How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children
Abstract
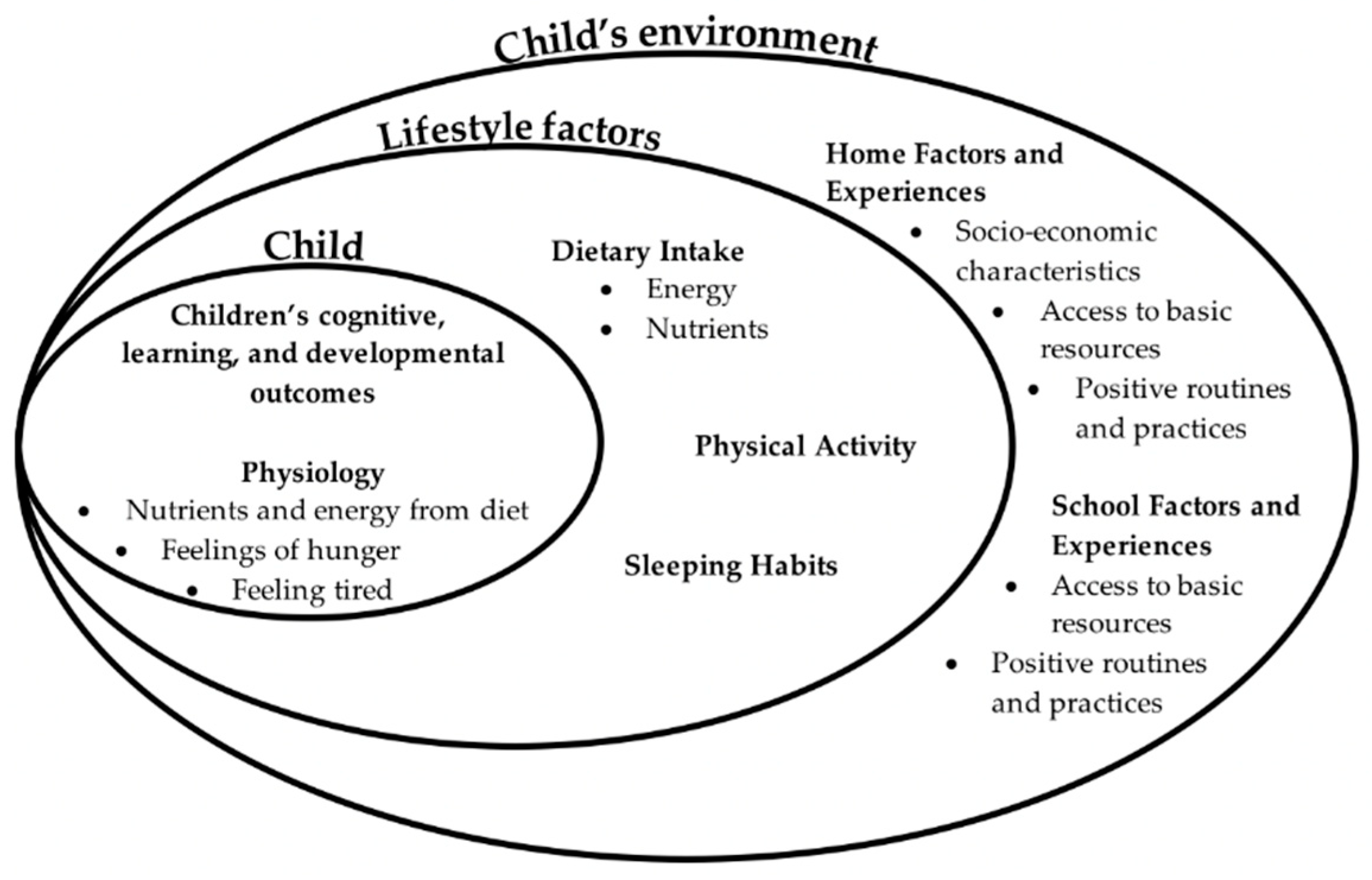
1. Introduction
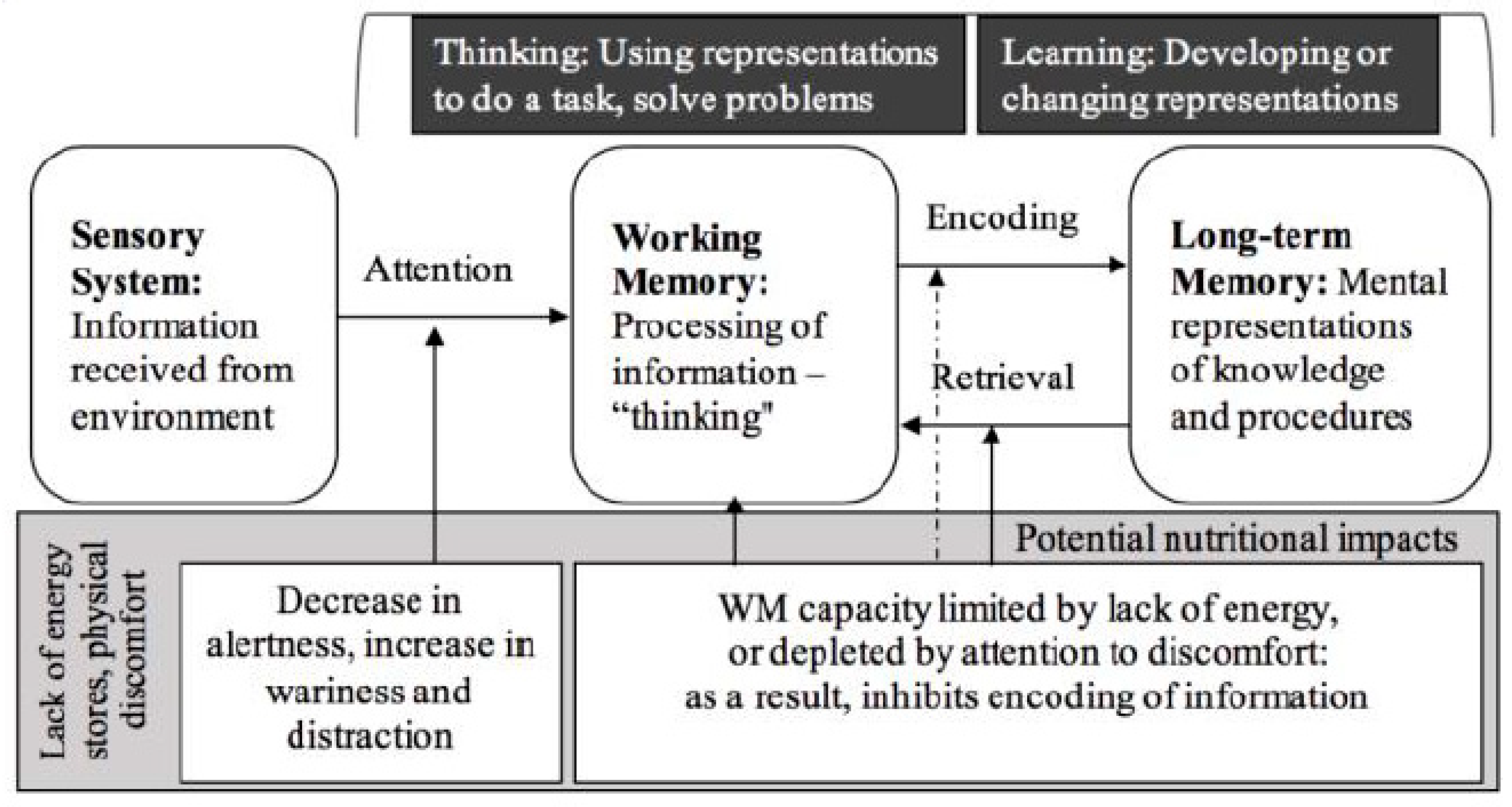
1.1. Children’s Cognition and Learning
1.2. Diet and Nutrition
1.3. Power Supply for Learning: Blood Glucose Availability
1.4. Glucose in the Brain
1.5. Nutrition and the Brain: Cognitive Processes and Learning
1.6. Nutritients and Brain Development
2. Physical Activity (PA) and Fitness
2.1. Physical Activity for Enhancing Cognition and Learning in Children
2.2. Improved Aerobic Fitness for Enhancing Cognition and Learning in Children
2.3. Role of Exercise Intensity
2.4. Interaction between Exercise Intensity and Cognitively Engaging Activities
2.5. The Interaction between Nutrition and Physical Activity for Enhancing Cognition and Learning in Children
3. The Role of Sleep on Cognition, Learning, and Development
4. The Role of Family Socioeconomic Status in Children’s Nutrition and Learning: Examining the Pathways
4.1. Considering Socio-Economic Status in Relation to Children’s Nutrition and Learning
4.2. How Are Young Children Around the World Faring with Regard to Nutrition and Learning?
4.3. Potential Pathways: SES and Access to Basic Resources
4.4. Potential Pathways: SES and Children’s Home Experiences
4.5. Potential Pathways: SES and Children’s Early Care and Education Experiences
5. Application and Practice Example
5.1. School Nutrition, Cognition and Learning
5.2. Specific Nutritional Components and Specific Cognitive Processes
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Academies of Sciences, Engineering, and Medicine. How People Learn II: Learners, Contexts, and Cultures; National Academies Press: Washington, DC, USA, 2018. [Google Scholar]
- Atkinson, R.; Shiffrin, R. Human Memory: A Proposed System and its Control Processes. Psychol. Learn. Motiv. 1968, 2, 89–195. [Google Scholar]
- Simon, H.A. Information processing models of cognition. Annu. Rev. Psychol. 1979, 30, 363–396. [Google Scholar] [CrossRef]
- Kemps, E.; Tiggemann, M.; Grigg, M. Food cravings consume limited cognitive resources. J. Exp. Psychol. Appl. 2008, 14, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Hoyland, A.; Dye, L.; Lawton, C.L. A systematic review of the effect of breakfast on the cognitive performance of children and adolescents. Nutr. Res. Rev. 2009, 22, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Hardman, R.J.; Kennedy, G.; Macpherson, H.; Scholey, A.B.; Pipingas, A. Adherence to a Mediterranean-Style Diet and Effects on Cognition in Adults: A Qualitative Evaluation and Systematic Review of Longitudinal and Prospective Trials. Front. Nutr. 2016, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Reichelt, A.C.; Stoeckel, L.E.; Reagan, L.P.; Winstanley, C.A.; Page, K.A. Dietary influences on cognition. Physiol. Behav. 2018, 192, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Monti, J.M.; Baym, C.L.; Cohen, N.J. Identifying and Characterizing the Effects of Nutrition on Hippocampal Memory. Adv. Nutr. 2014, 5, 337S–343S. [Google Scholar] [CrossRef]
- Kelsey, C.; Dreisbach, C.; Alhusen, J.; Grossmann, T. A primer on investigating the role of the microbiome in brain and cognitive development. Dev. Psychobiol. 2018, 61, 341–349. [Google Scholar] [CrossRef]
- Murakami, K.; McCaffrey, T.A.; Livingstone, M.B.E. Dietary glycaemic index and glycaemic load in relation to food and nutrient intake and indices of body fatness in British children and adolescents. Br. J. Nutr. 2013, 110, 1512–1523. [Google Scholar] [CrossRef]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef]
- Mergenthaler, P.; Lindauer, U.; Dienel, G.A.; Meisel, A. Sugar for the brain: The role of glucose in physiological and pathological brain function. Trends Neurosci. 2013, 36, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Camps, S.G.; Lim, J.; Ishikado, A.; Inaba, Y.; Suwa, M.; Matsumoto, M.; Henry, C.J. Co-Ingestion of Rice Bran Soymilk or Plain Soymilk with White Bread: Effects on the Glycemic and Insulinemic Response. Nutrients 2018, 10, 449. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.; Matthan, N.R.; Ausman, L.M.; Lichtenstein, A.H. Effect of prior meal macronutrient composition on postprandial glycemic responses and glycemic index and glycemic load value determinations. Am. J. Clin. Nutr. 2017, 106, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.S.; Jenkins, A.L.; Vuksan, V.; Campbell, J. The glycaemic index values of foods containing fructose are affected by metabolic differences between subjects. Eur. J. Clin. Nutr. 2009, 63, 1106–1114. [Google Scholar] [CrossRef] [PubMed]
- Soh, N.L.; Brand-Miller, J. The glycaemic index of potatoes: The effect of variety, cooking method and maturity. Eur. J. Clin. Nutr. 1999, 53, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.L.; Gellar, L.; Nathanson, B.H.; Pbert, L.; Ma, Y.; Ockene, I.; Rosal, M.C. Decrease in Glycemic Index Associated with Improved Glycemic Control among Latinos with Type 2 Diabetes. J. Acad. Nutr. Diet. 2015, 115, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Monro, J.A.; Shaw, M. Glycemic impact, glycemic glucose equivalents, glycemic index, and glycemic load: Definitions, distinctions, and implications. Am. J. Clin. Nutr. 2008, 87, 237S–243S. [Google Scholar] [CrossRef] [PubMed]
- Augustin, L.S.; Kendall, C.W.; Jenkins, D.J.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Bjorck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef]
- Dienel, G.A. Brain Glucose Metabolism: Integration of Energetics with Function. Physiol. Rev. 2019, 99, 949–1045. [Google Scholar] [CrossRef]
- Han, P.; Bagenna, B.; Fu, M. The sweet taste signalling pathways in the oral cavity and the gastrointestinal tract affect human appetite and food intake: A review. Int. J. Food Sci. Nutr. 2019, 70, 125–135. [Google Scholar] [CrossRef]
- Olszewski, P.K.; Wood, E.L.; Klockars, A.; Levine, A.S. Excessive Consumption of Sugar: An Insatiable Drive for Reward. Curr. Nutr. Rep. 2019, 8, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Killen, S.S.; Marras, S.; McKenzie, D.J. Fuel, fasting, fear: Routine metabolic rate and food deprivation exert synergistic effects on risk-taking in individual juvenile European sea bass. J. Anim. Ecol. 2011, 80, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.W.; Woods, S.C.; Porte, D., Jr.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Cheon, B.; Sim, A.; Lee, L.; Forde, C. Avoiding hunger or attaining fullness? Implicit goals of satiety guide portion selection and food intake patterns. Appetite 2019, 138, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Vilela, S.; Hetherington, M.M.; Oliveira, A.; Lopes, C. Tracking diet variety in childhood and its association with eating behaviours related to appetite: The generation XXI birth cohort. Appetite 2018, 123, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Paterson, C.; Lacroix, E.; von Ranson, K.M. Conceptualizing addictive-like eating: A qualitative analysis. Appetite 2019, 141, 104326. [Google Scholar] [CrossRef] [PubMed]
- Willem, C.; Gandolphe, M.-C.; Roussel, M.; Verkindt, H.; Pattou, F.; Nandrino, J.-L. Difficulties in emotion regulation and deficits in interoceptive awareness in moderate and severe obesity. Eat. Weight Disord. Stud. Anorex. Bulim. Obes. 2019, 24, 633–644. [Google Scholar] [CrossRef]
