Food and Beverage Consumption and Melanoma Risk: A Population-Based Case-Control Study in Northern Italy
Abstract
1. Introduction
2. Methods
2.1. Dietary Assessment
2.2. Additional Variables
2.3. Statistical Analysis
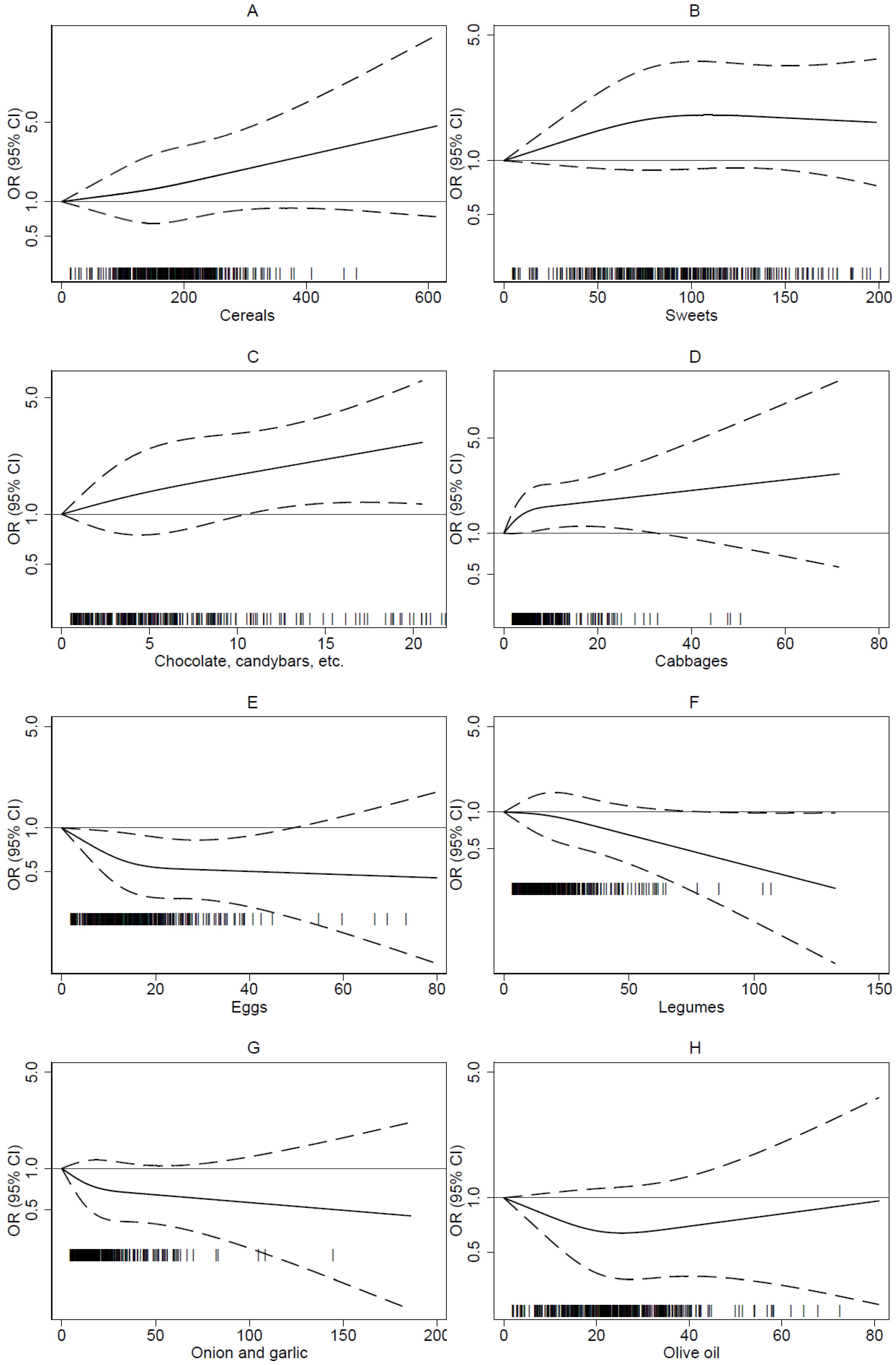
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Laversanne, M.; Brewster, D.H.; Gombe Mbalawa, C.; Kohler, B.; Pineros, M.; Steliarova-Foucher, E.; Swaminathan, R.; Antoni, S.; et al. Cancer Incidence in Five Continents: Inclusion criteria, highlights from Volume X and the global status of cancer registration. Int. J. Cancer 2015, 137, 2060–2071. [Google Scholar] [CrossRef] [PubMed]
- Dimitriou, F.; Krattinger, R.; Ramelyte, E.; Barysch, M.J.; Micaletto, S.; Dummer, R.; Goldinger, S.M. The World of Melanoma: Epidemiologic, Genetic, and Anatomic Differences of Melanoma across the Globe. Curr. Oncol. Rep. 2018, 20, 87. [Google Scholar] [CrossRef] [PubMed]
- Haenssle, H.A.; Mograby, N.; Ngassa, A.; Buhl, T.; Emmert, S.; Schon, M.P.; Rosenberger, A.; Bertsch, H.P. Association of Patient Risk Factors and Frequency of Nevus-Associated Cutaneous Melanomas. JAMA Dermatol. 2016, 152, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Ballotari, P.; Steinmaus, C.; Malagoli, C.; Luberto, F.; Malavolti, M.; Giorgi Rossi, P. Long-term mortality patterns in a residential cohort exposed to inorganic selenium in drinking water. Environ. Res. 2016, 150, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Vicentini, M.; Wise, L.A.; Sacchettini, C.; Malagoli, C.; Ballotari, P.; Filippini, T.; Malavolti, M.; Rossi, P.G. Cancer incidence following long-term consumption of drinking water with high inorganic selenium content. Sci. Total Environ. 2018, 635, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Malavolti, M.; Malagoli, C.; Crespi, C.M.; Brighenti, F.; Agnoli, C.; Sieri, S.; Krogh, V.; Fiorentini, C.; Farnetani, F.; Longo, C.; et al. Glycaemic index, glycaemic load and risk of cutaneous melanoma in a population-based, case-control study. Br. J. Nutr. 2017, 117, 432–438. [Google Scholar] [CrossRef]
- Malagoli, C.; Malavolti, M.; Agnoli, C.; Crespi, C.M.; Fiorentini, C.; Farnetani, F.; Longo, C.; Ricci, C.; Albertini, G.; Lanzoni, A.; et al. Diet Quality and Risk of Melanoma in an Italian Population. J. Nutr. 2015, 145, 1800–1807. [Google Scholar] [CrossRef]
- Tong, L.X.; Young, L.C. Nutrition: The future of melanoma prevention? J. Am. Acad. Dermatol. 2014, 71, 151–160. [Google Scholar] [CrossRef]
- Vinceti, M.; Malagoli, C.; Iacuzio, L.; Crespi, C.M.; Sieri, S.; Krogh, V.; Marmiroli, S.; Pellacani, G.; Venturelli, E. Serum Fatty acids and risk of cutaneous melanoma: A population-based case-control study. Dermatol. Res. Pr. 2013, 2013, 659394. [Google Scholar] [CrossRef]
- Malavolti, M.; Malagoli, C.; Fiorentini, C.; Longo, C.; Farnetani, F.; Ricci, C.; Albertini, G.; Lanzoni, A.; Reggiani, C.; Virgili, A.; et al. Association between dietary vitamin C and risk of cutaneous melanoma in a population of northern Italy. Int. J. Vitam. Nutr. Res. 2013, 83, 291–298. [Google Scholar] [CrossRef]
- Vinceti, M.; Malagoli, C.; Fiorentini, C.; Longo, C.; Crespi, C.M.; Albertini, G.; Ricci, C.; Lanzoni, A.; Reggiani, M.; Virgili, A.; et al. Inverse association between dietary vitamin D and risk of cutaneous melanoma in a northern Italy population. Nutr. Cancer 2011, 63, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Hohmann, C.B.; Bonamigo, R.R.; Segatto, M.M.; Costa, M.M.; Mastroeni, S.; Fortes, C. Could a specific dietary intake be a risk factor for cutaneous melanoma? Cutis 2016, 97, 421–425. [Google Scholar] [PubMed]
- Fortes, C.; Mastroeni, S.; Melchi, F.; Pilla, M.A.; Antonelli, G.; Camaioni, D.; Alotto, M.; Pasquini, P. A protective effect of the Mediterranean diet for cutaneous melanoma. Int. J. Epidemiol. 2008, 37, 1018–1029. [Google Scholar] [CrossRef] [PubMed]
- De Waure, C.; Quaranta, G.; Gualano, M.R.; Cadeddu, C.; Jovic-Vranes, A.; Djikanovic, B.; La Torre, G.; Ricciardi, W. Systematic review of studies investigating the association between dietary habits and cutaneous malignant melanoma. Public Health 2015, 129, 1099–1113. [Google Scholar] [CrossRef] [PubMed]
- Caini, S.; Masala, G.; Saieva, C.; Kvaskoff, M.; Savoye, I.; Sacerdote, C.; Hemmingsson, O.; Hammer Bech, B.; Overvad, K.; Tjonneland, A.; et al. Coffee, tea and melanoma risk: Findings from the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 2017, 140, 2246–2255. [Google Scholar] [CrossRef]
- Gandini, S.; Masala, G.; Palli, D.; Cavicchi, B.; Saieva, C.; Ermini, I.; Baldini, F.; Gnagnarella, P.; Caini, S. Alcohol, alcoholic beverages, and melanoma risk: A systematic literature review and dose-response meta-analysis. Eur. J. Nutr. 2018, 57, 2323–2332. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Fung, T.T.; Nan, H. An Epidemiological Review of Diet and Cutaneous Malignant Melanoma. Cancer Epidemiol. Biomarkers Prev. 2018, 27, 1115–1122. [Google Scholar] [CrossRef]
- Sieri, S.; Agnoli, C.; Pala, V.; Grioni, S.; Brighenti, F.; Pellegrini, N.; Masala, G.; Palli, D.; Mattiello, A.; Panico, S.; et al. Dietary glycemic index, glycemic load, and cancer risk: Results from the EPIC-Italy study. Sci. Rep. 2017, 7, 9757. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Human Basal Metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef]
- Pisani, P.; Faggiano, F.; Krogh, V.; Palli, D.; Vineis, P.; Berrino, F. Relative validity and reproducibility of a food frequency dietary questionnaire for use in the Italian EPIC centres. Int. J. Epidemiol. 1997, 26 (Suppl. 1), S152–S160. [Google Scholar] [CrossRef]
- Pala, V.; Sieri, C.; Palli, D.; Salvini, S.; Berrino, F.; Bellegotti, M.; Frasca, G.; Tumino, R.; Sacerdote, C.; Fiorini, L.; et al. Diet in the Italian EPIC cohorts: Presentation of data and methodological issues. Tumori J. 2003, 89, 594–607. [Google Scholar] [CrossRef]
- DNFCS. Dutch National Food Consumption Survey 2007–2010—Part 2, Foods NEVO Codes. Available online: https://www.rivm.nl/en/Documents_and_publications/Scientific/Tables_graphs/DNFCS/DNFCS_Core_survey_Part_2_Foods_NEVO_codes (accessed on 26 August 2019).
