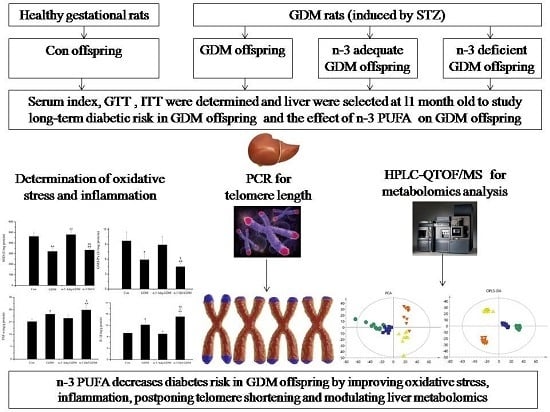
n-3 Polyunsaturated Fatty Acids Decrease Long-Term Diabetic Risk of Offspring of Gestational Diabetes Rats by Postponing Shortening of Hepatic Telomeres and Modulating Liver Metabolism
Abstract
1. Introduction
2. Materials and Methods
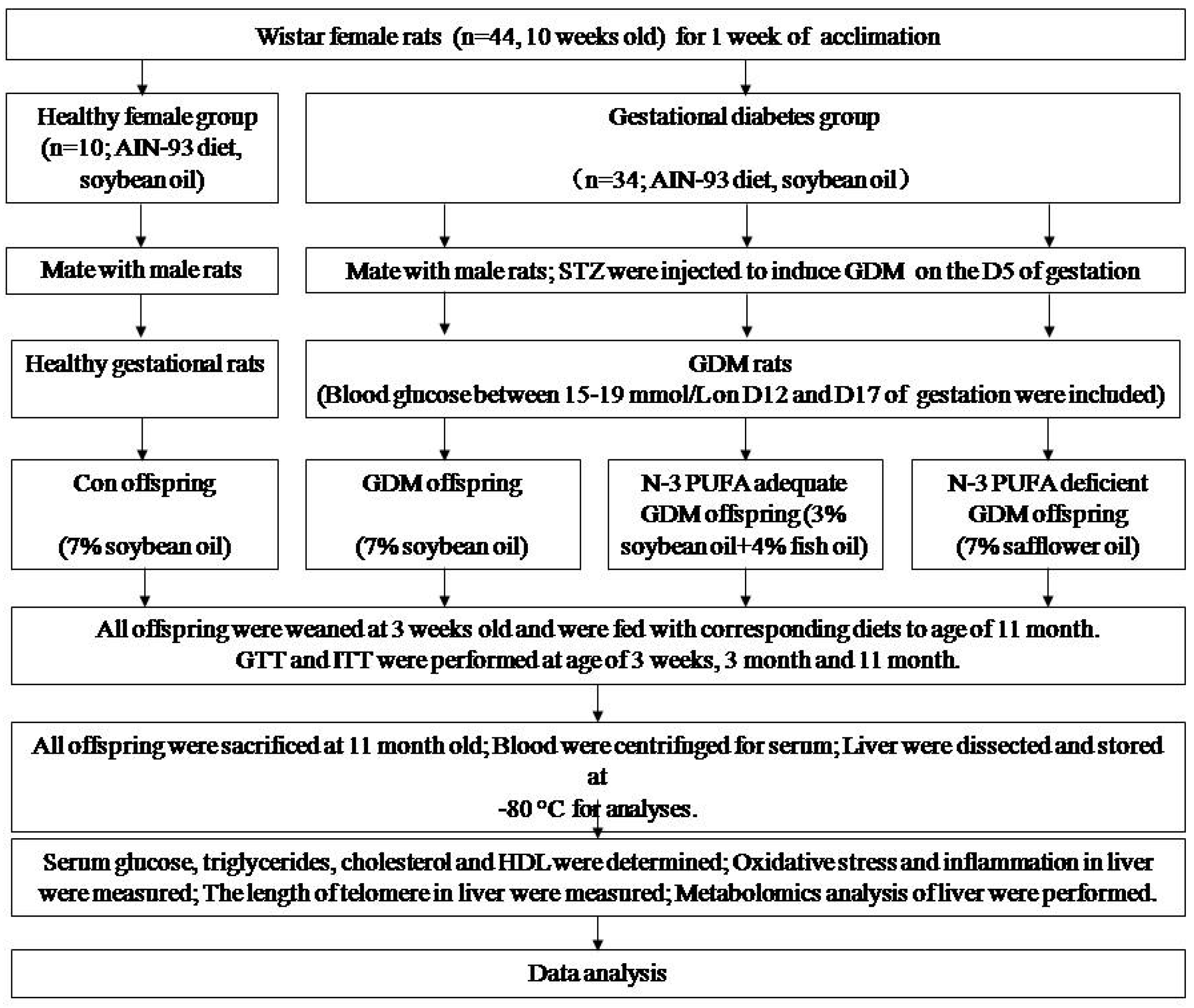
2.1. Animal Model and Offspring Diet Intervention
2.2. Determination of Serum Factors
2.3. Glucose and Insulin Tolerance Tests
2.4. Measurement of Oxidative Stress
2.5. Enzyme-Linked Immunosorbent Assay for Inflammatory Factors
2.6. Telomere Length Measurement
2.7. Metabolomics Analysis
2.7.1. Sample Preparation
2.7.2. High Performance Lipid Chromatography Coupled with Quadrupole-Time of Flight Mass Spectrometry (HPLC-QTOF-MS) Analysis
2.7.3. Data Processing and Metabolites Identification
2.8. Statistical Analysis
3. Results
3.1. Serum Biochemical Index of GDM Offspring and Effect of n-3 PUFA
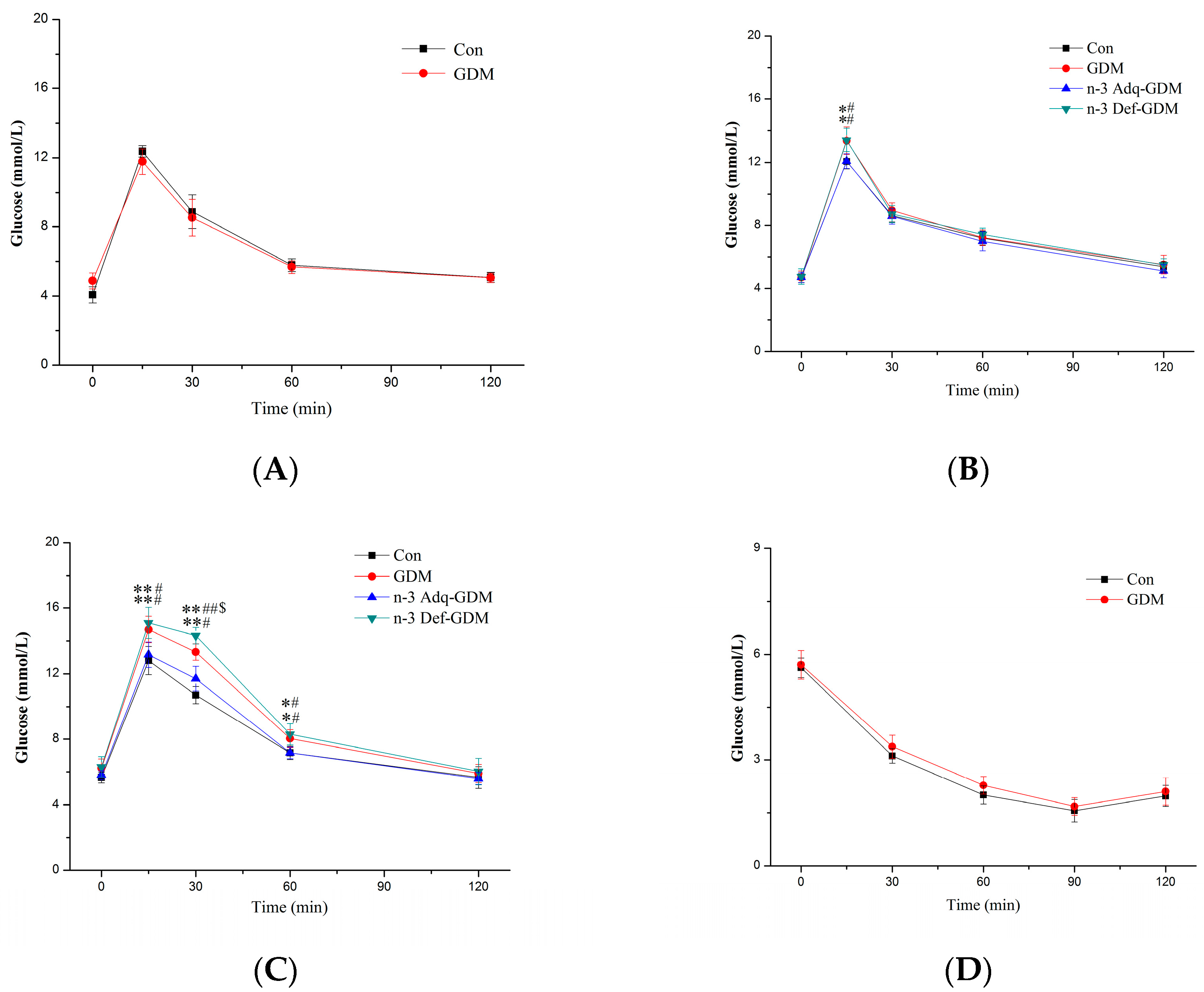
3.2. Glucose and Insulin Tolerance Test during Growth of GDM Offspring and Effect of n-3 PUFA
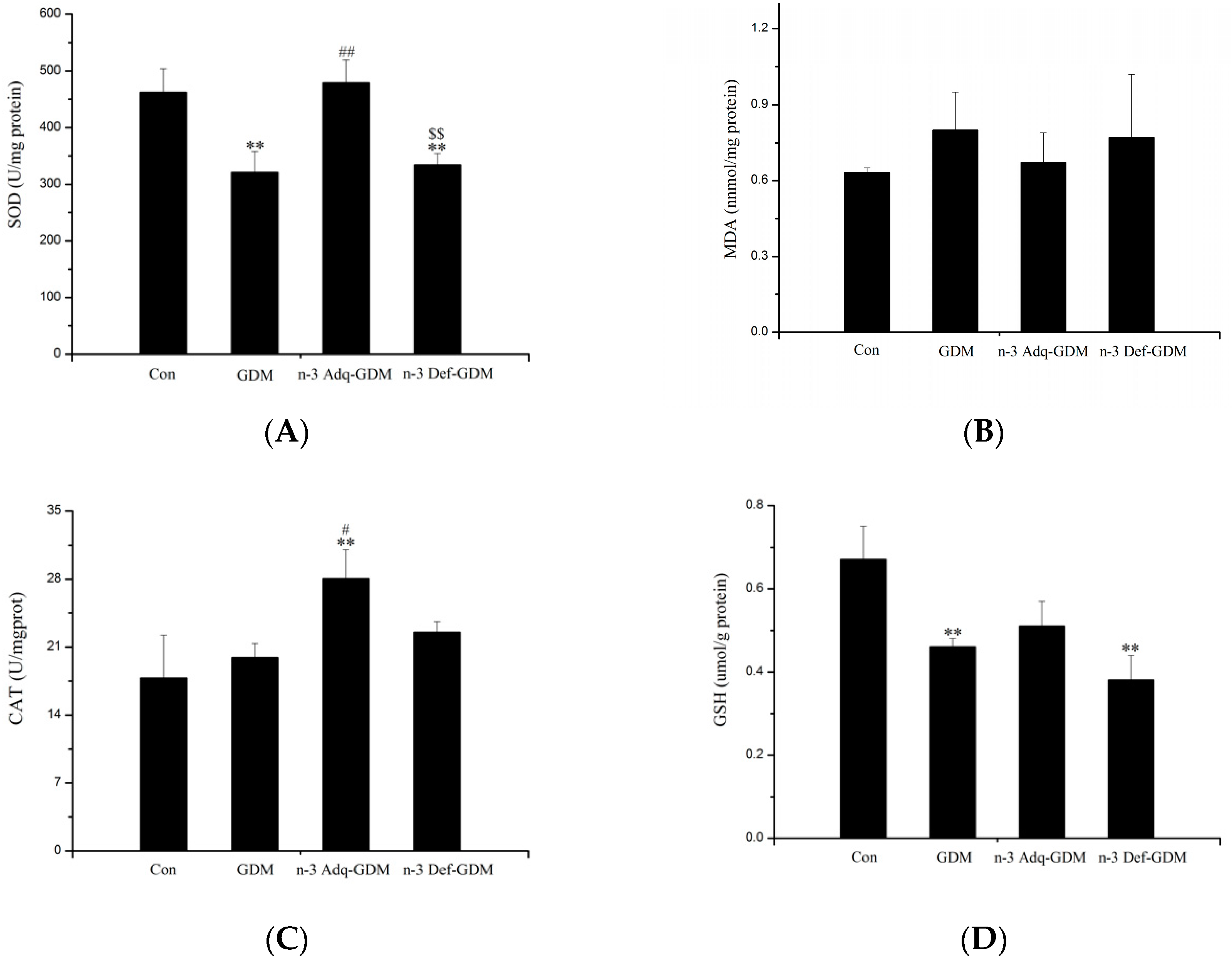
3.3. Effect of n-3 PUFA on Oxidative Stress of the Liver of GDM Offspring
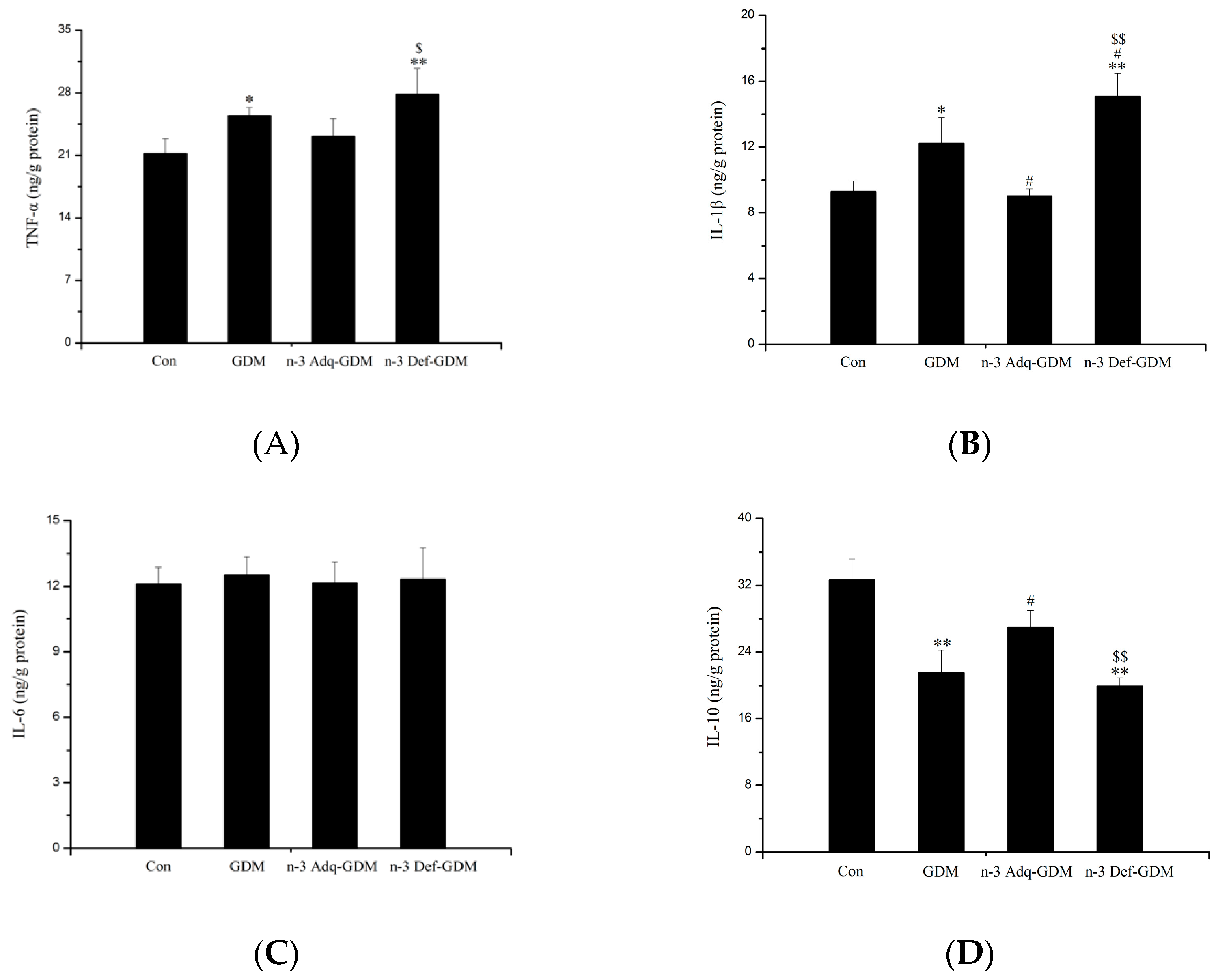
3.4. Effect of n-3 PUFA on Inflammatory Factors in the Liver of GDM Offspring
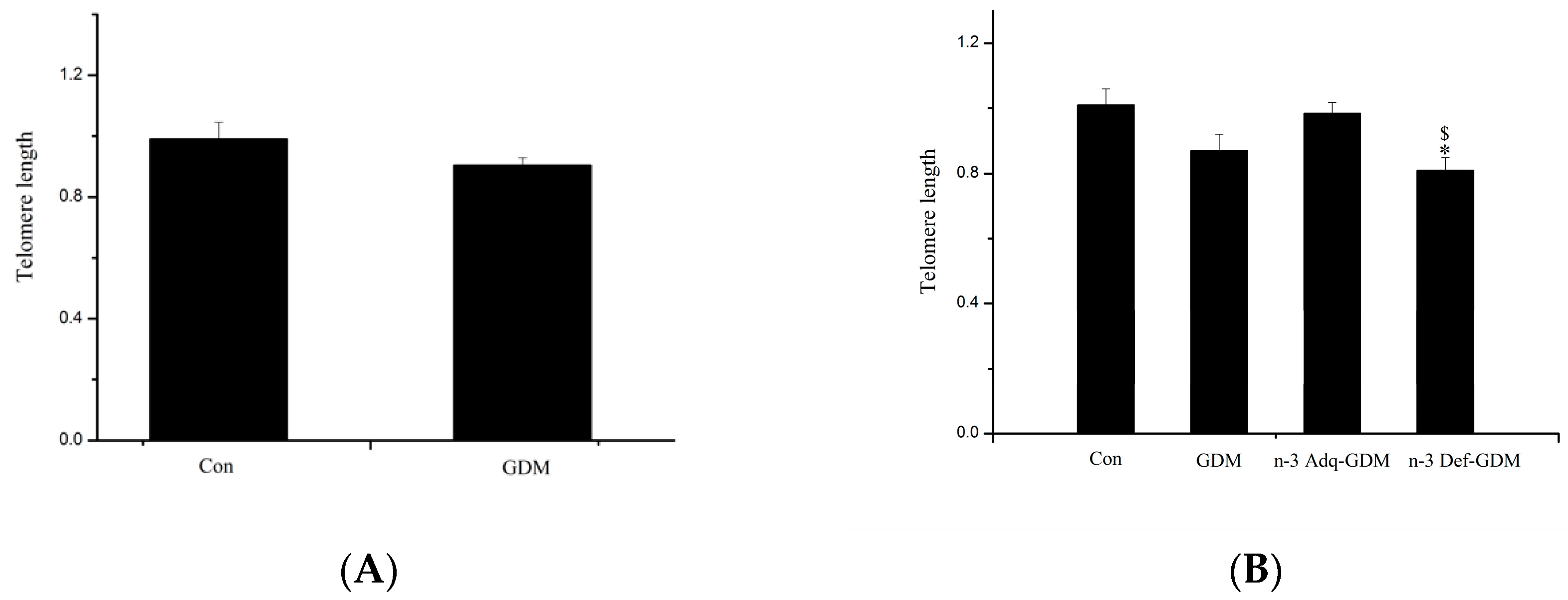
3.5. Influence of GDM on Telomere Length of the Liver of Offspring and the Effect of n-3 PUFA and n-6 PUFA on Telomere Length
3.6. Metabolomics Analysis of the Liver of GDM Offspring at Old Age and the Modulating Effect of n-3 PUFA on the Liver of GDM Offspring
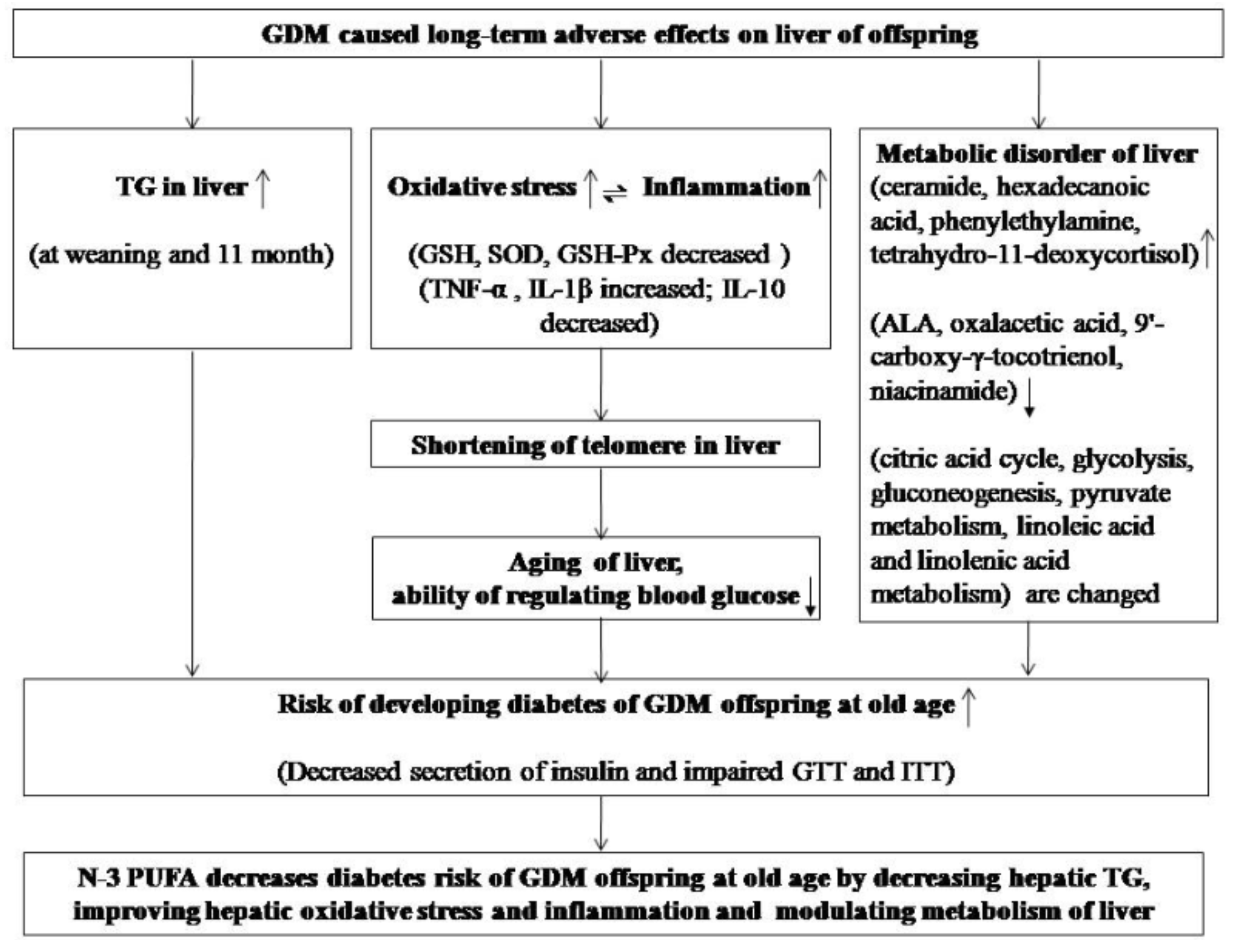
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Pereira, T.J.; Moyce, B.L.; Kereliuk, S.M.; Dolinsky, V.W. Influence of maternal overnutrition and gestational diabetes on the programming of metabolic health outcomes in the offspring: Experimental evidence. Biochem. Cell Biol. 2015, 93, 438–451. [Google Scholar] [CrossRef] [PubMed]
- Riediger, N.D.; Othman, R.A.; Suh, M.; Moghadasian, M.H. A Systemic review of the roles of n-3 Fatty Acids in Health and Disease. J. Am. Diet. Assoc. 2009, 109, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Nettleton, J.; Katz, R. N-3 long-chain polyunsaturated fatty acids in type 2 diabetes: A review. J. Am. Diet. Assoc. 2005, 105, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Poreba, M.; Mostowik, M.; Siniarski, A.; Golebiowska-Wiatrak, R.; Malinowski, K.P.; Haberka, M.; Konduracka, E.; Nessler, J.; Undas, A.; Gajos, G. Treatment with high-dose n-3 PUFAs has no effect on platelet function, coagulation, metabolic status or inflammation in patients with atherosclerosis and type 2 diabetes. Cardiovasc. Diabetol. 