Metabolic Syndrome and Its Associated Early-Life Factors among Chinese and Spanish Adolescents: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
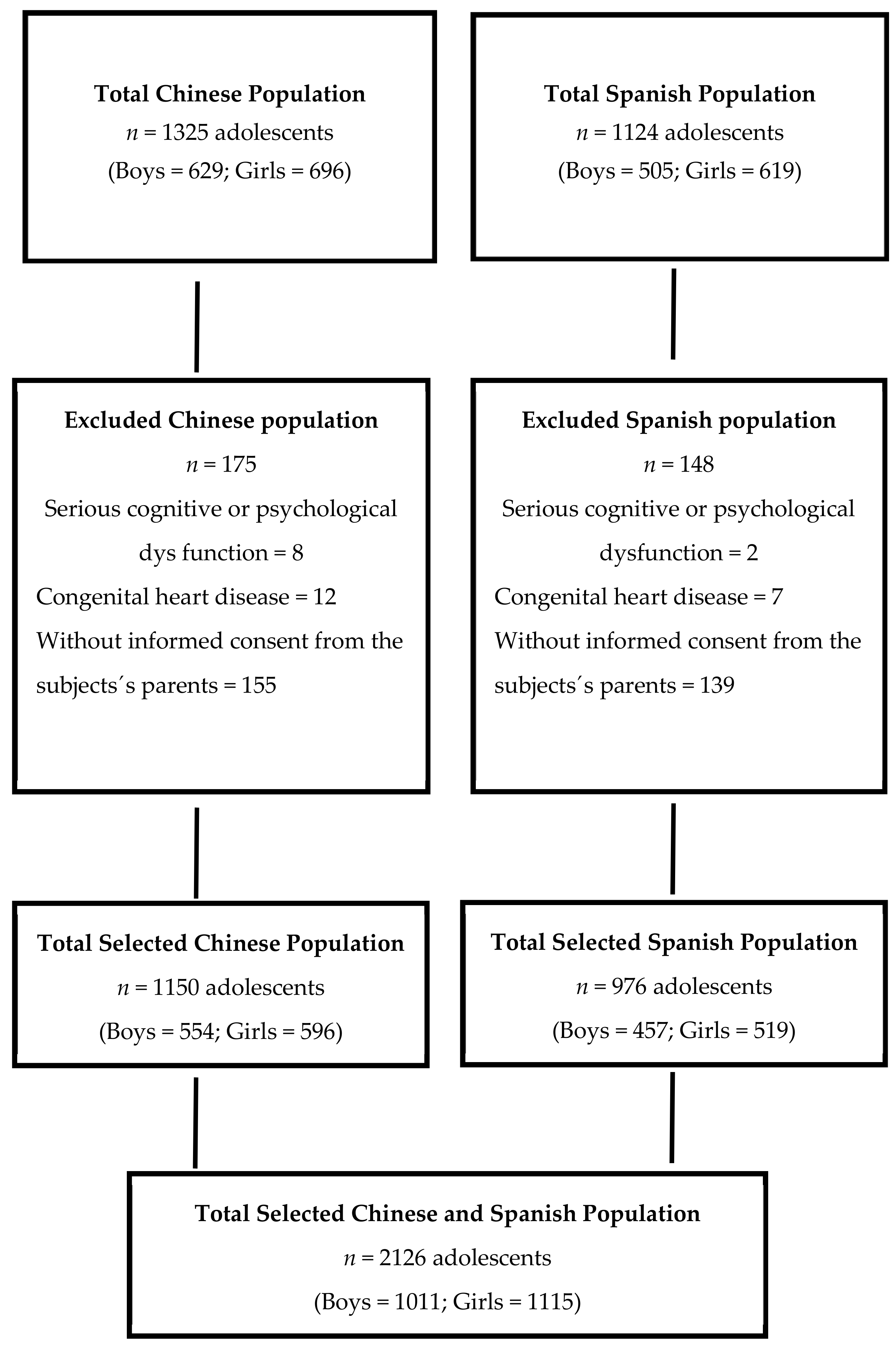
2.1. Study Design and Participants
2.2. Data Collection and Measurements
2.3. Serum Biochemical Examination
2.4. Diagnostic Criteria of Metabolic Syndrome according to the International Diabetes Federation, IDF
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weiss, R.; Bremer, A.A.; Lustig, R.H. What is metabolic syndrome, and why are children getting it? Ann. N. Y. Acad. Sci. 2013, 1281, 123–140. [Google Scholar] [CrossRef] [PubMed]
- Schubert, C.M.; Sun, S.S.; Burns, T.L.; Morrison, J.A.; Huang, T.T. Predictive ability of childhood metabolic components for adult metabolic syndrome and type 2 diabetes. J. Pediatr. 2009, 155, S1–S6. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.T.; Sun, S.S.; Daniels, S.R. Understanding the nature of metabolic syndrome components in children and what they can and cannot do to predict adult disease. J. Pediatr. 2009, 155, e13–e14. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, F.; Wang, Y.; Shan, X.; Cheng, H.; Hou, D.; Zhao, X.; Wang, T.; Zhao, D.; Mi, J. Association between childhood obesity and metabolic syndrome: Evidence from a large sample of Chinese children and adolescents. PLoS ONE 2012, 7, e47380. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.M.; Kaylor, M.B.; Johannsson, M.; Bay, C.; Churilla, J.R. Prevalence of metabolic syndrome and individual criterion in US adolescents: 2001–2010 national health and nutrition examination survey. Metab. Syndr. Relat. Disord. 2014, 12, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Jimenez, E.; Montero-Alonso, M.A.; Schmidt-RioValle, J.; Garcia-Garcia, C.J.; Padez, C. Metabolic syndrome in Spanish adolescents and its association with birth weight, breastfeeding duration, maternal smoking, and maternal obesity: A cross-sectional study. Eur. J. Nutr. 2015, 54, 589–597. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xu, H.; Li, Y.; Liu, A.; Zhang, Q.; Hu, X.; Fang, H.; Li, T.; Guo, H.; Li, Y.; Xu, G.; et al. Prevalence of the metabolic syndrome among children from six cities of China. BMC Public Health 2012, 12, 13. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhu, Y.; Cai, L.; Jing, J.; Chen, Y.; Mai, J.; Ma, L.; Ma, Y.; Ma, J. Metabolic syndrome and its associated early-life factors in children and adolescents: A cross-sectional study in Guangzhou, China. Public Health Nutr. 2016, 19, 1147–1154. [Google Scholar] [CrossRef]
- Ahmadi, A.; Gharipour, M.; Nouri, F.; Sarrafzadegan, N. Metabolic syndrome in Iranian youths: A population-based study on junior and high schools students in rural and urban areas. J. Diabetes Res. 2012, 2013, 738485. [Google Scholar] [CrossRef][Green Version]