- Filippini, T.; Michalke, B.; Wise, L.A.; Malagoli, C.; Malavolti, M.; Vescovi, L.; Salvia, C.; Bargellini, A.; Sieri, S.; Krogh, V.; et al. Diet composition and serum levels of selenium species: A cross-sectional study. Food Chem. Toxicol. 2018, 115, 482–490. [Google Scholar] [CrossRef]
- Salvini, S.; Parpinel, M.; Gnagnarella, P. Banca Dati di Composizione Degli Alimenti Per Studi Epidemiologici in Italia, 269th ed.; European Institute of Oncology: Milan, Italy, 1998. [Google Scholar]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Adherence to Mediterranean diet and risk of cancer: A systematic review and meta-analysis of observational studies. Int. J. Cancer 2014, 135, 1884–1897. [Google Scholar] [CrossRef]
- Willett, W.; Stampfer, M.J. Total energy intake: Implications for epidemiologic analyses. Am. J. Epidemiol. 1986, 124, 17–27. [Google Scholar] [CrossRef]
- Rothman, K.J. Six persistent research misconceptions. J. Gen. Intern. Med. 2014, 29, 1060–1064. [Google Scholar] [CrossRef]
- Orsini, N.; Greenland, S. A procedure to tabulate and plot results after flexible modeling of a quantitative covariate. Stata J. 2011, 11, 1–29. [Google Scholar] [CrossRef]
- Harrell, F.E. Regression Modeling Strategies; Springer: New York, NY, USA, 2001. [Google Scholar]
- Sieri, S.; Krogh, V.; Agnoli, C.; Ricceri, F.; Palli, D.; Masala, G.; Panico, S.; Mattiello, A.; Tumino, R.; Giurdanella, M.C.; et al. Dietary glycemic index and glycemic load and risk of colorectal cancer: Results from the EPIC-Italy study. Int. J. Cancer 2015, 136, 2923–2931. [Google Scholar] [CrossRef]
- Sieri, S.; Krogh, V. Dietary glycemic index, glycemic load and cancer: An overview of the literature. Nutr. Metab. Cardiovasc. Dis. NMCD 2017, 27, 18–31. [Google Scholar] [CrossRef]
- Wu, D.; Hu, D.; Chen, H.; Shi, G.; Fetahu, I.S.; Wu, F.; Rabidou, K.; Fang, R.; Tan, L.; Xu, S.; et al. Glucose-regulated phosphorylation of TET2 by AMPK reveals a pathway linking diabetes to cancer. Nature 2018, 559, 637–641. [Google Scholar] [CrossRef]
- Gogas, H.; Trakatelli, M.; Dessypris, N.; Terzidis, A.; Katsambas, A.; Chrousos, G.P.; Petridou, E.T. Melanoma risk in association with serum leptin levels and lifestyle parameters: A case-control study. Ann. Oncol. 2008, 19, 384–389. [Google Scholar] [CrossRef]
- Grasgruber, P.; Hrazdira, E.; Sebera, M.; Kalina, T. Cancer Incidence in Europe: An Ecological Analysis of Nutritional and Other Environmental Factors. Front. Oncol. 2018, 8, 151. [Google Scholar] [CrossRef]
- Verhoeven, D.T.; Verhagen, H.; Goldbohm, R.A.; van den Brandt, P.A.; van Poppel, G. A review of mechanisms underlying anticarcinogenicity by brassica vegetables. Chem. Biol. Interact. 1997, 103, 79–129. [Google Scholar] [CrossRef]
- Zhang, Y. The molecular basis that unifies the metabolism, cellular uptake and chemopreventive activities of dietary isothiocyanates. Carcinogenesis 2012, 33, 2–9. [Google Scholar] [CrossRef]
- Filippini, T.; Cilloni, S.; Malavolti, M.; Violi, F.; Malagoli, C.; Tesauro, M.; Bottecchi, I.; Ferrari, A.; Vescovi, L.; Vinceti, M. Dietary intake of cadmium, chromium, copper, manganese, selenium and zinc in a Northern Italy community. J. Trace Elem. Med. Biol. 2018, 50, 508–517. [Google Scholar] [CrossRef]
- Matthews, N.H.; Fitch, K.; Li, W.Q.; Morris, J.S.; Christiani, D.C.; Qureshi, A.A.; Cho, E. Exposure to Trace Elements and Risk of Skin Cancer: A Systematic Review of Epidemiologic Studies. Cancer Epidemiol. Biomark. Prev. 2019, 28, 3–21. [Google Scholar] [CrossRef]
- Filippini, T.; Malagoli, C.; Wise, L.A.; Malavolti, M.; Pellacani, G.; Vinceti, M. Dietary cadmium intake and risk of cutaneous melanoma: An Italian population-based case-control study. J. Trace Elem. Med. Biol. 2019, 56, 100–106. [Google Scholar] [CrossRef]
- Kouris-Blazos, A.; Belski, R. Health benefits of legumes and pulses with a focus on Australian sweet lupins. Asia Pac. J. Clin. Nutr. 2016, 25, 1–17. [Google Scholar] [CrossRef]
- Visioli, F.; Franco, M.; Toledo, E.; Luchsinger, J.; Willett, W.C.; Hu, F.B.; Martinez-Gonzalez, M.A. Olive oil and prevention of chronic diseases: Summary of an International conference. Nutr. Metab. Cardiovasc. Dis. NMCD 2018, 28, 649–656. [Google Scholar] [CrossRef]
- Ruzzolini, J.; Peppicelli, S.; Andreucci, E.; Bianchini, F.; Scardigli, A.; Romani, A.; la Marca, G.; Nediani, C.; Calorini, L. Oleuropein, the Main Polyphenol of Olea europaea Leaf Extract, Has an Anti-Cancer Effect on Human BRAF Melanoma Cells and Potentiates the Cytotoxicity of Current Chemotherapies. Nutrients 2018, 10, 1950. [Google Scholar] [CrossRef]
- Fogli, S.; Arena, C.; Carpi, S.; Polini, B.; Bertini, S.; Digiacomo, M.; Gado, F.; Saba, A.; Saccomanni, G.; Breschi, M.C.; et al. Cytotoxic Activity of Oleocanthal Isolated from Virgin Olive Oil on Human Melanoma Cells. Nutr. Cancer 2016, 68, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.B.; Fan, J.; Lin, R.; Elf, S.; Ji, Q.; Zhao, L.; Jin, L.; Seo, J.H.; Shan, C.; Arbiser, J.L.; et al. Metabolic Rewiring by Oncogenic BRAF V600E Links Ketogenesis Pathway to BRAF-MEK1 Signaling. Mol. Cell 2015, 59, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Tomas, N.; Blanco Mejia, S.; Viguiliouk, E.; Khan, T.; Kendall, C.W.C.; Kahleova, H.; Rahelic, D.; Sievenpiper, J.L.; Salas-Salvado, J. Mediterranean diet, cardiovascular disease and mortality in diabetes: A systematic review and meta-analysis of prospective cohort studies and randomized clinical trials. Crit. Rev. Food Sci. Nutr. 2019. [Google Scholar] [CrossRef] [PubMed]
