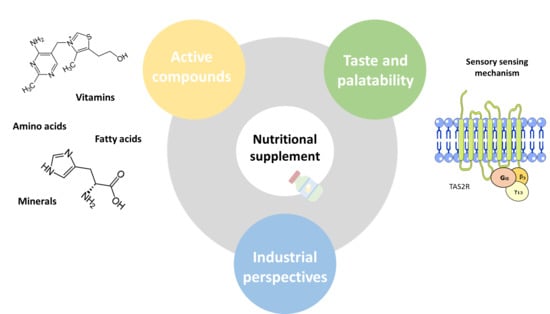
Taste Perception of Nutrients Found in Nutritional Supplements: A Review
Abstract
1. Introduction
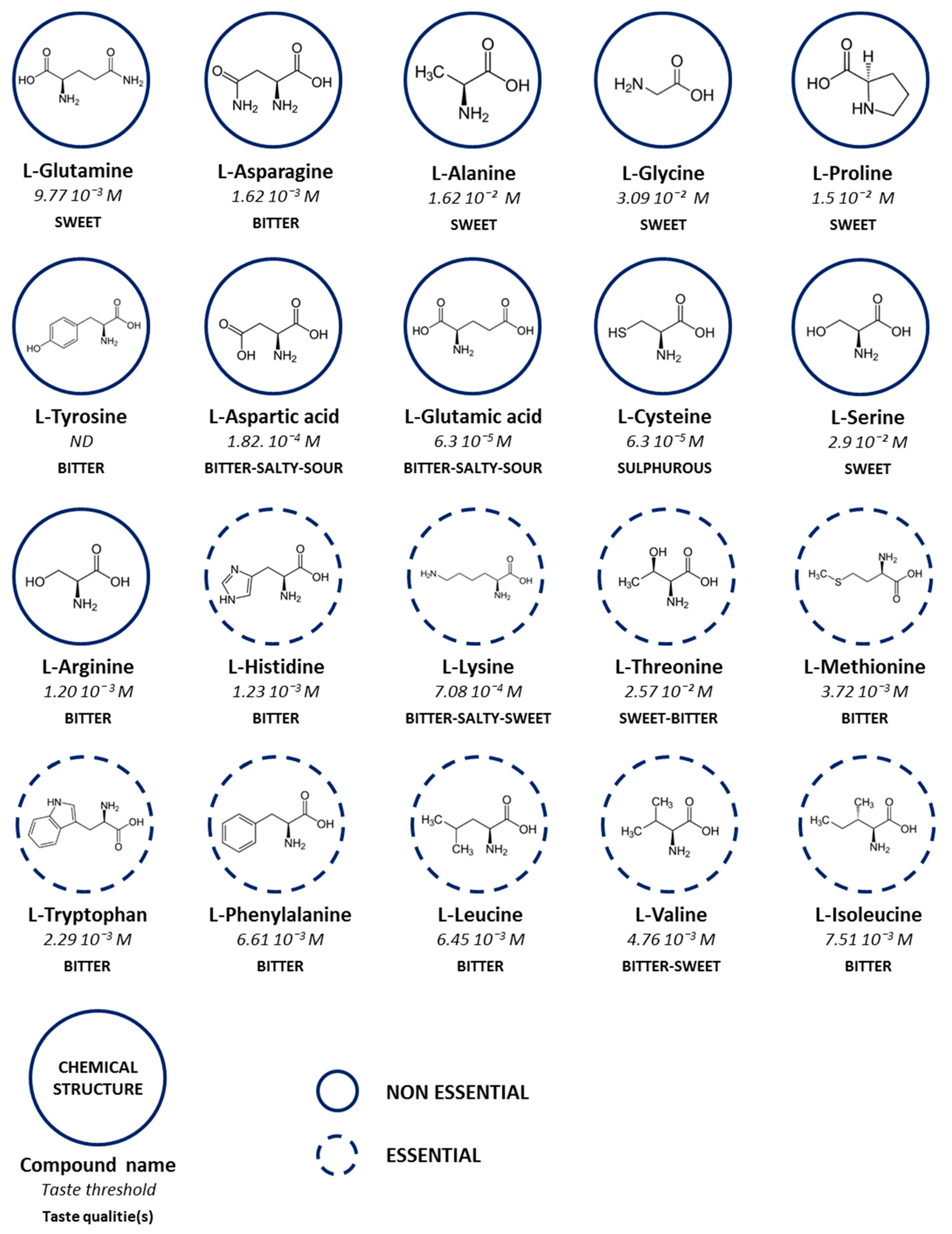
2. Taste of Amino Acids and Their Influence on Nutritional Supplement Taste
3. The Taste of Polyunsaturated Fatty Acids and Their Influence on Nutritional Supplement Taste
4. Taste of Minerals and Their Influence on Nutritional Supplement Taste
5. Taste of Vitamins and Their Influence on Nutritional Supplement Taste
6. Interactions Between Nutrients of Nutritional Supplements
7. Conclusion and Perspectives
Funding
Conflicts of Interest
References
- Bolton, J.; Shannon, L.; Smith, V.; Abbott, R.; Bell, S.J.; Stubbs, L.; Slevin, M.L. Comparison of short-term and long-term palatability of six commercially available oral supplements. J. Hum. Nutr. Diet. 1990, 3, 317–321. [Google Scholar] [CrossRef]
- Tisdale, M.J. Cancer cachexia: Metabolic alterations and clinical manifestations. Nutrition 1997, 13, 1–7. [Google Scholar] [CrossRef]
- Devi, A.; Chennakesavulu, S.; Suresh, C.; Reddy, A.B.M. Nutraceuticals and Their Role in Human Health and Disease. In Functional Food and Human Health; Rani, V., Yadav, U.C.S., Eds.; Springer: Singapore, 2018; pp. 375–403. [Google Scholar]
- Stratton, R.J. Summary of a systematic review on oral nutritional supplement use in the community. Proc. Nutr. Soc 2000, 59, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Van Thuyne, W.; Van Eenoo, P.; Delbeke, F.T. Nutritional supplements: Prevalence of use and contamination with doping agents. Nutr. Res. Rev. 2006, 19, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Thomas-Danguin, T. Flavor. In Encyclopedia of Neuroscience; Binder, M.D., Hirokawa, N., Windhorst, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1580–1582. [Google Scholar]
- Briand, L.; Salles, C. Taste perception and integration. In Flavor: From Food to Behaviors, Wellbeing and Health; Etiévant, P., Guichard, E., Salles, C., Voilley, A., Eds.; Elservier Ltd.: Cambridge, UK, 2016; pp. 101–119. [Google Scholar]
- Cayeux, I.; Starkenman, C. Sensory characterization of compounds with a trigeminal effect for taste modulation purposes. In Flavour, From Food to Perception; Guichard, E., Salles, C., Morzel, M., Le Bon, A.M., Eds.; Wiley Blackweel: Oxford, UK, 2016; pp. 192–207. [Google Scholar]
- Greer, B.K.; Woodard, J.L.; White, J.P.; Arguello, E.M.; Haymes, E.M. Branched-Chain Amino Acid Supplementation and Indicators of Muscle Damage after Endurance Exercise. Int. J. Sport. Nutr. Exerc. Metab. 2007, 17, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Miriel, V.; Bertun, E. Amino Acid Supplementation and Exercise Performance. Sports. Med. 1993, 16, 190–209. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Donohoe, R.T. The effects of nutrients on mood. Public. Health. Nutr. 1999, 2, 403–409. [Google Scholar] [CrossRef]
- Parker, G.; Brotchie, H. Mood effects of the amino acids tryptophan and tyrosine. Acta. Psychiatr. Scand. 2011, 124, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Yaqoob, P. Glutamine and the immune system. Amino Acids 1999, 17, 227–241. [Google Scholar] [CrossRef]
