d-amino Acids in Health and Disease: A Focus on Cancer
Abstract
1. Introduction
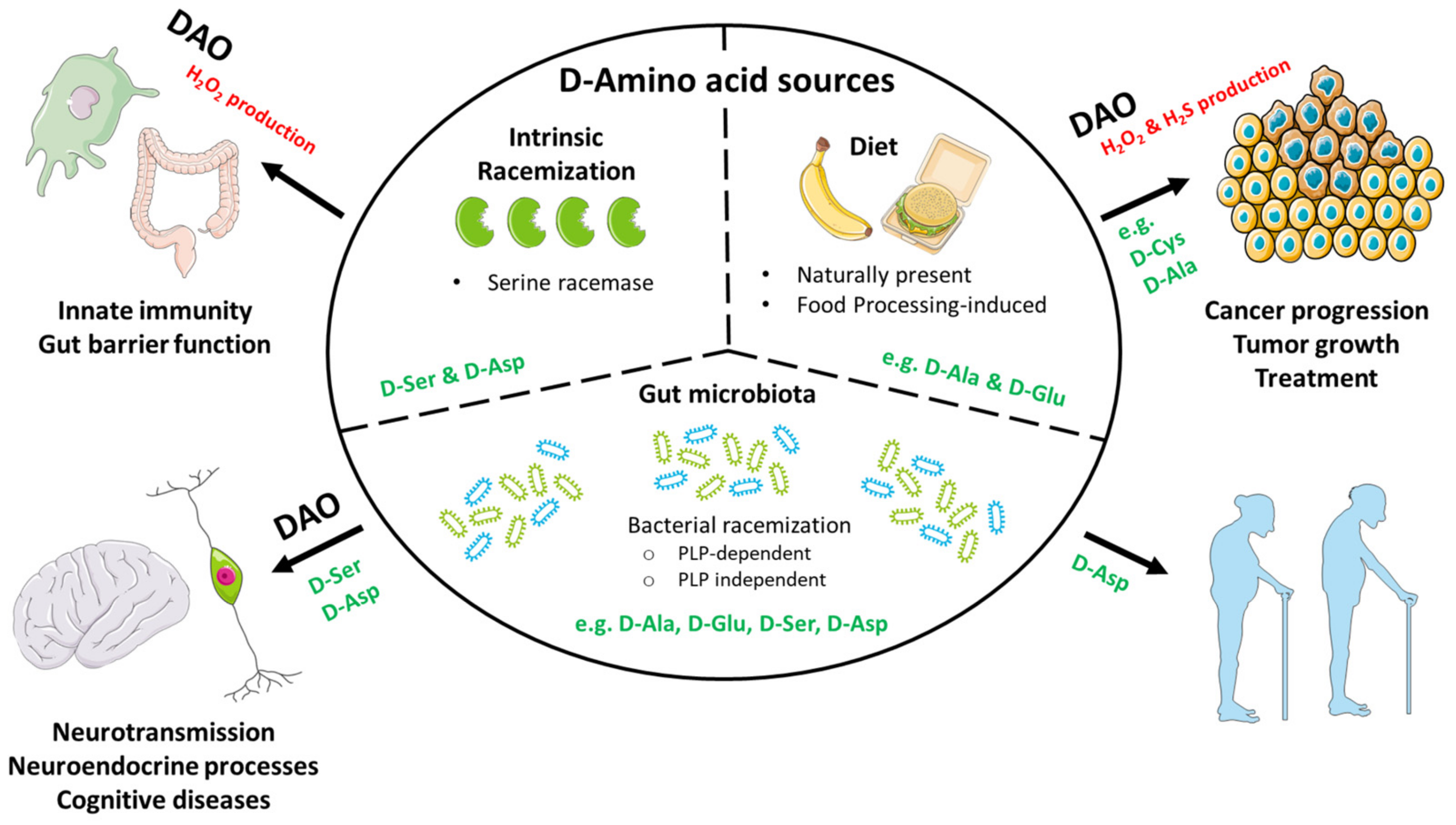
2. Origin and Sources
2.1. Microbially Produced d-Amino Acids
2.2. Dietary d-amino Acids
2.3. Intrinsic Racemization
2.4. Degradation
3. Physiological Roles of d-Amino Acids
3.1. Neurological
3.2. Aging
3.3. Innate Immune Defence
4. d-Amino Acids in Disease
4.1. Neurological Disorders
4.2. d-amino Acids in Cancer
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Auclair, J.L.; Patton, R.L. On the occurrence of d-Alanine in the haemolymph of the milkweed bug, oncopeltus fasciatus. Rev. Can. Biol. 1950, 9, 3–8. [Google Scholar] [PubMed]
- Stevens, C.M.; Halpern, P.E.; Gigger, R.P. Occurrence of d-amino acids in some natural materials. J. Biol. Chem. 1951, 190, 705–710. [Google Scholar] [PubMed]
- Robinson, T. d-amino acids in higher plants. Life Sci. 1976, 19, 1097–1102. [Google Scholar] [CrossRef]
- Corrigan, J.J. d-Amino Acids in Animals. Science 1969, 164, 142. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, A.; Giuditta, A. Presence of d-aspartate in squid axoplasm and in other regions of the cephalopod nervous system. J. Neurochem. 1978, 31, 1107–1108. [Google Scholar] [CrossRef] [PubMed]
- Montecucchi, P.C.; de Castiglione, R.; Piani, S.; Gozzini, L.; Erspamer, V. Amino acid composition and sequence of dermorphin, a novel opiate-like peptide from the skin of Phyllomedusa sauvagei. Int. J. Pept. Protein Res. 1981, 17, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Fukuda, M.; Sasaoka, K. Occurrence of d-amino acid aminotransferase in pea seedlings. Biochem. Biophys. Res. Commun. 1973, 52, 998–1002. [Google Scholar] [CrossRef]
- Man, E.H.; Sandhouse, M.E.; Burg, J.; Fisher, G.H. Accumulation of d-aspartic acid with age in the human brain. Science 1983, 220, 1407–1408. [Google Scholar] [CrossRef]
- Helfman, P.M.; Bada, J.L. Aspartic acid racemization in tooth enamel from living humans. Proc. Natl. Acad. Sci. USA 1975, 72, 2891–2894. [Google Scholar] [CrossRef]
- Masters, P.M.; Bada, J.L.; Zigler, J.S., Jr. Aspartic acid racemisation in the human lens during ageing and in cataract formation. Nature 1977, 268, 71–73. [Google Scholar] [CrossRef]
- Fisher, G.H.; Garcia, N.M.; Payan, I.L.; Cadilla-Perezrios, R.; Sheremata, W.A.; Man, E.H. d-aspartic acid in purified myelin and myelin basic protein. Biochem. Biophys. Res. Commun. 1986, 135, 683–687. [Google Scholar] [CrossRef]
- Masters, P.M.; Bada, J.L.; Zigler, J.S., Jr. Aspartic acid racemization in heavy molecular weight crystallins and water insoluble protein from normal human lenses and cataracts. Proc. Natl. Acad. Sci. USA 1978, 75, 1204–1208. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Masui, R.; Akino, T. The presence of free d-serine, d-alanine and d-proline in human plasma. Experientia 1992, 48, 986–988. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.W.; Gasper, M.; Lee, S.H.; Zukowski, J.; Ercal, N. d-amino acid levels in human physiological fluids. Chirality 1993, 5, 375–378. [Google Scholar] [CrossRef]
- Nagata, Y.; Higashi, M.; Ishii, Y.; Sano, H.; Tanigawa, M.; Nagata, K.; Noguchi, K.; Urade, M. The presence of high concentrations of free d-amino acids in human saliva. Life Sci. 2006, 78, 1677–1681. [Google Scholar] [CrossRef] [PubMed]
- Powell, J.T.; Vine, N.; Crossman, M. On the accumulation of d-aspartate in elastin and other proteins of the ageing aorta. Atherosclerosis 1992, 97, 201–208. [Google Scholar] [CrossRef]
- Ritz-Timme, S.; Laumeier, I.; Collins, M.J. Aspartic acid racemization: Evidence for marked longevity of elastin in human skin. Br. J. Dermatol. 2003, 149, 951–959. [Google Scholar] [CrossRef]
- Cloos, P.A.; Fledelius, C. Collagen fragments in urine derived from bone resorption are highly racemized and isomerized: A biological clock of protein aging with clinical potential. Biochem. J. 2000, 345, 473–480. [Google Scholar] [CrossRef]
- Kiriyama, Y.; Nochi, H. d-Amino Acids in the Nervous and Endocrine Systems. Scientifica 2016. [Google Scholar] [CrossRef]
- Ohide, H.; Miyoshi, Y.; Maruyama, R.; Hamase, K.; Konno, R. d-Amino acid metabolism in mammals: Biosynthesis, degradation and analytical aspects of the metabolic study. J. Chromatogr. 2011, 879, 3162–3168. [Google Scholar] [CrossRef]
- Wolosker, H.; Blackshaw, S.; Snyder, S.H. Serine racemase: A glial enzyme synthesizing d-serine to regulate glutamate-N-methyl-d-aspartate neurotransmission. Proc. Natl. Acad. Sci. USA 1999, 96, 13409. [Google Scholar] [CrossRef]
- Foltyn, V.N.; Bendikov, I.; De Miranda, J.; Panizzutti, R.; Dumin, E.; Shleper, M.; Li, P.; Toney, M.D.; Kartvelishvily, E.; Wolosker, H. Serine racemase modulates intracellular d-serine levels through an alpha,beta-elimination activity. J. Biol. Chem. 2005, 280, 1754–1763. [Google Scholar] [CrossRef]
- Raboni, S.; Marchetti, M.; Faggiano, S.; Campanini, B.; Bruno, S.; Marchesani, F.; Margiotta, M.; Mozzarelli, A. The Energy Landscape of Human Serine Racemase. Front. Mol. Biosci. 2019, 5, 112. [Google Scholar] [CrossRef]
