Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults
Abstract
1. Introduction
2. Methods
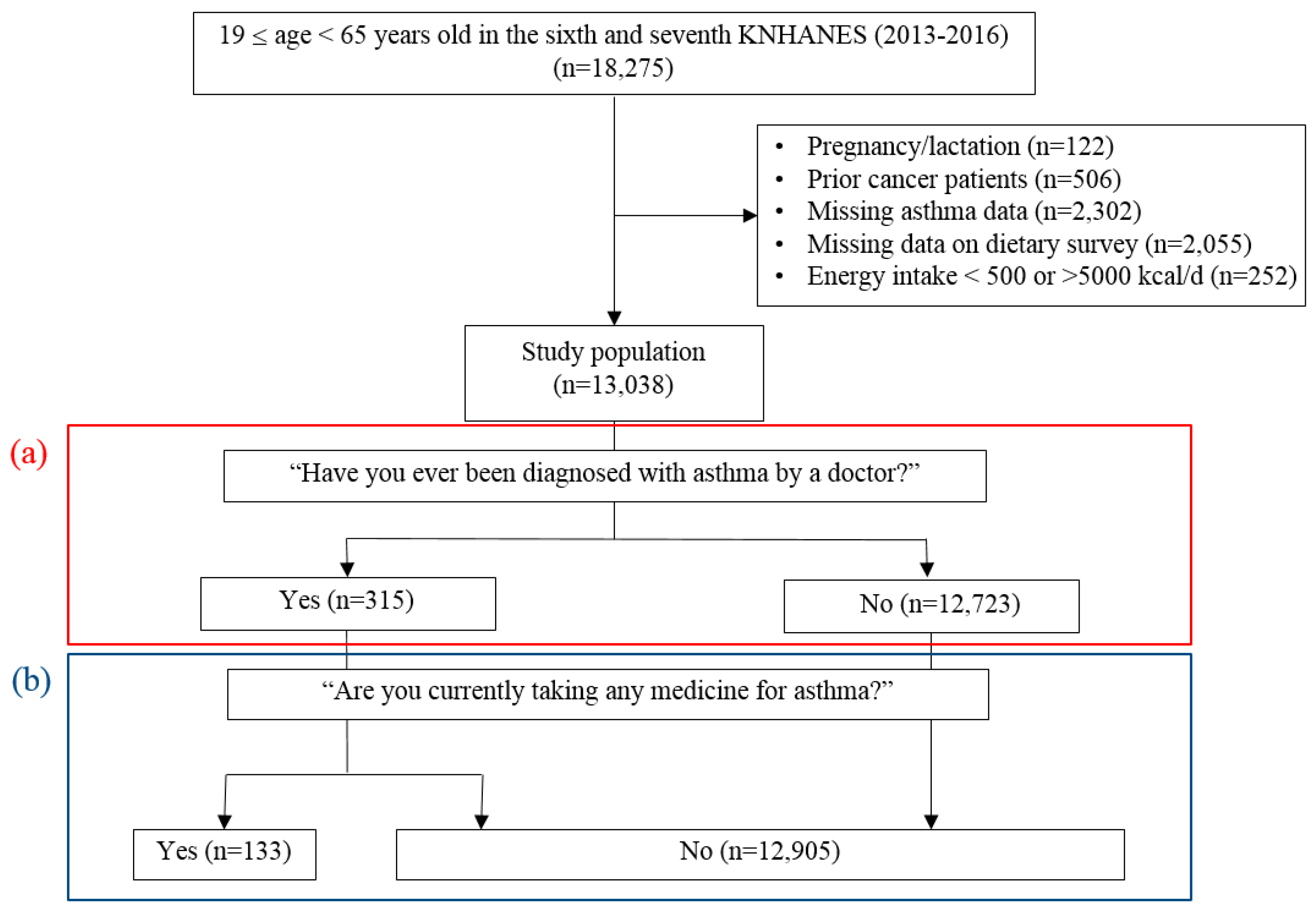
2.1. Study Population
2.2. Definition of Asthma
2.3. Dietary Assessment
2.4. Covariates
2.5. Statistical Analysis
3. Results
3.1. Characteristics Based on the Presence of Asthma
3.2. Daily Food and Nutrient Intake Based on the Presence of Asthma
3.3. Association between Dietary Intake and Asthma
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Backman, H.; Raisanen, P.; Hedman, L.; Stridsman, C.; Andersson, M.; Lindberg, A.; Lundback, B.; Ronmark, E. Increased prevalence of allergic asthma from 1996 to 2006 and further to 2016-results from three population surveys. Clin. Exp. Allergy 2017, 47, 1426–1435. [Google Scholar] [CrossRef] [PubMed]
- Loftus, P.A.; Wise, S.K. Epidemiology of asthma. Curr. Opin. Otolaryngol. Head Neck Surg. 2016, 24, 245–249. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Stanojevic, S.; Moores, G.; Gershon, A.S.; Bateman, E.D.; Cruz, A.A.; Boulet, L.P. Global asthma prevalence in adults: Findings from the cross-sectional world health survey. BMC Public Health 2012, 12, 204. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Jung, J.Y.; Park, M.S.; Kang, Y.A.; Kim, E.Y.; Kim, S.K.; Chang, J.; Kim, Y.S. Increased prevalence of self-reported asthma among Korean adults: An analysis of KNHANES I and IV data. Lung 2013, 191, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Barros, R.; Moreira, A.; Fonseca, J.; Ferraz de Oliveira, J.; Delgado, L.; Castel-Branco, M.; Haahtela, T.; Lopes, C.; Moreira, P. Adherence to the Mediterranean diet and fresh fruit intake are associated with improved asthma control. Allergy 2008, 63, 917–923. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.D.; Welch, A.A.; Bingham, S.A.; Luben, R.N.; Day, N.E.; Khaw, K.T.; Lomas, D.A.; Wareham, N.J. Dietary antioxidants and asthma in adults. Thorax 2006, 61, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Miyake, Y.; Sasaki, S.; Tanaka, K.; Ohya, Y.; Matsunaga, I.; Yoshida, T.; Oda, H.; Ishiko, O.; Hirota, Y. Fat and fish intake and asthma in Japanese women: Baseline data from the Osaka Maternal and Child Health Study. Int. J. Tuberc. Lung Dis. 2007, 11, 103–109. [Google Scholar]
- Miyata, J.; Arita, M. Role of omega-3 fatty acids and their metabolites in asthma and allergic diseases. Allergol. Int. 2015, 64, 27–34. [Google Scholar] [CrossRef]
- Li, J.; Xun, P.; Zamora, D.; Sood, A.; Liu, K.; Daviglus, M.; Iribarren, C.; Jacobs, D., Jr.; Shikany, J.M.; He, K. Intakes of long-chain omega-3 (n− 3) PUFAs and fish in relation to incidence of asthma among American young adults: The CARDIA study. Am. J. Clin. Nutr. 2012, 97, 173–178. [Google Scholar] [CrossRef]