2017, 16, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, R.S.; Westgate, J.A.; Beattie, J.; Pattison, N.S.; Gamble, G.; Mildenhall, L.F.J.; Breier, B.H.; Johnstone, F.D. Inverse changes in fetal insulin-like growth factor (IGF)-1 and IGF binding protein-1 in association with higher birth weight in maternal diabetes. Clin. Endocrinol. 2007, 66, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Yessoufou, A.; Soulaimann, N.; Merzouk, S.; Moutairou, K.; Ahissou, H.; Prost, H.; Simmoni, A.M.; Merzouk, A.; Khan, N.A. N-3 Fatty acids modulate antioxidant status in diabetic rats and their macrosomic offspring. Int. J. Obes. 2006, 30, 739–750. [Google Scholar] [CrossRef]
- Pereira, T.J.; Fonseca, M.A.; Campbell, K.E.; Moyce, B.L.; Cole, L.K.; Hatch, G.M.; Doucette, C.A.; Klein, J.; Aliani, M.; Dolinsky, V.W. Maternal obesity characterized by gestational diabetes increases the susceptibility of rat offspring to hepatic steatosis via a disrupted liver metabolome. J. Physiol. 2015, 593, 3181–3197. [Google Scholar] [CrossRef]
- Ruchat, S.; Hivert, M.; Bouchard, L. Epigenetic programming of obesity and diabetes by in utero exposure to gestational diabetes mellitus. Nutr. Rev. 2013, 71, S88–S94. [Google Scholar] [CrossRef]
- Aubert, G.; Lansdorp, P.M. Telomeres and Aging. Physiol. Rev. 2008, 88, 557–579. [Google Scholar] [CrossRef]
- Epel, E.S.; Blackburn, E.H.; Lin, J.; Dhabhar, F.S.; Adler, N.E.; Morrow, J.D.; Cawthon, R.M. Accelerated telomere shortening in response to life stress. Proc. Natl. Acad. Sci. USA 2004, 101, 17312–17315. [Google Scholar] [CrossRef]
- Blasco, M.A. Telomeres and human disease: Ageing, cancer and beyond. Nat. Rev. Genet. 2005, 6, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Li, H.; Chung, A.C.K.; Xiang, L.; Li, X.; Zheng, Y.; Luan, H.; Zhu, L.; Liu, W.; Peng, Y.; et al. Large-scale longitudinal metabolomics study reveals different trimester-specific alterations of metabolites in relation to gestational diabetes mellitus. J. Proteome Res. 2018, 18, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Huo, X.; Cao, Y.; Li, S.; Du, Z.; Shao, P.; Leng, J.; Zhang, C.; Sun, X.; Ma, R.C.W.; et al. Bile acid metabolites in early pregnancy and risk of gestational diabetes in Chinese women: A nested case-control study. EBioMedicine 2018, 35, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Gelaye, B.; Clish, C.B.; Denis, M.; Larrabure, G.; Tadesse, M.G.; Deik, A.; Pierce, K.; Bullock, K.; Dennis, C.; Enquobahrie, D.A.; et al. Metabolomics signatures associated with an oral glucose challenge in pregnant women. Diabetes Metab. 