The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health
Abstract
1. Introduction
2. Intestinal Fate and Bioavailability
2.1. Intestinal Metabolism: In Vitro Evidence
2.2. Bioavailability in Humans
3. Effects on Microbiota Composition
3.1. In Vitro Studies
3.2. Animal and Human Studies
4. Effects on Host Parameters Related to Gastrointestinal Health
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Molina-Montes, E.; Sanchez, M.J.; Zamora-Ros, R.; Bueno-de-Mesquita, H.B.; Wark, P.A.; Obon-Santacana, M.; Kuhn, T.; Katzke, V.; Travis, R.C.; Ye, W.; et al. Flavonoid and lignan intake and pancreatic cancer risk in the European prospective investigation into cancer and nutrition cohort. Int. J. Cancer 2016, 139, 1480–1492. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Chanet, A.; Milenkovic, D.; Manach, C.; Mazur, A.; Morand, C. Citrus flavanones: What is their role in cardiovascular protection? J. Agric. Food Chem. 2012, 60, 8809–8822. [Google Scholar] [CrossRef]
- Brat, P.; George, S.; Bellamy, A.; Du Chaffaut, L.; Scalbert, A.; Mennen, L.; Arnault, N.; Amiot, M.J. Daily polyphenol intake in France from fruit and vegetables. J. Nutr. 2006, 136, 2368–2373. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A.; Clifford, M.N. Flavanones, chalcones and dihydrochalcones—Nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1073–1080. [Google Scholar] [CrossRef]
- Gattuso, G.; Barreca, D.; Gargiulli, C.; Leuzzi, U.; Caristi, C. Flavonoid composition of Citrus juices. Molecules 2007, 12, 1641–1673. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Forouhi, N.G.; Sharp, S.J.; Gonzalez, C.A.; Buijsse, B.; Guevara, M.; van der Schouw, Y.T.; Amiano, P.; Boeing, H.; Bredsdorff, L.; et al. The association between dietary flavonoid and lignan intakes and incident type 2 diabetes in European populations: The EPIC-InterAct study. Diabetes Care 2013, 36, 3961–3970. [Google Scholar] [CrossRef]
- Cassidy, A.; Rimm, E.B.; O’Reilly, E.J.; Logroscino, G.; Kay, C.; Chiuve, S.E.; Rexrode, K.M. Dietary flavonoids and risk of stroke in women. Stroke 2012, 43, 946–951. [Google Scholar] [CrossRef]
- Mink, P.J.; Scrafford, C.G.; Barraj, L.M.; Harnack, L.; Hong, C.P.; Nettleton, J.A.; Jacobs, D.R., Jr. Flavonoid intake and cardiovascular disease mortality: A prospective study in postmenopausal women. Am. J. Clin. Nutr. 2007, 85, 895–909. [Google Scholar] [CrossRef]
- Knekt, P.; Kumpulainen, J.; Jarvinen, R.; Rissanen, H.; Heliovaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef]
- Goetz, M.E.; Judd, S.E.; Hartman, T.J.; McClellan, W.; Anderson, A.; Vaccarino, V. Flavanone intake is inversely associated with risk of incident ischemic stroke in the REasons for Geographic and Racial Differences in Stroke (REGARDS) study. J. Nutr. 2016, 146, 2233–2243. [Google Scholar] [CrossRef]
- Cutler, G.J.; Nettleton, J.A.; Ross, J.A.; Harnack, L.J.; Jacobs, D.R., Jr.; Scrafford, C.G.; Barraj, L.M.; Mink, P.J.; Robien, K. Dietary flavonoid intake and risk of cancer in postmenopausal women: The Iowa Women’s Health Study. Int. J. Cancer 2008, 123, 664–671. [Google Scholar] [CrossRef]
- Rossi, M.; Garavello, W.; Talamini, R.; La Vecchia, C.; Franceschi, S.; Lagiou, P.; Zambon, P.; Dal Maso, L.; Bosetti, C.; Negri, E. Flavonoids and risk of squamous cell esophageal cancer. Int. J. Cancer 2007, 120, 1560–1564. [Google Scholar] [CrossRef]
- Yi, L.Z.; Ma, S.S.; Ren, D.B. Phytochemistry and bioactivity of Citrus flavonoids: A focus on antioxidant, anti-inflammatory, anticancer and cardiovascular protection activities. Phytochem. Rev. 2017, 16, 479–511. [Google Scholar] [CrossRef]
- Testai, L.; Calderone, V. Nutraceutical value of citrus flavanones and their implications in cardiovascular disease. Nutrients 2017, 9, 502. [Google Scholar] [CrossRef]
- Barreca, D.; Gattuso, G.; Bellocco, E.; Calderaro, A.; Trombetta, D.; Smeriglio, A.; Lagana, G.; Daglia, M.; Meneghini, S.; Nabavi, S.M. Flavanones: Citrus phytochemical with health-promoting properties. Biofactors 2017, 43, 495–506. [Google Scholar] [CrossRef]
- Duque, A.L.R.F.; Monteiro, M.; Adorno, M.A.T.; Sakamoto, I.K.; Sivieri, K. An exploratory study on the influence of orange juice on gut microbiota using a dynamic colonic model. Food Res. Int. 2016, 84, 160–169. [Google Scholar] [CrossRef]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef]
- Wu, H.J.; Wu, E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes 2012, 3, 4–14. [Google Scholar] [CrossRef]
- Belkaid, Y.; Hand, T.W. Role of the microbiota in immunity and inflammation. Cell 2014, 157, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Romier, B.; Schneider, Y.J.; Larondelle, Y.; During, A. Dietary polyphenols can modulate the intestinal inflammatory response. Nutr. Rev. 2009, 67, 363–378. [Google Scholar] [CrossRef] [PubMed]
- Tojo, R.; Suarez, A.; Clemente, M.G.; de los Reyes-Gavilan, C.G.; Margolles, A.; Gueimonde, M.; Ruas-Madiedo, P. Intestinal microbiota in health and disease: Role of bifidobacteria in gut homeostasis. World J. Gastroenterol. 2014, 20, 15163–15176. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Wang, Z.; Zhang, J. Pathomechanisms of oxidative stress in inflammatory bowel disease and potential antioxidant therapies. Oxid. Med. Cell. Longev. 2017, 2017, 4535194. [Google Scholar] [CrossRef] [PubMed]