- Nagel, G.; Linseisen, J. Dietary intake of fatty acids, antioxidants and selected food groups and asthma in adults. Eur. J. Clin. Nutr. 2005, 59, 8. [Google Scholar] [CrossRef]
- Shaheen, S.O.; Sterne, J.A.; Thompson, R.L.; Songhurst, C.E.; Margetts, B.M.; Burney, P.G. Dietary antioxidants and asthma in adults: Population-based case–control study. Am. J. Respir. Crit. Care Med. 2001, 164, 1823–1828. [Google Scholar] [CrossRef] [PubMed]
- Øien, T.; Storrø, O.; Johnsen, R. Do early intake of fish and fish oil protect against eczema and doctor-diagnosed asthma at 2 years of age? A cohort study. J. Epidemiol. Community Health 2010, 64, 124–129. [Google Scholar] [CrossRef] [PubMed]
- SanGiovanni, J.P.; Chew, E.Y. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog. Retin. Eye Res. 2005, 24, 87–138. [Google Scholar] [CrossRef] [PubMed]
- Lumia, M.; Luukkainen, P.; Tapanainen, H.; Kaila, M.; Erkkola, M.; Uusitalo, L.; Niinistö, S.; Kenward, M.G.; Ilonen, J.; Simell, O. Dietary fatty acid composition during pregnancy and the risk of asthma in the offspring. Pediatr. Allergy Immunol. 2011, 22, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Oh, S.Y.; Kang, M.H.; Kim, K.N.; Kim, Y.; Chang, N. Association between kimchi intake and asthma in Korean adults: The fourth and fifth Korea National Health and Nutrition Examination Survey (2007–2011). J. Med. Food 2014, 17, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Park, B.J.; Jung, D.H.; Kwon, Y.J. Association between Household Food Insecurity and Asthma in Korean Adults. Int. J. Environ. Res. Public Health 2019, 16, 2115. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Del Giacco, S.; Moreira, A.; Bonini, M.; Charles, D.; Reeves, T.; Carlsen, K.H.; Haahtela, T.; Bonini, S.; Fonseca, J. Asthma and dietary intake: An overview of systematic reviews. Allergy 2016, 71, 433–442. [Google Scholar] [CrossRef]
- Romieu, I.; Torrent, M.; Garcia-Esteban, R.; Ferrer, C.; Ribas-Fitó, N.; Anto, J.; Sunyer, J. Maternal fish intake during pregnancy and atopy and asthma in infancy. Clin. Exp. Allergy 2007, 37, 518–525. [Google Scholar] [CrossRef]
- Salam, M.T.; Li, Y.F.; Langholz, B.; Gilliland, F.D. Maternal fish consumption during pregnancy and risk of early childhood asthma. J. Asthma 2005, 42, 513–518. [Google Scholar] [CrossRef]
- Sausenthaler, S.; Koletzko, S.; Schaaf, B.; Lehmann, I.; Borte, M.; Herbarth, O.; von Berg, A.; Wichmann, H.E.; Heinrich, J.; LISA Study Group. Maternal diet during pregnancy in relation to eczema and allergic sensitization in the offspring at 2 y of age. Am. J. Clin. Nutr. 2007, 85, 530–537. [Google Scholar]