2018, 45, 39–46. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, K.; Alexander, J.; Azuma, R.; Xiao, R.; Snyder, N.; Mesaros, C.; Blair, I.; Pinney, S. Gestational diabetes alters the metabolomic profile in 2nd trimester amniotic fluid in a sex-specific manner. Int. J. Mol. Sci. 2018, 19, 2696. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Reichetzeder, C.; Prehn, C.; von Websky, K.; Slowinski, T.; Chen, Y.; Yin, L.; Kleuser, B.; Yang, X.; Adamski, J.; et al. Fetal serum metabolites are independently associated with gestational diabetes mellitus. Cell. Physiol. Biochem. 2018, 45, 625–638. [Google Scholar] [CrossRef]
- Han, H.; Kang, G.; Kim, J.S.; Choi, B.H.; Koo, S. Regulation of glucose metabolism from a liver-centric perspective. Exp. Mol. Med. 2016, 48, e218. [Google Scholar] [CrossRef]
- Barker, D.J.P. The origins of the developmental origins theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Hocher, B. More than genes: The advanced fetal programming hypothesis. J. Reprod. Immunol. 2014, 104, 8–11. [Google Scholar] [CrossRef]
- Gauster, M.; Desoye, G.; Tötsch, M.; Hiden, U. The placenta and gestational diabetes mellitus. Curr. Diabetes Rep. 2012, 12, 16–23. [Google Scholar] [CrossRef]
- Wang, K.; Jin, X.; Li, Q.; Sawaya, A.C.H.F.; Le Leu, R.K.; Conlon, M.A.; Wu, L.; Hu, F. Propolis from Different Geographic Origins Decreases Intestinal Inflammation and Bacteroides spp. Populations in a Model of DSS-Induced Colitis. Mol. Nutr. Food Res. 2018, 62, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Liu, Y.; Ren, G.; Yin, L.; Liang, X.; Geng, T.; Dang, H.; An, R. Resveratrol limits diabetes-associated cognitive decline in rats by preventing oxidative stress and inflammation and modulating hippocampal structural synaptic plasticity. Brain Res. 2016, 1650, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.B.; Backlund, L.; Wegener, G.; Mathe, A.A.; Lavebratt, C. Telomerase dysregulation in the hippocampus of a rat model of depression: Normalization by lithium. Int. J. Neuropsychopharmacol. 2015, 18, 1–8. [Google Scholar] [CrossRef]