- Agius, R.; Savona-Ventura, C.; Vassallo, J. Transgenerational metabolic determinants of fetal birth weight. Exp. Clin. Endocrinol. Diabetes. 2013, 121, 431–435. [Google Scholar] [CrossRef]
- Stuebe, A. The risks of not breastfeeding for mothers and infants. Rev. Obstet. Gynecol. 2009, 2, 222–231. [Google Scholar] [PubMed]
- Yang, Z.; Huffman, S.L. Nutrition in pregnancy and early childhood and associations with obesity in developing countries. Matern. Child. Nutr. 2013, 9, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liang, L.; Junfen, F.U.; Lizhong, D.U. Metabolic syndrome in obese children born large for gestational age. Indian J. Pediatr. 2007, 74, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Marfell-Jones, M.; Olds, T.; Stewart, A. International Standards for Anthropometric Assessment; ISAK: Potchefstroom, South Africa, 2006. [Google Scholar]
- Pickering, T.G.; Hall, J.E.; Appel, L.J.; Falkner, B.; Graves, J.; Hill, M.; Jones, D.W.; Kurtz, T.; Sheps, S.G.; Roccella, E.J. Subcommittee of professional and public education of the American heart association council on high blood pressure research. Recommendations for blood pressure measurement in humans and experimental animals, part 1: Blood pressure measurement in humans: A statement for professionals from the subcommittee of professional and public education of the American heart association council on high blood pressure research. Hypertension 2005, 45, 142–161. [Google Scholar] [PubMed]
- Zimmet, P.; Alberti, K.G.; Kaufman, F.; Tajima, N.; Silink, M.; Arslanian, S.; Wong, G.; Bennett, P.; Shaw, J.; Caprio, S.; et al. The metabolic syndrome in children and adolescents—An IDF consensus report. Pediatr. Diabetes 2007, 8, 299–306. [Google Scholar] [CrossRef]
- Kelishadi, R. Childhood overweight, obesity, and the metabolic syndrome in developing countries. Epidemiol. Rev. 2007, 29, 62–76. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Shah, P.; Nayyar, S.; Misra, A. Childhood obesity and the metabolic syndrome in developing countries. Indian J. Pediatr. 2013, 80, S28–S37. [Google Scholar] [CrossRef]
- Faienza, M.F.; Wang, D.Q.; Fruhbeck, G.; Garruti, G.; Portincasa, P. The dangerous link between childhood and adulthood predictors of obesity and metabolic syndrome. Intern. Emerg. Med. 2016, 11, 175–182. [Google Scholar] [CrossRef]
- Alvarez, L.E.; Ribas, B.L.; Serra, M.L. Prevalence of the metabolic syndrome in the population of Canary Islands, Spain. Med. Clin. 2003, 120, 172–174. [Google Scholar]
- Holst-Schumacher, I.; Nunez-Rivas, H.; Monge-Rojas, R.; Barrantes-Santamaria, M. Components of the metabolic syndrome among a sample of overweight and obese Costa Rican schoolchildren. Food Nutr. Bull. 2009, 30, 161–170. [Google Scholar] [CrossRef]