- Galbete, C.; Schwingshackl, L.; Schwedhelm, C.; Boeing, H.; Schulze, M.B. Evaluating Mediterranean diet and risk of chronic disease in cohort studies: An umbrella review of meta-analyses. Eur. J. Epidemiol. 2018, 33, 909–931. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.B. Vitamin D, cancer, and dysregulated phosphate metabolism. Endocrine 2019, 65, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Han, J.; Feskanich, D.; Cho, E.; Stampfer, M.J.; Willett, W.C.; Qureshi, A.A. Citrus Consumption and Risk of Cutaneous Malignant Melanoma. J. Clin. Oncol. 2015, 33, 2500–2508. [Google Scholar] [CrossRef]
- Feskanich, D.; Willett, W.C.; Hunter, D.J.; Colditz, G.A. Dietary intakes of vitamins A, C, and E and risk of melanoma in two cohorts of women. Br. J. Cancer 2003, 88, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Gavrieli, A.; Trichopoulou, A.; Valsta, L.M.; Ioannidou, S.; Berry, R.; Roe, M.; Harvey, L.; Finglas, P.; Glibetic, M.; Gurinovic, M.; et al. Identifying sources of measurement error in assessing dietary intakes—Results of a multi-country ring-trial. Nutr. Metab. Cardiovasc. Dis. NMCD 2019, 29, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Johansson, E.; Hussain, A.; Kuktaite, R.; Andersson, S.C.; Olsson, M.E. Contribution of organically grown crops to human health. Int. J. Environ. Res. Public Health 2014, 11, 3870–3893. [Google Scholar] [CrossRef]
- Baudry, J.; Assmann, K.E.; Touvier, M.; Alles, B.; Seconda, L.; Latino-Martel, P.; Ezzedine, K.; Galan, P.; Hercberg, S.; Lairon, D.; et al. Association of Frequency of Organic Food Consumption With Cancer Risk: Findings From the NutriNet-Sante Prospective Cohort Study. JAMA Intern. Med. 2018, 178, 1597–1606. [Google Scholar] [CrossRef]
- SINAB–Sistema D’Informazione Nazionale sull’Agricoltura Biologica. Facts and Figures on Organic Farming in Italy. 2018. Available online: http://www.sinab.it/sites/default/files/share/EN%20Bio%20in%20cifre%202018%20231118_%20Preview%20FINALE.pdf (accessed on 26 August 2019).
- Giampaoli, S.; Krogh, V.; Grioni, S.; Palmieri, L.; Gulizia, M.M.; Stamler, J.; Vanuzzo, D. Eating behaviours of italian adults: Results of the Osservatorio epidemiologico cardiovascolare/Health Examination Survey. Epidemiol. Prev. 2015, 39, 373–379. [Google Scholar] [PubMed]
| Cases | Controls | |||
|---|---|---|---|---|
| n | (%) | n | (%) | |
| Participants | 380 | (34.6) | 719 | (65.4) |
| Sex | ||||
| Male | 175 | (46.1) | 319 | (44.4) |
| Female | 205 | (53.9) | 400 | (55.6) |
| Age (years) | ||||
| <50 | 146 | (38.4) | 272 | (37.8) |
| ≥50 | 234 | (61.6) | 447 | (62.2) |
| Education (years) | ||||
| ≤5 | 91 | (24.1) | 170 | (23.8) |
| 6–8 | 95 | (25.1) | 176 | (24.6) |
| 9–13 | 136 | (36.0) | 266 | (37.2) |
| ≥14 | 56 | (14.8) | 103 | (14.4) |
| Marital status | ||||
| Married | 257 | (67.6) | 493 | (68.7) |
| Unmarried/single | 68 | (17.9) | 103 | (14.3) |
| Divorced | 23 | (6.0) | 48 | (6.6) |
| Widowed | 31 | (8.2) | 74 | (10.3) |
| Unknown | 1 | (0.3) | 1 | (0.1) |
| Body mass index (kg/m2) | ||||
| ≤19 | 31 | (8.2) | 45 | (6.3) |
| 20–24 | 164 | (43.3) | 306 | (42.5) |
| 25–29 | 133 | (35.0) | 287 | (39.9) |
| ≥30 | 52 | (13.7) | 81 | (11.3) |
| Phototype a | ||||
| I | 105 | (27.6) | 109 | (15.2) |
| II | 136 | (35.8) | 238 | (33.1) |
| III | 122 | (32.1) | 312 | (43.4) |
| IV | 17 | (4.5) | 60 | (8.3) |
| Sunburn history | ||||
| Never | 182 | (47.9) | 452 | (62.9) |
| Before 18 years | 108 | (28.4) | 164 | (22.8) |
| After 18 years | 90 | (23.7) | 103 | (14.3) |
| Greek Mediterranean index | ||||
| ≤2 | 61 | (16.1) | 102 | (14.2) |
| 3–4 | 140 | (36.8) | 250 | (34.8) |
| 5–6 | 128 | (33.7) | 282 | (39.2) |
| ≥7 | 51 | (13.4) | 85 | (11.8) |
| Glycemic load | ||||
| 50th (IQR) | 118.1 | (89.7–150.1) | 112.7 | (87.0–148.1) |
| Vitamin C (mg/day) | ||||
| 50th (IQR) | 113.7 | (78.0–154.1) | 121.0 | (83.66–162.6) |
| Vitamin D (mg/day) | ||||
| 50th (IQR) | 2.3 | (1.7–3.1) | 2.4 | (1.8–3.4) |
| Energy (kcal/day) | ||||
| 50th (IQR) | 1.928 | (1490–2443) | 1.906 | (1538–2365) |
| Food Items | Cases | Controls | ||||
|---|---|---|---|---|---|---|
| % Non Consumers | 50th | IQR | % Non Consumers | 50th | IQR | |
| Cereals and cereal products | 0.0 | 162.6 | 119.2–201.9 | 0.0 | 147.8 | 110.5–190.4 |
| Pasta, other grain | 0.5 | 56.0 | 37.8–80.0 | 1.3 | 55.9 | 37.1–76.9 |
| Rice | 17.9 | 3.9 | 2.4–6.9 | 20.4 | 3.8 | 1.9–6.3 |
| Bread | 5.5 | 86.4 | 45.6–124.0 | 8.1 | 76.4 | 44.4–113.7 |
| Crackers, crispbread, salty snacks | 9.5 | 4.6 | 2.2–17.4 | 10.4 | 4.5 | 2.1–16.3 |
| Meat and meat products | 0.0 | 117.3 | 88.7–157.6 | 0.1 | 126.2 | 90.6–161.5 |
| Red meat | 1.1 | 61.3 | 37.8–87.2 | 1.8 | 64.5 | 40.4–89.9 |
| White meat | 4.2 | 22.2 | 13.1–42.1 | 4.7 | 24.5 | 13.4–42.4 |
| Processed meat | 0.5 | 27.5 | 18.1–39.7 | 0.3 | 26.4 | 16.9–39.9 |
| Offal | 59.5 | 0.7 | 0.3–1.9 | 56.2 | 0.7 | 0.3–2.5 |
| Milk and dairy products | 0.0 | 176.9 | 100.0–298.6 | 0.0 | 209.9 | 111.3–306.9 |
| Milk | 35.3 | 110.8 | 30.3–224.3 | 32.7 | 134.4 | 36.5–223.4 |
| Yogurt | 36.3 | 27.4 | 13.8–70.6 | 33.8 | 41.2 | 13.5–108.3 |
| Cheeses (including fresh cheeses) | 0.0 | 42.4 | 27.9–58.0 | 0.8 | 38.4 | 27.1–54.9 |
| Eggs | 0.8 | 11.4 | 7.1–17.1 | 2.1 | 13.5 | 7.9–20.1 |
| Fish and seafood | 2.4 | 28.8 | 15.9–42.7 | 2.1 | 29.9 | 17.7–45.2 |
| Fish | 2.6 | 22.7 | 12.8–34.9 | 2.5 | 23.4 | 13.8–35.9 |
| Crustaceans and molluscs | 22.6 | 3.9 | 1.8–9.7 | 21.8 | 4.6 | 1.8–11.4 |