- Rhee, K.E.; Kessl, S.; Manzano, M.A.; Strong, D.R.; Boutelle, K.N. Cluster randomized control trial promoting child self-regulation around energy-dense food. Appetite 2019, 133, 156–165. [Google Scholar] [CrossRef]
- De Lauzon-Guillain, B.; Koudou, Y.A.; Botton, J.; Forhan, A.; Carles, S.; Pelloux, V.; Clement, K.; Ong, K.K.; Charles, M.A.; Heude, B.; et al. Association between genetic obesity susceptibility and mother-reported eating behaviour in children up to 5 years. Pediatr. Obes. 2019, 14, e12496. [Google Scholar] [CrossRef]
- Kamijo, K.; Pontifex, M.B.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Evans, E.M.; Castelli, D.M.; Hillman, C.H. The association of childhood obesity to neuroelectric indices of inhibition. Psychophysiology 2012, 49, 1361–1371. [Google Scholar] [CrossRef]
- Kamijo, K.; Khan, N.A.; Pontifex, M.B.; Scudder, M.R.; Drollette, E.S.; Raine, L.B.; Evans, E.M.; Castelli, D.M.; Hillman, C.H. The Relation of Adiposity to Cognitive Control and Scholastic Achievement in Preadolescent Children. Obesity 2012, 20, 2406–2411. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Dai, Q.; Jackson, J.C.; Zhang, J. Overweight Is Associated with Decreased Cognitive Functioning among School-age Children and Adolescents. Obesity 2008, 16, 1809–1815. [Google Scholar] [CrossRef]
- Kranz, S.; Brauchla, M.; Campbell, W.W.; Mattes, R.D.; Schwichtenberg, A.J. High-Protein and High-Dietary Fiber Breakfasts Result in Equal Feelings of Fullness and Better Diet Quality in Low-Income Preschoolers Compared with Their Usual Breakfast. J. Nutr. 2017, 147, 445–452. [Google Scholar] [CrossRef] [PubMed]
- De Lange, A.G.; Brathen, A.C.S.; Rohani, D.A.; Fjell, A.M.; Walhovd, K.B. The Temporal Dynamics of Brain Plasticity in Aging. Cereb. Cortex 2018, 28, 1857–1865. [Google Scholar] [PubMed]
- Clarke, D.D.; Sokoloff, L. Circulation and energy metabolism of the brain. In Basic Neurochemistry: Molecular, Cellular, and Medical Aspects; Siegel, G., Aranoff, B., Albers, R., Eds.; Lippincott-Raven: Philadelphia, PA, USA, 1999. [Google Scholar]
- Bryan, J.; Osendarp, S.; Hughes, D.; Calvaresi, E.; Baghurst, K.; Van Klinken, J.-W.; Klinken, J.-W.; Klinken, J. Nutrients for Cognitive Development in School-aged Children. Nutr. Rev. 2004, 62, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Derbyshire, E. Brain Health across the Lifespan: A Systematic Review on the Role of Omega-3 Fatty Acid Supplements. Nutrients 2018, 10, 1094. [Google Scholar] [CrossRef] [PubMed]
- Achanta, L.B.; Rowlands, B.D.; Thomas, D.S.; Housley, G.D.; Rae, C.D. Beta-Hydroxybutyrate Boosts Mitochondrial and Neuronal Metabolism but is not Preferred Over Glucose Under Activated Conditions. Neurochem. Res. 2017, 42, 1710–1723. [Google Scholar] [CrossRef]
- Sunram-Lea, S.I.; Owen, L. The impact of diet-based glycaemic response and glucose regulation on cognition: Evidence across the lifespan. Proc. Nutr. Soc. 2017, 76, 466–477. [Google Scholar] [CrossRef]
- Adeva-Andany, M.M.; Gonzalez-Lucan, M.; Donapetry-Garcia, C.; Fernandez-Fernandez, C.; Ameneiros-Rodriguez, E. Glycogen metabolism in humans. BBA Clin. 2016, 5, 85–100. [Google Scholar] [CrossRef]
- Settergren, G.; Lindblad, B.S.; Persson, B. Cerebral blood flow and exchange of oxygen, glucose, ketone bodies, lactate, pyruvate and amino acids in infants. Acta Paediatr. Scand. 1976, 65, 343–353. [Google Scholar] [CrossRef]
- Nugent, S.; Tremblay, S.; Chen, K.W.; Ayutyanont, N.; Roontiva, A.; Castellano, C.-A.; Fortier, M.; Roy, M.; Courchesne-Loyer, A.; Bocti, C.; et al. Brain glucose and acetoacetate metabolism: A comparison of young and older adults. Neurobiol. Aging 2014, 35, 1386–1395. [Google Scholar] [CrossRef] [PubMed]
- Croteau, E.; Castellano, C.A.; Fortier, M.; Bocti, C.; Fulop, T.; Paquet, N.; Cunnane, S.C. A cross-sectional comparison of brain glucose and ketone metabolism in cognitively healthy older adults, mild cognitive impairment and early Alzheimer’s disease. Exp. Gerontol. 2018, 107, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Ingwersen, J.; Defeyter, M.A.; Kennedy, D.O.; Wesnes, K.A.; Scholey, A.B. A low glycaemic index breakfast cereal preferentially prevents children’s cognitive performance from declining throughout the morning. Appetite 2007, 49, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Brindal, E.; Baird, D.; Slater, A.; Danthiir, V.; Wilson, C.; Bowen, J.; Noakes, M. The effect of beverages varying in glycaemic load on postprandial glucose responses, appetite and cognition in 10-12-year-old school children. Br. J. Nutr. 2013, 110, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Tabet, F.; Cochran, B.J.; Torres, L.F.C.; Wu, B.J.; Barter, P.J.; Rye, K.-A. Apolipoprotein A-I enhances insulin-dependent and insulin-independent glucose uptake by skeletal muscle. Sci. Rep. 2019, 9, 1350. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Richards, E.M.; Raizada, M.K.; Sumners, C. Angiotensin II increases glucose uptake and glucose transporter-1 mRNA levels in astroglia. Am. J. Physiol. Metab. 1995, 268, E384–E390. [Google Scholar] [CrossRef] [PubMed]
- Taki, Y.; Hashizume, H.; Sassa, Y.; Takeuchi, H.; Asano, M.; Asano, K.; Kawashima, R. Breakfast Staple Types Affect Brain Gray Matter Volume and Cognitive Function in Healthy Children. PLoS ONE 2010, 5, e15213. [Google Scholar] [CrossRef] [PubMed]
- Micha, R.; Rogers, P.J.; Nelson, M. Glycaemic index and glycaemic load of breakfast predict cognitive function and mood in school children: A randomised controlled trial. Br. J. Nutr. 2011, 106, 1552–1561. [Google Scholar] [CrossRef]
- Cooper, S.B.; Bandelow, S.; Nute, M.L.; Morris, J.G.; Nevill, M.E. Breakfast glycaemic index and cognitive function in adolescent school children. Br. J. Nutr. 2012, 107, 1823–1832. [Google Scholar] [CrossRef]
- Ludwig, D.S.; Majzoub, J.A.; Al-Zahrani, A.; Dallal, G.E.; Blanco, I.; Roberts, S.B. High Glycemic Index Foods, Overeating, and Obesity. Pediatrics 1999, 103, E26. [Google Scholar] [CrossRef]
- Rouhani, M.H.; Salehi-Abargouei, A.; Azadbakht, L. Effect of glycemic index and glycemic load on energy intake in children. Nutrients 2013, 29, 1100–1105. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.B.; Bandelow, S.; Nevill, M.E. Breakfast consumption and cognitive function in adolescent schoolchildren. Physiol. Behav. 2011, 103, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Parillo, M.; Licenziati, M.R.; Vacca, M.; De Marco, D.; Iannuzzi, A. Metabolic changes after a hypocaloric, low-glycemic-index diet in obese children. J. Endocrinol. Investig. 2012, 35, 629–633. [Google Scholar]
- Lacombe, A.; Ganji, V. Influence of two breakfast meals differing in glycemic load on satiety, hunger, and energy intake in preschool children. Nutr. J. 2010, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Warren, J.M.; Henry, C.J.; Simonite, V. Low glycemic index breakfasts and reduced food intake in preadolescent children. Pediatrics 2003, 112, e414. [Google Scholar] [CrossRef] [PubMed]
- USDA. Updated Child and Adult Care Food Program Meal Patterns: Child and Adult Meals; United States Department of Agriculture: Washington, DC, USA, 2017.