- Uda, K.; Abe, K.; Dehara, Y.; Mizobata, K.; Sogawa, N.; Akagi, Y.; Saigan, M.; Radkov, A.D.; Moe, L.A. Distribution and evolution of the serine/aspartate racemase family in invertebrates. Amino Acids 2016, 48, 387–402. [Google Scholar] [CrossRef]
- Matsuda, S.; Katane, M.; Maeda, K.; Kaneko, Y.; Saitoh, Y.; Miyamoto, T.; Sekine, M.; Homma, H. Biosynthesis of d-aspartate in mammals: The rat and human homologs of mouse aspartate racemase are not responsible for the biosynthesis of d-aspartate. Amino Acids 2015, 47, 975–985. [Google Scholar] [CrossRef]
- Friedman, M. Origin, Microbiology, nutrition, and pharmacology of d-amino acids. Chem. Biodivers. 2010, 7, 1491–1530. [Google Scholar] [CrossRef]
- Mutaguchi, Y.; Ohmori, T.; Akano, H.; Doi, K.; Ohshima, T. Distribution of d-amino acids in vinegars and involvement of lactic acid bacteria in the production of d-amino acids. SpringerPlus 2013, 2, 691. [Google Scholar] [CrossRef]
- Jin, D.; Miyahara, T.; Oe, T.; Toyo’oka, T. Determination of d-amino acids labeled with fluorescent chiral reagents, R(-)- and S(+)-4-(3-isothiocyanatopyrrolidin-1-yl)-7-(N,N-dimethylaminosulfonyl)-2,1,3-benzoxadiazoles, in biological and food samples by liquid chromatography. Anal. Biochem. 1999, 269, 124–132. [Google Scholar] [CrossRef]
- Aliashkevich, A.; Alvarez, L.; Cava, F. New Insights Into the Mechanisms and Biological Roles of d-Amino Acids in Complex Eco-Systems. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Sasabe, J.; Miyoshi, Y.; Rakoff-Nahoum, S.; Zhang, T.; Mita, M.; Davis, B.M.; Hamase, K.; Waldor, M.K. Interplay between microbial d-amino acids and host d-amino acid oxidase modifies murine mucosal defence and gut microbiota. Nat. Microbiol. 2016, 1, 16125. [Google Scholar] [CrossRef]
- Sasabe, J.; Suzuki, M. Distinctive Roles of d-Amino Acids in the Homochiral World: Chirality of Amino Acids Modulates Mammalian Physiology and Pathology. Keio J. Med. 2018. [Google Scholar] [CrossRef]
- Fuchs, S.A.; Berger, R.; Klomp, L.W.; de Koning, T.J. d-amino acids in the central nervous system in health and disease. Mol. Genet. Metab. 2005, 85, 168–180. [Google Scholar] [CrossRef]
- Paul, P.; de Belleroche, J. The role of d-serine and glycine as co-agonists of NMDA receptors in motor neuron degeneration and amyotrophic lateral sclerosis (ALS). Front. Synaptic Neurosci. 2014, 6, 10. [Google Scholar] [CrossRef]
- Shleper, M.; Kartvelishvily, E.; Wolosker, H. d-serine is the dominant endogenous coagonist for NMDA receptor neurotoxicity in organotypic hippocampal slices. J. Neurosci. 2005, 25, 9413–9417. [Google Scholar] [CrossRef]
- Guercio, G.D.; Panizzutti, R. Potential and Challenges for the Clinical Use of d-Serine As a Cognitive Enhancer. Front. Psychiatry 2018, 9, 14. [Google Scholar] [CrossRef]
- Klatte, K.; Kirschstein, T.; Otte, D.; Pothmann, L.; Muller, L.; Tokay, T.; Kober, M.; Uebachs, M.; Zimmer, A.; Beck, H. Impaired d-serine-mediated cotransmission mediates cognitive dysfunction in epilepsy. J. Neurosci. 2013, 33, 13066–13080. [Google Scholar] [CrossRef]
- Errico, F.; Napolitano, F.; Nistico, R.; Usiello, A. New insights on the role of free d-aspartate in the mammalian brain. Amino Acids 2012, 43, 1861–1871. [Google Scholar] [CrossRef]
- D’Aniello, A. d-Aspartic acid: An endogenous amino acid with an important neuroendocrine role. Brain Res. Rev. 2007, 53, 215–234. [Google Scholar] [CrossRef]
- Roshanzamir, F.; Safavi, S.M. The putative effects of d-Aspartic acid on blood testosterone levels: A systematic review. Int. J. Reprod. Biomed. 2017, 15, 1–10. [Google Scholar] [CrossRef]
- Kimura, T.; Hamase, K.; Miyoshi, Y.; Yamamoto, R.; Yasuda, K.; Mita, M.; Rakugi, H.; Hayashi, T.; Isaka, Y. Chiral amino acid metabolomics for novel biomarker screening in the prognosis of chronic kidney disease. Sci. Rep. 2016, 6, 26137. [Google Scholar] [CrossRef]
- Sasabe, J.; Suzuki, M. Emerging Role of d-Amino Acid Metabolism in the Innate Defense. Front. Microbiol. 2018, 9, 933. [Google Scholar] [CrossRef]
- Cava, F.; Lam, H.; de Pedro, M.A.; Waldor, M.K. Emerging knowledge of regulatory roles of d-amino acids in bacteria. CMLS 2011, 68, 817–831. [Google Scholar] [CrossRef]
- Lam, H.; Oh, D.C.; Cava, F.; Takacs, C.N.; Clardy, J.; de Pedro, M.A.; Waldor, M.K. d-amino acids govern stationary phase cell wall remodeling in bacteria. Science 2009, 325, 1552–1555. [Google Scholar] [CrossRef]
- Typas, A.; Banzhaf, M.; Gross, C.A.; Vollmer, W. From the regulation of peptidoglycan synthesis to bacterial growth and morphology. Nat. Rev. Microbiol. 2011, 10, 123–136. [Google Scholar] [CrossRef]
- Leiman, S.A.; May, J.M.; Lebar, M.D.; Kahne, D.; Kolter, R.; Losick, R. d-Amino Acids Indirectly Inhibit Biofilm Formation in Bacillus subtilis by Interfering with Protein Synthesis. J. Bacteriol. 2013, 195, 5391–5395. [Google Scholar] [CrossRef]
