Biological Effect of Soy Isoflavones in the Prevention of Civilization Diseases
Abstract
1. Introduction
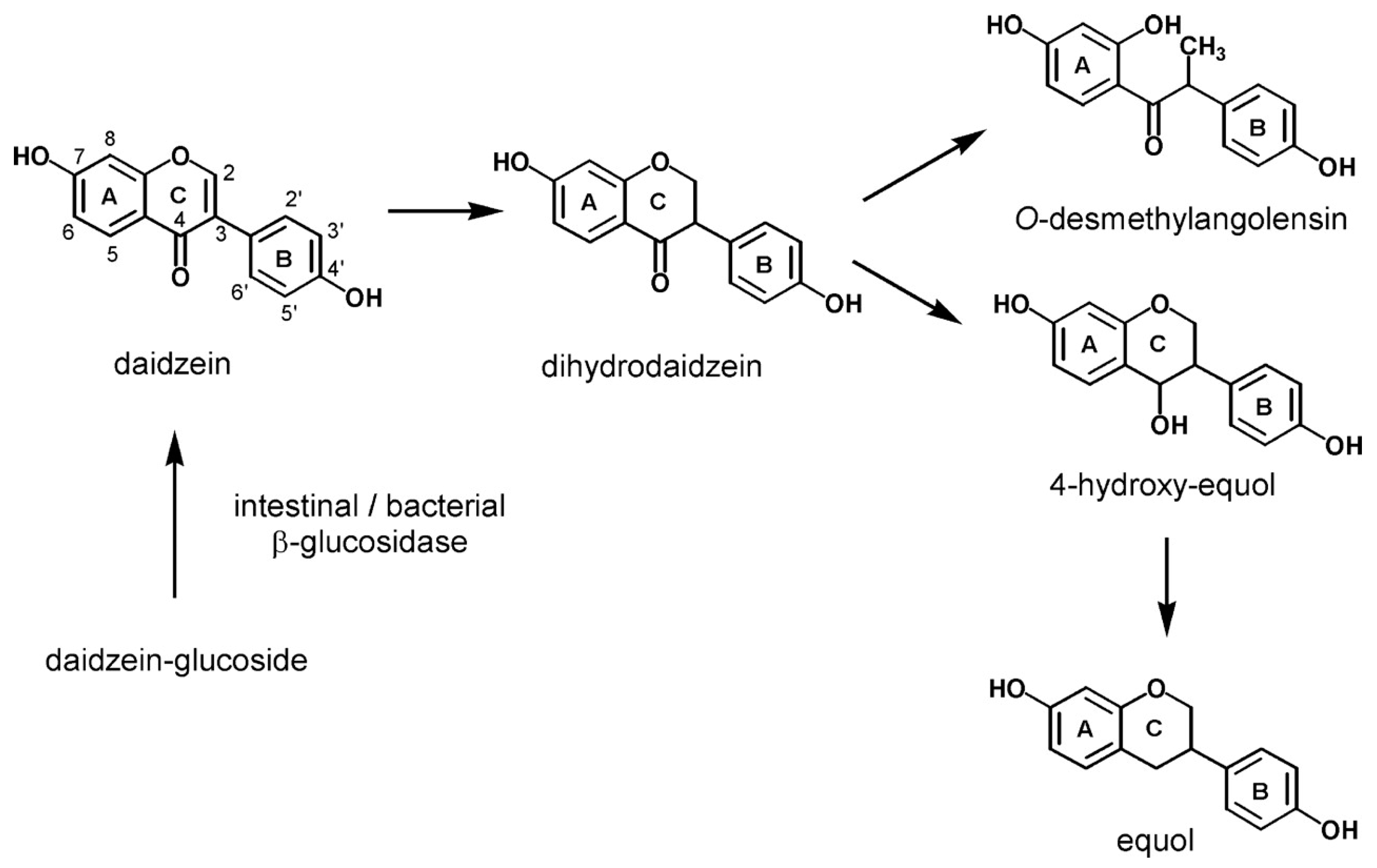
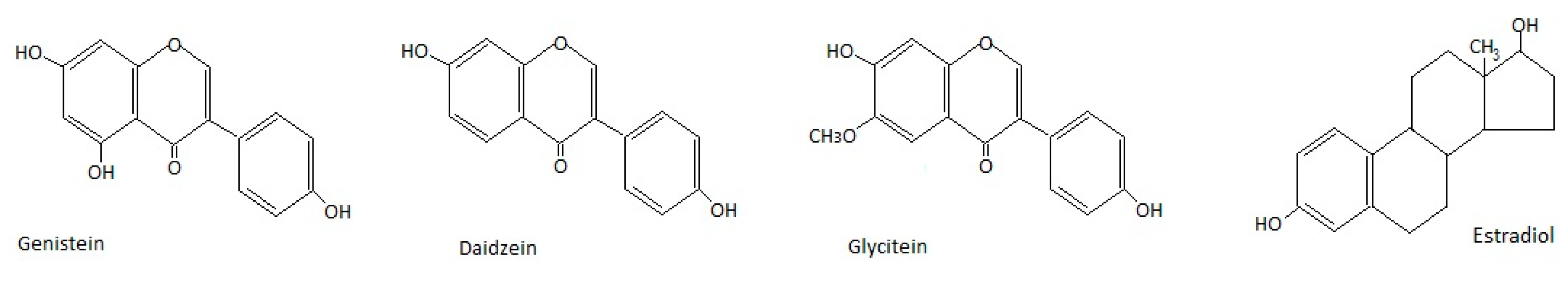
2. Metabolism of Isoflavones
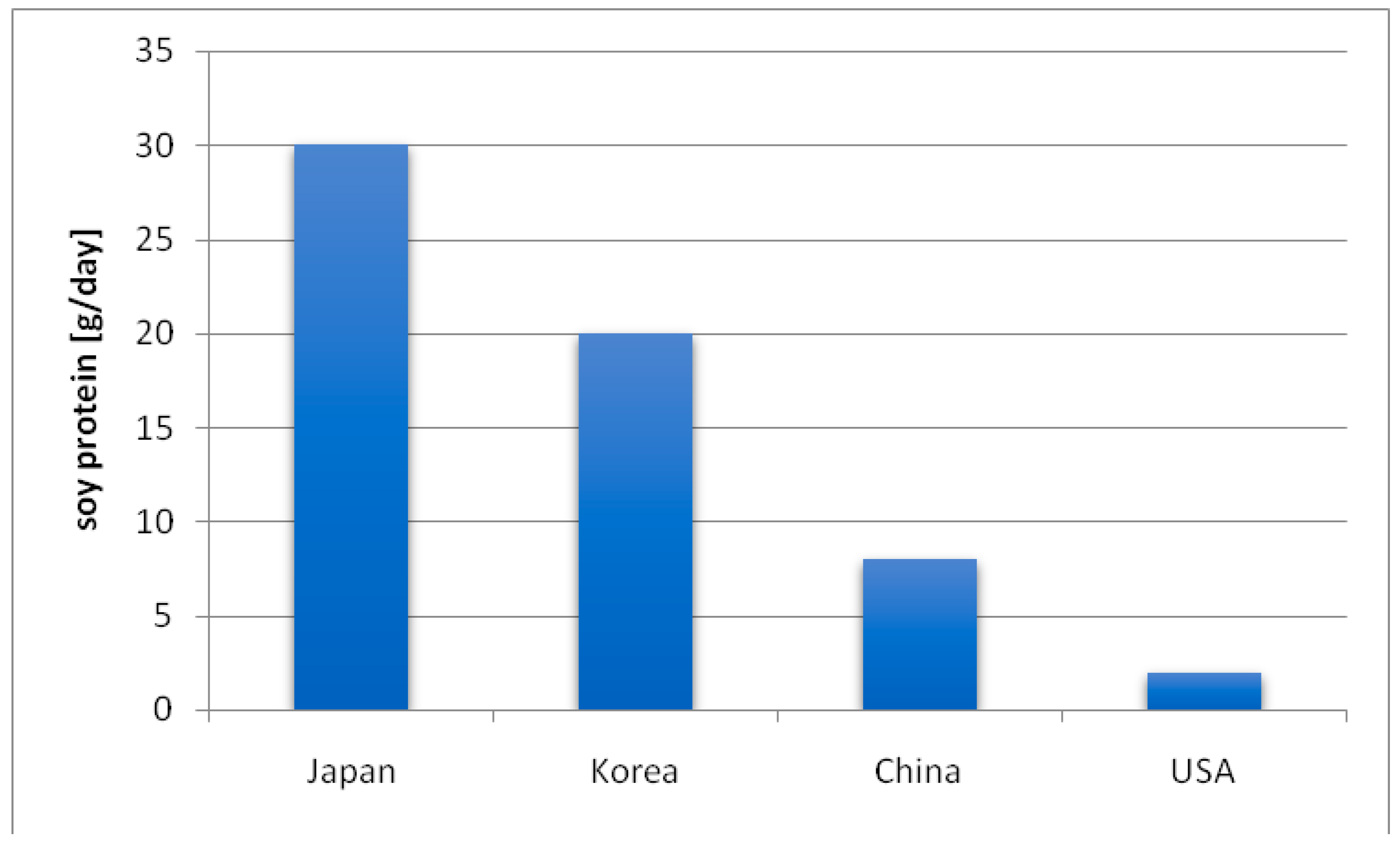
3. Consumption of Soy
4. Isoflavones in Type II Diabetes Mellitus (DM)
5. Isoflavones and Osteoporosis
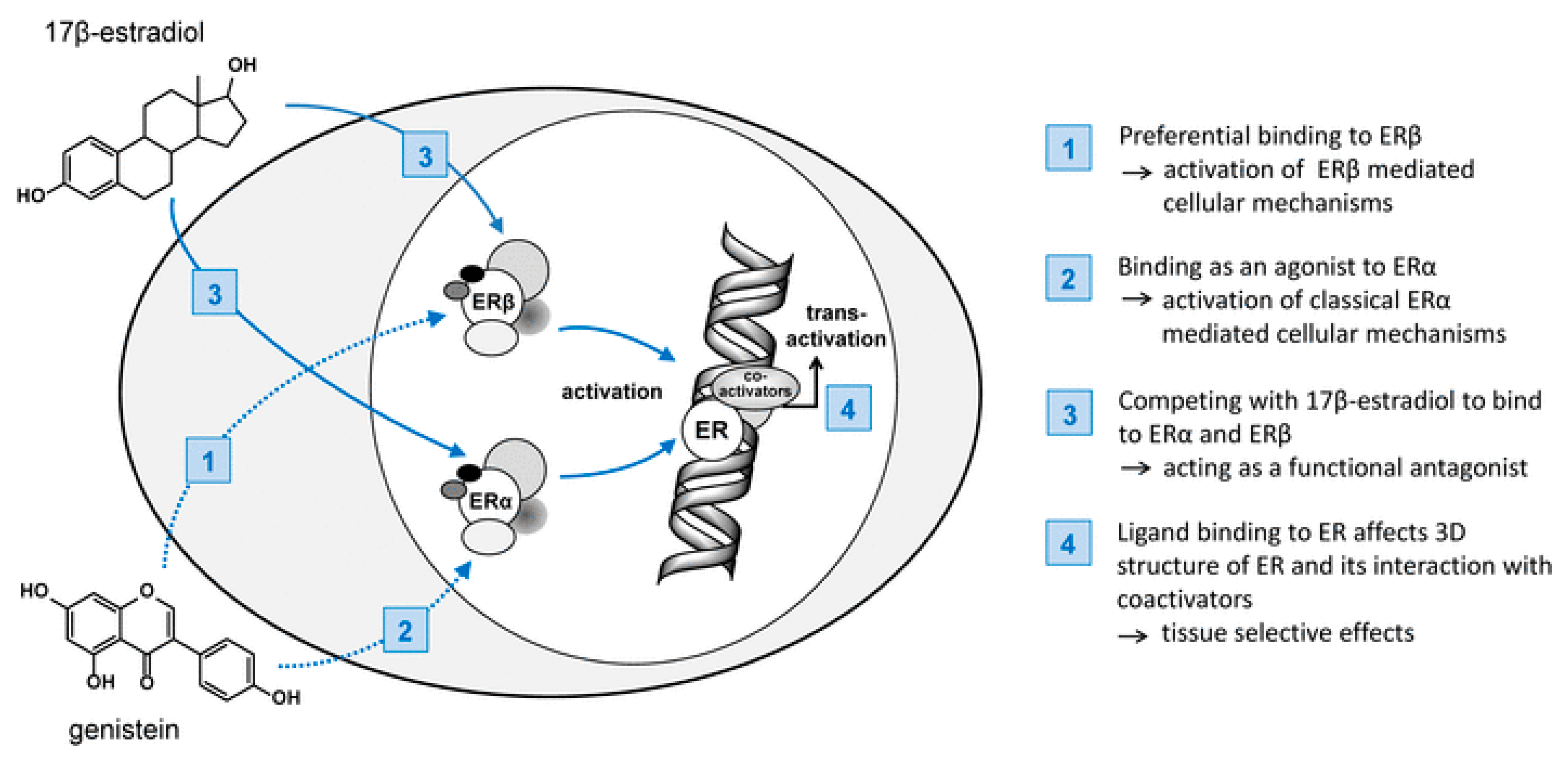
6. Hormone-Associated Cancers
7. Effect on Cardiovascular Disease
8. Conclusions
Author Contributions