- Tu, Y.H.; Cooper, A.J.; Teng, B.; Chang, R.B.; Artiga, D.J.; Turner, H.N.; Mulhall, E.M.; Ye, W.; Smith, A.D.; Liman, E.R. An evolutionarily conserved gene family encodes proton-selective ion channels. Science 2018, 359, 1047–1050. [Google Scholar] [CrossRef]
- Bassoli, A.; Borgonovo, G.; Caremoli, F.; Mancuso, G. The taste of D and L-amino acids: In vitro binding assays with cloned human bitter (TAS2Rs) and sweet (TAS1R2/TAS1R3) receptors. Food Chem. 2014, 150, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Rhue, M.R.; Nishimura, T. Role of Free Amino Acids and Peptides in Food Taste. In Flavor Chemistry Trends and Developments; Teranishi, R., Buterry, R.G., Shahidi, F., Eds.; American chemical society: Washington, DC, UK, 1989; Volume 388, pp. 158–175. [Google Scholar]
- Pin, J.-P.; Kniazeff, J.; Binet, V.; Liu, J.; Maurel, D.; Galvez, T.; Duthey, B.; Havlickova, M.; Blahos, J.; Prézeau, L.; et al. Activation mechanism of the heterodimeric GABAB receptor. Biochem. Pharmacol. 2004, 68, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, S.S.; Sennewald, K.; Gagnon, J. Comparison of taste qualities and thresholds of D and L-amino acids. Physiol. Behav. 1981, 27, 51–59. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef]
- Nelson, G.; Chandrashekar, J.; Hoon, M.A.; Feng, L.; Zhao, G.; Ryba, N.J.; Zuker, C.S. An amino-acid taste receptor. Nature 2002, 416, 199–202. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Hoon, M.A.; Chandrashekar, J.; Zhang, Y.; Ryba, N.J.; Zuker, C.S. Mammalian sweet taste receptors. Cell 2001, 106, 381–390. [Google Scholar] [CrossRef]
- Meyerhof, W.; Batram, C.; Kuhn, C.; Brockhoff, A.; Chudoba, E.; Bufe, B.; Appendino, G.; Behrens, M. The molecular receptive ranges of human TAS2R bitter taste receptors. Chem. Senses 2010, 35, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Meyerhof, W. Elucidation of mammalian bitter taste. Rev. Physiol. Biochem. Pharmacol. 2005, 154, 37–72. [Google Scholar]
- Kohl, S.; Behrens, M.; Dunkel, A.; Hofmann, T.; Meyerhof, W. Amino acids and peptides activate at least five members of the human bitter taste receptor family. J. Agric. Food Chem. 2013, 61, 53–60. [Google Scholar] [CrossRef]
- Ueda, Y.; Yonemitsu, M.; Tsubuku, T.; Sakaguchi, M.; Miyajima, R. Flavor characteristics of glutathione in raw and cooked foodstuffs. Biosci. Biotechnol. Biochem. 1997, 61, 1977–1980. [Google Scholar] [CrossRef]
- Ohsu, T.; Amino, Y.; Nagasaki, H.; Yamanaka, T.; Takeshita, S.; Hatanaka, T.; Maruyama, Y.; Miyamura, N.; Eto, Y. Involvement of the calcium-sensing receptor in human taste perception. J. Biol. Chem. 2010, 285, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, A.S.; Uneyama, H.; Maekawa, T.; Torii, K. The calcium-sensing receptor in taste tissue. Biochem. Biophys. Res. Commun. 2009, 378, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Neyraud, E.; Palicki, O.; Schwartz, C.; Nicklaus, S.; Feron, G. Variability of human saliva composition: Possible relationships with fat perception and liking. Arch. Oral Biol. 2012, 57, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Fazzalari, F.A. Compilation of Odor and Taste Threshold Values Data; Stahl, W.H., Ed.; American Society for Testing and Materials: Philadelphia, PA, USA, 1973; Volume DS 48A, p. 250. [Google Scholar]