- Willers, S.; Devereux, G.; Craig, L.; McNeill, G.; Wijga, A.; El-Magd, W.A.; Turner, S.; Helms, P.; Seaton, A. Maternal food consumption during pregnancy and asthma, respiratory and atopic symptoms in 5-year-old children. Thorax 2007, 62, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Netting, M.J.; Middleton, P.F.; Makrides, M. Does maternal diet during pregnancy and lactation affect outcomes in offspring? A systematic review of food-based approaches. Nutrition 2014, 30, 1225–1241. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.Y.; Forno, E.; Holguin, F.; Celedón, J.C. Diet and asthma: An update. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 369. [Google Scholar] [CrossRef] [PubMed]
- Hodge, L.; Salome, C.M.; Peat, J.K.; Haby, M.M.; Xuan, W.; Woolcock, A.J. Consumption of oily fish and childhood asthma risk. Med. J. Aust. 1996, 164, 137–140. [Google Scholar] [PubMed]
- Takemura, Y.; Sakurai, Y.; Honjo, S.; Tokimatsu, A.; Gibo, M.; Hara, T.; Kusakari, A.; Kugai, N. The relationship between fish intake and the prevalence of asthma: The Tokorozawa childhood asthma and pollinosis study. Prev. Med. 2002, 34, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Xun, P.; He, K. Fish and fish oil intake in relation to risk of asthma: A systematic review and meta-analysis. PLoS ONE 2013, 8, e80048. [Google Scholar] [CrossRef] [PubMed]
- Stratakis, N.; Roumeliotaki, T.; Oken, E.; Ballester, F.; Barros, H.; Basterrechea, M.; Cordier, S.; De Groot, R.; den Dekker, H.T.; Duijts, L. Fish and seafood consumption during pregnancy and the risk of asthma and allergic rhinitis in childhood: A pooled analysis of 18 European and US birth cohorts. Int. J. Epidemiol. 2017, 46, 1465–1477. [Google Scholar] [CrossRef]
- De Luis, D.A.; Armentia, A.; Aller, R.; Asensio, A.; Sedano, E.; Izaola, O.; Cuellar, L. Dietary intake in patients with asthma: A case control study. Nutrition 2005, 21, 320–324. [Google Scholar] [CrossRef]
- Pelé, F.; Bajeux, E.; Gendron, H.; Monfort, C.; Rouget, F.; Multigner, L.; Viel, J.F.; Cordier, S. Maternal fish and shellfish consumption and wheeze, eczema and food allergy at age two: A prospective cohort study in Brittany, France. Environ. Health 2013, 12, 102. [Google Scholar] [CrossRef]
- Miyake, Y.; Tanaka, K.; Okubo, H.; Sasaki, S.; Arakawa, M. Maternal fat intake during pregnancy and wheeze and eczema in Japanese infants: The Kyushu Okinawa Maternal and Child Health Study. Ann. Epidemiol. 2013, 23, 674–680. [Google Scholar] [CrossRef]