Conflicts of Interest
References
- Kitajewska, W.; Szeląg, W.; Kopański, Z.; Maslyak, Z.; Sklyarov, I. Choroby cywilizacyjne i ich prewencja. J. Clin. Healthc. 2014, 1, 3–7. [Google Scholar]
- Czerwiecki, L. Współczesne poglądy na rolę przeciwutleniaczy roślinnych w profilaktyce chorób cywilizacyjnych. Rocz. Państ. Zakł. Hig. 2009, 60, 201–206. Available online: http://agro.icm.edu.pl/agro/element/bwmeta1.element.dl-catalog-2dabcbf8-1c2c-4cf3-ab91-daf86239ec07 (accessed on 30 June 2019).
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Paszkiewicz, M.; Budzyńska, A.; Różalska, B.; Sadowska, B. Immunomodulacyjna rola polifenoli roślinnych. Post. Hig. Med. Dośw. 2012, 66, 637–646. [Google Scholar] [CrossRef]
- Koszowska, A.; Dittfeld, A.; Puzoń-Brończyk, A.; Nowak, J.; Zubelewicz-Szkodzińska, B. Polifenole w profilaktyce chorób cywilizacyjnych. Post. Fitoter. 2013, 4, 263–266. Available online: http://www.czytelniamedyczna.pl/4658,polifenole-w-profilaktyce-chorob-cywilizacyjnych.html (accessed on 30 June 2019).