- Tonkiss, J.; Galler, J.; Morgane, P.J.; Bronzino, J.D.; Austin-LaFrance, R.J. Prenatal protein malnutrition and postnatal brain function. Ann. N. Y. Acad. Sci. 1993, 678, 215–227. [Google Scholar] [CrossRef]
- Strupp, B.J.; Levitsky, D.A. Enduring cognitive effects of early malnutrition: A theoretical reappraisal. J. Nutr. 1995, 125, 2221S–2232S. [Google Scholar] [CrossRef]
- Bellisle, F. Effects of diet on behaviour and cognition in children. Br. J. Nutr. 2004, 92 (Suppl. 2), S227–S232. [Google Scholar] [CrossRef]
- Hassevoort, K.M.; Khan, N.A.; Hillman, C.H.; Cohen, N.J. Childhood Markers of Health Behavior Relate to Hippocampal Health, Memory, and Academic Performance. Mind Brain Educ. 2016, 10, 162–170. [Google Scholar] [CrossRef]
- Smart, J.L.; Adlard, B.P.; Dobbing, J. Further studies of body growth and brain development in “small-for-dates” rats. Biol. Neonate 1974, 25, 135–150. [Google Scholar] [CrossRef]
- Morgane, P.J.; Austin-LaFrance, R.; Bronzino, J.; Tonkiss, J.; Díaz-Cintra, S.; Cintra, L.; Kemper, T.; Galler, J.R.; Kemper, T. Prenatal malnutrition and development of the brain. Neurosci. Biobehav. Rev. 1993, 17, 91–128. [Google Scholar] [CrossRef]
- Winick, M.; Rosso, P. Head circumference and cellular growth of the brain in normal and marasmic children. J. Pediatr. 1969, 74, 774–778. [Google Scholar] [CrossRef]
- Dickerson, J.W.T. Nutrition, brain growth and development. In Maturation and Development: Biological and Psychological Perspectives KK Connoly and HFR Prechtl; The Lavenham Press: Suffolk, UK, 1981; pp. 110–130. [Google Scholar]
- Morgan, B.L.G.; Winick, M. Pathologic effects of malnutrition on the central nervous system. In Nutritional Pathology: Pathobiochemistry of Dietary Imbalances; Sidransky, H., Ed.; Dekker: New York, NY, USA, 1985; pp. 161–206. [Google Scholar]
- Winick, M. Malnutrition and Brain Development; Oxfrod University Press: New York, NY, USA, 1976. [Google Scholar]
- Nokes, C.C.; van den, B.; Bundy, D. The Effects of Iron Deficiency and Anemia on Mental and Motor Performance, Educational Achievement, and Behavior in Children; International Nutritional Anemia Consulting Group: Washington, DC, USA, 1998. [Google Scholar]
- Honig, A.S.; Oski, F.A. Solemnity: A clinical risk index for iron deficient infants. Early Child Dev. 1984, 16, 16–84. [Google Scholar] [CrossRef]
- Lozoff, B.; Wolf, A.W.; Urrutia, J.J.; Viteri, F.E. Abnormal Behavior and Low Developmental Test Scores in Iron-Deficient Anemic Infants. J. Dev. Behav. Pediatr. 1985, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Klein, N.K.; Nelson, E.C.; McClish, D.K.; Manuel, M.; Chacon, M.E. Behavior of Infants with Iron-Deficiency Anemia. Child Dev. 1998, 69, 24–36. [Google Scholar] [CrossRef]
- Lozoff, B.; Wolf, A.W.; Jimenez, E. Iron-deficiency anemia and infant development: Effects of extended oral iron therapy. J. Pediatr. 1996, 129, 382–389. [Google Scholar] [CrossRef]
- Lozoff, B.; Klein, N.K.; Prabucki, K.M. Iron-deficient anemic infants at play. J. Dev. Behav. Pediatr. 1986, 7, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Walter, T.; Kovalskys, J.; Stekel, A. Effect of mild iron deficiency on infant mental development scores. J. Pediatr. 1983, 102, 519–522. [Google Scholar] [CrossRef]
- Walter, T.; De Andraca, I.; Chadud, P.; Perales, C.G. Iron deficiency anemia: Adverse effects on infant psychomotor development. Pediatrics 1989, 84, 7–17. [Google Scholar] [CrossRef]
- Walker, S.P.; Wachs, T.D.; Gardner, J.M.; Lozoff, B.; Wasserman, G.A.; Pollitt, E.; Carter, J.A. Child development: Risk factors for adverse outcomes in developing countries. Lancet 2007, 369, 145–157. [Google Scholar] [CrossRef]
- Iannotti, L.L.; Tielsch, J.M.; Black, M.M.; Black, R.E. Iron supplementation in early childhood: Health benefits and risks. Am. J. Clin. Nutr. 2006, 84, 1261–1276. [Google Scholar] [CrossRef] [PubMed]
- Aukett, M.A.; Parks, Y.A.; Scott, P.H.; Wharton, B.A. Treatment with iron increases weight gain and psychomotor development. Arch. Dis. Child. 1986, 61, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Brittenham, G.M.; Wolf, A.W.; McClish, D.K.; Kuhnert, P.M.; Jimenez, E.; Jimenez, R.; Mora, L.A.; Gomez, I.; Krauskoph, D. Iron deficiency anemia and iron therapy effects on infant developmental test performance. Pediatrics 1987, 79, 981–995. [Google Scholar] [PubMed]
- Dommergues, J.P.; Archambeaud, M.P.; Ducot, B.; Gerval, Y.; Hiard, C.; Rossignol, C.; Tchernia, G. Iron deficiency and psychomotor development tests. Longitudinal study between 10 months and 4 years of age. Arch. Fr. Pediatr. 1989, 46, 487–490. [Google Scholar] [PubMed]
- Lozoff, B.; Jimenez, E.; Wolf, A.W. Long-Term Developmental Outcome of Infants with Iron Deficiency. N. Engl. J. Med. 1991, 325, 687–694. [Google Scholar] [CrossRef]
- Palti, H.; Pevsner, B.; Adler, B. Does anemia in infancy affect achievement on developmental and intelligence tests? Hum. Biol. 1983, 55, 183–194. [Google Scholar] [PubMed]
- Palti, H.; Meijer, A.; Adler, B. Learning achievement and behavior at school of anemic and non-anemic infants. Early Hum. Dev. 1985, 10, 217–223. [Google Scholar] [CrossRef]
- Walter, T.I.; De Andraca, M.; Rivera, C.F.; Cobo, C. Cognitive effect at 5 years of age in infants who were anemic at 12 months: A longitudinal study. Pediatr. Res. 1990, 28, 295. [Google Scholar] [CrossRef][Green Version]
- Idjradinata, P.; Pollitt, E. Reversal of developmental delays in iron-deficient anaemic infants treated with iron. Lancet 1993, 341, 1–4. [Google Scholar] [CrossRef]
- Winick, M.; Meyer, K.; Harris, R. Malnutrition and environmental enrichment by early adoption. Science 1975, 190, 1173–1175. [Google Scholar] [CrossRef]
- Gomez-Pinilla, F.; Tyagi, E. Diet and cognition: Interplay between cell metabolism and neuronal plasticity. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Lintas, C. Linking genetics to epigenetics: The role of folate and folate-related pathways in neurodevelopmental disorders. Clin. Genet. 2019, 95, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, T.K.; Yngve, A.; Böttiger, A.K.; Hurtig-Wennlöf, A.; Sjöström, M. High Folate Intake Is Related to Better Academic Achievement in Swedish Adolescents. Pediatrics 2011, 128, e358–e365. [Google Scholar] [CrossRef] [PubMed]
- Breimer, L.H.; Nilsson, T.K. Has folate a role in the developing nervous system after birth and not just during embryogenesis and gestation? Scand. J. Clin. Lab. Investig. 2012, 72, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Adamo, A.M.; Oteiza, P.I. Zinc deficiency and neurodevelopment: The case of neurons. Biofactors 2010, 36, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Taneja, S. Zinc and cognitive development. Br. J. Nutr. 2001, 85 (Suppl. 2), S139–S145. [Google Scholar] [CrossRef] [PubMed]
- Di Girolamo, A.M.; Ramirez-Zea, M. Role of zinc in maternal and child mental health. Am. J. Clin. Nutr. 2009, 89, 940S–945S. [Google Scholar] [CrossRef] [PubMed]
- Saint, S.E.; Renzi-Hammond, L.M.; Khan, N.A.; Hillman, C.H.; Frick, J.E.; Hammond, B.R. The Macular Carotenoids are Associated with Cognitive Function in Preadolescent Children. Nutrients 2018, 10, 193. [Google Scholar] [CrossRef] [PubMed]