- Standl, M.; Demmelmair, H.; Koletzko, B.; Heinrich, J. Cord blood LC-PUFA composition and allergic diseases during the first 10 yr. Results from the LISA plus study. Pediatr. Allergy Immunol. 2014, 25, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Soutar, A.; Seaton, A.; Brown, K. Bronchial reactivity and dietary antioxidants. Thorax 1997, 52, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Trak-Fellermeier, M.; Brasche, S.; Winkler, G.; Koletzko, B.; Heinrich, J. Food and fatty acid intake and atopic disease in adults. Eur. Respir. J. 2004, 23, 575–582. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.L.; Pan, W.H. Dietary fats and asthma in teenagers: Analyses of the first Nutrition and Health Survey in Taiwan (NAHSIT). Clin. Exp. Allergy 2001, 31, 1875–1880. [Google Scholar] [CrossRef] [PubMed]
- Dunder, T.; Kuikka, L.; Turtinen, J.; Räsänen, L.; Uhari, M. Diet, serum fatty acids, and atopic diseases in childhood. Allergy 2001, 56, 425–428. [Google Scholar] [CrossRef]
| Variables | Doctor-Diagnosed Asthma | Medication-Prescribed Asthma | ||||
|---|---|---|---|---|---|---|
| Without (n = 12,723) | With (n = 315) | p-Value | Without (n = 12,905) | With (n = 133) | p-Value | |
| Sex | ||||||
| Male | 5113 (50.6) | 105 (45.1) | 0.1049 | 5175 (50.5) | 43 (45.0) | 0.2904 |
| Female | 7610 (49.4) | 210 (54.9) | 7730 (49.5) | 90 (55.0) | ||
| Age (years) | 41.2 ± 0.2 | 39.2 ± 1.0 | 0.0335 | 41.1 ± 0.2 | 44.9 ± 1.4 | 0.0063 |
| BMI (kg/m2) | 23.8 ± 0.0 | 24.1 ± 0.2 | 0.1387 | 23.8 ± 0.0 | 24.8 ± 0.4 | 0.0106 |
| Residential area | ||||||
| Urban | 10611 (85.7) | 256 (84.2) | 0.4917 | 10765 (85.7) | 102 (81.4) | 0.2037 |
| Rural | 2112 (14.3) | 59 (15.8) | 2140 (14.3) | 31 (18.6) | ||
| Education level | ||||||
| <High school graduate | 6170 (43.5) | 161 (42.3) | 0.8637 | 6241 (43.3) | 90 (58.0) | 0.0185 |
| High school graduate | 2090 (17.7) | 51 (19.0) | 2129 (17.8) | 12 (11.8) | ||
| ≥College graduate | 4429 (38.8) | 102 (38.7) | 4500 (38.9) | 31 (30.1) | ||
| Household income | ||||||
| Low | 1192 (9.0) | 42 (11.2) | 0.1535 | 1210 (9.0) | 24 (15.7) | 0.0993 |
| Moderately low | 3142 (24.3) | 65 (21.3) | 3176 (24.2) | 31 (22.6) | ||
| Moderately high | 3963 (31.5) | 111 (36.4) | 4033 (31.6) | 41 (32.2) | ||
| High | 4389 (35.3) | 97 (31.0) | 4449 (35.2) | 37 (29.5) | ||
| Alcohol consumption | ||||||
| Never | 1786 (13.2) | 59 (16.4) | 0.4215 | 1814 (13.2) | 31 (21.5) | 0.1548 |
| ≤1 drink/month | 3902 (32.1) | 97 (33.2) | 3960 (32.2) | 39 (31.0) | ||
| 2–4 drink/month | 3192 (29.3) | 69 (28.7) | 3241 (29.3) | 20 (21.1) | ||
| 2–3 drink/week | 2032 (18.7) | 47 (17.5) | 2060 (18.6) | 19 (19.3) | ||
| ≥4 drink/week | 747 (6.7) | 12 (4.2) | 752 (6.6) | 7 (7.1) | ||
