Effects of Daily Low-Dose Date Consumption on Glycemic Control, Lipid Profile, and Quality of Life in Adults with Pre- and Type 2 Diabetes: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion and Exclusion Criteria
2.3. Randomization
2.4. Procedures
2.5. Outcomes
2.6. Experimental Design and Diets
2.7. Study Visits and Blood Samples
2.8. Health-Related Quality of Life
2.9. Dietary and Physical Activity Assessments
2.10. Sample Size
2.11. Statistical Analysis
3. Results
3.1. HbA1c and Serum Lipid Profiles
3.2. Quality of Life
3.3. Safety
3.4. Monitoring the Physical Activity
3.5. Dietary Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Al Jasser, M.S. Physicochemical Composition of Date Fruit (Phoenix dactyifera L.) from Offshoots and Cultured Cells at Different Stages. J. Food Technol. 2009, 7, 102–105. [Google Scholar]
- Miller, C.J.; Dunn, E.V.; Hashim, I.B. The glycaemic index of dates and date/yoghurt mixed meals. Are dates the candy that grows on trees? Eur. J. Clin. Nutr. 2003, 57, 427–430. [Google Scholar] [CrossRef] [PubMed]
- Assirey, E.A. Nutritional composition of fruit of 10 date palm (Phoenix dactylifera L.) cultivars grown in Saudi Arabia. J. Taibah Univ. Sci. 2015, 9, 75–79. [Google Scholar] [CrossRef]
- Al-Farsi, M.; Alasalvar, C.; Morris, A.; Baron, M.; Shahidi, F. Compositional and Sensory Characteristics of Three Native Sun-Dried Date Varieties Grown in Oman L. Varieties Grown in Oman. J. Agric. Food Chem. 2005, 53, 7586–7591. [Google Scholar] [CrossRef]
- Vayalil, P.K. Date Fruits (Phoenix dactylifera Linn): An Emerging Medicinal Food. Crit. Rev. Food Sci. Nutr. 2012, 52, 249–271. [Google Scholar] [CrossRef] [PubMed]
- Taleb, H.; Maddocks, S.E.; Morris, R.K.; Kanekanian, A.D. Chemical characterisation and the anti-inflammatory, anti-angiogenic and antibacterial properties of date fruit (Phoenix dactylifera L.). J. Ethnopharmacol. 2016, 194, 457–468. [Google Scholar] [CrossRef]
- Rahmani, A.H.; Aly, S.M.; Ali, H.; Babiker, A.Y.; Srikar, S.; Khan, A.A. Therapeutic effects of date fruits (Phoenix dactylifera) in the prevention of diseases via modulation of anti-inflammatory, anti-oxidant and anti-tumour activity. Int. J. Clin. Exp. Med. 2014, 7, 483–491. [Google Scholar]
- Yasin, B.R.; El-Fawal, H.A.N.; Mousa, S.A.; Segura-Carretero, A.; Gómez Caravaca, A.M. Date (Phoenix dactylifera) Polyphenolics and Other Bioactive Compounds: A Traditional Islamic Remedy’s Potential in Prevention of Cell Damage, Cancer Therapeutics and Beyond. Int. J. Mol. Sci. 2015, 16, 30075–30090. [Google Scholar] [CrossRef]
- IDF. IDF Diabetes Atlas—8th Edition 2017. Available online: https://diabetesatlas.org/ (accessed on 26 July 2019).
