Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
2.2. Experimental Protocol and Evaluation Plan
2.3. Incremental Power Tests
2.4. Blood Lactate Concentrations
2.5. Determination of Thresholds
2.6. Anthropometry
2.7. Dietary Assessment
2.8. Statistical Analysis
3. Results
4. Discussion
4.1. Limitations, Strengths and Future Research
4.2. Practical Application
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. American College of Sports Medicine Joint Position Statement. Nutrition and Athletic Performance. Med. Sci. Sports Exerc. 2016, 48, 543–568. [Google Scholar] [PubMed]
- Calleja-González, J.; Mielgo-Ayuso, J.; Sampaio, J.; Delextrat, A.; Ostojic, S.M.; Marques-Jiménez, D.; Arratibel, I.; Sánchez-Ureña, B.; Dupont, G.; Schelling, X.; et al. Brief ideas about evidence-based recovery in team sports. J. Exerc. Rehabil. 2018, 14, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, N.R.; DiMarco, N.M.; Langley, S. Position of the American Dietetic Association, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and athletic performance. J. Am. Diet. Assoc. 2013, 109, 509–527. [Google Scholar]
- Jeukendrup, A.E. Periodized Nutrition for Athletes. Sports Med. 2017, 47, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Terrados, N.; Mielgo-Ayuso, J.; Delextrat, A.; Ostojic, S.M.; Calleja-Gonzalez, J. Dietetic-nutritional, physical and physiological recovery methods post-competition in team sports. A review. J. Sports Med. Phys. Fit. 2018. epub ahead of print. [Google Scholar]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef]
- Wilson, J.M.; Fitschen, P.J.; Campbell, B.; Wilson, G.J.; Zanchi, N.; Taylor, L.; Wilborn, C.; Kalman, D.S.; Stout, J.R.; Hoffman, J.R.; et al. International Society of Sports Nutrition Position Stand: Beta-hydroxy-beta-methylbutyrate (HMB). J. Int. Soc. Sports Nutr. 2013, 10, 6. [Google Scholar] [CrossRef]
- Silva, V.R.; Belozo, F.L.; Micheletti, T.O.; Conrado, M.; Stout, J.R.; Pimentel, G.D.; Gonzalez, A.M. β-Hydroxy-β-Methylbutyrate Free Acid Supplementation May Improve Recovery and Muscle Adaptations After Resistance Training: A Systematic Review. Nutr. Res. 2017, 45, 1–9. [Google Scholar] [CrossRef]
- Stecker, R.A.; Harty, P.S.; Jagim, A.R.; Candow, D.G.; Kerksick, C.M. Timing of ergogenic aids and micronutrients on muscle and exercise performance. J. Int. Soc. Sports Nutr. 2019, 16, 37. [Google Scholar] [CrossRef]
- Chwalbinska-Moneta, J. Effect of creatine supplementation on aerobic performance and anaerobic capacity in elite rowers in the course of endurance training. Int. J. Sport. Exerc. Metab. 2003, 13, 173–183. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Calleja-Gonzalez, J.; Marqués-Jiménez, D.; Caballero-García, A.; Córdova, A.; Fernández-Lázaro, D. Effects of creatine supplementation on athletic performance in soccer players: A systematic review and meta-analysis. Nutrients 2019, 11, 757. [Google Scholar] [CrossRef] [PubMed]