- Stephan, A.; Steinhart, H. Bitter taste of unsaturated free fatty acids in emulsions: Contribution to the off-flavour of soybean lecithins. Eur. Food Res. Technol. 2000, 212, 17–25. [Google Scholar] [CrossRef]
- Chale-Rush, A.; Burgess, J.R.; Mattes, R.D. Evidence for human orosensory (taste ?) sensitivity to free fatty acids. Chem. Senses 2007, 32, 423–431. [Google Scholar] [CrossRef]
- Running, C.A.; Mattes, R.D. Humans are more sensitive to the taste of linoleic and alpha-linolenic than oleic acid. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, G442–G449. [Google Scholar] [CrossRef] [PubMed]
- Running, C.A.; Mattes, R.D. Different oral sensitivities to and sensations of short-, medium-, and long-chain fatty acids in humans. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G381–G389. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.P.; Newman, L.P.; Keast, R.S.J. Effects of Salt and Fat Combinations on Taste Preference and Perception. Chem. Senses 2016, 41, 189–195. [Google Scholar] [CrossRef]
- Stewart, J.E.; Feinle-Bisset, C.; Golding, M.; Delahunty, C.; Clifton, P.M.; Keast, R.S.J. Oral sensitivity to fatty acids, food consumption and BMI in human subjects. Br. J. Nut. 2010, 104, 145–152. [Google Scholar] [CrossRef]
- Poette, J.; Mekoué, J.; Neyraud, E.; Berdeaux, O.; Renault, A.; Guichard, E.; Genot, C.; Feron, G. Fat sensitivity in humans: Oleic acid detection threshold is linked to saliva composition and oral volume. Flavour Fragr. J. 2014, 29, 39–49. [Google Scholar] [CrossRef]
- Neyraud, E.; Cabaret, S.; Brignot, H.; Chabanet, C.; Laboure, H.; Guichard, E.; Berdeaux, O. The basal free fatty acid concentration in human saliva is related to salivary lipolytic activity. Sci. Rep. 2017, 7, 5969. [Google Scholar] [CrossRef] [PubMed]
- Pepino, M.Y.; Love-Gregory, L.; Klein, S.; Abumrad, N.A. The fatty acid translocase gene CD36 and lingual lipase influence oral sensitivity to fat in obese subjects. J. Lipid Res. 2012, 53, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Heinze, J.M.; Preissl, H.; Fritsche, A.; Frank, S. Controversies in fat perception. Physiol. Behav. 2015, 152, 479–493. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Keast, R.S.J. Recent fat intake modulates fat taste sensitivity in lean and overweight subjects. Int. J. Obes. 2012, 36, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D. Oral thresholds and suprathreshold intensity ratings for free fatty acids on 3 tongue sites in humans: Implications for transduction mechanisms. Chem. Senses 2009, 34, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Besnard, P.; Passilly-Degrace, P.; Khan, N.A. Taste of fat: A sixth taste modality? Physiol. Rev. 2016, 96, 151–176. [Google Scholar] [CrossRef]
- Laugerette, F.; Passilly-Degrace, P.; Patris, B.; Niot, I.; Febbraio, M.; Montmayeur, J.P.; Besnard, P. CD36 involvement in orosensory detection of dietary lipids, spontaneous fat preference, and digestive secretions. J. Clin. Invest. 2005, 115, 3177–3184. [Google Scholar] [CrossRef] [PubMed]
- Sollai, G.; Melis, M.; Mastinu, M.; Pani, D.; Cosseddu, P.; Bonfiglio, A.; Crnjar, R.; Tepper, B.J.; Tomassini Barbarossa, I. Human Tongue Electrophysiological Response to Oleic Acid and Its Associations with PROP Taster Status and the CD36 Polymorphism (rs1761667). Nutrients 2019, 11, 315. [Google Scholar] [CrossRef] [PubMed]