- Muldoon, M.F.; Kritchevsky, S.B. Flavonoids and heart disease. BMJ 1996, 312, 458–459. [Google Scholar] [CrossRef]
- Kchaou, W.; Abbès, F.; Attia, H.; Besbes, S. In Vitro Antioxidant Activities of Three Selected Dates from Tunisia (Phoenix dactylifera L.). J. Chem. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Al-Mamary, M.; Al-Habori, M.; Al-Zubairi, A.S. The In Vitro antioxidant activity of different types of palm dates (Phoenix dactylifera) syrups. Arab. J. Chem. 2014, 7, 964–971. [Google Scholar] [CrossRef]
- Hamad, I.; AbdElgawad, H.; Al Jaouni, S.; Zinta, G.; Asard, H.; Hassan, S.; Hegab, M.; Hagagy, N.; Selim, S. Metabolic Analysis of Various Date Palm Fruit (Phoenix dactylifera L.) Cultivars from Saudi Arabia to Assess Their Nutritional Quality. Molecules 2015, 20, 13620–13641. [Google Scholar] [CrossRef] [PubMed]
- Alsaif, M.A.; Khan, L.K.; Alhamdan, A.A.H.; Alorf, S.M.; Harfi, S.H.; Al-Othman, A.M.; Arif, Z. Effect of Dates and Gahwa (Arabian Coffee) Supplementation on Lipids in Hypercholesterolemic Hamsters. Int. J. Pharmacol. 2007, 3, 123–129. [Google Scholar] [CrossRef]
- Arshad, M.S.; Batool, S.M.; Khan, M.K.; Imran, M.; Ahmad, M.H.; Anjum, F.M.; Hussain, S. Bio-evaluation of functional date bars using rats as model organism against hypercholesterolemia. Lipids Health Dis. 2019, 18, 148. [Google Scholar] [CrossRef]
- Verma, P. Effect of Dates in Lowering LDL Level in Geriatric Cases. Health Sci. J. 2016, 10, 1–3. [Google Scholar] [CrossRef]
- Andlauer, W.; Fürst, P. Special Characteristics of Non-Nutrient Food Constituents of Plants—Phytochemicals Introductary Lecture. Int. J. Vitam. Nutr. Res. 2003, 73, 55–62. [Google Scholar] [CrossRef]
- Nasir, M.; Hussain, S.; Jabbar, S.; Rashid, F.; Khalid, N.; Mehmood, A. A review on the nutritional content, functional properties and medicinal potential of dates. Sci. Lett. 2015, 3, 17–22. [Google Scholar]
- Hussein, A.M.; El-Mousalamy, A.M.D.; Hussein, S.A.M.; Mahmoud, S.A. Effects of Palm Dates (Phoenix Dactylifera L.) Extracts on Hepatic Dysfunctions in Type 2 Diabetic Rat Model. World J. Pharm. Pharm. Sci. 2015, 4, 62–79. [Google Scholar]
- Masmoudi-Allouche, F.; Touati, S.; Mnafgui, K.; Gharsallah, N.; El Feki, A.; Allouche, N. Phytochemical profile, antioxidant, antibacterial, antidiabetic and anti-obesity activities of fruits and pits from date palm (Phoenix dactylifera L.) grown in south of Tunisia. J. Pharmacogn. Phytochem. 2016, 5, 15–22. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, without Use of the Preparative Ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar]
- Ware, J.E.; Snow, K.K.; Kosinski, M.; Gandek, B. SF-36 Health Survey Manual and Interpretation Guide; The Health Institute, New England Medical Center: Boston, MA, USA, 1993. [Google Scholar]
- Musaiger, A. Food Composition Tables for Bahrain; Arab Center for Nutrition: Manama, Bahrain, 2011. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-Country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Consort Group the Consort Statement. Available online: http://www.consort-statement.org/ (accessed on 1 September 2019).