| Vegetables | 0.0 | 136.7 | 98.8–197.5 | 0.0 | 147.4 | 105.1–204.0 |
| Leafy vegetables | 0.5 | 23.7 | 12.3–43.5 | 0.1 | 25.5 | 14.6–44.2 |
| Other vegetables | 0.8 | 19.4 | 10.8–29.8 | 0.8 | 20.5 | 11.0–34.2 |
| Tomatoes | 0.8 | 53.3 | 31.8–85.4 | 0.7 | 54.3 | 33.5–86.5 |
| Root vegetables | 11.8 | 8.2 | 3.9–18.1 | 12.0 | 8.8 | 3.8–20.9 |
| Cabbages | 21.1 | 2.5 | 0.9–7.2 | 26.0 | 2.2 | 0.7–6.3 |
| Mushrooms | 22.4 | 1.6 | 0.8–3.3 | 20.7 | 1.7 | 0.8–3.8 |
| Onion and garlic | 0.5 | 10.8 | 6.4–20.5 | 0.3 | 12.6 | 7.3–21.9 |
| Legumes | 4.5 | 13.9 | 7.82–22.4 | 3.6 | 15.5 | 8.3–24.1 |
| Potatoes | 1.3 | 19.3 | 12.0–31.6 | 1.4 | 20.4 | 12.5–32.7 |
| Fresh fruit | 0.3 | 244.7 | 167.8–357.5 | 0.8 | 257.1 | 174.4–361.8 |
| Citrus fruits | 4.2 | 53.7 | 30.0–81.2 | 3.8 | 59.7 | 31.7–85.9 |
| All other fruits | 0.3 | 191.2 | 126.8–286.3 | 0.8 | 199.5 | 130.5–279.5 |
| Dried fruit, nuts and seeds | 10.0 | 0.5 | 0.3–1.63 | 11.0 | 0.5 | 0.3–1.8 |
| Sweets | 1.3 | 84.6 | 56.7–113.1 | 1.9 | 80.0 | 55.3–113.5 |
| Chocolate, candy bars, etc. | 25.0 | 4.4 | 2.6–8.5 | 31.4 | 3.6 | 2.1–7.4 |
| Sugar, honey, jam, confectionery | 13.2 | 14.9 | 7.2–27.61 | 12.2 | 14.5 | 7.8–27.6 |
| Ice-cream | 11.3 | 11.7 | 6.4–20.4 | 12.5 | 10.8 | 5.7–19.6 |
| Cakes, pies and pastries | 16.6 | 30.5 | 17.9–54.4 | 20.2 | 30.4 | 16.5–56.2 |
| Biscuits, dry cakes | 22.6 | 12.2 | 3.7–27.9 | 28.8 | 10.1 | 3.2–24.5 |
| Oils and fats | 0.0 | 25.2 | 19.2–32.4 | 0.1 | 26.3 | 20.6–33.2 |
| Vegetable fats and non-olive oils | 26.8 | 0.7 | 0.4–1.4 | 19.9 | 0.8 | 0.4–1.8 |
| Olive oil | 2.6 | 20.3 | 13.2–28.0 | 1.3 | 21.2 | 15.3–28.5 |
| Butter and other animal fats | 8.7 | 2.3 | 1.3–4.2 | 5.3 | 2.4 | 1.3–3.9 |
| Coffee | 8.2 | 79.1 | 49.7–116.6 | 9.9 | 83.9 | 49.3–123.2 |
| Tea | 39.7 | 21.8 | 2.7–86.5 | 47.3 | 10.6 | 1.7–65.4 |
| Red wine | 34.2 | 42.3 | 22.3–135.7 | 32.7 | 40.6 | 18.5–134.1 |
| White wine | 38.2 | 20.5 | 11.0–60.6 | 37.8 | 19.6 | 9.6–71.9 |
| Aperitif wines and beers | 31.6 | 26.1 | 13.3–41.2 | 30.2 | 26.0 | 13.8–50.7 |
| Spirits and liqueurs | 56.3 | 1.4 | 0.7–2.3 | 57.7 | 1.3 | 0.7–2.2 |
| Fruit juices | 38.7 | 45.9 | 25.7–107.3 | 39.6 | 43.5 | 21.8–102.0 |
| Soft drinks | 57.4 | 30.7 | 15.3–55.6 | 55.8 | 30.4 | 15.2–67.4 |
| Food Items | Cases/Controls | Median | OR a | (95% CI) | OR b | (95% CI) |
|---|---|---|---|---|---|---|
| Cereals and cereal products | ||||||
| 1st tertile (ref.) | 111/249 | 92.1 | 1.00 | - | 1.00 | - |
| 2nd tertile | 105/235 | 147.9 | 0.90 | (0.64–1.27) | 0.84 | (0.58–1.21) |
| 3nd tertile | 164/235 | 215.0 | 1.57 | (1.14–2.17) | 1.32 | (0.89–1.96) |
| Linear trend | 1.03 | (1.01–1.05) | 1.02 | (1.00–1.05) | ||
| Pasta, other grain | ||||||
| 1st tertile (ref.) | 129/259 | 30.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 123/230 | 55.5 | 1.13 | (0.81–1.56) | 1.06 | (0.76–1.48) |
| 3nd tertile | 128/230 | 90.2 | 1.33 | (0.95–1.85) | 1.15 | (0.82–1.63) |
| Linear trend | 1.04 | (1.00–1.08) | 1.02 | (0.98–1.06) | ||
| Rice | ||||||
| 1st tertile (ref.) | 133/273 | 1.2 | 1.00 | - | 1.00 | - |
| 2nd tertile | 122/223 | 3.8 | 1.14 | (0.83–1.56) | 1.11 | (0.80–1.54) |
| 3nd tertile | 125/223 | 9.7 | 1.20 | (0.88–1.64) | 1.20 | (0.88–1.65) |
| Linear trend | 1.01 | (0.84–1.21) | 1.02 | (0.85–1.22) | ||
| Bread | ||||||
| 1st tertile (ref.) | 131/273 | 35.0 | 1.00 | - | 1.00 | - |
| 2nd tertile | 101/223 | 76.7 | 0.89 | (0.63–1.26) | 0.89 | (0.62–1.27) |
| 3nd tertile | 148/223 | 133.2 | 1.33 | (0.97–1.82) | 1.21 | (0.83–1.77) |
| Linear trend | 1.02 | (1.00–1.04) | 1.01 | (0.99–1.04) | ||
| Crackers, crispbread, salty snacks | ||||||
| 1st tertile (ref.) | 137/296 | 1.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 127/212 | 4.5 | 1.44 | (1.04–2.01) | 1.37 | (0.98–1.92) |
| 3nd tertile | 116/211 | 22.7 | 1.20 | (0.87–1.65) | 1.11 | (0.79–1.55) |
| Linear trend | 1.04 | (0.95–1.14) | 1.01 | (0.92–1.12) | ||
| Meat and meat products | ||||||
| 1st tertile (ref.) | 151/247 | 76.7 | 1.00 | - | 1.00 | - |
| 2nd tertile | 117/236 | 125.9 | 0.85 | (0.61–1.19) | 0.83 | (0.58–1.19) |
| 3nd tertile | 112/236 | 177.6 | 0.80 | (0.57–1.12) | 0.77 | (0.53–1.14) |
| Linear trend | 0.99 | (0.97–1.02) | 1.00 | (0.97–1.03) | ||
| Red meat | ||||||
| 1st tertile (ref.) | 150/253 | 32.9 | 1.00 | - | 1.00 | - |
| 2nd tertile | 116/233 | 64.5 | 0.91 | (0.66–1.26) | 0.90 | (0.64–1.26) |
| 3nd tertile | 114/233 | 104.9 | 0.90 | (0.65–1.24) | 0.87 | (0.60–1.25) |
| Linear trend | 1.00 | (0.96–1.03) | 1.00 | (0.96–1.04) | ||
| White meat | ||||||
| 1st tertile (ref.) | 144/258 | 10.2 | 1.00 | - | 1.00 | - |
| 2nd tertile | 112/231 | 23.8 | 0.88 | (0.64–1.22) | 0.91 | (0.65–1.26) |
| 3nd tertile | 124/230 | 51.4 | 0.97 | (0.70–1.33) | 1.09 | (0.78–1.52) |
| Linear trend | 0.99 | (0.94–1.04) | 1.01 | (0.95–1.06) | ||
| Processed meat | ||||||
| 1st tertile (ref.) | 130/257 | 13.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 131/231 | 26.7 | 1.19 | (0.85–1.67) | 1.11 | (0.79–1.57) |
| 3nd tertile | 119/231 | 46.5 | 1.03 | (0.74–1.44) | 1.01 | (0.70–1.44) |
| Linear trend | 1.00 | (0.93–1.06) | 0.99 | (0.92–1.06) | ||
| Offal | ||||||
| 1st tertile (ref.) | 192/354 | 0.2 | 1.00 | - | 1.00 | - |
| 2nd tertile | 106/183 | 0.7 | 1.07 | (0.76–1.51) | 1.10 | (0.77–1.55) |
| 3nd tertile | 82/182 | 4.0 | 0.89 | (0.64–1.25) | 0.98 | (0.70–1.38) |
| Linear trendc | 1.00 | (0.97–1.03) | 1.01 | (0.98–1.04) | ||
| Milk and dairy products | ||||||
| 1st tertile (ref.) | 170/259 | 79.9 | 1.00 | - | 1.00 | - |