- Zilm, P.S.; Butnejski, V.; Rossi-Fedele, G.; Kidd, S.P.; Edwards, S.; Vasilev, K. d-amino acids reduce Enterococcus faecalis biofilms in vitro and in the presence of antimicrobials used for root canal treatment. PLoS ONE 2017, 12, e0170670. [Google Scholar] [CrossRef]
- Bucher, T.; Oppenheimer-Shaanan, Y.; Savidor, A.; Bloom-Ackermann, Z.; Kolodkin-Gal, I. Disturbance of the bacterial cell wall specifically interferes with biofilm formation. Environ. Microbiol. Rep. 2015, 7, 990–1004. [Google Scholar] [CrossRef]
- Hernandez, S.B.; Cava, F. Environmental roles of microbial amino acid racemases. Environ. Microbiol. 2016, 18, 1673–1685. [Google Scholar] [CrossRef]
- Radkov, A.D.; Moe, L.A. Bacterial synthesis of d-amino acids. Appl. Microbiol. Biotechnol. 2014, 98, 5363–5374. [Google Scholar] [CrossRef]
- Espaillat, A.; Carrasco-Lopez, C.; Bernardo-Garcia, N.; Pietrosemoli, N.; Otero, L.H.; Alvarez, L.; de Pedro, M.A.; Pazos, F.; Davis, B.M.; Waldor, M.K.; et al. Structural basis for the broad specificity of a new family of amino-acid racemases. Acta Crystallogr. Sect. D Biol. Crystallogr. 2014, 70, 79–90. [Google Scholar] [CrossRef]
- Genchi, G. An overview on D-amino acids. Amino Acids 2017, 49, 1521–1533. [Google Scholar] [CrossRef]
- Mutaguchi, Y.; Kobayashi, J.; Oikawa, T.; Ohshima, T. d-Amino Acids in Fermentative Foods. In d-Amino Acids: Physiology, Metabolism, and Application; Yoshimura, T., Nishikawa, T., Homma, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Gobbetti, M.; Simonetti, M.S.; Rossi, J.; Cossignani, L.; Corsetti, A.; Damiani, P. Free d- and l-Amino Acid Evolution During Sourdough Fermentation and Baking. J. Food Sci. 1994, 59, 881–884. [Google Scholar] [CrossRef]
- Bruckner, H.; Westhauser, T. Chromatographic determination of l- and d-amino acids in plants. Amino Acids 2003, 24, 43–55. [Google Scholar] [CrossRef]
- Nasreddine, L.; Tamim, H.; Itani, L.; Nasrallah, M.P.; Isma’eel, H.; Nakhoul, N.F.; Abou-Rizk, J.; Naja, F. A minimally processed dietary pattern is associated with lower odds of metabolic syndrome among Lebanese adults. Public Health Nutr. 2018, 21, 160–171. [Google Scholar] [CrossRef]
- Lin, T.K.; Teymourian, Y.; Tursini, M.S. The effect of sugar and processed food imports on the prevalence of overweight and obesity in 172 countries. Glob. Health 2018, 14, 35. [Google Scholar] [CrossRef]
- Friedman, M.; Masters, P.M. Kinetics of Racemization of Amino Acid Residues in Casein. J. Food Sci. 1982, 47, 760–764. [Google Scholar] [CrossRef]
- Hayase, F.; Kato, H.; Fujimaki, M. Racemization of Amino Acid Residues in Proteins during Roasting. Agric. Biol. Chem. 1975, 23, 491–494. [Google Scholar] [CrossRef]
- Jenkins, W.L.; Raul Tovar, L.; Schwass, D.E.; Liardon, R.; Carpenter, K.J. Nutritional characteristics of alkali-treated zein. J. Agric. Food Chem. 1984, 32, 1035–1041. [Google Scholar] [CrossRef]
- Csapo, J.; Varga-Visi, E.; Loki, K.; Albert, C.; Salamon, S. The influence of extrusion on loss and racemization of amino acids. Amino Acids 2008, 34, 287–292. [Google Scholar] [CrossRef]
- Friedman, M.; Gumbmann, M.R.; Masters, P.M. Protein-alkali reactions: Chemistry, toxicology, and nutritional consequences. Adv. Exp. Med. Biol. 1984, 177, 367–412. [Google Scholar]
- Schwass, D.E.; Finley, J.W. Heat and alkaline damage to proteins: Racemization and lysinoalanine formation. J. Agric. Food Chem. 1984, 32, 1377–1382. [Google Scholar] [CrossRef]
- Friedman, M.; Liardon, R. Racemization kinetics of amino acid residues in alkali-treated soybean protein. J. Agric. Food Chem. 1985, 33, 666–672. [Google Scholar] [CrossRef]
- Kawai, M.; Sekine-Hayakawa, Y.; Okiyama, A.; Ninomiya, Y. Gustatory sensation of (l)- and (d)-amino acids in humans. Amino Acids 2012, 43, 2349–2358. [Google Scholar] [CrossRef]
- De Vrese, M.; Frik, R.; Roos, N.; Hagemeister, H. Protein-bound d-amino acids, and to a lesser extent lysinoalanine, decrease true ileal protein digestibility in minipigs as determined with (15)N-labeling. J. Nutr. 2000, 130, 2026–2031. [Google Scholar] [CrossRef]
- Balu, D.T.; Takagi, S.; Puhl, M.D.; Benneyworth, M.A.; Coyle, J.T. d-serine and serine racemase are localized to neurons in the adult mouse and human forebrain. Cell. Mol. Neurobiol. 2014, 34, 419–435. [Google Scholar] [CrossRef]
- Suzuki, M.; Sasabe, J.; Miyoshi, Y.; Kuwasako, K.; Muto, Y.; Hamase, K.; Matsuoka, M.; Imanishi, N.; Aiso, S. Glycolytic flux controls d-serine synthesis through glyceraldehyde-3-phosphate dehydrogenase in astrocytes. Proc. Natl. Acad. Sci. USA 2015, 112, E2217–E2224. [Google Scholar] [CrossRef]
- De Miranda, J.; Panizzutti, R.; Foltyn, V.N.; Wolosker, H. Cofactors of serine racemase that physiologically stimulate the synthesis of the n-methyl-d-aspartate (NMDA) receptor coagonist d-serine. Proc. Natl. Acad. Sci. USA 2002, 99, 14542–14547. [Google Scholar] [CrossRef]
- Wolosker, H.; Sheth, K.N.; Takahashi, M.; Mothet, J.P.; Brady, R.O., Jr.; Ferris, C.D.; Snyder, S.H. Purification of serine racemase: Biosynthesis of the neuromodulator d-serine. Proc. Natl. Acad. Sci. USA 1999, 96, 721–725. [Google Scholar] [CrossRef]