- Saks, V.A.; Kongas, O.; Vendelin, M.; Kay, L. Role of the creatine/phosphocreatine system in the regulation of mitochondrial respiration. Acta Physiol. Scand. 2000, 168, 635–641. [Google Scholar] [CrossRef]
- Roberts, P.A.; Fox, J.; Peirce, N.; Jones, S.W.; Casey, A.; Greenhaff, P.L. Creatine ingestion augments dietary carbohydrate mediated muscle glycogen supercompensation during the initial 24 h of recovery following prolonged exhaustive exercise in humans. Amino Acids 2016, 48, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Young, J.F.; Bertram, H.C.; Theil, P.K.; Petersen, A.G.D.; Poulsen, K.A.; Rasmussen, M.; Malmendal, A.; Nielsen, N.C.; Vestergaard, M.; Oksbjerg, N. In vitro and in vivo studies of creatine monohydrate supplementation to Duroc and Landrace pigs. Meat Sci. 2007, 76, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Arazi, H.; Rahmaninia, F.; Hosseini, K.; Asadi, A. Effects of short term creatine supplementation and resistance exercises on resting hormonal and cardiovascular responses. Sci. Sports 2015, 30, 105–109. [Google Scholar] [CrossRef]
- Oliver, J.M.; Joubert, D.P.; Martin, S.E.; Crouse, S.F. Oral creatine supplementation’s decrease of blood lactate during exhaustive, incremental cycling. Int. J. Sport Exerc. Metab. 2013, 23, 252–258. [Google Scholar] [CrossRef]
- Bassit, R.; Pinheiro, C.; Vitzel, K.; Sproesser, A.; Silveira, L.; Curi, R. Effect of short-term creatine supplementation on markers of skeletal muscle damage after strenuous contractile activity. Eur. J. Appl. Physiol. 2010, 108, 945–955. [Google Scholar] [CrossRef]
- Durkalec-Michalski, K.; Jeszka, J.; Podgórski, T. The effect of a 12-week beta-hydroxy-beta-methylbutyrate (HMB) supplementation on highly-trained combat sports athletes: A randomised, double-blind, placebo-controlled crossover study. Nutrients 2017, 9, 753. [Google Scholar] [CrossRef]
- Durkalec-Michalski, K.; Jeszka, J. The efficacy of a β-hydroxy-β-methylbutyrate supplementation on physical capacity, body composition and biochemical markers in elite rowers: A randomised, double-blind, placebo-controlled crossover study. J. Int. Soc. Sports Nutr. 2015, 12, 31. [Google Scholar] [CrossRef]
- Lamboley, C.R.H.; Royer, D.; Dionne, I.J. Effects of β-hydroxy-beta-methylbutyrate on aerobic-performance components and body composition in college students. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 56–69. [Google Scholar] [CrossRef]
- He, X.; Duan, Y.; Yao, K.; Li, F.; Hou, Y.; Wu, G.; Yin, Y. β-Hydroxy-β-methylbutyrate, mitochondrial biogenesis, and skeletal muscle health. Amino Acids 2016, 48, 653–664. [Google Scholar] [CrossRef]
- Pinheiro, C.; Gerlinger-Romero, F.; Guimarães-Ferreira, L.; de Souza, A.J.; Vitzel, K.; Nachbar, R.; Nunes, M.; Curi, R. Metabolic and functional effects of beta-hydroxy-beta-methylbutyrate (HMB) supplementation in skeletal muscle. Eur. J. Appl. Physiol. 2012, 112, 2531–2537. [Google Scholar] [CrossRef] [PubMed]