| 2nd tertile | 104/230 | 210.4 | 0.65 | (0.47–0.90) | 0.67 | (0.48–0.92) |
| 3nd tertile | 106/230 | 365.8 | 0.70 | (0.50–0.97) | 0.72 | (0.51–1.01) |
| Linear trend | 1.00 | (0.99–1.00) | 1.00 | (0.99–1.00) | ||
| Milk | ||||||
| 1st tertile (ref.) | 188/313 | 23.8 | 1.00 | - | 1.00 | - |
| 2nd tertile | 82/203 | 131.5 | 0.68 | (0.48–0.96) | 0.71 | (0.50–1.01) |
| 3nd tertile | 110/203 | 256.8 | 0.93 | (0.68–1.29) | 0.96 | (0.69–1.33) |
| Linear trend | 1.00 | (0.99–1.01) | 1.00 | (0.99–1.01) | ||
| Yogurt | ||||||
| 1st tertile (ref.) | 174/318 | 9.0 | 1.00 | - | 1.00 | - |
| 2nd tertile | 133/201 | 36.8 | 1.21 | (0.88–1.66) | 1.28 | (0.93–1.77) |
| 3nd tertile | 73/200 | 128.1 | 0.69 | (0.48–0.98) | 0.76 | (0.53–1.09) |
| Linear trend | 0.98 | (0.97–1.00) | 0.99 | (0.97–1.00) | ||
| Cheeses (including fresh cheeses) | ||||||
| 1st tertile (ref.) | 125/259 | 20.9 | 1.00 | - | 1.00 | - |
| 2nd tertile | 118/230 | 38.7 | 1.03 | (0.74–1.45) | 1.01 | (0.71–1.45) |
| 3nd tertile | 137/230 | 65.0 | 1.11 | (0.80–1.54) | 1.17 | (0.81–1.70) |
| Linear trend | 1.03 | (0.98–1.07) | 1.03 | (0.98–1.09) | ||
| Eggs | ||||||
| 1st tertile (ref.) | 176/249 | 6.2 | 1.00 | - | 1.00 | - |
| 2nd tertile | 120/235 | 13.5 | 0.71 | (0.52–0.96) | 0.73 | (0.53–0.99) |
| 3nd tertile | 84/235 | 23.4 | 0.53 | (0.38–0.74) | 0.58 | (0.41–0.82) |
| Linear trendc | 0.98 | (0.97–0.99) | 0.98 | (0.97–1.00) | ||
| Fish and seafood | ||||||
| 1st tertile (ref.) | 143/250 | 13.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 123/235 | 30.0 | 0.88 | (0.64–1.20) | 1.01 | (0.72–1.43) |
| 3nd tertile | 114/234 | 54.6 | 0.77 | (0.56–1.07) | 1.11 | (0.73–1.70) |
| Linear trend | 0.95 | (0.90–1.00) | 1.01 | (0.94–1.08) | ||
| Fish | ||||||
| 1st tertile (ref.) | 141/251 | 10.7 | 1.00 | - | 1.00 | - |
| 2nd tertile | 124/234 | 23.4 | 0.90 | (0.66–1.23) | 1.08 | (0.77–1.51) |
| 3nd tertile | 115/234 | 43.9 | 0.81 | (0.58–1.11) | 1.24 | (0.80–1.92) |
| Linear trend | 0.94 | (0.88–1.01) | 1.04 | (0.94–1.14) | ||
| Crustaceans and molluscs | ||||||
| 1st tertile (ref.) | 162/290 | 1.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 117/215 | 4.6 | 1.02 | (0.74–1.42) | 1.08 | (0.77–1.50) |
| 3nd tertile | 101/214 | 14.9 | 0.85 | (0.62–1.18) | 0.97 | (0.69–1.37) |
| Linear trendc | 0.99 | (0.97–1.00) | 0.99 | (0.98–1.01) | ||
| Vegetables | ||||||
| 1st tertile (ref.) | 148/243 | 87.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 111/238 | 147.0 | 0.74 | (0.54–1.02) | 0.80 | (0.57–1.14) |
| 3nd tertile | 121/238 | 235.3 | 0.81 | (0.58–1.12) | 0.95 | (0.63–1.43) |
| Linear trend | 0.99 | (0.97–1.00) | 1.00 | (0.98–1.02) | ||
| Leafy vegetables | ||||||
| 1st tertile (ref.) | 144/248 | 10.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 121/236 | 25.2 | 0.80 | (0.58–1.10) | 0.85 | (0.60–1.21) |
| 3nd tertile | 115/235 | 54.5 | 0.80 | (0.57–1.11) | 0.94 | (0.64–1.37) |
| Linear trend | 0.98 | (0.93–1.04) | 1.02 | (0.96–1.09) | ||
| Other vegetables | ||||||
| 1st tertile (ref.) | 137/250 | 8.4 | 1.00 | - | 1.00 | - |
| 2nd tertile | 141/235 | 20.5 | 1.12 | (0.81–1.55) | 1.24 | (0.88–1.73) |
| 3nd tertile | 102/234 | 40.6 | 0.81 | (0.58–1.13) | 0.96 | (0.66–1.39) |
| Linear trend | 0.95 | (0.88–1.02) | 0.98 | (0.90–1.07) | ||
| Tomatoes | ||||||
| 1st tertile (ref.) | 131/255 | 25.1 | 1.00 | - | 1.00 | - |
| 2nd tertile | 130/232 | 54.1 | 1.08 | (0.78–1.48) | 1.13 | (0.81–1.57) |
| 3nd tertile | 119/232 | 103.1 | 0.96 | (0.70–1.32) | 1.02 | (0.72–1.46) |
| Linear trend | 0.99 | (0.97–1.02) | 1.00 | (0.97–1.03) | ||
| Root vegetables | ||||||
| 1st tertile (ref.) | 145/284 | 2.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 135/218 | 8.6 | 1.23 | (0.88–1.70) | 1.31 | (0.93–1.83) |
| 3nd tertile | 100/217 | 28.9 | 0.82 | (0.58–1.16) | 0.90 | (0.62–1.32) |
| Linear trendc | 1.00 | (0.99–1.00) | 1.00 | (0.99–1.01) | ||
| Cabbages | ||||||
| 1st tertile (ref.) | 140/297 | 0.4 | 1.00 | - | 1.00 | - |
| 2nd tertile | 120/211 | 2.3 | 1.22 | (0.89–1.67) | 1.38 | (1.00–1.92) |
| 3nd tertile | 120/211 | 8.9 | 1.25 | (0.91–1.73) | 1.52 | (1.08–2.14) |
| Linear trendc | 1.01 | (0.99–1.03) | 1.02 | (1.00–1.04) | ||
| Mushrooms | ||||||
| 1st tertile (ref.) | 151/291 | 0.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 130/214 | 1.6 | 1.25 | (0.90–1.73) | 1.34 | (0.96–1.86) |
| 3nd tertile | 99/214 | 4.7 | 0.90 | (0.64–1.25) | 1.09 | (0.76–1.55) |
| Linear trendc | 0.97 | (0.93–1.01) | 0.99 | (0.95–1.03) | ||
| Onion and garlic | ||||||
| 1st tertile (ref.) | 176/270 | 5.7 | 1.00 | - | 1.00 | - |
| 2nd tertile | 98/225 | 12.5 | 0.67 | (0.48–0.94) | 0.70 | (0.50–0.98) |
| 3nd tertile | 106/224 | 27.9 | 0.71 | (0.51–1.00) | 0.80 | (0.56–1.14) |
| Linear trend | 0.92 | (0.86–0.99) | 0.94 | (0.87–1.02) | ||
| Legumes | ||||||
| 1st tertile (ref.) | 148/256 | 6.0 | 1.00 | - | 1.00 | - |
| 2nd tertile | 130/232 | 15.4 | 0.97 | (0.71–1.34) | 1.01 | (0.73–1.41) |
| 3nd tertile | 102/231 | 29.6 | 0.71 | (0.50–1.00) | 0.77 | (0.52–1.13) |
| Linear trend | 0.90 | (0.82–0.98) | 0.91 | (0.82–1.00) | ||
| Potatoes | ||||||
| 1st tertile (ref.) | 142/262 | 9.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 134/229 | 20.1 | 1.11 | (0.80–1.53) | 1.14 | (0.82–1.58) |
| 3nd tertile | 104/228 | 41.6 | 0.81 | (0.58–1.12) | 0.83 | (0.60–1.16) |
| Linear trend | 0.98 | (0.92–1.04) | 0.99 | (0.93–1.05) | ||
| Fresh fruit | ||||||
| 1st tertile (ref.) | 138/245 | 137.9 | 1.00 | - | 1.00 | - |
| 2nd tertile | 125/237 | 255.0 | 0.88 | (0.63–1.22) | 0.98 | (0.69–1.39) |