- Ito, T.; Hayashida, M.; Kobayashi, S.; Muto, N.; Hayashi, A.; Yoshimura, T.; Mori, H. Serine racemase is involved in d-aspartate biosynthesis. J. Biochem. 2016, 160, 345–353. [Google Scholar] [CrossRef]
- Graham, D.L.; Beio, M.L.; Nelson, D.L.; Berkowitz, D.B. Human Serine Racemase: Key Residues/Active Site Motifs and Their Relation to Enzyme Function. Front. Mol. Biosci. 2019, 6, 8. [Google Scholar] [CrossRef]
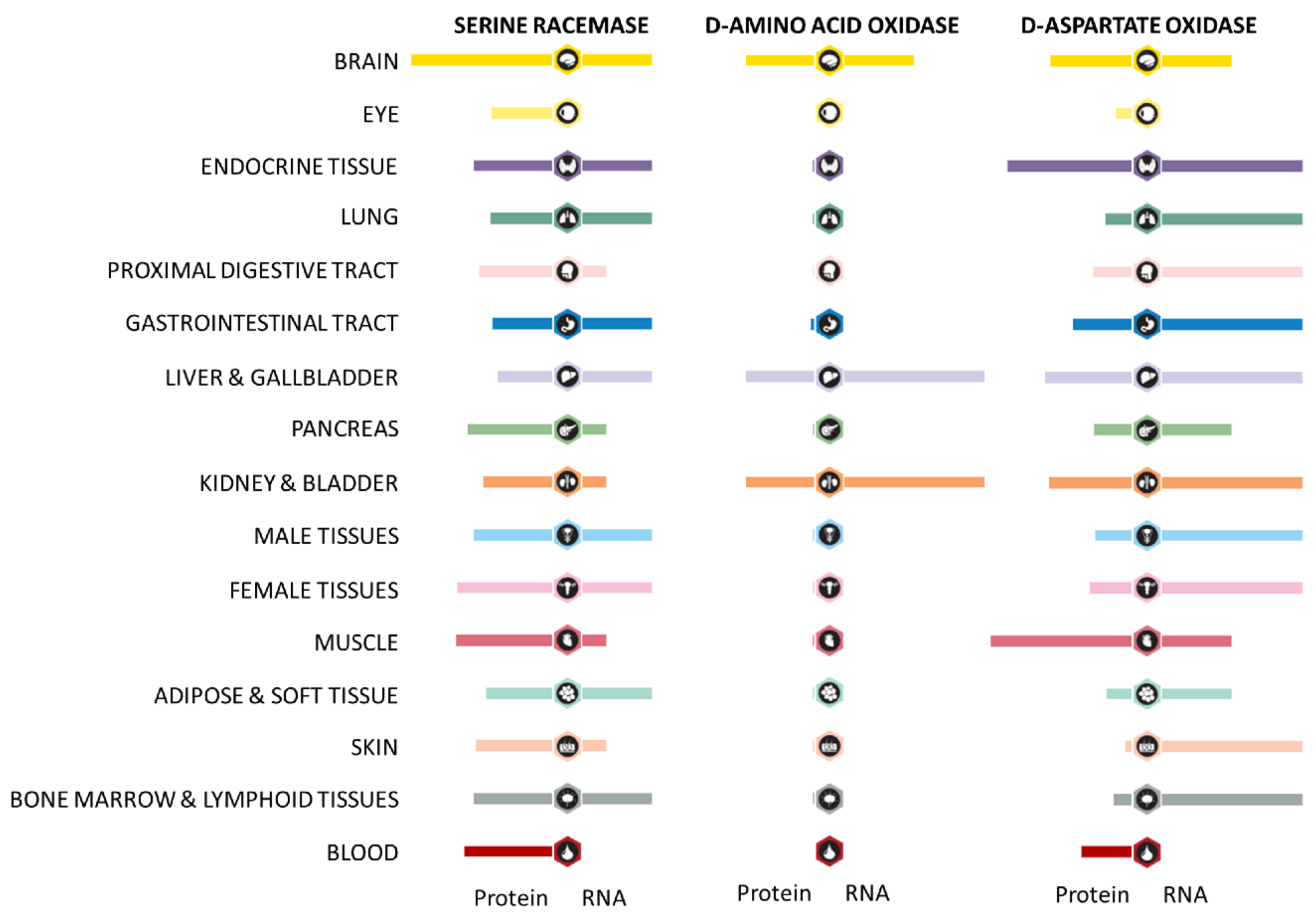
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; et al. Tissue-based map of the human proteome. Science 2015, 347. [Google Scholar] [CrossRef] [PubMed]
- Krebs, H.A. Metabolism of amino-acids: Deamination of amino-acids. Biochem. J. 1935, 29, 1620–1644. [Google Scholar] [CrossRef] [PubMed]
- Sacchi, S.; Cappelletti, P.; Murtas, G. Biochemical Properties of Human d-amino Acid Oxidase Variants and Their Potential Significance in Pathologies. Front. Mol. Biosci. 2018, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Murtas, G.; Sacchi, S.; Valentino, M.; Pollegioni, L. Biochemical Properties of Human d-Amino Acid Oxidase. Front. Mol. Biosci. 2017, 4, 88. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Abe, K.; Kera, Y. Bacterial d-amino acid oxidases: Recent findings and future perspectives. Bioengineered 2015, 6, 237–241. [Google Scholar] [CrossRef]
- Koga, R.; Miyoshi, Y.; Sakaue, H.; Hamase, K.; Konno, R. Mouse d-Amino-Acid Oxidase: Distribution and Physiological Substrates. Front. Mol. Biosci. 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.; Nardacci, R.; Cimini, A.; Cer, M.P. Immunocytochemical localization of d-amino acid oxidase in rat brain. J. Neurocytol. 1999, 28, 169–185. [Google Scholar] [CrossRef]
- Usuda, N.; Yokota, S.; Hashimoto, T.; Nagata, T. Immunocytochemical localization of d-amino acid oxidase in the central clear matrix of rat kidney peroxisomes. J. Histochem. Cytochem. 1986, 34, 1709–1718. [Google Scholar] [CrossRef]
- Perotti, M.E.; Gavazzi, E.; Trussardo, L.; Malgaretti, N.; Curti, B. Immunoelectron microscopic localization of d-amino acid oxidase in rat kidney and liver. Histochem. J. 1987, 19, 157–169. [Google Scholar] [CrossRef]
- Horiike, K.; Tojo, H.; Arai, R.; Yamano, T.; Nozaki, M.; Maeda, T. Localization of d-amino acid oxidase in Bergmann glial cells and astrocytes of rat cerebellum. Brain Res. Bull. 1987, 19, 587–596. [Google Scholar] [CrossRef]
- Luks, L.; Sacchi, S.; Pollegioni, L.; Dietrich, D.R. Novel insights into renal d-amino acid oxidase accumulation: Propiverine changes DAAO localization and peroxisomal size in vivo. Arch. Toxicol. 2017, 91, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, P.; Campomenosi, P.; Pollegioni, L.; Sacchi, S. The degradation (by distinct pathways) of human d-amino acid oxidase and its interacting partner pLG72--two key proteins in d-serine catabolism in the brain. FEBS J. 2014, 281, 708–723. [Google Scholar] [CrossRef]
- Hashimoto, A.; Oka, T.; Nishikawa, T. Anatomical distribution and postnatal changes in endogenous free d-aspartate and d-serine in rat brain and periphery. Eur. J. Neurosci. 1995, 7, 1657–1663. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, A.; Nishikawa, T.; Oka, T.; Takahashi, K. Endogenous D-serine in rat brain: n-methyl-d-aspartate receptor-related distribution and aging. J. Neurochem. 1993, 60, 783–786. [Google Scholar] [CrossRef]
- Hamase, K.; Homma, H.; Takigawa, Y.; Fukushima, T.; Santa, T.; Imai, K. Regional distribution and postnatal changes of d-amino acids in rat brain. Biochim. Biophys. Acta 1997, 1334, 214–222. [Google Scholar] [CrossRef]
- D’Ascenzo, M.; Podda, M.V.; Grassi, C. The role of d-serine as co-agonist of NMDA receptors in the nucleus accumbens: Relevance to cocaine addiction. Front. Synaptic Neurosci. 2014, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Imanishi, N.; Mita, M.; Hamase, K.; Aiso, S.; Sasabe, J. Heterogeneity of d-Serine Distribution in the Human Central Nervous System. ASN Neuro 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, Y.; Konno, R.; Sasabe, J.; Ueno, K.; Tojo, Y.; Mita, M.; Aiso, S.; Hamase, K. Alteration of intrinsic amounts of d-serine in the mice lacking serine racemase and D-amino acid oxidase. Amino Acids 2012, 43, 1919–1931. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.C.; Tsai, G.E.; Ma, C.L.; Ehmsen, J.T.; Mustafa, A.K.; Han, L.; Jiang, Z.I.; Benneyworth, M.A.; Froimowitz, M.P.; Lange, N.; et al. Targeted disruption of serine racemase affects glutamatergic neurotransmission and behavior. Mol. Psychiatry 2009, 14, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Coyle, J.T.; Balu, D.T. The Role of Serine Racemase in the Pathophysiology of Brain Disorders. Adv. Pharm. 2018, 82, 35–56. [Google Scholar] [CrossRef]
- Yang, Y.; Ge, W.; Chen, Y.; Zhang, Z.; Shen, W.; Wu, C.; Poo, M.; Duan, S. Contribution of astrocytes to hippocampal long-term potentiation through release of d-Serine. Proc. Natl. Acad. Sci. USA 2003, 100, 15194. [Google Scholar] [CrossRef] [PubMed]
