The Emerging Role of l-Glutamine in Cardiovascular Health and Disease
Abstract
1. Introduction
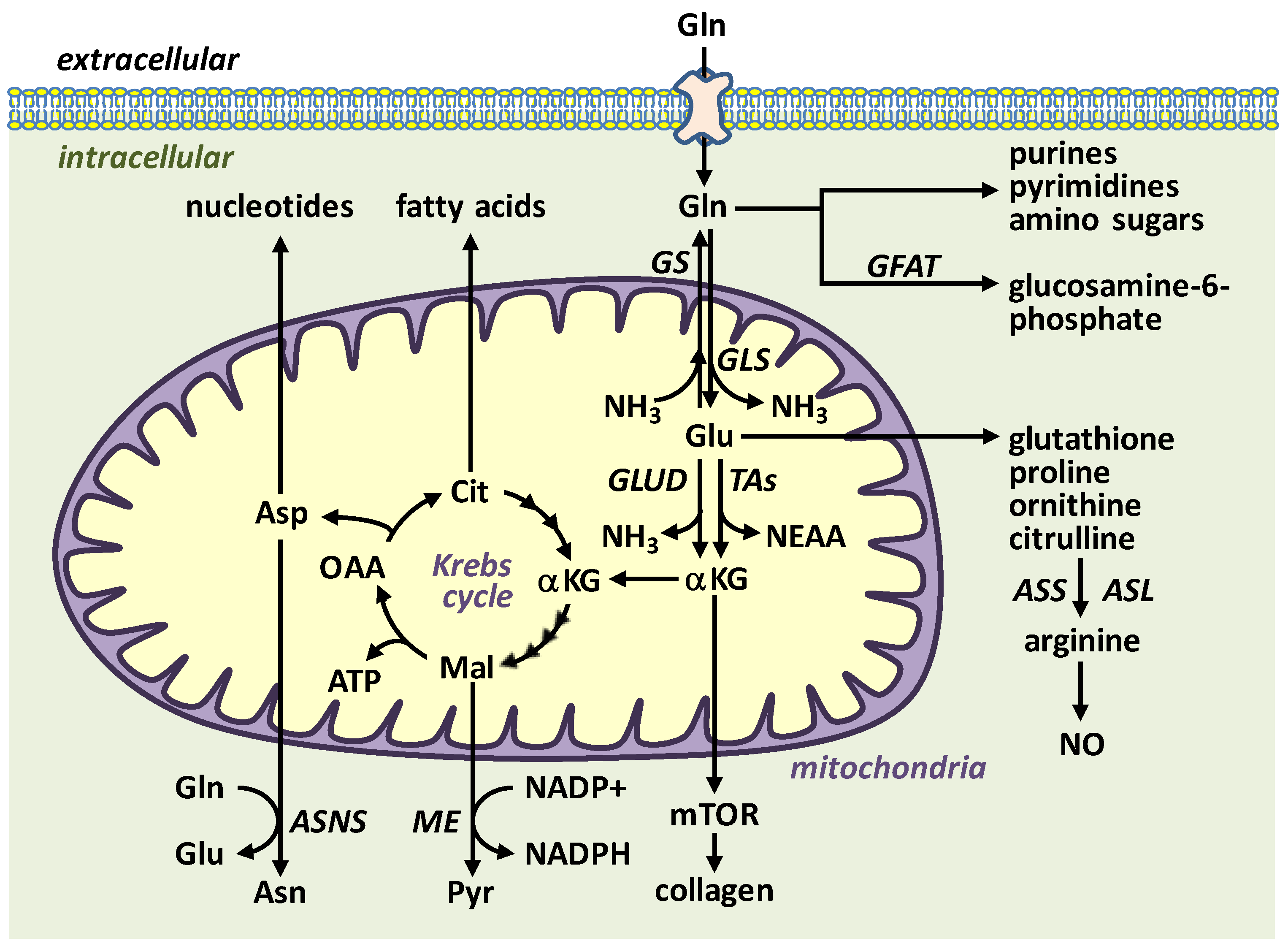
2. l-Glutamine Metabolism
3. l-Glutamine and Cardiometabolic Disease
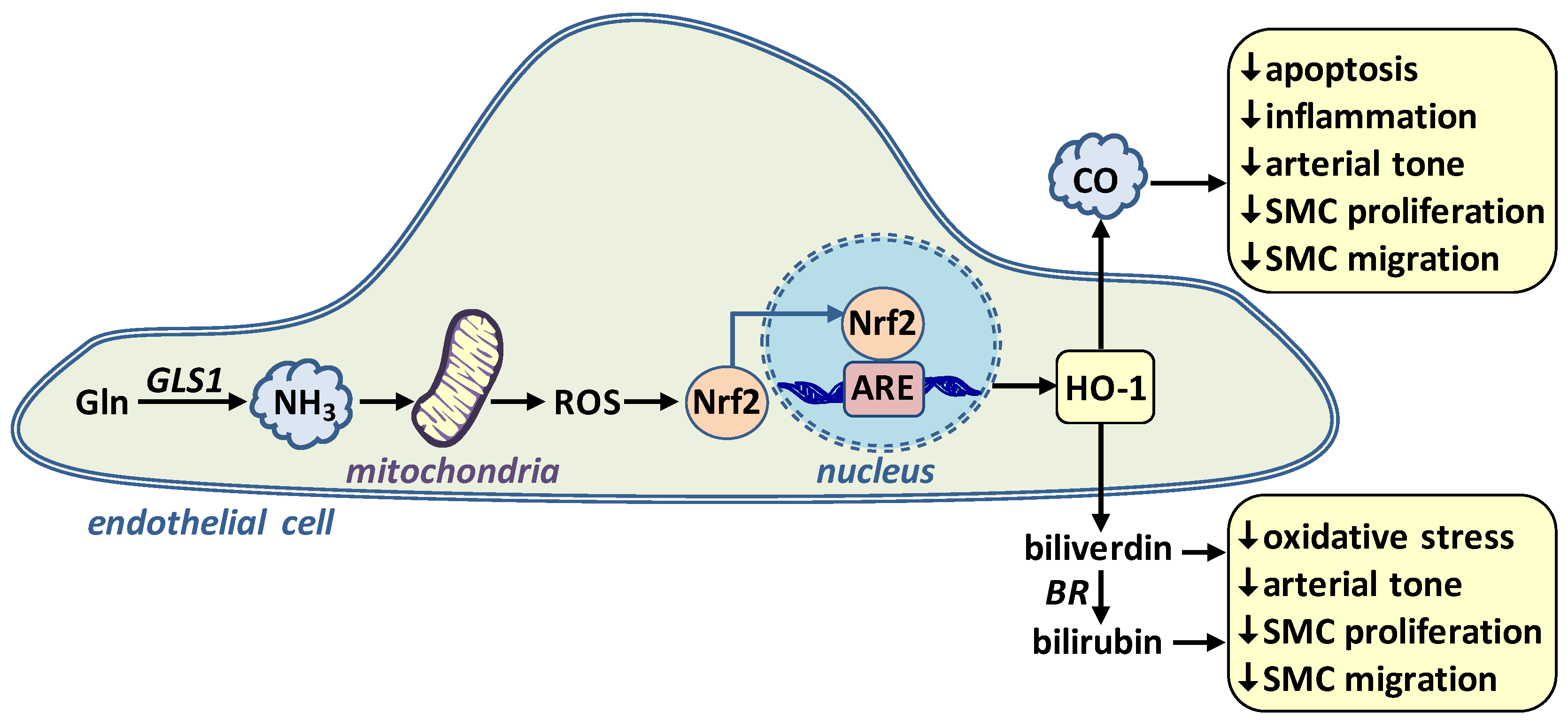
4. l-Glutamine and Endothelial Cell Function
5. l-Glutamine and Pulmonary Arterial Hypertension
6. l-Glutamine and other Cardiovascular Disorders
7. Therapeutic Targeting of l-Glutamine in Cardiovascular Disease
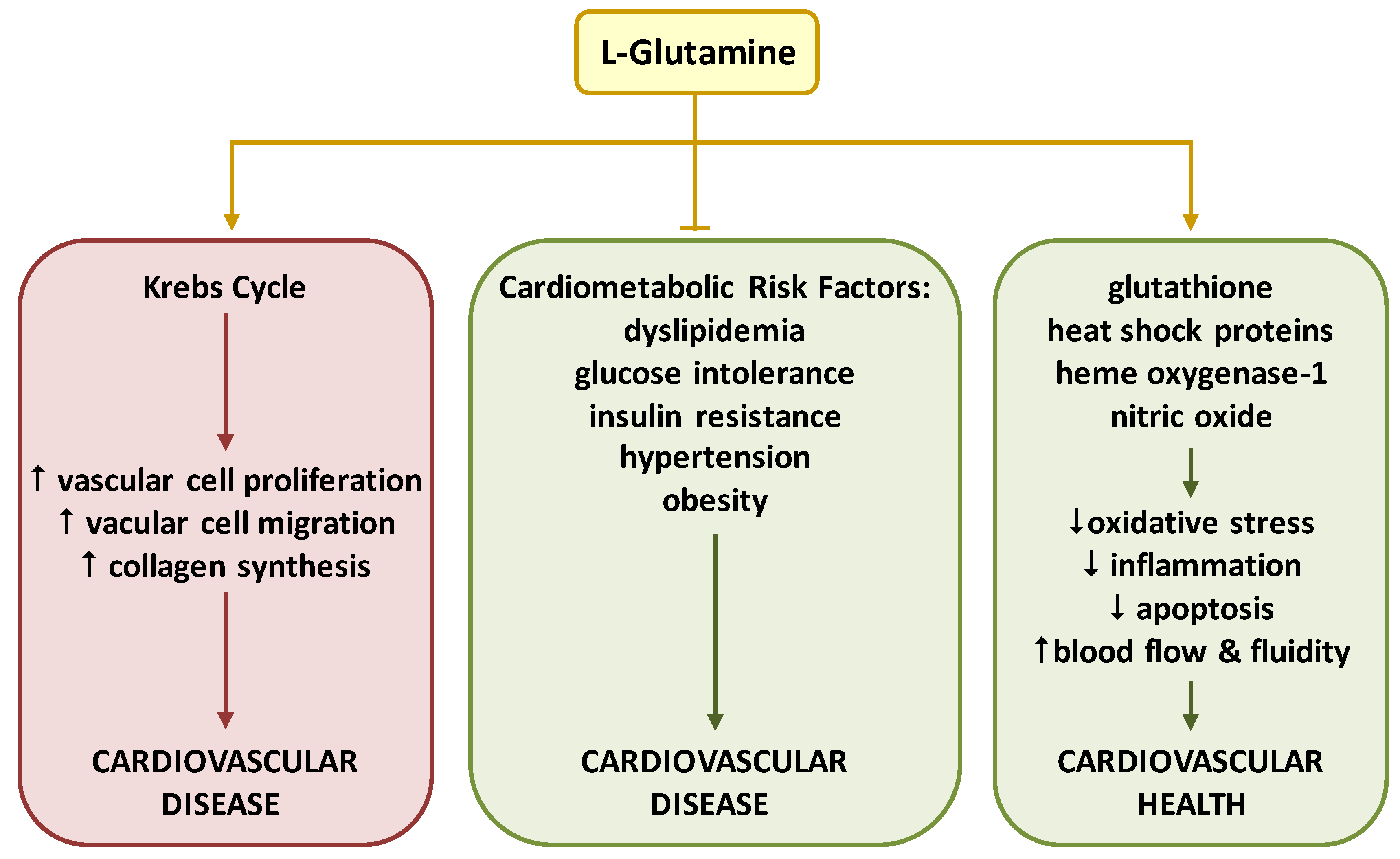
8. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics-2017 update. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Tarride, J.E.; Lim, M.; Desmeules, M.; Luo, W.; Burke, N.; O’Reilly, D.; Bowen, J.; Goeree, R. A review of the cost of cardiovascular disease. Can. J. Cardiol. 2009, 25, e195–e202. [Google Scholar] [CrossRef]
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: nitric oxide and beyond. Biochem. J. 1998, 336, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Durante, W. Regulation of l-arginine transport and metabolism in vascular smooth muscle cells. Cell Biophys. 2001, 35, 19–34. [Google Scholar] [CrossRef]
- Forstermann, U.; Sessa, W.C. Nitric oxide synthase: regulation and function. Eur. Heart J. 2012, 33, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Palfrey, H.A.; Pathak, R.; Kadowitz, P.J.; Gettys, T.W.; Murthy, S.N. The metabolism and significance of homocysteine in health and disease. Nutr. Metab. 2017, 14, 78. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Ramprasath, T.; Wang, H.; Zou, M.H. Abnormal kynurenine pathway of tryptophan catabolism in cardiovascular diseases. Cell. Mol. Life Sci. 2017, 74, 2899–2916. [Google Scholar] [CrossRef]
- Bertero, T.; Perk, D.; Chan, S.Y. The molecular rationale for therapeutic targeting of glutamine metabolism in pulmonary hypertension. Expert Opin. Ther. Targets 2019, 23, 511–524. [Google Scholar] [CrossRef]
- Xi, P.; Jiang, Z.; Zheng, C.; Lin, Y.; Wu, G. Regulation of protein metabolism by glutamine: Implications for nutrition and health. Front. Biosci. 2011, 16, 578–597. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Cheng, T. Q’s next: The diverse functions of glutamine in metabolism, cell biology, and cancer. Oncogene 2010, 29, 313–324. [Google Scholar] [CrossRef]
- Cruzat, V.; Rogero, M.M.; Keane, K.N.; Curi, R.; Newsholme, P. Glutamine: metabolism and immune function, supplementation, and clinical translation. Nutrients 2018, 10, 1564. [Google Scholar] [CrossRef] [PubMed]
- Bertero, T.; Oldham, W.M.; Cottrill, K.A.; Pisano, S.; Vanderpool, R.R.; Yu, Q.; Zhao, J.; Tai, Y.; Tang, Y.; Zhang, Y.Y.; et al. Vascular stiffness mechanoactivates YAP/TAZ-dependent glutaminolysis to drive pulmonary hypertension. J. Clin. Invest. 2016, 126, 3313–3335. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Y.; Deng, Y.J.; Li, X.Q.; Ning, Y.; Lin, X.P.; Guo, S.M.; Chen, M.X.; Han, M. Glutaminolysis was induced by TGF-β1 through PP2Ac regulated Raf-MEK-ERK signaling in endothelial cells. PLoS ONE 2016, 11, e0162658. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.; Rumsey, W.L.; Erecinska, M. Glutamine catabolism by heart muscle: Properties of phosphate-activated glutaminase. Biochem. J. 1992, 24, S26. [Google Scholar]
- Boelens, P.G.; van Leeuwen, P.A.; Dejong, C.H.; Deutz, N.E. Intestinal renal metabolism of l-citrulline and l-arginine following enteral or parenteral infusion of l-alanyl-L[2,15N]glutamine or l-[2,15N]glutamine in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 289, G679–G685. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Giles, W.H.; Dietz, W.H. Prevalence of the metabolic syndrome among US adults: Findings from the Third National Health and Nutrition Examination Survey. Obstet. Gynecol. Surv. 2002, 57, 356–359. [Google Scholar] [CrossRef]
- McNeill, A.M.; Rosamond, W.D.; Girman, C.J.; Golden, S.H.; Schmidt, M.I.; East, H.E.; Ballantyne, C.M.; Heiss, G. The metabolic syndrome and 11-year risk of incident cardiovascular disease in the atherosclerosis risk in communities study. Diabetes Care 2005, 28, 385–390. [Google Scholar] [CrossRef]