- Mulder, K.A.; Innis, S.M.; Rasmussen, B.F.; Wu, B.T.; Richardson, K.J.; Hasman, D. Plasma lutein concentrations are related to dietary intake, but unrelated to dietary saturated fat or cognition in young children. J. Nutr. Sci. 2014, 3, e11. [Google Scholar] [CrossRef]
- Lieblein-Boff, J.C.; Johnson, E.J.; Kennedy, A.D.; Lai, C.S.; Kuchan, M.J. Exploratory Metabolomic Analyses Reveal Compounds Correlated with Lutein Concentration in Frontal Cortex, Hippocampus, and Occipital Cortex of Human Infant Brain. PLoS ONE 2015, 10, e0136904. [Google Scholar] [CrossRef]
- Jia, Y.-P.; Sun, L.; Yu, H.-S.; Liang, L.-P.; Li, W.; Ding, H.; Song, X.-B.; Zhang, L.-J. The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases. Molecules 2017, 22, 610. [Google Scholar] [CrossRef] [PubMed]
- Bonatto, F.; Polydoro, M.; Andrades, M.E.; Júnior, M.L.C.D.F.; Dal-Pizzol, F.; Rotta, L.N.; Souza, D.O.; Perry, M.L.; Moreira, J.C.F. Effects of maternal protein malnutrition on oxidative markers in the young rat cortex and cerebellum. Neurosci. Lett. 2006, 406, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Grantham-McGregor, S.; Baker-Henningham, H. Review of the evidence linking protein and energy to mental development. Public Health Nutr. 2005, 8, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Baym, C.L.; Khan, N.A.; Monti, J.M.; Raine, L.B.; Drollette, E.S.; Moore, R.D.; Scudder, M.R.; Kramer, A.F.; Hillman, C.H.; Cohen, N.J. Dietary lipids are differentially associated with hippocampal-dependent relational memory in prepubescent children. Am. J. Clin. Nutr. 2014, 99, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Handeland, K.; Øyen, J.; Skotheim, S.; Graff, I.E.; Baste, V.; Kjellevold, M.; Frøyland, L.; Lie, Ø.; Dahl, L.; Stormark, K.M. Fatty fish intake and attention performance in 14–15 year old adolescents: FINS-TEENS—A randomized controlled trial. Nutr. J. 2017, 16, 64. [Google Scholar] [CrossRef]
- Taylor, R.M.; Fealy, S.M.; Bisquera, A.; Smith, R.; Collins, C.E.; Evans, T.J.; Hure, A.J. Effects of Nutritional Interventions during Pregnancy on Infant and Child Cognitive Outcomes: A Systematic Review and Meta-Analysis. Nutrients 2017, 9, 1265. [Google Scholar] [CrossRef]
- Chaddock, L.; Pontifex, M.B.; Hillman, C.H.; Kramer, A.F. A review of the relation of aerobic fitness and physical activity to brain structure and function in children. J. Int. Neuropsychol. Soc. 2011, 17, 975–985. [Google Scholar] [CrossRef]
- Donnelly, J.E.; Hillman, C.H.; Castelli, D.; Etnier, J.L.; Lee, S.; Tomporowski, P.; Lambourne, K.; Szabo-Reed, A.N. This summary was written for the American College of Sports Medicine b: Physical Activity, Fitness, Cognitive Function, and Academic Achievement in Children: A Systematic Review. Med. Sci. Sports Exerc. 2016, 48, 1223–1224. [Google Scholar] [CrossRef]
- Marques, A.; Santos, D.A.; Hillman, C.H.; Sardinha, L.B. How does academic achievement relate to cardiorespiratory fitness, self-reported physical activity and objectively reported physical activity: A systematic review in children and adolescents aged 6–18 years. Br. J. Sports Med. 2018, 52, 1039. [Google Scholar] [CrossRef]
- Martin, A.; Booth, J.N.; Laird, Y.; Sproule, J.; Reilly, J.J.; Saunders, D.H. Physical activity, diet and other behavioural interventions for improving cognition and school achievement in children and adolescents with obesity or overweight. Cochrane Database Syst. Rev. 2018, 3, CD009728. [Google Scholar]
- Drollette, E.; Raine, L.; Kao, S.-C.; Kahn, N.; Westfall, D.; Hamil, M.; Shorin, R.; Calobrisi, E.; John, D. Moving fast, thinking fast: The relations of physical activity levels and bouts to neuroelectric indies of inhibitory control in preadolescents. J. Sport Health Sci. 2019, 8, 1–14. [Google Scholar]
- Egger, F.; Benzing, V.; Conzelmann, A.; Schmidt, M. Boost your brain, while having a break! The effects of long-term cognitively engaging physical activity breaks on children’s executive functions and academic achievement. PLoS ONE 2019, 14, e0212482. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.; Timperio, A.; Brown, H.; Best, K.; Hesketh, K.D. Effect of classroom-based physical activity interventions on academic and physical activity outcomes: A systematic review and meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 114. [Google Scholar] [CrossRef] [PubMed]
- Asigbee, F.M.; Whitney, S.D.; Peterson, C.E. The Link between Nutrition and Physical Activity in Increasing Academic Achievement. J. Sch. Heal. 2018, 88, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Resaland, G.K.; Moe, V.F.; Aadland, E.; Steene-Johannessen, J.; Glosvik, O.; Andersen, J.R.; Kvalheim, O.M.; McKay, H.A.; Anderssen, S.A.; Group ASKs. Active Smarter Kids (ASK): Rationale and design of a cluster-randomized controlled trial investigating the effects of daily physical activity on children’s academic performance and risk factors for non-communicable diseases. BMC Public Health 2015, 15, 709. [Google Scholar] [CrossRef] [PubMed]
- Ardoy, D.N.; Fernandez-Rodriguez, J.M.; Jimenez-Pavon, D.; Castillo, R.; Ruiz, J.R.; Ortega, F.B. A physical education trial improves adolescents’ cognitive performance and academic achievement: The EDUFIT study. Scand. J. Med. Sci. Sports 2014, 24, e52–e61. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Pontifex, M.B.; Castelli, D.M.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Moore, R.D.; Wu, C.-T.; Kamijo, K. Effects of the FITKids Randomized Controlled Trial on Executive Control and Brain Function. Pediatrics 2014, 134, e1063–e1071. [Google Scholar] [CrossRef] [PubMed]
- Alhassan, S.; Laurent, C.W.S.; Burkart, S.; Greever, C.J.; Ahmadi, M.N. Feasibility of Integrating Physical Activity into Early Education Learning Standards on Preschooler’s Physical Activity Levels. J. Phys. Act. Heal. 2019, 16, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Ullman, M.T. Contributions of memory circuits to language: The declarative/procedural model. Cognition 2004, 92, 231–270. [Google Scholar] [CrossRef]
- Evans, T.M.; Ullman, M.T. An Extension of the Procedural Deficit Hypothesis from Developmental Language Disorders to Mathematical Disability. Front. Psychol. 2016, 7, 1318. [Google Scholar] [CrossRef]
- Eichenbaum, H.; Yonelinas, A.; Ranganath, C. The Medial Temporal Lobe and Recognition Memory. Annu. Rev. Neurosci. 2007, 30, 123–152. [Google Scholar] [CrossRef] [PubMed]
- Ranganath, C. A unified framework for the functional organization of the medial temporal lobes and the phenomenology of episodic memory. Hippocampus 2010, 20, 1263–1290. [Google Scholar] [CrossRef] [PubMed]
- Voss, M.W.; Soto, C.; Yoo, S.; Sodoma, M.; Vivar, C.; Van Praag, H. Exercise and Hippocampal Memory Systems. Trends Cogn. Sci. 2019, 23, 318–333. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Cornejo, I.; Rodriguez-Ayllon, M.; Verdejo-Roman, J.; Cadenas-Sanchez, C.; Mora-Gonzalez, J.; Chaddock-Heyman, L.; Raine, L.B.; Stillman, C.M.; Kramer, A.F.; Erickson, K.I.; et al. Physical Fitness, White Matter Volume and Academic Performance in Children: Findings from the ActiveBrains and FITKids2 Projects. Front. Psychol. 2019, 10, 208. [Google Scholar] [CrossRef] [PubMed]
