Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Dietary Index
2.4. Statistical Analyses
3. Results
3.1. Participants
3.2. Evaluation by Diet Group
3.3. Evaluation by Diet Quality
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schwingshackl, L.; Hoffmann, G.; Lampousi, A.M.; Knüppel, S.; Iqbal, K.; Schwedhelm, C.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food groups and risk of type 2 diabetes mellitus: A systematic review and meta-analysis of prospective studies. Eur. J. Epidemiol. 2017, 32, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zuurmond, M.G.; van der Schaft, N.; Nano, J.; Wijnhoven, H.A.H.; Ikram, M.A.; Franco, O.H.; Voortman, T. Plant versus animal based diets and insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Eur. J. Epidemiol. 2018, 33, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Micha, R.; Peñalvo, J.L.; Cudhea, F.; Imamura, F.; Rehm, C.D.; Mozaffarian, D. Association between dietary factors and mortality from heart disease, stroke, and type 2 diabetes in the United States. JAMA 2017, 317, 912–924. [Google Scholar] [CrossRef]
- Kane-Diallo, A.; Srour, B.; Sellem, L.; Deschasaux, M.; Latino-Martel, P.; Hercberg, S.; Galan, P.; Fassier, P.; Guéraud, F.; Pierre, F.H.; et al. Association between a pro plant-based dietary score and cancer risk in the prospective NutriNet-santé cohort. Int. J. Cancer 2018, 143, 2168–2176. [Google Scholar] [CrossRef] [PubMed]
- Pearson, K.E.; Wadley, V.G.; McClure, L.A.; Shikany, J.M.; Unverzagt, F.W.; Judd, S.E. Dietary patterns are associated with cognitive function in the REasons for Geographic and Racial Differences in Stroke (REGARDS) cohort. J. Nutr. Sci. 2016, 5, e38. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Xiao, R.; Cai, C.; Xu, Z.; Wang, S.; Pan, L.; Yuan, L. Diet, lifestyle and cognitive function in old Chinese adults. Arch. Gerontol. Geriatr. 2016, 63, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Scapagnini, G.; Willcox, B.J. Healthy aging diets other than the Mediterranean: A focus on the Okinawan diet. Mech. Ageing Dev. 2014, 136, 148–162. [Google Scholar] [CrossRef]