- Ford, E.S. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome. Diabetes Care 2005, 28, 1769–1778. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Rhee, E.P.; Larson, M.G.; Lewis, G.D.; McCabe, E.L.; Shen, D.; Palma, M.J.; Roberts, L.D.; Dejam, A.; Souza, A.L.; et al. Metabolite profiling identifies pathways associated with metabolic risk in humans. Circulation 2012, 125, 2222–2231. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferre, M.; Hruby, A.; Toledo, E.; Clish, C.B.; Martinez-Gonzalez, M.A.; Salas-Salvado, J.; Hu, F.B. Metabolomics in prediabetes and diabetes: A systemic review and meta-analysis. Diabetes Care 2016, 39, 833–846. [Google Scholar] [CrossRef]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed]
- Würtz, P.; Mäkinen, V.P.; Soininen, P.; Kangas, A.J.; Tukiainen, T.; Kettunen, J.; Savolainen, M.J.; Tammelin, T.; Viikari, J.S.; Rönnemaa, T.; et al. Metabolic signatures of insulin resistance in 7,098 young adults. Diabetes 2012, 61, 1372–1380. [Google Scholar] [CrossRef] [PubMed]
- Wijekoon, E.P.; Skinner, C.; Brosnan, M.E.; Brosnan, J.T. Amino acid metabolism in the Zucker fatty rat: Effects of insulin resistance and type 2 diabetes. Can. J. Physiol. Pharmacol. 2004, 82, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yang, R.; Wang, M.; Ji, F.; Li, H.; Tang, Y.; Chen, W.; Dong, J. Identification of serum metabolites associated with obesity and traditional risk factors for metabolic disease in Chinese adults. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yu, X.; Zhang, W.; Ji, F.; Wang, M.; Yang, R.; Li, H.; Chen, W.; Dong, J. Association of serum metabolites with impaired fasting glucose/diabetes and traditional risk factors for metabolic disease in Chinese adults. Clin. Chim. Acta 2018, 487, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Ntzouvani, A.; Nomikos, T.; Panagiotakos, D.; Fragopoulou, E.; Pitsavos, C.; McCann, A.; Ueland, P.; Antonopoulou, S. Amino acid profile and metabolic syndrome in a male Mediterranean population: A cross-sectional study. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1021–1030. [Google Scholar] [CrossRef]
- Vangipurapu, J.; Stancáková, A.; Smith, U.; Kuusisto, J.; Laakso, M. Nine amino acids are associated with decreased insulin secretion and elevated glucose levels in a 7.4-year follow-up study of 5,181 Finnish men. Diabetes 2019, 68, 1353–1358. [Google Scholar] [CrossRef]
- Greenfield, J.R.; Farooqi, I.S.; Keogh, J.M.; Henning, E.; Habib, A.M.; Blackwood, A.; Reimann, F.; Holst, J.J.; Gribble, F.M. Oral glutamine increases circulating glucagon-like peptide 1, glucagon, and insulin concentrations in lean, obese, type 2 diabetic patients. Am. J. Clin. Nutr. 2009, 89, 106–113. [Google Scholar] [CrossRef]
- Mansour, A.; Tehrani, M.R.M.; Qorbani, M.; Heshmat, R.; Larijani, B.; Hosseini, S. Effect of glutamine supplementation on cardiovascular risk factors in patients with type 2 diabetes. Nutrition 2015, 31, 119–126. [Google Scholar] [CrossRef]
- Torres-Santiago, L.; Mauras, N.; Hossain, J.; Weltman, A.L.; Darmaun, D. Does oral glutamine improve insulin sensitivity in adolescents with type 1 diabetes? Nutrition 2017, 34, 1–6. [Google Scholar] [CrossRef]
- Hissa, M.N.; De Vasconcelos, R.C.; Guimarães, S.B.; Silva, R.P.; Garcia, J.H.P.; De Vasconcelos, P.R.L. Preoperative glutamine infusion improves glycemia in heart surgery patients. Acta Cir. Bras. 2011, 26, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Bakalar, B.; Duska, F.; Pachl, J.; Fric, M.; Otahal, M.; Pazout, J.; Andel, M. Parenterally administered dipeptide alanyl-glutamine prevents worsening of insulin sensitivity in multiple-trauma patients. Crit. Care Med. 2006, 34, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Iwashita, S.; Williams, P.; Jabbour, K.; Ueda, T.; Kobayashi, H.; Baier, S.; Flakoll, P.J. Impact of glutamine supplementation on glucose homeostasis during and after exercise. J. Appl. Physiol. 2005, 99, 1858–1865. [Google Scholar] [CrossRef] [PubMed]