- Ahmed, S.; Alam Khan, R.; Jamil, S. Anti-hyperlipidemic and hepatoprotective effects of native date fruit variety “Aseel” (Phoenix dactylifera). Pak. J. Pharm. Sci. 2016, 29, 1945–1950. [Google Scholar] [PubMed]
- Ali, Z.; Ma, H.; Ayim, I.; Wali, A. Efficacy of new beverage made of dates vinegar and garlic juice in improving serum lipid profile parameters and inflammatory biomarkers of mildly hyperlipidemic adults: A double-blinded, randomized, placebo-controlled study. J. Food Biochem. 2018, 42, 1–10. [Google Scholar] [CrossRef]
- Rock, W.; Rosenblat, M.; Borochov-Neori, H.; Volkova, N.; Judeinstein, S.; Elias, M.; Aviram, M. Effects of Date (Phoenix dactylifera L., Medjool or Hallawi Variety) Consumption by Healthy Subjects on Serum Glucose and Lipid Levels and on Serum Oxidative Status: A Pilot Study. J. Agric. Food Chem. 2009, 57, 8010–8017. [Google Scholar] [CrossRef] [PubMed]
- Freha, G.; Cherifa, H. Study of the Effect of Dates on Blood Glucose and Lipid Profile in Healthy Human Subjects. Int. J. Pharm. Chem. Biol. Sci. 2013, 3, 826–833. [Google Scholar]
- Al-Mssallem, M.Q.; Elmulthum, N.A.; Elzaki, R.M. Nutrition elements secured by date palm fruit consumption. In Proceedings of the Saudi FDA Annual Conference and Exhibition (SFDACE 2018), Riyadh, Saudi Arabia, 25 September 2018. [Google Scholar]
- Alalwan, T.A.; Mandeel, Q.A.; Al-Sarhani, L. Traditional plant-based foods and beverages in Bahrain. J. Ethn. Foods 2017, 4, 274–283. [Google Scholar] [CrossRef]
- Alkaabi, J.M.; Al-Dabbagh, B.; Ahmad, S.; Saadi, H.F.; Gariballa, S.; Al Ghazali, M. Glycemic indices of five varieties of dates in healthy and diabetic subjects. Nutr. J. 2011, 10, 59. [Google Scholar] [CrossRef]
- Alkaabi, J.; Al-Dabbagh, B.; Saadi, H.; Gariballa, S.; Yasin, J. Effect of traditional Arabic coffee consumption on the glycemic index of Khalas dates tested in healthy and diabetic subjects. Asia Pac. J. Clin. Nutr. 2013, 22, 565–573. [Google Scholar] [CrossRef]