- Sathyapalan, T.; Aye, M.; Rigby, A.; Thatcher, N.J.; Dargham, S.R.; Kilpatrick, E.S.; Atkin, S.L. Soy isoflavones improve cardiovascular disease risk markers in women during the early menopause. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Abdelrazek, H.M.; Mahmoud, M.M.; Tag, H.M.; Greish, S.M.; Eltamany, D.A.; Soliman, M.T. Soy Isoflavones Ameliorate Metabolic and Immunological Alterations of Ovariectomy in Female Wistar Rats: Antioxidant and Estrogen Sparing Potential. Oxid. Med. Cell Longev. 2019, 2019, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.M.; Silva, T.E.; Lemes, A.C.; Boldrin, M.C.F.; Silva Pereira, M.A.; Silva, F.G.; Egea, M.B. Okara: A soybean by-product as an alternative to enrich vegetable paste. LWT Food Sci. Technol. 2018, 92, 593–599. [Google Scholar] [CrossRef]
- Cederroth, C.R.; Nef, S. Soy, phytoestrogens and metabolism: A review. Mol. Cell. Endocrinol. 2009, 304, 30–42. [Google Scholar] [CrossRef]
- Das, D.; Sarkar, S.; Bordoloi, J.; Wann, S.B.; Kalita, J.; Manna, P. Daidzein, its effects on impaired glucose and lipid metabolism and vascular inflammation associated with type 2 diabetes. BioFactors 2018, 44, 407–417. [Google Scholar] [CrossRef]
- Hüser, S.; Guth, S.; Joost, H.G.; Soukup, S.T.; Köhrle, J.; Kreienbrock, L.; Diel, P.; Lachenmeier, D.W.; Eisenbrand, G.; Vollmer, G.; et al. Effects of isoflavones on breast tissue and the thyroid hormone system in humans: A comprehensive safety evaluation. Arch. Toxicol. 2018, 92, 2703–2748. [Google Scholar] [CrossRef]
- Bhathena, S.J.; Velasquez, M.T. Beneficial role of dietary phytoestrogens in obesity and diabetes. Am. J. Clin. Nutr. 2002, 76, 1191–1201. [Google Scholar] [CrossRef]
- Sivoňová, M.K.; Kaplán, P.; Tatarková, Z.; Lichardusová, L.; Dušenka, R.; Jurečeková, J. Androgen receptor and soy isoflavones in prostate cancer (Review). Mol. Clin. Oncol. 2019, 10, 191–204. [Google Scholar] [CrossRef]
- Kalaiselvan, V.; Kalaivani, M.; Vijayakumar, A.; Sureshkumar, K.; Venkateskumar, K. Current knowledge and future direction of research on soy isoflavones as a therapeutic agents. Pharmacogn. Rev. 2010, 4, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Rüfer, C.E.; Glatt, H.; Kulling, S.E. Structural elucidation of hydroxylated metabolites of the isoflavan equol by gas chromatography-mass spectrometry and high-performance liquid chromatography-mass spectrometry. Drug Metab. Dispos. 2006, 34, 51–60. [Google Scholar] [CrossRef]
- Messina, M. Soy and health update: Evaluation of the clinical and epidemiologic literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef] [PubMed]
- Castelo-Branco, C.; Hidalgo, M.J.C. Isoflavones: Effects on bone health. Climacteric 2011, 14, 204–211. [Google Scholar] [CrossRef]
- Soy Isoflavones. Available online: https://lpi.oregonstate.edu/mic/dietary-factors/phytochemicals/soy-isoflavones (accessed on 22 June 2019).