- Kaijser, M.; EdstedtBonamy, A.K.; Akre, O.; Cnattingius, S.; Granath, F.; Norman, M.; Ekbom, A. Perinatal Risk Factors for Diabetes in Later Life. Diabetes 2009, 58, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Gastaldelli, A.; Cusi, K.; Pettiti, M.; Hardies, J.; Miyazaki, Y.; Berria, R.; Buzzigoli, E.; Sironi, A.M.; Cersosimo, E.; Ferrannini, E.; et al. Relationship Between Hepatic/Visceral Fat and Hepatic Insulin Resistance in Nondiabetic and Type 2 Diabetic Subjects. Gastroenterology 2007, 133, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Aref, A.M.; Ahmed, O.M.; Ali, L.A.; Semmler, M. Maternal rat diabetes mellitus deleteriously affects insulin sensitivity and beta-cell function in the offspring. J. Diabetes Res. 2013, 2013, 429154. [Google Scholar] [CrossRef] [PubMed]
- Ding, G.; Wang, F.; Shu, J.; Tian, S.; Jiang, Y.; Zhang, D.; Wang, N.; Luo, Q.; Zhang, Y.; Jin, F.; et al. Transgenerational glucose intolerance with Igf2/H19 epigenetic alterations in mouse islet induced by intrauterine hyperglycemia. Diabetes 2012, 61, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Kamel, M.A.; Helmy, M.H.; Hanafi, M.Y.; Mahmoud, S.A.; Abo Elfetooh, H. Impaired peripheral glucose sensing in F1 offspring of diabetic pregnancy. J. Physiol. Biochem. 2014, 70, 685–699. [Google Scholar] [CrossRef]
- Malcolm, J.C.; Lawson, M.L.; Gaboury, I.; Lough, G.; Keely, E. Glucose tolerance of offspring of mother with gestational diabetes mellitus in a low-risk population. Diabet. Med. 2006, 23, 565–570. [Google Scholar] [CrossRef]
- Franks, P.W.; Looker, H.C.; Kobes, S.; Touger, L.; Tataranni, P.A.; Hanson, R.L.; Knowler, W.C. Gestational glucose tolerance and risk of type 2 diabetes in young Pima Indian offspring. Diabetes 2006, 55, 460–465. [Google Scholar] [CrossRef][Green Version]
- Dabelea, D.; Knowler, W.C.; Pettitt, D.J. Effect of diabetes in pregnancy on offspring: Follow-up research in the Pima Indians. J. Matern.-Fetal Med. 2000, 9, 83–88. [Google Scholar]
- Moussa, S.A. Oxidative stress in diabetes mellitus. Romanian. J. Biophys. 2008, 18, 225–236. [Google Scholar]
- Ouassila, A.; Saad, S.; Meriem, K.; Amira, M.; Imen, M. The investigation of the oxidative stress-related parameters in type 2 diabetes mellitue. Can. J. Diabetes 2015, 39, 44–49. [Google Scholar]
- Weitao, C.; Dandan, R.; Yuanhu, X.; Chao, N.; Youli, T.; Yang, W.; Kungao, Z.; Lu, C.; Litai, J.; Yi, T. Cardiac-specific overexpression of catalase prevents diabetes-induced pathological changes by inhibiting NF-κB signaling activation in the heart. J. Mol. Cell. Cardiol. 2015, 89, 314–325. [Google Scholar]