- AlGeffari, M.A.; Almogbel, E.S.; Alhomaidan, H.T.; El-Mergawi, R.; Barrimah, I.A. Glycemic indices, glycemic load and glycemic response for seventeen varieties of dates grown in Saudi Arabia. Ann. Saudi Med. 2016, 36, 397–403. [Google Scholar] [CrossRef]
- Al-Mssallem, M.Q. Consumption of Dates among Saudi Adults and its Association with the Prevalence of Type 2 Diabetes. Asian J. Clin. Nutr. 2018, 10, 58–64. [Google Scholar] [CrossRef]
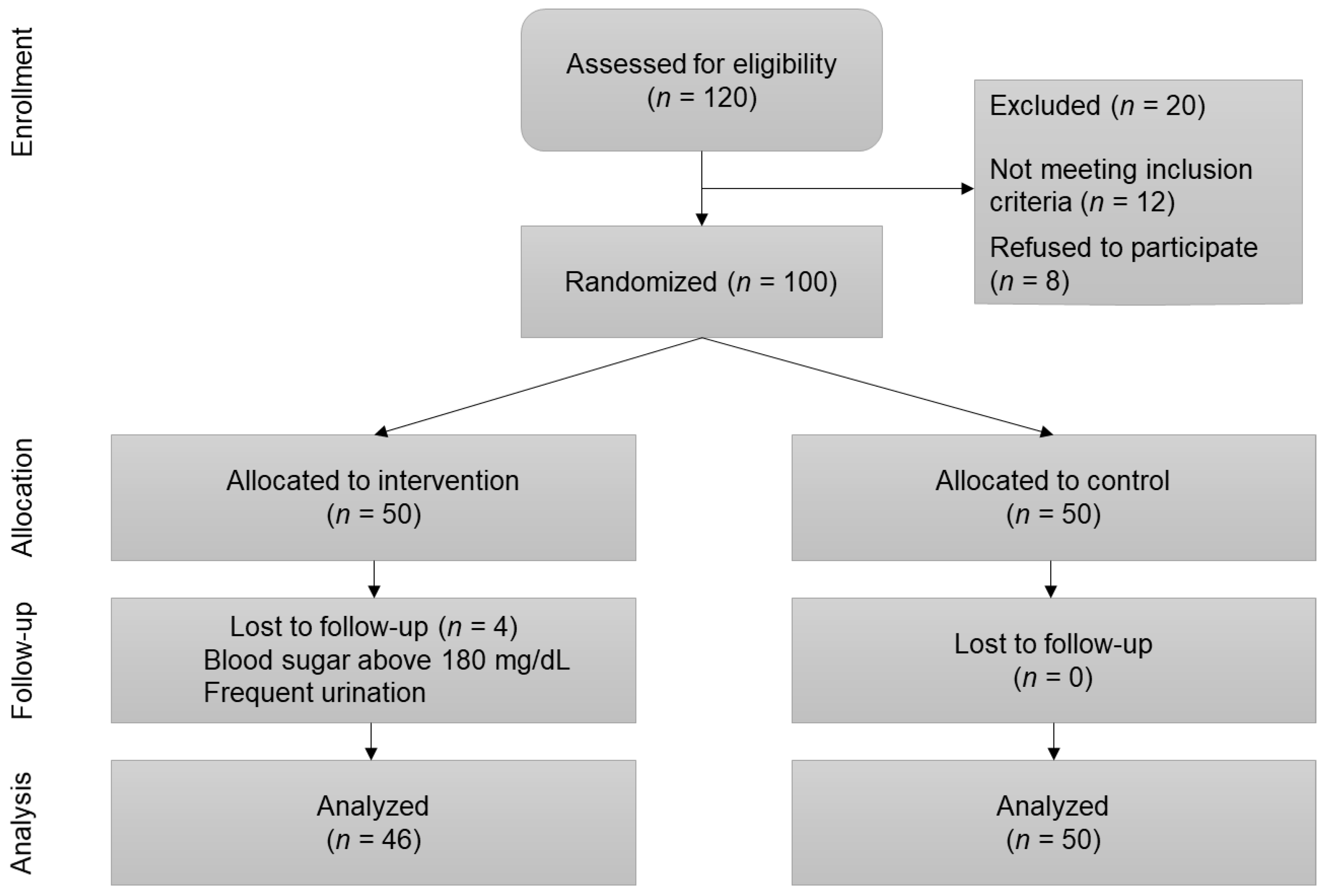
| Variable | Control (n = 50) | Intervention (n = 50) | Total Sample (n = 100, Female = 61, Male = 39) | p-Value between Groups |
|---|---|---|---|---|
| Age (years) | 56.86 ± 4.41 | 55.25 ± 2.71 | 56.00 ± 3.35 | 0.404 |
| Total cholesterol (mmol/L) | 3.68 ± 0.95 | 4.127 ± 1.04 | 3.39 ± 0.74 | 0.111 |
| Triglyceride (mmol/L) | 1.48 ± 0.57 | 1.693 ± 0.88 | 1.60 ± 0.78 | 0.125 |
| HbA1c (%) | 6.62 ± 0.67 | 6.59 ± 0.81 | 6.61 ± 74 | 0.584 |
| HDL (mmol/L) | 1.21 ± 0.29 | 1.149 ± 0.30 | 1.20 ± 0.36 | 0.794 |
| LDL (mmol/L) | 2.36 ± 0.70 | 2.3035 ± 0.92 | 2.30 ± 0.78 | 0.443 |
| BMI (kg/m2) | 29.92 ± 4.11 | 28.45 ± 7.69 | 29.22 ± 6.11 | 0.244 |
| Total SF-36 (score) | 284.89 ± 126.72 | 349.16 ± 146.71 | 315.47 ± 138.76 | 0.139 |
| SF-36 mental health | 48.00 ± 17.90 | 54.00 ± 16.20 | 51.00 ± 17.20 | 0.211 |
| Glycemia (mg/dL) | 109.44 ± 14.79 | 104.71 ± 13.16 | 107.38 ± 13.85 | 0.511 |
| Variable | Control Intra-Group ∆ Change (SD) | Intervention Intra-Group Δ Change (SD) | Intervention Effect between Groups p-Value |
|---|---|---|---|
| HbA1c (%) | −0.044 (−0.164; 0.076) | 0.043 (−0.082; 0.169) | 0.087 (−0.086; 0.261) |
| Total cholesterol (mmol/L) | −0.024 (−0.167; 0.119) | −0.209 (−0.358; −0.059) | −0.185 (−0.392; 0.022) |
| Triglyceride (mmol/L) | −0.006 (−0.102; 0.090) | 0.004 (−0.096; 0.105) | 0.010 (−0.128; 0.149) |
| HDL (mmol/L) | 0.032 (−0.060; 0.123) | −0.077 (−0.173; 0.018) | −0.109 (−0.241; 0.023) |
| LDL (mmol/L) | 0.055 (−0.125; 0.235) | −0.171 (−0.358; 0.016) | −0.226 (−0.485; 0.034) |