- Tolonen, M. Minerals and trace elements. In Vitamins and Minerals in Health and Nutrition; Woodhead Publishing: Cambridge, UK, 1996; pp. 148–197. [Google Scholar]
- Saini, R.K.; Nile, S.H.; Keum, Y.-S. Food science and technology for management of iron deficiency in humans: A review. Trends Food. Sci. Tech. 2016, 53, 13–22. [Google Scholar] [CrossRef]
- Young, W.F.; Horth, H.; Crane, R.; Ogden, T.; Arnott, M. Taste and odour threshold concentrations of potential potable water contaminants. Water Res. 1996, 30, 331–340. [Google Scholar] [CrossRef]
- Lopez, P.; Perez-Rodriguez, I.; Estrany, F.; Devesa, R. Effects of sulfate and nitrate on the taste of water: A study with a trained panel. J. Water Supply Res. T. 2017, 66, 598–605. [Google Scholar] [CrossRef]
- Lim, J.; Lawless, H.T. Detection thresholds and taste qualities of iron salts. Food Qual. Prefer. 2006, 17, 513–521. [Google Scholar] [CrossRef]
- Epke, E.M.; Lawless, H.T. Retronasal smell and detection thresholds of iron and copper salts. Physiol. Behav. 2007, 92, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.M.; Kamphake, L.J.; Harris, E.K.; Woodward, R.L. Taste threshold concentrations of metals in drinking water. J. Am. Water Works Ass. 1960, 52, 660–670. [Google Scholar] [CrossRef]
- Vanderklaauw, N.J.; Smith, D.V. Taste quality profiles for 15 organic and inorganic salts. Physiol. Behav. 1995, 58, 295–306. [Google Scholar] [CrossRef]
- Ben Abu, N.; Harries, D.; Voet, H.; Niv, M.Y. The taste of KCl - What a difference a sugar makes. Food Chem. 2018, 255, 165–173. [Google Scholar] [CrossRef]
- Keast, R.S.J. The effect of zinc on human taste perception. J. Food Sci. 2003, 68, 1871–1877. [Google Scholar] [CrossRef]
- Omur-Ozbek, P.; Dietrich, A.M.; Duncan, S.E.; Lee, Y. Role of Lipid Oxidation, Chelating Agents, and Antioxidants in Metallic Flavor Development in the Oral Cavity. J. Agric. Food Chem. 2012, 60, 2274–2280. [Google Scholar] [CrossRef]
- Lawless, H.T.; Schlake, S.; Smythe, J.; Lim, J.Y.; Yang, H.D.; Chapman, K.; Bolton, B. Metallic taste and retronasal smell. Chem. Senses 2004, 29, 25–33. [Google Scholar] [CrossRef]
- Yang, H.H.L.; Lawless, H.T. Time-intensity characteristics of iron compounds. Food Qual. Prefer. 2006, 17, 337–343. [Google Scholar] [CrossRef]
- Zacarias, I.; Yanez, C.G.; Araya, M.; Oraka, C.; Olivares, M.; Uauy, R. Determination of the taste threshold of copper in water. Chem. Senses 2001, 26, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Pearson, W.N. Blood and urinary vitamin levels as potential indices of body stores. Am. J. Clin. Nutr. 1967, 20, 514–527. [Google Scholar] [CrossRef] [PubMed]
- Jacob, R.A.; Sotoudeh, G. Vitamin C function and status in chronic disease. Nutr. Clin. Care 2002, 5, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F.; McClung, J.P. The Vitamins, 5th ed.; Combs, G.F., McClung, J.P., Eds.; Academic Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Lathrop, L.B.; Clark, R.W. Masking the Taste of the Vitamin B Complex: A preliminary study. J. Am. Pharm. Assoc. 1950, 11, 90–94. [Google Scholar]
- Schiffman, S.S.; Dackis, C. Taste of nutrients: Amino acids, vitamins, and fatty acids. Percept. Psychophys. 1975, 17, 140–146. [Google Scholar] [CrossRef]