| 3nd tertile | 117/237 | 411.6 | 0.83 | (0.60–1.16) | 1.09 | (0.72–1.66) |
| Linear trend | 0.99 | (0.98–1.00) | 1.00 | (0.99–1.01) | ||
| Citrus fruits | ||||||
| 1st tertile (ref.) | 150/255 | 19.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 122/232 | 58.7 | 0.91 | (0.66–1.26) | 0.97 | (0.70–1.36) |
| 3nd tertile | 108/232 | 104.5 | 0.77 | (0.55–1.07) | 0.93 | (0.64–1.37) |
| Linear trend | 0.99 | (0.96–1.01) | 1.01 | (0.97–1.04) | ||
| All other fruits | ||||||
| 1st tertile (ref.) | 140/245 | 103.1 | 1.00 | - | 1.00 | - |
| 2nd tertile | 112/237 | 198.4 | 0.81 | (0.58–1.14) | 0.90 | (0.64–1.27) |
| 3nd tertile | 128/237 | 319.7 | 0.88 | (0.63–1.23) | 1.13 | (0.76–1.69) |
| Linear trend | 0.99 | (0.98–1.00) | 1.00 | (0.99–1.01) | ||
| Dried fruit, nuts and seeds | ||||||
| 1st tertile (ref.) | 145/277 | 0.2 | 1.00 | - | 1.00 | - |
| 2nd tertile | 123/221 | 0.5 | 1.16 | (0.83–1.62) | 1.21 | (0.86–1.70) |
| 3nd tertile | 112/221 | 3.2 | 1.00 | (0.72–1.38) | 1.08 | (0.77–1.52) |
| Linear trendc | 1.01 | (0.97–1.04) | 1.01 | (0.98–1.05) | ||
| Sweets | ||||||
| 1st tertile (ref.) | 128/258 | 42.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 124/231 | 81.2 | 1.20 | (0.85–1.70) | 1.19 | (0.84–1.69) |
| 3nd tertile | 128/230 | 130.7 | 1.21 | (0.86–1.69) | 1.22 | (0.84–1.76) |
| Linear trend | 1.01 | (0.98–1.03) | 1.01 | (0.98–1.04) | ||
| Chocolate, candy bars, etc. | ||||||
| 1st tertile (ref.) | 133/321 | 1.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 121/199 | 3.7 | 1.54 | (1.08–2.17) | 1.50 | (1.05–2.13) |
| 3nd tertile | 126/199 | 10.5 | 1.55 | (1.12–2.13) | 1.51 | (1.09–2.09) |
| Linear trend | 1.06 | (0.89–1.26) | 1.05 | (0.88–1.26) | ||
| Sugar, honey, jam, confectionery | ||||||
| 1st tertile (ref.) | 155/277 | 5.8 | 1.00 | - | 1.00 | - |
| 2nd tertile | 112/221 | 14.8 | 0.98 | (0.70–1.36) | 1.00 | (0.71–1.41) |
| 3nd tertile | 113/221 | 34.0 | 0.88 | (0.63–1.23) | 0.82 | (0.58–1.16) |
| Linear trend | 1.00 | (0.94–1.07) | 0.98 | (0.92–1.05) | ||
| Ice-cream | ||||||
| 1st tertile (ref.) | 139/275 | 4.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 123/222 | 11.0 | 1.23 | (0.89–1.70) | 1.24 | (0.89–1.71) |
| 3nd tertile | 118/222 | 25.7 | 1.08 | (0.77–1.51) | 1.12 | (0.79–1.58) |
| Linear trend | 1.03 | (0.94–1.13) | 1.04 | (0.94–1.14) | ||
| Cakes, pies and pastries | ||||||
| 1st tertile (ref.) | 152/309 | 12.0 | 1.00 | - | 1.00 | - |
| 2nd tertile | 116/205 | 30.3 | 1.18 | (0.84–1.67) | 1.23 | (0.87–1.74) |
| 3nd tertile | 112/205 | 70.8 | 1.14 | (0.83–1.57) | 1.26 | (0.89–1.78) |
| Linear trend | 1.00 | (0.97–1.03) | 1.01 | (0.98–1.05) | ||
| Biscuits, dry cakes | ||||||
| 1st tertile (ref.) | 147/300 | 2.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 112/210 | 10.1 | 1.09 | (0.78–1.51) | 1.10 | (0.79–1.53) |
| 3nd tertile | 121/209 | 31.8 | 1.22 | (0.89–1.68) | 1.14 | (0.82–1.58) |
| Linear trend | 1.04 | (0.96–1.13) | 1.02 | (0.95–1.11) | ||
| Oils and fats | ||||||
| 1st tertile (ref.) | 142/245 | 17.7 | 1.00 | - | 1.00 | - |
| 2nd tertile | 124/237 | 26.3 | 0.83 | (0.60–1.14) | 0.85 | (0.61–1.20) |
| 3nd tertile | 114/237 | 36.8 | 0.81 | (0.59–1.13) | 0.89 | (0.59–1.34) |
| Linear trend | 0.92 | (0.82–1.04) | 0.95 | (0.81–1.13) | ||
| Vegetable fats and non-olive oils | ||||||
| 1st tertile (ref.) | 180/322 | 0.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 119/199 | 0.8 | 1.09 | (0.77–1.54) | 1.10 | (0.77–1.57) |
| 3nd tertile | 81/198 | 6.0 | 0.75 | (0.52–1.06) | 0.72 | (0.50–1.04) |
| Linear trendc | 1.00 | (0.98–1.02) | 1.00 | (0.98–1.03) | ||
| Olive oil | ||||||
| 1st tertile (ref.) | 147/248 | 12.1 | 1.00 | - | 1.00 | - |
| 2nd tertile | 121/236 | 21.3 | 0.81 | (0.59–1.11) | 0.81 | (0.58–1.14) |
| 3nd tertile | 112/235 | 32.4 | 0.74 | (0.53–1.03) | 0.77 | (0.51–1.16) |
| Linear trend | 0.92 | (0.82–1.03) | 0.95 | (0.81–1.10) | ||
| Butter and other animal fats | ||||||
| 1st tertile (ref.) | 164/282 | 0.9 | 1.00 | - | 1.00 | - |
| 2nd tertile | 99/219 | 2.3 | 0.87 | (0.63–1.21) | 0.87 | (0.62–1.21) |
| 3nd tertile | 117/218 | 4.8 | 0.94 | (0.69–1.29) | 0.91 | (0.64–1.28) |
| Linear trendc | 1.00 | (0.96–1.04) | 1.00 | (0.96–1.05) | ||
| Coffee | ||||||
| 1st tertile (ref.) | 138/264 | 36.4 | 1.00 | - | 1.00 | - |
| 2nd tertile | 138/228 | 83.5 | 1.20 | (0.88–1.63) | 1.22 | (0.89–1.67) |
| 3nd tertile | 104/227 | 149.0 | 0.90 | (0.65–1.24) | 0.89 | (0.64–1.24) |
| Linear trend | 0.99 | (0.98–1.01) | 0.99 | (0.98–1.01) | ||
| Tea | ||||||
| 1st tertile (ref.) | 143/321 | 1.1 | 1.00 | - | 1.00 | - |
| 2nd tertile | 116/199 | 12.3 | 1.31 | (0.95–1.80) | 1.32 | (0.95–1.83) |
| 3nd tertile | 121/199 | 148.8 | 1.32 | (0.96–1.81) | 1.32 | (0.96–1.82) |
| Linear trend | 1.00 | (0.98–1.01) | 1.00 | (0.98–1.01) | ||
| Red wine | ||||||
| 1st tertile (ref.) | 168/323 | 13.0 | 1.00 | - | 1.00 | - |
| 2nd tertile | 103/198 | 39.5 | 1.09 | (0.76–1.57) | 1.18 | (0.81–1.71) |
| 3nd tertile | 109/198 | 206.7 | 1.13 | (0.79–1.61) | 1.17 | (0.80–1.69) |
| Linear trend | 1.00 | (0.99–1.01) | 1.00 | (0.99–1.01) | ||
| White wine | ||||||
| 1st tertile (ref.) | 165/333 | 6.4 | 1.00 | - | 1.00 | - |
| 2nd tertile | 123/193 | 19.7 | 1.44 | (1.01–2.06) | 1.38 | (0.95–1.99) |
| 3nd tertile | 92/193 | 128.1 | 1.03 | (0.73–1.45) | 1.05 | (0.73–1.49) |
| Linear trend | 0.99 | (0.98–1.01) | 0.99 | (0.98–1.01) | ||
| Aperitif wines and beers | ||||||
| 1st tertile (ref.) | 187/347 | 10.3 | 1.00 | - | 1.00 | - |