- Katz, D.L.; Meller, S. Can we say what diet is best for health? Annu. Rev. Public Health 2014, 35, 83–103. [Google Scholar] [CrossRef]
- McEvoy, C.T.; Temple, N.; Woodside, J.V. Vegetarian diets, low-meat diets and health: A review. Public Health Nutr. 2012, 15, 2287–2294. [Google Scholar] [CrossRef]
- Satija, A.; Bhupathiraju, S.N.; Rimm, E.B.; Spiegelman, D.; Chiuve, S.E.; Borgi, L.; Willett, W.C.; Manson, J.E.; Sun, Q.; Hu, F.B. Plant-Based Dietary Patterns and Incidence of Type 2 Diabetes in US Men and Women: Results from Three Prospective Cohort Studies. PLoS Med. 2016, 13, e1002039. [Google Scholar] [CrossRef]
- Satija, A.; Bhupathiraju, S.N.; Spiegelman, D.; Chiuve, S.E.; Manson, J.E.; Willett, W.; Rexrode, K.M.; Rimm, E.B.; Hu, F.B. Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. J. Am. Coll. Cardiol. 2017, 70, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Wirt, A.; Collins, C.E. Diet quality—What is it and does it matter? Public Health Nutr. 2009, 12, 2473–2492. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.B. Dietary pattern analysis: a new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, E.T.; Ohls, J.; Carlson, S.; Fleming, K. The Healthy Eating Index: Design and applications. J. Am. Diet Assoc. 1995, 95, 1103–1108. [Google Scholar] [CrossRef]
- Gans, K.M.; Risica, P.M.; Wylie-Rosett, J.; Ross, E.M.; Strolla, L.O.; McMurray, J.; Eaton, C.B. Development and evaluation of the nutrition component of the Rapid Eating and Activity Assessment for Patients (REAP): A new tool for primary care providers. J. Nutr. Educ. Behav. 2006, 38, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Segal-Isaacson, C.; Wylie-Rosett, J.; Gans, K.M. Validation of a short dietary assessment questionnaire: The rapid eating and activity assessment for participants short version (REAP-S). Diabetes Educ. 2004, 30, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.S.; Bliss, C.; Knurick, J.R.; Scholtz, C. Rapid Eating Assessment for Participants [shortened version] scores are associated with Healthy Eating Index-2010 scores and other indices of diet quality in healthy adult omnivores and vegetarians. Nutr. J. 2018, 17, 89. [Google Scholar] [CrossRef]