- Liu, K.; Jaggupilli, A.; Premnath, D.; Chelikani, P. Plasticity of the ligand binding pocket in the bitter taste receptor T2R7. Biochim. Biophys. Acta Biomembr. 2018, 1860, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Aboul-Enein, H.Y.; Loutfy, M.A. Pyridoxine Hydrochloride. In Analytical Profiles of Drug Substances; Florey, K., Ed.; Academic Press: Cambridge, MA, USA, 1984; Volume 13, pp. 447–486. [Google Scholar]
- Windholz, M.; Budavari, S.; Stroumtsos, L.Y.; Fertig, M.N. The Merck Index. An Encyclopedia of Chemicals and Drugs; Merck & Co.: Rahway, NJ, USA, 1976. [Google Scholar]
- Keast, R.S.J.; Breslin, P.A.S. An overview of binary taste–taste interactions. Food Qual. Prefer. 2003, 14, 111–124. [Google Scholar] [CrossRef]
- Iwasaki, K.; Sato, M. Inhibitory effects of some heavy-metal ions on taste nerve responses in mice. Jpn. J. Physiol. 1984, 34, 907–918. [Google Scholar] [CrossRef]
- Keast, R.S.J. Modification of the bitterness of caffeine. Food Qual. Prefer. 2008, 19, 465–472. [Google Scholar] [CrossRef]
- Keast, R.S.J.; Breslin, P.A.S. Bitterness suppression with zinc sulfate and Na-cyclamate: A model of combined peripheral and central neural approaches to flavor modification. Pharm. Res. 2005, 22, 1970–1977. [Google Scholar] [CrossRef]
- Keast, R.S.J.; Canty, T.M.; Breslin, P.A.S. Oral zinc sulfate solutions inhibit sweet taste perception. Chem. Senses 2004, 29, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Keast, R.S.J.; Breslin, P.A.S. Modifying the bitterness of selected oral pharmaceuticals with cation and anion series of salts. Pharm. Res. 2002, 19, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Behrens, M.; Redel, U.; Blank, K.; Meyerhof, W. The human bitter taste receptor TAS2R7 facilitates the detection of bitter salts. Biochem. Biophys. Res. Commun. 2019, 512, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zajac, A.L.; Lei, W.; Christensen, C.M.; Margolskee, R.F.; Bouysset, C.; Golebiowski, J.; Zhao, H.; Fiorucci, S.; Jiang, P. Metal Ions Activate the Human Taste Receptor TAS2R7. Chem. Senses 2019, 44, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Lawless, H.T.; Rapacki, F.; Horne, J.; Hayes, A.; Wang, G. The taste of calcium chloride in mixtures with NaCl, sucrose and citric acid. Food Qual. Prefer. 2004, 15, 83–89. [Google Scholar] [CrossRef]
- Koriyama, T.; Wongso, S.; Watanabe, K.; Abe, H. Fatty acid compositions of oil species affect the 5 basic taste perceptions. J. Food Sci. 2002, 67, 868–873. [Google Scholar] [CrossRef]
- Koriyama, T.; Kohata, T.; Watanabe, K.; Abe, H. Effects of docosahexaenoic acid content in triacylglycerol on human taste perception. J. Food Sci. 2002, 67, 2352–2356. [Google Scholar] [CrossRef]
- Ishida, K.; Yokoo, Y.; Koyama, Y. The influence of the addition of calcium pantothenate upon the taste of foods. J. Jpn. Soc. Food Nut. 1965, 17, 423–428. [Google Scholar]
- Challaner, C.A. Selecting excipients for controlled release. Pharm. Technol. 2018, 42, 24–26. [Google Scholar]
- Stevenson, R.J.; Prescott, J.; Boakes, R.A. Confusing tastes and smell: How odours can influence the perception of sweet and sour tastes. Chem. Senses 1999, 24, 627–635. [Google Scholar] [CrossRef]
 water-soluble vitamins,
water-soluble vitamins,  fat-soluble vitamins.