| 2nd tertile | 103/186 | 26.2 | 0.94 | (0.66–1.36) | 0.93 | (0.64–1.34) |
| 3nd tertile | 90/186 | 72.0 | 0.83 | (0.58–1.19) | 0.81 | (0.56–1.18) |
| Linear trend | 0.99 | (0.97–1.00) | 0.99 | (0.97–1.00) | ||
| Spirits and liqueurs | ||||||
| 1st tertile (ref.) | 204/372 | 0.5 | 1.00 | - | 1.00 | - |
| 2nd tertile | 91/174 | 1.4 | 0.93 | (0.64–1.36) | 0.93 | (0.63–1.36) |
| 3nd tertile | 85/173 | 4.5 | 0.92 | (0.63–1.35) | 0.94 | (0.63–1.39) |
| Linear trend c | 0.99 | (0.98–1.01) | 0.93 | (0.79–1.09) | ||
| Fruit juices | ||||||
| 1st tertile (ref.) | 172/331 | 15.6 | 1.00 | - | 1.00 | - |
| 2nd tertile | 101/194 | 42.9 | 1.04 | (0.73–1.48) | 1.11 | (0.77–1.59) |
| 3nd tertile | 107/194 | 136.1 | 1.07 | (0.77–1.49) | 1.29 | (0.90–1.86) |
| Linear trend | 0.99 | (0.98–1.01) | 0.99 | (0.98–1.01) | ||
| Soft drinks | ||||||
| 1st tertile (ref.) | 194/361 | 10.7 | 1.00 | - | 1.00 | - |
| 2nd tertile | 101/179 | 30.7 | 1.04 | (0.73–1.49) | 1.00 | (0.70–1.44) |
| 3nd tertile | 85/179 | 91.7 | 0.82 | (0.58–1.17) | 0.75 | (0.52–1.08) |
| Linear trend | 0.99 | (0.98–1.01) | 0.99 | (0.97–1.00) | ||
| Cases/Controls (n) | Men | Women | <50 years | ≥50 years | ||||
|---|---|---|---|---|---|---|---|---|
| 175/319 | 205/400 | 146/272 | 234/447 | |||||
| OR | (95% CI) | OR | (95% CI) | OR | (95% CI) | OR | (95% CI) | |
| Cereals and cereal products | 1.03 | (0.99–1.07) | 1.02 | (0.98–1.06) | 1.02 | (0.98–1.07) | 1.03 | (0.99–1.06) |
| Pasta, other grain | 1.01 | (0.96–1.07) | 1.03 | (0.97–1.10) | 1.00 | (0.93–1.07) | 1.02 | (0.97–1.08) |
| Rice | 1.01 | (0.78–1.31) | 1.04 | (0.79–1.38) | 0.98 | (0.71–1.35) | 1.08 | (0.86–1.35) |
| Bread | 1.03 | (0.98–1.08) | 1.00 | (0.96–1.04) | 1.04 | (0.99–1.09) | 1.01 | (0.97–1.05) |
| Crackers, crispbread, salty snacks | 0.98 | (0.84–1.15) | 1.05 | (0.92–1.20) | 0.89 | (0.73–1.09) | 1.06 | (0.94–1.19) |
| Meat and meat products | 1.01 | (0.97–1.06) | 0.99 | (0.95–1.03) | 1.00 | (0.96–1.04) | 1.00 | (0.96–1.03) |
| Red meat | 1.02 | (0.97–1.07) | 0.98 | (0.92–1.03) | 1.00 | (0.94–1.06) | 0.99 | (0.94–1.04) |
| White meat | 1.02 | (0.94–1.10) | 1.01 | (0.93–1.09) | 1.03 | (0.94–1.12) | 1.00 | (0.93–1.08) |
| Processed meat | 1.01 | (0.91–1.12) | 0.98 | (0.88–1.09) | 0.93 | (0.83–1.05) | 1.02 | (0.92–1.13) |
| Offal b | 0.95 | (0.90–1.01) | 1.04 | (1.00–1.09) | 1.06 | (0.99–1.14) | 0.99 | (0.95–1.03) |
| Milk and dairy products | 1.00 | (0.98–1.01) | 1.00 | (0.99–1.01) | 1.00 | (0.99–1.01) | 0.99 | (0.98–1.00) |
| Milk | 1.00 | (0.98–1.01) | 1.00 | (0.99–1.01) | 1.01 | (0.99–1.02) | 0.99 | (0.98–1.01) |
| Yogurt | 0.99 | (0.97–1.02) | 0.98 | (0.96–1.00) | 0.97 | (0.94–1.00) | 0.99 | (0.97–1.01) |
| Cheeses (including fresh cheeses) | 1.06 | (0.98–1.16) | 1.02 | (0.95–1.10) | 1.02 | (0.94–1.11) | 1.07 | (0.99–1.16) |
| Eggs b | 0.98 | (0.96–1.00) | 0.98 | (0.96–1.00) | 0.99 | (0.97–1.02) | 0.98 | (0.96–0.99) |
| Fish and seafood | 0.99 | (0.88–1.10) | 1.03 | (0.93–1.15) | 0.96 | (0.85–1.09) | 1.02 | (0.92–1.12) |
| Fish | 0.97 | (0.84–1.12) | 1.10 | (0.96–1.26) | 0.93 | (0.79–1.11) | 1.08 | (0.95–1.22) |
| Crustaceans and molluscs b | 1.00 | (0.98–1.03) | 0.99 | (0.97–1.01) | 1.00 | (0.98–1.02) | 0.99 | (0.96–1.01) |
| Vegetables | 0.98 | (0.95–1.02) | 1.00 | (0.97–1.03) | 1.01 | (0.97–1.04) | 0.99 | (0.97–1.02) |
| Leafy vegetables | 0.97 | (0.87–1.07) | 1.04 | (0.96–1.13) | 0.99 | (0.89–1.10) | 1.06 | (0.97–1.15) |
| Other vegetables | 1.01 | (0.87–1.17) | 0.98 | (0.88–1.08) | 0.99 | (0.85–1.16) | 0.97 | (0.87–1.07) |
| Tomatoes | 0.98 | (0.94–1.02) | 1.01 | (0.97–1.06) | 1.02 | (0.97–1.07) | 0.98 | (0.94–1.03) |
| Root vegetables b | 1.00 | (0.99–1.01) | 1.00 | (0.99–1.01) | 1.00 | (0.99–1.01) | 1.00 | (0.98–1.01) |
| Cabbages b | 1.02 | (0.99–1.05) | 1.02 | (0.99–1.05) | 1.02 | (0.99–1.06) | 1.03 | (1.00–1.06) |
| Mushrooms b | 0.97 | (0.91–1.03) | 1.00 | (0.95–1.05) | 1.07 | (0.99–1.16) | 0.95 | (0.89–1.01) |
| Onion and garlic | 0.93 | (0.83–1.05) | 0.94 | (0.83–1.05) | 0.94 | (0.82–1.08) | 0.94 | (0.85–1.04) |
| Legumes | 0.97 | (0.84–1.11) | 0.84 | (0.72–0.98) | 1.03 | (0.86–1.24) | 0.87 | (0.77–0.98) |
| Potatoes | 1.02 | (0.94–1.11) | 0.93 | (0.84–1.03) | 1.09 | (0.98–1.20) | 0.91 | (0.83–1.00) |
| Fresh fruit | 1.01 | (0.99–1.03) | 0.99 | (0.98–1.01) | 0.99 | (0.97–1.02) | 1.01 | (0.99–1.02) |
| Citrus fruits | 1.03 | (0.98–1.08) | 1.00 | (0.96–1.05) | 1.03 | (0.97–1.09) | 1.00 | (0.95–1.04) |
| All other fruits | 1.01 | (0.99–1.04) | 0.99 | (0.97–1.01) | 0.99 | (0.96–1.01) | 1.01 | (0.99–1.03) |
| Dried fruit, nuts and seeds b | 1.03 | (0.97–1.09) | 0.99 | (0.93–1.06) | 1.06 | (0.96–1.17) | 1.01 | (0.97–1.05) |
| Sweets | 0.99 | (0.95–1.04) | 1.02 | (0.99–1.06) | 1.01 | (0.97–1.05) | 1.01 | (0.97–1.05) |
| Chocolate, candy bars, etc. | 1.02 | (0.76–1.36) | 1.08 | (0.86–1.37) | 0.97 | (0.73–1.27) | 1.11 | (0.86–1.43) |
| Sugar, honey, jam, confectionery | 0.94 | (0.83–1.06) | 1.00 | (0.93–1.08) | 1.06 | (0.93–1.21) | 0.96 | (0.88–1.04) |
| Ice-cream | 1.07 | (0.94–1.23) | 1.01 | (0.88–1.16) | 0.92 | (0.79–1.07) | 1.15 | (1.01–1.31) |
| Cakes, pies and pastries | 0.99 | (0.93–1.05) | 1.02 | (0.97–1.07) | 1.01 | (0.96–1.07) | 1.00 | (0.95–1.06) |
| Biscuits, dry cakes | 0.98 | (0.85–1.13) | 1.08 | (0.98–1.20) | 1.00 | (0.88–1.14) | 1.05 | (0.95–1.17) |
| Oils and fats | 0.90 | (0.70–1.15) | 0.95 | (0.75–1.20) | 1.01 | (0.77–1.33) | 0.91 | (0.73–1.13) |
| Vegetable fats and non-olive oils | 1.00 | (0.97–1.03) | 1.01 | (0.97–1.04) | 1.02 | (0.98–1.06) | 1.00 | (0.97–1.03) |