- Mishra, K. Structure activity relationship of antioxidative property of hesperidin. Int. J. Pharm. Erud. 2013, 2, 40–53. [Google Scholar]
- Kumar, S.; Pandey, A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Porrini, M.; Riso, P. Factors influencing the bioavailability of antioxidants in foods: A critical appraisal. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 647–650. [Google Scholar] [CrossRef]
- Manach, C.; Morand, C.; Gil-Izquierdo, A.; Bouteloup-Demange, C.; Remesy, C. Bioavailability in humans of the flavanones hesperidin and narirutin after the ingestion of two doses of orange juice. Eur. J. Clin. Nutr. 2003, 57, 235–242. [Google Scholar] [CrossRef]
- Erlund, I.; Meririnne, E.; Alfthan, G.; Aro, A. Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice. J. Nutr. 2001, 131, 235–241. [Google Scholar] [CrossRef]
- Nielsen, I.L.; Chee, W.S.; Poulsen, L.; Offord-Cavin, E.; Rasmussen, S.E.; Frederiksen, H.; Enslen, M.; Barron, D.; Horcajada, M.N.; Williamson, G. Bioavailability is improved by enzymatic modification of the citrus flavonoid hesperidin in humans: A randomized, double-blind, crossover trial. J. Nutr. 2006, 136, 404–408. [Google Scholar] [CrossRef]
- Kanaze, F.I.; Bounartzi, M.I.; Georgarakis, M.; Niopas, I. Pharmacokinetics of the citrus flavanone aglycones hesperetin and naringenin after single oral administration in human subjects. Eur. J. Clin. Nutr. 2007, 61, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Braune, A.; Blaut, M. Bacterial species involved in the conversion of dietary flavonoids in the human gut. Gut Microbes 2016, 7, 216–234. [Google Scholar] [CrossRef] [PubMed]
- Borges, G.; Lean, M.E.; Roberts, S.A.; Crozier, A. Bioavailability of dietary (poly)phenols: A study with ileostomists to discriminate between absorption in small and large intestine. Food Funct. 2013, 4, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Wang, W.; Yang, H.; Wang, D.; Ling, W. Influence of intestinal microbiota on the catabolism of flavonoids in mice. J. Food Sci. 2016, 81, H3026–H3034. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Tanabe, S.; Sugiyama, M.; Konishi, Y. Transepithelial transport of hesperetin and hesperidin in intestinal Caco-2 cell monolayers. Biochim. Biophys. Acta 2008, 1778, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Caro, G.; Borges, G.; Ky, I.; Ribas, A.; Calani, L.; Del Rio, D.; Clifford, M.N.; Roberts, S.A.; Crozier, A. In vitro colonic catabolism of orange juice (poly)phenols. Mol. Nutr. Food Res. 2015, 59, 465–475. [Google Scholar] [CrossRef]
- Hervert-Hernandez, D.; Goni, I. Dietary polyphenols and human gut microbiota: A review. Food Rev. Int. 2011, 27, 154–169. [Google Scholar] [CrossRef]
- Liu, M.; Zou, W.; Yang, C.; Peng, W.; Su, W. Metabolism and excretion studies of oral administered naringin, a putative antitussive, in rats and dogs. Biopharm. Drug Dispos. 2012, 33, 123–134. [Google Scholar] [CrossRef]
- Kim, D.H.; Jung, E.A.; Sohng, I.S.; Han, J.A.; Kim, T.H.; Han, M.J. Intestinal bacterial metabolism of flavonoids and its relation to some biological activities. Arch. Pharm. Res. 1998, 21, 17–23. [Google Scholar] [CrossRef]
- Pereira-Caro, G.; Fernandez-Quiros, B.; Ludwig, I.A.; Pradas, I.; Crozier, A.; Moreno-Rojas, J.M. Catabolism of citrus flavanones by the probiotics Bifidobacterium longum and Lactobacillus rhamnosus. Eur. J. Nutr. 2018, 57, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Van Rymenant, E.; Salden, B.; Voorspoels, S.; Jacobs, G.; Noten, B.; Pitart, J.; Possemiers, S.; Smagghe, G.; Grootaert, C.; Van Camp, J. A critical evaluation of in vitro hesperidin 2S bioavailability in a model combining luminal (microbial) digestion and Caco-2 cell absorption in comparison to a randomized controlled human trial. Mol. Nutr. Food Res. 2018, 62, e1700881. [Google Scholar] [CrossRef] [PubMed]
- Gao, K.; Xu, A.; Krul, C.; Venema, K.; Liu, Y.; Niu, Y.; Lu, J.; Bensoussan, L.; Seeram, N.P.; Heber, D.; et al. Of the major phenolic acids formed during human microbial fermentation of tea, citrus, and soy flavonoid supplements, only 3,4-dihydroxyphenylacetic acid has antiproliferative activity. J. Nutr. 2006, 136, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.D.; Pereira-Caro, G.; Ludwig, I.A.; Clifford, M.N.; Crozier, A. Anthocyanins and flavanones are more bioavailable than previously perceived: A review of recent evidence. Annu. Rev. Food Sci. Technol. 2017, 8, 155–180. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Su, W.; Yan, Z.; Wu, H.; Zeng, X.; Peng, W.; Gan, L.; Zhang, Y.; Yao, H. Identification of naringin metabolites mediated by human intestinal microbes with stable isotope-labeling method and UFLC-Q-TOF-MS/MS. J. Pharm. Biomed. Anal. 2018, 161, 262–272. [Google Scholar] [CrossRef]