- Petro, A.; Tevrizian, A.; Feinglos, M.N.; Surwit, R.S.; Opara, E.C. L-Glutamine supplementation of a high fat diet reduces body weight and attenuates hyperglycemia and hyperinsulinemia in C57BL/6J mice. J. Nutr. 1996, 126, 273–279. [Google Scholar]
- Prada, P.O.; Hirabara, S.M.; De Souza, C.T.; Schenka, A.A.; Zecchin, H.G.; Vassallo, J.; Velloso, L.A.; Carneiro, E.; Carvalheira, J.B.C.; Curi, R.; et al. RETRACTED ARTICLE: l-glutamine supplementation induces insulin resistance in adipose tissue and improves insulin signalling in liver and muscle of rats with diet-induced obesity. Diabetologia 2007, 50, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Buettger, C.; Kwagh, J.; Matter, A.; Daikhin, Y.; Nissim, I.B.; Collins, H.W.; Yudkoff, M.; Stanley, C.A.; Matschinsky, F.M. A signaling role for glutamine in insulin secretion. J. Biol. Chem. 2004, 279, 13393–13401. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.; Brown, I.J.; Daviglus, M.L.; Chan, Q.; Kesteloot, H.; Ueshima, H.; Zhao, L.; Elliot, P. For the INTERMAP Research Group. Glutamic acid, the main dietary amino acid, and blood pressure: The INTERMAP study (International Collaboration Study of Macronutrients, Micronutrients, and Blood Pressure). Circulation 2009, 120, 221–228. [Google Scholar] [CrossRef]
- Cabrera, O.; Jacques-Silva, M.C.; Speier, S.; Yang, S.-N.; Köhler, M.; Fachado, A.; Vieira, E.; Zierath, J.R.; Kibbey, R.; Berman, D.M.; et al. Glutamate is a positive autocrine signal for glucagon release. Cell Metab. 2008, 7, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Cynober, L.A. The use of alpha-ketoglutarate salts in clinical nutrition and metabolic care. Curr. Opin. Clin. Nutr. Metab. Care 1999, 2, 33–39. [Google Scholar] [CrossRef]
- Karaca, M.; Martin-Levilain, J.; Grimaldi, M.; Li, L.; Dizin, E.; Emre, Y.; Maechler, P. Liver glutamate dehydrogenase controls whole body-body energy partitioning through amino acid-derived gluconeogenesis and ammonia homeostasis. Diabetes 2018, 67, 1949–1961. [Google Scholar] [CrossRef]
- Zheng, Y.; Hu, F.B.; Ruiz-Canela, M.; Clish, C.B.; Dennis, C.; Salas-Salvado, J.; Hruby, A.; Liang, L.; Toledo, E.; Corella, D.; et al. Metabolites of glutamate metabolism are associated with incident cardiovascular events in the PREDIMED PREvencion con DIeta MEDiterranea PREDIMED) trial. J. Am. Heart Assoc. 2016, 5, e003755. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Qi, Q.; Prudente, S.; Mendonca, C.; Andreozzi, F.; Di Pietro, N.; Sturma, M.; Novelli, V.; Mannino, G.C.; Formoso, G.; et al. Association between a genetic variant of related to glutamic acid metabolism and coronary heart disease in type 2 diabetes. JAMA 2013, 310, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Khogali, S.E.; Pringle, S.D.; Weryk, B.V.; Rennie, M.J. Is glutamine beneficial in ischemic heart disease? Nutrition 2002, 18, 123–126. [Google Scholar] [CrossRef]
- Sufit, A.; Weitzel, L.B.; Hamiel, C.; Queensland, K.; Dauber, I.; Rooyackers, O.; Wischmeyer, P.E. Pharmacologically dosed oral glutamine reduces myocardial injury in patients undergoing cardiac surgery: A randomized pilot feasibility trial. Enteral. Nutr. 2012, 36, 556–561. [Google Scholar] [CrossRef]
- Chavez-Tostado, M.; Carrill-Llamas, F.; Martinez-Gutierrez, P.E.; Alvarado-Ramirez, A.; Lopez-Taylor, J.G.; Vasquez-Jiminez, J.C.; Fuentes-Orozco, C.; Rendón-Félix, J.; Irusteta-Jiménez, L.; Calil-Romero, V.C.; et al. Oral glutamine reduces myocardial damage after coronary revascularization under cardiopulmonary bypass. A random clinical trial. Nutr. Hosp. 2017, 34, 277–283. [Google Scholar] [CrossRef]