| Olive oil | 0.93 | (0.74–1.18) | 0.93 | (0.76–1.15) | 0.93 | (0.72–1.22) | 0.94 | (0.77–1.14) |
| Butter and other animal fats b | 0.99 | (0.93–1.05) | 1.01 | (0.95–1.08) | 1.00 | (0.93–1.07) | 1.00 | (0.94–1.06) |
| Coffee | 1.00 | (0.97–1.03) | 0.99 | (0.97–1.01) | 1.01 | (0.98–1.03) | 0.98 | (0.96–1.01) |
| Tea | 1.01 | (0.99–1.03) | 0.99 | (0.98–1.01) | 1.00 | (0.97–1.02) | 1.00 | (0.98–1.01) |
| Red wine | 1.00 | (0.98–1.01) | 1.00 | (0.97–1.02) | 1.00 | (0.97–1.03) | 1.00 | (0.98–1.01) |
| White wine | 1.00 | (0.98–1.01) | 0.99 | (0.96–1.01) | 1.00 | (0.97–1.03) | 0.99 | (0.98–1.01) |
| Aperitif wines and beers | 1.00 | (0.97–1.02) | 0.97 | (0.94–1.01) | 0.98 | (0.96–1.01) | 0.99 | (0.96–1.01) |
| Spirits and liqueurs b | 0.99 | (0.98–1.01) | 0.93 | (0.86–1.00) | 0.94 | (0.90–1.00) | 1.00 | (0.98–1.02) |
| Fruit juices | 0.97 | (0.94–1.00) | 1.02 | (1.00–1.04) | 1.00 | (0.97–1.02) | 1.01 | (0.99–1.04) |
| Soft drinks | 0.98 | (0.96–1.01) | 0.99 | (0.97–1.01) | 0.98 | (0.96–1.00) | 1.00 | (0.97–1.02) |
| Cases/Controls (n) | Greek Mediterranean Index = 0–4 | Greek Mediterranean Index = 5–9 | ||
|---|---|---|---|---|
| 201/352 | 179/367 | |||
| OR | (95% CI) | OR | (95% CI) | |
| Cereals and cereal products | 1.04 | (0.99–1.08) | 1.00 | (0.95–1.06) |
| Pasta, other grain | 1.04 | (0.97–1.13) | 0.98 | (0.91–1.06) |
| Rice | 1.55 | (1.04–2.31) | 0.79 | (0.54–1.15) |
| Bread | 1.01 | (0.96–1.06) | 1.01 | (0.95–1.07) |
| Crackers, crispbread, salty snacks | 1.05 | (0.87–1.27) | 1.08 | (0.90–1.28) |
| Meat and meat products | 1.07 | (1.01–1.13) | 1.00 | (0.95–1.05) |
| Red meat | 1.09 | (1.01–1.17) | 0.95 | (0.88–1.02) |
| White meat | 1.10 | (0.99–1.21) | 0.99 | (0.89–1.10) |
| Processed meat | 0.91 | (0.80–1.03) | 1.20 | (1.04–1.37) |
| Offal b | 1.02 | (0.97–1.06) | 0.97 | (0.90–1.05) |
| Milk and dairy products | 1.00 | (0.99–1.01) | 0.98 | (0.96–1.00) |
| Milk | 1.00 | (0.99–1.02) | 0.99 | (0.96–1.01) |
| Yogurt | 1.00 | (0.97–1.03) | 0.97 | (0.93–1.01) |
| Cheeses (including fresh cheeses) | 1.04 | (0.96–1.14) | 1.00 | (0.89–1.12) |
| Eggs b | 0.98 | (0.95–1.01) | 0.99 | (0.96–1.01) |
| Fish and seafood | 0.91 | (0.76–1.09) | 1.05 | (0.92–1.19) |
| Fish | 0.92 | (0.74–1.13) | 1.08 | (0.92–1.27) |
| Crustaceans and molluscs b | 0.99 | (0.95–1.02) | 1.00 | (0.97–1.02) |
| Vegetables | 0.97 | (0.93–1.01) | 0.99 | (0.95–1.03) |
| Leafy vegetables | 0.97 | (0.85–1.10) | 1.06 | (0.95–1.18) |
| Other vegetables | 1.06 | (0.86–1.31) | 0.85 | (0.73–1.00) |
| Tomatoes | 0.93 | (0.87–0.99) | 0.99 | (0.94–1.05) |
| Root vegetables b | 1.00 | (0.98–1.02) | 1.00 | (0.99–1.01) |
| Cabbages b | 1.03 | (0.98–1.09) | 1.03 | (0.99–1.06) |
| Mushrooms b | 1.14 | (1.03–1.25) | 0.96 | (0.89–1.03) |
| Onion and garlic | 0.98 | (0.81–1.19) | 0.93 | (0.82–1.04) |
| Legumes | 0.99 | (0.82–1.20) | 0.84 | (0.70–1.01) |
| Potatoes | 0.89 | (0.78–1.02) | 1.03 | (0.92–1.14) |
| Fresh fruit | 1.01 | (0.98–1.03) | 1.01 | (0.99–1.03) |
| Citrus fruits | 1.06 | (0.99–1.14) | 1.03 | (0.97–1.09) |
| All other fruits | 1.00 | (0.97–1.03) | 1.00 | (0.98–1.03) |
| Dried fruit, nuts and seeds b | 1.03 | (0.94–1.14) | 1.00 | (0.95–1.04) |
| Sweets | 0.99 | (0.95–1.04) | 1.00 | (0.96–1.05) |
| Chocolate, candy bars, etc. | 0.90 | (0.67–1.23) | 1.24 | (0.89–1.72) |
| Sugar, honey, jam, confectionery | 1.13 | (0.97–1.31) | 0.95 | (0.87–1.05) |
| Ice-cream | 0.96 | (0.81–1.14) | 1.07 | (0.88–1.31) |
| Cakes, pies and pastries | 0.98 | (0.92–1.05) | 1.02 | (0.96–1.09) |
| Biscuits, dry cakes | 0.96 | (0.84–1.10) | 1.00 | (0.86–1.17) |
| Oils and fats | 0.88 | (0.63–1.25) | 0.91 | (0.68–1.22) |
| Vegetable fats and non-olive oils b | 1.01 | (0.97–1.06) | 1.00 | (0.96–1.04) |
| Olive oil | 0.77 | (0.56–1.07) | 0.97 | (0.75–1.25) |
| Butter and other animal fats b | 1.06 | (0.98–1.15) | 0.97 | (0.88–1.07) |
| Coffee | 1.01 | (0.98–1.03) | 0.99 | (0.96–1.02) |
| Tea | 0.99 | (0.96–1.01) | 0.99 | (0.97–1.01) |
| Red wine | 0.98 | (0.96–1.01) | 0.99 | (0.97–1.02) |
| White wine | 0.99 | (0.96–1.02) | 1.03 | (1.00–1.07) |
| Aperitif wines and beers | 0.98 | (0.96–1.01) | 1.01 | (0.98–1.05) |
| Spirits and liqueurs b | 0.99 | (0.96–1.02) | 1.00 | (0.97–1.02) |
| Fruit juices | 1.01 | (0.98–1.05) | 0.99 | (0.96–1.02) |
| Soft drinks | 1.00 | (0.98–1.01) | 0.99 | (0.94–1.03) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malagoli, C.; Malavolti, M.; Farnetani, F.; Longo, C.; Filippini, T.; Pellacani, G.; Vinceti, M. Food and Beverage Consumption and Melanoma Risk: A Population-Based Case-Control Study in Northern Italy. Nutrients 2019, 11, 2206. https://doi.org/10.3390/nu11092206
Malagoli C, Malavolti M, Farnetani F, Longo C, Filippini T, Pellacani G, Vinceti M. Food and Beverage Consumption and Melanoma Risk: A Population-Based Case-Control Study in Northern Italy. Nutrients. 2019; 11(9):2206. https://doi.org/10.3390/nu11092206
Chicago/Turabian StyleMalagoli, Carlotta, Marcella Malavolti, Francesca Farnetani, Caterina Longo, Tommaso Filippini, Giovanni Pellacani, and Marco Vinceti. 2019. "Food and Beverage Consumption and Melanoma Risk: A Population-Based Case-Control Study in Northern Italy" Nutrients 11, no. 9: 2206. https://doi.org/10.3390/nu11092206
APA StyleMalagoli, C., Malavolti, M., Farnetani, F., Longo, C., Filippini, T., Pellacani, G., & Vinceti, M. (2019). Food and Beverage Consumption and Melanoma Risk: A Population-Based Case-Control Study in Northern Italy. Nutrients, 11(9), 2206. https://doi.org/10.3390/nu11092206