- Zeng, X.; Bai, Y.; Peng, W.; Su, W. Identification of naringin metabolites in human urine and feces. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 647–656. [Google Scholar] [CrossRef]
- Pereira-Caro, G.; Ludwig, I.A.; Polyviou, T.; Malkova, D.; Garcia, A.; Moreno-Rojas, J.M.; Crozier, A. Identification of plasma and urinary metabolites and catabolites derived from orange juice (poly)phenols: Analysis by high-performance liquid chromatography-high-resolution mass spectrometry. J. Agric. Food Chem. 2016, 64, 5724–5735. [Google Scholar] [CrossRef]
- Roowi, S.; Mullen, W.; Edwards, C.A.; Crozier, A. Yoghurt impacts on the excretion of phenolic acids derived from colonic breakdown of orange juice flavanones in humans. Mol. Nutr. Food Res. 2009, 53 (Suppl. 1), S68–S75. [Google Scholar] [CrossRef]
- Pereira-Caro, G.; Borges, G.; van der Hooft, J.; Clifford, M.N.; Del Rio, D.; Lean, M.E.; Roberts, S.A.; Kellerhals, M.B.; Crozier, A. Orange juice (poly)phenols are highly bioavailable in humans. Am. J. Clin. Nutr. 2014, 100, 1378–1384. [Google Scholar] [CrossRef]
- Schar, M.Y.; Curtis, P.J.; Hazim, S.; Ostertag, L.M.; Kay, C.D.; Potter, J.F.; Cassidy, A. Orange juice-derived flavanone and phenolic metabolites do not acutely affect cardiovascular risk biomarkers: A randomized, placebo-controlled, crossover trial in men at moderate risk of cardiovascular disease. Am. J. Clin. Nutr. 2015, 101, 931–938. [Google Scholar] [CrossRef]
- Vallejo, F.; Larrosa, M.; Escudero, E.; Zafrilla, M.P.; Cerda, B.; Boza, J.; Garcia-Conesa, M.T.; Espin, J.C.; Tomas-Barberan, F.A. Concentration and solubility of flavanones in orange beverages affect their bioavailability in humans. J. Agric. Food Chem. 2010, 58, 6516–6524. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Rios-Covian, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; de Los Reyes-Gavilan, C.G.; Salazar, N. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed]
- van der Beek, C.M.; Dejong, C.H.C.; Troost, F.J.; Masclee, A.A.M.; Lenaerts, K. Role of short-chain fatty acids in colonic inflammation, carcinogenesis, and mucosal protection and healing. Nutr. Rev. 2017, 75, 286–305. [Google Scholar] [CrossRef] [PubMed]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Bakar, N.S.; Zin, N.M.; Basri, D.F. Synergy of flavone with vancomycin and oxacillin against vancomycin-intermediate Staphyloccus aureus. Pak. J. Pharm. Sci. 2012, 25, 633–638. [Google Scholar]
- Abuelsaad, A.S.; Mohamed, I.; Allam, G.; Al-Solumani, A.A. Antimicrobial and immunomodulating activities of hesperidin and ellagic acid against diarrheic Aeromonas hydrophila in a murine model. Life Sci. 2013, 93, 714–722. [Google Scholar] [CrossRef]
- Celiz, G.; Daz, M.; Audisio, M.C. Antibacterial activity of naringin derivatives against pathogenic strains. J. Appl. Microbiol. 2011, 111, 731–738. [Google Scholar] [CrossRef]
- Basile, A.; Sorbo, S.; Giordano, S.; Ricciardi, L.; Ferrara, S.; Montesano, D.; Castaldo Cobianchi, R.; Vuotto, M.L.; Ferrara, L. Antibacterial and allelopathic activity of extract from Castanea sativa leaves. Fitoterapia 2000, 71 (Suppl. 1), S110–S116. [Google Scholar] [CrossRef]
- Gwiazdowska, D.; Jus, K.; Jasnowska-Malecka, J.; Kluczynska, K. The impact of polyphenols on Bifidobacterium growth. Acta Biochim. Pol. 2015, 62, 895–901. [Google Scholar] [CrossRef]
- Cueva, C.; Moreno-Arribas, M.V.; Martin-Alvarez, P.J.; Bills, G.; Vicente, M.F.; Basilio, A.; Rivas, C.L.; Requena, T.; Rodriguez, J.M.; Bartolome, B. Antimicrobial activity of phenolic acids against commensal, probiotic and pathogenic bacteria. Res. Microbiol. 2010, 161, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Kabir, F.; Katayama, S.; Tanji, N.; Nakamura, S. Antimicrobial Effects of Chlorogenic Acid and Related Compounds. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 359–365. [Google Scholar] [CrossRef]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Barcenilla, A.; Pryde, S.E.; Martin, J.C.; Duncan, S.H.; Stewart, C.S.; Henderson, C.; Flint, H.J. Phylogenetic relationships of butyrate-producing bacteria from the human gut. Appl. Environ. Microbiol. 2000, 66, 1654–1661. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Zhu, W. Effects of six flavonoid compounds addition on short-chain fatty acids production and human fecal microbial community change during in vitro fermentation. Afr. J. Microbiol. Res. 2011, 5, 4484–4491. [Google Scholar] [CrossRef][Green Version]
- Unno, T.; Hisada, T.; Takahashi, S. Hesperetin modifies the composition of fecal microbiota and increases cecal levels of short-chain fatty acids in rats. J. Agric. Food Chem. 2015, 63, 7952–7957. [Google Scholar] [CrossRef] [PubMed]
- Estruel-Amades, S.; Massot-Cladera, M.; Perez-Cano, F.J.; Franch, A.; Castell, M.; Camps-Bossacoma, M. Hesperidin effects on gut microbiota and gut-associated lymphoid tissue in healthy rats. Nutrients 2019, 11, 324. [Google Scholar] [CrossRef]
- Salden, B.N.; Troost, F.J.; Possemiers, S.; Stevens, Y.; Masclee, A.A. Maastricht University: Maastricht, The Netherlands, Unpublished work. 2019.