- Anderson, J.J.; Anthony, M.S.; Cline, J.M.; Washburn, S.A.; Garner, S.C. Health potential of soy isoflavones for menopausal women. Publ. Health Nutr. 1999, 2, 489–504. [Google Scholar] [CrossRef]
- Xiao, C.W. Health Effects of Soy Protein and Isoflavones in Humans. J. Nutr. 2008, 138, 1244S–1249S. [Google Scholar] [CrossRef]
- Klein, C.B.; King, A.A. Genistein genotoxicity: Critical considerations of in vitro exposure dose. Toxicol. Appl. Pharmacol. 2007, 224, 1–11. [Google Scholar] [CrossRef]
- Rizzo, G.; Baroni, L. Soy, Soy Foods and Their Role in Vegetarian Diets. Nutrients 2018, 10, 43. [Google Scholar] [CrossRef]
- Zheng, X.L.S.; Lee, S.K.; Chun, O.K. Soy isoflavones and osteoporotic bone loss: A review with an emphasis on modulation of bone remodeling. J. Med. Food 2016, 19, 1–14. [Google Scholar] [CrossRef]
- Chen, M.; Rao, Y.; Zheng, Y.; Wei, S.; Li, Y.; Guo, T.; Yin, P. Association between soy isoflavone intake and breast cancer risk for pre- and post-menopausal women: A meta-analysis of epidemiological studies. PLoS ONE 2014, 9, e89288. [Google Scholar] [CrossRef]
- Jayagopal, V.; Albertazzi, P.; Kilpatrick, E.S.; Howarth, E.M.; Jennings, P.E.; Hepburn, D.A.; Jennings, P.E.; Hepburn, D.A.; Atkins, S.L. Beneficial effects of soy phytoestrogen intake in postmenopausal women with type 2 diabetes. Diabetes Care 2002, 25, 1709–1714. [Google Scholar] [CrossRef]
- Ademiluyi, A.O.; Oboh, G. Soybean phenolic-rich extracts inhibit key-enzymes linked to type 2 diabetes (α-amylase and α-glucosidase) and hypertension (angiotensin I converting enzyme) in vitro. Exp. Toxicol. Pathol. 2013, 65, 305–309. [Google Scholar] [CrossRef]
- Mezei, O.; Banz, W.J.; Steger, R.W.; Peluso, M.R.; Winters, T.A.; Shay, N. Soy Isoflavones Exert Antidiabetic and Hypolipidemic Effects through the PPAR Pathways in Obese Zucker Rats and Murine RAW 264.7 Cells. J. Nutr. 2003, 133, 1238–1243. [Google Scholar] [CrossRef]
- Ding, M.; Franke, A.A.; Rosner, B.A.; Giovannucci, E.; van Dam, R.M.; Tworoger, S.S.; Hu, F.B.; Sun, Q. Urinary isoflavonoids and risk of type 2 diabetes: A prospective investigation in US women. Br. J. Nutr. 2015, 114, 1694–1701. [Google Scholar] [CrossRef][Green Version]
- Duru, K.C.; Kovaleva, E.G.; Danilova, I.G.; van der Bijl, P.; Belousova, A.V. The potential beneficial role of isoflavones in type 2 diabetes mellitus. Nutr. Res. 2018, 59, 1–15. [Google Scholar] [CrossRef]
- Nguyen, C.T.; Pham, N.M.; Do, V.V.; Binns, C.W.; Hoang, V.M.; Dang, D.A.; Lee, A.H. Soyfood and isoflavone intake and risk of type 2 diabetes in Vietnamese adults. Eur. J. Clin. Nutr. 2017, 71, 1186–1192. [Google Scholar] [CrossRef]
- Chi, X.X.; Zhang, T.; Zhang, D.J.; Yu, W.; Wang, Q.Y.; Zhen, J.L. Effects of isoflavones on lipid and apolipoprotein levels in patients with type 2 diabetes in Heilongjiang Province in China. J. Clin. Biochem. Nutr. 2016, 59, 134–138. [Google Scholar] [CrossRef]
- Ho, S.C.; Chen, Y.M.; Ho, S.S.; Woo, J.L. Soy isoflavone supplementation and fasting serum glucose and lipid profile among postmenopausal Chinese women: A double-blind, randomized, placebo-controlled trial. Menopause 2007, 14, 905–912. [Google Scholar] [CrossRef]
- Li, Z.; Hong, K.; Saltsman, P.; DeShields, S.; Bellman, M.; Thames, G.; Liu, Y.; Wang, H.J.; Elashoff, R.; Heber, D. Long-term efficacy of soy-based meal replacements vs an individualized diet plan in obese type II DM patients: Relative effects on weight loss, metabolic parameters, and Creactive protein. Eur. J. Clin. Nutr. 2005, 59, 411–418. [Google Scholar] [CrossRef]
- Villegas, R.; Gao, Y.T.; Yang, G.; Li, H.L.; Elasy, T.A.; Zheng, W.; Shu, X.O. Legume and soy food intake and the incidence of type 2 diabetes in the Shanghai Women’s Health Study. Am. J. Clin. Nutr. 2008, 87, 162–167. [Google Scholar] [CrossRef]
- Charles, C.; Yuskavage, J.; Carlson, O.; John, M.; Tagalicud, A.S.; Maggio, M.; Muller, D.C.; Egan, J.; Basaria, S. Effects of high-dose isoflavones on metabolic and inflammatory markers in healthy postmenopausal women. Menopause 2009, 16, 395–400. [Google Scholar] [CrossRef]
- Talaei, M.; Lee, B.L.; Ong, C.N.; van Dam, R.M.; Jian, M.Y.J.M.; Koh, W.P.; Pan, A. Urine phyto-oestrogen metabolites are not significantly associated with risk of type 2 diabetes: The Singapore Chinese health study. Br. J. Nutr. 2016, 115, 1607–1615. [Google Scholar] [CrossRef]
- Janiszewska, M.; Kulik, T.; Dziedzic, M. Osteoporoza jako problem społeczny—Patogeneza, objawy i czynniki ryzyka osteoporozy pomenopauzalnej. Probl. Hig. Epidemiol. 2015, 96, 106–114. [Google Scholar]
- Wang, Q.; Ge, X.; Tian, X.; Zhang, Y.; Zhang, J.; Zhang, P. Soy isoflavone: The multipurpose phytochemical. Biomed. Rep. 2013, 1, 697–701. [Google Scholar] [CrossRef]
- Suchecka-Rachoń, K.; Rachoń, D. Rola hormonalnej terapii zastępczej (HTZ) u kobiet w okresie pomenopauzalnym. Chor. Serca Naczyń 2005, 2, 115–124. Available online: https://journals.viamedica.pl/choroby_serca_i_naczyn/article/view/12197 (accessed on 10 June 2019).