- Chaddock-Heyman, L.; Erickson, K.I.; Chappell, M.A.; Johnson, C.L.; Kienzler, C.; Knecht, A.; Drollette, E.S.; Raine, L.B.; Scudder, M.R.; Kao, S.C.; et al. Aerobic fitness is associated with greater hippocampal cerebral blood flow in children. Dev. Cogn. Neurosci. 2016, 20, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.D.; Arena, R.; Riebe, D.; Pescatello, L.S. American College of Sports M: ACSM’s new preparticipation health screening recommendations from ACSM’s guidelines for exercise testing and prescription, ninth edition. Curr. Sports Med. Rep. 2013, 12, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Moreau, D.; Kirk, I.J.; Waldie, K.E. High-intensity training enhances executive function in children in a randomized, placebo-controlled trial. eLife 2017, 6, e25062. [Google Scholar] [CrossRef]
- Jeon, Y.K.; Ha, C.H. The effect of exercise intensity on brain derived neurotrophic factor and memory in adolescents. Environ. Health Prev. Med. 2017, 22, 27. [Google Scholar] [CrossRef]
- Miller, A.L.; Miller, S.E.; LeBourgeois, M.K.; Sturza, J.; Rosenblum, K.L.; Lumeng, J.C. Sleep duration and quality are associated with eating behavior in low-income toddlers. Appetite 2019, 135, 100–107. [Google Scholar] [CrossRef]
- Hill, C.M.; Hogan, A.M.; Karmiloff-Smith, A. To sleep, perchance to enrich learning? Arch. Dis. Child. 2007, 92, 637–643. [Google Scholar] [CrossRef][Green Version]
- Bernier, A.; Beauchamp, M.H.; Bouvette-Turcot, A.A.; Carlson, S.M.; Carrier, J. Sleep and cognition in preschool years: Specific links to executive functioning. Child Dev. 2013, 84, 1542–1553. [Google Scholar] [CrossRef] [PubMed]
- Botting, N.; Baraka, N. Sleep behaviour relates to language skills in children with and without communication disorders. Int. J. Dev. Disabil. 2017, 64, 238–243. [Google Scholar] [CrossRef]
- Cremone, A.; de Jong, D.M.; Kurdziel, L.B.F.; Desrochers, P.; Sayer, A.; LeBourgeois, M.K.; Spencer, R.M.C.; McDermott, J.M. Sleep Tight, Act Right: Negative Affect, Sleep and Behavior Problems during Early Childhood. Child Dev. 2018, 89, e42–e59. [Google Scholar] [CrossRef] [PubMed]
- Touchette, E.; Petit, D.; Séguin, J.R.; Boivin, M.; Tremblay, R.E.; Montplaisir, J.Y. Associations between Sleep Duration Patterns and Behavioral/Cognitive Functioning at School Entry. Sleep 2007, 30, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Ravid, S.; Afek, I.; Suraiya, S.; Shahar, E.; Pillar, G. Sleep Disturbances are Associated with Reduced School Achievements in First-Grade Pupils. Dev. Neuropsychol. 2009, 34, 574–587. [Google Scholar] [CrossRef] [PubMed]
- Buckhalt, J.A. Children’s Sleep, Sleepiness, and Performance on Cognitive Tasks. WMF Press Bull. 2011, 2011, 1–12. [Google Scholar] [PubMed]
- Cronin, A.; Halligan, S.L.; Murray, L. Maternal Psychosocial Adversity and the Longitudinal Development of Infant Sleep. Infancy 2008, 13, 469–495. [Google Scholar] [CrossRef]
- El-Sheikh, M.; Bagley, E.J.; Keiley, M.; Elmore-Staton, L.; Chen, E.; Buckhalt, J.A. Economic adversity and children’s sleep problems: Multiple indicators and moderation of effects. Health Psychol. 2013, 32, 849–859. [Google Scholar] [CrossRef]
- Bagley, E.J.; Kelly, R.J.; Buckhalt, J.A.; El-Sheikh, M. What keeps low-SES children from sleeping well: The role of presleep worries and sleep environment. Sleep Med. 2015, 16, 496–502. [Google Scholar] [CrossRef]
- Allen, S.L.; Howlett, M.D.; Coulombe, J.A.; Corkum, P.V.; Information, P.E.K.F.C. ABCs of SLEEPING: A review of the evidence behind pediatric sleep practice recommendations. Sleep Med. Rev. 2016, 29, 1–14. [Google Scholar] [CrossRef]
- Hale, L.; Berger, L.M.; LeBourgeois, M.K.; Brooks-Gunn, J. Social and Demographic Predictors of Preschoolers’ Bedtime Routines. J. Dev. Behav. Pediatr. 2009, 30, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W.; Eckenrode, J.; Marcynyszyn, L. Chaos and the Macrosetting: The Role of Poverty and Socioeconomic Status. In Decade of Behavior (Science Conference). Chaos and Its Influence on Children’s Development: An Ecological Perspective; Evans, G.W., Wachs, T.D., Eds.; American Psychological Association: Washington, DC, USA, 2010; pp. 225–238. [Google Scholar]
- Garaulet, M.; Ortega, F.B.; Ruiz, J.R.; Rey-Lopez, J.P.; Beghin, L.; Manios, Y.; Cuenca-García, M.; Plada, M.; Diethelm, K.; Kafatos, A.; et al. Short sleep duration is associated with increased obesity markers in European adolescents: Effect of physical activity and dietary habits. The HELENA study. Int. J. Obes. 2011, 35, 1308–1317. [Google Scholar] [CrossRef] [PubMed]
- Yoong, S.L.; Chai, L.K.; Williams, C.M.; Wiggers, J.; Finch, M.; Wolfenden, L. Systematic review and meta-analysis of interventions targeting sleep and their impact on child body mass index, diet, and physical activity. Obesity 2016, 24, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Case, A.; Fertig, A.; Paxson, C. The lasting impact of childhood health and circumstance. J. Health Econ. 2005, 24, 365–389. [Google Scholar] [CrossRef] [PubMed]
- Case, A.; Paxson, C. Causes and consequences of early-life health. Demography 2010, 47, S65–S85. [Google Scholar] [CrossRef] [PubMed]
- Haas, S.A. Health selection and the process of social stratification: The effect of childhood health on socioeconomic attainment. J. Health Soc. Behav. 2006, 47, 339–354. [Google Scholar] [CrossRef]
- Palloni, A. Reproducing inequalities: Luck, wallets, and the enduring effects of childhood health. Demography 2006, 43, 587–615. [Google Scholar] [CrossRef]
- Haas, S.A. The long-term effects of poor childhood health: An assessment and application of retrospective reports. Demography 2007, 44, 113–135. [Google Scholar] [CrossRef]
- Smith, J.P. Reconstructing childhood health histories. Demography 2009, 46, 387–403. [Google Scholar] [CrossRef]
- Streisand, R.M.; Zawacki, T.; Joseph, K.E.; Adams, C.D. Living with a Chronic Illness: A Measure of Social Functioning for Children and Adolescents. J. Pediatr. Psychol. 2002, 27, 593–605. [Google Scholar]
- Jackson, M.I. A Life Course Perspective on Child Health, Academic Experiences and Occupational Skill Qualifications in Adulthood: Evidence from a British Cohort. Soc. Forces 2010, 89, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Bradley, R.H.; Corwyn, R.F.; McAdoo, H.P.; Coll, C.G. The Home Environments of Children in the United States Part I: Variations by Age, Ethnicity, and Poverty Status. Child Dev. 2001, 72, 1844–1867. [Google Scholar] [CrossRef] [PubMed]
- Hackman, D.A.; Farah, M.J.; Meaney, M.J. Socioeconomic status and the brain: Mechanistic insights from human and animal research. Nat. Rev. Neurosci. 2010, 11, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.P.W.T.; Grantham-McGregor, S.; Black, M.M.; Nelson, C.A.; Huffman, S.L.; Baker-Henningham, H.; Chang, S.M.; Hamadani, J.D.; Lozoff, B.; Gardner, J.M.M. Inequality in early childhood: Risk and protective factors for early child development. Lancet 2011, 378, 1325–1338. [Google Scholar] [CrossRef]
- Hackman, D.A.; Farah, M.J. Socioeconomic status and the developing brain. Trends Cogn. Sci. 2009, 13, 65–73. [Google Scholar] [CrossRef] [PubMed]