fat-soluble vitamins.
 water-soluble vitamins,
water-soluble vitamins,  fat-soluble vitamins.
fat-soluble vitamins.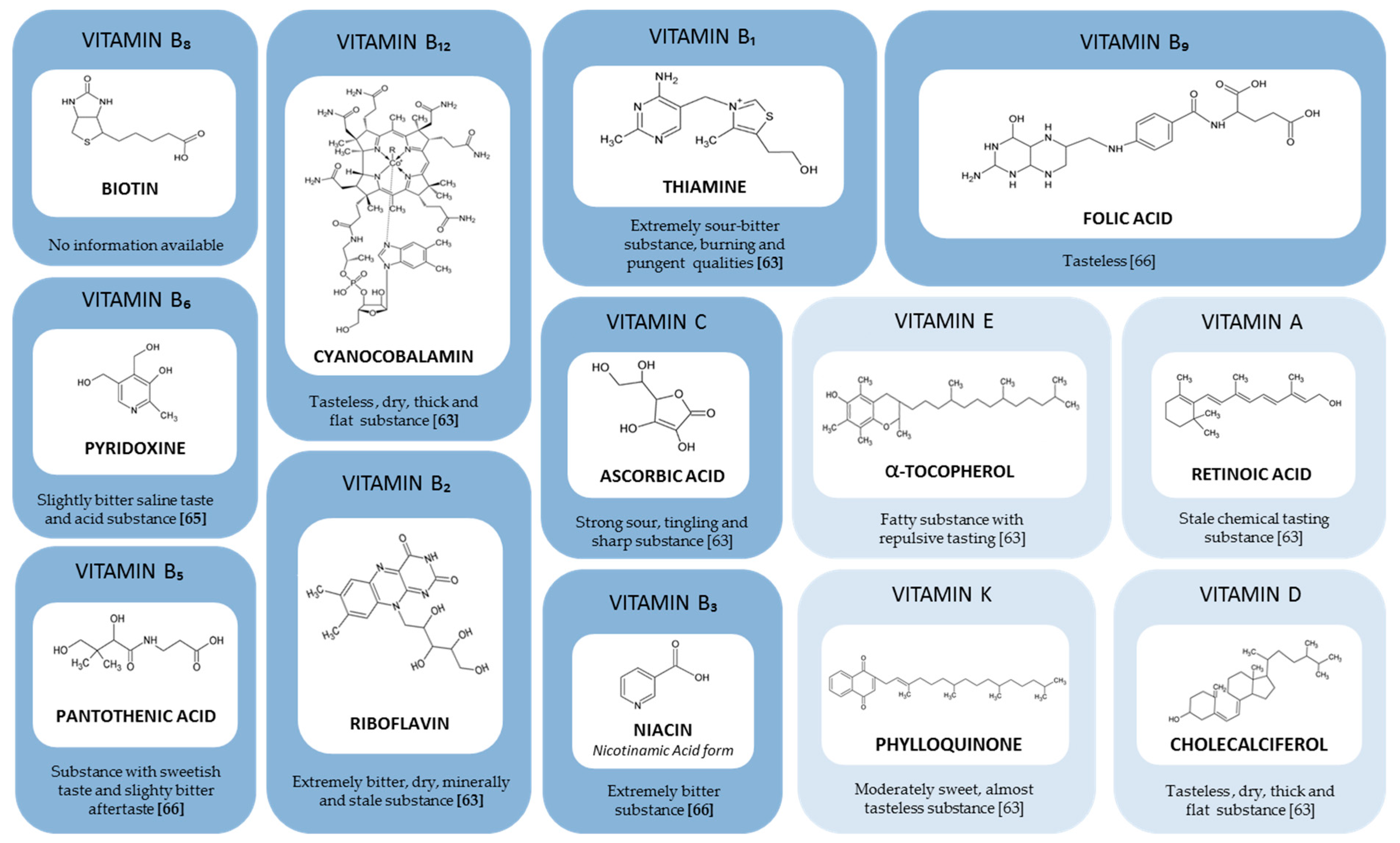
| Amino Acids | Threshold Concentration (mol/L) | Amino Acids Concentration (mol/L) | |
|---|---|---|---|
| l-Alanine | 1.62 × 10−2 | < | 1.72 × 10−2 |
| l-Arginine | 1.20 × 10−3 | < | 4.31 × 10−3 |
| l-Aspartic acid | 1.82 × 10−4 | < | 2.55 × 10−2 |
| l-Cysteine | 6.3 × 10−5 | < | 5.5 × 10−3 |
| l-Glutamic acid | 6.3 × 10−5 | < | 4.1 × 10−2 |
| l-Glycine | 3.09 × 10−2 | > | 6.3 × 10−3 |
| l-Histidine | 1.13 × 10−3 | < | 3.85 × 10−3 |
| l-Isoleucine | 7.51 × 10−3 | < | 1.5 × 10−2 |
| l-Leucine | 6.45 × 10−3 | < | 2.58 × 10−2 |
| l-Lysine | 7.08 × 10−4 | < | 2.07 × 10−2 |
| l-Methionine | 3.72 × 10−3 | < | 4.99 × 10−3 |
| l-Phenylalanine | 6.61 × 10−3 | < | 6.4 × 10−3 |
| l-Proline | 1.5 × 10−2 | < | 1.73 × 10−2 |
| l-Serine | 2.9 × 10−2 | > | 1.46 × 10−2 |
| l-Threonine | 2.57 × 10−3 | < | 1.72 × 10−2 |
| l-Tryptophan | 2.29 × 10−3 | > | 2.19 × 10−3 |
| l-Valine | 4.76 × 10−3 | < | 1.63 × 10−2 |
| TAS2R1 | TAS2R4 | TAS2R8 | TAS2R39 | TAS2R43 | TAS2R49 | |
|---|---|---|---|---|---|---|
| l-Phenylalanine | + | + | + | + | − | − |
| l-Tryptophan | + | + | − | + | + | + |
| d-Phenylalanine | + | − | − | + | − | − |
| d-Tryptophan | − | + | − | + | − | − |
| Formula | Name | Taste Qualities | Threshold Concentration (M) | Medium |
|---|---|---|---|---|
| C12H24O2 | Lauric acid | ND | 0.035 × 10−3 [29] | X |
| C14H28O2 | Myristic acid | ND | 0.22 × 10−3 [29] | X |
| C16H32O2 | Palmitic acid | ND | 3.9 × 10−5 [29] | X |
| C18H36O2 | Stearic acid | Irritant, metallic, bitter | 1.4 × 10−5 [30] | UHT milk 3.5% fat |
| ND | 0.01 × 10−3 [31] | Water, 5% gum acacia | ||
| ND | 0.05 × 10−3 [29] | Emulsion, 5% gum acacia | ||
| C18H34O2 | Oleic acid | ND | * 1.99 × 10−2 [32] | 10% gum arabic, 0.05% xanthan |
| Irritant, bitter, sour | * 2.57 × 10−2 [33] | 10% gum arabic, 0.05% xanthan | ||
| Astringent, bitter | 3.9 × 10−3 [30] | UHT milk 3.5% fat | ||
| ND | 2.0 × 10−2 [34] | Skim milk 5% gum acacia | ||
| ND | * 2.2 × 10−3 [35] | Non-fat milk | ||
| ND | * 3.5 × 10−3 [36] | 10% skimmed milk powder | ||