- Ma, W.; Heianza, Y.; Huang, T.; Wang, T.; Sun, D.; Zheng, Y.; Hu, F.B.; Rexrode, K.M.; Manson, J.; Qi, L. Dietary glutamine, glutamate, and mortality: two large prospective studies in US men and women. Int. J. Epidemiol. 2018, 47, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Epstein, F.H.; Vane, J.R.; Anggärd, E.E.; Botting, R.M. Regulatory functions of the vascular endothelium. N. Engl. J. Med. 1990, 323, 27–36. [Google Scholar] [CrossRef]
- Peyton, K.J.; Liu, X.M.; Yu, Y.; Yates, B.; Behnammanesh, G.; Durante, W. Glutaminase-1 stimulates the proliferation, migration, and survival of human endothelial cells. Biochem. Pharmacol. 2018, 156, 204–214. [Google Scholar] [CrossRef]
- Kim, B.; Li, J.; Jang, C.; Arany, Z. Glutamine fuels proliferation but not migration of endothelial cells. EMBO J. 2017, 36, 2321–2333. [Google Scholar] [CrossRef]
- Huang, H.; Vandekeere, S.; Kalucka, J.; Bierhansl, L.; Zecchin, A.; Bruning, U.; Visnagri, A.; Yuldasheva, N.; Goveia, J.; Cruys, B.; et al. Role of glutamine and interlinked asparagine metabolism in vessel formation. EMBO J. 2017, 36, 2334–2352. [Google Scholar] [CrossRef]
- Eelen, G.; Dubois, C.; Cantelmo, A.R.; Goveia, J.; Brüning, U.; DeRan, M.; Jarugumilli, G.; van Rijssel, J.; Saladino, G.; Comitani, F.; et al. Role of glutamine synthetase in angiogenesis beyond glutamine synthesis. Nature 2018, 561, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Unterluggauer, H.; Mazurek, S.; Lener, B.; Hütter, E.; Eigenbrodt, E.; Zwerschke, W.; Jansen-Dürr, P. Premature senescence of human endothelial cells induced by inhibition of glutaminase. Biogerontology 2008, 9, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, H.; Ogando, D.G.; Li, S.; Feng, M.; Price, F.W.; Tennessen, J.M.; Bonanno, J.A. Glutaminolysis is essential for energy production and ion transport in human corneal endothelium. EBioMedicine 2017, 16, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Hinshaw, D.B.; Burger, J.M. Protective effect of glutamine on endothelial cell ATP in oxidant injury. J. Surg. Res. 1990, 49, 222–227. [Google Scholar] [CrossRef]
- Parolari, A.; Sala, R.; Antona, C.; Bussolati, O.; Alamanni, F.; Mezzadri, P.; Dall’Asta, V.; Gazzola, G.C.; Biglioli, P. Hypertonicity induces injury to cultured human endothelium: attenuation by glutamine. Ann. Thorac. Surg. 1997, 64, 1770–1775. [Google Scholar] [CrossRef]
- Sanchez, E.L.; Carroll, P.A.; Thalhofer, A.B.; Lagunoff, M. Latent KSHV infected endothelial cells are glutamine addicted and require glutaminolysis for survival. PLoS Pathog. 2015, 11, e1005052. [Google Scholar] [CrossRef] [PubMed]
- Safi, S.Z.; Batumalaie, K.; Mansor, M.; Chinna, K.; Kumar, S.; Karimian, H.; Qvist, R.; Ashraf, M.A.; Yan, G.O. Glutamine treatment attenuates hyperglycemia-induced mitochondrial stress and apoptosis in umbilical vein endothelial cells. Clinics (Sao Paulo) 2015, 70, 569–576. [Google Scholar] [CrossRef]
- Parfenova, H.; Basuroy, S.; Bhattacharya, S.; Tcheranova, D.; Qu, Y.; Regan, R.F.; Leffler, C.W. Glutamate induces oxidative stress and apoptosis in cerebral vascular endothelial cells: Contributions of HO-1 and HO-2 to cytoprotection. Am. J. Physiol. Physiol. 2006, 290, C1399–C1410. [Google Scholar] [CrossRef]
- Hsu, C.S.; Chou, S.Y.; Liang, S.J.; Chang, C.Y.; Yeh, S.L. Effect of physiologic levels of glutamine on ICAM-1 expression in endothelial cells activated by preeclamptic plasma. J. Reprod. Med. 2006, 51, 193–198. [Google Scholar]
- Hou, Y.C.; Hsu, C.S.; Yeh, C.L.; Chiu, W.C.; Pai, M.H.; Yeh, S.L. Effects of glutamine on adhesion molecule expression and leukocyte transmigration in endothelial cells exposed to arsenic. J. Nutr. Biochem. 2005, 16, 700–704. [Google Scholar] [CrossRef]