| Dietary supplement use | ||||||
| Yes | 5861 (44.4) | 133 (41.1) | 0.2846 | 5938 (44.3) | 56 (40.8) | 0.4951 |
| Smoking status | ||||||
| Current smokers | 2172 (25.8) | 54 (25.0) | 0.6101 | 2202 (25.8) | 24 (29.4) | 0.8019 |
| Ex-smokers | 379 (4.4) | 5 (2.9) | 382 (4.4) | 2 (3.5) | ||
| Nonsmokers | 7974 (69.8) | 207 (72.0) | 81 (67.1) | |||
| Variables | Doctor-Diagnosed Asthma | Medication-Prescribed Asthma | ||||
|---|---|---|---|---|---|---|
| Without (n = 12,723) | With (n = 315) | p-Value | Without (n = 12,905) | With (n = 133) | p-Value | |
| Food intake (g) | ||||||
| Cereals/potatoes/sugar products | 349.3 ± 2.3 | 333.2 ± 11.3 | 0.2961 | 349.1 ± 2.2 | 321.7 ± 16.1 | 0.0006 |
| Beans/nuts/seeds | 44.0 ± 0.9 | 38.3 ± 4.1 | 0.5653 | 43.9 ± 0.9 | 41.5 ± 7.2 | 0.6520 |
| Meats and eggs | 146.8 ± 1.9 | 132.7 ± 10.5 | 0.9874 | 146.6 ± 1.9 | 127.6 ± 17.5 | 0.8904 |
| Fishes and shellfishes | 98.2 ± 1.9 | 83.1 ± 10.2 | 0.5890 | 97.9 ± 1.9 | 90.0 ± 20.8 | 0.8761 |
| Fishes | 27.6 ± 0.7 | 22.4 ± 3.3 | 0.0923 | 27.5 ± 0.7 | 27.1 ± 5.8 | 0.5785 |
| Milk and dairy products | 88.9 ± 1.8 | 110.2 ± 14.8 | 0.2664 | 89.1 ± 1.8 | 118.4 ± 29.9 | 0.3723 |
| Fruit and vegetable | 518.1 ± 4.6 | 494.5 ± 24.9 | 0.8628 | 517.3 ± 4.6 | 534.1 ± 39.9 | 0.9614 |
| Mushrooms | 6.7 ± 0.3 | 4.5 ± 0.8 | 0.0964 | 6.7 ± 0.3 | 4.9 ± 1.4 | 0.3942 |
| Seaweeds | 24.7 ± 1.1 | 17.4 ± 3.3 | 0.0638 | 24.6 ± 1.1 | 13.5 ± 4.1 | 0.0110 |
| Energy and nutrient intake | ||||||
| Energy (kcal) | 2128.8 ± 9.8 | 2028.3 ± 58.3 | 0.0705 | 2127.0 ± 9.7 | 2056.5 ± 97.6 | 0.6463 |
| Carbohydrate (g) | 312.0 ± 1.4 | 302.7 ± 8.3 | 0.7192 | 311.8 ± 1.4 | 302.2 ± 12.7 | 0.0195 |
| Protein (g) | 75.2 ± 0.4 | 71.6 ± 3.0 | 0.4817 | 75.2 ± 0.4 | 74.3 ± 5.4 | 0.3215 |
| Fat (g) | 50.6 ± 0.4 | 47.6 ± 2.0 | 0.7848 | 50.5 ± 0.4 | 47.2 ± 3.3 | 0.3143 |
| Calcium (mg) | 501.8 ± 3.3 | 496.7 ± 25.5 | 0.4282 | 501.5 ± 3.3 | 524.7 ± 46.1 | 0.5543 |
| Iron (mg) | 17.7 ± 0.2 | 16.0 ± 0.6 | 0.1972 | 17.6 ± 0.2 | 16.1 ± 1.0 | 0.0246 |
| Thiamin (mg) | 2.1 ± 0.0 | 2.0 ± 0.1 | 0.9728 | 2.1 ± 0.0 | 2.1 ± 0.1 | 0.5263 |
| Riboflavin (mg) | 1.5 ± 0.0 | 1.5 ± 0.1 | 0.1498 | 1.5 ± 0.0 | 1.6 ± 0.2 | 0.1721 |
| Niacin (mg) | 17.4 ± 0.1 | 16.5 ± 0.7 | 0.8362 | 17.4 ± 0.1 | 17.2 ± 1.4 | 0.6666 |
| Vitamin C (mg) | 100.5 ± 1.5 | 95.6 ± 7.5 | 0.8166 | 100.3 ± 1.5 | 110.3 ± 13.6 | 0.8952 |
| Vitamin A (μgRE) | 751.9 ± 11.1 | 780.7 ± 75.2 | 0.6336 | 752.1 ± 11.1 | 798.2 ± 160.8 | 0.6804 |
| Fatty acids | ||||||
| SFA (g) | 14.7 ± 0.1 | 14.3 ± 0.7 | 0.3194 | 14.7 ± 0.1 | 14.5 ± 1.3 | 0.1252 |
| MUFA (g) | 16.2 ± 0.1 | 15.3 ± 0.8 | 0.7624 | 16.2 ± 0.1 | 15.4 ± 1.2 | 0.1925 |
