1) | 317 (87.1) | <0.001 |
| Energy (kcal/day), mean (SD) | 1891.4 (573.3) | 1934.3 (601.7) | 0.20 |
| Alcohol consumption (gr/day), mean (SD)d | 12.0 (17.1) | 9.5 (13.8) | 0.008 |
| Ever had diabetes, N (%) | 236 (14.6) | 46 (12.8) | 0.38 |
| Smoking, N (%) | 0.85 | ||
| Never | 722 (44.1) | 164 (45.1) | |
| Current smoker | 290 (17.7) | 60 (16.5) | |
| Former smoker | 626 (38.2) | 140 (38.5) | |
| Physical activity, N (%)e | 0.62 | ||
| Inactive | 668 (41.6) | 134 (38.0) | |
| Low | 227 (14.1) | 55 (15.6) | |
| Moderate | 196 (12.2) | 47 (13.3) | |
| Very active | 514 (32.0) | 117 (33.1) | |
| Ever worked in agriculture, N (%) | 325 (19.8) | 107 (29.3) | <0.001 |
| First-degree family history of haematological cancer, N (%) | 53 (3.5) | 36 (10.3) | <0.001 |
| Treated for CLL, N (%) | N/A | 79 (21.8) | N/A |
| Disease severity, N (%)f | N/A | ||
| Rai 0 | N/A | 207 (58.0) | |
| Rai I–IV | N/A | 150 (42.0) | |
| Time from diagnosis to recruitment | N/A | ||
| <1 year | N/A | 97 (26.5) | |
| ≥1year | N/A | 269 (73.5) |
| By Tertiles of E-DII | P-value Trend | One-Unit Increase in E-DII | P-value Trend | |||
|---|---|---|---|---|---|---|
| T(min, max) | T1 (−5.64, −1.31) | T2 (−1.31, 0.59) | T3 (0.59, 5.47) | |||
| N controls/cases | 547/114 | 549/131 | 547/121 | 1643/366 | ||
| OR a and 95% CI | Ref | 1.20 (0.90, 1.59) | 1.21 (0.90, 1.62) | 0.21 c | 1.05 (0.99, 1.12) | 0.09 |
| N controls/cases | 1586/352 | |||||
| OR b and 95% CI | Ref | 1.24 (0.93, 1.66) | 1.24 (0.91, 1.68) | 0.17 c | 1.06 (1.00, 1.13) | 0.07 |
| E-DII | One-Unit Increase in E-DII | ||||||
|---|---|---|---|---|---|---|---|
| OR a and 95% CI | OR a and 95% CI | ||||||
| T1 | T2 | T3 | p-Value for Trend b | p-Value | p-Value for Heterogeneity c | ||
| Rai stages | |||||||
| N controls/C1/C2 | 547/69/43 | 549/66/61 | 547/72/46 | ||||
| C1: Rai 0 | Ref | 1.04 (0.72, 1.50) | 1.30 (0.90, 1.89) | 0.17 | 1.05 (0.97, 1.13) | 0.27 | |
| C2: Rai I–IV | Ref | 1.42 (0.94, 2.14) | 1.09 (0.69, 1.72) | 0.70 | 1.06 (0.97, 1.16) | 0.17 | 0.70 |
| Time from diagnosis to recruitment | |||||||
| N controls/C1/C2 | 547/30/84 | 549/36/95 | 547/31/90 | ||||
| C1: <1 year | Ref | 1.25 (0.75, 2.07) | 1.14 (0.66, 1.95) | 0.63 | 1.03 (0.93, 1.15) | 0.55 | |
| C2: ≥1 year | Ref | 1.18 (0.86, 1.63) | 1.23 (0.88, 1.72) | 0.22 | 1.06 (0.99, 1.14) | 0.09 | 0.67 |
| 1–3 years | Ref | 0.93 (0.60 to 1.47) | 1.14 (0.72 to 1.78) | 0.58 | 1.03 (0.94 to 1.14) | 0.51 | |
| ≥3 years | Ref | 1.45 (0.95 to 2.22) | 1.33 (0.84 to 2.12) | 0.21 | 1.09 (0.99 to 1.20) | 0.07 | |
| Treated for CLL | |||||||
| N controls/C1/C2 | 547/95/19 | 549/94/35 | 547/95/25 | ||||
| C1: No | Ref | 1.05 (0.76, 1.43) | 1.18 (0.86, 1.64) | 0.31 | 1.04 (0.97, 1.12) | 0.22 | |
| C2: Yes | Ref | 1.80 (1.01, 3.20) | 1.30 (0.69, 2.44) | 0.46 | 1.08 (0.96, 1.22) | 0.20 | 0.56 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores, J.C.; Gracia-Lavedan, E.; Benavente, Y.; Amiano, P.; Romaguera, D.; Costas, L.; Robles, C.; Gonzalez-Barca, E.; de la Banda, E.; Alonso, E.; et al. The Dietary Inflammatory Index and Chronic Lymphocytic Leukaemia in the MCC Spain Study. Nutrients 2020, 12, 48. https://doi.org/10.3390/nu12010048
Flores JC, Gracia-Lavedan E, Benavente Y, Amiano P, Romaguera D, Costas L, Robles C, Gonzalez-Barca E, de la Banda E, Alonso E, et al. The Dietary Inflammatory Index and Chronic Lymphocytic Leukaemia in the MCC Spain Study. Nutrients. 2020; 12(1):48. https://doi.org/10.3390/nu12010048
Chicago/Turabian StyleFlores, José Carlos, Esther Gracia-Lavedan, Yolanda Benavente, Pilar Amiano, Dora Romaguera, Laura Costas, Claudia Robles, Eva Gonzalez-Barca, Esmeralda de la Banda, Esther Alonso, and et al. 2020. "The Dietary Inflammatory Index and Chronic Lymphocytic Leukaemia in the MCC Spain Study" Nutrients 12, no. 1: 48. https://doi.org/10.3390/nu12010048
APA StyleFlores, J. C., Gracia-Lavedan, E., Benavente, Y., Amiano, P., Romaguera, D., Costas, L., Robles, C., Gonzalez-Barca, E., de la Banda, E., Alonso, E., Aymerich, M., Campo, E., Dierssen-Sotos, T., Marcos-Gragera, R., Rodriguez-Suarez, M. M., Solans, M., Gimeno, E., Garcia Martin, P., Aragones, N., ... Casabonne, D. (2020). The Dietary Inflammatory Index and Chronic Lymphocytic Leukaemia in the MCC Spain Study. Nutrients, 12(1), 48. https://doi.org/10.3390/nu12010048