- Akash, M.S.H.; Rehman, K.; Liaqat, A. Tumor necrosis factor-alpha: Role in development of insulin resistance and pathogenesis of type 2 diabetes mellitus. J. Cell. Biochem. 2018, 119, 105–110. [Google Scholar] [CrossRef]
- Cai, D.; Yuan, M.; Frantz, D.F.; Melendez, P.A.; Hansen, L.; Lee, J.; Shoelson, S.E. Local and systemic insulin resistance resulting from hepatic activation of IKK-beta andNF-kappa B. Nat. Med. 2005, 11, 183–190. [Google Scholar] [CrossRef]
- Vasconcelos-Moreno, M.P.; Fries, G.R.; Gubert, C.; dos Santos, B.T.M.Q.; Fijtman, A.; Sartori, J.; Ferrari, P.; Grun, L.K.; Parisi, M.M.; Guma, F.T.C.R.; et al. Telomere length, oxidative stress, inflammation and BDNF levels in siblings of patients with bipolar disorder: Implications for accelerated cellular aging. Int. J. Neuropsychopharmacol. 2017, 6, 445–454. [Google Scholar] [CrossRef]
- Zhang, J.; Rane, G.; Dai, X.; Shanmugam, M.K.; Arfuso, F.; Samy, R.P.; Lai, M.K.P.; Kappei, D.; Kumar, A.P.; Sethi, G. Aging and the telomere connections: An intimate relationship with inflammation. Ageing Res. Rev. 2016, 25, 55–69. [Google Scholar] [CrossRef]
- Xu, J.; Ye, J.; Wu, Y.; Zhang, H.; Luo, Q.; Han, C.; Ye, X.; Wang, H.; He, J.; Huang, H.; et al. Reduced fetal telomere length in gestational diabetes. PLoS ONE 2014, 9, e86161. [Google Scholar] [CrossRef]
- Cross, J.A.; Brennan, C.; Gray, T.; Temple, R.C.; Dozio, N.; Hughes, J.C.; Levell, N.J.; Murphy, H.; Fowler, D.; Hughes, D.A.; et al. Absence of telomere shortening and oxidative DNA damage in the young adult offspring of women with pre-gestational type 1 diabetes. Diabetologia 2009, 52, 226–234. [Google Scholar] [CrossRef][Green Version]
- Gilfillan, C.; Naidu, P.; Gunawan, F.; Hassan, F.; Tian, P.; Elwood, N. Leukocyte telomere length in the neonatal offspring of mothers with gestational and pre-gestational diabetes. PLoS ONE 2016, 11, e163824. [Google Scholar] [CrossRef]
- Chavez, J.A.; Summers, S.A. A Ceramide-centric view of insulin resistance. Cell Metab. 2012, 15, 585–594. [Google Scholar] [CrossRef]
- Pardo, V.; González-Rodríguez, Á.; Muntané, J.; Kozma, S.C.; Valverde, Á.M. Role of hepatocyte S6K1 in palmitic acid-induced endoplasmic reticulum stress, lipotoxicity, insulin resistance and in oleic acid-induced protection. Food Chem. Toxicol. 2015, 80, 298–309. [Google Scholar] [CrossRef]
- Boden, G.; Chen, X.; Stein, T.P. Gluconeogenesis in moderately and severely hyperglycemic patients with type 2 diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E23–E30. [Google Scholar] [CrossRef]