- Taku, K.; Melby, M.K.; Nishi, N.; Omori, T.; Kurzer, M.S. Soy isoflavones for osteoporosis: An evidence-based approach. Maturitas 2011, 70, 333–338. [Google Scholar] [CrossRef]
- Tit, D.M.; Bungau, S.; Iovan, C.; Nistor Cseppento, D.C.; Endres, L.; Sava, C.; Sabau, A.M.; Furau, G.; Furau, C. Effects of the hormone replacement therapy and of soy isoflavones on bone resorption in postmenopause. J. Clin. Med. 2018, 7, 297. [Google Scholar] [CrossRef]
- Lewandowski, B.; Kita, K.; Kita, J.; Sulik, A.; Klimiuk, P.A.; Sierakowski, S.; Domysławska, I.; Chwieśko, Z. Osteoporoza—Część 2. Badanie gęstości mineralnej kości oraz badania obrazowe w diagnostyce osteoporozy. Nowa Med. 2004, 3. Available online: http://www.czytelniamedyczna.pl/1541,osteoporoza-czesc-2-badanie-gestosci-mineralnej-kosci-oraz-badania-obrazowe-w-d.html (accessed on 10 June 2019).
- Morabito, N.; Crisafulli, A.; Vergara, C.; Gaudio, A.; Lasco, A.; Frisina, N.; D’Anna, R.; Corrado, F.; Pizzoleo, M.A.; Cincotta, M.; et al. Effects of genistein and hormone-replacement therapy on bone loss in early postmenopausal women: A randomized double-blind placebo-controlled study. J. Bone Min. Res. 2002, 17, 1904–1912. [Google Scholar] [CrossRef]
- Turhan, N.; Bolkan, F.; Iltemir, D.; Ardicoglu, Y. The effect of isoflavones on bone mass and bone remodeling markers in postmenopausal women. Turk. J. Med. Sci. 2008, 38, 145–152. [Google Scholar] [CrossRef]
- Lee, H.; Choue, R.; Lim, H. Effect of soy isoflavones supplement on climacteric symptoms, bone biomarkers, and quality of life in Korean postmenopausal women: A randomized clinical trial. Nutr. Res. Prac. 2017, 11, 223–231. [Google Scholar] [CrossRef]
- Sathyapalan, T.; Aye, M.; Rigby, A.S.; Fraser, W.D.; Thatcher, N.J.; Kilpatrick, E.S.; Atkin, S.L. Soy Reduces Bone Turnover Markers in Women During Early Menopause: A Randomized Controlled Trial. J. Bone Miner. Res. 2017, 32, 157–164. [Google Scholar] [CrossRef]
- Gherbi, E. Znaczenie fitoestrogenów roślinnych w profilaktyce osteoporozy. Post. Fitoter. 2012, 3, 192–196. [Google Scholar]
- Goluch-Koniuszy, Z.; Radziszewska, M.; Dęga, S. Ocena sposobu żywienia kobiet w okresie menopauzalnym—Zdrowych i z leczoną osteoporozą. Folia Pomer. Univ. Technol. Stetin. Agric. Aliment. Pisc. Zootech. 2009, 269, 5–18. [Google Scholar]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Varinska, L.; Gal, P.; Mojzisova, G.; Mirossay, L.; Mojzis, J. Soy and breast cancer: Focus on angiogenesis. Int. J. Mol. Sci. 2015, 16, 11728–11749. [Google Scholar] [CrossRef]
- Zalega, J.; Szostak-Węgierek, D. Żywienie w profilaktyce nowotworów. Część III. Diety o właściwościach przeciwnowotworowych. Probl. Hig. Epidemiol. 2013, 94, 59–70. [Google Scholar]
- Ziaei, S.; Halaby, R. DietaryIsoflavonesand Breast CancerRisk. Medicines 2017, 4, 18. [Google Scholar] [CrossRef]
- Wada, K.; Nakamura, K.; Tamai, Y.; Tsuji, M.; Kawachi, T.; Hori, A.; Takeyama, N.; Tanabashi, S.; Matsushita, S.; Tokimitsu, N.; et al. Soy isoflavone intake and breast cancer risk in Japan: From the Takayama study. Int. J. Cancer 2013, 133, 952–960. [Google Scholar] [CrossRef]
- Shu, X.O.; Jin, F.; Dai, Q.; Wen, W.; Potter, J.D.; Kushi, L.H.; Ruan, Z.; Gao, Y.T.; Zheng, W. Soyfood intake during adolescence and subsequent risk of breast cancer among Chinese women. Cancer Epidemiol. Biomark. Prev. 2001, 10, 483–488. Available online: https://www.ncbi.nlm.nih.gov/pubmed/?term=Soyfood+intake+during+adolescence+and+subsequent+risk+of+breast+cancer+among+Chinese+women (accessed on 28 June 2019).