- Bado, P.; Madeira, C.; Vargas-Lopes, C.; Moulin, T.C.; Wasilewska-Sampaio, A.P.; Maretti, L.; de Oliveira, R.V.; Amaral, O.B.; Panizzutti, R. Effects of low-dose d-serine on recognition and working memory in mice. Psychopharmacology 2011, 218, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Shimazaki, T.; Kaku, A.; Chaki, S. d-Serine and a glycine transporter-1 inhibitor enhance social memory in rats. Psychopharmacology 2010, 209, 263–270. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, S.; Somorjai, I.; Garcia-Fernandez, J.; Topo, E.; D’Aniello, A. d-Aspartic acid is a novel endogenous neurotransmitter. FASEB J. 2011, 25, 1014–1027. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, A.; Di Cosmo, A.; Di Cristo, C.; Annunziato, L.; Petrucelli, L.; Fisher, G. Involvement of d-aspartic acid in the synthesis of testosterone in rat testes. Life Sci. 1996, 59, 97–104. [Google Scholar] [CrossRef]
- D’Aniello, G.; Ronsini, S.; Guida, F.; Spinelli, P.; D’Aniello, A. Occurrence of d-aspartic acid in human seminal plasma and spermatozoa: Possible role in reproduction. Fertil. Steril. 2005, 84, 1444–1449. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, A.; Kumashiro, S.; Nishikawa, T.; Oka, T.; Takahashi, K.; Mito, T.; Takashima, S.; Doi, N.; Mizutani, Y.; Yamazaki, T.; et al. Embryonic development and postnatal changes in free d-aspartate and d-serine in the human prefrontal cortex. J. Neurochem. 1993, 61, 348–351. [Google Scholar] [CrossRef] [PubMed]
- Ota, N.; Shi, T.; Sweedler, J.V. d-Aspartate acts as a signaling molecule in nervous and neuroendocrine systems. Amino Acids 2012, 43, 1873–1886. [Google Scholar] [CrossRef]
- Kim, P.M.; Duan, X.; Huang, A.S.; Liu, C.Y.; Ming, G.L.; Song, H.; Snyder, S.H. Aspartate racemase, generating neuronal d-aspartate, regulates adult neurogenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 3175–3179. [Google Scholar] [CrossRef]
- Wang, L.; Ota, N.; Romanova, E.V.; Sweedler, J.V. A novel pyridoxal 5′-phosphate-dependent amino acid racemase in the Aplysia californica central nervous system. J. Biol. Chem. 2011, 286, 13765–13774. [Google Scholar] [CrossRef]
- Katane, M.; Kawata, T.; Nakayama, K.; Saitoh, Y.; Kaneko, Y.; Matsuda, S.; Saitoh, Y.; Miyamoto, T.; Sekine, M.; Homma, H. Characterization of the enzymatic and structural properties of human d-aspartate oxidase and comparison with those of the rat and mouse enzymes. Biol. Pharm. Bull. 2015, 38, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Katane, M.; Homma, H. d-Aspartate—An important bioactive substance in mammals: A review from an analytical and biological point of view. J. Chromatogr. B 2011, 879, 3108–3121. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, A.; Di Fiore, M.M.; Fisher, G.H.; Milone, A.; Seleni, A.; D’Aniello, S.; Perna, A.F.; Ingrosso, D. Occurrence of d-aspartic acid and n-methyl-d-aspartic acid in rat neuroendocrine tissues and their role in the modulation of luteinizing hormone and growth hormone release. FASEB J. 2000, 14, 699–714. [Google Scholar] [CrossRef]
- Topo, E.; Soricelli, A.; D’Aniello, A.; Ronsini, S.; D’Aniello, G. The role and molecular mechanism of d-aspartic acid in the release and synthesis of LH and testosterone in humans and rats. Reprod. Biol. Endocrinol. 2009, 7, 120. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Takata, T.; Fujii, N.; Aki, K.; Sakaue, H. d-Amino acids in protein: The mirror of life as a molecular index of aging. BBA Proteins Proteom. 2018, 1866, 840–847. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, S.; Matsushima, Y.; Kobayashi, Y.; Yamamoto, T. Age estimation by measuring the racemization of aspartic acid from total amino acid content of several types of bone and rib cartilage: A preliminary account. J. Forensic Sci. 2002, 47, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Aki, K.; Kuge, K.; Tajima, S.; Yamanaka, N.; Kaji, Y.; Yamamoto, N.; Nagai, R.; Yoshii, H.; Fujii, N.; et al. UV B-irradiation enhances the racemization and isomerizaiton of aspartyl residues and production of Nepsilon-carboxymethyl lysine (CML) in keratin of skin. J. Chromatogr. B 2011, 879, 3303–3309. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Ishibashi, Y.; Satoh, K.; Fujino, M.; Harada, K. Simultaneous racemization and isomerization at specific aspartic acid residues in alpha b-crystallin from the aged human lens. BBA 1994, 1204, 157–163. [Google Scholar] [CrossRef]
- Fujii, N.; Sakaue, H.; Sasaki, H.; Fujii, N. A rapid, comprehensive liquid chromatography-mass spectrometry (LC-MS)-based survey of the Asp isomers in crystallins from human cataract lenses. J. Biol. Chem. 2012, 287, 39992–40002. [Google Scholar] [CrossRef]
- Fujii, N.; Tajima, S.; Tanaka, N.; Fujimoto, N.; Takata, T.; Shimo-Oka, T. The presence of d-beta-aspartic acid-containing peptides in elastic fibers of sun-damaged skin: A potent marker for ultraviolet-induced skin aging. Biochem. Biophys. Res. Commun. 2002, 294, 1047–1051. [Google Scholar] [CrossRef]