- Lima, A.C.D.; Cecatti, C.; Fidelix, M.P.; Adorno, M.A.T.; Sakamoto, I.K.; Cesar, T.B.; Sivieri, K. Effect of daily consumption of orange juice on the levels of blood glucose, lipids, and gut microbiota metabolites: Controlled clinical trials. J. Med. Food 2019, 22, 202–210. [Google Scholar] [CrossRef]
- Brasili, E.; Hassimotto, N.M.A.; Del Chierico, F.; Marini, F.; Quagliariello, A.; Sciubba, F.; Miccheli, A.; Putignani, L.; Lajolo, F. Daily consumption of orange juice from Citrus sinensis L. Osbeck cv. Cara Cara and cv. Bahia differently affects gut microbiota profiling as unveiled by an integrated meta-omics approach. J. Agric. Food Chem. 2019, 67, 1381–1391. [Google Scholar] [CrossRef]
- Bischoff, S.C. ‘Gut health’: A new objective in medicine? BMC Med. 2011, 9, 24. [Google Scholar] [CrossRef]
- Vancamelbeke, M.; Vermeire, S. The intestinal barrier: A fundamental role in health and disease. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Noda, S.; Tanabe, S.; Suzuki, T. Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells. J. Agric. Food Chem. 2012, 60, 4628–4633. [Google Scholar] [CrossRef] [PubMed]
- Noda, S.; Tanabe, S.; Suzuki, T. Naringenin enhances intestinal barrier function through the expression and cytoskeletal association of tight junction proteins in Caco-2 cells. Mol. Nutr. Food Res. 2013, 57, 2019–2028. [Google Scholar] [CrossRef] [PubMed]
- Fukui, H. Increased intestinal permeability and decreased barrier function: Does it really influence the risk of inflammation? Inflamm. Intest. Dis. 2016, 1, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Azuma, T.; Shigeshiro, M.; Kodama, M.; Tanabe, S.; Suzuki, T. Supplemental naringenin prevents intestinal barrier defects and inflammation in colitic mice. J. Nutr. 2013, 143, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Shigeshiro, M.; Tanabe, S.; Suzuki, T. Dietary polyphenols modulate intestinal barrier defects and inflammation in a murine model of colitis. J. Funct. Foods 2013, 5, 949–955. [Google Scholar] [CrossRef]
- Xu, L.; Yang, Z.L.; Li, P.; Zhou, Y.Q. Modulating effect of Hesperidin on experimental murine colitis induced by dextran sulfate sodium. Phytomedicine 2009, 16, 989–995. [Google Scholar] [CrossRef]
- Crespo, M.E.; Galvez, J.; Cruz, T.; Ocete, M.A.; Zarzuelo, A. Anti-inflammatory activity of diosmin and hesperidin in rat colitis induced by TNBS. Planta Med. 1999, 65, 651–653. [Google Scholar] [CrossRef]
- Khan, R.A.; Mallick, N.; Feroz, Z. Anti-inflammatory effects of Citrus sinensis L., Citrus paradisi L. and their combinations. Pak. J. Pharm. Sci. 2016, 29, 843–852. [Google Scholar]
- Impellizzeri, D.; Bruschetta, G.; Di Paola, R.; Ahmad, A.; Campolo, M.; Cuzzocrea, S.; Esposito, E.; Navarra, M. The anti-inflammatory and antioxidant effects of bergamot juice extract (BJe) in an experimental model of inflammatory bowel disease. Clin. Nutr. 2015, 34, 1146–1154. [Google Scholar] [CrossRef]
- Kamboh, A.A.; Zhu, W.Y. Individual and combined effects of genistein and hesperidin on immunity and intestinal morphometry in lipopolysacharide-challenged broiler chickens. Poult. Sci. 2014, 93, 2175–2183. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, H.; Rogoll, D.; Scheppach, W.; Melcher, R.; Richling, E. The Ussing type chamber model to study the intestinal transport and modulation of specific tight-junction genes using a colonic cell line. Mol. Nutr. Food Res. 2009, 53, 1211–1225. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Luceri, C.; Vivoli, E.; Pagliuca, C.; Lodovici, M.; Moneti, G.; Dolara, P. Polyphenol metabolites from colonic microbiota exert anti-inflammatory activity on different inflammation models. Mol. Nutr. Food Res. 2009, 53, 1044–1054. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Liu, F.; Xu, L.; Yin, P.; Li, D.; Mei, C.; Jiang, L.; Ma, Y.; Xu, J. Protective Effects of Ferulic Acid against Heat Stress-Induced Intestinal Epithelial Barrier Dysfunction In Vitro and In Vivo. PLoS ONE 2016, 11, e0145236. [Google Scholar] [CrossRef] [PubMed]