- De Weerth, C.; Zijl, R.H.; Buitelaar, J.K. Development of cortisol circadian rhythm in infancy. Early Hum. Dev. 2003, 73, 39–52. [Google Scholar] [CrossRef]
- Phillips, D.I.; Barker, D.J.; Fall, C.H.; Seckl, J.R.; Whorwood, C.B.; Wood, P.J.; Walker, B.R. Elevated plasma cortisol concentrations: A link between low birth weight and the insulin resistance syndrome? J. Clin. Endocrinol. Metab. 1998, 83, 757–760. [Google Scholar]
- Wu, J.H.; Micha, R.; Imamura, F.; Pan, A.; Biggs, M.L.; Ajaz, O.; Djousse, L.; Hu, F.B.; Mozaffarian, D. Omega-3 fatty acids and incident type 2 diabetes: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, S214–S227. [Google Scholar] [CrossRef]
- Muramatsu, T.; Yatsuya, H.; Toyoshima, H.; Sasaki, S.; Li, Y.; Otsuka, R.; Wada, K.; Hotta, Y.; Mitsuhashi, H.; Matsushita, K. Higher dietary intake of alpha-linolenic acid is associated with lower insulin resistance in middle-aged Japanese. Prev. Med. 2010, 50, 272–276. [Google Scholar] [CrossRef]
- Djoussé, L.; Biggs, M.L.; Lemaitre, R.N.; King, I.B.; Song, X.; Ix, J.H.; Mukamal, K.J.; Siscovick, D.S.; Mozaffarian, D. Plasma omega-3 fatty acids and incident diabetes in older adults. Am. J. Clin. Nutr. 2011, 94, 527–533. [Google Scholar] [CrossRef]
- Behme, M.T. Nicotinamide and diabetes prevention. Nutr. Rev. 1995, 53, 137–139. [Google Scholar] [CrossRef]
- Olmos, P.R.; Hodgson, M.I.; Maiz, A.; Manrique, M.; De Valdés, M.D.; Foncea, R.; Acosta, A.M.; Emmerich, M.V.; Velasco, S.; Muñiz, O.P.; et al. Nicotinamide protected first-phase insulin response (FPIR) and prevented clinical disease in first-degree relatives of type-1 diabetics. Diabetes Res. Clin. Pract. 2006, 71, 320–333. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, B.A.; Harmon, B.V.; Cameron, D.P.; Allan, D.J. Nicotinamide prevents the development of diabetes in the cyclophosphamide-induced NOD mouse model by reducing beta-cell apoptosis. J. Pathol. 2000, 191, 86–92. [Google Scholar] [CrossRef]
- Baliarsingh, S.; Beg, Z.H.; Ahmad, J. The therapeutic impacts of tocotrienols in type 2 diabetic patients with hyperlipidemia. Atherosclerosis 2005, 182, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Kuhad, A.; Bishnoi, M.; Tiwari, V.; Chopra, K. Suppression of NF-κβ signaling pathway by tocotrienol can prevent diabetes associated cognitive deficits. Pharmacol. Biochem. Behav. 2009, 92, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Erdinest, N.; Shmueli, O.; Grossman, Y.; Ovadia, H.; Solomon, A. Anti-inflammatory effects of alpha linolenic acid on human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2012, 53, 4396–4406. [Google Scholar] [CrossRef]