- Holeček, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopenia Muscle 2017, 8, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.J.; Wilson, J.M.; Manninen, A.H. Effects of beta-hydroxy-beta-methylbutyrate (HMB) on exercise performance and body composition across varying levels of age, sex, and training experience: A review. Nutr. Metab. 2008, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; Givan, A.H.; Graybeal, A.J.; Villarreal, M.I.; Cross, A.G. β-Hydroxy β-methylbutyrate free acid alters cortisol responses, but not myofibrillar proteolysis, during a 24-h fast. Br. J. Nutr. 2018, 119, 517–526. [Google Scholar] [CrossRef]
- Knitter, A.E.; Panton, L.; Rathmacher, J.A.; Petersen, A.; Sharp, R. Effects of β-hydroxy-β-methylbutyrate on muscle damage after a prolonged run. J. Appl. Physiol. 2000, 89, 1340–1344. [Google Scholar] [CrossRef]
- Kresta, J.Y.; Oliver, J.M.; Jagim, A.R.; Fluckey, J.; Riechman, S.; Kelly, K.; Meininger, C.; Mertens-Talcott, S.U.; Rasmussen, C.; Kreider, R.B. Effects of 28 days of beta-alanine and creatine supplementation on muscle carnosine, body composition and exercise performance in recreationally active females. J. Int. Soc. Sports Nutr. 2014, 11, 55. [Google Scholar] [CrossRef]
- Stout, J.; Cramer, J.; Mielke, M.; O’Kroy, J.; Torok, D.; Zoeller, F. Effects of twenty-eight days of beta-alanine and creatine monohydrate supplementation on the physical working capacity at neuromuscular fatigue threshold. J. Strength Cond. Res. 2006, 20, 928–931. [Google Scholar]
- Jówko, E.; Ostaszewski, P.; Jank, M.; Sacharuk, J.; Zieniewicz, A.; Wilczak, J.; Nissen, S. Creatine and β-hydroxy-β-methylbutyrate (HMB) additively increase lean body mass and muscle strength during a weight-training program. Nutrition 2001, 17, 558–566. [Google Scholar] [CrossRef]
- Crowe, M.J.; O’Connor, D.M.; Lukins, J.E. The effects of β-hydroxy-β-methylbutyrate (HMB) and HMB/creatine supplementation on indices of health in highly trained athletes. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 184–197. [Google Scholar] [CrossRef]
- O’Connor, D.M.; Crowe, M.J. Effects of six weeks of beta-hydroxy-beta-methylbutyrate (HMB) and HMB/creatine supplementation on strength, power, and anthropometry of highly trained athletes. J. Strength Cond. Res. 2007, 21, 419–423. [Google Scholar] [CrossRef]
- Zajac, A.; Waskiewicz, Z.; Poprzecki, S.; Cholewa, J. Effects of creatine and HMß supplementation on anaerobic power and body composition in basketball players. J. Hum. Kinet. 2003, 10, 95–108. [Google Scholar]
- Faramarzi, M.; Nuri, R.; Banitalebi, E.; Sciences, S. The effect of short–term combination of HMB (beta-hydroxy-beta-methylbutyrate) and creatine supplementation on anaerobic performance and muscle injury markers in soccer players. Braz. J. Biomotricity 2009, 3, 366–375. [Google Scholar]