- Aura, A.M. Microbial metabolism of dietary phenolic compounds in the colon. Phytochem. Rev. 2008, 7, 407–429. [Google Scholar] [CrossRef]
- Brett, G.M.; Hollands, W.; Needs, P.W.; Teucher, B.; Dainty, J.R.; Davis, B.D.; Brodbelt, J.S.; Kroon, P.A. Absorption, metabolism and excretion of flavanones from single portions of orange fruit and juice and effects of anthropometric variables and contraceptive pill use on flavanone excretion. Br. J. Nutr. 2009, 101, 664–675. [Google Scholar] [CrossRef] [PubMed]
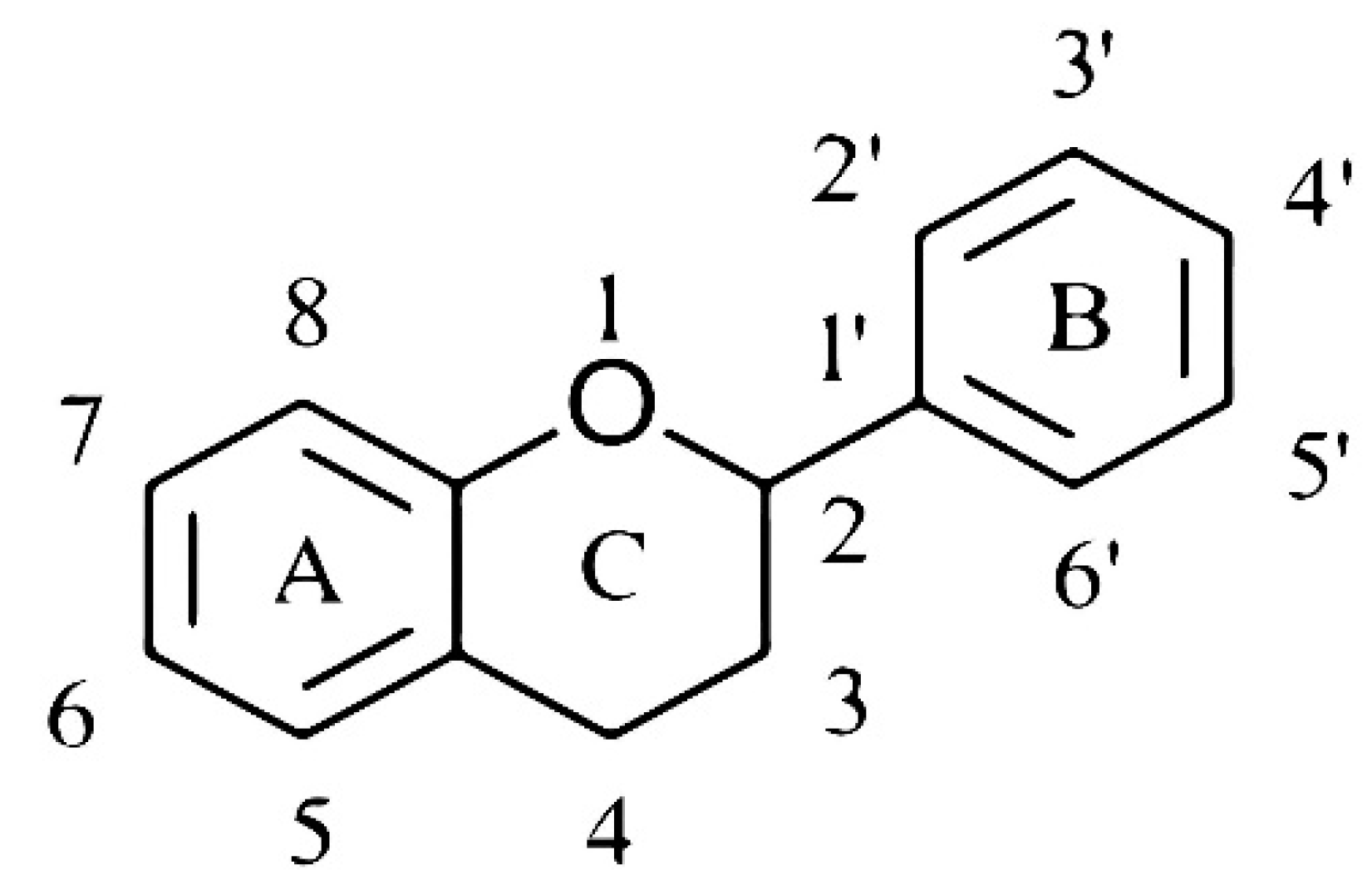
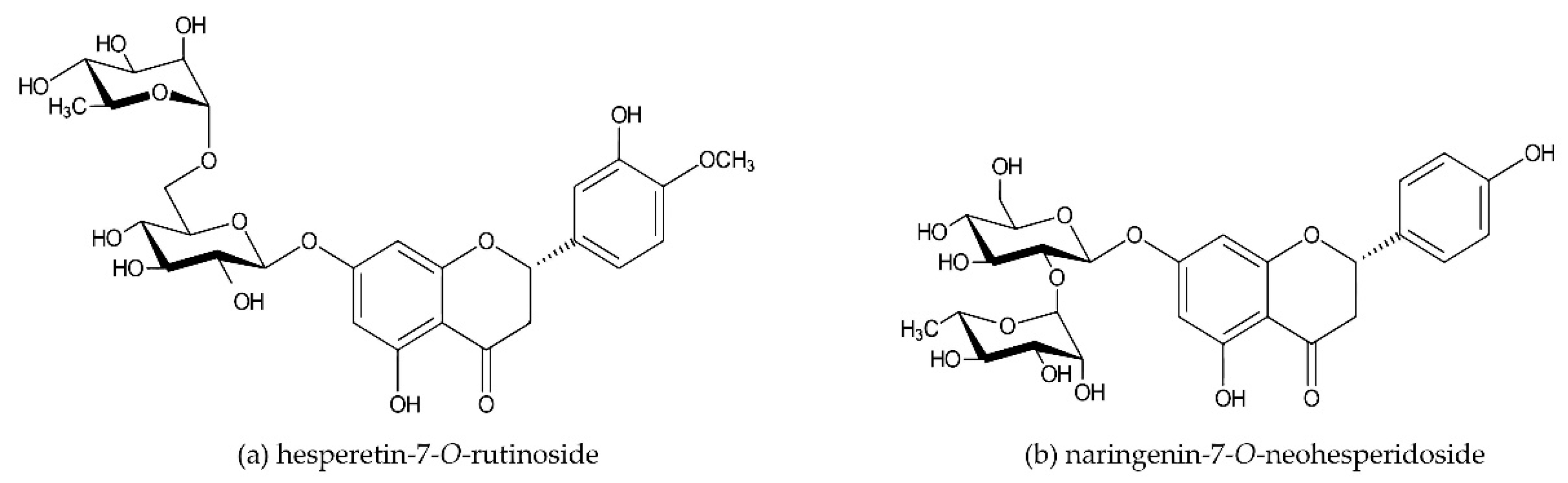
| In Vitro Model System [Ref.] | Sampling Time (Extra Sampling) | Treatment (Dosage) | Metabolites Formed | Comments |
|---|---|---|---|---|
| Batch incubation, fecal samples from four healthy volunteers [37] | 24 h (1 h, 2 h, 4 h, 6 h, 8 h, 24 h) | Hesperetin (50 µmol in 50 mL) Naringenin (50 µmol in 50 mL) | Isoferulic acid, dihydrocaffeic acid, hydrocinnamic acid, 3-(3′-hydroxyphenyl)propionic acid, phloretic acid, 4-hydroxyphenylacetic acid Phloretic acid, hydrocinnamic acid, 4-hydroxyphenylacetic acid | Metabolite concentrations vary between time points and donors |
| Batch incubation, bacteria isolated from fresh human fecal samples [40] | 12 h | Hesperidin (5 mg in 50 mL) Naringin (5 mg in 50 mL) | Hesperetin, resorcinol, phloroglucinol, 2,4-dihydroxyphenylacetic acid Naringenin, 4-hydroxybenzoic acid, phloroglucinol, phloroglucinic acid, 4-hydroxyphenylacetic acid | |
| Batch incubation, probiotic bacteria (Bifidobacterium longum R0175 and Lactobacillus rhamnosus subsp. rhamnosus NCTC 10302) [41] | 48 h (12 h, 24 h, 36 h, 48 h) | Hesperidin (410 nmol in 10 mL) Naringenin (430 nmol in 10 mL) Hesperetin (820 nmol in 10 mL) Naringenin (920 nmol in 10 mL) | - - Isoferulic acid, dihydrocaffeic acid, 3-(3′-hydroxyphenyl)propionic acid, hydrocinnamic acid Phloretic acid, hydrocinnamic acid | Metabolite concentrations vary between time points and bacteria |
| SHIME, fecal sample from one healthy volunteer [42] | 3 weeks (1 wk, 2 wks, 3 wks) | Hesperidin (500 mg) | Hesperetin, dihydrocaffeic acid, isoferulic acid, 4-hydroxyphenylacetic acid, dihydroferulic acid, ferulic acid, protocatechuic acid, vanillic acid, caffeic acid | |
| TIM-2, fecal samples from 10 healthy volunteers (pooled) [43] | 28 h (0 h, 4 h, 8 h, 12 h, 16 h, 24 h, and 28 h) | Citrus + rutin supplement (284 mg rutin, 430 mg naringin, 88 mg hesperidin, 4.4 mg eriodictyol). | Phloretic acid, isoferulic acid, dihydroferulic acid, dihydrocaffeic acid, homovanillic acid, 3-hydroxyphenylacetic acid, 4-hydroxyphenylacetic acid, 2,4-dihydroxyphenylacetic acid, 3,4-dihydroxyphenylacetic acid, hippuric acid, resorcinol, phloroglucinol | |