| PUFA (g) | 12.2 ± 0.1 | 11.4 ± 0.6 | 0.9407 | 12.2 ± 0.1 | 11.0 ± 0.8 | 0.9465 |
| n-3 (g) | 1.7 ± 0.0 | 1.5 ± 0.1 | 0.8966 | 1.7 ± 0.0 | 1.5 ± 0.1 | 0.6403 |
| n-6 (g) | 10.6 ± 0.1 | 9.8 ± 0.5 | 0.9783 | 10.6 ± 0.1 | 9.6 ± 0.7 | 0.8517 |
| n3/n6 | 0.18 ± 0.00 | 0.17 ± 0.01 | 0.0708 | 0.18 ± 0.00 | 0.16 ± 0.01 | 0.0275 |
| EPA (mg) | 108.2 ± 2.6 | 96.2 ± 15.7 | 0.9384 | 107.6 ± 2.6 | 135.9 ± 37.0 | 0.3860 |
| DHA (mg) | 187.7 ± 5.2 | 152.1 ± 22.5 | 0.5155 | 186.6 ± 5.1 | 205.4 ± 50.5 | 0.5818 |
| Cholesterol (mg) | 283.5 ± 2.9 | 287.6 ± 19.0 | 0.4395 | 283.5 ± 2.8 | 291.7 ± 31.0 | 0.3442 |
| Variable | Range (g) | Median (g) | Doctor-Diagnosed Asthma | Medication-Prescribed Asthma | ||||
|---|---|---|---|---|---|---|---|---|
| Prevalence (%) | Crude OR (95% CI) | Adjusted OR (95% CI) | Prevalence (%) | Crude OR (95% CI) | Adjusted OR (95% CI) | |||
| Fish and shellfish | ||||||||
| Q1 | <4.07 | 0 | 94/3259 (2.88) | 1 | 1 | 41/3259 (1.26) | 1 | 1 |
| Q2 | 4.07–35.20 | 14.82 | 78/3260 (2.39) | 0.82 (0.60–1.13) | 0.90 (0.62–1.32) | 32/3260 (0.98) | 0.71 (0.42–1.20) | 0.77 (0.41–1.44) |
| Q3 | 35.21–125.14 | 67.05 | 71/3260 (2.18) | 0.82 (0.58–1.17) | 0.75 (0.49–1.15) | 35/3260 (1.07) | 0.93 (0.56–1.55) | 0.71 (0.38–1.34) |
| Q4 | >125.14 | 233.34 | 72/3259 (2.21) | 0.77 (0.54–1.10) | 0.85 (0.57–1.27) | 25/3259 (0.77) | 0.55 (0.31–0.98) | 0.57 (0.29–1.12) |
| P for trend | 0.2705 | 0.6894 | 0.0501 | 0.1005 | ||||
| Fish | ||||||||
| Q1 | 0 | 0 | 135/4782 (2.82) | 1 | 1 | 51/4782 (1.07) | 1 | 1 |
| Q2 | <2.49 | 1.09 | 43/1737 (2.48) | 0.73 (0.49–1.11) | 0.78 (0.49–1.24) | 17/1737 (0.98) | 0.78 (0.42–1.43) | 0.80 (0.38–1.69) |
| Q3 | 2.49–24.01 | 7.46 | 74/3260 (2.27) | 0.65 (0.47–0.91) | 0.71 (0.48–1.04) | 36/3260 (1.10) | 0.71 (0.45–1.12) | 0.60 (0.34–1.07) |
| Q4 | >24.01 | 69.32 | 63/3259 (1.93) | 0.71 (0.51–0.99) | 0.63 (0.41–0.97) | 29/3259 (0.89) | 0.91 (0.54–1.53) | 0.77 (0.40–1.50) |
| P for trend | 0.1617 | 0.0637 | 0.9453 | 0.6532 | ||||
| Seaweeds | ||||||||
| Q1 | 0 | 0 | 141/5627 (2.51) | 1 | 1 | 62/5627 (1.10) | 1 | 1 |
| Q2 | <1.00 | 0.50 | 27/896 (3.01) | 0.82 (0.50–1.34) | 0.78 (0.43–1.40) | 10/896 (1.12) | 0.84 (0.38–1.84) | 0.82 (0.30–2.24) |
| Q3 | 1.01–7.47 | 3.07 | 75/3256 (2.30) | 0.87 (0.64–1.19) | 0.95 (0.66–1.36) | 34/3256 (1.04) | 0.88 (0.56–1.41) | 0.99 (0.57–1.75) |
| Q4 | >7.47 | 28.84 | 72/3259 (2.21) | 0.72 (0.53–0.98) | 0.66 (0.44–1.00) | 27/3259 (0.83) | 0.52 (0.32–0.85) | 0.37 (0.19–0.70) |
| P for trend | 0.0346 | 0.0300 | 0.0018 | <0.0001 | ||||
| Variable | Range (g) | Median (g) | Doctor-Diagnosed Asthma | Medication-Prescribed Asthma | ||||
|---|---|---|---|---|---|---|---|---|