- O’Connor, D.M.; Crowe, M.J. Effects of beta-hydroxy-beta-methylbutyrate and creatine monohydrate supplementation on the aerobic and anaerobic capacity of highly trained athletes. J. Sports Med. Phys. Fit. 2003, 43, 64–68. [Google Scholar]
- Fernández-Landa, J.; Calleja-González, J.; León-Guereño, P.; Caballero-García, A.; Córdova, A.; Mielgo-Ayuso, J. Effect of the combination of creatine monohydrate plus HMB supplementation on sports performance, body composition, markers of muscle damage and hormone status: A systematic review. Nutrients 2019, 11, 2528. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and Athletic Performance. J. Acad. Nutr. Diet. 2016, 116, 501–528. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. J. Am. Med. Assoc. 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed]
- Ingham, S.; Whyte, G.; Jones, K.; Nevill, A. Determinants of 2000 m rowing ergometer performance in elite rowers. Eur. J. Appl. Physiol. 2002, 88, 243–246. [Google Scholar]
- Beelen, M.; Burke, L.M.; Gibala, M.J.; van Loon, L.J. Nutritional strategies to promote postexercise recovery. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 515–532. [Google Scholar] [CrossRef]
- Izquierdo-Gabarren, M.; De Txabarri Expósito, R.G.; García-PallarIs, J.; Sánchez-Medina, L.; De Villarreal, E.S.S.; Izquierdo, M. Concurrent endurance and strength training not to failure optimizes performance gains. Med. Sci. Sports Exerc. 2010, 42, 1191–1199. [Google Scholar] [CrossRef]
- Mejuto, G.; Arratibel, I.; Cámara, J.; Puente, A.; Iturriaga, J.; Calleja-González, J. The effect of a 6-week individual anaerobic threshold based programme in a traditional rowing crew. Biol. Sport 2012, 29, 297–301. [Google Scholar] [CrossRef]
- De Campos Mello, F.; Bertuzzi, R.; Franchini, E.; Candau, R. Rowing ergometer with the slide is more specific to rowers’ physiological evaluation. Res. Sports Med. 2014, 22, 136–146. [Google Scholar] [CrossRef]
- Tanner, R.K.; Fuller, K.L.; Ross, M.L.R. Evaluation of three portable blood lactate analysers: Lactate Pro, Lactate Scout and Lactate Plus. Eur. J. Appl. Physiol. 2010, 109, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.G.; Furber, M.J.W.; Van Someren, K.A.; Antón-Solanas, A.; Swart, J. The Physiological Profile of a Multiple Tour de France Winning Cyclist. Med. Sci. Sports Exerc. 2017, 49, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Weston, S.B. Reliability of using the D-max method to define physiological responses to incremental exercise testing. Physiol. Meas. 1997, 18, 145–154. [Google Scholar] [CrossRef]
- Stewart, A.; Marfell-Jones, M.; Olds, T.; de Ridder, H. International Standards for Anthropometric Assessment, 3rd ed.; ISAK: Lower Hutt, New Zealand, 2011. [Google Scholar]