- Su, S.T.; Yeh, C.L.; Hou, Y.C.; Pai, M.H.; Yeh, S.L. Dietary glutamine supplementation enhances endothelial progenitor cell mobilization in streptozotocin-induced diabetic mice subjected to limb ischemia. J. Nutr. Biochem. 2017, 40, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Pai, M.H.; Shih, Y.M.; Shih, J.M.; Yeh, C.L. Glutamine administration modulates endothelial progenitor cell and lung injury in septic mice. Shock 2016, 46, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Addabbo, F.; Chen, Q.; Patel, D.P.; Rabadi, M.; Ratliff, B.; Zhang, F.; Jasmin, J.F.; Wolin, M.; Lisanti, M.; Goligorsky, M.S. Glutamine supplementation alleviates vasculopathy and corrects metabolic profile in an in vivo model of endothelial cell dysfunction. PLoS ONE 2013, 8, e65458. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.C.; Patterson, M.; Dudenbostel, T.; Ccalhoun, D.; Gower, B. Effects of 6-month supplementation with β-hydroxy-β-methybutyrate, glutamine and arginine on vascular endothelial function of older adults. Eur. J. Clin. Nutr. 2016, 70, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Mitchell, J.A.; Swierkosz, T.A.; Sessa, W.C.; Vane, J.R. Inhibition by l-glutamine of the release of endothelium-derived relaxing factor from cultured endothelial cells. Br. J. Pharmacol. 1990, 101, 237–239. [Google Scholar] [CrossRef] [PubMed]
- Meininger, C.J.; Wu, G. L-glutamine inhibits nitric oxide synthesis in bovine venular endothelial cells. J. Pharmacol. Exp. Ther. 1997, 281, 448–453. [Google Scholar]
- Wu, G.; Haynes, T.E.; Li, H.; Yan, W.; Meininger, C.J. Glutamine metabolism to glucosamine is necessary for glutamine inhibition of endothelial nitric oxide synthase. Biochem. J. 2001, 353, 245–252. [Google Scholar] [CrossRef]
- Wu, G.; Haynes, T.E.; Yan, W.; Meininger, C.J. Presence of glutamine:fructose-6-phosphate amidotransferase for glucosamine-6-phosphate synthesis in endothelial cells: Effect of hyperglycemia and glutamine. Diabetologia 2001, 44, 196–202. [Google Scholar] [CrossRef]
- Leighton, B.; Curi, R.; Hussein, A.; Newsholme, E.A. Maximum activities of some key enzymes of glycolysis, glutaminolysis, Krebs cycle and fatty acid utilization in bovine pulmonary endothelial cells. FEBS Lett. 1987, 225, 93–96. [Google Scholar] [CrossRef]
- Wu, G.; Haynes, T.E.; Li, H.; Meininger, C.J. Glutamine metabolism in endothelial cells: Ornithine synthesis from glutamine via pyrroline-5-carboxylate synthase. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2000, 126, 115–123. [Google Scholar] [CrossRef]
- Liu, X.M.; Peyton, K.J.; Durante, W. Ammonia promotes endothelial cell survival via the heme oxygenase-1 mediated release of carbon monoxide. Free Radic. Biol. Med. 2017, 102, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Durante, W. Role of carbon monoxide in cardiovascular function. J. Cell. Mol. Med. 2006, 10, 672–686. [Google Scholar] [CrossRef] [PubMed]
- Durante, W.; Johnson, F.K.; Johnson, R.A. Targeting heme oxygenase-1 in vascular disease. Curr. Drug Targets 2010, 11, 1504–1516. [Google Scholar] [CrossRef] [PubMed]
- Durante, W. Protective role of heme oxygenase-1 against inflammation in atherosclerosis. Front. Biosci. 2011, 16, 2372. [Google Scholar] [CrossRef]
- Peyton, K.J.; Reyna, S.V.; Chapman, G.B.; Ensenat, D.; Liu, X.M.; Wang, H.; Schafer, A.I.; Durante, W. Heme oxygenase-1-derived carbon monoxide is an autocrine inhibitor of vascular smooth muscle cell growth. Blood 2002, 99, 4443–4448. [Google Scholar] [CrossRef] [PubMed]
- Peyton, K.J.; Shebib, A.R.; Azam, M.A.; Liu, X.-M.; Tulis, D.A.; Durante, W. Bilirubin inhibits neointima formation and vascular smooth muscle cell proliferation and migration. Front. Pharmacol. 2012, 3, 48. [Google Scholar] [CrossRef] [PubMed]
- Eng, C.H.; Yu, K.; Lucas, J.; White, E.; Abraham, R.T. Ammonia derived from glutaminolysis is a diffusible regulator of autophagy. Sci. Signal 2010, 3, ra31. [Google Scholar] [CrossRef] [PubMed]
- Abusneina, A.; Gauthier, E.R. Ammonium ions improve the survival of glutamine-starved hybridoma cells. Cell Biosci. 2016, 6, 23. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitch, M. Molecular pathogenesis of pulmonary arterial hypertension. J. Clin. Invest. 2012, 122, 4306–4313. [Google Scholar] [CrossRef]
- Chan, Y.; Loscalzo, J. Pathogenic mechanisms of pulmonary arterial hypertension. J. Mol. Cell. Cardiol. 2008, 44, 14–30. [Google Scholar] [CrossRef]