| Total SF-36 (score) | −2.23 (−44.45; 47.29) | 28.34 (17.45; 37.59) | 30.66 (12.45; 48.23) |
| SF-36 mental health | −0.04 (−3.83; 6.75) | 5.60 (1.24; 7.96) | 5.64 (0.57; 10.70) |
| Glycemia (mg/dL) | 6.71 (−2.51; 15.94) | −7.06 (−17.70; 3.57) | −13.77 (−28.73; 1.18) |
| BMI (kg/m2) | −0.50 (−2.00; 1.01) | 0.53 (−0.25; 0.81) | −0.03 (−0.39; 0.33) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alalwan, T.A.; Perna, S.; Mandeel, Q.A.; Abdulhadi, A.; Alsayyad, A.S.; D’Antona, G.; Negro, M.; Riva, A.; Petrangolini, G.; Allegrini, P.; et al. Effects of Daily Low-Dose Date Consumption on Glycemic Control, Lipid Profile, and Quality of Life in Adults with Pre- and Type 2 Diabetes: A Randomized Controlled Trial. Nutrients 2020, 12, 217. https://doi.org/10.3390/nu12010217
Alalwan TA, Perna S, Mandeel QA, Abdulhadi A, Alsayyad AS, D’Antona G, Negro M, Riva A, Petrangolini G, Allegrini P, et al. Effects of Daily Low-Dose Date Consumption on Glycemic Control, Lipid Profile, and Quality of Life in Adults with Pre- and Type 2 Diabetes: A Randomized Controlled Trial. Nutrients. 2020; 12(1):217. https://doi.org/10.3390/nu12010217
Chicago/Turabian StyleAlalwan, Tariq A., Simone Perna, Qaher A. Mandeel, Aalaa Abdulhadi, Adel Salman Alsayyad, Giuseppe D’Antona, Massimo Negro, Antonella Riva, Giovanna Petrangolini, Pietro Allegrini, and et al. 2020. "Effects of Daily Low-Dose Date Consumption on Glycemic Control, Lipid Profile, and Quality of Life in Adults with Pre- and Type 2 Diabetes: A Randomized Controlled Trial" Nutrients 12, no. 1: 217. https://doi.org/10.3390/nu12010217
APA StyleAlalwan, T. A., Perna, S., Mandeel, Q. A., Abdulhadi, A., Alsayyad, A. S., D’Antona, G., Negro, M., Riva, A., Petrangolini, G., Allegrini, P., & Rondanelli, M. (2020). Effects of Daily Low-Dose Date Consumption on Glycemic Control, Lipid Profile, and Quality of Life in Adults with Pre- and Type 2 Diabetes: A Randomized Controlled Trial. Nutrients, 12(1), 217. https://doi.org/10.3390/nu12010217