- St John, A.M.; Kibbe, M.; Tarullo, A.R. A systematic assessment of socioeconomic status and executive functioning in early childhood. J. Exp. Child Psychol. 2019, 178, 352–368. [Google Scholar] [CrossRef] [PubMed]
- Ursache, A.; Noble, K.G. Socioeconomic status, white matter, and executive function in children. Brain Behav. 2016, 6, e00531. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; LoCasale-Crouch, J.; Kranz, S. Eating Patterns Among Head Start Children: Do They Relate to School Readiness Skills. In Biennial Meeting of the Society for Research in Child Development; Society for Research in Child Development: Baltimore, MD, USA, 2019. [Google Scholar]
- Grantham-McGregor, S.; Cheung, Y.B.; Cueto, S.; Glewwe, P.; Richter, L.; Strupp, B.; International Child Development Steering Group. Developmental potential in the first 5 years for children in developing countries. Lancet 2007, 369, 60–70. [Google Scholar] [CrossRef]
- Toczydlowska, E. Children in the Bottom of Income Distribution in Europe: Risks and Composition; UNICEF: New York, NY, USA, 2016. [Google Scholar]
- Fund UNICEF. Building the Future: Children and the Sustainable Development Goals in Rich Countries; Innocenti Report Card; UNICEF: New York, NY, USA, 2017. [Google Scholar]
- UNICEF-WHO-The World Bank Group. Joint Child Malnutrition Estimates—Levels and Trends; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World; UNICEF: New York, NY, USA, 2018. [Google Scholar]
- Fund UNICEF. The State of the Worlds’ Children; UNICEF: New York, NY, USA, 2017. [Google Scholar]
- Ogden, C.L.; Fakhouri, T.H.; Carroll, M.D.; Hales, C.M.; Fryar, C.D.; Li, X.; Freedman, D.S. Prevalence of Obesity Among Adults, by Household Income and Education—United States, 2011–2014. Morb. Mortal. Wkly. Rep. 2017, 66, 1369–1373. [Google Scholar] [CrossRef]
- UNESCO. More than One-Half of Children and Adolescents Are Not Learning Worldwide; Publication No UIS/FS/2017/ED/46; UNESCO: Paris, France, 2017. [Google Scholar]
- UNESCO. Sustainable Development Goal 4; UNESCO: Paris, France, 2019. [Google Scholar]
- Latham, S. Changes in School Readiness of America’s Entering Kindergarteners (1998–2010); Springer: Berlin/Heidelberg, Germany, 2018; pp. 111–138. [Google Scholar]
- Duncan, G.J.; Magnuson, K. Socioeconomic status and cognitive functioning: Moving from correlation to causation. Wiley Interdiscip. Rev. Cogn. Sci. 2012, 3, 377–386. [Google Scholar] [CrossRef]
- UNICEF. Inequities in early childhood development: What the data say. In Evidence from the Multiple Indicator Cluster Surveys; UNICEF: New York, NY, USA, 2012. [Google Scholar]
- Evans, G.W. The environment of childhood poverty. Am. Psychol. 2004, 59, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Lamb, M.M.; Ogden, C.L. The Association between Food Insecurity and Obesity in Children—The National Health and Nutrition Examination Survey. J. Acad. Nutr. Diet. 2015, 115, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, T.W.; Oellerich, D.; Meade, E.; Simms, J.; Stock, A. Neighborhood poverty and children’s food insecurity. Child. Youth Serv. Rev. 2016, 66, 85–93. [Google Scholar] [CrossRef]
- Widener, M.J. Spatial access to food: Retiring the food desert metaphor. Physiol. Behav. 2018, 193, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Black, C.; Moon, G.; Baird, J. Dietary inequalities: What is the evidence for the effect of the neighbourhood food environment? Health Place 2014, 27, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.E.; Keane, C.R.; Burke, J.G. Disparities and access to healthy food in the United States: A review of food deserts literature. Health Place 2010, 16, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, D.; Smith, C.; Eikenberry, N. Fruit and vegetable access in four low-income food deserts communities in Minnesota. Agric. Hum. Values 2006, 23, 371–383. [Google Scholar] [CrossRef]
- Committee on Environmental Health. The built environment: Designing communities to promote physical activity in children. Pediatrics 2009, 123, 1591–1598. [Google Scholar] [CrossRef]
- Bates, C.R.; Buscemi, J.; Nicholson, L.M.; Cory, M.; Jagpal, A.; Bohnert, A.M. Links between the organization of the family home environment and child obesity: A systematic review. Obes. Rev. 2018, 19, 716–727. [Google Scholar] [CrossRef]
- Schrempft, S.; Van Jaarsveld, C.H.; Fisher, A.; Fildes, A.; Wardle, J. Maternal characteristics associated with the obesogenic quality of the home environment in early childhood. Appetite 2016, 107, 392–397. [Google Scholar] [CrossRef]
- De Jong, E.; Visscher, T.L.; HiraSing, R.A.; Seidell, J.C.; Renders, C.M. Home environmental determinants of children’s fruit and vegetable consumption across different SES backgrounds. Pediatr. Obes. 2015, 10, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Muñiz, E.I.; Silver, E.J.; Stein, R.E. Family Routines and Social-Emotional School Readiness Among Preschool-Age Children. J. Dev. Behav. Pediatr. 2014, 35, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, L.K.; Bub, K.L. Family Routines and School Readiness during the Transition to Kindergarten. Early Educ. Dev. 2017, 28, 59–77. [Google Scholar] [CrossRef]
- Bradley, R.H.; Corwyn, R.F.; Burchinal, M.; McAdoo, H.P.; Coll, C.G. The Home Environments of Children in the United States Part II: Relations with Behavioral Development through Age Thirteen. Child Dev. 2001, 72, 1868–1886. [Google Scholar] [CrossRef] [PubMed]
- Duncan, G.J.; Magnuson, K.; Votruba-Drzal, E. Moving Beyond Correlations in Assessing the Consequences of Poverty. Annu. Rev. Psychol. 2017, 68, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Bradley, R.H.; Corwyn, R.F. Home Life and the Development of Competence in Mathematics: Implications of Research with the HOME Inventory. In Early Childhood Mathematics Skill Development in the Home Environment; Blevins-Knabe, A.A., Ed.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Mol, S.E.; Bus, A.G. To read or not to read: A meta-analysis of print exposure from infancy to early adulthood. Psychol. Bull. 2011, 137, 267–296. [Google Scholar] [CrossRef]
- Masarik, A.S.; Conger, R.D. Stress and child development: A review of the Family Stress Model. Curr. Opin. Psychol. 2017, 13, 85–90. [Google Scholar] [CrossRef]
- Mistry, R.S.; Vandewater, E.A.; Huston, A.C.; McLoyd, V.C. Economic well-being and children’s social adjustment: The role of family process in an ethnically diverse low-income sample. Child Dev. 2002, 73, 935–951. [Google Scholar] [CrossRef]
- Melhuish, E.; Ereky-Stevens, K.; Petrogiannis, K.; Ariescu Penderi, E.; Rentzou, K.; Tawell, A.; Slot, B.; Leseman, P. A Review of Research on the Effects of Early Childhood Education and Care (ECEC) upon Child Development; CARE Project; CARE: Utrecht, The Netherlands, 2015. [Google Scholar]
- Zaslow, M.; Burchinal, M.; Tarullo, L.; Martinez-Beck, I.V. Quality thresholds, features, and dosage in early care and education: discussion and conclusions. Monogr. Soc. Res. Child Dev. 2016, 81, 75–87. [Google Scholar] [CrossRef]
- Cappella, E.; Aber, J.L.; Kim, H.Y.; Gitomer, D.H.; Bell, C.A. Teaching beyond Achievement Tests: Perspectives from Developmental and Education Science. In Handbook of Research on Teaching; ERIC: Washington, DC, USA, 2016; pp. 249–347. [Google Scholar]