- Ge, J.; Cui, H.; Xie, N.; Banerjee, S.; Guo, S.; Dubey, S.; Barnes, S.; Liu, G. Glutminolysis promotes collagen translation and stability via α-ketoglutarate-mediate mTOR activation and proline hydroxylation. Am. J. Respir. Cell Mol. Biol. 2018, 58, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Piao, L.; Fang, Y.-H.; Parikh, K.; Ryan, J.J.; Toth, P.T.; Archer, S.L. Cardiac glutaminolysis: a maladaptive cancer metabolism pathway in the right ventricle in pulmonary hypertension. J. Mol. Med. 2013, 91, 1185–1197. [Google Scholar] [CrossRef] [PubMed]
- Egnatchik, R.A.; Brittain, E.L.; Shah, A.; Farres, W.H.; Ford, H.J.; Monahan, K.; Kang, C.J.; Kocurek, E.G.; Zhu, S.; Luong, T.; et al. Dysfunctional BMPR2 signaling drives an abnormal endothelial requirement for glutamine in pulmonary arterial hypertension. Pulm. Circ. 2017, 7, 186–199. [Google Scholar] [CrossRef] [PubMed]
- Zabot, G.P.; Carvalhal, G.F.; Marroni, N.P.; Hartmann, R.M.; Da Silva, V.D.; Fillmann, H.S. Glutamine prevents oxidative stress in a model of mesenteric ischemia and reperfusion. World J. Gastroenterol. 2014, 20, 11406–11414. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Suh, G.J.; Kwon, W.Y.; Lee, H.J.; Jeong, K.Y.; Jung, S.K.; Kwak, Y.H. The effect of glutamine on cerebral ischaemic injury after cardiac arrest. Resuscitation 2013, 84, 1285–1290. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.M.; Shih, J.M.; Pai, M.H.; Hou, Y.C.; Yeh, C.L.; Yeh, S.L. Glutamine administration after sublethal lower limb ischemia reduces inflammatory reaction and offers organ protection in ischemia/reperfusion injury. J. Parenter. Enter. Nutr. 2016, 40, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Prem, J.T.; Eppinger, M.; Lemmon, G.; Miller, S.; Nolan, D.; Peoples, J. The role of glutamine in skeletal muscle ischemia/reperfusion injury in the rat hind limb model. Am. J. Surg. 1999, 178, 147–150. [Google Scholar] [CrossRef]
- Zhang, S.C.; Shi, Q.; Feng, Y.N.; Fang, J. Tissue protective effect of glutamine on hepatic ischemia-reperfusion injury via the induction of heme oxygenase-1. Pharmacology 2013, 91, 59–68. [Google Scholar] [CrossRef]
- Esposito, E.; Mondello, S.; Di Paola, R.; Mazzon, E.; Italiano, D.; Paterniti, I.; Mondello, P.; Aloisi, C.; Cuzzocrea, S. Glutamine contributes to ameliorate inflammation after renal ischemia/reperfusion injury in rats. Naunyn Schmiedeberg’s Arch. Pharmacol. 2011, 383, 493–508. [Google Scholar] [CrossRef]
- Bolotin, G.; Raman, J.; Williams, U.; Bacha, E.; Kocherginsky, M.; Jeevanandam, V. Glutamine improves myocardial infarction following ischemia-reperfusion injury. Asian Cardiovasc. Thorac. Ann. 2007, 15, 463–467. [Google Scholar] [CrossRef]
- Pai, M.H.; Lei, C.S.; Su, S.T.; Yeh, S.L.; Hou, Y.C. Effects of dietary glutamine supplementation on immune cell polarization and muscle regeneration in diabetic mice with limb ischemia. Eur. J. Nutr. 2019, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Stangl, R.; Szijártó, A.; Onody, P.; Tamas, J.; Tátrai, M.; Hegedüs, V.; Blázovics, A.; Lotz, G.; Kiss, A.; Módis, K.; et al. Reduction of liver ischemia-reperfusion injury via glutamine pretreatment. J. Surg. Res. 2011, 166, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.L.; Li, Y.F.; Shan, H.M.; Wang, L.P.; Yuan, F.; Ma, Y.Y.; Li, W.L.; He, T.T.; Wang, Y.Y.; Qu, M.J.; et al. L-glutamine protects mouse brain from ischemic injury via up-regulating heat shock protein 70. CNS Neurosci. Ther. 2019, 25, 1030–1041. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zou, Z.; Li, Y.; Yuan, H.; Shi, X.Y.; Shi, X. Glutamine-induced heat shock protein protects against renal ischaemia-reperfusion injury in rats. Nephrology 2009, 14, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.L.; Niu, Q.; Shi, N.; Wang, J.; Jia, X.F.; Lian, H.F.; Liu, Z.; Liu, C.X. Glutamine ameliorates intestinal ischemia-reperfusion Injury in rats by activating the Nrf2/Are signaling pathway. Int. J. Clin. Exp. Pathol. 2015, 8, 7896–7904. [Google Scholar]