- Alvarez, M.M.; Vieira, A.C.; Sichieri, R.; Veiga, G.V. Prevalence of metabolic syndrome and of its specific components among adolescents from Niteroi city, Rio de Janeiro state, Brazil. Arq. Bras. Endocrinol. Metab. 2011, 55, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Fu, J.; Jiang, Y.; Dong, G.; Wang, X.; Wu, W. TriGlycerides and high-density lipoprotein cholesterol ratio compared with homeostasis model assessment insulin resistance indexes in screening for metabolic syndrome in the chinese obese children: A cross section study. BMC Pediatr. 2015, 15, 138. [Google Scholar] [CrossRef] [PubMed]
- Yakubov, R.; Nadir, E.; Stein, R.; Klein-Kremer, A. The duration of breastfeeding and its association with metabolic syndrome among obese children. Sci. World J. 2015, 2015, 731319. [Google Scholar] [CrossRef] [PubMed]
- Owen, C.G.; Martin, R.M.; Whincup, P.H.; Smith, G.D.; Cook, D.G. Effect of infant feeding on the risk of obesity across the life course: A quantitative review of published evidence. Pediatrics 2005, 115, 1367–1377. [Google Scholar] [CrossRef] [PubMed]
- Harder, T.; Bergmann, R.; Kallischnigg, G.; Plagemann, A. Duration of breastfeeding and risk of overweight: A meta-analysis. Am. J. Epidemiol. 2005, 162, 397–403. [Google Scholar] [CrossRef]
- De Armas, M.G.; Megías, S.M.; Modino, S.C.; Bolaños, P.I.; Guardiola, P.D.; Alvarez, T.M. Importance of breastfeeding in the prevalence of metabolic syndrome and degree of childhood obesity. Endocrinol. Nutr. 2009, 56, 400–403. [Google Scholar] [CrossRef]
- Ekelund, U.; Anderssen, S.; Andersen, L.B.; Riddoch, C.J.; Sardinha, L.B.; Luan, J.; Froberg, K.; Brage, S. Prevalence and correlates of the metabolic syndrome in a population-based sample of European youth. Am. J. Clin. Nutr. 2009, 89, 90–96. [Google Scholar] [CrossRef]
- Sun, K.; Chen, M.; Yin, Y.; Wu, L.; Gao, L. Why Chinese mothers stop breastfeeding: Mothers’ self-reported reasons for stopping during the first six months. J. Child Health Care 2017, 21, 353–363. [Google Scholar] [CrossRef]
- De Jersey, S.J.; Mallan, K.; Forster, J.; Daniels, L.A. A prospective study of breastfeeding intentions of healthy weight and overweight women as predictors of breastfeeding outcomes. Midwifery 2017, 53, 20–27. [Google Scholar] [CrossRef]
- Amir, L.H.; Donath, S. A systematic review of maternal obesity and breastfeeding intention, initiation and duration. BMC Pregnancy Childbirth 2007, 7, 9. [Google Scholar] [CrossRef]