- Godin, G.; Shephard, R.J. A simple method to assess exercise behavior in the community. Can. J. Appl. Sport Sci. 1985, 10, 141–146. [Google Scholar]
- Weinstein, S.J.; Vogt, T.M.; Gerrior, S.A. Healthy Eating Index scores are associated with blood nutrient concentrations in the third National Health and Nutrition Examination Survey. J. Am. Diet Assoc. 2004, 104, 576–584. [Google Scholar] [CrossRef]
- Murray, E.K.; Baker, S.; Auld, G. Nutrition Recommendations from the US Dietary Guidelines Critical to Teach Low-Income Adults: Expert Panel Opinion. J. Acad. Nutr. Diet 2018, 118, 201–210. [Google Scholar] [CrossRef]
- Karlsen, M.C.; Lichtenstein, A.H.; Economos, C.D.; Folta, S.C.; Rogers, G.; Jacques, P.F.; Livingston, K.A.; Rancaño, K.M.; McKeown, N.M. Web-Based Recruitment and Survey Methodology to Maximize Response Rates from Followers of Popular Diets: The Adhering to Dietary Approaches for Personal Taste (ADAPT) Feasibility Survey. Curr. Dev. Nut 2018, 2. [Google Scholar] [CrossRef] [PubMed]
- Povey, R.; Wellens, B.; Conner, M. Attitudes towards following meat, vegetarian and vegan diets: An examination of the role of ambivalence. Appetite 2001, 37, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Lea, E.; Worsley, A. The factors associated with the belief that vegetarian diets provide health benefits. Asia Pac. J. Clin. Nutr. 2003, 12, 296–303. [Google Scholar] [PubMed]
- Mullee, A.; Vermeire, L.; Vanaelst, B.; Mullie, P.; Deriemaeker, P.; Leenaert, T.; De Henauw, S.; Dunne, A.; Gunter, M.J.; Clarys, P.; et al. Vegetarianism and meat consumption: A comparison of attitudes and beliefs between vegetarian, semi-vegetarian, and omnivorous subjects in Belgium. Appetite 2017, 114, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, M.C.; Rogers, G.; Miki, A.; Lichtenstein, A.H.; Folta, S.C.; Economos, C.D.; Jacques, P.F.; Livingston, K.A.; McKeown, N.M. Theoretical Food and Nutrient Composition of Whole-Food Plant-Based and Vegan Diets Compared to Current Dietary Recommendations. Nutrients 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Baden, M.Y.; Satija, A.; Hu, F.B.; Huang, T. Change in Plant-Based Diet Quality Is Associated with Changes in Plasma Adiposity-Associated Biomarker Concentrations in Women. J. Nutr. 2019, 149, 676–686. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.F.; Hsu, C.C.; Chiu, T.H.; Lee, C.Y.; Liu, T.T.; Tsao, C.K.; Chuang, S.C.; Hsiung, C.A. Cross-sectional and longitudinal comparisons of metabolic profiles between vegetarian and non-vegetarian subjects: A matched cohort study. Br. J. Nutr. 2015, 114, 1313–1320. [Google Scholar] [CrossRef]
- Robberecht, H.; De Bruyne, T.; Hermans, N. Effect of various diets on biomarkers of the metabolic syndrome. Int. J. Food Sci. Nutr. 2017, 68, 627–641. [Google Scholar] [CrossRef]
- Kim, H.; Caulfield, L.E.; Rebholz, C.M. Healthy Plant-Based Diets Are Associated with Lower Risk of All-Cause Mortality in US Adults. J. Nutr. 2018, 148, 624–631. [Google Scholar] [CrossRef]
- Viguiliouk, E.; Kendall, C.W.; Kahleová, H.; Rahelić, D.; Salas-Salvadó, J.; Choo, V.L.; Mejia, S.B.; Stewart, S.E.; Leiter, L.A.; Jenkins, D.J.; et al. Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. 2019, 38, 1133–1145. [Google Scholar] [CrossRef]
- Picasso, M.C.; Lo-Tayraco, J.A.; Ramos-Villanueva, J.M.; Pasupuleti, V.; Hernandez, A.V. Effect of vegetarian diets on the presentation of metabolic syndrome or its components: A systematic review and meta-analysis. Clin. Nutr. 2019, 38, 1117–1132. [Google Scholar] [CrossRef] [PubMed]
- USDA Food and Nutrition Service. HEI Scores for Americans. Available online: https://www.fns.usda.gov/hei-scores-americans (accessed on 16 June 2019).
- Marlow, H.J.; Harwatt, H.; Soret, S.; Sabaté, J. Comparing the water, energy, pesticide and fertilizer usage for the production of foods consumed by different dietary types in California. Public Health Nutr. 2015, 18, 2425–2432. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.; Curran, T.P.; Holden, N.M.; O’Brien, D.; Upton, J. Water footprinting of pasture-based farms; beef and sheep. Animal 2018, 12, 1068–1076. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.D.; Hoey, L.; Blesh, J.; Miller, L.; Green, A.; Shapiro, L.F. A Systematic Review of the Measurement of Sustainable Diets. Adv. Nutr. 2016, 7, 641–664. [Google Scholar] [CrossRef] [PubMed]