| Batch incubation, fecal samples from 30 healthy volunteers [45] | 24 h (4 h, 8 h, 12 h, 24 h) | [2′,3′,5′,6′-D4]naringin (10 µL (20 mg/mL) in 990 µL) | [2′,3′,5′,6′-D4]naringenin, 5-Oac-[2′,3′,5′,6′-D4]naringin, [2′,3′,5′,6′-D4]apiforol-7-O-rhamnoglucoside, 6/8-hydroxyl-[2′,3′,5′,6′-D4]naringin, 8/6-hydroxyl-[2′,3′,5′,6′-D4]naringin, [2′,5′,6′-D3]neoeriocitrin, [2′,3′,5′,6′-D4]apigenin, [2′,5′,6′-D3]eriodictyol, 6/8-hydroxyl-[2′,3′,5′,6′-D4]naringenin, 8/6-hydroxyl-[2′,3′,5′,6′-D4]naringenin, [2′,3′,5′,6′-D4]apiforol, 3-(4′-hydroxyphenyl)-[2′,3′,5′,6′-D4]propanoic acid, 3-phenyl-[2′,3′,5′,6′-D4]propanoic acid | Metabolism varies between donors |
| Population (Design) [Ref.] | Treatment (Dosage/Flavanone Concentration) | Sample and Sampling Time (Extra Sampling) | Metabolites Formed | Comments |
|---|---|---|---|---|
| Healthy subjects, n = 7 (pre- and post-test) [46] | 320 mg naringin | 72 h urine (0–4 h, 4–8 h, 8–12 h, 12–24 h, 24–36 h, 36–48 h, 48–60 h, 60–72 h), 72 h feces | Urine and feces: 4-hydroxybenzoic acid, 4-hydroxyhippuric acid, hippuric acid, phoretic acid, phloretic acid sulfate, naringin, naringenin, naringenin diglucuronide Urine: naringenin diglucuronide, naringenin glucoside glucuronide, naringenin glucoside sulfate, naringenin glucuronide sulfate, naringin glucuronide, naringenin sulfate, hydroxylated naringenin glucuronide, naringenin glucuronide, naringenin glucuronide dimer, hydroxylated naringenin sulfate | Excretion of metabolites varied between individuals |
| Healthy volunteers, n = 12 (pre- and post-test) [47] | Orange juice (500 mL /398 μmol (poly)phenols, of which 246 μmol was hesperidin) | 24 h plasma (0 h, 1 h, 2 h, 3 h, 4 h, 5 h, 6 h, 7 h, 8 h, 24 h) | Plasma: naringenin-4′-O-glucuronide, naringenin-7-O-glucuronide, naringenin-4′-sulfate, hesperetin-3′,7-O-diglucuronide, hesperetin-5,7-O-diglucuronide, hesperetin-3′,5-O-diglucuronide, hesperetin-O-glucuronyl-sulfate, hesperetin-7-O-glucuronide, hesperetin-3′-O-glucuronide, hesperetin-sulfate, hesperetin-3′-sulfate, eriodictyol sulfate, eriodictyol-O-glucuronyl-sulfate, caffeic acid-3′-sulfate, caffeic acid-4′-sulfate, ferulic acid, ferulic acid-4′-O-glucuronide, ferulic acid-4′-sulfate, isoferulic acid, isoferulic acid-3′-O-glucuronide, 3-(3′-hydroxyphenyl)hydracrylic acid, 3-(3′,4′-dihydroxyphenyl)propionic acid, 3-(3′-hydroxyphenyl)propionic acid-4′-sulfate, 3-(4′-hydroxyphenyl)propionic acid-3′-sulfate, 3-(3′-methoxy-4′-hydroxyphenyl)propionic acid, 3-(3′-methoxyphenyl)propionic acid-4′-O-glucuronide, 3-(3′-hydroxy-4′-methoxyphenyl)propionic acid, 3-(4′-methoxyphenyl)propionic acid-3′-O-glucuronide, 3-(3′-methoxyphenyl)propionic acid-4′-sulfate, 3-(4′-methoxyphenyl)propionic acid-3′-sulfate, 3-(4′-hydroxyphenyl)propionic acid, 3-(phenyl)propionic acid, hydroxyphenylacetic acid-O-glucuronide, hydroxyphenyl acetic acid-3′-sulfate, methoxyphenylacetic acid-O-glucuronide, 3′-methoxyphenylacetic acid-4′-sulfate, 4′-methoxyphenylacetic acid-3′-sulfate, 3′-hydroxyphenylacetic acid, 4′-hydroxyphenylacetic acid, 3,4-dihydroxybenzoic acid, benzoic acid-4-sulfate, 3′-methoxy-4′-hydroxymandelic acid, 4′-hydroxymandelic acid, hippuric acid-O-glucuronide, 3′-hydroxyhippuric acid, 4′-hydroxyhippuric acid, hippuric acid | |
| 24 h urine (baseline, 0–5 h, 5–8 h, 8–10 h, 10–24 h) | Urine: same as plasma, but also naringenin-4′,7-O-diglucuronide, naringenin-5,7-O-diglucuronide, naringenin-4′,5-O-diglucuronide, naringenin-O-glucuronyl-sulfate, hesperetin-5-O-glucuronide, hesperetin-O-glucosyl-sulfate, 3′-hydroxycinnamic acid, coumaric acid-3′-O-glucuronide, 4′-hydroxycinnamic acid, coumaric acid-4′-O-glucuronide, coumaric acid-4′-sulfate, caffeic acid-3′-O-glucuronide, caffeic acid-4′-O-glucuronide, 3-(3′-hydroxyphenyl)propionic acid-4′-O-glucuronide, 3-(phenyl)propionic acid-4′-O-glucuronide, 