| ND | 2.23 × 10−3 [36] | 10% skimmed milk powder | ||
| ND | 0.78 × 10−3 [31] | Emulsion, 5% oil, 5% gum acacia | ||
| ND | 0.28 × 10−3 [29] | X | ||
| C18H32O2 | Linoleic acid | Metallic, astringent, bitter | 2.39 × 10−3 [30] | UHT milk 3.5% fat |
| ND | * 1.5 × 10−3 [35] | Non-fat milk | ||
| ND | 1.21 × 10−3 [31] | Emulsion, 5% oil, 5% gum acacia | ||
| ND | 1.55 × 10−3 [32] | 10% gum arabic, 0.05% xanthan | ||
| ND | 0.039 × 10−3 [29] | X | ||
| C18H30O2 | Alpha-linolenic acid | ND | * 3.15 × 10−3 [32] | 10% gum arabic, 0.05% xanthan |
| Irritant, metallic, bitter | 0.41 × 10−3 [30] | UHT milk 3.5% fat |
| Formula | Name | Taste Qualities | Threshold Concentration (M) | Medium |
|---|---|---|---|---|
| NaCl | Sodium chloride | Salty | * 8.0 × 10−3 [29] | Water |
| KCl | Potassium chloride | Salty, bitter, metallic | * 1.7 × 10−2 [29] | Water |
| CaCl2 | Calcium chloride | Bitter, salty | * 1.0 × 10−2 [29] | Water |
| MgCl2 | Magnesium chloride | Bitter | * 1.5 × 10−2 [29] | Water |
| MgSO4 | Magnesium sulphate | ND | * 4.6 × 10−3 [29] | Water |
| LiCl2 | Lithium chloride | Salty, sour | * 2.5 × 10−2 [29] | Water |
| NaI | Sodium iodide | ND | * 2.8 × 10−2 [29] | Water |
| CuSO4 | Copper sulphate | ND | * 6.2 × 10−6 [47] | Water |
| Na2SO4 | Sodium sulphate | Salty, bitter | * 1.7 × 10−3 [48] | Water |
| CaSO4 | Calcium sulphate | Salty, bitter | * 8.3 × 10−4 [48] | Water |
| Na2NO3 | Sodium nitrate | Untasty | * 1.6 × 10−3 [48] | Water |
| CaNO3 | Calcium nitrate | Untasty | * 1.6 × 10−3 [48] | Water |
| FeSO4 | Ferrous sulphate | ND | * 9.9 × 10−5 [49] | Deionized water |
| FeSO4 | Ferrous sulphate | Metallic | * 3.0 × 10−5 [50] | Deionized water |
| FeSO4 | Ferrous sulphate | ND | 1.6 × 10−4 [50] | Deionized water |
| FeCl2 | Ferrous chloride | ND | * 6.6 × 10−5 [49] | Deionized water |
| FeCl2 | Ferrous chloride | ND | * 6.4 × 10−5 [50] | Deionized water |
| FeCl2 | Ferrous chloride | ND | 2.27 × 10−4 [50] | Deionized water |
| CuSO4 | Copper sulphate | Bitter, astringent, metallic | * 7.8 × 10−6 [50] | Deionized water |
| CuSO4 | Copper sulphate | Bitter, astringent, metallic | 2.46 × 10−5 [50] | Deionized water |