| Prevalence (%) | Crude OR (95% CI) | Adjusted OR (95% CI) | Prevalence (%) | Crude OR (95% CI) | Adjusted OR (95% CI) | |||
| Total fat | ||||||||
| Q1 | <24.52 | 16.94 | 89/3259 (2.73) | 1 | 1 | 42/3259 (1.29) | 1 | 1 |
| Q2 | 24.52–39.69 | 32.00 | 73/3260 (2.24) | 0.79 (0.55–1.14) | 0.75 (0.48–1.17) | 32/3260 (0.98) | 0.78 (0.46–1.32) | 0.76 (0.38–1.50) |
| Q3 | 39.70–60.98 | 48.90 | 83/3260 (2.55) | 0.90 (0.64–1.26) | 0.92 (0.59–1.43) | 31/3260 (0.95) | 0.74 (0.47–1.17) | 0.96 (0.48–1.93) |
| Q4 | >60.98 | 83.53 | 70/3259 (2.15) | 0.80 (0.55–1.16) | 1.04 (0.60–1.81) | 28/3259 (0.86) | 0.75 (0.44–1.27) | 1.61 (0.65–3.99) |
| P for trend | 0.3792 | 0.6269 | 0.3702 | 0.2090 | ||||
| SFA | ||||||||
| Q1 | <6.36 | 4.21 | 783259 (2.39) | 1 | 1 | 41/3259 (1.26) | 1 | 1 |
| Q2 | 6.36–11.02 | 8.67 | 82/3260 (2.52) | 1.18 (0.82–1.69) | 1.21 (0.78–1.89) | 26/3260 (0.80) | 0.67 (0.38-1.20) | 0.59 (0.83-1.15) |
| Q3 | 11.03-17.73 | 13.96 | 77/3260 (2.36) | 1.15 (0.80-1.65) | 1.33 (0.84-2.11) | 38/3260 (1.17) | 1.05 (0.66-1.68) | 1.59 (0.83-3.08) |
| Q4 | >17.73 | 25.05 | 78/3259 (2.39) | 1.11 (0.77-1.60) | 1.63 (0.97-2.73) | 28/3259 (0.86) | 0.80 (0.46-1.37) | 1.58 (0.74-3.38) |
| P for trend | 0.7875 | 0.0743 | 0.7171 | 0.0871 | ||||
| MUFA | ||||||||
| Q1 | <6.90 | 4.55 | 87/3259 (2.67) | 1 | 1 | 42/3259 (1.29) | 1 | 1 |
| Q2 | 6.90-12.04 | 9.48 | 79/3260 (2.42) | 0.98 (0.68-1.39) | 0.99 (0.63-1.55) | 31/3260 (0.95) | 0.89 (0.52-1.50) | 1.14 (0.57-2.27) |
| Q3 | 12.05–19.76 | 15.24 | 78/3260 (2.39) | 0.91 (0.63–1.32) | 0.87 (0.54–1.40) | 31/3260 (0.95) | 0.75 (0.46–1.23) | 0.92 (0.41–2.08) |
| Q4 | >19.76 | 27.87 | 71/3259 (2.18) | 0.87 (0.60–1.26) | 1.05 (0.57–1.93) | 29/3259 (0.89) | 0.84 (0.50–1.43) | 1.77 (0.66–4.76) |
| P for trend | 0.4361 | 0.8685 | 0.5791 | 0.2505 | ||||
| PUFA | ||||||||
| Q1 | <5.57 | 3.76 | 95/3259 (2.92) | 1 | 1 | 48/3259 (1.47) | 1 | 1 |
| Q2 | 5.57–9.24 | 7.36 | 64/3260 (1.96) | 0.67 (0.46–0.97) | 0.72 (0.45–1.16) | 27/3260 (0.83) | 0.56 (0.33–0.94) | 0.58 (0.28–1.18) |
| Q3 | 9.25–14.92 | 11.71 | 87/3260 (2.67) | 1.04 (0.74–1.46) | 1.09 (0.70–1.69) | 30/3260 (0.92) | 0.85 (0.52–1.40) | 0.86 (0.42–1.75) |
| Q4 | >14.92 | 21.0 | 69/3259 (2.12) | 0.74 (0.51–1.07) | 1.10 (0.66–1.84) | 28/3259 (0.86) | 0.55 (0.33–0.93) | 0.85 (0.37–1.99) |
| P for trend | 0.3328 | 0.3753 | 0.0803 | 0.9308 | ||||
| n–3 PUFA | ||||||||
| Q1 | <0.65 | 0.41 | 91/3259 (2.79) | 1 | 1 | 49/3259 (1.50) | 1 | 1 |
| Q2 | 0.65–1.19 | 0.90 | 77/3260 (2.36) | 0.82 (0.58–1.16) | 0.86 (0.57–1.29) | 28/3260 (0.86) | 0.55 (0.32–0.93) | 0.62 (0.31–1.22) |