- Boon, J.; Hoy, A.J.; Stark, R.; Brown, R.D.; Meex, R.C.; Henstridge, D.C.; Schenk, S.; Meikle, P.J.; Horowitz, J.F.; Kingwell, B.A.; et al. Ceramides contained in LDL are elevated in type 2 diabetes and promote inflammation and skeletal muscle insulin resistance. Diabetes 2013, 62, 401–410. [Google Scholar] [CrossRef]
- Berglund, T.; Wallström, A.; Nguyen, T.V.; Laurell, C.; Ohlsson, A.B. Nicotinamide: Antioxidative and DNA hypomethylation effects in plant cells. Plant Physiol. Biochem. 2017, 118, 551–560. [Google Scholar] [CrossRef]
- Ying, W.; Min, Z. Nicotinamide improves sevoflurane-induced cognitive impairment through suppression of inflammation and anti-apoptosis in rat. Int. J. Clin. Exp. Med. 2015, 8, 20079–20085. [Google Scholar]
- Abu-Bakar, A.; Hakkola, J.; Juvonen, R.; Rahnasto-Rilla, M.; Raunio, H.; A. Lang, M. Function and regulation of the Cyp2a5/CYP2A6 genes in response to toxic insults in the liver. Curr. Drug Metab. 2013, 14, 137–150. [Google Scholar] [CrossRef]
- Skowrońska, M.; Albrecht, J. Alterations of blood brain barrier function in hyperammonemia: An overview. Neurotox. Res. 2012, 21, 236–244. [Google Scholar] [CrossRef]



| 3 Week (Weaning) | Con | GDM |
|---|---|---|
| Fasting glucose (mmol/L) | 6.17 ± 1.24 | 6.01 ± 1.3 |
| Postprandial glucose (mmol/L) | 7.34 ± 0.61 | 7.57 ± 0.49 |
| Fasting insulin (mU/L) | 12.01 ± 0.59 | 9.7 ± 0.46 * |
| Postprandial insulin (mU/L) | 13.23 ± 0.78 | 20.17 ± 1.45 ** |
| TG (mmol/L) | 0.45 ± 0.1 | 0.46 ± 0.11 |
| TC (mmol/L) | 1.76 ± 0.19 | 1.98 ± 0.16 |
| 11 Months old | Con | GDM | n-3 Adq-GDM | n-3 Def-GDM |
|---|---|---|---|---|
| Glucose (mmol/L) | 6.05 ± 0.37 | 8.39 ± 2.07 | 6.3 ± 0.54 | 8.38 ± 1.66 |
| Insulin (mU/L) | 11.73 ± 0.55 | 8.03 ± 0.47 ** | 10 ± 0.46 *## | 7.8 ± 0.78 ** |
| TG (mmol/L) | 0.79 ± 0.13 | 0.63 ± 0.19 | 0.43 ± 0.19 *# | 0.61 ± 0.19 |
| TC (mmol/L) | 1.87 ± 0.04 | 1.79 ± 0.36 | 0.69 ± 0.16 **## | 1.7 ± 0.17 |
| HDL (mmol/L) | 0.53 ± 0.03 | 0.52 ± 0.18 | 0.27 ± 0.04 **## | 0.47 ± 0.03 |
| TC/HDL | 3.52 ± 0.12 | 3.44 ± 0.66 | 2.55 ± 0.28 **## | 3.62 ± 0.19 |
| Identification | RT (Min) | m/z | Changing Trend | Significance | ||
|---|---|---|---|---|---|---|
| GDM vs. Con | n-3adq vs. GDM | n-3def vs. GDM | ||||
| Ceramide (d18:1/16:0) | 17.78 | 560.5073 | ↑ | ↓* | - | Biomarker for diabetes; impair insulin signaling and cause insulin resistance; increase oxidative stress; promote inflammation; contribute to non-alcohol fatty liver disease |