- Yoshikawa, H.; Kabay, S. The Evidence Base on Early Childhood Care and Education in Global Contexts; UNESCO: Paris, France, 2015. [Google Scholar]
- OCED. New Insights from TALIS 2013: Teaching and Learning in Primary and Upper Secondary Education; OECD: Paris, France, 2014. [Google Scholar]
- Heckman, J.J. Skill Formation and the Economics of Investing in Disadvantaged Children. Science 2006, 312, 1900–1902. [Google Scholar] [CrossRef]
- Sammons, P.; Anders, Y.; Sylva, K.; Melhuish, E.; Siraj-Blatchford, I.; Taggart, B.; Barreau, S. Children’s Cognitive Attainment and Progress in English Primary Schools during Key Stage 2: Investigating the potential continuing influences of pre-school education. Fruhpadagogische Ford. Inst. 2008. [Google Scholar] [CrossRef]
- Sylva, K.; Melhuish, E.; Sammons, P.; Siraj-Blatchford, I.; Taggart, B. The Effective Provision of Pre-School Education (EPPE) Project: Final Report: A Longitudinal Study Funded by the DfES 1997–2004; UCL Press: London, UK, 2004. [Google Scholar]
- Arnold, D.H.; Doctoroff, G.L. The early education of socioeconomically disadvantaged children. Annu. Rev. Psychol. 2003, 54, 517–545. [Google Scholar] [CrossRef] [PubMed]
- Aboud, F.E.; Yousafzai, A.K. Global health and development in early childhood. Annu. Rev. Psychol. 2015, 66, 433–457. [Google Scholar] [CrossRef] [PubMed]
- Grantham-McGregor, S.M.; Fernald, L.C.; Kagawa, R.M.; Walker, S. Effects of integrated child development and nutrition interventions on child development and nutritional status. Ann. N. Y. Acad. Sci. 2014, 1308, 11–32. [Google Scholar] [CrossRef] [PubMed]
- OCED. Starting Strong 2017: Key OECD Indicators on Early Childhood Education and Care; OECD Publishing: Paris, France, 2018. [Google Scholar]
- Vargas-Baron, E. Policies on Early Childhood Care and Education: Their Evolution and Some Impacts; Background Paper Prepared for the Education for All Global Monitoring Report 2015; UNESCO: Paris, France, 2015. [Google Scholar]
- Levy, S.; Schady, N. Latin America’s Social Policy Challenge: Education, Social Insurance, Redistribution. J. Econ. Perspect. 2013, 27, 193–218. [Google Scholar] [CrossRef]
- The World Bank. World Development Report: Learning to Realize Education’s Promise; The World Bank: Washington, DC, USA, 2018. [Google Scholar]
- Burchinal, M.; Vandergrift, N.; Pianta, R.; Mashburn, A. Threshold analysis of association between child care quality and child outcomes for low-income children in pre-kindergarten programs. Early Child. Res. Q. 2010, 25, 166–176. [Google Scholar] [CrossRef]
- Herman, J.; Post, S.; O’Halloran, S. The United States is Far behind Other Countries on Pre-K. Dietetics 2013, 115, 751–758. [Google Scholar]
- McFarland, L.; Hussar, B.; Wang, X.; Zhang, J.; Wang, K.; Rathbun, A.; Mann, F.B. The Condition of Education National Center for Education Statistics 2018. NCES 2018-144; National Center for Education Statistics: Washington, DC, USA, 2018. [Google Scholar]
- European Commission. Tackling Social and Educational Inequalities through Early Childhood Education and Care in Europe; European Commission: Brussels, Belgium, 2009. [Google Scholar]
- Britto, P.R.; Yoshikawa, H.; Boller, K. Quality of Early Childhood Development Programs in Global Contexts: Rationale for Investment, Conceptual Framework and Implications for Equity and commentaries. Soc. Policy Rep. 2011, 25, 1–31. [Google Scholar] [CrossRef]
- Leu, H.R.; Schelle, R. Between education and care? Critical reflections on early childhood policies in Germany. Early Years 2009, 29, 5–18. [Google Scholar] [CrossRef]
- Lo Casale-Crouch, J.; Konold, T.; Pianta, R.; Howes, C.; Burchinal, M.; Bryant, D.; Clifford, R.; Early, D.; Barbarin, O.; Konold, T. Observed classroom quality profiles in state-funded pre-kindergarten programs and associations with teacher, program, and classroom characteristics. Early Child. Res. Q. 2007, 22, 3–17. [Google Scholar] [CrossRef]
- Stewart, K.; Waldfogel, J. Closing Gaps Early: The Role of Early Years Policy in Promoting Social Mobility in England; The Sutton Trust: London, UK, 2017. [Google Scholar]
- Vandenbroeck, S.; Jorissen, M. Endoscopic septoplasty: Literature review, surgical techniques and retrospective analysis at the University Hospitals Leuven. J. Laryngol. Otol. 2019, 133, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Zachrisson, H.D.; Dearing, E.; Lekhal, R.; Toppelberg, C.O. Little Evidence That Time in Child Care Causes Externalizing Problems During Early Childhood in Norway. Child Dev. 2013, 84, 1152–1170. [Google Scholar] [CrossRef] [PubMed]
- Taras, H. Nutrition and student performance at school. J. Sch. Health 2005, 75, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Widenhorn-Muller, K.; Hille, K.; Klenk, J.; Weiland, U. Influence of having breakfast on cognitive performance and mood in 13- to 20-year-old high school students: Results of a crossover trial. Pediatrics 2008, 122, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Boschloo, A.; Ouwehand, C.; Dekker, S.; Lee, N.; De Groot, R.; Krabbendam, L.; Jolles, J. The Relation between Breakfast Skipping and School Performance in Adolescents. Mind Brain Educ. 2012, 6, 81–88. [Google Scholar] [CrossRef]
- Ells, L.J.; Hillier, F.C.; Shucksmith, J.; Crawley, H.; Harbige, L.; Shield, J.; Wiggins, A.; Summerbell, C.D. A systematic review of the effect of dietary exposure that could be achieved through normal dietary intake on learning and performance of school-aged children of relevance to UK schools. Br. J. Nutr. 2008, 100, 927–936. [Google Scholar] [CrossRef][Green Version]
- Rampersaud, G.C.; Pereira, M.A.; Girard, B.L.; Adams, J.; Metzl, J.D. Breakfast Habits, Nutritional Status, Body Weight, and Academic Performance in Children and Adolescents. J. Am. Diet. Assoc. 2005, 105, 743–760. [Google Scholar] [CrossRef]
- Richter, L.M.; Rose, C.; Griesel, R.D. Cognitive and behavioural effects of a school breakfast. S. Afr. Med. J. 1997, 87, 93–100. [Google Scholar]
- Lien, L. Is breakfast consumption related to mental distress and academic performance in adolescents? Public Health Nutr. 2007, 10, 422–428. [Google Scholar] [CrossRef]
- Edwards, J.U.; Mauch, L.; Winkelman, M.R. Relationship of Nutrition and Physical Activity Behaviors and Fitness Measures to Academic Performance for Sixth Graders in a Midwest City School District. J. Sch. Health 2011, 81, 65–73. [Google Scholar] [CrossRef]
- Murphy, J.M.; Pagano, M.E.; Nachmani, J.; Sperling, P.; Kane, S.; Kleinman, R.E. The relationship of school breakfast to psychosocial and academic functioning: Cross-sectional and longitudinal observations in an inner-city school sample. Arch. Pediatr. Adolesc. Med. 1998, 152, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Wahlstrom, K.L.; Begalle, M.S. More than test scores-results of the Universal School breakfast pilot in Minnesota. Top. Clin. Nutr. 1999, 15, 7–29. [Google Scholar] [CrossRef]
- Kleinman, R.E.; Hall, S.; Green, H.; Korzec-Ramirez, D.; Patton, K.; Pagano, M.E.; Murphy, J.M. Diet, breakfast, and academic performance in children. Ann. Nutr. Metab. 2002, 46 (Suppl. 1), 24–30. [Google Scholar] [CrossRef] [PubMed]