- Dos Santos Alves, P.J.; Henriques, A.C.; Pinto, L.R.; Mota, R.M.; Alencar, C.H.; Alves, R.S.; Carvalho, F.H. Endothelial and metabolic disorders in adolescence: Low birth weight is not an isolated risk factor. J. Pediatr. Endocrinol. Metab. 2015, 28, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.P.; Yang, M.; Liang, L.; Fu, J.F.; Xiong, F.; Liu, G.L.; Gong, C.X.; Luo, F.H.; Chen, S.K.; Zhang, D.D.; et al. Possible role of birth weight on general and central obesity in Chinese children and adolescents: A cross-sectional study. Ann. Epidemiol. 2015, 25, 748–752. [Google Scholar] [CrossRef] [PubMed]
- Zabeen, B.; Tayyeb, S.; Naz, F.; Ahmed, F.; Rahman, M.; Nahar, J.; Nahar, N.; Azad, K. Prevalence of obesity and central obesity among adolescent girls in a district school in Bangladesh. Indian J. Endocrinol. Metab. 2015, 19, 649–652. [Google Scholar] [CrossRef] [PubMed]
- Schroder, H.; Ribas, L.; Koebnick, C.; Funtikova, A.; Gomez, S.F.; Fíto, M.; Perez-Rodrigo, C.; Serra-Majem, L. Prevalence of abdominal obesity in Spanish children and adolescents. Do we need waist circumference measurements in pediatric practice? PLoS ONE 2014, 9, e87549. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.M.; Huxley, R.R.; Wildman, R.P.; Woodward, M. Indices of abdominal obesity are better discriminators of cardiovascular risk factors than BMI: A meta-analysis. J. Clin. Epidemiol. 2008, 61, 646–653. [Google Scholar] [CrossRef]
- Haldar, S.; Chia, S.C.; Henry, C.J. Body composition in Asians and caucasians: Comparative analyses and influences on cardiometabolic outcomes. Adv. Food Nutr. Res. 2015, 75, 97–154. [Google Scholar] [PubMed]
- Papoutsakis, C.; Yannakoulia, M.; Ntalla, I.; Dedoussis, G.V. Metabolic syndrome in a Mediterranean pediatric cohort: Prevalence using international diabetes federation-derived criteria and associations with adiponectin and leptin. Metabolism 2012, 61, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Tai, E.S.; Lau, T.N.; Ho, S.C.; Fok, A.C.; Tan, C.E. Body fat distribution and cardiovascular risk in normal weight women. Associations with insulin resistance, lipids and plasma leptin. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 751–757. [Google Scholar] [CrossRef]
- Park, Y.W.; Allison, D.B.; Heymsfield, S.B.; Gallagher, D. Larger amounts of visceral adipose tissue in Asian Americans. Obes. Res. 2001, 9, 381–387. [Google Scholar] [CrossRef]
- Victora, C.G.; Horta, B.L.; Post, P.; Lima, R.C.; De Leon Elizalde, J.W.; Gerson, B.M.; Barros, F.C. Breast feeding and blood lipid concentrations in male Brazilian adolescents. J. Epidemiol. Community Health 2006, 60, 621–625. [Google Scholar] [CrossRef][Green Version]
- Lawlor, D.A.; Riddoch, C.J.; Page, A.S.; Andersen, L.B.; Wedderkopp, N.; Harro, M.; Stansbie, D.; Smith, G.D. Infant feeding and components of the metabolic syndrome: Findings from the European youth heart study. Arch. Dis. Child. 2005, 90, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Silva, I.; Rivera, J.A.; Trejo-Valdivia, B.; Martorell, R.; Stein, A.D.; Romieu, I.; Ramakrishnan, U. Breastfeeding status at age 3 months is associated with adiposity and cardiometabolic markers at age 4 years in Mexican children. J. Nutr. 2015, 145, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