- Wu, A.H.; Wan, P.; Hankin, J.; Tseng, C.C.; Yu, M.C.; Pike, M.C. Adolescent and adult soy intake and risk of breast cancer in Asian-Americans. Carcinogenesis 2002, 23, 1491–1496. [Google Scholar] [CrossRef]
- Thanos, J.; Cotterchio, M.; Boucher, B.A.; Kreiger, N.; Thompson, L.U. Adolescent dietary phytoestrogen intake and breast cancer risk (Canada). Cancer Causes Control. 2006, 17, 1253–1261. [Google Scholar] [CrossRef]
- Delmanto, A.; Nahas-Neto, J.; Traiman, P.; Uemura, G.; Pessoa, E.C.; Nahas, E.A. Effects of soy isoflavones on mammographic density and breast parenchyma in postmenopausal women: A randomized, double-blind, placebo-controlled clinical trial. Menopause 2013, 20, 1049–1054. [Google Scholar] [CrossRef]
- Khan, S.A.; Chatterton, R.T.; Michel, N.; Bryk, M.; Lee, O.; Ivancic, D.; Heinz, R.; Zalles, C.M.; Helenowski, I.B.; Jovanovic, B.D.; et al. Soy isoflavone supplementation for breast cancer risk reduction: A randomized phase II trial. Cancer Prev. Res. (Phila.) 2012, 5, 309–319. [Google Scholar] [CrossRef]
- Murkes, D.; Conner, P.; Leifland, K.; Tani, E.; Beliard, A.; Lundstrom, E.; Soderqvist, G. Effects of percutaneous estradiol-oral progesterone versus oral conjugated equine estrogens-medroxyprogesterone acetate on breast cell proliferation and BCL-2 protein in healthy women. Fertil. Steril. 2011, 95, 1188–1191. [Google Scholar] [CrossRef]
- Johnson, K.A.; Vemuri, S.; Alsahafi, S.; Castillo, R.; Cheriyath, V. Glycone-rich Soy Isoflavone Extracts Promote Estrogen Receptor Positive Breast Cancer Cell Growth. Nutr. Cancer. 2016, 68, 622–633. [Google Scholar] [CrossRef]
- Applegate, C.C.; Rowles, J.L.; Ranard, K.M.; Jeon, S.; Erdman, J.W. Soy Consumption and the Risk of Prostate Cancer: An Updated Systematic Review and Meta-Analysis. Nutrients 2018, 10, 40. [Google Scholar] [CrossRef]
- Jian, L. Soy, isoflavones, and prostate cancer. Mol. Nutr. Food Res. 2009, 53, 217–226. [Google Scholar] [CrossRef]
- Rebbeck, T.R. Prostate cancer genetics: Variation by race, ethnicity, and geography. Semin. Radiat. Oncol. 2017, 27, 3–10. [Google Scholar] [CrossRef]
- Russo, G.I.; Di Mauro, M.; Regis, F.; Reale, G.; Campisi, D.; Marranzano, M.; Lo Giudice, A.; Solinas, T.; Madonia, M.; Cimino, S.; et al. Association between dietary phytoestrogens intakes and prostate cancer risk in Sicily. Aging Male 2018, 21, 48–54. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, L.; Na, R.; Xu, J.; Xiong, Z.; Zhang, N.; Dai, W.; Jiang, H.; Ding, Q. Plasma genistein and risk of prostate cancer in Chinese population. Int. Urol. Nephrol. 2015, 47, 965–970. [Google Scholar] [CrossRef]
- Nagata, Y.; Sugiyama, Y.; Fukuta, F.; Takayanagi, A.; Masumori, N.; Tsukamoto, T.; Akasaka, H.; Ohnishi, H.; Saitoh, S.; Miura, T.; et al. Relationship of serum levels and dietary intake of isoflavone, and the novel bacterium Slackia sp. strain NATTS with the risk of prostate cancer: A case-control study among Japanese men. Int. Urol. Nephrol. 2016, 48, 1453–1460. [Google Scholar] [CrossRef]
- Oldewage-Theron, W.; Egal, A. The effect of consumption of soy foods on the blood lipid profile of women: A pilot study from Qwa-Qwa. J. Nutr. Sci. Vitaminol. 2013, 59, 431–436. [Google Scholar] [CrossRef]
- Malek Rivan, N.F.; Shahar, S.; Haron, H.; Ambak, R.; Othman, F. Association between intake of soy isoflavones and blood pressure among urban and rural Malaysian adults. Mal. J. Nutr. 2018, 24, 381–393. [Google Scholar]
- Tokede, O.A.; Onabanjo, T.A.; Yansane, A.; Gaziano, J.M.; Djoussé, L. Soya products and serum lipids: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2015, 114, 831–943. [Google Scholar] [CrossRef]
- Ramdath, D.D.; Padhi, E.M.; Sarfaraz, S.; Renwick, S.; Duncan, A.M. Beyond the Cholesterol-Lowering Effect of Soy Protein: A Review of the Effects of Dietary Soy and Its Constituents on Risk Factors for Cardiovascular Disease. Nutrients 2017, 9, 324. [Google Scholar] [CrossRef]