- Korthuis, R.J.; Durante, W. Heme oxygenase-1: A pluripotent sentinel limiting the systemic inflammatory response to extremity ischemia and reperfusion. Crit. Care Med. 2005, 33, 2701–2703. [Google Scholar] [CrossRef]
- Morris, C.R.; Suh, J.H.; Hagar, W.; Larkin, S.; Bland, D.A.; Steinberg, M.H.; Vichinsky, E.P.; Shigenaga, M.; Ames, B.; Kuypers, F.A.; et al. Erythrocyte glutamine depletion, altered redox environment, and pulmonary hypertension in sickle cell disease. Blood 2008, 111, 402–410. [Google Scholar] [CrossRef]
- Niihara, Y.; Matsui, N.M.; Shen, Y.M.; Akiyama, D.A.; Johnson, C.S.; Sunga, M.A.; Magpayo, J.; Embury, S.H.; Kalra, V.K.; Cho, S.H.; et al. L-Glutamine therapy reduces endothelial adhesion of sickle red blood cells to human umbilical vein endothelial cells. BMC Blood Disord. 2005, 5, 4. [Google Scholar] [CrossRef]
- Niihara, Y.; Miller, S.T.; Kanter, J.; Lanzkron, S.; Smith, W.R.; Hsu, L.L.; Gordeuk, V.R.; Viswanathan, K.; Sarnaik, S.; Osunkwo, I.; et al. A phase 3 clinical trial of l-glutamine in sickle cell disease. N. Engl. J. Med. 2018, 379, 226–235. [Google Scholar] [CrossRef]
- Cao, Y.; Kennedy, R.; Klimberg, V. Glutamine protects against doxorubicin-induced cardiotoxicity. J. Surg. Res. 1999, 85, 178–182. [Google Scholar] [CrossRef]
- Todorova, V.; Vanderpool, D.; Blossom, S.; Nwokedi, E.; Hennings, L.; Mrak, R.; Klimberg, V.S. Oral glutamine protects against cyclophosphamide-induced cardiotoxicity in experimental rats through increase of cardiac glutathione. Nutrition 2009, 25, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, Y.; Lv, S.J.; Wang, L.; Liang, G.P.; Wan, Q.X.; Peng, X. Effects of glutamine treatment on myocardial damage and cardiac function in rats after severe burn injury. Int. J. Clin. Exp. Pathol. 2012, 5, 651–659. [Google Scholar] [PubMed]
- Badole, S.L.; Jangam, G.B.; Chaudhari, S.M.; Ghule, A.E.; Zanwar, A.A. L-Glutamine supplementation prevents the development of experimental diabetic cardiomyopathy in streptozotocin-nicotinamide induced diabetic rats. PLoS ONE 2014, 9, e92697. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.; Huang, C.; Li, Z.; Yang, H.; Feng, Q. Effects of glutamine and valsartan on the brain natriuretic peptide and N-terminal pro-B-type natriuretic peptide of patients with chronic heart failure. Pak. J. Med. Sci. 2015, 31, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Kato, T.S.; Ji, R.; Zizola, C.; Brunjes, D.L.; Deng, Y.; Akashi, H.; Armstrong, H.F.; Kennel, P.J.; Thomas, T.; et al. Supplementation of l-alanyl-l-glutamine and fish oil improves body composition and quality of life in patients with chronic heart failure. Circ. Hear. Fail. 2015, 8, 1077–1087. [Google Scholar] [CrossRef] [PubMed]
- Hankard, R.G.; Darmaun, D.; Sager, B.K.; D’Amore, D.; Parsons, W.R.; Haymond, M. Response of glutamine metabolism to exogenous glutamine in humans. Am. J. Physiol. Metab. 1995, 269, E663–E670. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.E.; Marano, M.A.; Campbell, R.G. Splanchnic bed utilization of glutamine and glutamic acid in humans. Am. J. Physiol. Metab. 1993, 264, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, T.R.; Benfell, K.; Smith, R.J.; Young, L.S.; Brown, E.; Ferrari-Baliviera, E.; Lowe, D.K.; Wilmore, D.W. Safety and metabolic effects of l-glutamine administration in Humans. J. Parenter. Enter. Nutr. 1990, 14, 137S–146S. [Google Scholar] [CrossRef]
- Klassen, P.; Mazariegos, M.; Solomons, N.W.; Furst, P. The pharmacokinetic responses of humans to 20 g of alanyl-glutamine dipeptide differ with the dosing protocol but not with gastric acidity or in patients with acute dengue fever. J. Nutr. 2000, 130, 177–182. [Google Scholar] [CrossRef]
- Cruzat, V.F.; Rogero, M.M.; Tirapegui, J. Effects of supplementation with free glutamine and the dipeptide alanyl-glutamine on parameters of muscle damage and inflammation in rats submitted to prolonged exercise. Cell Biochem. Funct. 2010, 28, 24–30. [Google Scholar] [CrossRef]