| Variable | Chinese Adolescents | Spanish Adolescents | p-Value |
|---|---|---|---|
| Demographic information | |||
| n | 1150 | 976 | |
| Boys (%) | 554 (48.2) | 457 (45.7) | 0.28 |
| Age (years) | 12.9 ± 1.8 | 13.1 ± 1.2 | 0.09 |
| Anthropometry | |||
| Height (m) | 1.55 ± 0.14 | 1.59 ± 0.10 | <0.01 ** |
| Weight (kg) | 46.28 ± 14.01 | 54.77 ± 12.71 | <0.01 ** |
| BMI (kg/m2) | 19.24 ± 7.25 | 21.22 ± 3.79 | <0.01 ** |
| HC (cm) | 81.06 ± 16.91 | 83.82 ± 9.35 | 0.21 |
| WHR | 0.84 ± 0.09 | 0.87 ± 0.16 | <0.01 ** |
| WHtR | 0.44 ± 0.07 | 0.45 ± 0.07 | 0.01 * |
| Metabolic syndrome features | |||
| WC (cm) | 66.7 ± 10.6 | 72.3 ± 10.8 | <0.01 ** |
| FG (mmol/L) | 4.62 ± 0.49 | 4.77 ± 1.68 | 0.01 * |
| HDL-C (mmol/L) | 1.4 ± 0.3 | 1.0 ± 0.1 | <0.01 ** |
| TG (mmol/L) | 0.97 ± 0.51 | 1.43 ± 0.60 | <0.01 ** |
| SBP (mmHg) | 99.9 ± 23.2 | 118.0 ± 15.5 | <0.01 ** |
| DBP (mmHg) | 63.6 ± 15.7 | 64.1 ± 9.0 | 0.35 |
| Information of early-life factors | |||
| Breastfeeding duration (month) | 4.2 ± 3.7 | 9.2 ± 2.7 | <0.01 ** |
| Breastfeeding duration range (month) | 0~13 | 0~13 | |
| Birth weight (kg) | 3.23 ± 0.47 | 3.20 ± 0.50 | 0.09 |
| Birth weight range (kg) | 1.4~4.9 | 2.0~5.7 |
| Characteristics | Chinese Adolescents (n = 1150) | Spanish Adolescents (n = 976) |
|---|---|---|
| Abdominal obesity | 65 (5.5) | 128 (13.1) ** |
| Hypertension | 11 (1.0) | 256 (26.2) ** |
| Hyperglycemia | 23 (2.0) | 65 (6.7) ** |
| Low HDL-C | 160 (13.9) | 176 (18.0) * |
| Hypertriglyceridemia | 84 (7.3) | 32 (3.3) ** |
| Pattern of risk factors clustering | ||
| 0 Component | 886 (77.0) | 568 (58.2) ** |
| 1 Component | 193 (16.8) | 234 (24.0) ** |
| 2 Components | 61 (5.6) | 120 (12.3) ** |
| ≥3 Components | 7 (0.6) | 54 (5.5) ** |
| MetS | 6 (0.5) | 24 (2.5) ** |
| Chinese Adolescents (n = 1150) | Spanish Adolescents (n = 976) | |||
|---|---|---|---|---|
| Breastfeeding Duration | Birth Weight | Breastfeeding Duration | Birth Weight | |
| WC | −0.04 | 0.15 ** | 0.05 | 0.10 ** |
| FG | 0.03 | 0.01 | −0.89 ** | 0.65 ** |
| HDL-C | −0.04 | −0.01 | 0.81 ** | −0.58 ** |
| TG | 0.09 ** | −0.04 | −0.64 ** | 0.48 ** |
| SBP | 0.03 | 0.08 * | −0.03 | 0.06 |
| DBP | −0.02 | 0.07 * | −0.02 | 0.07 * |
| Chinese Adolescents (n = 1150) | Spanish Adolescents (n = 976) | |||
|---|---|---|---|---|
| Breastfeeding Duration (month) | Birth Weight (100 g) | Breastfeeding Duration (month) | Birth Weight (100 g) | |
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | |
| Abdominal obesity | 0.95 (0.88, 1.02) | 1.09 (1.03, 1.16) | 0.98 (0.90, 1.07) | 1.04 (0.99, 1.10) |