3-(4′-hydroxyphenyl)propionic acid-3-O-glucuronide, 3-(3′-hydroxyphenyl)propionic acid, 3-(phenyl)propionic acid-3′-O-glucuronide, 3-(phenyl)propionic acid-4′-sulfate,3-(phenyl)propionic acid-3′-sulfate, 3′,4′-dihydroxyphenylacetic acid, hydroxyphenylacetic acid-4′-sulfate, 3′-methoxy-4′-hydroxyphenylacetic acid, 3′,4′-dimethoxyphenylacetic acid, phenylacetic acid, hydroxybenzoic acid-O-glucuronide, 3-hydroxybenzoic acid-4-sulfate, 4-hydroxybenzoic acid-3-sulfate, 3-methoxy-4-hydroxybenzoic acid, 3-hydroxy-4-methoxybenzoic acid, 3-hydroxybenzoic acid, 4-hydroxybenzoic acid, benzoic acid-3-sulfate, 1,3,5-trihydroxyphenol, 1,2,3-trihydroxyphenol, 1,2-dihydroxyphenol | |||
| Healthy subjects, n = 5 (controlled cross over) [48] | Orange juice (250 mL/168 µmol hesperidin, 12 µmol narirutin) | 24 h urine (0–2 h, 2–5 h, 5–10 h, 10–24 h) | 3-hydroxyphenylacetic acid, 3-hydroxyphenylhydracrylic acid, dihydroferulic acid, 3-methoxy-4-hydroxyphenylhydracrylic acid, 3-hydroxyhippuric acid | |
| Healthy volunteers, n = 12 (controlled cross over) [49] | Pulp-enriched orange juice (250 mL/537 µmol flavanones, of which 329 µmol was hesperidin) | 24 h urine (0 h, 0–2 h, 2–5 h, 5–10 h, 10–24 h) | Hesperetin-O-diglucuronide, hesperetin-O-sulfate-O-glucuronide, hesperetin-O-sulfate-O-glucoside, hesperetin-7-O-glucuronide, hesperetin-3-O-glucuronide, hesperetin-3-O-sulfate, naringenin-O-diglucuronide, naringenin-4′-O-glucuronide, naringenin-7-O-glucuronide, eriodictyol-O-sulfate 3-(3′-hydroxy-4′-methoxyphenyl)hydracrylic acid, isoferulic acid, dihydroferulic acid, 3′-hydroxyhippuric acid, 4′-hydroxyhippuric acid, 3-(3′-hydroxyphenyl)hydracrylic acid, 3-methoxy-4-hydroxyphenylacetic acid, hippuric acid | Excretion of metabolites varied between individuals |
| Men at moderate CVD risk, n = 16 (controlled cross over) [50] | Orange juice, (767 mL/ 320 mg hesperidin) | 5 h plasma | Hesperetin-glucuronide, naringenin-7-glucuronide, hesperetin-glucuronide, naringenin-glucuronide, hesperetin, naringenin Hippuric acid, dihydroferulic acid, dihydroferulic acid-3-glucuronide, 4-hydroxyphenylacetic acid, vanillic acid, hydroxyhippuric acid, iso/ferulic acid-glucuronide, 3- hydroxyhippuric acid, isovanillic acid, 3-hydroxyphenylacetic acid, vanillic acid-glucuronide, isovanillic acid-glucuronide, iso/vanillic acid-glucuronide, 4-hydroxy-benzoic acid, benzoic acid-4-glucuronide |
| Model [Ref.] | Treatment | Treatment Duration | Dose and Administration | Change in Main Outcomes vs. Control (Relevant Concentrations) |
|---|---|---|---|---|
| Caco-2 cell monolayers [73] | Hesperetin vs. control Naringenin vs. control | 24 h | 100 µM | Barrier function: TEER, occludin expression, claudin-4 expression, cytoskeletal association of occludin and claudin-1, and -3 FITC–dextran flux Barrier function: TEER, occludin expression, claudin-4 expression, cytoskeletal association of ZO-2, occludin and claudin-1, -3, and -4 FITC–dextran flux |
| Caco-2 cell monolayers [74] | Naringenin vs. control | 48 h | 10, 30, 100 µM | Barrier function: TEER (30, 100 µM)†, claudin-4 expression (30, 100 µM)†, cytoskeletal association of occludin, claudin-1, claudin-4 and ZO-2 (100 µM) FITC–dextran flux (30, 100 µM)† |
| DSS-induced colitis in male BALB/c mice [76] | Naringenin vs. control diet | 9 days | 0.3% of the diet, oral administration | Barrier function: Colonic permeability, claudin-1 expression Occludin, junctional adhesion molecule-A, claudin-3 expression Inflammation: DAI, colonic shortening, expression of cytokines (IFN-ϒ, IL-6, MIP-2, and IL-17A) |