- Carter, J.E.L. Body composition of montreal olympic athletes. In Physical Structure of Olympic Athletes Part I the Montreal Olympic Games Anthropological Project; Karger: Basel, Switzerland, 1982; pp. 107–116. [Google Scholar]
- Lee, R.C.; Wang, Z.; Heo, M.; Ross, R.; Jannsen, I.; Heymsfield, S.B. Total-body skeletal muscle mass: Development and cross-validation of anthropometric prediction models. Am. J. Clin. Nutr. 2000, 72, 796–803. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Zourdos, M.C.; Calleja-González, J.; Urdampilleta, A.; Ostojic, S.M. Dietary intake habits and controlled training on body composition and strength in elite female volleyball players during the season. Appl. Physiol. Nutr. Metab. 2015, 40, 827–834. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Collado, P.S.; Urdampilleta, A.; Martínez-Sanz, J.M.; Seco, J. Changes induced by diet and nutritional intake in the lipid profile of female professional volleyball players after 11 weeks of training. J. Int. Soc. Sports Nutr. 2013, 10, 55. [Google Scholar] [CrossRef]
- Farrán, A.; Zamora, R.; Cervera, P. Tablas de Composición de Alimentos del Centre D’Ensenyament Superior de Nutrició i Dietètica (CESNID); Universitat de Barcelona: Barcelona, Spain, 2004. [Google Scholar]
- Ferguson, C.J. An Effect Size Primer: A Guide for Clinicians and Researchers. Prof. Psychol. Res. Pract. 2009, 40, 532–538. [Google Scholar] [CrossRef]
- Lloyd, A.; Hodder, S.; Havenith, G. The interactive effect of cooling and hypoxia on forearm fatigue development. Eur. J. Appl. Physiol. 2015, 115, 2007–2018. [Google Scholar] [CrossRef][Green Version]
- Izquierdo-Gabarren, M.; Expósito, R.G.; de Villarreal, E.S.; Izquierdo, M. Physiological factors to predict on traditional rowing performance. Eur. J. Appl. Physiol. 2010, 108, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Xia, Y.; Jiang, J.; Du, H.; Guo, X.; Liu, X.; Li, C.; Huang, G.; Niu, K. Effect of beta-hydroxy-beta-methylbutyrate supplementation on muscle loss in older adults: A systematic review and meta-analysis. Arch. Gerontol. Geriatr. 2015, 61, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Chilibeck, P.; Kaviani, M.; Candow, D.; Zello, G.A. Effect of creatine supplementation during resistance training on lean tissue mass and muscular strength in older adults: A meta-analysis. Open Access J. Sports Med. 2017, 8, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Stallknecht, B.; Vissing, J.; Galbo, H. Lactate production and clearance in exercise. Effects of training. A mini-review. Scand. J. Med. Sci. Sports 1998, 8, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Baar, K. Nutrition and the adaptation to endurance training. Sports Med. 2014, 44, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Soultanakis, H.; Mandaloufas, M.; Platanou, T. Lactate threshold and performance adaptations to 4 weeks of training in untrained swimmers: Volume vs. intensity. J. Strength Cond. Res. 2012, 26, 131–137. [Google Scholar] [CrossRef]