- Robinson, J.M.; Briggs, R.T.; Karnovsky, M.J. Localization of d-amino acid oxidase on the cell surface of human polymorphonuclear leukocytes. J. Cell Biol. 1978, 77, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, M.R.; Baehner, R.L.; Nathan, D.G. Amino acid oxidase of leukocytes in relation to H2O2-mediated bacterial killing. J. Clin. Investig. 1971, 50, 1985–1991. [Google Scholar] [CrossRef] [PubMed][Green Version]
- DeChatelet, L.R.; McCall, C.E.; Cooper, M.R. Amino acid oxidase in leukocytes: Evidence against a major role in phagocytosis. Infect. Immun. 1972, 5, 632–633. [Google Scholar] [PubMed]
- Nakamura, H.; Fang, J.; Maeda, H. Protective role of d-amino acid oxidase against Staphylococcus aureus infection. Infect. Immun. 2012, 80, 1546–1553. [Google Scholar] [CrossRef] [PubMed]
- Tuinema, B.R.; Reid-Yu, S.A.; Coombes, B.K. Salmonella Evades Amino Acid Oxidase To Promote Infection in Neutrophils. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Brachet, P.; Puigserver, A. Regional differences for the d-amino acid oxidase-catalysed oxidation of d-methionine in chicken small intestine. Comp. Biochem. Physiol. B 1992, 101, 509–511. [Google Scholar] [CrossRef]
- Canfora, E.E.; Meex, R.C.R.; Venema, K.; Blaak, E.E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 2019, 15, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Verrall, L.; Burnet, P.W.; Betts, J.F.; Harrison, P.J. The neurobiology of d-amino acid oxidase and its involvement in schizophrenia. Mol. Psychiatry 2010, 15, 122–137. [Google Scholar] [CrossRef]
- Yang, H.-C.; Liu, C.-M.; Liu, Y.-L.; Chen, C.-W.; Chang, C.C.; Fann, C.S.J.; Chiou, J.-J.; Yang, U.-C.; Chen, C.-H.; Faraone, S.V.; et al. The DAO Gene Is Associated with Schizophrenia and Interacts with Other Genes in the Taiwan Han Chinese Population. PLoS ONE 2013, 8, e60099. [Google Scholar] [CrossRef]
- Sacchetti, E.; Scassellati, C.; Minelli, A.; Valsecchi, P.; Bonvicini, C.; Pasqualetti, P.; Galluzzo, A.; Pioli, R.; Gennarelli, M. Schizophrenia susceptibility and NMDA-receptor mediated signalling: An association study involving 32 tagSNPs of DAO, DAOA, PPP3CC, and DTNBP1 genes. BMC Med Genet. 2013, 14, 33. [Google Scholar] [CrossRef]
- Liu, Y.L.; Wang, S.C.; Hwu, H.G.; Fann, C.S.; Yang, U.C.; Yang, W.C.; Hsu, P.C.; Chang, C.C.; Wen, C.C.; Tsai-Wu, J.J.; et al. Haplotypes of the d-Amino Acid Oxidase Gene Are Significantly Associated with Schizophrenia and Its Neurocognitive Deficits. PLoS ONE 2016, 11, e0150435. [Google Scholar] [CrossRef] [PubMed]
- Burnet, P.W.J.; Eastwood, S.L.; Bristow, G.C.; Godlewska, B.R.; Sikka, P.; Walker, M.; Harrison, P.J. d-amino acid oxidase activity and expression are increased in schizophrenia. Mol. Psychiatry 2008, 13, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Madeira, C.; Freitas, M.E.; Vargas-Lopes, C.; Wolosker, H.; Panizzutti, R. Increased brain d-amino acid oxidase (DAAO) activity in schizophrenia. Schizophr. Res. 2008, 101, 76–83. [Google Scholar] [CrossRef]
- MacKay, M.-A.B.; Kravtsenyuk, M.; Thomas, R.; Mitchell, N.D.; Dursun, S.M.; Baker, G.B. d-Serine: Potential Therapeutic Agent and/or Biomarker in Schizophrenia and Depression? Front. Psychiatry 2019, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Labrie, V.; Wong, A.H.; Roder, J.C. Contributions of the d-serine pathway to schizophrenia. Neuropharmacology 2012, 62, 1484–1503. [Google Scholar] [CrossRef] [PubMed]
- Tsai, G.E.; Lin, P.Y. Strategies to enhance n-methyl-d-aspartate receptor-mediated neurotransmission in schizophrenia, a critical review and meta-analysis. Curr. Pharm. Des. 2010, 16, 522–537. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.H.; Wykes, T.; Kurtz, M.M. Adjunctive pharmacotherapy for cognitive deficits in schizophrenia: Meta-analytical investigation of efficacy. Br. J. Psychiatry 2013, 203, 172–178. [Google Scholar] [CrossRef]
- Singh, S.P.; Singh, V. Meta-analysis of the efficacy of adjunctive NMDA receptor modulators in chronic schizophrenia. CNS Drugs 2011, 25, 859–885. [Google Scholar] [CrossRef]
- Errico, F.; Nuzzo, T.; Carella, M.; Bertolino, A.; Usiello, A. The Emerging Role of Altered d-Aspartate Metabolism in Schizophrenia: New Insights From Preclinical Models and Human Studies. Front. Psychiatry 2018, 9. [Google Scholar] [CrossRef]
- Nuzzo, T.; Sacchi, S.; Errico, F.; Keller, S.; Palumbo, O.; Florio, E.; Punzo, D.; Napolitano, F.; Copetti, M.; Carella, M.; et al. Decreased free d-aspartate levels are linked to enhanced d-aspartate oxidase activity in the dorsolateral prefrontal cortex of schizophrenia patients. NPJ Schizophr. 2017, 3, 16. [Google Scholar] [CrossRef]