| Tetrahydro-11-deoxycortisol | 11.56 | 337.2756 | ↑ | ↓** | - | Impact cortisol and further impact insulin production and glucose metabolism; inhibit glycogen synthesis; cause insulin resistance |
| 9’-Carboxy-γ-tocotrienol | 16.50 | 395.2234 | ↓ | - | ↓** | Antioxidant effect; improve glycemic control; prevent hyperlipidemia; suppress inflammation |
| α-Linolenic acid | 11.93 | 570.3029 | ↓ | - | ↓** | Decreases diabetic risk; improve insulin resistance; improve oxidative stress and inflammation; regulate lipid metabolism; improve non-alcohol fatty liver disease |
| Hexadecenoic acid | 15.99 | 271.2649 | ↑ | - | - | Induce endoplasmic reticulum stress and insulin resistance; lipotoxicity; enhance oxidative stress and inflammation; contribute to non-alcohol fatty liver disease |
| Niacinamide | 0.95 | 123.0515 | ↓ | - | - | Prevent diabetes; protect β cell; antioxidative role; anti-inflammatory effect |
| Oxalacetic acid | 18.17 | 154.9962 | ↓ | - | ↓* | Impact citric acid cycle and glucose and lipid metabolism; decrease of it indicate gluconeogenesis |
| Phenylethylamine | 2.52 | 122.0956 | ↑ | - | ↑** | Indicate possibility of hepatic damage and hepatic encephalopathy |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, J.; Xiao, H.; Li, J.; Guo, X.; Cai, W.; Li, D. n-3 Polyunsaturated Fatty Acids Decrease Long-Term Diabetic Risk of Offspring of Gestational Diabetes Rats by Postponing Shortening of Hepatic Telomeres and Modulating Liver Metabolism. Nutrients 2019, 11, 1699. https://doi.org/10.3390/nu11071699
Gao J, Xiao H, Li J, Guo X, Cai W, Li D. n-3 Polyunsaturated Fatty Acids Decrease Long-Term Diabetic Risk of Offspring of Gestational Diabetes Rats by Postponing Shortening of Hepatic Telomeres and Modulating Liver Metabolism. Nutrients. 2019; 11(7):1699. https://doi.org/10.3390/nu11071699
Chicago/Turabian StyleGao, Jinlong, Hailong Xiao, Jiaomei Li, Xiaofei Guo, Wenwen Cai, and Duo Li. 2019. "n-3 Polyunsaturated Fatty Acids Decrease Long-Term Diabetic Risk of Offspring of Gestational Diabetes Rats by Postponing Shortening of Hepatic Telomeres and Modulating Liver Metabolism" Nutrients 11, no. 7: 1699. https://doi.org/10.3390/nu11071699
APA StyleGao, J., Xiao, H., Li, J., Guo, X., Cai, W., & Li, D. (2019). n-3 Polyunsaturated Fatty Acids Decrease Long-Term Diabetic Risk of Offspring of Gestational Diabetes Rats by Postponing Shortening of Hepatic Telomeres and Modulating Liver Metabolism. Nutrients, 11(7), 1699. https://doi.org/10.3390/nu11071699