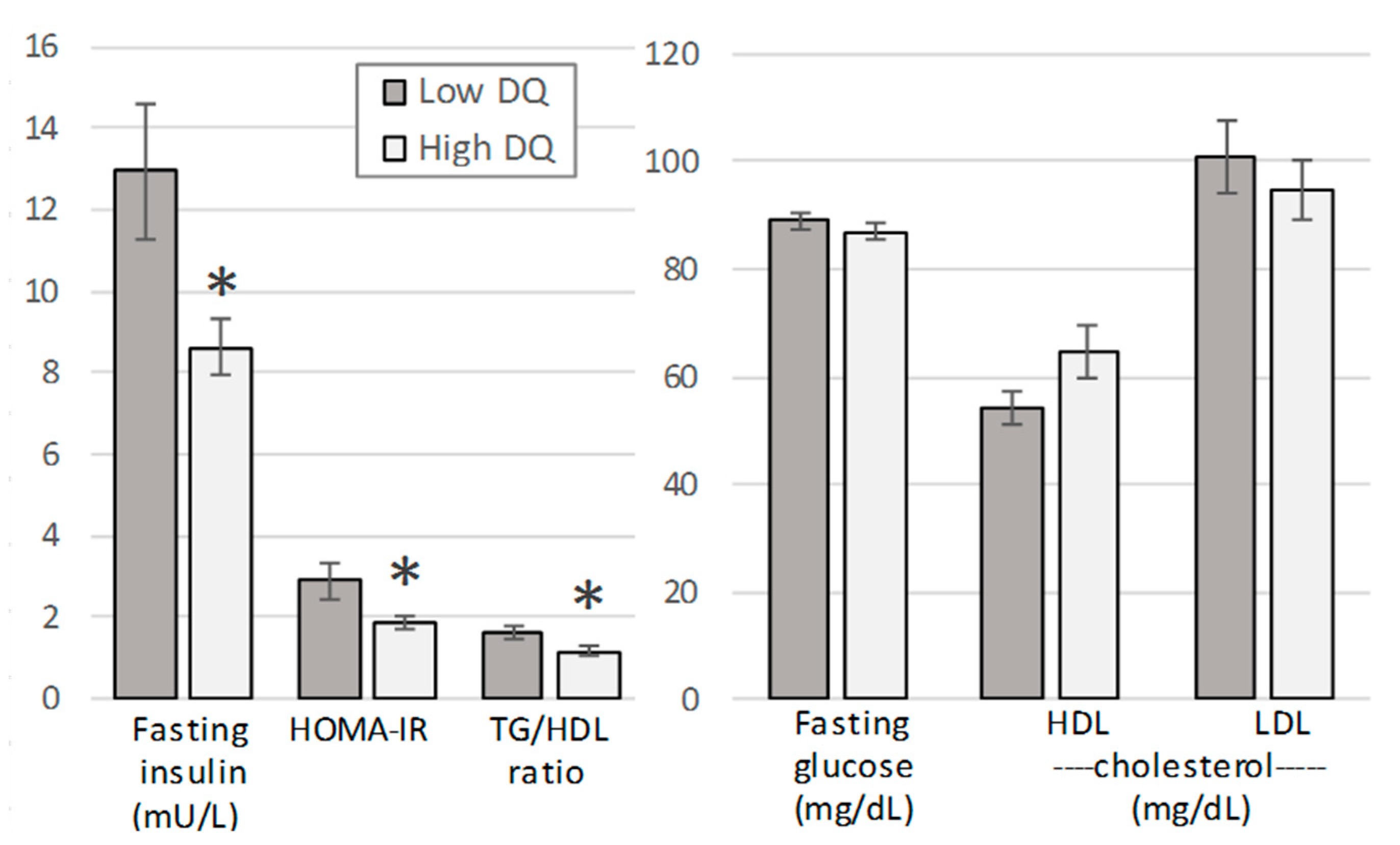
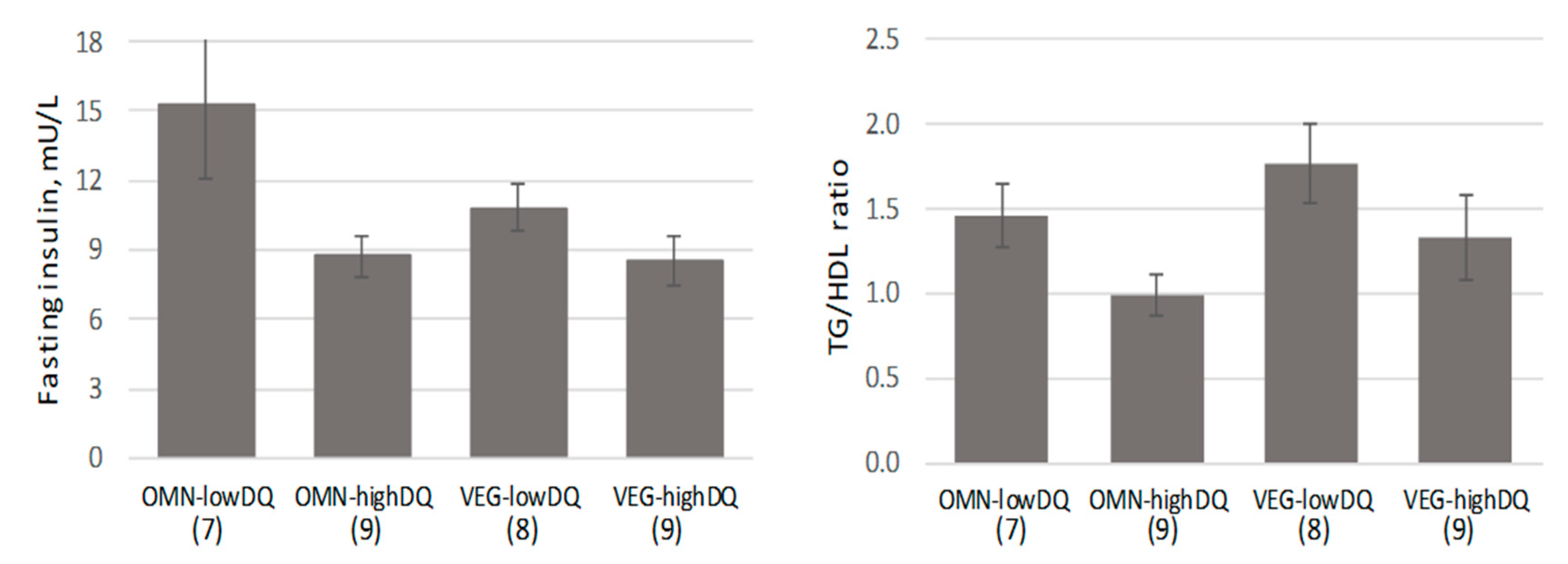
| Characteristic | Total Sample (n = 33; 6 M, 27 F) | Vegetarians (n = 17; 3 M, 14 F) | Omnivores (n = 16; 3 M, 13 F) |
|---|---|---|---|
| Age, year | 28.2 ± 8.9 | 27.1 ± 8.9 | 29.4 ± 9.1 |
| Body weight, kg | 63.4 ± 8.8 | 62.0 ± 8.2 | 64.8 ± 9.4 |
| Body mass index, kg/m2 | 22.5 ± 2.7 | 21.9 ± 2.5 | 23.2 ± 2.8 |
| Waist circumference, cm | 77.0 ± 11.9 | 76.4 ± 15.1 | 77.6 ± 7.7 |
| METS, kcal/kg·wk | 52.2 ± 27.3 | 53.9 ± 24.6 | 50.5 ± 30.7 |
| Diet Quality, score | 37.8 ± 2.9 | 37.8 ± 2.8 | 37.7 ± 3.1 |
| Plasma folate, nmol/L | 33.3 ± 12.3 | 34.8 ± 13.9 | 31.7 ± 10.6 |
| Fasting glucose, mg/dL | 87.8 ± 5.5 | 85.6 ± 4.7 | 90.2 ± 5.5 |
| Fasting insulin, mU/L | 10.6 ± 5.2 | 9.6 ± 3.2 | 11.6 ± 6.7 |
| HOMA-IR, score | 2.3 ± 1.3 | 2.0 ± 0.7 | 2.6 ± 1.7 |
| Triglycerides, mg/dL | 75.2 ± 25.3 | 77.9 ± 26.4 | 72.4 ± 24.5 |
| Total cholesterol, mg/dL | 165.6 ± 31.7 | 163.1 ± 29.8 | 168.3 ± 34.4 |
| HDL cholesterol, mg/dL | 60.0 ± 17.8 | 56.2 ± 16.2 | 64.0 ± 18.9 |
| LDL cholesterol, mg/dL | 97.5 ± 23.9 | 100.6 ± 25.1 | 94.2 ± 23.0 |
| TG/HDL ratio | 1.37 ± 0.63 | 1.54 ± 0.73 | 1.19 ± 0.47 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayra, S.; Ugarte, N.; Johnston, C.S. Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization. Nutrients 2019, 11, 1427. https://doi.org/10.3390/nu11061427
Mayra S, Ugarte N, Johnston CS. Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization. Nutrients. 2019; 11(6):1427. https://doi.org/10.3390/nu11061427
Chicago/Turabian StyleMayra, Selicia, Noel Ugarte, and Carol S. Johnston. 2019. "Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization" Nutrients 11, no. 6: 1427. https://doi.org/10.3390/nu11061427
APA StyleMayra, S., Ugarte, N., & Johnston, C. S. (2019). Health Biomarkers in Adults Are More Closely Linked to Diet Quality Attributes Than to Plant-Based Diet Categorization. Nutrients, 11(6), 1427. https://doi.org/10.3390/nu11061427