- Simeon, D.T. School feeding in Jamaica: A review of its evaluation. Am. J. Clin. Nutr. 1998, 67, 790S–794S. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, R.T.; Benton, D. Cognitive functioning is susceptible to the level of blood glucose. Psychopharmacology 1999, 145, 378–385. [Google Scholar] [CrossRef]
- Pollitt, E.; Mathews, R. Breakfast and cognition: An integrative summary. Am. J. Clin. Nutr. 1998, 67, 804S–813S. [Google Scholar] [CrossRef] [PubMed]
- Meyers, A.F.; Sampson, A.E.; Weitzman, M.; Rogers, B.L.; Kayne, H. School Breakfast Program and School Performance. Arch. Pediatr. Adolesc. Med. 1989, 143, 1234. [Google Scholar] [CrossRef]
- Shemilt, I.; Harvey, I.; Shepstone, L.; Swift, L.; Reading, R.; Mugford, M.; Belderson, P.; Norris, N.; Thoburn, J.; Robinson, J. A national evaluation of school breakfast clubs: Evidence from a cluster randomized controlled trial and an observational analysis. Child Care Health Dev. 2004, 30, 413–427. [Google Scholar] [CrossRef]
- Herrero Lozano, R.; Fillat Ballesteros, J.C. A study on breakfast and school performance in a group of adolescents. Nutr. Hosp. 2006, 21, 346–352. [Google Scholar]
- Lopez-Sobaler, A.M.; Ortega, R.M.; Quintas, M.E.; Navia, B.; Requejo, A.M. Relationship between habitual breakfast and intellectual performance (logical reasoning) in well-nourished schoolchildren of Madrid (Spain). Eur. J. Clin. Nutr. 2003, 57 (Suppl. 1), S49–S53. [Google Scholar] [CrossRef][Green Version]
- Sigman, M.; McDonald, M.A.; Neumann, C.; Bwibo, N. Prediction of Cognitive Competence in Kenyan Children from Toddler Nutrition, Family Characteristics and Abilities. J. Child Psychol. Psychiatry 1991, 32, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Frank, D.A.; Zeisel, S.H. Failure to thrive. Pediatr. Clin. N. Am. 1988, 35, 1187–1206. [Google Scholar] [CrossRef]
- Hirsh-Pasek, K.B.L.; Singer, D. Applying the Scientific Evidence; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Foster, F.P. Nutrition and educational experience: Interrelated variables in children’s learning. Young Children 1972, 27, 284–288. [Google Scholar]
- Goldberger, L.I.; Ausman, L.M.; Boelkins, R.C. Incidental learning, attention, and curiosity in squirrel monkeys calorie deprived as infants. Learn. Motiv. 1980, 11, 185–207. [Google Scholar] [CrossRef]
- Jirout, J.; Vitiello, V.; Zumbrunn, S. Curiosity in Schools. In The New Science of Curiosity; Gordon, G., Ed.; Nova: Hauppauge, NY, USA, 2018. [Google Scholar]
- Kristjansson, A.L.; Sigfusdottir, I.D.; Allegrante, J.P. Health behavior and academic achievement among adolescents: The relative contribution of dietary habits, physical activity, body mass index, and self-esteem. Health Educ. Behav. 2010, 37, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Sandstead, H.H.; Penland, J.G.; Alcock, N.W.; Dayal, H.H.; Chen, X.C.; Li, J.S.; Zhao, F.; Yang, J.J. Effects of repletion with zinc and other micronutrients on neuropsychologic performance and growth of Chinese children. Am. J. Clin. Nutr. 1998, 68, 470S–475S. [Google Scholar] [CrossRef]
- Benton, D.; Roberts, G. Effect of vitamin and mineral supplementation on intelligence of a sample of schoolchildren. Lancet 1988, 331, 140–143. [Google Scholar] [CrossRef]
- Schoenthaler, S.J.; Amos, S.P.; Eysenck, H.J.; Peritz, E.; Yudkin, J. Controlled trial of vitamin-mineral supplementation: Effects on intelligence and performance. Personal. Individ. Differ. 1991, 12, 351–362. [Google Scholar] [CrossRef]
- Crombie, I. Effect of vitamin and mineral supplementation on verbal and non-verbal reasoning of schoolchildren. Lancet 1990, 335, 744–747. [Google Scholar] [CrossRef]
- Prinz, R.J.; Roberts, W.A.; Hantman, E. Dietary correlates of hyperactive behavior in children. J. Consult. Clin. Psychol. 1980, 48, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Wolraich, M.L.; Stumbo, P.J.; Milich, R.; Chenard, C.; Schultz, F.; Milch, R. Dietary characteristics of hyperactive and control boys. J. Am. Diet. Assoc. 1986, 86, 500–504. [Google Scholar] [PubMed]
- Wolraich, M.; Milich, R.; Stumbo, P.; Schultz, F. Effects of sucrose ingestion on the behavior of hyperactive boys. J. Pediatr. 1985, 106, 675–682. [Google Scholar] [CrossRef]
- Ferguson, H.B.; Stoddart, C.; Simeon, J.G. Double-blind challenge studies of behavioral and cognitive effects of sucrose-aspartame ingestion in normal children. Nutr. Rev. 1986, 44, 144–150. [Google Scholar] [CrossRef]
- Roshon, M.S.; Hagen, R.L. Sugar consumption, locomotion, task orientation, and learning in preschool children. J. Abnorm. Child Psychol. 1989, 17, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Mahan, L.K.; Chase, M.; Furukawa, C.T.; Sulzbacher, S.; Shapiro, G.G.; Pierson, W.E.; Bierman, C.W. Sugar “allergy” and children’s behavior. Ann. Allergy 1988, 61, 453–458. [Google Scholar] [CrossRef]
- Wender, E.H.; Solanto, M.V. Effects of sugar on aggressive and inattentive behavior in children with attention deficit disorder with hyperactivity and normal children. Pediatrics 1991, 88, 960–966. [Google Scholar] [PubMed]
- Behar, D.; Adams, A.A.; Berg, C.K.; Cornblath, M. Sugar challenge testing with children considered behaviourally ‘sugar active’. Nutr. Behav. 1984, 1, 277–288. [Google Scholar]
- Saravis, S.; Schachar, R.; Zlotkin, S.; Leiter, L.A.; Anderson, G.H. Aspartame: Effects on learning, behavior, and mood. Pediatrics 1990, 86, 75–83. [Google Scholar] [PubMed]
- Logue, A.W. The Psychology of Eating and Drinking: An Introduction; WH Freeman: New York, NY, USA, 1991. [Google Scholar]
- Deheeger, M.; Rolland-Cachera, M.F.; Fontvieille, A.M. Physical activity and body composition in 10 year old French children: Linkages with nutritional intake? Int. J. Obes. Relat. Metab. Disord. 1997, 21, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Benton, D.; Brett, V.; Brain, P.F. Glucose improves attention and reaction to frustration in children. Boil. Psychol. 1987, 24, 95–100. [Google Scholar] [CrossRef]
- Foster, J.K.; Lidder, P.G.; Sunram, S.I. Glucose and memory: Fractionation of enhancement effects? Psychopharmacology 1998, 137, 259–270. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jirout, J.; LoCasale-Crouch, J.; Turnbull, K.; Gu, Y.; Cubides, M.; Garzione, S.; Evans, T.M.; Weltman, A.L.; Kranz, S. How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children. Nutrients 2019, 11, 1953. https://doi.org/10.3390/nu11081953
Jirout J, LoCasale-Crouch J, Turnbull K, Gu Y, Cubides M, Garzione S, Evans TM, Weltman AL, Kranz S. How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children. Nutrients. 2019; 11(8):1953. https://doi.org/10.3390/nu11081953
Chicago/Turabian StyleJirout, Jamie, Jennifer LoCasale-Crouch, Khara Turnbull, Yin Gu, Mayaris Cubides, Sarah Garzione, Tanya M. Evans, Arthur L. Weltman, and Sibylle Kranz. 2019. "How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children" Nutrients 11, no. 8: 1953. https://doi.org/10.3390/nu11081953
APA StyleJirout, J., LoCasale-Crouch, J., Turnbull, K., Gu, Y., Cubides, M., Garzione, S., Evans, T. M., Weltman, A. L., & Kranz, S. (2019). How Lifestyle Factors Affect Cognitive and Executive Function and the Ability to Learn in Children. Nutrients, 11(8), 1953. https://doi.org/10.3390/nu11081953