| DSS-induced colitis in male BALB/c mice [77] | Naringenin vs. control diet Hesperetin vs. control diet | 12 days | 0.3% of the diet, oral administration 0.3% of the diet, oral administration | Barrier function: Colonic permeability Occludin expression Inflammation: Weight loss, colonic damage Colon length Barrier function: Colonic permeability, occludin expression Inflammation: Weight loss, colonic damage Colon length |
| DSS-induced colitis in male BALB/c mice [78] | Hesperidin vs. control | 7 days | 10, 40, 80 mg/kg, oral administration | Inflammation: DAI, MPO, MDA, IL-6, colonic wet weight (10, 40, 80 mg/kg)† Mucosal cell damage (80 mg/kg) IL-4 (10, 40, 80 mg/kg) |
| TNBS-induced colitis in female Wistar rats [79] | Hesperidin vs. control | Twice (48 h pre- + 48 h post-colitis induction) | 2.5, 5, 10, 25, 50 mg/kg, oral administration | Inflammation: Colonic damage, colonic weight, colonic MPO (10, 25 mg/kg) Glutathione levels (10, 25 mg/kg), colonic fluid absorption (10–50 mg/kg) MDA, LTB4 (2.5–50 mg/kg) |
| TNBS- induced colitis in male Wistar rats [80] | Orange juice vs. control Grapefruit juice vs. control Combination vs. control | 15 days | 2 mL/kg, 5 mL/kg, 8 mL/kg, oral administration 0.1 mL/kg, 0.3 mL/kg, 0.5 mL/kg, oral administration 2 mL/kg OJ + 0.1 mL/kg GJ (low dose), 5 mL/kg OJ + 0.3 mL/kg GJ (high dose), oral administration | Inflammation: Colonic damage (2, 5, 8 mL/kg), MPO, CRP (5, 8 mL/kg), ALP (8 mg/kg) GSH (8 mL/kg) Inflammation: Colonic damage (0.1, 0.3, 0.5 mL/kg) MPO, CRP (0.3, 0.5 mL/kg), ALP (0.3, 0.5 mL/kg) GSH (0.3 mL/kg) Inflammation: Colonic damage, MPO, CRP, ALP (low dose, high dose) GSH (high dose) |
| DNBS- induced colitis in Male CD1 mice [81] | Bergamot juice extract vs. control | 4 days | 5, 10, 20 mg/kg, oral administration | Inflammation: Colonic damage, weight loss, MPO, TNF-α, IL-1β, ICAM-1, p-selectin, nitrotyrosine, PAR, nuclear NF-kB translocation, p-JNK activation (20 mg/kg) Colon length (20 mg/kg) |
| LPS-challenged broiler chickens [82] | Hesperidin vs. control diet | 42 days | 20 mg/kg diet, oral administration | Inflammation: Phagocytic index, villus length, villus width, villus length/crypt depth Crypt depth Body weight gain, feed intake feed conversion ratio |
| Human subjects with features of metabolic syndrome [68] | Citrus extract (>80% hesperidin-2S and <4% of naringin) vs. placebo | 12 weeks | 500 mg, oral administration | Inflammation: Calprotectin |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stevens, Y.; Rymenant, E.V.; Grootaert, C.; Camp, J.V.; Possemiers, S.; Masclee, A.; Jonkers, D. The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health. Nutrients 2019, 11, 1464. https://doi.org/10.3390/nu11071464
Stevens Y, Rymenant EV, Grootaert C, Camp JV, Possemiers S, Masclee A, Jonkers D. The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health. Nutrients. 2019; 11(7):1464. https://doi.org/10.3390/nu11071464
Chicago/Turabian StyleStevens, Yala, Evelien Van Rymenant, Charlotte Grootaert, John Van Camp, Sam Possemiers, Adrian Masclee, and Daisy Jonkers. 2019. "The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health" Nutrients 11, no. 7: 1464. https://doi.org/10.3390/nu11071464
APA StyleStevens, Y., Rymenant, E. V., Grootaert, C., Camp, J. V., Possemiers, S., Masclee, A., & Jonkers, D. (2019). The Intestinal Fate of Citrus Flavanones and Their Effects on Gastrointestinal Health. Nutrients, 11(7), 1464. https://doi.org/10.3390/nu11071464