| CuCl2 | Copper chloride | ND | * 8.2 × 10−6 [50] | Deionized water |
| CuCl2 | Copper chloride | ND | 1.56 × 10−5 [50] | Deionized water |
| Formula | Name | Taste Qualities | Threshold Concentration (mol cation/L) | Medium |
|---|---|---|---|---|
| ZnSO4 | Zinc sulphate | ND | * 4.12 × 10−4 | Spring water |
| ZnSO4 | Zinc sulphate | ND | * 2.75 × 10−4 | Distilled water |
| ZnNO3 | Zinc nitrate | ND | * 3.36 × 10−4 | Distilled water |
| ZnCl2 | Zinc chloride | ND | * 4.12 × 10−4 | Spring water |
| ZnCl2 | Zinc chloride | ND | * 5.04 × 10−4 | Distilled water |
| CuCl2 | Copper chloride | ND | * 2.04 × 10−4 | Spring water |
| CuCl2 | Copper chloride | ND | * 1.03 × 10−4 | Distilled water |
| FeSO4 | Ferrous sulphate | ND | * 3.21 × 10−5 | Spring water |
| FeSO4 | Ferrous sulphate | ND | * 6.07 × 10−5 | Distilled water |
| Fe2H204 | Hydrous ferric oxide | ND | * 1.57 × 10−4 | Distilled water |
| MnSO4 | Manganese sulphate | ND | * 8.19 × 10−4 | Distilled water |
| CuSO4 | Copper sulphate | ND | * 3.78 × 10−5 | Distilled deionized water |
| CuSO4 | Copper sulphate | ND | * 5.51 × 10−5 | Uncarbonated water |
| CuCl2 | Copper chloride | ND | * 3.94 × 10−5 | Distilled deionized water |
| CuCl2 | Copper chloride | ND | * 5.98 × 10−5 | Uncarbonated water |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delompré, T.; Guichard, E.; Briand, L.; Salles, C. Taste Perception of Nutrients Found in Nutritional Supplements: A Review. Nutrients 2019, 11, 2050. https://doi.org/10.3390/nu11092050
Delompré T, Guichard E, Briand L, Salles C. Taste Perception of Nutrients Found in Nutritional Supplements: A Review. Nutrients. 2019; 11(9):2050. https://doi.org/10.3390/nu11092050
Chicago/Turabian StyleDelompré, Thomas, Elisabeth Guichard, Loïc Briand, and Christian Salles. 2019. "Taste Perception of Nutrients Found in Nutritional Supplements: A Review" Nutrients 11, no. 9: 2050. https://doi.org/10.3390/nu11092050
APA StyleDelompré, T., Guichard, E., Briand, L., & Salles, C. (2019). Taste Perception of Nutrients Found in Nutritional Supplements: A Review. Nutrients, 11(9), 2050. https://doi.org/10.3390/nu11092050