| Q3 | 1.20–2.10 | 1.56 | 81/3260 (2.48) | 0.80 (0.56–1.14) | 0.82 (0.53–1.26) | 27/3260 (0.83) | 0.47 (0.27–0.82) | 0.43 (0.21–0.89) |
| Q4 | >2.10 | 3.07 | 66/3259 (2.03) | 0.74 (0.51–1.10) | 0.90 (0.57–1.40) | 29/3259 (0.89) | 0.63 (0.37–1.07) | 0.71 (0.33–1.51) |
| P for trend | 0.1918 | 0.7609 | 0.2290 | 0.4776 | ||||
| n–6 PUFA | ||||||||
| Q1 | <4.65 | 3.09 | 93/3259 (2.85) | 1 | 1 | 48/3259 (1.47) | 1 | 1 |
| Q2 | 4.65–7.85 | 6.24 | 72/3260 (2.21) | 0.82 (0.56–1.19) | 0.92 (0.58–1.45) | 28/3260 (0.86) | 0.64 (0.38–1.09) | 0.69 (0.35–1.36) |
| Q3 | 7.86–12.93 | 10.04 | 81/3260 (2.48) | 0.99 (0.70–1.41) | 1.03 (0.65–1.63) | 29/3260 (0.89) | 0.79 (0.47–1.31) | 0.82 (0.38–1.74) |
| Q4 | >12.93 | 18.30 | 69/3259 (2.12) | 0.75 (0.52–1.09) | 1.07 (0.63–1.82) | 28/3259 (0.86) | 0.54 (0.32–0.89) | 0.80 (0.32–1.98) |
| P for trend | 0.2015 | 0.6872 | 0.0305 | 0.7295 | ||||
| n–3/n–6 | ||||||||
| Q1 | <0.10 | 0.08 | 92/3259 (2.82) | 1 | 1 | 45/3259 (1.38) | 1 | 1 |
| Q2 | 0.10–0.13 | 0.12 | 68/3259 (2.09) | 0.78 (0.54–1.15) | 0.92 (0.59–1.43) | 29/3259 (0.89) | 0.60 (0.35–1.02) | 0.61 (0.32–1.16) |
| Q3 | 0.14–0.19 | 0.16 | 75/3260 (2.30) | 0.89 (0.63–1.27) | 1.01 (0.66–1.58) | 33/3260 (1.01) | 0.75 (0.45–1.25) | 0.69 (0.36–1.31) |
| Q4 | >0.19 | 0.30 | 80/3259 (2.45) | 0.92 (0.65–1.30) | 0.91 (0.58–1.43) | 26/3259 (0.80) | 0.59 (0.33–1.06) | 0.47 (0.22–0.99) |
| P for trend | 0.9761 | 0.7471 | 0.1475 | 0.0563 | ||||
| Cholesterol | ||||||||
| Q1 | <93.07 | 48.68 | 97/3259 (2.98) | 1 | 1 | 45/3259 (1.38) | 1 | 1 |
| Q2 | 93.07–202.34 | 142.84 | 62/3260 (1.90) | 0.61 (0.42–0.90) | 0.64 (0.41–0.99) | 33/3260 (1.01) | 0.76 (0.48–1.19) | 0.84 (0.48–1.49) |
| Q3 | 202.35–361.02 | 272.54 | 81/3260 (2.48) | 0.86 (0.61–1.21) | 1.05 (0.73–1.51) | 27/3260 (0.83) | 0.60 (0.35–1.04) | 0.72 (0.37–1.42) |
| Q4 | >361.02 | 534.53 | 75/3259 (2.30) | 0.87 (0.61–1.24) | 0.85 (0.56–1.31) | 28/3259 (0.86) | 0.88 (0.53–1.46) | 0.96 (0.50–1.85) |
| P for trend | 0.9132 | 0.9354 | 0.8583 | 0.9555 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, E.-k.; Ju, S.-Y. Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults. Nutrients 2019, 11, 2187. https://doi.org/10.3390/nu11092187
Kim E-k, Ju S-Y. Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults. Nutrients. 2019; 11(9):2187. https://doi.org/10.3390/nu11092187
Chicago/Turabian StyleKim, Eun-kyung, and Se-Young Ju. 2019. "Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults" Nutrients 11, no. 9: 2187. https://doi.org/10.3390/nu11092187
APA StyleKim, E.-k., & Ju, S.-Y. (2019). Asthma and Dietary Intake of Fish, Seaweeds, and Fatty Acids in Korean Adults. Nutrients, 11(9), 2187. https://doi.org/10.3390/nu11092187