- Kornasio, R.; Riederer, I.; Butler-Browne, G.; Mouly, V.; Uni, Z.; Halevy, O. β-hydroxy-β-methylbutyrate (HMB) stimulates myogenic cell proliferation, differentiation and survival via the MAPK/ERK and PI3K/Akt pathways. Biochim. Biophys. Acta Mol. Cell Res. 2009, 1793, 755–763. [Google Scholar] [CrossRef]
- Gerlinger-Romero, F.; Guimarães-Ferreira, L.; Giannocco, G.; Nunes, M.T. Chronic supplementation of beta-hydroxy-beta methylbutyrate (HMβ) increases the activity of the GH/IGF-I axis and induces hyperinsulinemia in rats. Growth Horm. IGF Res. 2011, 21, 57–62. [Google Scholar] [CrossRef]
- Smith, H.J.; Mukerji, P.; Tisdale, M.J. Attenuation of proteasome-induced proteolysis in skeletal muscle by β-hydroxy-β-methylbutyrate in cancer-induced muscle loss. Cancer Res. 2005, 65, 277–283. [Google Scholar]
- Nissen, S.; Sharp, R.; Ray, J.; Rathmacher, J.A.; Rice, D.; Fuller, J.C.; Connelly, A.S.; Abumrad, N. Effect of leucine metabolite β-hydroxy-β-methylbutyrate on muscle metabolism during resistance-exercise training. J. Appl. Physiol. 1996, 81, 2095–2104. [Google Scholar] [CrossRef]
- Nissen, S.; Abumrad, N. Nutritional role of the leucine metabolite β-hydroxy β-methylbutyrate (HMB). J. Nutr. Biochem. 1997, 8, 300–311. [Google Scholar] [CrossRef]
- Hespel, P.; Op’t Eijnde, B.; Van Leemputte, M.; Ursø, B.; Greenhaff, P.L.; Labarque, V.; Dymarkowski, S.; Van Hecke, P.; Richter, E.A. Oral creatine supplementation facilitates the rehabilitation of disuse atrophy and alters the expression of muscle myogenic factors in humans. J. Physiol. 2001, 536, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.; Aagaard, P.; Kadi, F.; Tufekovic, G.; Verney, J.; Olesen, J.L.; Suetta, C.; Kjær, M. Creatine supplementation augments the increase in satellite cell and myonuclei number in human skeletal muscle induced by strength training. J. Physiol. 2006, 573, 525–534. [Google Scholar] [CrossRef] [PubMed]
| PLG | CrMG | HMBG | CrM-HMBG | |
|---|---|---|---|---|
| Energy (kcal) | 3340 ± 350 | 3358 ± 358 | 3290 ± 410 | 3375 ± 395 |
| Energy (kcal/kg) | 44 ± 6 | 45 ± 6 | 45 ± 6 | 45 ± 7 |
| Protein (g) | 143 ± 24 | 145 ± 26 | 141 ± 29 | 143 ± 26 |
| Protein (%) | 17 ± 3 | 18 ± 3 | 18 ± 3 | 17 ± 3 |
| Protein (g/kg) | 2 ± 0 | 2 ± 1 | 2 ± 1 | 2 ± 0 |
| Animal protein (g) | 84 ± 23 | 86 ± 25 | 81 ± 19 | 86 ± 25 |
| Vegetal protein (g) | 60 ± 11 | 58 ± 15 | 62 ± 19 | 59 ± 17 |
| Fat (g) | 101 ± 20 | 103 ± 21 | 99 ± 21 | 101 ± 22 |
| Fat (%) | 27 ± 4 | 28 ± 4 | 27 ± 5 | 28 ± 5 |
| Fat (g/kg) | 2 ± 0 | 2 ± 1 | 2 ± 1 | 2 ± 1 |
| Total carbohydrates (g) | 450 ± 55 | 460 ± 60 | 459 ± 58 | 453 ± 61 |
| Carbohydrates (%) | 54 ± 5 | 55 ± 5 | 55 ± 6 | 54 ± 5 |
| Carbohydrates (g/kg) | 6 ± 1 | 6 ± 1 | 6 ± 1 | 6 ± 1 |
| Fe (mg) | 24 ± 7 | 24 ± 7 | 24 ± 7 | 23 ± 8 |
| Group | T1 | T2 | P (TxG) | η2p |