| Low HDL-C | 1.03 (0.98, 1.08) | 1.17 (0.98, 1.06) | 0.18 (0.13, 0.26) | 1.06 (0.93, 1.21) |
| Hyperglycemia | 0.96 (0.86, 1.08) | 0.96 (0.88, 1.05) | 0.17 (0.04, 0.72) | 6.65 (1.83, 24.19) |
| Hypertriglyceridemia | 1.03 (0.97, 1.10) | 0.99 (0.94, 1.04) | 0.52 (0.40, 0.70) | 1.09 (0.93, 1.27) |
| Hypertension | 1.10 (0.91, 1.32) | 1.07 (0.92, 1.23) | 0.96 (0.90, 1.03) | 1.02 (0.99, 1.07) |
| MetS | 1.07 (0.87, 1.32) | 0.93 (0.80, 1.08) | 0.62 (0.51, 0.76) | 0.98 (0.86, 1.12) |
| Variables (Reference) | Abdominal Obesity | Low HDL-C | Hyperglycemia | Hypertriglyceridemia | Hypertension | MetS |
|---|---|---|---|---|---|---|
| OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | OR (95% CI) | |
| Age | 1.20 (1.09, 1.32) | 1.07 (0.99, 1.15) | 0.96 (0.80, 1.16) | 0.92 (0.83, 1.02) | 1.04 (0.93, 1.17) | 1.01 (0.80, 1.28) |
| Gender (Boy) | 2.40 (1.72, 3.35) | 0.78 (0.58,1.07) | 0.30 (0.12, 0.73) | 0.79 (0.53, 1.20) | 0.77 (0.58, 1.02) | 1.76 (0.79, 3.89) |
| Country (China) | 12.04 (0.58, 24.23) | 3.84 (2.68, 5.49) | 26.85 (11.76, 61.34) | 0.70 (0.43, 1.15) | 45.05 (21.71, 93.45) | 13.6 (4.16, 44.54) |
| Breastfeeding duration (month) | 0.91 (0.77, 1.08) | 0.81 (0.78, 0.85) | 0.60 (0.53, 0.68) | 0.87 (0.82, 0.92) | 1.10 (0.91, 1.32) | 0.74 (0.65, 0.84) |
| Birth weight (100 g) | 1.15 (1.01, 1.31) | 0.98 (0.84, 1.10) | 1.96 (1.88, 3.08) | 0.99 (0.95, 1.04) | 1.07 (0.92, 1.23) | 0.94 (0.80, 1.09) |
| p value | p value | p value | p value | p value | p value | |
| Breastfeeding duration*Country | 0.504 | <0.001 | <0.017 | <0.001 | 0.187 | <0.001 |
| Birth weight*Country | 0.211 | 0.578 | <0.001 | 0.213 | 0.605 | 0.743 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Perona, J.S.; Schmidt-RioValle, J.; Chen, Y.; Jing, J.; González-Jiménez, E. Metabolic Syndrome and Its Associated Early-Life Factors among Chinese and Spanish Adolescents: A Pilot Study. Nutrients 2019, 11, 1568. https://doi.org/10.3390/nu11071568
Wang J, Perona JS, Schmidt-RioValle J, Chen Y, Jing J, González-Jiménez E. Metabolic Syndrome and Its Associated Early-Life Factors among Chinese and Spanish Adolescents: A Pilot Study. Nutrients. 2019; 11(7):1568. https://doi.org/10.3390/nu11071568
Chicago/Turabian StyleWang, Jiao, Javier S. Perona, Jacqueline Schmidt-RioValle, Yajun Chen, Jin Jing, and Emilio González-Jiménez. 2019. "Metabolic Syndrome and Its Associated Early-Life Factors among Chinese and Spanish Adolescents: A Pilot Study" Nutrients 11, no. 7: 1568. https://doi.org/10.3390/nu11071568
APA StyleWang, J., Perona, J. S., Schmidt-RioValle, J., Chen, Y., Jing, J., & González-Jiménez, E. (2019). Metabolic Syndrome and Its Associated Early-Life Factors among Chinese and Spanish Adolescents: A Pilot Study. Nutrients, 11(7), 1568. https://doi.org/10.3390/nu11071568