- Rietjens, I.M.; Louisse, J.; Beekmann, K. The potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 2017, 174, 1263–1280. [Google Scholar] [CrossRef]
- Messina, M. Soy foods, isoflavones, and the health of postmenopausal women. Am. J. Clin. Nutr. 2014, 100 (Suppl. 1), 423S–430S. [Google Scholar] [CrossRef]
- Anderson, J.W.; Johnstone, B.M.; Cook-Newell, M.E. Meta-analysis of the effects of soy protein intake on serum lipids. N. Engl. J. Med. 1995, 333, 276–282. [Google Scholar] [CrossRef]
- Zhan, S.; Ho, S.C. Meta-analysis of the effects of soy protein containing isoflavones on the lipid profile. Am. J. Clin. Nutr. 2005, 81, 397–408. [Google Scholar] [CrossRef]
- Hazim, S.; Curtis, P.J.; Schär, M.Y.; Ostertag, L.M.; Kay, C.D.; Minihane, A.-M.; Cassidy, A. Acute benefits of the microbial-derived isoflavone metabolite equol on arterial stiffness in men prospectively recruited according to equol producer phenotype: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2016, 103, 694–702. [Google Scholar] [CrossRef]
- Richardson, S.I.; Steffen, L.M.; Swett, K.; Smith, C.; Burke, L.; Zhou, X.; Shikany, J.M.; Rodriguez, C.J. Dietary total isoflavone intake is associated with lower systolic blood pressure: The coronary artery risk development in young adults (CARDIA) study. J. Clin. Hypertens 2016, 18, 778–783. [Google Scholar] [CrossRef]
- Yu, J.; Bi, X.; Yu, B.; Chen, D. Isoflavones: Anti-inflammatory benefits and possible caveats. Nutrients 2016, 8, 361. [Google Scholar] [CrossRef]
- Liu, X.X.; Li, S.H.; Chen, J.Z.; Sun, K.; Wang, X.J.; Wang, X.G.; Hui, R.T. Effect of soy isoflavones on blood pressure: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 463–470. [Google Scholar] [CrossRef]
- Beavers, D.P.; Beavers, K.M.; Miller, M.; Stamey, J.; Messina, M.J. Exposure to isoflavone-containing soy products and endothelial function: A Bayesian meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 182–191. [Google Scholar] [CrossRef]
| Model | Number of Participants | Duration | Isoflavone Intake | Result | Reference |
|---|---|---|---|---|---|
| Vietnamese adults aged 40–65 years | Case: 599 people with newly diagnosed T2DM Control: 599 people in the hospital | 24 months | 5.2–9.8 mg daidzein/day | Reducing risk of T2DM | [30] |
| Chinese women | Case: 80 T2DM women Control: 40 women | 2 months | 435 mg/day | Exposure ofT2DM women to isoflavone supplementation showed reduced risk of diabetes | [31] |
| Postmenopausal Chinese women, aged 48–62 years. | Mild-dose n = 68, High-dose n = 67, Control n = 68 | 12 months | Mild-dose 40 mg/day; High-dose 80 mg/day (daidzein 46.7%) | Beneficial effect on reducing fasting glucose by higher doses of daidzein supplementation | [32] |
| American obese women and men with diagnosed and pharmacologically treated T2DM | 104 male and female | 12 months | Individual nutrition plan versus 3-1 soy-based meals during the day | plasma glucose concentration reduction by 26.17 mg/dL at 6 months but not at 12 months, HbA1c reduction by 0.49 ± 0.22% at 3 and 6 months, but not at 12 months | [33] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pabich, M.; Materska, M. Biological Effect of Soy Isoflavones in the Prevention of Civilization Diseases. Nutrients 2019, 11, 1660. https://doi.org/10.3390/nu11071660
Pabich M, Materska M. Biological Effect of Soy Isoflavones in the Prevention of Civilization Diseases. Nutrients. 2019; 11(7):1660. https://doi.org/10.3390/nu11071660
Chicago/Turabian StylePabich, Marzena, and Małgorzata Materska. 2019. "Biological Effect of Soy Isoflavones in the Prevention of Civilization Diseases" Nutrients 11, no. 7: 1660. https://doi.org/10.3390/nu11071660
APA StylePabich, M., & Materska, M. (2019). Biological Effect of Soy Isoflavones in the Prevention of Civilization Diseases. Nutrients, 11(7), 1660. https://doi.org/10.3390/nu11071660