|---|---|---|---|---|
| Body mass (Kg) | ||||
| PLG | 81.9 ± 6.3 | 80.0 ± 5.3 * | 0.883 | 0.028 |
| CrMG | 81.2 ± 5.0 | 78.6 ± 5.4 * | ||
| HMBG | 79.9 ± 12.2 | 77.6 ± 11.1 * | ||
| CrM-HMBG | 78.0 ± 4.7 | 75.5 ± 4.5 * | ||
| 6 Skinfolds (mm) | ||||
| PLG | 51.6 ± 18.9 | 48.8 ± 16.3 | 0.790 | 0.050 |
| CrMG | 57.0 ± 6.5 | 54.7 ± 14.1 | ||
| HMBG | 54.2 ± 11.4 | 52.0 ± 13.0 | ||
| CrM-HMBG | 50.4 ± 7.1 | 47.4 ± 4.9 | ||
| Fat mass (kg) | ||||
| PLG | 7.3 ± 2.7 | 6.4 ± 2.3 * | 0.207 | 0.255 |
| CrMG | 6.1 ± 0.7 | 5.8 ± 0.7 * | ||
| HMBG | 6.8 ± 1.3 | 6.3 ± 1.1 * | ||
| CrM-HMBG | 6.4 ± 0.8 | 6.2 ± 0.4 * | ||
| Muscle mass (kg) | ||||
| PLG | 33.3 ± 4.3 | 32.7 ± 4.1 * | 0.442 | 0.160 |
| CrMG | 31.5 ± 1.9 | 31.2 ± 2.3 | ||
| HMBG | 32.8 ± 1.5 | 32.2 ± 1.1 | ||
| CrM-HMBG | 34.6 ± 1.3 | 34.6 ± 1.1 | ||
| Group | T1 | T2 | P (TxG) | η2p |
|---|---|---|---|---|
| WAT (W) | ||||
| PLG | 254 ± 34 | 259 ± 20 | 0.228 | 0.168 |
| CrMG | 242 ± 21 | 253 ± 12 | ||
| HMBG | 238 ± 40 | 253 ± 35 * | ||
| CrM-HMBG | 238 ± 22 | 264 ± 19 * | ||
| W4 (W) | ||||
| PLG | 242 ± 16 | 241 ± 31 | 0.196 | 0.181 |
| CrMG | 243 ± 20 | 247 ± 21 | ||
| HMBG | 238 ± 45 | 251 ± 36 | ||
| CrM-HMBG | 236 ± 29 | 262 ± 19 * | ||
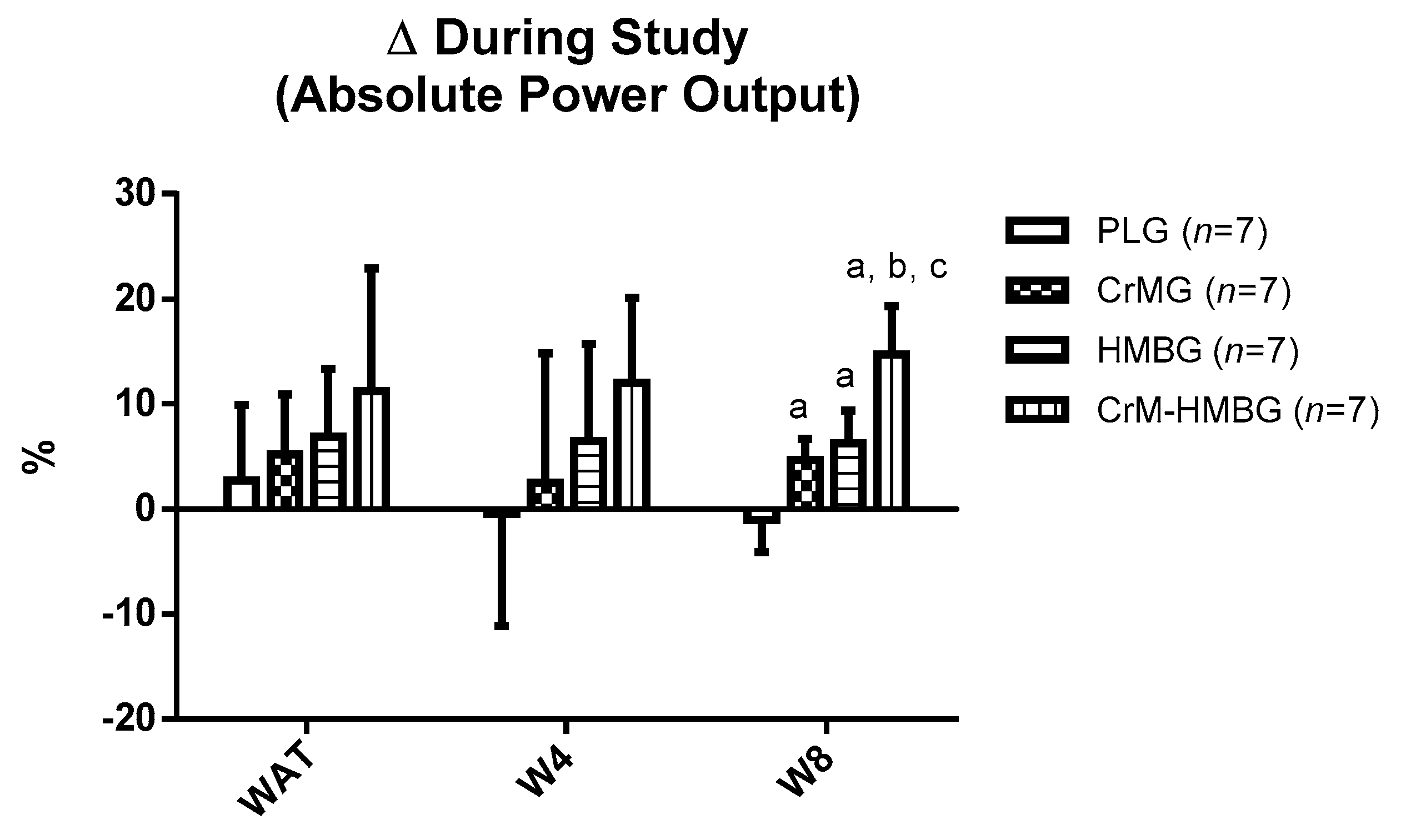
| W8 (W) | ||||
| PLG | 317 ± 19 | 314 ± 24 | <0.001 | 0.766 |
| CrMG | 300 ± 20 | 314 ± 21 * | ||
| HMBG | 295 ± 45 | 31 ± 48 * | ||
| CrM-HMBG | 288 ± 21 | 331 ± 35 * | ||
| Group | CrMG (A) | HMBG (B) | A + B | CrM-HMBG | CrM-HMBG − (A + B) |
|---|---|---|---|---|---|
| WAT | 134% | 203% | 338% | 403% | 66% |
| W4 | 397% | 1309% | 1705% | 2736% | 1031% |
| W8 | 364% | 514% | 878% | 1293% | 364% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Landa, J.; Fernández-Lázaro, D.; Calleja-González, J.; Caballero-García, A.; Córdova Martínez, A.; León-Guereño, P.; Mielgo-Ayuso, J. Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes. Nutrients 2020, 12, 193. https://doi.org/10.3390/nu12010193
Fernández-Landa J, Fernández-Lázaro D, Calleja-González J, Caballero-García A, Córdova Martínez A, León-Guereño P, Mielgo-Ayuso J. Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes. Nutrients. 2020; 12(1):193. https://doi.org/10.3390/nu12010193
Chicago/Turabian StyleFernández-Landa, Julen, Diego Fernández-Lázaro, Julio Calleja-González, Alberto Caballero-García, Alfredo Córdova Martínez, Patxi León-Guereño, and Juan Mielgo-Ayuso. 2020. "Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes" Nutrients 12, no. 1: 193. https://doi.org/10.3390/nu12010193
APA StyleFernández-Landa, J., Fernández-Lázaro, D., Calleja-González, J., Caballero-García, A., Córdova Martínez, A., León-Guereño, P., & Mielgo-Ayuso, J. (2020). Effect of Ten Weeks of Creatine Monohydrate Plus HMB Supplementation on Athletic Performance Tests in Elite Male Endurance Athletes. Nutrients, 12(1), 193. https://doi.org/10.3390/nu12010193