- Errico, F.; Napolitano, F.; Squillace, M.; Vitucci, D.; Blasi, G.; de Bartolomeis, A.; Bertolino, A.; D’Aniello, A.; Usiello, A. Decreased levels of d-aspartate and NMDA in the prefrontal cortex and striatum of patients with schizophrenia. J. Psychiatr. Res. 2013, 47, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Curcio, L.; Podda, M.V.; Leone, L.; Piacentini, R.; Mastrodonato, A.; Cappelletti, P.; Sacchi, S.; Pollegioni, L.; Grassi, C.; D’Ascenzo, M. Reduced d-serine levels in the nucleus accumbens of cocaine-treated rats hinder the induction of NMDA receptor-dependent synaptic plasticity. Brain A J. Neurol. 2013, 136, 1216–1230. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.Q.; Gu, X.H.; Yang, Y.J.; Yin, X.P.; Xu, L.J.; Wang, W. d-Serine in the nucleus accumbens region modulates behavioral sensitization and extinction of conditioned place preference. Pharmacol. Biochem. Behav. 2016, 143, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Harai, T.; Inoue, R.; Fujita, Y.; Tanaka, A.; Horio, M.; Hashimoto, K.; Hongou, K.; Miyawaki, T.; Mori, H. Decreased susceptibility to seizures induced by pentylenetetrazole in serine racemase knockout mice. Epilepsy Res. 2012, 102, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Wu, Y.; Chen, B.; Zhang, W.; Jin, L.; Shen, C.; Wang, Y.; Liu, Y. d-Serine Contributes to Seizure Development via ERK Signaling. Front. Neurosci. 2019, 13, 254. [Google Scholar] [CrossRef] [PubMed]
- Sasabe, J.; Miyoshi, Y.; Suzuki, M.; Mita, M.; Konno, R.; Matsuoka, M.; Hamase, K.; Aiso, S. d-amino acid oxidase controls motoneuron degeneration through d-serine. Proc. Natl. Acad. Sci. USA 2012, 109, 627–632. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Lobner, D.; Banack, S.A.; Cox, G.A.; Nunn, P.B.; Wyatt, P.B.; Cox, P.A. Analysis of BMAA enantiomers in cycads, cyanobacteria, and mammals: In vivo formation and toxicity of d-BMAA. Amino Acids 2017, 49, 1427–1439. [Google Scholar] [CrossRef] [PubMed]
- Madeira, C.; Lourenco, M.V.; Vargas-Lopes, C.; Suemoto, C.K.; Brandão, C.O.; Reis, T.; Leite, R.E.P.; Laks, J.; Jacob-Filho, W.; Pasqualucci, C.A.; et al. d-serine levels in Alzheimer’s disease: Implications for novel biomarker development. Transl. Psychiatry 2015, 5, e561. [Google Scholar] [CrossRef] [PubMed]
- Kögl, F.; Erxleben, H. Zur ätiologie der malignen tumoren. Zeitschrift für Physiologische Chemie 1939, 258, 57–95. [Google Scholar] [CrossRef]
- Miller, J.A. Do tumor proteins contain d-amino acids? A review of the controversy. Cancer Res. 1950, 10, 65–72. [Google Scholar]
- Stroud, E.D.; Smith, G.G. A search for d-amino acids in tumor tissue. Biochem. Med. 1984, 31, 254–256. [Google Scholar] [CrossRef]
- Fisher, G.H.; Torres, D.; Bruna, J.; Cerwinski, S.; Martin, T.; Bergljung, C.; Gruneiro, A.; Chou, S.J.; Man, E.H.; Pappatheodorou, S. Presence of d-aspartate and d-glutamate in tumor proteins. Cancer Biochem. Biophys. 1995, 15, 79–82. [Google Scholar] [PubMed]
- Nagata, Y.; Sato, T.; Enomoto, N.; Ishii, Y.; Sasaki, K.; Yamada, T. High concentrations of d-amino acids in human gastric juice. Amino Acids 2007, 32, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, Y.; Liu, P.; Yang, L.; Jiang, X.; Luo, D.; Yang, D. Non-invasive detection of gastric cancer relevant d-amino acids with luminescent DNA/silver nanoclusters. Nanoscale 2017, 9, 19367–19373. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Xie, M.; Han, J.; Yuan, D.; Yang, T.; Xie, Y. Development and validation of a rapid, selective, and sensitive LC-MS/MS method for simultaneous determination of d- and l-amino acids in human serum: Application to the study of hepatocellular carcinoma. Anal. Bioanal. Chem. 2018, 410, 2517–2531. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhong, J.; Wang, S.; Zhou, Y.; Wang, L.; Zhang, Y.; Yuan, Y. Serum metabolomics differentiating pancreatic cancer from new-onset diabetes. Oncotarget 2017, 8, 29116–29124. [Google Scholar] [CrossRef] [PubMed]
- Tamemasa, O.; Goto, R.; Suzuki, T. Preferential incorporation of some 14C-labeled d-amino acids into tumor-bearing animals. Gan 1978, 69, 517–523. [Google Scholar]
- Stegman, L.D.; Zheng, H.; Neal, E.R.; Ben-Yoseph, O.; Pollegioni, L.; Pilone, M.S.; Ross, B.D. Induction of cytotoxic oxidative stress by d-alanine in brain tumor cells expressing Rhodotorula gracilis d-amino acid oxidase: A cancer gene therapy strategy. Hum. Gene. Ther. 1998, 9, 185–193. [Google Scholar] [CrossRef]
- Sasamura, T.; Matsuda, A.; Kokuba, Y. Nutritional effects of a d-methionine-containing solution on AH109A hepatoma-bearing rats. Biosci. Biotechnol. Biochem. 1998, 62, 2418–2420. [Google Scholar] [CrossRef]
- Sasamura, T.; Matsuda, A.; Kokuba, Y. Tumor growth inhibition and nutritional effect of d-amino acid solution in AH109A hepatoma-bearing rats. J. Nutr. Sci. Vitaminol. 1998, 44, 79–87. [Google Scholar] [CrossRef]