- Adibi, S.A. Regulation of expression of the intestinal oligopeptide transporter (Pept-1) in health and disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.R.; Hamilton-Reeves, J.; Martindale, R.G.; Sarav, M.; Gautier, J.B.O. Acquired amino acid deficiencies: a focus on arginine and glutamine. Nutr. Clin. Pract. 2017, 32, 30S–47S. [Google Scholar] [CrossRef] [PubMed]
- Johnson, F.K.; Peyton, K.J.; Liu, X.M.; Azam, M.A.; Shebib, A.R.; Johnson, R.A.; Durante, W. Arginase promotes endothelial dysfunction and hypertension in obesity. Obesity 2015, 23, 383–390. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Wang, Z.; Cho, L.; Brennan, D.M.; Hazen, S.L. Diminished global arginine bioavailability and increased arginine catabolism as metabolic profile of increased cardiovascular risk. J. Am. Coll. Cardiol. 2009, 53, 2061–2067. [Google Scholar] [CrossRef] [PubMed]
- Heyland, D.; Muscedere, J.; Wischmeyer, P.E.; Cook, D.; Jones, G.; Albert, M.; Elke, G.; Berger, M.M.; Day, A.G. A randomized trial of glutamine and antioxidants in critical ill patients. N. Engl. J. Med. 2013, 368, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Van Zanten, A.R.; Sztark, F.; Kaisers, U.X.; Zielmann, S.; Felbinger, T.W.; Sablotzki, A.R.; De Waele, J.J.; Timsit, J.F.; Honing, M.L.; Keh, D.; et al. High-protein enteral nutrition enriched with immune-modulating nutrients vs standard high-protein enteral nutrition and nosocomial infections in the ICU: A randomized clinical trial. JAMA 2014, 312, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Motterlini, R.; Otterbein, L.E. The therapeutic potential of carbon monoxide. Nat. Rev. Drug Discov. 2010, 9, 728–743. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-H.; Choi, S. Therapeutic aspects of carbon monoxide in cardiovascular disease. Int. J. Mol. Sci. 2018, 19, 2381. [Google Scholar] [CrossRef]
- Yu, X.H.; Cui, L.B.; Wu, K.; Zheng, X.L.; Cayabyab, F.S.; Chen, Z.W.; Tang, C.K. Hydrogen sulfide as a potent cardiovascular protective agent. Clin. Chim. Acta 2014, 437, 78–87. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, Y.; Li, Y.; Li, L.; Xu, S.; Feng, X.; Liu, S. Hydrogen sulfide (H2S)-releasing compounds: therapeutic potential in cardiovascular diseases. Front. Pharmacol. 2018, 9, 1066. [Google Scholar] [CrossRef]
- Zimmerman, S.C.; Duvall, B.; Tsukamoto, T. Recent progress in the discovery of allosteric inhibitors of kidney type glutaminase. J. Med. Chem. 2019, 62, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.I.; Demo, S.D.; Dennison, J.B.; Chen, L.; Chernov-Rogan, T.; Goyal, B.; Janes, J.R.; Laidig, G.J.; Lewis, E.R.; Li, J.; et al. Antitumor activity of the glutaminase inhibitor CB-839 in triple-negative breast cancer. Mol. Cancer Ther. 2014, 13, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Park, K.G. Targeting glutamine metabolism for cancer treatment. Biomol. Ther. 2018, 26, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Korangath, P.; Teo, W.W.; Sadik, H.; Han, L.; Mori, N.; Huijts, C.M.; Wildes, F.; Bharti, S.; Zhang, Z.; Santa-Maria, C.A.; et al. Targeting glutamine metabolism in breast cancer with aminooxyacetate. Clin. Cancer Res. 2015, 21, 3263–3273. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durante, W. The Emerging Role of l-Glutamine in Cardiovascular Health and Disease. Nutrients 2019, 11, 2092. https://doi.org/10.3390/nu11092092
Durante W. The Emerging Role of l-Glutamine in Cardiovascular Health and Disease. Nutrients. 2019; 11(9):2092. https://doi.org/10.3390/nu11092092
Chicago/Turabian StyleDurante, William. 2019. "The Emerging Role of l-Glutamine in Cardiovascular Health and Disease" Nutrients 11, no. 9: 2092. https://doi.org/10.3390/nu11092092
APA StyleDurante, W. (2019). The Emerging Role of l-Glutamine in Cardiovascular Health and Disease. Nutrients, 11(9), 2092. https://doi.org/10.3390/nu11092092