- Sasamura, T.; Matsuda, A.; Kokuba, Y. Effects of d-methionine-containing solution on tumor cell growth in vitro. Arzneimittelforschung 1999, 49, 541–543. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.K.; Araujo, T.S.; Sousa, N.A.; Sousa, F.B.; Nogueira, K.M.; Nicolau, L.A.; Medeiros, J.V. Evidence that d-cysteine protects mice from gastric damage via hydrogen sulfide produced by d-amino acid oxidase. Nitric Oxide 2017, 64, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, N.; Kimura, H. Production of hydrogen sulfide from d-cysteine and its therapeutic potential. Front. Endocrinol. 2013, 4, 87. [Google Scholar] [CrossRef] [PubMed]
- Hellmich, M.R.; Szabo, C. Hydrogen Sulfide and Cancer. Handb. Exp. Pharmacol. 2015, 230, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Huang, D.; An, N.; Chen, D.; Zhao, D. A novel pathway for the production of H2 S by DAO in rat jejunum. Neurogastroenterol. Motil. 2016, 28, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Wang, Y.; Alatrash, N.; Weatherly, C.A.; Roy, D.; MacDonnell, F.M.; Armstrong, D.W. Altered profiles and metabolism of l- and d-amino acids in cultured human breast cancer cells vs. non-tumorigenic human breast epithelial cells. J. Pharm. Biomed. Anal. 2019, 164, 421–429. [Google Scholar] [CrossRef] [PubMed]
- North, W.G.; Gao, G.; Memoli, V.A.; Pang, R.H.; Lynch, L. Breast cancer expresses functional NMDA receptors. Breast Cancer Res. Treat. 2010, 122, 307–314. [Google Scholar] [CrossRef]
- Piossek, C.; Thierauch, K.H.; Schneider-Mergener, J.; Volkmer-Engert, R.; Bachmann, M.F.; Korff, T.; Augustin, H.G.; Germeroth, L. Potent inhibition of angiogenesis by d,l-peptides derived from vascular endothelial growth factor receptor 2. Thromb. Haemost. 2003, 90, 501–510. [Google Scholar] [CrossRef]
- Pallerla, S.; Naik, H.; Singh, S.; Gauthier, T.; Sable, R.; Jois, S.D. Design of cyclic and d-amino acids containing peptidomimetics for inhibition of protein-protein interactions of HER2-HER3. J. Pept. Sci. 2018, 24. [Google Scholar] [CrossRef]
- Wang, H.; Feng, Z.; Xu, B. d-amino acid-containing supramolecular nanofibers for potential cancer therapeutics. Adv. Drug Deliv. Rev. 2017, 110, 102–111. [Google Scholar] [CrossRef]
- Borrelli, A.; Tornesello, A.L.; Tornesello, M.L.; Buonaguro, F.M. Cell Penetrating Peptides as Molecular Carriers for Anti-Cancer Agents. Molecules 2018, 23, 295. [Google Scholar] [CrossRef]
- Jiang, T.; Olson, E.S.; Nguyen, Q.T.; Roy, M.; Jennings, P.A.; Tsien, R.Y. Tumor imaging by means of proteolytic activation of cell-penetrating peptides. Proc. Natl. Acad. Sci. USA 2004, 101, 17867–17872. [Google Scholar] [CrossRef]
- Rosini, E.; Pollegioni, L.; Ghisla, S.; Orru, R.; Molla, G. Optimization of d-amino acid oxidase for low substrate concentrations—Towards a cancer enzyme therapy. FEBS J. 2009, 276, 4921–4932. [Google Scholar] [CrossRef]
- El Sayed, S.M.; El-Magd, R.M.; Shishido, Y.; Yorita, K.; Chung, S.P.; Tran, D.H.; Sakai, T.; Watanabe, H.; Kagami, S.; Fukui, K. d-Amino acid oxidase-induced oxidative stress, 3-bromopyruvate and citrate inhibit angiogenesis, exhibiting potent anticancer effects. J. Bioenerg. Biomembr. 2012, 44, 513–523. [Google Scholar] [CrossRef]
- Bava, A.; Gornati, R.; Cappellini, F.; Caldinelli, L.; Pollegioni, L.; Bernardini, G. d-amino acid oxidase-nanoparticle system: A potential novel approach for cancer enzymatic therapy. Nanomedicine 2013, 8, 1797–1806. [Google Scholar] [CrossRef]
- Huang, J.L.; Chen, X.L.; Guo, C.; Wang, Y.X. Contributions of spinal d-amino acid oxidase to bone cancer pain. Amino Acids 2012, 43, 1905–1918. [Google Scholar] [CrossRef]
- Zhao, W.J.; Gao, Z.Y.; Wei, H.; Nie, H.Z.; Zhao, Q.; Zhou, X.J.; Wang, Y.X. Spinal d-amino acid oxidase contributes to neuropathic pain in rats. J. Pharmacol. Exp. Ther. 2010, 332, 248–254. [Google Scholar] [CrossRef]
- Zhao, W.; Konno, R.; Zhou, X.J.; Yin, M.; Wang, Y.X. Inhibition of d-amino-Acid oxidase activity induces pain relief in mice. Cell. Mol. Neurobiol. 2008, 28, 581–591. [Google Scholar] [CrossRef]
- Smith, S.M.; Uslaner, J.M.; Hutson, P.H. The Therapeutic Potential of d-Amino Acid Oxidase (DAAO) Inhibitors. Open Med. Chem. J. 2010, 4, 3–9. [Google Scholar] [CrossRef]
- Miyamoto, T.; Homma, H. Detection and quantification of d-amino acid residues in peptides and proteins using acid hydrolysis. Biochim. Biophys. Acta Proteins Proteom. 2018, 1866, 775–782. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bastings, J.J.A.J.; van Eijk, H.M.; Olde Damink, S.W.; Rensen, S.S. d-amino Acids in Health and Disease: A Focus on Cancer. Nutrients 2019, 11, 2205. https://doi.org/10.3390/nu11092205
Bastings JJAJ, van Eijk HM, Olde Damink SW, Rensen SS. d-amino Acids in Health and Disease: A Focus on Cancer. Nutrients. 2019; 11(9):2205. https://doi.org/10.3390/nu11092205
Chicago/Turabian StyleBastings, Jacco J.A.J., Hans M. van Eijk, Steven W. Olde Damink, and Sander S. Rensen. 2019. "d-amino Acids in Health and Disease: A Focus on Cancer" Nutrients 11, no. 9: 2205. https://doi.org/10.3390/nu11092205
APA StyleBastings, J. J. A. J., van Eijk, H. M., Olde Damink, S. W., & Rensen, S. S. (2019). d-amino Acids in Health and Disease: A Focus on Cancer. Nutrients, 11(9), 2205. https://doi.org/10.3390/nu11092205




